Convalescent Plasma was adopted
in 16 countries.
Jun 7 |
Convalescent Plasma for COVID-19: real-time meta-analysis of 58 studies (Version 45) | |
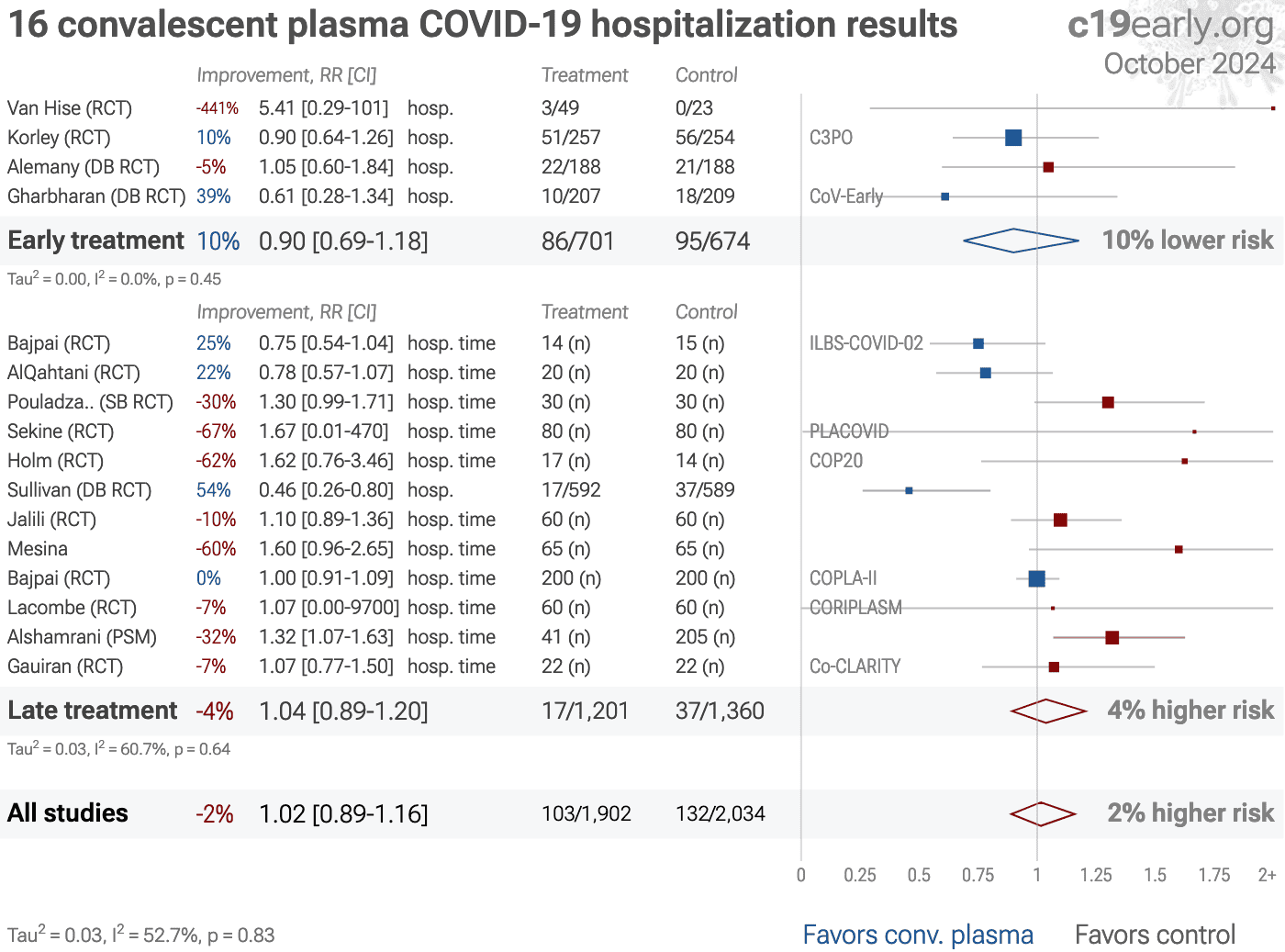
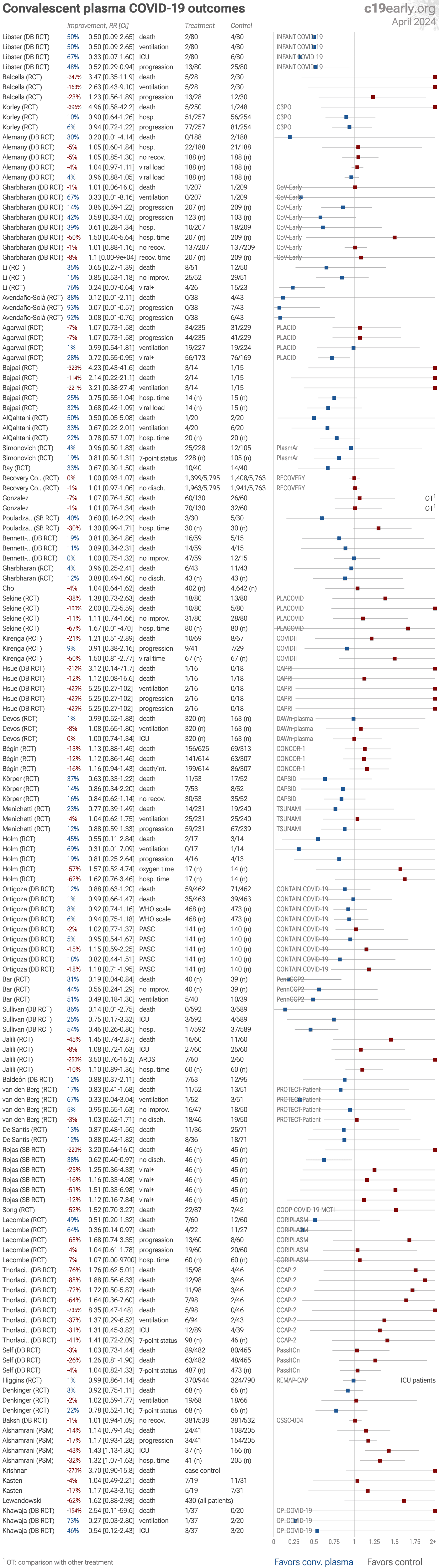
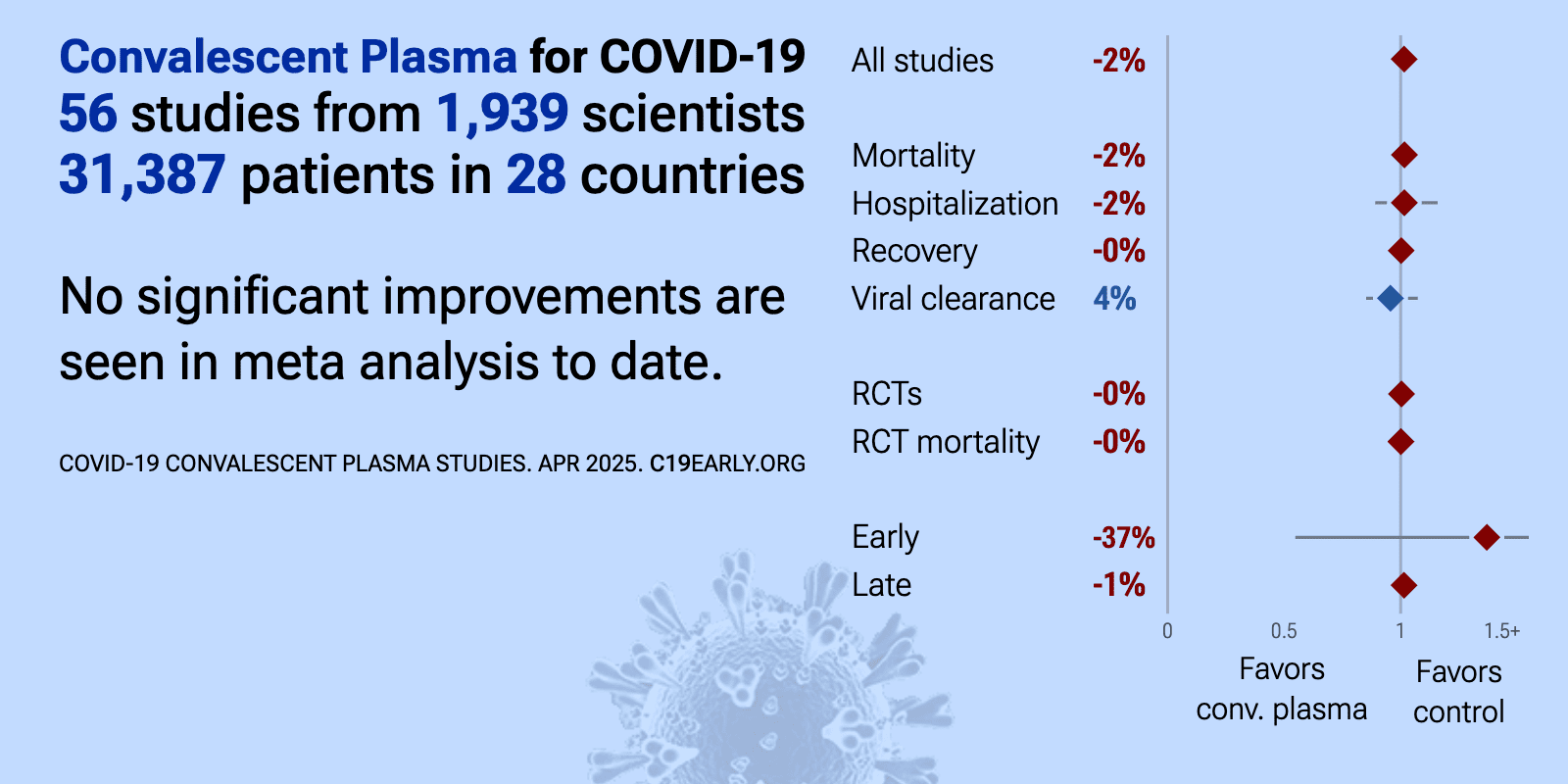
| Meta-analysis using the most serious outcome reported shows 2% [-2‑6%] higher risk, without reaching statistical significance. Control Convalescent PlasmaConv. Plasma 29 RCTs with 3,534 patients have not reported results (up to.. | ||
Nov 11 2025 |
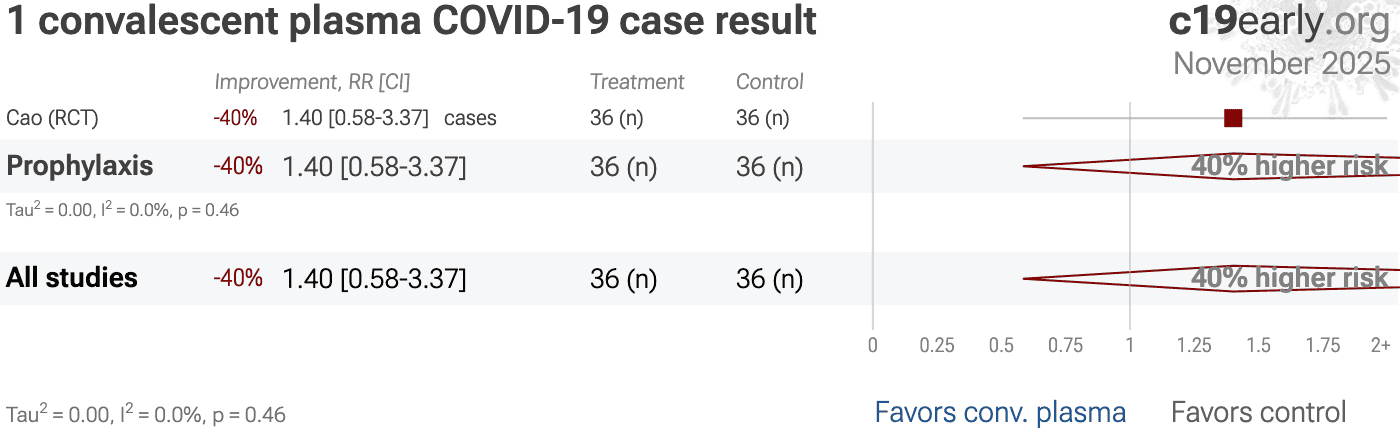
et al., Frontiers in Immunology, doi:10.3389/fimmu.2025.162677 | Convalescent Plasma Therapy for COVID-19 Prophylaxis in Adults Early Post-Hematopoietic Stem Cell Transplantation: One-Year Outcomes from a Randomized Controlled Trial |
| 224% higher mortality (p=0.04) and 40% more cases (p=0.46). RCT 72 hematopoietic stem cell transplantation (HSCT) recipients, showing higher one-year mortality with COVID-19 convalescent plasma prophylaxis treatment. | ||
Sep 2 2025 |
et al., medRxiv, doi:10.1101/2025.08.29.25334732 | Long-term follow-up of treatment comparisons in RECOVERY: a randomised, open-label, platform trial for patients hospitalised with COVID-19 |
| 6-month followup of RECOVERY patients. Results are reported within the respective trials for each treatment. | ||
Mar 31 2025 |
et al., Saudi Journal of Medicine, doi:10.36348/sjm.2025.v10i03.00X | Phase-II RCT Convalescent Plasma Transfusion in Severe COVID-19 Patients -Evaluation of Efficacy and Tolerability |
| no change in mortality (p=1). RCT 60 severe COVID-19 patients showing no benefit with convalescent plasma. | ||
Feb 27 2025 |
et al., eBioMedicine, doi:10.1016/j.ebiom.2025.105613 | Early, very high-titre convalescent plasma therapy in clinically vulnerable individuals with mild COVID-19: an international, randomised, open-label trial |
| 91% lower combined mortality/hospitalization (p=0.03). RCT 117 immunocompromised patients with mild COVID-19 showing lower hospitalization or death with early administration of very high-titre COVID-19 convalescent plasma (CCP). The trial was terminated early due to declining enrollment. | ||
Oct 24 2024 |
et al., PLOS ONE, doi:10.1371/journal.pone.0309449 | Impact of variable titer COVID-19 convalescent plasma and recipient SARS-CoV2-specific humoral immunity on survival in hospitalized patients |
| 26% higher mortality (p=0.14), 1% higher ventilation (p=1), and no change in progression (p=1). Retrospective propensity-matched analysis of 290 hospitalized COVID-19 patients who received convalescent plasma (CCP) compared to 290 controls, showing no significant difference in 30-day mortality, ECMO/mechanical ventilation, or hospit.. | ||
Mar 21 2024 |
et al., Transfusion, doi:10.1111/trf.17777 | Quality of life and cost‐effectiveness of convalescent plasma compared to standard care for hospitalized COVID‐19 patients in the CONCOR‐1 trial |
| Analysis of the CONCUR-1 RCT showing that convalescent plasma was associated with higher costs, longer hospital stays, and lower quality-adjusted life days. | ||
Mar 21 2024 |
et al., Infectious Diseases, doi:10.1080/23744235.2024.2329957 | Double-blinded, randomised, placebo-controlled trial of convalescent plasma for COVID-19: analyses by neutralising antibodies homologous to donors’ variants |
| RCT 57 hospitalized COVID-19 patients showing no significant difference in outcomes with convalescent plasma treatment. | ||
Mar 7 2024 |
et al., Biomedicines, doi:10.3390/biomedicines12030605 | Insulin and Metformin Administration: Unravelling the Multifaceted Association with Mortality across Various Clinical Settings Considering Type 2 Diabetes Mellitus and COVID-19 |
| 62% higher mortality (p=0.12). Retrospective 430 hospitalized COVID-19 patients with type 2 diabetes in Poland showing lower mortality with metformin and higher mortality with remdesivir, convalescent plasma, and aspirin in univariable analysis. These results were not.. | ||
