Oral Nirmatrelvir for High-Risk, Nonhospitalized Adults with Covid-19
et al., New England Journal of Medicine, doi:10.1056/NEJMoa2118542, EPIC-HR, NCT04960202, Feb 2022
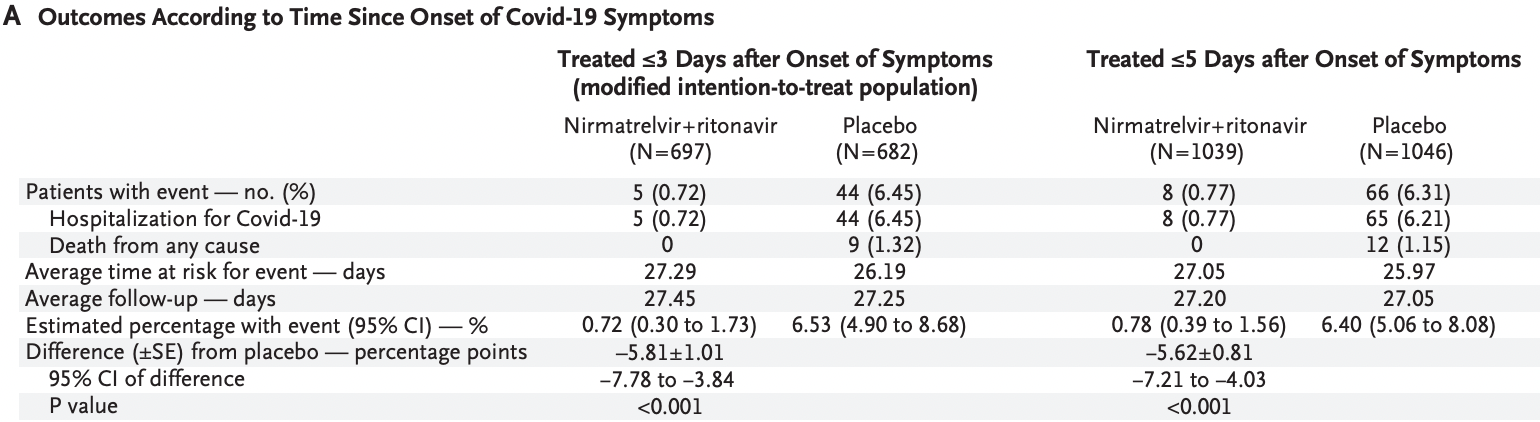
EPIC-HR RCT, 1,039 higher risk patients treated with paxlovid (PF-07321332 + ritonavir) and 1,046 control patients, showing significantly lower mortality and hospitalization with treatment. Some results were reported later in Hammond et al., and some results are only available in the registry.
|
risk of death, 96.0% lower, RR 0.04, p < 0.001, treatment 0 of 1,039 (0.0%), control 12 of 1,046 (1.1%), NNT 87, relative risk is not 0 because of continuity correction due to zero events (with reciprocal of the contrasting arm), within 5 days.
|
|
risk of death, 94.8% lower, RR 0.05, p = 0.002, treatment 0 of 697 (0.0%), control 9 of 682 (1.3%), NNT 76, relative risk is not 0 because of continuity correction due to zero events (with reciprocal of the contrasting arm), within 3 days.
|
|
risk of hospitalization, 87.6% lower, RR 0.12, p < 0.001, treatment 8 of 1,039 (0.8%), control 65 of 1,046 (6.2%), NNT 18, within 5 days.
|
|
risk of hospitalization, 88.9% lower, RR 0.11, p < 0.001, treatment 5 of 697 (0.7%), control 44 of 682 (6.5%), NNT 17, within 3 days.
|
|
risk of progression, 10.8% higher, OR 1.11, p = 0.45, treatment 686, control 674, from registry results, day 28, RR approximated with OR.
|
|
risk of no recovery, 16.7% lower, HR 0.83, p = 0.002, treatment 686, control 674, inverted to make HR<1 favor treatment, sustained resolution, day 28.
|
|
risk of no recovery, 21.3% lower, HR 0.79, p < 0.001, treatment 686, control 674, inverted to make HR<1 favor treatment, sustained alleviation, day 28.
|
|
viral load, 12.1% lower, relative load 0.88, p < 0.001, treatment mean 4.53 (±2.1) n=529, control mean 3.98 (±2.11) n=526, from registry results, day 10.
|
|
viral load, 5.1% lower, relative load 0.95, p = 0.048, treatment mean 5.1 (±2.13) n=529, control mean 4.84 (±2.11) n=526, from registry results, day 14.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Hammond et al., 16 Feb 2022, Double Blind Randomized Controlled Trial, placebo-controlled, multiple countries, peer-reviewed, 12 authors, study period 16 July, 2021 - 9 December, 2021, average treatment delay 2.89 days, trial NCT04960202 (history) (EPIC-HR).
