Concerns have been raised over potential harm with the use of ibuprofen for COVID-191 due to the suppression of beneficial immune and inflammatory responses during early infection, ACE2 upregulation, and delaying further care. There is limited clinical data currently, especially with regard to acute usage at onset of infection, however current results suggest harm with early treatment and benefit with late treatment.
Ibuprofen was adopted
in 16 countries.
Jun 4 |
Ibuprofen for COVID-19: real-time meta-analysis of 13 studies (Version 11) | |
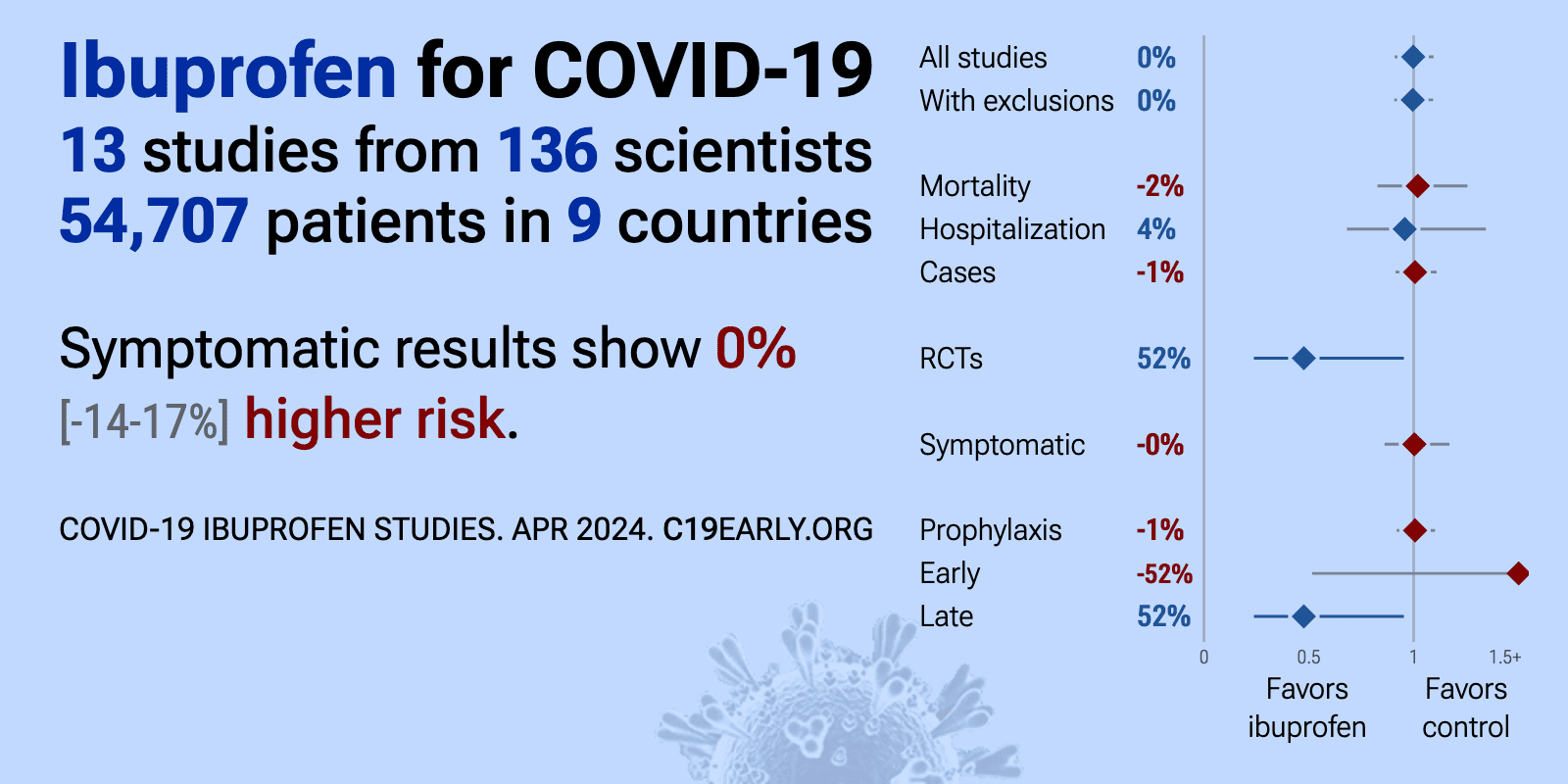
| Meta-analysis using the most serious outcome reported shows 0% [-9‑9%] lower risk, without reaching statistical significance. 4 studies from 4 independent teams in 4 countries show significant benefit. Control Ibuprofen Concern.. | ||
Dec 8 2024 |
et al., medRxiv, doi:10.1101/2024.12.07.24318645 | Effect of background therapy with Non-steroidal anti-inflammatory drugs (NSAIDs) and other anti-inflammatory agents on COVID-19 outcomes |
| Retrospective 485,779 osteoarthritis patients in the US showing lower mortality with non-aspirin NSAIDs and celecoxib, and higher mortality with aspirin. Aspirin was associated with higher hospitalization in COVID-positive and COVID-negat.. | ||
Feb 2 2024 |
et al., Heliyon, doi:10.1016/j.heliyon.2024.e25734 | Evaluating NSAIDs in SARS-CoV-2: Immunomodulatory mechanisms and future therapeutic strategies |
| Review of NSAIDs and their complex immunomodulatory roles in SARS-CoV-2 infection. Authors discuss the mechanisms by which NSAIDs impact viral replication, immune responses, and inflammation. They note NSAIDs can reduce neutrophil recruit.. | ||
Jan 3 2024 |
et al., BMJ Open, doi:10.1136/bmjopen-2023-077365 | NSAID prescribing and adverse outcomes in common infections: a population-based cohort study |
| 219% higher combined mortality/hospitalization (p<0.0001). Retrospective 142,925 outpatients in the UK showing significantly higher risk of hospitalization or death with prescription of NSAIDs for respiratory tract infections (RTI) or urinary tract infections (UTI). Practice-level analysis also f.. | ||
Oct 18 2023 |
et al., Journal of Infection, doi:10.1016/j.jinf.2023.10.009 | Analysis of blood and nasal epithelial transcriptomes to identify mechanisms associated with control of SARS-CoV-2 viral load in the upper respiratory tract |
| Blood and nasal epithelial transcriptomic analysis suggesting potential benefits of several treatments including: Vitamin E: transcriptomic analysis revealed that the AIPL1 module, which contains genes connected to vitamin E (α-tocopherol.. | ||
May 16 2023 |
et al., Clinical and Translational Discovery, doi:10.1002/ctd2.204 | Repurposing Inhaled Ibuprofenate, a Non Steroidal Anti‐Inflammatory Drug, as a Potential Adjuvant Treatment for Pneumonia, CARDS and its Aetiological Agent SARS‐CoV‐2 |
| Review of the potential mechanisms of action and therapeutic effects of inhaled sodium ibuprofenate for the treatment of COVID-19 pneumonia. Authors describe potential anti-inflammatory, immunomodulatory, antiangiogenic, and virucidal eff.. | ||
Apr 19 2023 |
et al., The Open Anesthesia Journal, doi:10.2174/25896458-v17-e230403-2022-26 | Early Use of Ibuprofen in Moderate Cases of COVID-19 Might be a Promising Agent to Attenuate the Severity of Disease: A Randomized Controlled Trial |
