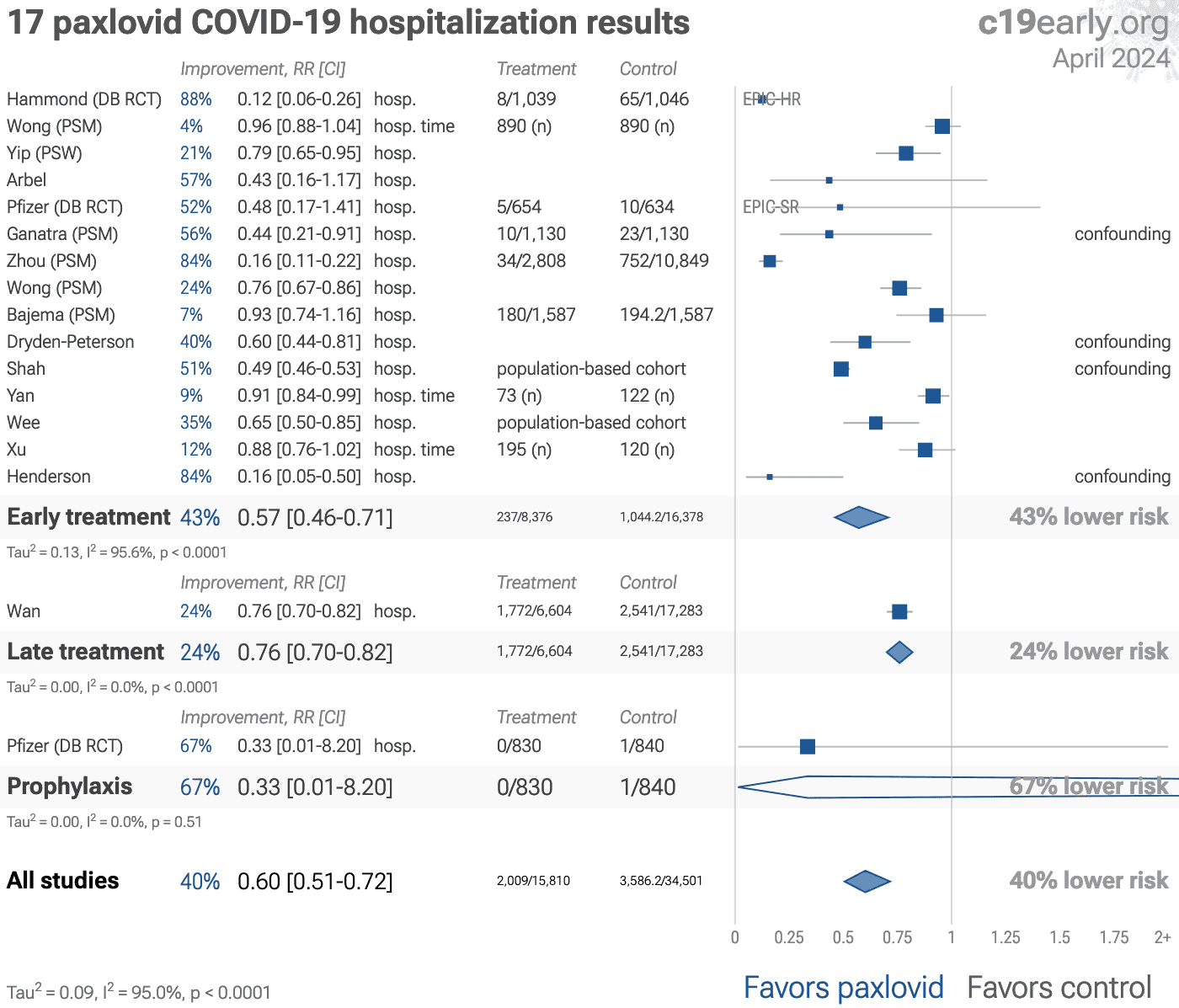
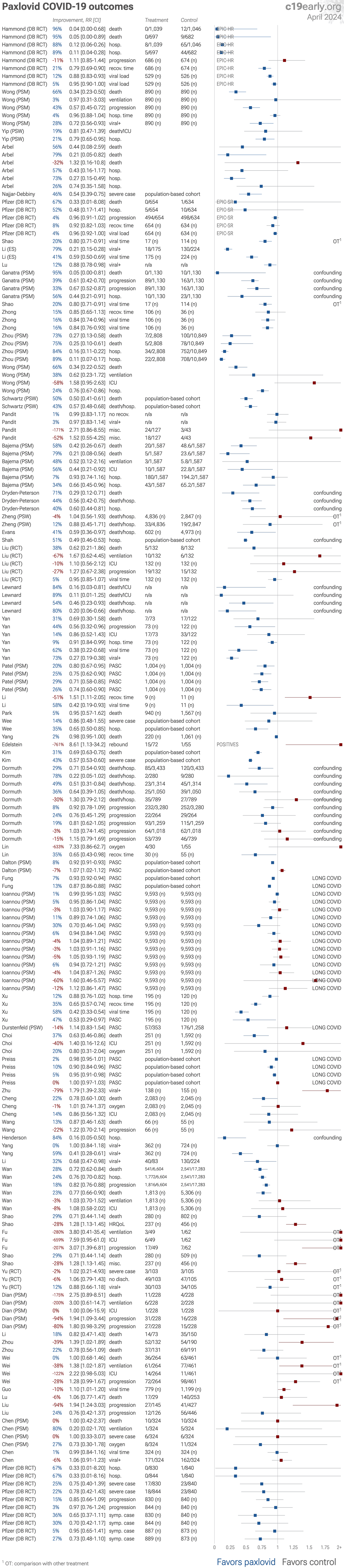
Pfizer has denied access to paxlovid for independent RCTs1. Pfizer RCTs report very good results, while non-Pfizer RCTs show relatively poor results2,3. Variants may be resistant to paxlovid4-12. Hoertel et al. find that >50% of patients that died had a contraindication for paxlovid. Retrospective studies that do not exclude contraindicated patients may significantly overestimate efficacy. Black box warning. The FDA notes that severe, life-threatening, and/or fatal adverse reactions due to drug interactions have been reported in patients treated with paxlovid14. Use may promote the emergence of variants that weaken host immunity and potentially contribute to long COVID15. Population studies often do not account for the different expected outcomes for the class of patients that seek out and receive early treatment. Studies show significantly increased risk of acute kidney injury16, liver injury17,18, and replication-competent viral rebound19-21.
Paxlovid is a combination of nirmatrelvir and ritonavir. Nirmatrelvir is a first generation SARS-CoV-2 3CL protease inhibitor22. Ritonavir is a HIV drug used to boost the levels of nirmatrelvir in the body by inhibiting its metabolism.
Paxlovid was adopted
in 64 countries.
Recent:Liu Kueper Chen Butler.
Jun 7 |
Meta-analysis of paxlovid studies | |
| Meta-analysis of paxlovid studies | ||
Jun 2 |
et al., Interactive Journal of Medical Research, doi:10.2196/80263 | COVID-19 Rebound in Nirmatrelvir Plus Ritonavir Treatment and Control Groups: Prospective Cohort Study |
| 87% worse recovery (p=0.02) and 148% worse viral clearance (p=0.01). Prospective observational study of 669 COVID-19 outpatients in the USA showing higher rates of viral and symptom rebound with paxlovid. | ||
May 29 |
et al., Medicine, doi:10.1097/MD.0000000000049099 | Independent mortality predictors in older COVID-19 patients with delayed hospitalization: A retrospective study from Eastern China, 2023 |
| Retrospective 300 elderly hospitalized COVID-19 patients in China, showing no mortality benefit with antivirals (paxlovid and molnupiravir) in multivariable analysis, OR 1.15 [0.28-4.74], without statistical significance. | ||
Apr 27 |
et al., Journal of Virology, doi:10.1128/jvi.02223-25 | SARS-CoV-2 3CLpro mutations T21I and E166A confer differential resistance to simnotrelvir, bofutrelvir, and ensitrelvir |
| In vitro and mouse study showing differential resistance of SARS-CoV-2 3CLpro T21I/E166A mutations to simnotrelvir, bofutrelvir, nirmatrelvir, and ensitrelvir. The resistant isolate SARS2-T21I/E166A showed 4.3-9.2-fold resistance to simno.. | ||
Mar 27 |
et al., NCT05965726 | RECOVER-VITAL: A Platform Protocol for Evaluation of Interventions for Viral Persistence, Viral Reactivation, and Immune Dysregulation in Post-Acute Sequelae of SARS-CoV-2 Infection (PASC) |
| 9% worse improvement (p=0.22). RCT 964 patients showing no difference in improvement of long COVID with 15-day or 25-day paxlovid compared with placebo. Results appeared in the registry 15 months after primary completion, with no publication or announcement [clinicaltr.. | ||
Mar 24 |
et al., Communications Medicine, doi:10.1038/s43856-026-01535-4 | Paxlovid shows organ-specific and age-specific impacts on risk of developing post-acute sequelae of COVID-19 |
| 7% higher long COVID (p=0.29). Retrospective 19,413 COVID-19 outpatient and hospitalized patients showing no overall reduction in long COVID risk with paxlovid. Stratification by organ system found a 37% reduction in gastrointestinal PASC (OR 0.63, p=0.002) - biologica.. | ||
Mar 19 |
et al., Health and Quality of Life Outcomes, doi:10.1186/s12955-026-02518-8 | Health-related quality of life in immunocompromised adults with mild–moderate COVID-19 treated with nirmatrelvir-ritonavir: results from the randomized, double-blinded EPIC-IC trial |
| RCT 150 immunocompromised adults with mild-to-moderate COVID-19 comparing 5, 10, or 15 days of nirmatrelvir/ritonavir. There was no control group. No dose-response relationship was observed: HRQoL recovery patterns were statistically.. | ||
Mar 17 |
et al., BMC Medicine, doi:10.1186/s12916-026-04791-2 | Effectiveness of nirmatrelvir/ritonavir and molnupiravir on post-COVID diabetes risk among an older adult cohort: a target trial emulation study |
| Retrospective analysis finding lower risk of post-COVID diabetes with nirmatrelvir/ritonavir but not molnupiravir. The effect may be entirely due to confounding. Authors excluded only 8% of non-diabetic patients for nirmatrelvir/ritonavir.. | ||
Mar 10 |
et al., Scientific Reports, doi:10.1038/s41598-026-42215-6 | A real-world retrospective analysis comparing the effectiveness of Azvudine and Nirmatrelvir/Ritonavir in COVID-19 patients with diabetes |
| Retrospective study of 400 hospitalized COVID-19 patients with diabetes in China showing no significant difference in the composite endpoint of disease progression (all-cause mortality, ICU admission, or invasive mechanical ventilation) b.. | ||
Mar 10 |
et al., Frontiers in Public Health, doi:10.3389/fpubh.2026.1760355 | Analysis of Long COVID characteristics and risk factors in individuals infected with COVID-19: a follow-up study based on a cohort of 2,792 participants |
| 10% higher long COVID (p=0.83). Prospective study of 2,792 COVID-19 patients in China. After propensity score matching, neither paxlovid nor azvudine showed a statistically significant association with long COVID. | ||
Feb 26 |
et al., medRxiv, doi:10.64898/2026.02.24.26347001 | Exploratory analyses of Immunologic Features in a Randomized, Placebo-Controlled Trial of Nirmatrelvir/Ritonavir for Long COVID |
| RCT 82 long COVID patients showing no significant differences with nirmatrelvir/ritonavir treatment. The study found no improvement in physical health summary scores, no changes in circulating SARS-CoV-2 spike protein levels, and no signi.. | ||
Feb 11 |
et al., Frontiers in Public Health, doi:10.3389/fpubh.2026.1698604 | Canadian adaptive platform trial of treatments for COVID in community settings (CanTreatCOVID): recruitment strategies of a decentralized, national randomized controlled trial for acute SARS-CoV-2 |
| Recruitment strategy analysis for the decentralized adaptive platform trial CanTreatCOVID. This is the second paper published reporting secondary analyses, while authors have not yet published the actual efficacy results of the trial. | ||
Jan 30 |
et al., Acta Cardiol Sin, doi:10.6515/ACS.202601_42(1).20250726A | Cardiovascular Outcomes in COVID-19 Patients Treated with Paxlovid: A Multicenter Retrospective Study |
| 11% lower mortality (p=0.21). Retrospective 606 COVID-19 patients treated with paxlovid and 1,809 propensity score-matched controls in Taiwan, showing short-term mortality benefits at 3 months, but reduced benefit at 6 months, and no significant benefit at 12 months. .. | ||
Jan 28 |
et al., medRxiv, doi:10.64898/2026.01.23.26343617 | Balancing data quality and participant burden: A comparative analysis of abbreviated vs extended symptom diaries in the CanTreatCOVID trial |
| Secondary analysis of the CanTreatCOVID paxlovid RCT comparing adherence and symptom reporting between a 9-item abbreviated diary and 34-item FLU-PRO Plus diary in 712 COVID-19 outpatients, showing no significant difference in compliance,.. | ||
Jan 11 |
et al., Open Forum Infectious Diseases, doi:10.1093/ofid/ofaf695.1825 | Temporal Trends in Serious Adverse Events Associated with Oral Antivirals During the COVID-19 Pandemic: Insights from the FAERS Database (2020–2023) |
| Retrospective 11,547 serious adverse event reports from the FDA database (2020-2023) showing significant safety signals with oral COVID-19 antivirals. Paxlovid showed the strongest signals for drug-drug interactions (ROR: 4.83) and liver .. | ||
Jan 11 |
et al., Open Forum Infectious Diseases, doi:10.1093/ofid/ofaf695.1809 | Disparities in the Use of nirmatrelvir/ritonavir for COVID-19: A Retrospective Cohort Study |
| Retrospective 3,498 patients receiving COVID-19 antivirals showing that female patients were significantly more likely to receive treatment. Studies show that female patients are significantly more likely to be "health-conscious".. | ||
Jan 11 |
et al., Open Forum Infectious Diseases, doi:10.1093/ofid/ofaf695.1812 | Comparative Effectiveness of Combination Therapy with Nirmatrelvir-Ritonavir and Molnupiravir versus Monotherapy with Molnupiravir or Nirmatrelvir-Ritonavir in Hospitalised COVID-19 Patients: A Target Trial Emulation Study |
| 3% lower mortality (p=0.8), 21% lower ventilation (p=0.39), and 28% higher ICU admission (p=0.46). IPTW retrospective target trial emulation of 28,355 hospitalized COVID-19 patients in Hong Kong showing no benefit and potential harm (higher mortality) with combined nirmatrelvir-ritonavir and molnupiravir compared to nirmatrelvir-ritona.. | ||
Jan 10 |
et al., Virology Journal, doi:10.1186/s12985-025-03057-2 | Viral kinetics in adults with Covid-19 treated with nirmatrelvir-ritonavir or molnupiravir: a population-based, observational cohort study |
| 22% worse viral clearance (p<0.0001). Observational cohort study of 113,399 COVID-19 outpatients in Vienna showing viral kinetics patterns with nirmatrelvir-ritonavir and molnupiravir treatment. Both antivirals showed improved viral clearance at 7 days, but worse viral cleara.. | ||
Jan 9 |
et al., The Lancet Microbe, doi:10.1016/j.lanmic.2025.101227 | SARS-CoV-2 infectious shedding and rebound among adults with and without oral antiviral use: two case-ascertained prospective household studies |
| 218% worse results (p=0.01). Prospective study of 160 non-hospitalized adults at high risk for severe COVID-19 showing paxlovid or molnupiravir associated with increased risk of viral rebound with infectious virus. Among treated participants, 25% experienced culture .. | ||
Jan 5 |
et al., Frontiers in Immunology, doi:10.3389/fimmu.2025.1698271 | Patient-reported improvements from use of IMC-2 alone and IMC-2 and Paxlovid® in a Long COVID cohort: a case series |
| Case series of 24 long COVID outpatients showing greater symptom improvement with a combination of IMC-2 (valacyclovir + celecoxib) plus 15-day paxlovid compared to IMC-2 alone over 120 days. | ||
Dec 31 2025 |
et al., NCT04483960 | A Multi-centre Randomised Adaptive Platform Clinical Trial to Assess Clinical, Virological and Immunological Outcomes in Patients With SARS-CoV-2 Infection (COVID-19) |
| Estimated 2,200 patient paxlovid late treatment RCT with results expected soon (estimated completion over 5 months ago). | ||
Oct 14 2025 |
et al., Infectious Diseases and Therapy, doi:10.1007/s40121-025-01228-w | Symptom Alleviation/Resolution and Returns to Usual Health/Activities in Immunocompromised Adults with COVID-19 Treated with Nirmatrelvir-Ritonavir: Results from the EPIC-IC Trial |
| RCT 156 immunocompromised COVID-19 patients comparing 5-day, 10-day, and 15-day paxlovid treatment showing no significant differences. | ||
Oct 14 2025 |
et al., Emerging Microbes & Infections, doi:10.1080/22221751.2025.2552716 | SARS-CoV-2 Mpro inhibitor ensitrelvir: asymmetrical cross-resistance with nirmatrelvir and emerging resistance hotspots |
| In vitro study showing that SARS-CoV-2 develops high-fitness resistance to ensitrelvir through Mpro mutations. Authors found asymmetrical cross-resistance, with ensitrelvir-resistant variants showing minimal cross-resistance to nirmatrelv.. | ||
Oct 8 2025 |
et al., Open Forum Infectious Diseases, doi:10.1093/ofid/ofaf634 | Longitudinal patient-reported outcome trajectories in Long COVID: Findings from the STOP-PASC Clinical Trial |
| 127% higher progression (p=0.14). Secondary analysis of NCT0557666. RCT 155 adults with long COVID showing no benefit from a 15-day course of paxlovid. Patients with worsening symptoms were more likely to be treated with paxlovid, without statistical significance. | ||