Oral Nirmatrelvir for High-Risk, Nonhospitalized Adults with Covid-19
New England Journal of Medicine, doi:10.1056/nejmoa2118542
BACKGROUND Nirmatrelvir is an orally administered severe acute respiratory syndrome coronavirus 2 main protease (M pro ) inhibitor with potent pan-human-coronavirus activity in vitro.
METHODS We conducted a phase 2-3 double-blind, randomized, controlled trial in which symptomatic, unvaccinated, nonhospitalized adults at high risk for progression to severe coronavirus disease 2019 (Covid-19) were assigned in a 1:1 ratio to receive either 300 mg of nirmatrelvir plus 100 mg of ritonavir (a pharmacokinetic enhancer) or placebo every 12 hours for 5 days. Covid-19-related hospitalization or death from any cause through day 28, viral load, and safety were evaluated.
RESULTS A total of 2246 patients underwent randomization; 1120 patients received nirmatrelvir plus ritonavir (nirmatrelvir group) and 1126 received placebo (placebo group). In the planned interim analysis of patients treated within 3 days after symptom onset (modified intention-to treat population, comprising 774 of the 1361 patients in the full analysis population), the incidence of Covid-19-related hospitalization or death by day 28 was lower in the nirmatrelvir group than in the placebo group by 6.32 percentage points (95% confidence interval [CI], −9.04 to −3.59; P<0.001; relative risk reduction, 89.1%); the incidence was 0.77% (3 of 389 patients) in the nirmatrelvir group, with 0 deaths, as compared with 7.01% (27 of 385 patients) in the placebo group, with 7 deaths. Efficacy was maintained in the final analysis involving the 1379 patients in the modified intention-to-treat population, with a difference of −5.81 percentage points (95% CI, −7.78 to −3.84; P<0.001; relative risk reduction, 88.9%). All 13 deaths occurred in the placebo group. The viral load was lower with nirmaltrelvir plus ritonavir than with placebo at day 5 of treatment, with an adjusted mean difference of −0.868 log 10 copies per milliliter when treatment was initiated within 3 days after the onset of symptoms. The incidence of adverse events that emerged during the treatment period was similar in the two groups (any adverse event, 22.6% with nirmatrelvir plus ritonavir vs. 23.9% with placebo; serious adverse events, 1.6% vs. 6.6%; and adverse events leading to discontinuation of the drugs or placebo, 2.1% vs. 4.2%). Dysgeusia (5.6% vs. 0.3%) and diarrhea (3.1% vs. 1.6%) occurred more frequently with nirmatrelvir plus ritonavir than with placebo.
CONCLUSIONS Treatment of symptomatic Covid-19 with nirmatrelvir plus ritonavir resulted in a risk of progression to severe Covid-19 that was 89% lower than the risk with placebo, without evident safety concerns. (Supported by Pfizer; ClinicalTrials.gov number, NCT04960202.
