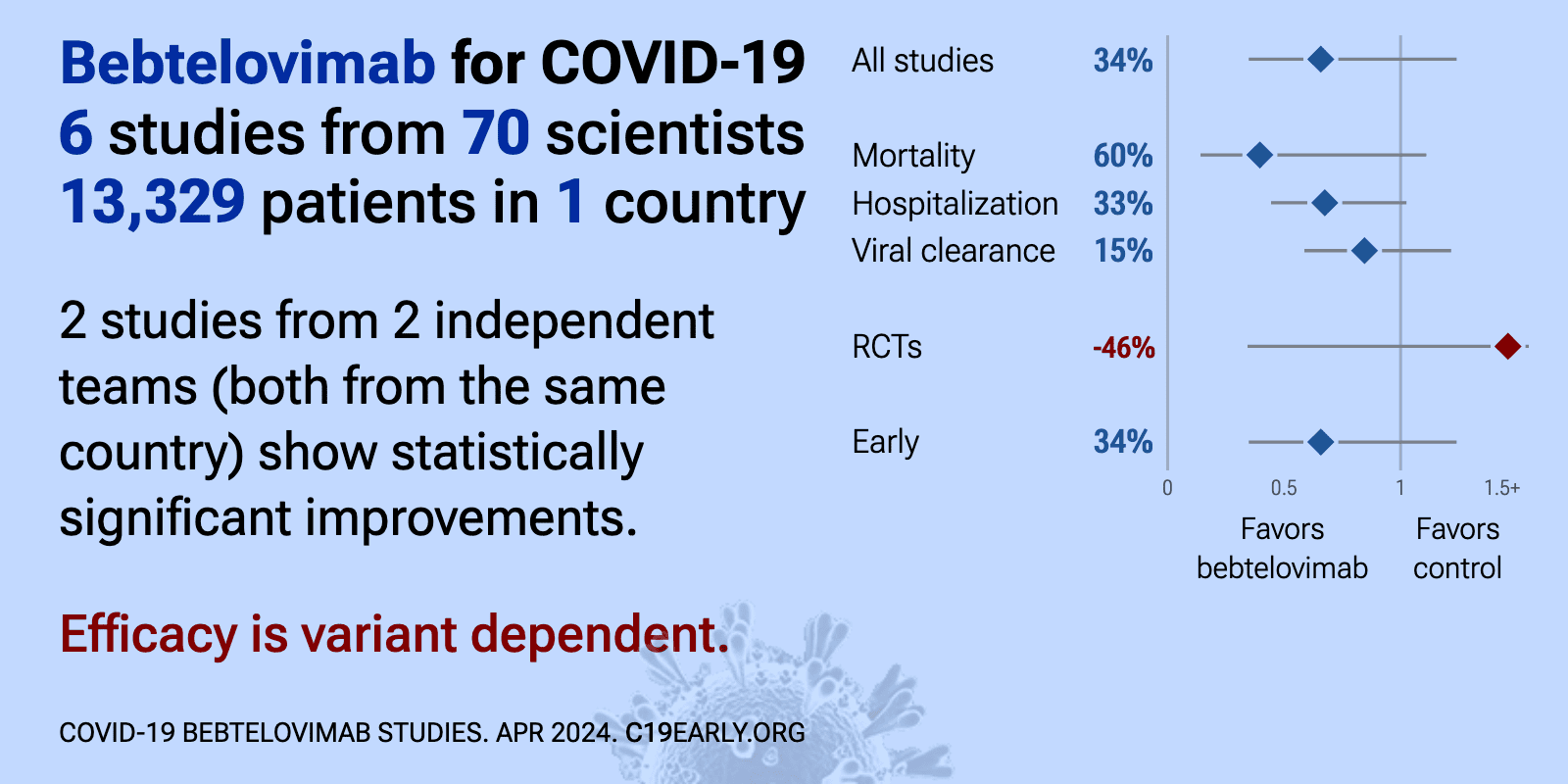
Efficacy is variant dependent. In Vitro research suggests a lack of efficacy for omicron BQ.1.11, BA.5, BA.2.75, XBB, XBB.1.5, XMM.1.9.12,3. mAb use may create new variants that spread globally4-6, and may be associated with increased risk of autoimmune disease7, prolonged viral loads, clinical deterioration, and immune escape5,8-12 .