Oct 6 2025 |
et al., Pulmonary Therapy, doi:10.1007/s41030-025-00316-z | A Retrospective Observational Study on COVID-19 Patients Receiving Treatment with Nirmatrelvir/Ritonavir (PAXLOVID) |
| Retrospective 3,011 outpatients in Bahrain showing lower hospitalization with paxlovid treatment. However, the study has significant methodological limitations that likely substantially overestimate paxlovid's benefit: Contraindicated/sic.. | ||
Sep 29 2025 |
et al., BMC Infectious Diseases, doi:10.1186/s12879-025-11651-6 | Comparative effectiveness of antiviral treatment on household transmission of SARS-CoV-2: a retrospective cohort study using administrative data |
| Retrospective 5,398 married couples in Japan showing no significant difference in household transmission rates between molnupiravir, ensitrelvir, and paxlovid. Hospitalized patients receiving antivirals showed a trend toward lower transmi.. | ||
Sep 10 2025 |
et al., New England Journal of Medicine, doi:10.1056/NEJMoa2502457 (results released 9/10/2025) | Oral Nirmatrelvir–Ritonavir for Covid-19 in Higher-Risk Outpatients |
| 18% higher combined mortality/hospitalization (p=0.69), 27% improved recovery (p<0.0001), and 81% improved viral clearance (p=0.006). PANORAMIC/CanTreatCOVID results showing that paxlovid did not reduce hospitalization or death, adjusted OR 1.18, 95% Bayesian credible interval [0.55-2.62] for PANORAMIC and adjusted OR 0.48, 95% Bayesian credible interval [0.08-2.23] for.. | ||
Sep 10 2025 |
et al., New England Journal of Medicine, doi:10.1056/NEJMoa2502457 (results released 9/10/2025) | Oral Nirmatrelvir–Ritonavir for Covid-19 in Higher-Risk Outpatients |
| 52% lower combined mortality/hospitalization (p=0.39) and 42% improved recovery (p<0.0001). Results for the CanTreatCOVID trial. See [Butler] for details. | ||
Sep 10 2025 |
et al., New England Journal of Medicine | Oral Nirmatrelvir–Ritonavir for Covid-19 in Higher-Risk Outpatients |
| Moved to [Butler] | ||
Sep 10 2025 |
et al., New England Journal of Medicine | Oral Nirmatrelvir–Ritonavir for Covid-19 in Higher-Risk Outpatients |
| Moved to [Butler] | ||
Sep 2 2025 |
et al., medRxiv, doi:10.1101/2025.08.29.25334732 | Long-term follow-up of treatment comparisons in RECOVERY: a randomised, open-label, platform trial for patients hospitalised with COVID-19 |
| 6-month followup of RECOVERY patients. Results are reported within the respective trials for each treatment. | ||
Aug 31 2025 |
et al., The Lancet Infectious Diseases, doi:10.1016/S1473-3099(25)00073-8 | Nirmatrelvir–ritonavir versus placebo–ritonavir in individuals with long COVID in the USA (PAX LC): a double-blind, randomised, placebo-controlled, phase 2, decentralised trial |
| 124% higher long COVID (p=0.56). RCT 100 outpatients with long COVID showing no significant difference with paxlovid. Authors hypothesize that viral reservoirs may have been inaccessible to the drug, or that the dose or duration may have been insufficient to eliminate pe.. | ||
Jul 1 2025 |
et al., BMC Infectious Diseases, doi:10.1186/s12879-025-11195-9 | Real-world effectiveness of simnotrelvir-ritonavir versus nirmatrelvir-ritonavir in hospitalized patients with COVID-19 during the omicron wave in China: a retrospective cohort study |
| Retrospective 585 hospitalized COVID-19 patients in China showing that xiannuoxin provided better clinical improvement than paxlovid. There were no significant differences for progression, mortality, or respiratory support. | ||
Jun 26 2025 |
et al., Pharmaceutics, doi:10.3390/pharmaceutics17070832 | Carboxylesterase Factors Influencing the Therapeutic Activity of Common Antiviral Medications Used for SARS-CoV-2 Infection |
| Review of how carboxylesterase (CES) enzymes influence the activity of certain COVID-19 antivirals, focusing on remdesivir, molnupiravir, nirmatrelvir, and favipiravir. Authors highlight that remdesivir requires activation by CES1, while .. | ||
Jun 5 2025 |
et al., Biomedicines, doi:10.3390/biomedicines13061387 | Comparative Pharmacovigilance Analysis of Approved and Repurposed Antivirals for COVID-19: Insights from EudraVigilance Data |
| Pharmacovigilance analysis of 64,776 adverse event reports from EudraVigilance comparing safety profiles of COVID-19 antivirals, showing remdesivir associated with more serious adverse events compared to paxlovid. Remdesivir showed consis.. | ||
Jun 3 2025 |
et al., The Journal of Infectious Diseases, doi:10.1093/infdis/jiaf294 | Functional and structural characterization of treatment-emergent nirmatrelvir resistance mutations at low frequencies in the main protease (Mpro) reveals a unique evolutionary route for SARS-CoV-2 to gain resistance |
| In vitro and in silico study showing that novel SARS-CoV-2 main protease (Mpro) mutations confer resistance to paxlovid. Authors identified new Mpro clinical variants, including D48D/L58F/P132H and D48D/L67V/K90R/P132H, in patients who re.. | ||
May 31 2025 |
et al., Open Forum Infectious Diseases, doi:10.1093/ofid/ofaf567 (date from preprint) | Risk of Post-COVID-19 Conditions Among Adolescents and Adults Who Received Nirmatrelvir-Ritonavir for Acute COVID-19: A Retrospective Cohort Study |
| Retrospective 291,433 paxlovid recipients matched 1:2 to 582,866 untreated COVID-19 outpatients in the USA reporting a modest reduction in long COVID in the primary model for patients 50+. Analysis requiring a positive laboratory test or .. | ||
May 31 2025 |
et al., American Journal of Respiratory and Critical Care Medicine, doi:10.1164/ajrccm.2025.211.Abstracts.A1193 | Toxic as FK - A Case of Paxlovid-Induced Tacrolimus (FK506) Toxicity in a Kidney Transplant Recipient with COVID-19 |
| Case report of a 71-year-old kidney transplant recipient who developed fatal tacrolimus toxicity after being prescribed paxlovid for COVID-19. | ||
May 21 2025 |
et al., The Lancet Infectious Diseases, doi:10.1016/S1473-3099(25)00482-7 (date from preprint) | Antiviral efficacy of oral ensitrelvir versus oral ritonavir-boosted nirmatrelvir in COVID-19 |
| 8% faster recovery (p=0.006) and 55% improved viral clearance (p<0.0001). RCT 604 low-risk adults with early COVID-19 symptoms showing significantly improved SARS-CoV-2 viral clearance with both ensitrelvir and paxlovid. Inclusion criteria selected for low-risk patients with high viral loads which may not gener.. | ||
May 8 2025 |
et al., Health Expectations, doi:10.1111/hex.70273 | Long Covid Symptom Clusters, Correlates and Predictors in a Highly Vaccinated Australian Population in 2023 |
| Retrospective 1,205 adults in Australa showing 20% reported long COVID symptoms during an Omicron-predominant period. In multivariable analysis, paxlovid and molnupiravir use showed higher risk of long COVID, without statistical significa.. | ||
May 2 2025 |
et al., medRxiv, doi:10.1101/2025.05.01.25326797 | A randomised-controlled Phase I de-escalation trial of Molnupiravir and Nirmatrelvir/Ritonavir combination for mild-moderate SARS-CoV-2 infection |
| 1% worse viral clearance (p=0.94). Randomized open-label phase I trial of 24 outpatients with mild-moderate COVID-19 showing safety and tolerability of combined molnupiravir and paxlovid therapy. Supplemental Table 2 shows greater decline of viral load in the control group.. | ||
May 1 2025 |
et al., Infectious Disease Reports, doi:10.3390/idr17030043 | Incidence of COVID-19 Symptom Rebound After Treatment with Remdesivir |
| Retrospective 155 patients with mild to moderate COVID-19 showing significantly lower symptom rebound rates with remdesivir compared with paxlovid. Authors provide an analysis of rebound with paxlovid, showing higher rates in prospective .. | ||
Apr 4 2025 |
et al., Biomedical Research, doi:10.2220/biomedres.46.37 | Possible involvement of neuropeptide Y sub-receptor 1 (NPY-Y1) in the anti-viral response of SARS-CoV-2 infection in Syrian hamster |
| Animal study showing that molnupiravir and remdesivir co-administration reduces SARS-CoV-2 viral load and inflammatory responses in Syrian hamsters, with involvement of neuropeptide Y sub-receptor 1 (NPY-Y1). Authors found that SARS-CoV-2.. | ||
Mar 31 2025 |
et al., Scientific Reports, doi:10.1038/s41598-025-85677-w | Efficacy and safety of azvudine versus nirmatrelvir/ritonavir in cancer patients with COVID-19 |
| PSM retrospective 596 cancer patients with COVID-19 showing that azvudine significantly reduced all-cause mortality and composite disease progression compared to paxlovid. | ||
Mar 17 2025 |
et al., JACC: Case Reports, doi:10.1016/j.jaccas.2025.103238 | Complete Heart Block Triggered by Nirmatrelvir-Ritonavir and Verapamil |
| Case report of a 58-year-old woman who developed complete heart block and shock due to a drug-drug interaction between paxlovid and verapamil, exacerbated by acute kidney injury and liver dysfunction. This case highlights potentially leth.. | ||
Mar 15 2025 |
et al., BMC Pulmonary Medicine, doi:10.1186/s12890-025-03524-0 | Risk prediction and early intervention strategies for persistent SARS-CoV-2 infection in patients with non-Hodgkin lymphoma: a retrospective cohort study |
| 5% worse viral clearance (p=0.83). Retrospective 660 patients with non-Hodgkin lymphoma (NHL) and SARS-CoV-2 infection, identifying risk factors for persistent COVID-19. There was no significant difference in persistent SARS-CoV-2 infection with paxlovid, molnupiravir, or .. | ||
Mar 13 2025 |
et al., Eurosurveillance, doi:10.2807/1560-7917.ES.2025.30.10.2400252 | Impact of SARS-CoV-2 variant mutations on susceptibility to monoclonal antibodies and antiviral drugs: a non-systematic review, April 2022 to October 2024 |
| Review of SARS-CoV-2 variant mutations and their impact on susceptibility to monoclonal antibodies and antivirals. Mutations in the ORF1ab region led to decreased susceptibility to paxlovid, while nsp12 mutations reduced efficacy for remd.. | ||
Mar 11 2025 |
et al., bioRxiv, doi:10.1101/2025.03.09.642200 | The protease inhibitor Nirmatrelvir synergizes with inhibitors of GRP78 to suppress SARS-CoV-2 replication |
| In vitro and animal study showing strong synergistic effects when combining nirmatrelvir with GRP78 inhibitors. | ||
Mar 11 2025 |
et al., Journal of Clinical Medicine, doi:10.3390/jcm14061886 | Comparative Analysis of Neuropsychiatric Adverse Reactions Associated with Remdesivir and Nirmatrelvir/Ritonavir in COVID-19 Treatment: Insights from EudraVigilance Data |
| Pharmacovigilance analysis of 8,078 paxlovid 3,934 remdesivir case safety reports from the EudraVigilance database, showing paxlovid associated with significantly higher incidence of neuropsychiatric adverse drug reactions. Paxlovid repor.. | ||
Mar 10 2025 |
et al., BMC Infectious Diseases, doi:10.1186/s12879-025-10684-1 | Development and validation of a nomogram to assess the occurrence of liver dysfunction in patients with COVID-19 pneumonia in the ICU |
| 27% worse results (p<0.0001). Retrospective 286 critically ill COVID-19 ICU patients developing a predictive model for liver dysfunction, showing significantly higher risk with azvudine and paxlovid use. | ||
Mar 3 2025 |
et al., BMC Infectious Diseases, doi:10.1186/s12879-025-10666-3 | Prognostic factors in hospitalized patients with COVID-19 pneumonia and effectiveness of prophylactic anticoagulant therapy: a single-center retrospective study |
| 28% higher mortality (p=0.14). Retrospective 2,520 hospitalized COVID-19 pneumonia patients focusing on prophylactic anticoagulation but also reporting results for azvudine and paxlovid. | ||
Feb 26 2025 |
et al., Expert Opinion on Drug Safety, doi:10.1080/14740338.2025.2471509 | Effects of Nirmatrelvir/ritonavir (paxlovid) on the nervous system: analysis on adverse events released by FDA |
| Adverse event disproportionality analysis for COVID-19 drugs showing significantly higher risk of neurological adverse events with paxlovid. | ||