References
Anand, Ziebuhr, Wadhwani, Mesters, Hilgenfeld, Coronavirus main proteinase (3CL pro ) structure: basis for design of anti-SARS drugs, Science
Docherty, Harrison, Green, Features of 20 133 UK patients in hospital with Covid-19 using the ISARIC WHO Clinical Characterisation Protocol: prospective observational cohort study, BMJ
Dougan, Nirula, Azizad, Bamlanivimab plus etesevimab in mild or moderate Covid-19, N Engl J Med
Gottlieb, Vaca, Paredes, Early remdesivir to prevent progression to severe Covid-19 in outpatients, N Engl J Med
Gupta, Gonzalez-Rojas, Juarez, Early treatment for Covid-19 with SARS-CoV-2 neutralizing antibody sotrovimab, N Engl J Med
Harvey, Carabelli, Jackson, SARS-CoV-2 variants, spike mutations and immune escape, Nat Rev Microbiol
Hilgenfeld, From SARS to MERS: crystallographic studies on coronaviral proteases enable antiviral drug design, FEBS J
Kim, Garg, 'halloran, Risk factors for intensive care unit admission and in-hospital mortality among hospitalized adults identified through the US Coronavirus Disease 2019 (COVID-19)-Associated Hospitalization Surveillance Network (COVID-NET), Clin Infect Dis
Lan, Demets, Discrete sequential boundaries for clinical trials, Biometrika
Mahase, Covid-19: what new variants are emerging and how are they being investigated?, BMJ
Merck, Merck and Ridgeback Biotherapeutics provide update on results from MOVe-OUT study of molnupiravir, an investigational oral antiviral medicine, in at risk adults with mild-to-moderate COV-ID-19
Merck, Merck and Ridgeback's investigational oral antiviral molnupiravir reduced the risk of hospitalization or death by approximately 50 percent compared to placebo for patients with mild or moderate COVID-19 in positive interim analysis of phase 3 study
Ncd-, RisC). Trends in adult body-mass index in 200 countries from 1975 to 2014: a pooled analysis of 1698 population-based measurement studies with 19•2 million participants, Lancet
Ning, Liu, Li, Novel coronavirus (SARS-CoV-2) infection in a renal transplant recipient: case report, Am J Transplant
O'brien, Fleming, A multiple testing procedure for clinical trials, Biometrics
Owen, Allerton, Anderson, An oral SARS-CoV-2 M pro inhibitor clinical candidate for the treatment of COVID-19, Science
Roth, Mensah, Johnson, Global burden of cardiovascular diseases and risk factors, 1990-2019: update from the GBD 2019 study, J Am Coll Cardiol
Sevrioukova, Poulos, Structure and mechanism of the complex between cytochrome P4503A4 and ritonavir, Proc Natl Acad Sci U S A
Shaughnessy, Re: emergency use authorization 091
Shaughnessy, Re: emergency use authorization 094
Shaughnessy, Re: emergency use authorization 100
Thakur, Dubey, Benitez, A systematic review and meta-analysis of geographic differences in comorbidities and associated severity and mortality among individuals with COVID-19, Sci Rep
Weinreich, Sivapalasingam, Norton, REGEN-COV antibody combination and outcomes in outpatients with Covid-19, N Engl J Med
Wu, Mcgoogan, Characteristics of and important lessons from the Coronavirus Disease 2019 (COVID-19) outbreak in China: summary of a report of 72 314 cases from the Chinese Center for Disease Control and Prevention, JAMA
Yang, Xie, Xue, Design of wide-spectrum inhibitors targeting coronavirus main proteases, PLoS Biol
Zheng, Peng, Xu, Risk factors of critical & mortal COVID-19 cases: a systematic literature review and metaanalysis, J Infect
DOI record:
{
"DOI": "10.1056/nejmoa2118542",
"ISSN": [
"0028-4793",
"1533-4406"
],
"URL": "http://dx.doi.org/10.1056/nejmoa2118542",
"alternative-id": [
"10.1056/NEJMoa2118542"
],
"author": [
{
"affiliation": [
{
"name": "From Global Product Development, Pfizer, Collegeville, PA (J.H., H.L.-T.); Global Product Development (A.G.) and Early Clinical Development (M.L.B.), Pfizer, Cambridge, MA; Global Product Development, Pfizer, New York (P.A., W.B., B.D., R.P.); Global Product Development, Pfizer, Lake Forest, IL (W.W.); Medical and Safety, Pfizer, Sandwich, United Kingdom (V.M.H.); Köhler and Milstein Research, Mérida, Yucatan, Mexico (A.S.-C.); and Global Product Development, Pfizer, Tampa, FL (J.M.R.)."
}
],
"family": "Hammond",
"given": "Jennifer",
"sequence": "first"
},
{
"affiliation": [
{
"name": "From Global Product Development, Pfizer, Collegeville, PA (J.H., H.L.-T.); Global Product Development (A.G.) and Early Clinical Development (M.L.B.), Pfizer, Cambridge, MA; Global Product Development, Pfizer, New York (P.A., W.B., B.D., R.P.); Global Product Development, Pfizer, Lake Forest, IL (W.W.); Medical and Safety, Pfizer, Sandwich, United Kingdom (V.M.H.); Köhler and Milstein Research, Mérida, Yucatan, Mexico (A.S.-C.); and Global Product Development, Pfizer, Tampa, FL (J.M.R.)."