Feb 15 2024 |
et al., Acta Medica Philippina, doi:10.47895/amp.vi0.4903 | Convalescent Plasma as Adjunctive Therapy for Hospitalized Patients with COVID-19: The Co-CLARITY Trial |
| 400% higher mortality (p=0.49), 100% higher ICU admission (p=1), and 7% longer hospitalization (p=0.7). Early terminated RCT 44 hospitalized COVID-19 patients showing no significant differences with convalescent plasma treatment. | ||
Dec 29 2023 |
et al., Open Forum Infectious Diseases, doi:10.1093/ofid/ofad686 | COVID-19 Convalescent Plasma Therapy: Long Term Implications |
| 2% higher long COVID (p=0.88). Long COVID (PASC) results for [Ortigoza] showing no significant difference with convalescent plasma treatment. | ||
Dec 1 2023 |
et al., Scientific Reports, doi:10.1038/s41598-023-48145-x | COVID-19 outcome is not affected by anti-CD20 or high-titer convalescent plasma in immunosuppressed patients |
| 4% higher mortality (p=1). Retrospective 144 immunocompromised patients treated with anti-CD20 therapy prior to contracting COVID-19. Among 50 patients hospitalized within 14 days, administration of high-titer convalescent plasma in the first 14 days was not associ.. | ||
Oct 31 2023 |
et al., mBio, doi:10.1128/mbio.00618-23 | Early antibody treatment, inflammation, and risk of post-COVID conditions |
| Long COVID results for [Baksh], showing early COVID-19 convalescent plasma (CCP) treatment within 5 days of symptom onset was associated with lower odds of post-COVID conditions (PCC) compared to late CCP treatment within a subset of pat.. | ||
Jun 10 2023 |
et al., Value in Health, doi:10.1016/j.jval.2023.03.2056 | Variation in Demographic Characteristics, Socioeconomic Status, Clinical Presentation and Selected Treatments in Mortality Among Patients with a Diagnosis of COVID-19 in the United States |
| Retrospective analysis of mortality for COVID-19 patients in the USA. Authors do not provide adjusted results, preventing any strong evidence. However it is notable that, despite comparable treatment frequencies, the mortality for patient.. | ||
Apr 5 2023 |
et al., The American Journal of Tropical Medicine and Hygiene, doi:10.4269/ajtmh.22-0705 | Predictors of Mortality among Patients Hospitalized with COVID-19 during the First Wave in India: A Multisite Case-Control Study |
| 270% higher mortality (p=0.07). Case control study with 2,431 hospitalized COVID-19 patients in India, showing higher mortality with convalescent plasma treatment, without statistical significance. | ||
Mar 23 2023 |
et al., NCT04412486 | An Open Label Trial of Transfusion of COVID-19 Convalescent Plasma (CCP) to Patients With Moderate to Severe COVID-19 |
| 86 patient convalescent plasma late treatment study with results not reported over 3 years after completion. | ||
Feb 15 2023 |
et al., Saudi Pharmaceutical Journal, doi:10.1016/j.jsps.2023.02.004 | Comprehensive evaluation of six interventions for hospitalized patients with COVID-19: A propensity score matching study |
| 14% higher mortality (p=0.39), 17% higher progression (p=0.05), 43% longer ICU admission (p=0.003), and 32% longer hospitalization (p=0.01). PSM retrospective 29 hospitals in Saudi Arabia, showing longer ICU and hospitalization time with convalescent plasma, but no significant difference in mortality. | ||
Jan 31 2023 |
et al., The Journal of Infectious Diseases, doi:10.1093/infdis/jiad023 | Symptom duration and resolution with early outpatient treatment of convalescent plasma for COVID-19: a randomized trial |
| 1% worse recovery (p=0.62) and 4% higher long COVID (p=0.78). RCT 1,070 outpatients in the USA, showing no significant difference in recovery with convalescent plasma treatment. Long COVID results are from [Gebo] | ||
Dec 31 2022 |