Feb 25 2025 |
et al., BMC Infectious Diseases, doi:10.1186/s12879-025-10643-w | Real-world effectiveness and safety of oral Azvudine versus Paxlovid for COVID-19 in patients with kidney disease: a multicenter, retrospective, cohort study |
| Retrospective 657 hospitalized COVID-19 patients with kidney disease showing no significant difference in all-cause mortality or disease progression between azvudine and paxlovid. Subgroup analysis showed lower disease progression with az.. | ||
Feb 18 2025 |
et al., Infectious Medicine, doi:10.1016/j.imj.2025.100169 | Clinical outcomes of patients with coronavirus disease 2019 and active tuberculosis co-infection in Beijing China: A retrospective single-center descriptive study |
| 337% higher mortality (p=0.02). Retrospective 102 COVID-19 patients with active tuberculosis co-infection showing a mortality rate of 9.8%, compared with 0.03% for patients with COVID-19 alone. Paxlovid was used for 19 patients due limited availability but resulted in h.. | ||
Feb 18 2025 |
et al., Emerging Microbes & Infections, doi:10.1080/22221751.2025.2469648 | Effectiveness of nirmatrelvir/ritonavir and molnupiravir on post-COVID-19 outcomes among outpatients: a target trial emulation investigation |
| 38% lower mortality (p<0.0001) and 15% lower hospitalization (p<0.0001). Retrospective target trial emulation of outpatients in Hong Kong showing reduced post-acute mortality and hospitalization with paxlovid, but no significant long-term benefit with molnupiravir. Figure 1 shows only 0.08% of patients were ex.. | ||
Feb 15 2025 |
et al., RPS Pharmacy and Pharmacology Reports, doi:10.1093/rpsppr/rqae028 | Potential drug interactions with nirmatrelvir/ritonavir in critically ill patients with COVID-19 – a retrospective observational study |
| Retrospective 500 critically ill COVID-19 patients in Germany showing potential drug-drug interactions with paxlovid in 48% of patients, with higher age and number of comorbidities significantly associated with drug-drug interactions. Aut.. | ||
Feb 5 2025 |
et al., Clinical Infectious Diseases, doi:10.1093/cid/ciaf046 | Long COVID Incidence Proportion in Adults and Children Between 2020 and 2024: An EHR-Based Study From the RECOVER Initiative |
| 16% higher long COVID (p=0.32). Retrospective 5.4 million patients across three US clinical research networks showing higher use of paxlovid (within 5 days of the index event) in patients with long COVID. Adjusted results for paxlovid’s impact on long COVID risk are not.. | ||
Jan 27 2025 |
et al., iScience, doi:10.1016/j.isci.2025.111907 | Real-world effectiveness of azvudine versus nirmatrelvir-ritonavir in hospitalized patients with COVID-19 and pre-existing diabetes |
| Retrospective 954 hospitalized patients with COVID-19 and pre-existing diabetes showing lower mortality with azvudine compared to paxlovid, but no significant difference for composite disease progression. | ||
Jan 27 2025 |
et al., bioRxiv, doi:10.1101/2025.01.24.634813 | Molecular Mechanisms of Drug Resistance and Compensation in SARS-CoV-2 Main Protease: The Interplay Between E166 and L50 |
| In vitro study showing that mutations at position E166 in the SARS-CoV-2 main protease (Mpro) confer resistance to nirmatrelvir, the active component of paxlovid, while preserving substrate cleavage. Authors found that E166A and E166V mut.. | ||
Dec 31 2024 |
et al., Acta Pharmaceutica Sinica B, doi:10.1016/j.apsb.2024.12.032 | A retrospective cohort study of the efficacy and safety of oral azvudine versus nirmatrelvir/ritonavir in elderly hospitalized COVID-19 patients aged over 60 years |
| Retrospective 5,131 elderly hospitalized COVID-19 patients in China showing lower mortality with azvudine compared to paxlovid. There was no significant difference in composite disease progression. Safety analysis showed azvudine had a lo.. | ||
Dec 25 2024 |
et al., Pulmonary Therapy, doi:10.1007/s41030-024-00284-w | Real-World Effectiveness of Bebtelovimab Versus Nirmatrelvir/Ritonavir in Outpatients with COVID-19 |
| Retrospective 5,827 matched pairs of non-hospitalized COVID-19 patients showing no significant differences between bebtelovimab and paxlovid treatment. Authors do not exclude patients with contraindications for paxlovid. | ||
Dec 12 2024 |
et al., American Journal of Gastroenterology, doi:10.14309/01.ajg.0001082744.48729.45 | Efficacy of Nirmatrelvir/Ritonavir (Paxlovid) for COVID-19 in Vaccinated Patients With Inflammatory Bowel Disease |
| no change in progression (p=0.97) and 33% lower hospitalization (p=0.14). PSM retrospective 2,773 IBD patients showing no significant difference in hospitalization or the composite outcome of ICU admission, intubation, or mortality with paxlovid treatment. Authors do not specify exclusion of contraindicated pat.. | ||
Nov 14 2024 |
et al., Pediatric Infectious Disease Journal, doi:10.1097/INF.0000000000004594 | Evolving COVID-19 Landscape: Assessing the Effectiveness and Safety Profile of Nirmatrelvir/Ritonavir in Adolescents |
| 813% higher mortality (p=0.06) and 360% higher progression (p=0.1). Retrospective 374 adolescent COVID-19 outpatients showing no significant differences with paxlovid treatment. | ||
Nov 12 2024 |
et al., Pharmacoepidemiology and Drug Safety, doi:10.1002/pds.70043 | Investigating the Safety Profile of Fast‐Track COVID‐19 Drugs Using the FDA Adverse Event Reporting System Database: A Comparative Observational Study |
| FAERS adverse event analysis for remdesivir, bebtelovimab, molnupiravir, and paxlovid. Top 10 signals for serious adverse drug reactions for remdesivir included death and acute kidney injury, for paxlovid: disease recurrence and rebound, .. | ||
Nov 11 2024 |
et al., Clinical Infectious Diseases, doi:10.1093/cid/ciae551 | Alleviation of COVID-19 Symptoms and Reduction in Healthcare Utilization Among High-Risk Patients Treated With Nirmatrelvir/Ritonavir (NMV/R): A phase 3 randomized trial |
| Additional analysis of [Hammond]. Results are shown with the main paper [Hammond]. | ||
Nov 8 2024 |
et al., Journal of Global Health, doi:10.7189/jogh.14.05032 | Low antiviral uptake of nirmatrelvir/ritonavir and molnupiravir in adult patients with COVID-19 in Taiwan in 2022 |
| Retrospective 96,398 adult COVID-19 patients in Taiwan showing very low use of approved antivirals nirmatrelvir/ritonavir (5.1%) and molnupiravir (1.9%) in 2022, especially in moderate-to-high risk groups eligible for treatment per WHO gu.. | ||
Nov 5 2024 |
et al., Blood, doi:10.1182/blood-2024-207488 | Nirmatrelvir/Ritonavir Reduces Infection Duration in Hospitalized Hematological Malignancies Patients with Mild-to-Moderate COVID-19: A Retrospective Study |
| 9% lower mortality (p=0.92) and 34% improved recovery (p=0.04). Retrospective 130 hematological malignancy patients with mild-to-moderate COVID-19 in China showing no significant difference in 90-day mortality, but shorter infection duration with paxlovid treatment within 5 days. | ||
Oct 28 2024 |
et al., Research Square, doi:10.21203/rs.3.rs-5314583/v1 | A randomized trial of at-home COVID-19 tests, telemedicine, and rapid prescription delivery for immunocompromised individuals |
| RCT 671 immunocompromised or aged 65+ participants in the US showing significantly reduced ICU admissions and cost of COVID care with access to at-home COVID tests, telemedicine, and rapid paxlovid delivery. There was no significant diffe.. | ||
Oct 19 2024 |
et al., bioRxiv, doi:10.1101/2024.10.18.619137 | Nirmatrelvir-Resistant Mutations in SARS-CoV-2 Mpro Enhance Host Immune Evasion via Cleavage of NF-κB Essential Modulator |
| In vitro study showing that paxlovid use may promote the emergence of SARS-CoV-2 variants that can weaken host immunity and potentially contribute to long COVID. Authors show that SARS-CoV-2 main protease (Mpro) mutations that confer resi.. | ||
Oct 17 2024 |
et al., International Journal of Clinical Pharmacy, doi:10.1007/s11096-024-01852-5 (date from preprint) | Frequency, type and severity of drug-related problems and pharmacist interventions in Paxlovid® prescribing: a descriptive analysis |
| Retrospective 140 hospitalized COVID-19 patients in Austria showing that pharmacist review identified a high rate of drug-related problems (DRPs) with paxlovid prescribing, including drug-drug interactions (DDIs) and inappropriate dosing .. | ||
Oct 8 2024 |
et al., Clinical Infectious Diseases, doi:10.1093/cid/ciaf548 (results released 10/8/2024) | Retreatment With Nirmatrelvir/Ritonavir Following Return of COVID-19 Symptoms and SARS-CoV-2 Positivity |
| 8% improved recovery (p=0.52) and 18% improved viral clearance (p=0.0004). RCT 436 patients with post-paxlovid rebound showing improved viral load at day 5 but no significant difference in recovery time or viral clearance with repeated paxlovid treatment vs. placebo/ritonavir. | ||
Oct 3 2024 |
et al., MMWR. Morbidity and Mortality Weekly Report, doi:10.15585/mmwr.mm7339a3 | Differences in COVID-19 Outpatient Antiviral Treatment Among Adults Aged ≥65 Years by Age Group — National Patient-Centered Clinical Research Network, United States, April 2022–September 2023 |
| Retrospective 393,390 COVID-19 patients aged ≥65 years showing underutilization of antiviral treatment, with lower use for older patients at higher risk for severe outcomes. | ||
Sep 27 2024 |
, M., Electronic Thesis and Dissertation Repository, 10427 | The Transfer of Molnupiravir and Nirmatrelvir Across the Human Placenta and Prediction of Drug Safety in Pregnancy |
| Ex vivo study showing placental transfer of molnupiravir (NHC) and nirmatrelvir in human placentas. Author used a placental perfusion model to assess drug transfer across the human placental barrier. Both compounds readily crossed the pla.. | ||
Sep 25 2024 |
et al., JAMA Network Open, doi:10.1001/jamanetworkopen.2024.35431 | Emerging SARS-CoV-2 Resistance After Antiviral Treatment |
| Prospective study of 156 outpatients showing emergent nirmatrelvir resistance mutations with nirmatrelvir treatment, especially in immunosuppressed individuals. Results support the use of combination antiviral therapy and host-directed tr.. | ||
Sep 25 2024 |
et al., PLOS ONE, doi:10.1371/journal.pone.0308205 | A SONAR report on Nirmatrelvir/ritonavir-associated rebound COVID-19: Using new databases for evaluating new diseases |
| Retrospective case series identifying 35 cases of presumed or documented paxlovid rebound COVID-19 infection from pre-print articles, social media posts, and news reports. Authors highlight a delay of 2-3 months for reports to appear in p.. | ||
Sep 18 2024 |
et al., Nature Communications, doi:10.1038/s41467-024-51924-3 | Emergence of transmissible SARS-CoV-2 variants with decreased sensitivity to antivirals in immunocompromised patients with persistent infections |
| Analysis of 15 immunocompromised patients with persistent SARS-CoV-2 infection showing emergence of putative antiviral resistance mutations in nsp5 and nsp12 following treatment with remdesivir and nirmatrelvir-ritonavir. An infectious SA.. | ||
Sep 7 2024 |
et al., Clinical Therapeutics, doi:10.1016/j.clinthera.2024.08.004 | Prevalence of Potential Drug Interactions With Direct-Acting Antivirals for COVID-19 Among Hospitalized Patients |
| Retrospective 788,238 hospitalized COVID-19 patients in the US, showing a very high prevalence of drug-drug interactions with paxlovid, with higher prevalence for older patients, patients with more comorbidities, and patients at high-risk.. | ||
Aug 31 2024 |
et al., Translational Cancer Research, doi:10.21037/tcr-24-70 | Effectiveness of Paxlovid in the treatment of the SARS-CoV-2 Omicron variant infection in children with hematologic malignancies: a retrospective cohort study |
| Retrospective 42 children with hematologic malignancies (HMs) and SARS-CoV-2 omicron infection showing no significant difference in clinical outcomes or viral clearance with paxlovid after propensity score matching. | ||