}
],
"family": "Leister-Tebbe",
"given": "Heidi",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Global Product Development, Pfizer, Collegeville, PA (J.H., H.L.-T.); Global Product Development (A.G.) and Early Clinical Development (M.L.B.), Pfizer, Cambridge, MA; Global Product Development, Pfizer, New York (P.A., W.B., B.D., R.P.); Global Product Development, Pfizer, Lake Forest, IL (W.W.); Medical and Safety, Pfizer, Sandwich, United Kingdom (V.M.H.); Köhler and Milstein Research, Mérida, Yucatan, Mexico (A.S.-C.); and Global Product Development, Pfizer, Tampa, FL (J.M.R.)."
}
],
"family": "Gardner",
"given": "Annie",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Global Product Development, Pfizer, Collegeville, PA (J.H., H.L.-T.); Global Product Development (A.G.) and Early Clinical Development (M.L.B.), Pfizer, Cambridge, MA; Global Product Development, Pfizer, New York (P.A., W.B., B.D., R.P.); Global Product Development, Pfizer, Lake Forest, IL (W.W.); Medical and Safety, Pfizer, Sandwich, United Kingdom (V.M.H.); Köhler and Milstein Research, Mérida, Yucatan, Mexico (A.S.-C.); and Global Product Development, Pfizer, Tampa, FL (J.M.R.)."
}
],
"family": "Abreu",
"given": "Paula",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Global Product Development, Pfizer, Collegeville, PA (J.H., H.L.-T.); Global Product Development (A.G.) and Early Clinical Development (M.L.B.), Pfizer, Cambridge, MA; Global Product Development, Pfizer, New York (P.A., W.B., B.D., R.P.); Global Product Development, Pfizer, Lake Forest, IL (W.W.); Medical and Safety, Pfizer, Sandwich, United Kingdom (V.M.H.); Köhler and Milstein Research, Mérida, Yucatan, Mexico (A.S.-C.); and Global Product Development, Pfizer, Tampa, FL (J.M.R.)."
}
],
"family": "Bao",
"given": "Weihang",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Global Product Development, Pfizer, Collegeville, PA (J.H., H.L.-T.); Global Product Development (A.G.) and Early Clinical Development (M.L.B.), Pfizer, Cambridge, MA; Global Product Development, Pfizer, New York (P.A., W.B., B.D., R.P.); Global Product Development, Pfizer, Lake Forest, IL (W.W.); Medical and Safety, Pfizer, Sandwich, United Kingdom (V.M.H.); Köhler and Milstein Research, Mérida, Yucatan, Mexico (A.S.-C.); and Global Product Development, Pfizer, Tampa, FL (J.M.R.)."
}
],
"family": "Wisemandle",
"given": "Wayne",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Global Product Development, Pfizer, Collegeville, PA (J.H., H.L.-T.); Global Product Development (A.G.) and Early Clinical Development (M.L.B.), Pfizer, Cambridge, MA; Global Product Development, Pfizer, New York (P.A., W.B., B.D., R.P.); Global Product Development, Pfizer, Lake Forest, IL (W.W.); Medical and Safety, Pfizer, Sandwich, United Kingdom (V.M.H.); Köhler and Milstein Research, Mérida, Yucatan, Mexico (A.S.-C.); and Global Product Development, Pfizer, Tampa, FL (J.M.R.)."
}
],
"family": "Baniecki",
"given": "MaryLynn",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Global Product Development, Pfizer, Collegeville, PA (J.H., H.L.-T.); Global Product Development (A.G.) and Early Clinical Development (M.L.B.), Pfizer, Cambridge, MA; Global Product Development, Pfizer, New York (P.A., W.B., B.D., R.P.); Global Product Development, Pfizer, Lake Forest, IL (W.W.); Medical and Safety, Pfizer, Sandwich, United Kingdom (V.M.H.); Köhler and Milstein Research, Mérida, Yucatan, Mexico (A.S.-C.); and Global Product Development, Pfizer, Tampa, FL (J.M.R.)."