, NCT04333251 | Evaluating Convalescent Plasma to Decrease Coronavirus Associated Complications. A Phase I Study Comparing the Efficacy and Safety of High-titer Anti-Sars-CoV-2 Plasma vs Best Supportive Care in Hospitalized Patients With Interstitial Pneumonia Due to COVID-19 |
| Estimated 115 patient convalescent plasma late treatment RCT with results not reported over 3 years after estimated completion. | ||
Dec 29 2022 |
et al., Nature Cancer, doi:10.1038/s43018-022-00503-w | Anti-SARS-CoV-2 antibody-containing plasma improves outcome in patients with hematologic or solid cancer and severe COVID-19: a randomized clinical trial |
| 8% lower mortality (p=0.39), 2% higher ventilation (p=1), and 22% improved 7-point scale results (p=0.22). RCT 134 hospitalized patients showing no significant difference in outcomes with convalescent plasma for all patients, however significantly improved mortality and time to improvement was seen for patients with cancer. | ||
Dec 16 2022 |
et al., JAMA, doi:10.1001/jama.2022.23257 | Long-term (180-Day) Outcomes in Critically Ill Patients With COVID-19 in the REMAP-CAP Randomized Clinical Trial |
| 1% lower mortality (p=0.9). Long-term followup for the REMAP-CAP very late stage ICU trial, showing no significant difference with convalescent plasma treatment. | ||
Nov 30 2022 |
et al., Chest, doi:10.1016/j.chest.2022.06.029 | Neutralizing COVID-19 Convalescent Plasma in Adults Hospitalized With COVID-19 |
| 3% higher mortality (p=0.86) and 4% worse 7-point scale results (p=0.76). RCT 947 hospitalized patients in the USA, showing no signficant difference with convalescent plasma treatment. | ||
Nov 22 2022 |
et al., BMC Infectious Diseases, doi:10.1186/s12879-022-07716-5 | Convalescent or standard plasma versus standard of care in the treatment of COVID-19 patients with respiratory impairment: short and long-term effects. A three-arm randomized controlled clinical trial |
| 25% higher mortality (p=0.54), 10% higher combined mortality/intubation (p=0.76), and 6% slower viral clearance (p=0.76). RCT 180 hospitalized COVID-19 patients with respiratory impairment in Italy showing no significant improvement in mortality or mechanical ventilation with either standard plasma or COVID-19 convalescent plasma compared to standard of care. | ||
Sep 30 2022 |
et al., Scientific Reports, doi:10.1038/s41598-022-19629-z | A randomized placebo-controlled trial of convalescent plasma for adults hospitalized with COVID-19 pneumonia |
| 76% higher mortality (p=0.43), 31% higher ICU admission (p=0.77), and 41% worse 7-point scale results (p=0.32). RCT 147 patients in Denmark, showing no significant difference in outcomes with convalescent plasma. The trial was terminated due to futility. | ||
Aug 26 2022 |
et al., medRxiv, doi:10.1101/2022.08.25.22279181 | International Multicenter Study Comparing Cancer to Non-Cancer Patients with COVID-19: Impact of Risk Factors and Treatment Modalities on Survivorship |
| 240% higher mortality (p<0.0001). Retrospective 3,966 COVID-19 patients, 1,115 with cancer, showing lower mortality with remdesivir and higher mortality with convalescent plasma. | ||
Aug 23 2022 |
et al., Clinical Microbiology and Infection, doi:10.1016/j.cmi.2022.08.005 | Outpatient convalescent plasma therapy for high-risk patients with early COVID-19. A randomized placebo-controlled trial |
| 14% lower progression (p=0.42), 39% lower hospitalization (p=0.22), and 1% worse recovery (p=0.92). RCT 416 outpatients in the Netherlands, showing no significant difference with convalesent plasma treatment. Hospitalization was lower, and improved results were seen with ≤5 days of symptoms, without statistical significance. | ||
Aug 10 2022 |
et al., medRxiv, doi:10.1101/2022.08.09.22278329 | COVID-19 convalescent plasma to treat hospitalised COVID-19 patients with or without underlying immunodeficiency |