Aug 20 2024 |
et al., BMC Infectious Diseases, doi:10.1186/s12879-024-09654-w | Predictors for cause-specific and timing of deaths in patients with COVID-19: a cohort study in Taiwan |
| 31% lower mortality (p=0.25). Retrospective 2,196 COVID-19 patients in Taiwan (49% mild cases, 44% moderate, 7% severe) showing lower mortality with paxlovid, without statistical significance. Authors report that all infected patients were hospitalized at the time of .. | ||
Aug 14 2024 |
et al., Age and Ageing, doi:10.1093/ageing/afae180 | Efficacy of COVID-19 Oral antivirals in hospitalised oldest-old with high morbidity burden: a target trial emulation study |
| 20% lower mortality (p=0.01). Target trial emulation study of 13,642 (molnupiravir) and 9,553 (paxlovid) elderly hospitalized patients in Hong Kong showing lower mortality with treatment. | ||
Aug 12 2024 |
et al., The Journal of Infectious Diseases, doi:10.1093/infdis/jiae385 | Persistence of an infectious form of SARS-CoV-2 post protease inhibitor treatment of permissive cells in vitro |
| In vitro study showing the persistence of an infectious form of SARS-CoV-2 after treatment with 3CLpro inhibitors nirmatrelvir and ensitrelvir, which may explain the rebound often seen with paxlovid. 3CLpro is crucial for processing viral.. | ||
Aug 5 2024 |
et al., BMC Infectious Diseases, doi:10.1186/s12879-024-09679-1 | Nirmatrelvir/ritonavir treatment and the risk of post-COVID condition over 180 days in Malaysia |
| 30% higher long COVID (p=0.34). Retrospective 2,524 adult COVID-19 outpatients in Malaysia showing no significant difference in post-COVID condition (PCC) at 3 months and 6 months with paxlovid treatment. | ||
Aug 1 2024 |
et al., BMC Infectious Diseases, doi:10.1186/s12879-024-09482-y | Paxlovid reduces the 28-day mortality of patients with COVID-19: a retrospective cohort study |
| Retrospective 1,018 hospitalized COVID-19 patients in China, showing lower 28-day mortality with paxlovid. This study contains significant confounding issues that could potentially account for the entire benefit seen: The control group in.. | ||
Jul 18 2024 |
et al., New England Journal of Medicine, doi:10.1056/NEJMoa2309002 | Oral Nirmatrelvir–Ritonavir as Postexposure Prophylaxis for Covid-19 |
| Delayed publication of the EPIC-PEP RCT showing no significant benefit with paxlovid for post-exposure prophylaxis. Results were available in 2023 on clinicaltrials.gov [Pfizer]. | ||
Jul 15 2024 |
et al., The Lancet Infectious Diseases, doi:10.1016/S1473-3099(24)00353-0 | Comparative effectiveness of combination therapy with nirmatrelvir–ritonavir and remdesivir versus monotherapy with remdesivir or nirmatrelvir–ritonavir in patients hospitalised with COVID-19: a target trial emulation study |
| 34% lower mortality (p=0.006), 37% lower ICU admission (p=0.26), and 45% lower need for oxygen therapy (p=0.03). Target trial emulation study of 18,196 hospitalized COVID-19 patients in Hong Kong showing significantly higher ICU admission and AKI with remdesivir + paxlovid compared with paxlovid alone, and lower mortality and ventilatory support wit.. | ||
Jul 6 2024 |
et al., Quantitative Biology, doi:10.1002/qub2.60 | Assessing the inhibition efficacy of clinical drugs against the main proteases of SARS‐CoV‐2 variants and other coronaviruses |
| In vitro study showing that leritrelvir and GC376 remained effective against some nirmatrelvir- and ensitrelvir-resistant Mpro mutants. Leritrelvir showed better broad-spectrum activity against other pathogenic coronaviruses compared to e.. | ||
Jul 5 2024 |
et al., Journal of Medicinal Chemistry, doi:10.1021/acs.jmedchem.4c01342 | Fixing the Achilles Heel of Pfizer’s Paxlovid for COVID-19 Treatment |
| Perspective article discussing the development of ibuzatrelvir (PF-07817883) as a SARS-CoV-2 main protease (Mpro) inhibitor which addresses the metabolic instability of nirmatrelvir and does not require co-administration with ritonavir, t.. | ||
Jun 27 2024 |
et al., In Vivo, doi:10.21873/invivo.13637 | Association of Antiviral Drugs for the Treatment of COVID-19 With Acute Renal Failure |
| Retrospective 176,197 adverse event reports in Japan showing paxlovid and remdesivir associated with increased risk of acute kidney injury (AKI) in COVID-19 patients. | ||
Jun 24 2024 |
et al., Journal of Zhejiang University - SCIENCE B (Biomedicine & Biotechnology, doi:10.1631/jzus.B2300538 | Clinical characteristics and outcomes of hospitalized kidney transplant recipients with COVID-19 infection in China during the Omicron wave: a single-center cohort study |
| 495% higher mortality (p<0.0001). Retrospective 324 hospitalized kidney transplant recipients with COVID-19 showing no significant benefit with molnupiravir, paxlovid, or azvudine. The study was conducted during the omicron wave in China between December 2022 and January .. | ||
Jun 24 2024 |
et al., Current Clinical Microbiology Reports, doi:10.1007/s40588-024-00229-6 | SARS-CoV-2 Resistance to Small Molecule Inhibitors |
| Review of resistance mutations in SARS-CoV-2 3CLpro and nsp12 that could reduce efficacy of antiviral therapies including nirmatrelvir, ensitrelvir, remdesivir, and favipiravir. Authors identify 39 single mutations across 17 critical resi.. | ||
Jun 7 2024 |
et al., JAMA Internal Medicine, doi:10.1001/jamainternmed.2024.2007 | Nirmatrelvir-Ritonavir and Symptoms in Adults With Postacute Sequelae of SARS-CoV-2 Infection |
| RCT 155 adults with long COVID (PASC) showing no significant benefit with paxlovid compared to ritonavir. | ||
Jun 3 2024 |
et al., bioRxiv, doi:10.1101/2024.06.02.596989 | Persistent Neurological Deficits in Mouse PASC Reveal Antiviral Drug Limitations |
| Mouse study showing persistent neurological deficits after SARS-CoV-2 infection, and failure of nirmatrelvir and molnupiravir to prevent neurological damage. Authors found decreased tyrosine hydroxylase expression in the olfactory bulb an.. | ||
May 24 2024 |
et al., The Lancet Infectious Diseases, doi:10.1016/S1473-3099(25)00093-3 (date from preprint) | Molnupiravir or nirmatrelvir-ritonavir versus usual care in patients admitted to hospital with COVID-19 (RECOVERY): a randomised, controlled, open-label, platform trial |
| 1% higher mortality (p=1), 35% lower hospital discharge (p=0.33), and 19% improved viral clearance (p<0.0001). RECOVERY RCT showing no significant differences in mortality, ventilation, or discharge with either molnupiravir (923 patients) or paxlovid (137 patients). Viral load was improved with treatment but did not translate into clinical benefit.. | ||
Sep 17 2024 |
et al., Journal of Virology, doi:10.1128/jvi.00905-24 (date from preprint) | Efficacy of late-onset antiviral treatment in immunocompromised hosts with persistent SARS-CoV-2 infection |
| Mouse study showing that late-onset treatment with paxlovid, molnupiravir, or 4'-FlU significantly reduced persistent SARS-CoV-2 infection in immunocompromised mice, with 4'-FlU being most effective. Authors used a CD4+ and CD8+ T cell-de.. | ||
Apr 30 2024 |
et al., Infections in the immunosuppressed and immunocompromised host, doi:10.1164/ajrccm-conference.2024.209.1_MeetingAbstracts.A2917 | Effectiveness of Azvudine and Nirmatrelvir-ritonavir in Kidney Transplant Recipients With COVID-19: A Retrospective Cohort Study |
| 272% higher progression (p=0.05). Retrospective 148 hospitalized kidney transplant patients with COVID-19 in China showing lower risk of disease progression with azvudine treatment compared, and higher risk with paxlovid treatment. | ||
Apr 28 2024 |
et al., Preprints, doi:10.20944/preprints202404.1825.v1 | In Silico Comparative Analysis of Ivermectin and Nirmatrelvir Inhibitors Interacting with the SARS-CoV-2 Main Protease |
| In silico study showing that ivermectin and nirmatrelvir interact with the SARS-CoV-2 main protease (Mpro). Authors used molecular docking and 100ns molecular dynamics simulations to investigate the binding interactions. Nirmatrelvir form.. | ||
Apr 5 2024 |
et al., Immunity, Inflammation and Disease, doi:10.1002/iid3.1232 | Nirmatrelvir and ritonavir combination against COVID‐19 caused by omicron BA.2.2 in the elderly: A single‐center large observational study |
| PSM retrospective 648 elderly COVID-19 patients showing no significant difference in time to viral clearance with paxlovid. However, in subgroup analysis of patients treated within 10 days of symptom onset, treatment was associated with f.. | ||
Apr 4 2024 |
et al., New England Journal of Medicine, doi:10.1056/NEJMoa2309003 | Nirmatrelvir for Vaccinated or Unvaccinated Adult Outpatients with Covid-19 |
| Delayed publication for EPIC-SR showing no significant difference in time to sustained alleviation. Selected results were first made available in December 2021 [Pfizer]. | ||
Apr 2 2024 |
et al., bioRxiv, doi:10.1101/2024.04.01.587566 | Distal Protein-Protein Interactions Contribute to SARS-CoV-2 Main Protease Substrate Binding and Nirmatrelvir Resistance |
| In vitro and crystallographic study reveals that the L50F mutation in SARS-CoV-2 main protease (Mpro) can restore the reduced enzymatic activity caused by nirmatrelvir resistance mutations E166A/L167F through enhanced protein-protein inte.. | ||
Mar 31 2024 |
et al., Pharmacoepidemiology and Risk Management, doi:10.56142/perm.24.0006 | Safety Monitoring of Oral Antiviral COVID-19 Treatment Using Korea Adverse Event Reporting System (KAERS) Database |
| Safety analysis of paxlovid and molnupiravir. Disproportionality analysis found significant signals for paxlovid with sensory abnormalities, interactions, fecal abnormalities, and signals not on the label including olfactory nerve disorde.. | ||
Mar 29 2024 |
et al., Infection and Drug Resistance, doi:10.2147/IDR.S443574 | Reduced Viral Shedding Time in High-Risk COVID-19 Patients Infected by Omicron and Treated with Paxlovid: A Real-World Study from China |
| Retrospective 3,159 high risk COVID-19 patients in China showing no significant difference for viral clearance with multivariable Cox regression, but significantly faster viral clearance with logistic regression. Cox results account for t.. | ||
Mar 29 2024 |
et al., Scientific Reports, doi:10.1038/s41598-024-57633-7 | Predictors of nirmatrelvir–ritonavir receipt among COVID-19 patients in a large US health system |
| Retrospective 319,900 treatment-eligible COVID-19 patients showing relatively low use of paxlovid and significant socioeconomic disparities. Treated patients were more likely to be from affluent neighborhoods, be up to date on vaccination.. | ||
Mar 13 2024 |
et al., JAMA Network Open, doi:10.1001/jamanetworkopen.2024.1765 | COVID-19 Rebound After VV116 vs Nirmatrelvir-Ritonavir Treatment |
| RCT showing high rates of viral and symptom rebound with both paxlovid and deuremidevir (VV116). There are multiple potential reasons, with one being the highly specific targets within viral replication (Mpro and RdRp respectively). Paxlo.. | ||
Mar 4 2024 |
et al., Medical Review, doi:10.1515/mr-2023-0068 | Clinical characteristics and pharmacokinetics of PAXLOVID in COVID-19 patients with hematological tumor |
| Retrospective 12 hospitalized COVID-19 patients with hematological malignancies showing one death and two cases of liver injury with paxlovid treatment. The study focused on pharmacokinetics, finding nirmatrelvir concentrations approximat.. | ||
Mar 2 2024 |
et al., Antiviral Research, doi:10.1016/j.antiviral.2024.105840 | The host-targeted antiviral drug Zapnometinib exhibits a high barrier to the development of SARS-CoV-2 resistance |
| In vitro study showing that molnupiravir and paxlovid induced resistant variants in SARS-CoV-2 during serial passaging, while the host-directed antiviral zapnometinib did not. Authors found that molnupiravir did not lead to abrogated vira.. | ||
Feb 29 2024 |