| 52% lower ICU admission (p=0.05), 52% lower need for oxygen therapy (p=0.05), and 26% shorter hospitalization (p=0.01). RCT 180 moderate hospitalized COVID-19 patients in Egypt, showing lower ICU admission and shorter hospitalization with ibuprofen compared with acetaminophen. | ||
Dec 31 2022 |
et al., European Journal of Respiratory Medicine, doi:10.31488/EJRM.132 | Safety and Efficacy of Nebulised Anti-Inflammatory Solution of Alkaline Hypertonic Ibuprofen (AHI) for Treatment of SARS-Cov-2 Infection: A Compassionate Study with a Comparator Arms |
| 79% lower mortality (p=0.0009). Retrospective 99 COVID-19 patients in Argentina showing significantly lower mortality with inhaled alkaline hypertonic ibuprofen (AHI) treatment. The treatment has a pH of 8.5. 3 times daily for 7-10 days. | ||
Sep 24 2022 |
et al., Hospital Pharmacy, doi:10.1177/00185787221125721 | How Much has COVID-19 Contributed to Increase the Worldwide Consumption of Paracetamol and Ibuprofen? Evidence From an Infodemiological Analysis |
| Infodemiological analysis showing a significant worldwide increase in Google searches for paracetamol (54%) and ibuprofen (24%) during the COVID-19 pandemic compared to pre-pandemic levels, suggesting dramatically increased consumption. | ||
Aug 16 2022 |
et al., Virology Journal, doi:10.1186/s12985-023-02195-9 (date from preprint) | Real-world evidence with a retrospective cohort of 15,968 COVID-19 hospitalized patients suggests 21 new effective treatments |
| 48% lower mortality (p=0.002). Retrospective 15,968 COVID-19 hospitalized patients in Spain, showing lower mortality with existing use of several medications including metformin, HCQ, azithromycin, aspirin, vitamin D, vitamin C, and budesonide. Since only hospitalized .. | ||
Jul 13 2022 |
et al., Drugs, doi:10.1007/s40265-022-01822-z (date from preprint) | Risk of COVID-19 Diagnosis and Hospitalization in Patients with Osteoarthritis or Back Pain Treated with Ibuprofen Compared to Other NSAIDs or Paracetamol: A Network Cohort Study |
| 12% higher hospitalization (p=0.26) and 8% more cases (p=0.25). PSM retrospective 1,697,522 osteoarthritis or back pain patients in the US, showing no significant differences in COVID-19 cases and hospitalization for ibuprofen vs. other NSAIDs. | ||
Jun 17 2022 |
et al., Nucleic Acids Research, doi:10.1093/nar/gkac513 | Body temperature variation controls pre-mRNA processing and transcription of antiviral genes and SARS-CoV-2 replication |
| In vitro and hamster study showing that higher temperature reduces SARS-CoV-2 replication. Authors show a 1.5°C increase in temperature (from 36.5 to 38°C) enhanced the expression of antiviral genes. Elevated temperatures were found to re.. | ||
May 31 2022 |
et al., National Bureau of Economic Research, doi:10.3386/w30084 | Causal Inference During a Pandemic: Evidence on the Effectiveness of Nebulized Ibuprofen as an Unproven Treatment for COVID-19 in Argentina |
| 49% lower mortality (p=0.01). Retrospective 5,146 hospitalized COVID-19 patients in Argentina, showing lower mortality associated with nebulized ibuprofen (NaIHS) treatment. Doubly robust inverse probability weighting estimators were used to control for confounding. A.. | ||
May 5 2022 |