| 49% lower mortality (p=0.16), 68% higher progression (p=0.18), and 7% longer hospitalization (p=0.99). RCT 120 hospitalized patients in France, showing no significant difference in outcomes with convalescent plasma treatment, with the exception of lower mortality in the subgroup of immunosuppressed patients. | ||
Jun 30 2022 |
et al., The Lancet Regional Health - Americas, doi:10.1016/j.lana.2022.100216 | Treatment of severe COVID-19 patients with either low- or high-volume of convalescent plasma versus standard of care: A multicenter Bayesian randomized open-label clinical trial (COOP-COVID-19-MCTI) |
| 52% higher mortality (p=0.37). RCT 129 severe COVID-19 patients in Brazil, showing no significant difference in outcomes with convalescent plasma. | ||
Jun 30 2022 |
et al., NCT04634422 | Plasma Exchange (PLEX) and Convalescent Plasma (CCP) in COVID-19 Patients With Multiorgan Failure - the COVID PLEX+CCP Trial |
| Estimated 220 patient convalescent plasma late treatment RCT with results not reported over 3 years after estimated completion. | ||
Jun 27 2022 |
et al., BMC Infectious Diseases, doi:10.1186/s12879-022-07560-7 | Safety and efficacy of convalescent plasma for severe COVID-19: a randomized, single blinded, parallel, controlled clinical study |
| 220% higher mortality (p=0.16), 38% higher hospital discharge (p=0.04), and 25% worse viral clearance (p=0.72). RCT 91 hospitalized patients in Colombia showing shorter time to discharge with convalescent plasma, but higher mortality (without statistical significance). | ||
Apr 7 2022 |
et al., NCT05077930 | Validation Protocol for The Clinical Use of Convalescent Plasma for Hospitalized Patients With COVID-19. A Prospective Study at a Hospital in Southern Brazil. |
| 38 patient convalescent plasma late treatment RCT with results not reported over 4 years after completion. | ||
Apr 6 2022 |
et al., BMJ Open, doi:10.1136/bmjopen-2021-055189 | Efficacy of convalescent plasma therapy in the patient with COVID-19: a randomised control trial (COPLA-II trial) |
| 14% higher mortality (p=0.62), 13% higher ventilation (p=0.76), 2% longer ICU admission (p=0.8), and no change in hospitalization (p=0.98). RCT 400 hospitalized severe COVID-19 patients in India showing no significant difference in time to clinical improvement, mortality, or other outcomes with convalescent plasma compared to standard treatment. In a subgroup analysis, result.. | ||
Mar 31 2022 |
et al., Emerging Infectious Diseases, doi:10.3201/eid2803.212299 | High-Dose Convalescent Plasma for Treatment of Severe COVID-19 |
| 13% lower mortality (p=0.67). RCT 110 hospitalized patients in Brazil, showing no significant difference in outcomes with high-dose convalescent plasma. | ||
Mar 1 2022 |
et al., medRxiv, doi:10.1101/2022.02.23.22271424 | Use of Convalescent Plasma Therapy with Best Available Treatment (BAT) among Hospitalized COVID-19 Patients: A Multi-Center Study |
| 29% higher mortality (p=0.54) and 60% longer hospitalization (p=0.07). Prospective study of 65 hospitalized COVID-19 patients in the Philippines treated with convalescent plasma and 65 matched controls showing no significant difference in mortality and longer hospitalization with treatment. | ||
Feb 15 2022 |
et al., Scientific Reports, doi:10.1038/s41598-022-06221-8 | Convalescent plasma in the treatment of moderate to severe COVID-19 pneumonia: a randomized controlled trial (PROTECT-Patient Trial) |
| 17% lower mortality (p=0.65), 67% lower ventilation (p=0.36), 5% greater improvement (p=1), and 3% lower hospital discharge (p=1). RCT 103 hospitalized patients in South Africa, showing no significant difference in outcomes with convalescent plasma. | ||
Feb 9 2022 |
et al., The Lancet Respiratory Medicine, doi:10.1016/S2213-2600(21)00545-2 | High-titre methylene blue-treated convalescent plasma as an early treatment for outpatients with COVID-19: a randomised, placebo-controlled trial |