et al., Antiviral Research, doi:10.1016/j.antiviral.2024.105814 | Generation and evaluation of protease inhibitor-resistant SARS-CoV-2 strains |
| In vitro and animal study showing that SARS-CoV-2 can develop resistance to nirmatrelvir and ensitrelvir. Authors generated resistant viral strains through repeated passaging with both drugs. For nirmatrelvir, they identified three differ.. | ||
Feb 21 2024 |
et al., Journal of Antimicrobial Chemotherapy, doi:10.1093/jac/dkae042 | COVID-19 hospitalization risk after outpatient nirmatrelvir/ritonavir use, January to August 2022, North Carolina |
| 84% lower hospitalization (p=0.002). EHR retrospective 44,671 patients with 4,948 receiving paxlovid, showing lower hospitalization with treatment. | ||
Feb 9 2024 |
et al., Scientific Reports, doi:10.1038/s41598-024-53862-y | Comparison of azvudine, molnupiravir, and nirmatrelvir/ritonavir in adult patients with mild-to-moderate COVID-19: a retrospective cohort study |
| Retrospective 157 hospitalized mild-to-moderate COVID-19 patients showing no significant differences between azvudine, molnupiravir, and paxlovid for time to viral clearance and length of hospitalization. | ||
Feb 9 2024 |
et al., eClinicalMedicine, doi:10.1016/j.eclinm.2024.102468 | Antiviral effectiveness and survival correlation of azvudine and nirmatrelvir/ritonavir in elderly severe patients with COVID-19: a retrospective real-world study |
| 13% lower mortality (p=0.67) and 22% higher progression (p=0.49). Retrospective 249 elderly patients with severe COVID-19, 128 treated with azvudine, 66 treated with paxlovid, and 55 receiving neither treatment, showing no significant differences for Ct value changes, progression, or survival for either.. | ||
Feb 9 2024 |
et al., Kidney International Reports, doi:10.1016/j.ekir.2024.02.009 | Effectiveness of molnupiravir and nirmatrelvir–ritonavir in CKD patients with COVID-19 |
| 22% lower mortality (p=0.05), 1% higher need for oxygen therapy (p=0.96), and 14% lower ICU admission (p=0.5). Retrospective emulated target trial of hospitalized COVID-19 patients with chronic kidney disease in Hong Kong showing lower mortality with molnupiravir and paxlovid treatment. No significant reduction was found in ICU admission or ventil.. | ||
Jan 31 2024 |
et al., The Journal of Infection in Developing Countries, doi:10.3855/jidc.18138 | Clinical characteristics and risk factors of non-mild outcomes in patients with Omicron variant COVID-19 in Shanghai, China |
| 79% worse viral clearance (p<0.0001). Retrospective 311 COVID-19 patients in China showing significantly slower viral clearance with paxlovid in multivariable analysis. | ||
Jan 31 2024 |
et al., Technology in Cancer Research & Treatment, doi:10.1177/15330338241248573 | Impact of Anti-angiogenic Drugs on Severity of COVID-19 in Patients with Non-Small Cell Lung Cancer |
| 79% higher mortality (p=0.42). Retrospective 166 hospitalized NSCLC patients with COVID-19 showing no significant difference in mortality with paxlovid or azvudine in univariate analysis. | ||
Jan 26 2024 |
et al., JAMA Health Forum, doi:10.1001/jamahealthforum.2023.5044 | Clinical Risk and Outpatient Therapy Utilization for COVID-19 in the Medicare Population |
| Analysis of Medicare beneficiaries in 2022 showing that outpatient COVID-19 treatments like antivirals and monoclonal antibodies were disproportionately used by patients at lower risk of severe infection and outcomes. Retrospective studie.. | ||
Jan 24 2024 |
et al., Research Square, doi:10.21203/rs.3.rs-3876022/v1 | Oral antivirals for COVID-19 among patients with cancer |
| Retrospective 67 cancer outpatients treated with nirmatrelvir/ritonavir or molnupiravir, compared to 56 untreated concurrent controls, reporting lower mortality with treatment. However, Figure 3 shows the opposite results for invasive mec.. | ||
Jan 24 2024 |
et al., npj Viruses, doi:10.1038/s44298-023-00013-1 | The anti-COVID-19 drug Paxlovid crosses biological barriers of the placenta and brain in rats |
| Animal study showing that paxlovid crosses biological barriers of the blood-placenta and blood-brain in rats. Authors developed a multisite microdialysis coupled with UHPLC-MS/MS to monitor nirmatrelvir levels in maternal blood, conceptus.. | ||
Jan 22 2024 |
et al., PLOS Medicine, doi:10.1371/journal.pmed.1004711 (date from preprint) | Effect of Paxlovid treatment during acute COVID-19 on Long COVID onset: An EHR-based target trial emulation from the N3C and RECOVER consortia |
| 1% lower long COVID (p=0.59). Retrospective 445,378 high-risk outpatients showing no significant difference in post-acute sequelae of COVID-19 (PASC) incidence with paxlovid treatment. Subgroup analysis showed benefits for cognitive and fatigue symptoms. The study use.. | ||
Jan 15 2024 |
et al., Toxics, doi:10.3390/toxics12010073 | Ritonavir Has Reproductive Toxicity Depending on Disrupting PI3K/PDK1/AKT Signaling Pathway |
| In vitro study on boar spermatozoa showing that the HIV drug ritonavir (part of paxlovid and xiannuoxin) causes reproductive toxicity by disrupting the PI3K/PDK1/AKT signaling pathway. Ritonavir suppressed sperm functions including motili.. | ||
Jan 8 2024 |
et al., SSRN, doi:10.2139/ssrn.4683854 | Comparative Efficacy of Combination Treatment with Nirmatrelvir-Ritonavir and Remdesivir Versus Remdesivir Monotherapy in Hospitalised COVID-19 Patients: A Target Trial Emulation Study |
| 37% lower mortality (p=0.004), 40% higher ICU admission (p=0.78), and 20% lower need for oxygen therapy (p=0.66). Retrospective 1,843 hospitalized COVID-19 patients in Hong Kong showing lower mortality with paxlovid. All patients received remdesivir. No significant difference was found for ICU admission or ventilatory support. | ||
Jan 4 2024 |
et al., Journal of Medical Virology, doi:10.1002/jmv.29333 | Association of nirmatrelvir for acute SARS‐CoV‐2 infection with subsequent Long COVID symptoms in an observational cohort study |
| 14% higher long COVID (p=0.4). Retrospective 4,684 COVID+ patients mostly in the USA, 988 treated with paxlovid, showing higher risk of long COVID with treatment, without statistical significance. | ||
Dec 31 2023 |
, E., Aging and disease, doi:10.14336/AD.2023.0318 | Mitigating COVID-19 Mortality and Morbidity in China's Aging Population: A Focus on Available Medications and Future Developments |
| Review focusing on 3CL protease inhibitors. First generation inhibitors like paxlovid and simnotrelvir require boosting with ritonavir, which can cause drug-drug interactions and other issues. Second generation inhibitors like ensitrelvir.. | ||
Dec 22 2023 |
et al., Medicine, doi:10.1097/MD.0000000000036714 | Impact of Paxlovid on in-hospital outcomes and post-COVID-19 condition in adult patients infected with SARS-CoV-2 Omicron variant: A non-randomized controlled clinical trial |
| 12% shorter hospitalization (p=0.09), 35% faster recovery (p<0.0001), 58% faster viral clearance (p<0.0001), and 47% lower long COVID (p=0.04). Prospective study of 320 COVID-19 patients infected with the SARS-CoV-2 Omicron variant in China, showing improved viral clearance and symptom resolution with 5 days of paxlovid treatment. Authors perform multivariable analysis for post-c.. | ||
Dec 19 2023 |
et al., Microorganisms, doi:10.3390/microorganisms12010002 | Assessing Genomic Mutations in SARS-CoV-2: Potential Resistance to Antiviral Drugs in Viral Populations from Untreated COVID-19 Patients |
| Analysis of naturally occurring SARS-CoV-2 mutations in genomic regions targeted by remdesivir, molnupiravir, and paxlovid in 4,155 antiviral-naive COVID-19 patients. Authors identified 84 amino-acid substitutions in Nsp12 (RdRp; target o.. | ||
Nov 30 2023 |
et al., Clinical Interventions in Aging, doi:10.2147/cia.s431271 | Factors Affecting Mortality in Elderly Hypertensive Hospitalized Patients with COVID-19: A Retrospective Study |
| 232% higher mortality (p=0.006). Retrospective 748 elderly hospitalized COVID-19 patients in China showing increased risk of death with paxlovid/molnupiravir. Multivariate analysis showed that paxlovid/molnupiravir was independently associated with higher in-hospital mor.. | ||
Nov 30 2023 |
et al., iScience, doi:10.1016/j.isci.2023.108147 | Antiviral efficacy against and replicative fitness of an XBB.1.9.1 clinical isolate |
| In vitro and animal study showing that the SARS-CoV-2 omicron subvariant XBB.1.9.1 has similar antigenicity, antiviral susceptibility, and replicative ability compared to XBB.1.5. Casirivimab, imdevimab, tixagevimab, cilgavimab, sotrovima.. | ||
Oct 31 2023 |
et al., Annals of Internal Medicine, doi:10.7326/M23-1394 | Effectiveness of Nirmatrelvir–Ritonavir Against the Development of Post–COVID-19 Conditions Among U.S. Veterans |
| 1% lower long COVID (p=0.75). Retrospective 9,593 veterans in the USA treated with paxlovid, matched to 9,593 untreated controls, showing no significant difference in post-COVID conditions across 31 different conditions. There was lower risk for the combination of 2 s.. | ||
Oct 31 2023 |
et al., International Journal of Infectious Diseases, doi:10.1016/j.ijid.2023.08.003 | Real-world nirmatrelvir-ritonavir outpatient treatment in reducing hospitalization for high-risk patients with COVID-19 during Omicron BA.4, BA.5 and XBB subvariants dominance in Malaysia: A retrospective cohort study |
| 36% lower hospitalization (p=0.03). Retrospective 31,483 high-risk COVID-19 outpatients in Malaysia showing lower risk of hospitalization with paxlovid. | ||
Oct 31 2023 |
et al., Biosafety and Health, doi:10.1016/j.bsheal.2023.08.004 | Viral rebound and safety of nirmatrelvir/ritonavir for lung-transplant recipients infected with SARS-CoV-2 |
| Prospective study of 4 lung transplant recipients showing viral rebound in all patients after initial paxlovid treatment. All four patients received paxlovid within 2 days of SARS-CoV-2 infection, with viral loads initially decreasing but.. | ||
Oct 23 2023 |
et al., JAMA Internal Medicine, doi:10.1001/jamainternmed.2023.5099 | Nirmatrelvir and Molnupiravir and Post–COVID-19 Condition in Older Patients |
| 7% lower long COVID (p<0.0001). Retrospective 51,658 paxlovid patients in the USA showing a small reduction in long COVID with treatment. Confounding is likely significant as below, and may eliminate the benefit. Results specific to the COVID-19 code should be closer to.. | ||
Oct 21 2023 |
et al., Heliyon, doi:10.1016/j.heliyon.2023.e21387 | Clinical characteristics, outcomes, and risk factors of SARS-CoV-2 breakthrough infections among 572 fully vaccinated (BBIBP-CorV) hospitalized patients |
| 94% higher progression (p=0.007). Retrospective 572 fully vaccinated hospitalized patients in China, showing higher risk with paxlovid use. The composite outcome included intubation, non-invasive respiratory support, ICU admission, and all-cause death. Details for analysi.. | ||
Oct 18 2023 |
et al., Infection and Drug Resistance, doi:10.2147/IDR.S430101 | Clinical Characteristics of Severe COVID-19 Patients During Omicron Epidemic and a Nomogram Model Integrating Cell-Free DNA for Predicting Mortality: A Retrospective Analysis |
| 6% higher mortality (p=0.84). Retrospective 282 severe COVID-19 patients, showing no significant difference in mortality with paxlovid in unadjusted results. | ||
Oct 18 2023 |
et al., BMC Infectious Diseases, doi:10.1186/s12879-023-08620-2 | In-hospital adverse outcomes and risk factors among chronic kidney disease patients infected with the omicron variant of SARS-CoV-2: a single-center retrospective study |
| 10% slower viral clearance (p=0.04). Retrospective 1,978 hospitalized patients in China, showing slower viral clearance with Paxlovid. Authors note improved results in the subgroup of non-severe patients with CKD. | ||
Oct 13 2023 |