et al., PLOS ONE, doi:10.1371/journal.pone.0267462 | Chronic use of non-steroidal anti-inflammatory drugs (NSAIDs) or acetaminophen and relationship with mortality among United States Veterans after testing positive for COVID-19 |
| no change in mortality (p=0.54). Retrospective 28,856 COVID-19 patients in the USA, showing no significant difference in mortality for chronic ibuprofen use vs. sporadic NSAID use. Since ibuprofen is available OTC and authors only tracked prescriptions, many patients cla.. | ||
Dec 31 2021 |
et al., Computational and Structural Biotechnology Journal, doi:10.1016/j.csbj.2020.12.005 | Temperature dependence of the SARS-CoV-2 affinity to human ACE2 determines COVID-19 progression and clinical outcome |
| In silico and in vitro study showing decreased SARS-CoV-2 binding affinity to the human ACE2 receptor at higher temperature (40°C vs. 37°C). Molecular dynamics simulations, surface plasmon resonance experiments, and pseudovirus cell entry.. | ||
Aug 30 2021 |
et al., Infectious Diseases and Therapy, doi:10.1007/s40121-021-00527-2 | Reversal of SARS-CoV2-Induced Hypoxia by Nebulized Sodium Ibuprofenate in a Compassionate Use Program |
| 45% lower mortality (p=0.009) and 14% shorter hospitalization (p<0.0001). Retrospective 383 hospitalized COVID-19 patients in Argentina showing signifcantly lower mortality and shorter hospital stay with nebulized sodium ibuprofenate compared to 195 contemporaneous controls. The treatment appears to be the same.. | ||
Aug 16 2021 |
et al., COVID, doi:10.3390/covid1010018 | Paracetamol Is Associated with a Lower Risk of COVID-19 Infection and Decreased ACE2 Protein Expression: A Retrospective Analysis |
| 3% fewer cases (p=0.29). UK Biobank retrospective showing no significant difference in cases with ibuprofen use. | ||
Jul 31 2021 |
et al., The Lancet Rheumatology, doi:10.1016/S2665-9913(21)00104-1 | Non-steroidal anti-inflammatory drug use and outcomes of COVID-19 in the ISARIC Clinical Characterisation Protocol UK cohort: a matched, prospective cohort study |
| 10% lower mortality (p=0.36). Prospective study of 78,674 COVID-19 patients, showing no significant difference in mortality with ibuprofen use. | ||
Apr 20 2021 |
et al., medRxiv, doi:10.1101/2021.04.13.21255438 | Cyclooxygenase inhibitor use is associated with increased COVID-19 severity |
| 9% lower mortality (p=0.65) and 303% higher severe cases (p<0.0001). N3C retrospective 250,533 patients showing higher COVID-19 severity with ibuprofen use. Note that results for individual treatments are not included in the journal version or v2 of this preprint. | ||
Jan 21 2021 |
et al., Annals of the Rheumatic Diseases, doi:10.1136/annrheumdis-2020-219517 | Use of non-steroidal anti-inflammatory drugs and risk of death from COVID-19: an OpenSAFELY cohort analysis based on two cohorts |
| 23% higher mortality (p=0.19). Retrospective 2,463,707 people in the UK, showing no significant difference in COVID-19 mortality with NSAID use. Current NSAID users were defined as those ever prescribed an NSAID in the 4 months prior to study start, and non-users were .. | ||
Nov 23 2020 |
et al., Evolution, Medicine, and Public Health, doi:10.1093/emph/eoaa044 | Let fever do its job |
| Review of fever focusing on COVID-19. Authors note that fever is a key component of the acute phase response to infection. Fever enhances immune cell performance, induces cellular stress on pathogens, and acts synergistically with other s.. | ||