| 80% lower mortality (p=0.5), 5% higher hospitalization (p=1), 5% worse recovery (p=0.67), and 4% worse viral clearance (p=0.33). RCT 188 convalescent plasma and 188 control patients, showing no significant difference in outcomes. | ||
Feb 7 2022 |
et al., Frontiers in Immunology, doi:10.3389/fimmu.2022.817829 | Convalescent Plasma Treatment in Patients with Covid-19: A Systematic Review and Meta-Analysis |
| Meta-analysis of 16 RCTs with 16,317 COVID-19 patients showing no significant difference in mortality, mechanical ventilation, time to clinical improvement, or time to discharge with convalescent plasma treatment. Subgroup analyses in cri.. | ||
Jan 30 2022 |
et al., NCT04528368 | Efficacy and Safety of Using Convalescent Plasma for Treating Patients With COVID-19 Pneumonia Without Indication of Ventilatory Support |
| Estimated 60 patient convalescent plasma late treatment RCT with results not reported over 4 years after estimated completion. | ||
Jan 26 2022 |
et al., NCT04649879 | Convalescent Plasma for Treatment of COVID-19: An Open Randomised Controlled Trial |
| 59 patient convalescent plasma late treatment RCT with results not reported over 4 years after completion. | ||
Jan 9 2022 |
et al., Transfusion Medicine, doi:10.1111/tme.12851 | Effect of convalescent plasma as complementary treatment in patients with moderate COVID-19 infection |
| 12% lower mortality (p=1). RCT 158 patients in Ecuador, showing no significant difference in mortality with convalescent plasma. Authors note indications of improved results for earlier treatment. | ||
Jan 1 2022 |
et al., Tanaffos 21:1 | Effect of Convalescent Plasma Therapy on Clinical Improvement of COVID-19 Patients: A Randomized Clinical Trial |
| 45% higher mortality (p=0.38), 8% higher ICU admission (p=0.85), 250% higher ARDS (p=0.16), and 10% longer hospitalization (p=0.39). RCT 120 hospitalized patients in Iran, showing no significant differences with convalescent plasma treatment. | ||
Dec 31 2021 |
et al., NCT04438694 | Assessment of the Effect of Convalescent Plasma Therapy in Patients With Life-threatening COVID19 Infection |
| Estimated 67 patient convalescent plasma late treatment RCT with results not reported over 4 years after estimated completion. | ||
Dec 31 2021 |
et al., NCT04873414 | Clinical Trial of Convalescent Plasma Administration as Adjunct Therapy for COVID-19 (Uji Klinik Pemberian Plasma Konvalesen Sebagai Terapi Tambahan COVID-19) |
| Estimated 364 patient convalescent plasma late treatment RCT with results not reported over 4 years after estimated completion. | ||
Dec 31 2021 |
et al., NCT05247307 | Clinical Efficacy of the Infusion of Donor Plasma, Convalescent From SARS-CoV-2 Infection, to Patients With Recent Infection. |
| 93 patient convalescent plasma late treatment RCT with results not reported over 4 years after completion. | ||
Dec 21 2021 |
et al., New England Journal of Medicine, doi:10.1056/NEJMoa2119657 (date from preprint) | Early Outpatient Treatment for Covid-19 with Convalescent Plasma |
| 86% lower mortality (p=0.12) and 54% lower hospitalization (p=0.005). RCT 1,181 outpatients in the USA, mean 6 days from symptom onset, showing lower hospitalization with treatment. | ||
Dec 15 2021 |
et al., Journal of Clinical Investigation, doi:10.1172/JCI155114 | A randomized controlled study of convalescent plasma for individuals hospitalized with COVID-19 pneumonia |
| 81% lower mortality (p=0.03), 44% greater improvement (p=0.18), and 51% lower ventilation (p=0.16). RCT 79 hospitalized patients in the USA, showing significant benefit in clinical severity score and 28-day mortality with convalescent plasma treatment. | ||
Dec 13 2021 |
et al., JAMA Internal Medicine, doi:10.1001/jamainternmed.2021.6850 | Efficacy and Safety of COVID-19 Convalescent Plasma in Hospitalized Patients |