et al., Frontiers in Pharmacology, doi:10.3389/fphar.2023.1274294 | Head-to-head comparison of azvudine and nirmatrelvir/ritonavir for the hospitalized patients with COVID-19: a real-world retrospective cohort study with propensity score matching |
| no change in mortality (p=1), 38% higher ventilation (p=0.04), 122% higher ICU admission (p=0.05), and 28% higher progression (p=0.07). PSM retrospective 725 hospitalized COVID-19 patients in China compared the effectiveness and safety of the oral antivirals azvudine and paxlovid. There was no significant difference in the risk of disease progression between groups, but a.. | ||
Oct 12 2023 |
et al., Frontiers in Microbiology, doi:10.3389/fmicb.2023.1280026 | Secondary pulmonary infection and co-infection in elderly COVID-19 patients during the pandemics in a tertiary general hospital in Beijing, China |
| 39% higher mortality (p=0.04). Retrospective 322 hospitalized patients ≥65 in China, showing higher mortality with paxlovid use. Details for analysis of confounding are not provided and authors note use may have been higher for more severe patients. The results for pax.. | ||
Oct 5 2023 |
et al., Journal of Infection and Public Health, doi:10.1016/j.jiph.2023.10.007 | Clinical outcomes of nirmatrelvir-ritonavir use in pregnant women during the Omicron wave of the coronavirus disease 2019 pandemic |
| 633% higher need for oxygen therapy (p=0.05) and 35% faster recovery (p=0.04). Retrospective 85 pregnant patients in Taiwan, 30 treated with paxlovid, showing higher oxygen requirements, not quite reaching statistical significance (p=0.05), and faster recovery. Patients taking paxlovid for less than three consecutiv.. | ||
Oct 2 2023 |
et al., JAMA Network Open, doi:10.1001/jamanetworkopen.2023.36678 | Nirmatrelvir-Ritonavir and COVID-19 Mortality and Hospitalization Among Patients With Vulnerability to COVID-19 Complications |
| 29% lower combined mortality/hospitalization (p=0.02) and 8% lower progression (p=0.37). Retrospective 3,433 high-risk patients and matched controls in Canada showing lower mortality with paxlovid use. Patients were divided into four groups based on risk, with improved results as risk increased. Authors did not exclude all.. | ||
Sep 28 2023 |
et al., Vaccines, doi:10.3390/vaccines11101533 | In Vitro Efficacy of Antivirals and Monoclonal Antibodies against SARS-CoV-2 Omicron Lineages XBB.1.9.1, XBB.1.9.3, XBB.1.5, XBB.1.16, XBB.2.4, BQ.1.1.45, CH.1.1, and CL.1 |
| In vitro study showing sharply reduced neutralization of SARS-CoV-2 variants XBB.1.9.1, XBB.1.9.3, XBB.1.5, XBB.1.16, XBB.2.4, BQ.1.1.45, CH.1.1, and CL.1 with monoclonal antibodies cilgavimab, tixagevimab, imdevimab, etsevimab, casirivim.. | ||
Sep 28 2023 |
et al., Frontiers in Medicine, doi:10.3389/fmed.2023.1238713 | Differences in the severity and mortality risk factors for patients hospitalized for COVID-19 pneumonia between the early wave and the very late stage of the pandemic |
| 18% lower mortality (p=0.61). Retrospective 223 hospitalized patients in China, showing no significant difference in mortality with paxlovid in unadjusted results. | ||
Aug 31 2023 |
et al., Journal of Infection, doi:10.1016/j.jinf.2023.05.012 | Azvudine versus Paxlovid for oral treatment of COVID-19 in Chinese patients with pre-existing comorbidities |
| 175% higher mortality (p=0.11), 200% higher ventilation (p=0.28), and 94% higher progression (p=0.03). Retrospective 2,118 hospitalized COVID-19 patients in China, showing improved results with azvudine vs. paxlovid. | ||
Aug 20 2023 |
et al., npj Antimicrobials and Resistance, doi:10.1038/s44259-023-00009-0 | Rapid resistance profiling of SARS-CoV-2 protease inhibitors |
| In vitro study of SARS-CoV-2 main protease (Mpro) variants showing distinct resistance profiles for the Mpro inhibitors nirmatrelvir, ensitrelvir, and FB2001. Results show significant resistance for multiple variants for nirmatrelvir and .. | ||
Aug 16 2023 |
et al., Phytomedicine, doi:10.1016/j.phymed.2023.155025 | Efficacy and safety of Huashi Baidu granule plus Nirmatrelvir-Ritonavir combination therapy in patients with high-risk factors infected with Omicron (B.1.1.529): A multi-arm single-center, open-label, randomized controlled trial |
| 2% higher severe cases (p=1), 6% lower hospital discharge (p=0.78), and 12% improved viral clearance (p=0.33). RCT 312 hospitalized COVID-19 patients in China, showing no significant difference between paxlovid and Huashi Baidu treatment. Combination therapy showed improved results to either treatment alone. | ||
Aug 7 2023 |
et al., BMJ Open, doi:10.1136/bmjopen-2022-069176 | Platform adaptive trial of novel antivirals for early treatment of COVID-19 In the community (PANORAMIC): protocol for a randomised, controlled, open-label, adaptive platform trial of community novel antiviral treatment of COVID-19 in people at increased risk of more severe disease |
| PANORAMIC protocol paper, published over 9 months after publication of the molnupiravir arm [Butler]. | ||
Jul 23 2023 |
et al., Microorganisms, doi:10.3390/microorganisms11071859 | Composite Interventions on Outcomes of Severely and Critically Ill Patients with COVID-19 in Shanghai, China |
| 29% lower mortality (p=0.16) and 28% worse results (p<0.0001). Retrospective 1,082 severely and critically ill COVID-19 patients in China showing lower 60 day mortality with azvudine. Mortality was also lower with paxlovid, but without statistical significance, and health related quality of life was.. | ||
Jul 10 2023 |
et al., Journal of Korean Medical Science, doi:10.3346/jkms.2023.38.e211 | Effectiveness of Paxlovid, an Oral Antiviral Drug, Against the Omicron BA.5 Variant in Korea: Severe Progression and Death Between July and November 2022 |
| 31% lower mortality (p<0.0001) and 43% lower severe cases (p<0.0001). Retrospective 1,936,925 COVID-19 patients in South Korea, showing lower mortality with paxlovid. | ||
Jun 27 2023 |
et al., medRxiv, doi:10.1101/2023.06.23.23288598 | SARS-CoV-2 virologic rebound with nirmatrelvir-ritonavir therapy |
| 761% worse viral clearance (p=0.04). Prospective study of 127 COVID-19 patients in the USA showing higher risk of replication-competent virologic rebound with paxlovid treatment. Authors note that rebound substantially increases the duration of shedding of replication-compet.. | ||
Jun 26 2023 |
et al., Authorea, Inc., doi:10.22541/au.168777909.90198442/v1 | Comparison of the Different Medications for COVID-19 in Kidney Transplant Recipients |
| 280% higher ventilation (p=0.32), 659% higher ICU admission (p=0.04), and 207% higher progression (p=0.005). Retrospective 140 kidney transplant patients, showing higher risk of AKI with paxlovid compared with azvudine. There were more severe cases in the paxlovid group at baseline. | ||
Jun 22 2023 |
et al., Elsevier BV, doi:10.2139/ssrn.4488145 | Clinical Prognosis and Risk Factors of Death for Covid-19 Patients Complicated with Coronary Heart Disease/Diabetes/Hypertension-A Retrospective, Real-World Study |
| 2% lower mortality (p=0.04). Retrospective 1,281 COVID-19 patients with comorbidities in China, showing 2% lower mortality with paxlovid. | ||
Jun 17 2023 |
et al., Clinical Microbiology and Infection, doi:10.1016/j.cmi.2023.06.016 | Real-world effectiveness of nirmatrelvir/ritonavir against COVID-19 hospitalisations and severe COVID-19 in community-dwelling elderly Singaporeans during Omicron BA.2, BA.4/5 and XBB transmission |
| 14% lower severe cases (p=0.63) and 35% lower hospitalization (p=0.002). Retrospective 3,959 paxlovid patients and 139,379 untreated controls, showing lower hospitalization with treatment. Contraindicted patients were excluded. | ||
Jun 16 2023 |
et al., Research Square, doi:10.21203/rs.3.rs-3003449/v1 | The association of mortality with vaccination and underlying disease among COVID-19 patients in long term care hospitals at Daegu and Gyeonsangbuk-do in Korea |
| 5% lower mortality (p=0.86). Retrospective 2,507 COVID-19 patients at 18 long term care hospitals with COVID-19 outbreaks in Korea, showing no significant difference in mortality with paxlovid treatment. Note that this study is less affected by the typical confoundin.. | ||
May 14 2023 |
et al., medRxiv, doi:10.1101/2023.05.10.23289325 | Composite interventions on outcomes of severely and critically ill patients with COVID-19 in Shanghai, China |
| 29% lower mortality (p=0.16) and 28% worse recovery (p<0.0001). Retrospective 1,082 hospitalized COVID-19 patients in China, showing lower mortality and worse quality of life with paxlovid. | ||
May 10 2023 |
et al., Elsevier BV, doi:10.2139/ssrn.4444431 | Real-Life Comparison of Mortality in Non-Hospitalised Patients with SARS-CoV-2 Infection at Risk for Clinical Progression Treated with Molnupiravir or Nirmatrevir Plus Ritonavir During the Omicron Era in Italy: A Nationwide, Observational Study |
| 32% lower mortality (p=0.0001). Prospective study of 17,977 outpatients treated with molnupiravir and 11,576 treated with paxlovid, showing significant mortality with both treatments, and lower mortality with paxlovid. | ||
Apr 30 2023 |
et al., Annals of Internal Medicine, doi:10.7326/M22-3057 | Effectiveness of Molnupiravir and Nirmatrelvir–Ritonavir in Hospitalized Patients With COVID-19 |
| 23% lower mortality (p=0.001), 3% higher ventilation (p=0.89), and 8% higher ICU admission (p=0.82). Target trial emulation retrospective with 7,119 patients in Hong Kong, showing lower mortality with paxlovid, but no significant difference for ventilation and ICU admission. See also [acpjournals.org], [acpjournals.org]. | ||
Apr 25 2023 |
et al., Frontiers in Pediatrics, doi:10.3389/fped.2023.1160929 | Clinical efficacy analysis of paxlovid in children with hematological diseases infected with the omicron SARS-CoV-2 new variant |
| 51% worse recovery (p=0.008) and 58% faster viral clearance (p=0.03). Retrospective 20 pediatric hematological disease patients in China, showing faster viral clearance with paxlovid, but slower resolution of fever. | ||
Apr 6 2023 |
et al., medRxiv, doi:10.1101/2023.04.05.23288196 | Incidence of Symptoms Associated with Post-Acute Sequelae of SARS-CoV-2 infection in Non-Hospitalized Vaccinated Patients Receiving Nirmatrelvir-Ritonavir |
| 20% lower long COVID (p=0.01). TriNetX retrospective 1,004 paxlovid patients and matched controls, showing lower risk of PASC with treatment. | ||
Mar 31 2023 |
et al., Science Advances, doi:10.1126/sciadv.ade8778 | Transmissible SARS-CoV-2 variants with resistance to clinical protease inhibitors |
| Analysis of naturally occurring SARS-CoV-2 main protease variants for resistance to nirmatrelvir and ensitrelvir. Authors identified multiple single amino acid mutations that confer significant resistance to these drugs. Phylogenetic anal.. | ||
Mar 23 2023 |
et al., Clinical Therapeutics, doi:10.1016/j.clinthera.2023.03.012 | Prevalence of Potential Drug-Drug Interactions with Ritonavir-Containing COVID-19 therapy in the United States: An Analysis of the National Health and Nutrition Examination Survey |
| Analysis of 15,685 adults in the USA estimating that 29.3% of the population risk a major or contraindicated drug interaction with paxlovid, which may increase significantly for patients over 60 or with several comorbidities that also inc.. | ||
Mar 22 2023 |
et al., Frontiers in Pharmacology, doi:10.3389/fphar.2023.1147980 | Nirmatrelvir/ritonavir for patients with SARS-CoV-2 infection and impaired kidney function during the Omicron surge |
| 31% lower mortality (p=0.5), 44% lower progression (p=0.04), 14% lower ICU admission (p=0.61), and 9% shorter hospitalization (p=0.03). Retrospective 195 patients with impaired kidney function in China, showing lower combined mortality/ICU/cardiovascular events, and improved viral clearance with paxlovid. | ||
Mar 15 2023 |
et al., The Lancet Infectious Diseases, doi:10.1016/S1473-3099(23)00118-4 | Effectiveness of nirmatrelvir–ritonavir in preventing hospital admissions and deaths in people with COVID-19: a cohort study in a large US health-care system |