}
],
"family": "Hendrick",
"given": "Victoria M.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Global Product Development, Pfizer, Collegeville, PA (J.H., H.L.-T.); Global Product Development (A.G.) and Early Clinical Development (M.L.B.), Pfizer, Cambridge, MA; Global Product Development, Pfizer, New York (P.A., W.B., B.D., R.P.); Global Product Development, Pfizer, Lake Forest, IL (W.W.); Medical and Safety, Pfizer, Sandwich, United Kingdom (V.M.H.); Köhler and Milstein Research, Mérida, Yucatan, Mexico (A.S.-C.); and Global Product Development, Pfizer, Tampa, FL (J.M.R.)."
}
],
"family": "Damle",
"given": "Bharat",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Global Product Development, Pfizer, Collegeville, PA (J.H., H.L.-T.); Global Product Development (A.G.) and Early Clinical Development (M.L.B.), Pfizer, Cambridge, MA; Global Product Development, Pfizer, New York (P.A., W.B., B.D., R.P.); Global Product Development, Pfizer, Lake Forest, IL (W.W.); Medical and Safety, Pfizer, Sandwich, United Kingdom (V.M.H.); Köhler and Milstein Research, Mérida, Yucatan, Mexico (A.S.-C.); and Global Product Development, Pfizer, Tampa, FL (J.M.R.)."
}
],
"family": "Simón-Campos",
"given": "Abraham",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Global Product Development, Pfizer, Collegeville, PA (J.H., H.L.-T.); Global Product Development (A.G.) and Early Clinical Development (M.L.B.), Pfizer, Cambridge, MA; Global Product Development, Pfizer, New York (P.A., W.B., B.D., R.P.); Global Product Development, Pfizer, Lake Forest, IL (W.W.); Medical and Safety, Pfizer, Sandwich, United Kingdom (V.M.H.); Köhler and Milstein Research, Mérida, Yucatan, Mexico (A.S.-C.); and Global Product Development, Pfizer, Tampa, FL (J.M.R.)."
}
],
"family": "Pypstra",
"given": "Rienk",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Global Product Development, Pfizer, Collegeville, PA (J.H., H.L.-T.); Global Product Development (A.G.) and Early Clinical Development (M.L.B.), Pfizer, Cambridge, MA; Global Product Development, Pfizer, New York (P.A., W.B., B.D., R.P.); Global Product Development, Pfizer, Lake Forest, IL (W.W.); Medical and Safety, Pfizer, Sandwich, United Kingdom (V.M.H.); Köhler and Milstein Research, Mérida, Yucatan, Mexico (A.S.-C.); and Global Product Development, Pfizer, Tampa, FL (J.M.R.)."
}
],
"family": "Rusnak",
"given": "James M.",
"sequence": "additional"
}
],
"container-title": [
"New England Journal of Medicine"
],
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2022,
2,
16
]
],
"date-time": "2022-02-16T22:00:58Z",
"timestamp": 1645048858000
},
"deposited": {
"date-parts": [
[
2022,
2,
16
]
],
"date-time": "2022-02-16T22:00:59Z",
"timestamp": 1645048859000
},
"funder": [
{
"DOI": "10.13039/100004319",
"doi-asserted-by": "publisher",
"name": "Pfizer"
}
],
"indexed": {
"date-parts": [
[
2022,
2,
16
]
],
"date-time": "2022-02-16T22:41:36Z",
"timestamp": 1645051296577
},
"is-referenced-by-count": 0,
"issn-type": [
{
"type": "print",
"value": "0028-4793"
},
{
"type": "electronic",
"value": "1533-4406"
}
],
"issued": {
"date-parts": [
[
2022,
2,
16
]
]
},
"language": "en",
"license": [
{
"URL": "http://www.nejmgroup.org/legal/terms-of-use.htm",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2022,
2,
16
]
],
"date-time": "2022-02-16T00:00:00Z",
"timestamp": 1644969600000
}
}
],
"link": [
{
"URL": "http://www.nejm.org/doi/pdf/10.1056/NEJMoa2118542",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "150",
"original-title": [],
"prefix": "10.1056",
"published": {
"date-parts": [
[
2022,
2,
16
]
]
},
"published-online": {
"date-parts": [
[
2022,
2,
16
]
]
},
"publisher": "Massachusetts Medical Society",
"reference-count": 21,
"references-count": 21,
"relation": {},
"score": 1,
"short-container-title": [
"N Engl J Med"
],
"short-title": [],
"source": "Crossref",
"subject": [
"General Medicine"
],
"subtitle": [],
"title": [
"Oral Nirmatrelvir for High-Risk, Nonhospitalized Adults with Covid-19"
],
"type": "journal-article"
}