| 12% lower mortality (p=0.45), 8% improved 7-point scale results (p=0.5), and 2% higher long COVID (p=0.88). RCT 941 hospitalized patients in the USA, showing no significant difference with convalescent plasma treatment. PASC results are from [Yoon] | ||
Dec 4 2021 |
et al., BMC Research Notes, doi:10.1186/s13104-021-05847-7 | Convalescence plasma treatment of COVID-19: results from a prematurely terminated randomized controlled open-label study in Southern Sweden |
| 19% lower progression (p=1), 57% higher need for oxygen therapy (p=0.43), and 62% longer hospitalization (p=0.21). RCT 31 hospitalized patients requiring supplemental oxygen in Sweden, showing no significant difference in outcomes with convalescent plasma. | ||
Dec 1 2021 |
et al., NCT04391101 | Efficacy of Convalescent Plasma for the Treatment of Severe SARS-CoV-2 Infection: A Randomized, Open Label Clinical Trial |
| Estimated 231 patient convalescent plasma late treatment RCT with results not reported over 4 years after estimated completion. | ||
Nov 29 2021 |
et al., JAMA Network Open, doi:10.1001/jamanetworkopen.2021.36246 | Effect of High-Titer Convalescent Plasma on Progression to Severe Respiratory Failure or Death in Hospitalized Patients With COVID-19 Pneumonia |
| 23% lower mortality (p=0.47), 4% higher ventilation (p=1), and 12% lower progression (p=0.54). RCT 487 patients in Italy, showing no significant difference in outcomes with convalescent plasma. | ||
Nov 20 2021 |
et al., PACTR202006760881890 | A Multi-Center, Randomized, Double-Blind, Placebo- Controlled Clinical Trial of the Safety and Efficacy of Convalescent Plasma for the Treatment of COVID-19 in Hospitalized Patients in Lagos State |
| 17% higher mortality (p=1). RCT 22 hospitalized patients, show no significant difference in mortality with convalescent plasma. Results are from [Axfors]. | ||
Nov 20 2021 |
et al., BMC Infectious Diseases, doi:10.1186/s12879-021-06829-7 | Association between convalescent plasma treatment and mortality in COVID-19: a collaborative systematic review and meta-analysis of randomized clinical trials |
| Meta analysis of 33 RCTs with 16,477 patients showing no significant reduction in all-cause mortality with convalescent plasma treatment for COVID-19. There was no difference between subgroups based on patient setting, timing of plasma.. | ||
Oct 29 2021 |
et al., NCT04681430 | Reconvalescent Plasma / Camostat Mesylate Early in Sars-CoV-2 Q-PCR (COVID-19) Positive High-risk Individuals |
| 22 patient convalescent plasma early treatment RCT with results not reported over 4 years after completion. | ||
Oct 15 2021 |
et al., Journal of Clinical Investigation, doi:10.1172/JCI152264 | Results of the CAPSID randomized trial for high-dose convalescent plasma in patients with severe COVID-19 |
| 37% lower mortality (p=0.19) and 16% improved recovery (p=0.32). RCT 105 hospitalized patients in Germany, 53 treated with convalescent plasma, showing no significant difference in mortality or the primary composite outcome of survival and no longer fulfilling criteria for severe COVID-19 on day 21. | ||
Sep 9 2021 |
et al., Nature Medicine, doi:10.1038/s41591-021-01488-2 | Convalescent plasma for hospitalized patients with COVID-19: an open-label, randomized controlled trial |
| 13% higher mortality (p=0.33) and 16% higher combined mortality/intubation (p=0.18). RCT 940 hospitalized patients, 614 assigned to convalescent plasma, showing no significant differences. | ||
Sep 1 2021 |
et al., NCT04712344 | Randomized Controlled Phase 2b Clinical Study in Parallel Groups for the Assessment of Efficacy and Safety of Therapy With COVID-19 Convalescent Plasma Plus Standard Treatment vs. Standard Treatment Alone of Subjects With Severe COVID-19 |
| Estimated 58 patient convalescent plasma late treatment RCT with results not reported over 4 years after estimated completion. | ||
Aug 26 2021 |