| 40% higher mortality (p=0.34), 84% lower combined mortality/ICU admission (p=0.03), and 54% lower combined mortality/hospitalization (p=0.03). Retrospective 7,274 outpatients in the USA treated with paxlovid and matched controls, showing lower combined hospitalization/death with treatment. With a small percentage of eligible patients receiving treatment, confounding by indicatio.. | ||
Mar 13 2023 |
et al., Proceedings of the National Academy of Sciences, doi:10.1073/pnas.2221857120 | Computational prediction of interactions between Paxlovid and prescription drugs |
| In silico analysis of drug-drug interactions for paxlovid. From 2,248 prescription drugs, 1,628 were predicted to have 2,445 interactions with nirmatrelvir and/or ritonavir (673 for nirmatrelvir and 1,403 ritonavir). For 873 drugs, author.. | ||
Feb 22 2023 |
et al., Journal of Infection, doi:10.1016/j.jinf.2023.02.029 | Molnupiravir and nirmatrelvir-ritonavir reduce mortality risk during post-acute COVID-19 phase |
| 28% lower mortality (p<0.0001), 24% lower hospitalization (p<0.0001), and 18% lower progression (p<0.0001). Retrospective 30,040 hospitalized patients in Hong Kong, showing lower mortality with paxlovid treatment. Patients with contraindications to paxlovid were not excluded. | ||
Feb 10 2023 |
et al., Journal of Antimicrobial Chemotherapy, doi:10.1093/jac/dkad027 | Efficacy comparison of 3CL protease inhibitors ensitrelvir and nirmatrelvir against SARS-CoV-2 in vitro and in vivo |
| In vitro and animal study comparing nirmatrelvir and ensitrelvir, showing similar efficacy in vitro, and equal or better efficacy of ensitrelvir in vivo (with similar unbound-drug plasma concentrations). | ||
Feb 9 2023 |
et al., The Lancet Infectious Diseases, doi:10.1016/S1473-3099(23)00011-7 | Real-world use of nirmatrelvir–ritonavir in outpatients with COVID-19 during the era of omicron variants including BA.4 and BA.5 in Colorado, USA: a retrospective cohort study |
| Retrospective 28,167 patients in the USA demonstrating confounding. The large difference shown in Figure 1 at day 0 indicates that the groups are not comparable (32 control hospitalizations versus 0 for paxlovid at day 0) and suggests imm.. | ||
Feb 8 2023 |
, A., Clinical and Experimental Medicine, doi:10.1007/s10238-023-01010-7 | On the choice of Molnupiravir and Paxlovid as the only antivirals permitted for COVID-19 infection in Australia |
| Letter to the editor discussing the choice of molnupiravir and paxlovid as the only approved COVID-19 antiviral therapies in Australia in 2022, despite other therapies showing higher efficacy and lower cost in clinical trials. The author .. | ||
Feb 5 2023 |
et al., The Lancent Regional Health, doi:10.1016/j.lanwpc.2023.100694 | Efficacy and safety of Paxlovid in severe adult patients with SARS-Cov-2 infection: a multicenter randomized controlled study |
| 38% lower mortality (p=0.57), 67% higher ventilation (p=0.44), 10% longer ICU admission (p=0.8), and 27% higher progression (p=0.58). RCT 264 patients in China, showing no significant difference in outcomes with paxlovid. | ||
Jan 31 2023 |
et al., American Journal of Transplantation, doi:10.1016/j.ajt.2022.12.004 | Paxlovid associated with decreased hospitalization rate among adults with COVID-19 — United States, April–September 2022 |
| 51% lower hospitalization (p<0.0001). Retrospective 699,848 outpatients with COVID-19 showing lower hospitalization with paxlovid. | ||
Jan 25 2023 |
et al., Journal of Infection, doi:10.1016/j.jinf.2023.02.012 | Real-world effectiveness of molnupiravir, nirmatrelvir-ritonavir, and sotrovimab on preventing hospital admission among higher-risk patients with COVID-19 in Wales: A retrospective cohort study |
| 41% lower combined mortality/hospitalization (p=0.04). Retrospective high risk outpatients in the UK, showing lower hospitalization/death with paxlovid treatment. Residual confounding is likely with adjustments having no detail on specific comorbidities. Patients with contraindications for pa.. | ||
Jan 22 2023 |
et al., medRxiv, doi:10.1101/2023.01.20.23284849 | Comparative effectiveness of Paxlovid versus sotrovimab and molnupiravir for preventing severe COVID-19 outcomes in non-hospitalised patients: observational cohort study using the OpenSAFELY platform |
| 4% higher combined mortality/hospitalization (p=0.91). OpenSAFELY retrospective 7,683 outpatients in the UK, showing no significant difference in hospitalization/death between paxlovid and sotrovimab. | ||
Jan 16 2023 |
et al., Cureus, doi:10.7759/cureus.33831 | Paxlovid-Induced Symptomatic Bradycardia and Syncope |
| Case report of a 71-year-old female who developed symptomatic bradycardia, syncopal episodes, and sinus pause after taking paxlovid (nirmatrelvir/ritonavir) for mild COVID-19 infection. | ||
Jan 11 2023 |
et al., Science Translational Medicine, doi:10.1126/scitranslmed.abq7360 | SARS-CoV-2 3CLpro mutations selected in a VSV-based system confer resistance to nirmatrelvir, ensitrelvir, and GC376 |
| In vitro and in silico study showing selection of resistant mutations with nirmatrelvir use. Several mutations were identified that confer resistance to 3CLpro inhibitors nirmatrelvir, ensitrelvir, and GC376. Authors note that most of the.. | ||
Jan 10 2023 |
et al., mBio, doi:10.1128/mbio.02815-22 | The Substitutions L50F, E166A, and L167F in SARS-CoV-2 3CLpro Are Selected by a Protease Inhibitor In Vitro and Confer Resistance To Nirmatrelvir |
| In vitro study showing that SARS-CoV-2 can develop resistance to 3CLpro inhibitors, including nirmatrelvir (paxlovid), through a combination of 3CLpro mutations (L50F, E166A, L167F). Authors passaged SARS-CoV-2 in the presence of increasi.. | ||
Dec 31 2022 |
et al., Journal of Infection, doi:10.1016/j.jinf.2022.10.002 | Efficacy of Paxlovid in patients with acute kidney injury who developed COVID-19 |
| 9% lower mortality (p=0.83), 30% lower ventilation (p=0.58), 11% lower ICU admission (p=0.55), and 20% shorter hospitalization (p=0.02). Retrospective 104 AKI patients with COVID-19 in China, 61 treated with paxlovid, showing faster viral clearance and shorter hospitalization with treatment, but no significant difference in morality or ICU admission. | ||
Dec 23 2022 |
et al., Science Advances, doi:10.1126/sciadv.add7197 | Nirmatrelvir-resistant SARS-CoV-2 variants with high fitness in an infectious cell culture system |
| In vitro study showing that SARS-CoV-2 can develop high-level resistance to the oral protease inhibitor nirmatrelvir while retaining fitness in cell culture. Authors identified combinations of substitutions in the SARS-CoV-2 main protease.. | ||
Dec 13 2022 |
et al., Annals of Internal Medicine, doi:10.7326/M22-2141 (date from preprint) | Nirmatrelvir Plus Ritonavir for Early COVID-19 in a Large U.S. Health System |
| 71% lower mortality (p=0.006), 44% lower combined mortality/hospitalization (p=0.0001), and 40% lower hospitalization (p=0.001). IPW retrospective 44,551 outpatients age 50+ in the USA, showing lower mortality and hospitalization with paxlovid treatment. | ||
Dec 6 2022 |
et al., medRxiv, doi:10.1101/2022.12.05.22283134 | Effectiveness of COVID-19 treatment with nirmatrelvir-ritonavir or molnupiravir among U.S. Veterans: target trial emulation studies with one-month and six-month outcomes |
| 58% lower mortality (p=0.0004), 56% lower ICU admission (p=0.03), and 7% lower hospitalization (p=0.53). Retrospective 112,380 high-risk patients in the USA, showing lower mortality with paxlovid treatment. The title and headers of Table S14 are conflicting but the data appears to match the title. | ||
Nov 15 2022 |
et al., JAMA Network Open, doi:10.1001/jamanetworkopen.2022.42140 | Prevalence of Contraindications to Nirmatrelvir-Ritonavir Among Hospitalized Patients With COVID-19 at Risk for Progression to Severe Disease |
| Retrospective 62,525 hospitalized COVID-19 patients in France, showing that over 50% of patients that died had a contraindication for the use of paxlovid. Retrospective studies that do not exclude contraindicated patients may significantl.. | ||
Nov 15 2022 |
et al., medRxiv, doi:10.1101/2022.11.14.22282195 | The Paxlovid Rebound Study: A Prospective Cohort Study to Evaluate Viral and Symptom Rebound Differences Between Paxlovid and Untreated COVID-19 Participants |
| 1% improved recovery (p=0.9), 3% improved viral clearance (p=0.73), and 171% worse results (p=0.06). Prospective study of 170 COVID-19 patients in the USA, showing no significant difference in symptomatic and viral recovery times, and higher risk of symptomatic rebound, without statistical significance. There were more elderly patients i.. | ||
Nov 5 2022 |
et al., medRxiv, doi:10.1101/2022.11.03.22281881 | Real-world effectiveness of nirmatrelvir/ritonavir use for COVID-19: A population-based cohort study in Ontario, Canada |
| 50% lower mortality (p<0.0001) and 43% lower combined mortality/hospitalization (p<0.0001). Retrospective 177,545 patients in Canada, 8,876 treated with paxlovid, showing lower mortality and hospitalization with treatment, and declining efficacy over the two time periods analyzed. | ||
Oct 8 2022 |
et al., The Lancet, doi:10.1016/S0140-6736(22)01586-0 | Real-world effectiveness of molnupiravir and nirmatrelvir plus ritonavir against mortality, hospitalisation, and in-hospital outcomes among community-dwelling, ambulatory patients with confirmed SARS-CoV-2 infection during the omicron wave in Hong Kong: an observational study |
| 66% lower mortality (p<0.0001), 38% lower ventilation (p=0.36), 58% higher ICU admission (p=0.08), and 24% lower hospitalization (p<0.0001). PSM retrospective 1,074,856 outpatients in Hong Kong, showing lower mortality and hospitalization with paxlovid. | ||
Oct 7 2022 |
et al., Journal of Medical Virology, doi:10.1002/jmv.28186 | Viral load decrease in SARS‐CoV‐2 BA.1 and BA.2 Omicron sublineages infection after treatment with monoclonal antibodies and direct antiviral agents |
| Observational study of 521 outpatients with mild-to-moderate COVID-19 showing paxlovid had stronger viral load reduction against Omicron BA.1 and BA.2 variants compared to sotrovimab, molnupiravir, and remdesivir, with the exception of co.. | ||
Sep 14 2022 |
et al., medRxiv, doi:10.1101/2022.09.13.22279908 | Real-World Effectiveness of Nirmatrelvir/Ritonavir in Preventing Hospitalization Among Patients With COVID-19 at High Risk for Severe Disease in the United States: A Nationwide Population-Based Cohort Study |
| 73% lower mortality (p<0.0001) and 84% lower hospitalization (p<0.0001). Pfizer retrospective 2,811 high risk COVID-19 patients treated with paxlovid in the US, and 10,849 matched controls, showing lower risk of mortality and hospitalization with treatment. | ||
Sep 6 2022 |
et al., Frontiers in Medicine, doi:10.3389/fmed.2022.980002 | The efficacy of paxlovid in elderly patients infected with SARS-CoV-2 omicron variants: Results of a non-randomized clinical trial |
| 15% faster recovery (p=0.26) and 16% faster viral clearance (p=0.009). Retrospective 106 paxlovid and 36 control patients in China, showing faster viral clearance with treatment. | ||
Aug 28 2022 |
et al., Vaccines, doi:10.3390/vaccines10091409 | Clinical Progression and Outcome of Hospitalized Patients Infected with SARS-CoV-2 Omicron Variant in Shanghai, China |
| 20% faster viral clearance (p=0.0006). Retrospective 226 patients in China, showing faster viral clearance with paxlovid. Age range and severity differed between groups. | ||
Aug 20 2022 |
et al., Clinical Infectious Diseases, doi:10.1093/cid/ciac673 | Oral Nirmatrelvir and Ritonavir in Non-hospitalized Vaccinated Patients with Covid-19 |
| 95% lower mortality (p=0.002), 39% lower progression (p=0.0001), and 56% lower hospitalization (p=0.02). TriNetX retrospective 1,131 vaccinated COVID-19 patients treated with paxlovid and matched controls, showing lower mortality and hospitalization with treatment. | ||