Bebtelovimab was adopted
in 2 countries.
Recent:Ashoor.
Jun 7 |
Bebtelovimab for COVID-19: real-time meta-analysis of 6 studies (Version 16) | |
| Meta-analysis using the most serious outcome reported shows 34% [-24‑65%] lower risk, without reaching statistical significance. Results are similar for peer-reviewed studies and worse for Randomized Controlled Trials. 2 studie.. | ||
Oct 1 2025 |
et al., Infection and Drug Resistance, doi:10.2147/IDR.S540928 | The Emergence of Escape Mutations in COVID-19 Following Anti-Spike Monoclonal Antibody Treatment: How Do We Tackle It? |
| Review of treatment-emergent resistance to anti-spike monoclonal antibodies in COVID-19 patients. Monotherapies like bamlanivimab and sotrovimab showed very high resistance rates, while combination approaches had lower risk. The rapid evo.. | ||
Dec 25 2024 |
et al., Pulmonary Therapy, doi:10.1007/s41030-024-00284-w | Real-World Effectiveness of Bebtelovimab Versus Nirmatrelvir/Ritonavir in Outpatients with COVID-19 |
| Retrospective 5,827 matched pairs of non-hospitalized COVID-19 patients showing no significant differences between bebtelovimab and paxlovid treatment. Authors do not exclude patients with contraindications for paxlovid. | ||
Nov 12 2024 |
et al., Pharmacoepidemiology and Drug Safety, doi:10.1002/pds.70043 | Investigating the Safety Profile of Fast‐Track COVID‐19 Drugs Using the FDA Adverse Event Reporting System Database: A Comparative Observational Study |
| FAERS adverse event analysis for remdesivir, bebtelovimab, molnupiravir, and paxlovid. Top 10 signals for serious adverse drug reactions for remdesivir included death and acute kidney injury, for paxlovid: disease recurrence and rebound, .. | ||
Sep 19 2024 |
et al., Antiviral Research, doi:10.1016/j.antiviral.2024.106006 | Identification of antibody-resistant SARS-CoV-2 mutants via N4-Hydroxycytidine mutagenesis |
| In vitro selection study identifying SARS-CoV-2 spike mutations that confer resistance to therapeutic antibodies. Using a Wuhan-like strain and omicron B.1.1.529, the authors used sequential mutagenesis with the molnupiravir active compou.. | ||
Aug 19 2024 |
, D., MDPI AG, doi:10.20944/preprints202408.1340.v1 | In Silico Evaluation of Ten Monoclonal Antibodies Neutralization Power of SARS-CoV-2 Variants EG.5, BA.2.86 and JN.1 |
| In silico study evaluating the neutralization power of 10 monoclonal antibodies (9 previously EUA-granted, 1 under clinical investigation) against SARS-CoV-2 variants EG.5, BA.2.86, and JN.1. Most available neutralizing antibodies have si.. | ||
Aug 11 2024 |
, D., Current Topics in Microbiology and Immunology, doi:10.1007/82_2024_268 | Monoclonal Antibody Therapies Against SARS-CoV-2: Promises and Realities |
| Review of monoclonal antibodies for SARS-CoV-2. Author notes that the omicron variant has reset achievements to date. | ||
Aug 8 2024 |
et al., Clinical Infectious Diseases, doi:10.1093/cid/ciae408 | Single monoclonal antibodies should not be used for COVID-19 therapy: a call for antiviral stewardship |
| Review arguing against use of single monoclonal antibodies for COVID-19 treatment, particularly in immunosuppressed patients, due to the risk of rapidly selecting for resistant viral variants. Authors suggest that while monoclonal antibod.. | ||
Aug 5 2024 |
et al., Viral Immunology, doi:10.1089/vim.2024.0034 | Effective Treatment of COVID-19 Infection with Repurposed Drugs: Case Reports |
| Review of the successful treatment of COVID-19 using existing medications including HCQ, AZ, ivermectin, famotidine, monoclonal antibodies, and others. Authors note that the typical treatment of severe viral infections with multiple thera.. | ||
Apr 30 2024 |