Nov 2 2020 |
et al., Infectious Diseases and Therapy, doi:10.1007/s40121-020-00363-w | Ibuprofen and NSAID Use in COVID-19 Infected Patients Is Not Associated with Worse Outcomes: A Prospective Cohort Study |
| 170% higher mortality (p=0.35), 45% higher need for oxygen therapy (p=0.64), 18% higher hospitalization (p=0.64), and 85% higher severe cases (p=0.42). Prospective study of 503 COVID-19 cases in Saudi Arabia, 40 using ibuprofen during infection, and 357 not using NSAIDs, showing no significant differences in outcomes. Results are subject to confounding by indication. | ||
Oct 21 2020 |
et al., Clinical and Translational Science, doi:10.1111/cts.12904 | Association Between Prescribed Ibuprofen and Severe COVID-19 Infection: A Nationwide Register-Based Cohort Study |
| 4% lower progression (p=0.78). Retrospective 4,002 COVID-19 patients in Denmark, 264 with ibuprofen prescriptions, showing no significant difference for COVID-19 severity. | ||
Sep 30 2020 |
et al., Clinical Microbiology and Infection, doi:10.1016/j.cmi.2020.06.003 | Ibuprofen use and clinical outcomes in COVID-19 patients |
| 21% higher mortality (p=0.73), 12% higher ventilation (p=0.77), and 40% higher ICU admission (p=0.56). Retrospective 403 COVID-19 cases in Israel, showing no significant difference in outcomes with ibuprofen use. Patients were asked about ibuprofen use starting a week before diagnosis of COVID-19 - treatment time may have been early, late,.. | ||
Jul 13 2020 |
et al., Archives of Clinical Infectious Diseases, doi:10.5812/archcid.106847 | The Association of Non-Steroidal Anti-Inflammatory Drugs with COVID-19 Severity and Mortality |
| 100% higher mortality (p=0.001), 428% higher severe cases (p=0.0007), and 13% higher progression (p=0.04). Retrospective 158 COVID-19 patients in Iran, showing higher risk of mortality with ibuprofen use. | ||
Jun 23 2020 |
et al., Journal of Clinical Medicine, doi:10.3390/jcm9061959 | Clinical Characteristics and Disease Progression in Early-Stage COVID-19 Patients in South Korea |
| 240% higher progression (p=0.26). Retrospective 293 patients in South Korea, showing higher risk of progression with ibuprofen use, without statistical significance. | ||
Mar 17 2020 |
, M., BMJ, doi:10.1136/bmj.m1086 | Covid-19: ibuprofen should not be used for managing symptoms, say doctors and scientists |
| Discussion of potential harms of ibuprofen use for COVID-19. | ||
Dec 1 1990 |
et al., Journal of Infectious Diseases, doi:10.1093/infdis/162.6.1277 | Adverse Effects of Aspirin, Acetaminophen, and Ibuprofen on Immune Function, Viral Shedding, and Clinical Status in Rhinovirus-Infected Volunteers |
| RCT examining the effects of aspirin, acetaminophen, and ibuprofen on immune function, virus shedding, and clinical symptoms in 60 healthy volunteers experimentally infected with rhinovirus type 2. The study found that aspirin and acetami.. | ||
Apr 30 1988 |
et al., Journal of Interferon Research, doi:10.1089/jir.1988.8.143 | Hyperthermia in Humans Enhances Interferon-γ Synthesis and Alters the Peripheral Lymphocyte Population |
| Analysis of induced hyperthermia (high body temperature) showing 10 times greater interferon gamma (IFN-γ) production and increased natural killer cells. This response was observed at a core body temperature of ~39°C, highlighting its pot.. | ||
References