Aug 9 2022 |
et al., Research Square, doi:10.21203/rs.3.rs-1915291/v1 | H172Y mutation perturbs the S1 pocket and nirmatrelvir binding of SARS-CoV-2 main protease through a nonnative hydrogen bond |
| In silico and in vitro study of the H172Y mutation which significantly reduces paxlovid's inhibitory activity. Monotherapy with paxlovid and selective pressure may favor resistance mutations. | ||
Aug 4 2022 |
et al., Emerging Microbes & Infections, doi:10.1080/22221751.2022.2109517 | Geriatric risk and protective factors for serious COVID-19 outcomes among older adults in Shanghai Omicron wave |
| 12% improved viral clearance (p=0.02). Retrospective 1,377 patients in China, showing significantly faster viral clearance with Paxlovid. Authors analyze progression to severe/critical disease, but do not provide results for Paxlovid. | ||
Aug 1 2022 |
et al., Open Forum Infectious Diseases, doi:10.1093/ofid/ofac389 | Prevalence of Medical Contraindications to Nirmatrelvir/Ritonavir in a Cohort of Hospitalized and Nonhospitalized Patients With COVID-19 |
| Analysis of 66,007 hospitalized and non-hospitalized patients showing a 14% prevalence of medical contraindications with nirmatrelvir/ritonavir for outpatients, 21% for inpatients, and 35% for patients that died. | ||
Jul 23 2022 |
et al., Clinical Infectious Diseases, doi:10.1093/cid/ciac600 | Association of nirmatrelvir/ritonavir treatment on upper respiratory SARS-CoV-2 RT-PCR negative conversion rates among high-risk patients with COVID-19 |
| 79% improved viral clearance (p<0.0001). Retrospective 258 paxlovid patients and 224 patients before paxlovid was available in China, showing significantly faster viral clearance with treatment. Adjusted results are only provided for subgroups (≤5, >5 days from onset). Patients .. | ||
Jul 6 2022 |
et al., medRxiv, doi:10.1101/2022.07.03.22277169 | Clinical advancement of patients infected with SARS-CoV-2 Omicron variant in Shanghai, China |
| 20% faster viral clearance (p=0.0006). Retrospective 17 paxlovid and 114 lianhuaqingwen patients in China, showing faster viral clearance with paxlovid. | ||
Jun 14 2022 |
, NCT05011513 | Evaluation of Protease Inhibition for COVID-19 in Standard-Risk Patients (EPIC-SR) |
| 52% lower hospitalization (p=0.2), 4% lower progression (p=0.21), 8% faster recovery (p=0.16), and 4% improved viral clearance (p=0.04). Results for the terminated and unpublished (until April 2024) EPIC-SR trial. | ||
Jun 2 2022 |
et al., Clinical Infectious Diseases, doi:10.1093/cid/ciac443 | Effectiveness of Paxlovid in Reducing Severe COVID-19 and Mortality in High Risk Patients |
| 46% lower severe cases (p=0.0002). Retrospective 180,351 patients in Israel, 4,737 treated with paxlovid, showing significantly lower combined severe COVID-19 / mortality with treatment. | ||
Jun 1 2022 |
et al., New England Journal of Medicine, doi:10.1056/NEJMoa2204919 (date from preprint) | Nirmatrelvir Use and Severe Covid-19 Outcomes during the Omicron Surge |
| 56% lower mortality (p=0.37) and 57% lower hospitalization (p=0.1). Retrospective 109,254 patients in Israel, 3,902 treated with nirmatrelvir, showing lower mortality and hospitalization with treatment for the subgroup of patients >65. Authors only provide subgroup results. | ||
May 24 2022 |
et al., Clinical Infectious Diseases, doi:10.1093/cid/ciac687 (date from preprint) | Impact of the use of oral antiviral agents on the risk of hospitalization in community COVID-19 patients |
| 19% lower combined mortality/ICU admission (p=0.45) and 21% lower hospitalization (p=0.01). Propensity score weighted retrospective of 93,883 outpatients in Hong Kong, 5,808 treated with molnupiravir and 4,921 treated with paxlovid, showing higher hospitalization and higher combined mortality/mechanical ventilation/ICU admission.. | ||
May 20 2022 |
et al., The Lancet Infectious Diseases, doi:10.1016/S1473-3099(22)00507-2 (date from preprint) | Real-world effectiveness of early molnupiravir or nirmatrelvir–ritonavir in hospitalised patients with COVID-19 without supplemental oxygen requirement on admission during Hong Kong's omicron BA.2 wave: a retrospective cohort study |
| 66% lower mortality (p<0.0001), 3% lower ventilation (p=0.96), 43% lower progression (p<0.0001), and 4% shorter hospitalization (p=0.32). PSM retrospective 40,776 patients in Hong Kong, showing lower mortality and lower combined mortality, ventilation, ICU, and oxygen therapy with paxlovid treatment. | ||
Apr 29 2022 |
, Press Release | Pfizer Shares Top-Line Results from Phase 2/3 EPIC-PEP Study of PAXLOVID™ for Post-Exposure Prophylactic Use |
| 25% lower severe cases (p=0.42), 15% lower progression (p=0.2), and 36% fewer symptomatic cases (p=0.12). PEP RCT showing no significant difference with paxlovid post-exposure prophylaxis. Results from [classic.clinicaltrials.gov]. | ||
Apr 4 2022 |
et al., Nature, doi:10.1038/d41586-022-00919-5 | African clinical trial denied access to key COVID drug Paxlovid |
| News reporting that Pfizer denied access to Paxlovid for an independent trial. The press release indicates that Pfizer has denied access for multiple groups [ANTICOV]. Also see [twitter.com]. | ||
Mar 15 2022 |
, Press Release | Pfizer blocking research to generate evidence on optimal use of novel antiviral for COVID-19 patients in low- and middle-income countries |
| ANTICOV reports that Pfizer has denied access to paxlovid for studies proposed by multiple groups. Also see [Ledford]. | ||
Feb 16 2022 |
et al., New England Journal of Medicine, doi:10.1056/NEJMoa2118542 | Oral Nirmatrelvir for High-Risk, Nonhospitalized Adults with Covid-19 |
| 96% lower mortality (p=0.0005), 88% lower hospitalization (p<0.0001), 11% higher progression (p=0.45), and 17% improved recovery (p=0.002). EPIC-HR RCT, 1,039 higher risk patients treated with paxlovid (PF-07321332 + ritonavir) and 1,046 control patients, showing significantly lower mortality and hospitalization with treatment. Some results were reported later in [Hammond], .. | ||
Dec 14 2021 |
, Press Release | Pfizer announces additional phase 2/3 study results confirming robust efficacy of novel COVID-19 oral antiviral treatment candidate in reducing risk or hospitalization or death |
| 70% lower hospitalization (p=0.05). EPIC-SR trial interim results, 428 patients treated with paxlovid (PF-07321332 + ritonavir) and 426 control patients, showing lower hospitalization with treatment. NCT05011513. | ||
Nov 30 2021 |
et al., bioRxiv, doi:10.1101/2021.11.28.4702264 | Main protease mutants of SARS-CoV-2 variants remain susceptible to PF-07321332 |
| In vitro study showing that PF-07321332 maintains efficacy against variants C.37 lambda, B.1.1.318, B.1.2, B.1.351 beta, and P.2 zeta. | ||
Nov 5 2021 |
et al., bioRxiv, doi:10.1101/2021.11.04.467077 | The oral protease inhibitor (PF-07321332) protects Syrian hamsters against infection with SARS-CoV-2 variants of concern |
| In vitro and hamster study showing paxlovid component PF-07321332 effective against four variants of concern, inibits replication of the alpha variant in primary human airway epithelial cells, protected Syrian hamsters against intranasal .. | ||
Jul 30 2021 |
et al., International Journal of Clinical Pharmacy, doi:10.1007/s11096-021-01311-5 | Drug-induced liver injury associated with lopinavir-ritonavir in patients with COVID-19: a disproportionality analysis of U.S. food and drug administration adverse event reporting system (FAERS) data |
| Disproportionality analysis showing higher risk of liver injury with lopinavir/ritonavir for COVID-19 patients. Paxlovid combines nirmatrelvir and ritonavir. | ||
Aug 22 2002 |
et al., Arteriosclerosis, Thrombosis, and Vascular Biology, doi:10.1161/01.atv.0000034707.40046.02 | HIV Protease Inhibitor Ritonavir Induces Cytotoxicity of Human Endothelial Cells |
| In vitro study showing that ritonavir (part of paxlovid) can cause endothelial mitochondrial DNA damage and cell death at concentrations near clinical plasma levels. | ||
References
Ledford et al., African clinical trial denied access to key COVID drug Paxlovid, Nature, doi:10.1038/d41586-022-00919-5.
Liu et al., Efficacy and safety of Paxlovid in severe adult patients with SARS-Cov-2 infection: a multicenter randomized controlled study, The Lancent Regional Health, doi:10.1016/j.lanwpc.2023.100694.
Yu et al., Efficacy and safety of Huashi Baidu granule plus Nirmatrelvir-Ritonavir combination therapy in patients with high-risk factors infected with Omicron (B.1.1.529): A multi-arm single-center, open-label, randomized controlled trial, Phytomedicine, doi:10.1016/j.phymed.2023.155025.
Zhou et al., Nirmatrelvir-resistant SARS-CoV-2 variants with high fitness in an infectious cell culture system, Science Advances, doi:10.1126/sciadv.add7197.
Moghadasi et al., Rapid resistance profiling of SARS-CoV-2 protease inhibitors, npj Antimicrobials and Resistance, doi:10.1038/s44259-023-00009-0.
Jochmans et al., The Substitutions L50F, E166A, and L167F in SARS-CoV-2 3CLpro Are Selected by a Protease Inhibitor In Vitro and Confer Resistance To Nirmatrelvir, mBio, doi:10.1128/mbio.02815-22.
Lopez et al., SARS-CoV-2 Resistance to Small Molecule Inhibitors, Current Clinical Microbiology Reports, doi:10.1007/s40588-024-00229-6.
Zvornicanin et al., Molecular Mechanisms of Drug Resistance and Compensation in SARS-CoV-2 Main Protease: The Interplay Between E166 and L50, bioRxiv, doi:10.1101/2025.01.24.634813.
Vukovikj et al., Impact of SARS-CoV-2 variant mutations on susceptibility to monoclonal antibodies and antiviral drugs: a non-systematic review, April 2022 to October 2024, Eurosurveillance, doi:10.2807/1560-7917.ES.2025.30.10.2400252.
Deschenes et al., Functional and structural characterization of treatment-emergent nirmatrelvir resistance mutations at low frequencies in the main protease (Mpro) reveals a unique evolutionary route for SARS-CoV-2 to gain resistance, The Journal of Infectious Diseases, doi:10.1093/infdis/jiaf294.
Zhou (B) et al., SARS-CoV-2 Mpro inhibitor ensitrelvir: asymmetrical cross-resistance with nirmatrelvir and emerging resistance hotspots, Emerging Microbes & Infections, doi:10.1080/22221751.2025.2552716.
Chen et al., SARS-CoV-2 3CLpro mutations T21I and E166A confer differential resistance to simnotrelvir, bofutrelvir, and ensitrelvir, Journal of Virology, doi:10.1128/jvi.02223-25.
Hoertel et al., Prevalence of Contraindications to Nirmatrelvir-Ritonavir Among Hospitalized Patients With COVID-19 at Risk for Progression to Severe Disease, JAMA Network Open, doi:10.1001/jamanetworkopen.2022.42140.
FDA, Fact sheet for healthcare providers: emergency use authorization for paxlovid, www.fda.gov/media/155050/download.
Thomas et al., Nirmatrelvir-Resistant Mutations in SARS-CoV-2 Mpro Enhance Host Immune Evasion via Cleavage of NF-κB Essential Modulator, bioRxiv, doi:10.1101/2024.10.18.619137.
Kamo et al., Association of Antiviral Drugs for the Treatment of COVID-19 With Acute Renal Failure, In Vivo, doi:10.21873/invivo.13637.
Wang et al., Development and validation of a nomogram to assess the occurrence of liver dysfunction in patients with COVID-19 pneumonia in the ICU, BMC Infectious Diseases, doi:10.1186/s12879-025-10684-1.
Siby et al., Temporal Trends in Serious Adverse Events Associated with Oral Antivirals During the COVID-19 Pandemic: Insights from the FAERS Database (2020–2023), Open Forum Infectious Diseases, doi:10.1093/ofid/ofaf695.1825.
Edelstein et al., SARS-CoV-2 virologic rebound with nirmatrelvir-ritonavir therapy, medRxiv, doi:10.1101/2023.06.23.23288598.
Shah et al., SARS-CoV-2 infectious shedding and rebound among adults with and without oral antiviral use: two case-ascertained prospective household studies, The Lancet Microbe, doi:10.1016/j.lanmic.2025.101227.