et al., European Respiratory Journal, doi:10.1183/13993003.01724-2021 | Early high antibody titre convalescent plasma for hospitalised COVID-19 patients: DAWn-plasma |
| 1% lower mortality (p=0.98), 8% higher ventilation (p=0.78), and no change in ICU admission (p=1). RCT 489 hospitalized COVID-19 patients in Belgium, showing no significant difference in outcomes with convalescent plasma. | ||
Aug 23 2021 |
et al., NCT04421404 | Effects of COVID-19 Convalescent Plasma (CCP) on Coronavirus-associated Complications in Hospitalized Patients (CAPRI) |
| 425% higher ventilation (p=0.21) and 425% higher progression (p=0.21). RCT 34 hospitalized patients in the USA, showing no significant difference with convalescent plasma treatment. | ||
Aug 18 2021 |
et al., NEJM, doi:10.1056/NEJMoa2103784 | Early Convalescent Plasma for High-Risk Outpatients with Covid-19 |
| 396% higher mortality (p=0.22), 10% lower hospitalization (p=0.59), and 6% lower progression (p=0.7). RCT 511 emergency department patients, 257 assigned to convalescent plasma, showing no significant difference in outcomes. | ||
Aug 12 2021 |
et al., NCT04438057 | Evaluating the Efficacy of Convalescent Plasma in Symptomatic Outpatients Infected With COVID-19 |
| 441% higher hospitalization (p=0.55). RCT 72 outpatients in the USA showing no significant difference with convalescent plasma treatment. | ||
Aug 9 2021 |
et al., NCT04374487 | A Phase II, Open Label, Randomized Controlled Trial to Assess the Safety and Efficacy of Convalescent Plasma to Limit COVID-19 Associated Complications |
| 100 patient convalescent plasma late treatment RCT with results not reported over 4 years after completion. | ||
Aug 9 2021 |
et al., BMJ Open Respiratory Research, doi:10.1136/bmjresp-2021-001017 | Efficacy of convalescent plasma for treatment of COVID-19 in Uganda |
| 21% higher mortality (p=0.8), 9% lower progression (p=1), no change in recovery (p=0.77), and 50% slower viral clearance (p=0.2). RCT 136 hospitalized COVID-19 patients in Uganda, showing no significant benefit with convalescent plasma treatment. | ||
Jul 8 2021 |
et al., European Respiratory Journal, doi:10.1183/13993003.01471-2021 | Convalescent plasma for COVID-19 in hospitalised patients: an open-label, randomised clinical trial |
| 38% higher mortality (p=0.42), 11% worse improvement (p=0.74), and 67% longer hospitalization (p=0.87). RCT 160 hospitalized patients in Brazil, showing no significant difference in outcomes with convalescent plasma. | ||
Jun 30 2021 |
et al., NCT04361253 | A Prospective, Randomized, Double-Masked, Placebo-Controlled Trial of High-Titer COVID-19 Convalescent Plasma (HT-CCP) for the Treatment of Hospitalized Patients With COVID-19 of Moderate Severity |
| 45 patient convalescent plasma late treatment RCT with results not reported over 4 years after completion. | ||
Jun 21 2021 |
et al., The Journal of Infectious Diseases, doi:10.1093/infdis/jiab330 | Early Convalescent Plasma Therapy and Mortality Among US Veterans Hospitalized With Nonsevere COVID-19: An Observational Analysis Emulating a Target Trial |
| 4% higher mortality (p=0.88). Target trial emulation with 4,755 patients showing no significant difference in 30-day mortality with convalescent plasma. | ||
Jun 19 2021 |
et al., NCT04468009 | Treatment of Critically Ill Patients With Covid-19 With Convalescent Plasma |
| 134 patient convalescent plasma late treatment RCT with results not reported over 4 years after completion. | ||
Jun 1 2021 |
et al., NCT04372979 | Evaluation Of Efficacy Of COVID-19 Convalescent Plasma Versus Standard Plasma In The Early Care Of COVID-19 Patients Hospitalized Outside Intensive Care Units. |
| 18 patient convalescent plasma late treatment RCT with results not reported over 5 years after completion. | ||
May 27 2021 |
et al., Nature Communications, doi:10.1038/s41467-021-23469-2 | Effects of potent neutralizing antibodies from convalescent plasma in patients hospitalized for severe SARS-CoV-2 infection |