et al., European Review for Medical and Pharmacological Sciences, doi:10.26355/eurrev_202404_35925 | The safety signal detection and analysis of monoclonal antibodies against SARS-CoV-2 based on real-world evidence – the suitable selectivity for different populations |
| FDA adverse event reporting analysis for bebtelovimab, tixagevimab/cilgavimab, and sotrovimab, showing distinct safety profiles with concerning adverse events for each drug. Bebtelovimab showed the highest associations with gastrointestin.. | ||
Mar 30 2024 |
et al., Infection, doi:10.1007/s15010-024-02223-y | SARS-CoV-2 journey: from alpha variant to omicron and its sub-variants |
| Review of SARS-CoV-2 variants showing increased transmissibility, disease severity, and immune escape with mutations in the spike protein receptor binding domain. Authors cover variants from the initial D614G mutation through omicron sub-.. | ||
Jan 26 2024 |
et al., JAMA Health Forum, doi:10.1001/jamahealthforum.2023.5044 | Clinical Risk and Outpatient Therapy Utilization for COVID-19 in the Medicare Population |
| Analysis of Medicare beneficiaries in 2022 showing that outpatient COVID-19 treatments like antivirals and monoclonal antibodies were disproportionately used by patients at lower risk of severe infection and outcomes. Retrospective studie.. | ||
Nov 30 2023 |
et al., iScience, doi:10.1016/j.isci.2023.108147 | Antiviral efficacy against and replicative fitness of an XBB.1.9.1 clinical isolate |
| In vitro and animal study showing that the SARS-CoV-2 omicron subvariant XBB.1.9.1 has similar antigenicity, antiviral susceptibility, and replicative ability compared to XBB.1.5. Casirivimab, imdevimab, tixagevimab, cilgavimab, sotrovima.. | ||
Sep 27 2023 |
et al., Microorganisms, doi:10.3390/microorganisms11102417 | Prevalence of SARS-CoV-2 Omicron Sublineages and Spike Protein Mutations Conferring Resistance against Monoclonal Antibodies in a Swedish Cohort during 2022–2023 |
| Analysis of 7,950 SARS-CoV-2 samples from central Sweden collected between March 2022 and May 2023 tracking the prevalence of omicron sublineages and mutations in the spike protein conferring resistance to monoclonal antibodies over time... | ||
Aug 10 2023 |
et al., Drug Resistance Updates, doi:10.1016/j.drup.2023.100991 | Analysis of SARS-CoV-2 mutations associated with resistance to therapeutic monoclonal antibodies that emerge after treatment |
| Review of reports of treatment-emergent resistance to COVID-19 monoclonal antibodies (mAbs), showing that some post-mAb treatment mutations appeared to spread globally soon after the mAb was introduced, raising concerns about transmission.. | ||
Apr 28 2023 |
et al., PLOS ONE, doi:10.1371/journal.pone.0279326 | Lack of effectiveness of Bebtelovimab monoclonal antibody among high-risk patients with SARS-Cov-2 Omicron during BA.2, BA.2.12.1 and BA.5 subvariants dominated era |
| 86% lower mortality (p=0.25), 25% lower combined mortality/hospitalization (p=0.31), and 11% lower hospitalization (p=0.78). PSM retrospective 19,778 high-risk outpatients in the USA, showing no significant difference in outcomes with bebtelovimab treatment. | ||
Apr 16 2023 |
et al., International Journal of Infectious Diseases, doi:10.1016/j.ijid.2023.04.396 | Real-World Evaluation of Bebtelovimab Effectiveness During the Period of COVID-19 Omicron Variants including BA.4/BA.5 |
| 57% lower mortality (p=0.14), 59% lower ICU admission (p=0.05), 56% lower hospitalization (p<0.0001), and 33% higher progression (p=0.001). Retrospective 3,739 patients treated with bebteloviman in the USA and matched controls, showing lower mortality and hospitalization with treatment, but higher emergency department visits. | ||
Apr 4 2023 |
