May 27 |
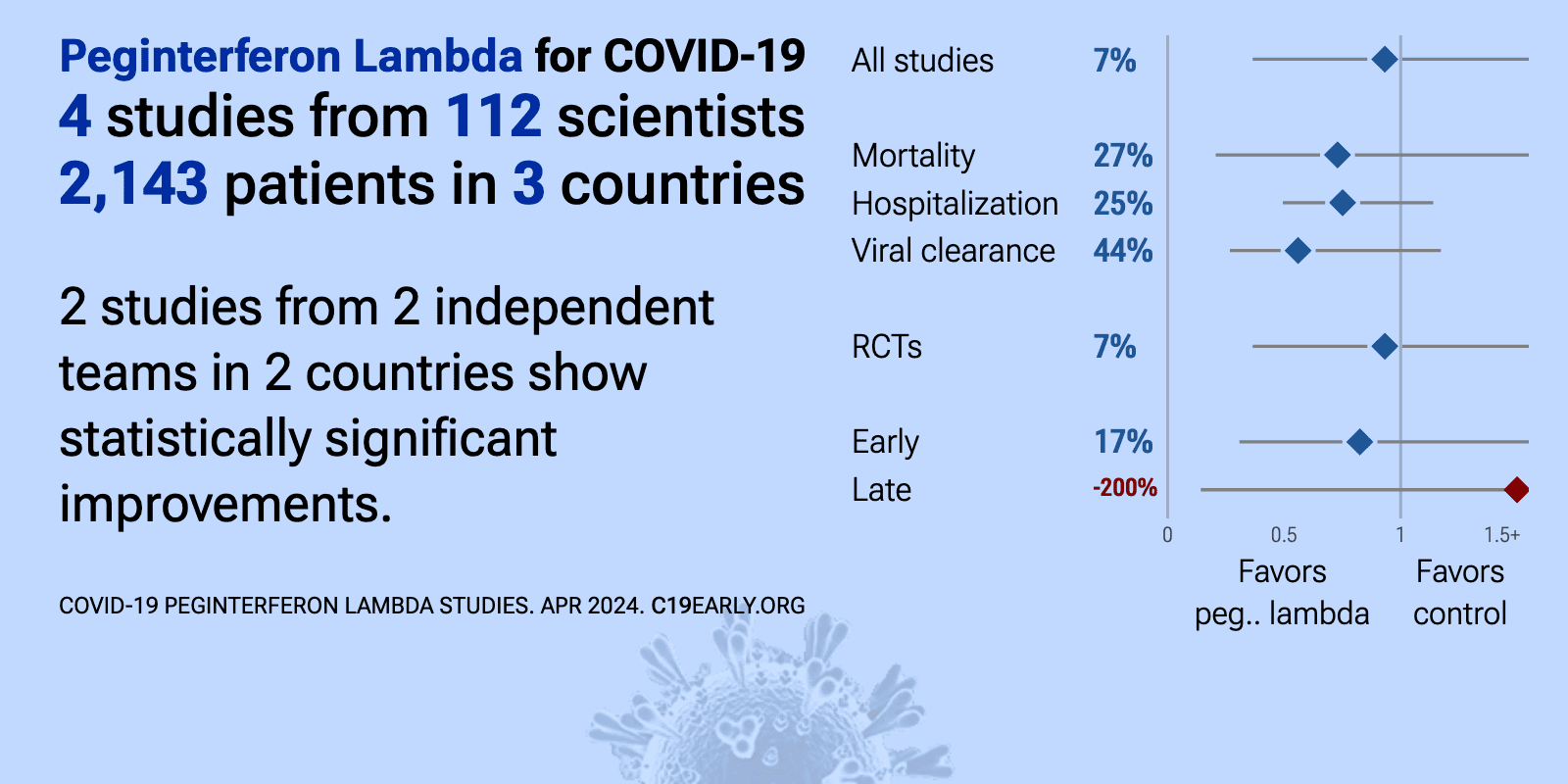
Peginterferon Lambda for COVID-19: real-time meta-analysis of 5 studies (Version 8) | |
| Meta-analysis using the most serious outcome reported shows 15% [-36‑107%] higher risk, without reaching statistical significance. Results are consistent with early treatment being more effective than late treatment. Currently .. | ||
Aug 14 2025 |
et al., Clinical Microbiology and Infection, doi:10.1016/j.cmi.2025.08.003 | Peginterferon lambda for the treatment of patients admitted to hospital with COVID-19: a phase 2, placebo-controlled randomised trial |
| 32% worse 7-point scale results (p=0.49). RCT 97 hospitalized COVID-19 patients showing no significant benefit with peginterferon lambda. | ||
Nov 6 2023 |
, E., Do Your Own Research | TOGETHER Files 2: Lawsuit reveals FTX bought effective control of TOGETHER trial, part of SBF's dream of a pharma empire |
| Analysis of legal documents showing that a non-profit controlled by FTX's SBF and a former colleague invested >$50 million in the Together Trial and had the right to potentially control the company. Author notes that incorrect and mislead.. | ||
Mar 16 2023 |
, M., Center for Open Science, doi:10.31219/osf.io/5xd6q | Peg-interferon Lambda Single Dose Treatment for COVID-19: A Call to Avoid another Hydroxychloroquine Fiasco |
| Review of critical issues with [Reis]. Author notes that NEJM declined to publish these issues without comment. | ||
Feb 24 2023 |
et al., Frontiers in Medicine, doi:10.3389/fmed.2023.1095828 | Peginterferon lambda for the treatment of hospitalized patients with mild COVID-19: A pilot phase 2 randomized placebo-controlled trial |
| 200% higher ICU admission (p=1), 25% longer hospitalization (p=0.59), and 12% improved viral clearance (p=1). Very small RCT with 14 hospitalized patients in the USA showing no significant differences with peginterferon lambda. Viral load was improved, however 86% of treatment versus 14% of control patients received remdesivir, and the median bas.. | ||
Feb 9 2023 |
et al., New England Journal of Medicine, doi:10.1056/NEJMoa2209760 | Early Treatment with Pegylated Interferon Lambda for Covid-19 |
| 27% lower mortality (p=0.76), 42% lower hospitalization (p=0.04), and 51% fewer combined hospitalization/ER visits (p=0.003). High-risk outpatient RCT with 931 peginterferon lambda patients and 1,018 control patients, showing significantly lower hospitalization/ER visits with treatment. Single subcutaneous injection. There were 85/931 and 286/1018 patients for w.. | ||
Mar 30 2021 |
et al., Nature Communications, doi:10.1038/s41467-021-22177-1 | Peginterferon Lambda-1a for treatment of outpatients with uncomplicated COVID-19: a randomized placebo-controlled trial |
| no change in hospitalization (p=1), 6% worse recovery (p=0.76), and 14% worse viral clearance (p=0.91). RCT 120 outpatients with mild/moderate COVID-19, showing no significant differences with peginterferon lambda-1a treatment. 180μg subcutaneous peginterferon lambda-1a. | ||
Nov 12 2020 |
et al., The Lancet Respiratory Medicine, doi:10.1016/S2213-2600(20)30566-X (date from preprint) | Peginterferon lambda for the treatment of outpatients with COVID-19: a phase 2, placebo-controlled randomised trial |
| 75% lower progression (p=0.35) and 66% improved viral clearance (p=0.03). Small outpatient RCT with 30 peginterferon lambda and 30 control patients, showing improved viral clearance with treatment. Single subcutaneous injection of peginterferon lambda 180μg. | ||
References