et al., Annals of Internal Medicine, doi:10.7326/M22-1286 | Evolving Real-World Effectiveness of Monoclonal Antibodies for Treatment of COVID-19 |
| 20% lower combined mortality/hospitalization (p=0.65). Retrospective 2,571 patients treated with mAbs in the USA, and 5,135 control patients, showing lower combined mortality/hospitalization for bamlanivimab, bamlanivimab/etesevimab, casirivimab/imdevimab, sotrovimab, and bebtelovimab, with s.. | ||
Nov 17 2022 |
et al., bioRxiv, doi:10.1101/2022.11.17.516888 | Resistance of Omicron subvariants BA.2.75.2, BA.4.6 and BQ.1.1 to neutralizing antibodies |
| In vitro study suggesting a lack of efficacy for bebtelovimab with BQ.1.1. | ||
Oct 27 2022 |
et al., Open Forum Infectious Diseases, doi:10.1093/ofid/ofac565 | Bebtelovimab for high-risk outpatients with early COVID-19 in a large US health system |
| 86% lower mortality (p=0.25), 43% lower combined mortality/hospitalization (p=0.14), and 29% lower hospitalization (p=0.53). Retrospective 377 outpatients in the USA and matched controls, showing lower hospitalization/mortality with bebtelovimab treatment, without statistical significance. Notably, none of the patients that died in the control group were hospit.. | ||
Mar 12 2022 |
et al., medRxiv, doi:10.1101/2022.03.10.22272100 | Bebtelovimab, alone or together with bamlanivimab and etesevimab, as a broadly neutralizing monoclonal antibody treatment for mild to moderate, ambulatory COVID-19 |
| 25% faster recovery and 4% improved viral clearance (p<0.0001). RCT showing improved viral clearance with bebtelovimab. Results refer to the placebo controlled portion of the trial. | ||
Feb 12 2022 |
, NCT04634409 | A Study of Immune System Proteins in Participants With Mild to Moderate COVID-19 Illness |
| 36% improved viral clearance (p=0.07). RCT with 127 bamlanivimab, etesevimab, and bebtelovimab patients, 125 bebtelovimab patients, and 128 control patients, showing no significant differences in hospitalization and mortality. Viral clearance was improved although not statisti.. | ||
References
Planas et al., Resistance of Omicron subvariants BA.2.75.2, BA.4.6 and BQ.1.1 to neutralizing antibodies, bioRxiv, doi:10.1101/2022.11.17.516888.
Haars et al., Prevalence of SARS-CoV-2 Omicron Sublineages and Spike Protein Mutations Conferring Resistance against Monoclonal Antibodies in a Swedish Cohort during 2022–2023, Microorganisms, doi:10.3390/microorganisms11102417.
Uraki et al., Antiviral efficacy against and replicative fitness of an XBB.1.9.1 clinical isolate, iScience, doi:10.1016/j.isci.2023.108147.
Focosi et al., Analysis of SARS-CoV-2 mutations associated with resistance to therapeutic monoclonal antibodies that emerge after treatment, Drug Resistance Updates, doi:10.1016/j.drup.2023.100991.
Leducq et al., Spike protein genetic evolution in patients at high-risk of severe COVID-19 treated by monoclonal antibodies, The Journal of Infectious Diseases, doi:10.1093/infdis/jiad523.
Bruhn et al., Somatic hypermutation shapes the viral escape profile of SARS-CoV-2 neutralising antibodies, eBioMedicine, doi:10.1016/j.ebiom.2025.105770.
Ngiam et al., Early administration of neutralising monoclonal antibodies and post-acute sequelae of COVID-19, International Journal of Infectious Diseases, doi:10.1016/j.ijid.2026.108435.
Choudhary et al., Emergence of SARS-CoV-2 Resistance with Monoclonal Antibody Therapy, medRxiv, doi:10.1101/2021.09.03.21263105.
Günther et al., Variant-specific humoral immune response to SARS-CoV-2 escape mutants arising in clinically severe, prolonged infection, medRxiv, doi:10.1101/2024.01.06.24300890.
Casadevall et al., Single monoclonal antibodies should not be used for COVID-19 therapy: a call for antiviral stewardship, Clinical Infectious Diseases, doi:10.1093/cid/ciae408.