| 4% lower mortality (p=0.95) and 12% higher hospital discharge (p=0.68). RCT 86 hospitalized patients, 43 treated with convalescent plasma, showing no significant differences with treatment. Authors conclude that the most likely explanation was already high antibody titers on the day of inclusion, and they rec.. | ||
May 26 2021 |
et al., NCT04374526 | Early transfusIon of COVID-19 Convalescent Plasma in Elderly COVID-19 Patients to Prevent Disease Progression |
| 100% higher mortality (p=1). RCT 12 patients in Italy, showing no significant difference with convalescent plasma treatment. Results are from [Axfors]. | ||
May 15 2021 |
et al., NCT04385043 | Efficacy and Safety of Hyperimmune Plasma Treatment in Patients With COVID-19 Severe Infection |
| Estimated 400 patient convalescent plasma late treatment RCT with results not reported over 5 years after estimated completion. | ||
May 5 2021 |
et al., NCT04442191 | Infusion of Convalescent Plasma for the Treatment of Patients Infected With Severe Acute Respiratory Syndrome-Coronavirus-2 (COVID-19): A Double-blinded, Placebo-controlled, Proof-of-concept Study |
| Estimated 50 patient convalescent plasma late treatment RCT with results not reported over 5 years after estimated completion. | ||
May 1 2021 |
et al., NCT04358783 | Phase II, Randomized, Double-blind, Controlled Clinical Trial Evaluating the Efficacy and Safety of Plasma From Patients Cured of COVID-19 Compared to the Best Available Therapy in Subjects With SARS-CoV-2 Pneumonia |
| 31 patient convalescent plasma late treatment RCT with results not reported over 5 years after completion. | ||
Apr 30 2021 |
et al., PLOS ONE, doi:10.1371/journal.pone.0251085 | Epidemiology, outcomes, and utilization of intensive care unit resources for critically ill COVID-19 patients in Libya: A prospective multi-center cohort study |
| 16% higher mortality (p=0.39). Prospective study of 465 COVID-19 ICU patients in Libya showing no significant differences with treatment. | ||
Apr 16 2021 |
et al., Critical Care Medicine, doi:10.1097/CCM.0000000000005066 | Severe Acute Respiratory Syndrome Coronavirus 2 Convalescent Plasma Versus Standard Plasma in Coronavirus Disease 2019 Infected Hospitalized Patients in New York |
| 19% lower mortality (p=0.75) and no change in improvement (p=1). RCT 74 hospitalized patients in the USA, showing no significant difference with convalescent plasma treatment. | ||
Apr 10 2021 |
et al., Internal and Emergency Medicine, doi:10.1007/s11739-021-02734-8 | A randomized clinical trial evaluating the immunomodulatory effect of convalescent plasma on COVID-19-related cytokine storm |
| 30% longer hospitalization (p=0.06). RCT 62 hospitalized patients in Iran, showing no significant difference in mortality and length of stay with convalescent plasma. | ||
Mar 31 2021 |
, NCT05578391 | PHASE II Clinical Trial Randomized And Controlled To Assess Need Of Oxygen On Day 14 After Administration Of Passive Immunotherapy In Patients Infected By SARS-CoV-2 With Risks Factors For Severe Disease |
| 61 patient convalescent plasma late treatment RCT with results not reported over 5 years after completion. | ||
Mar 31 2021 |
et al., medRxiv, doi:10.1101/2021.03.28.21254507 | Efficacy and safety of convalescent plasma and intravenous immunoglobulin in critically ill COVID-19 patients. A controlled clinical trial |
| 7% higher mortality (p=0.76). RCT 190 hospitalized severe condition patients in Mexico, showing no significant difference between convalescent plasma and human immunoglobulin treatment. | ||
Mar 18 2021 |
et al., New England Journal of Medicine, doi:10.1056/NEJMoa2031893 | Convalescent Plasma Antibody Levels and the Risk of Death from Covid-19 |
| Retrospective 3,082 hospitalized COVID-19 patients in the U.S. showing lower 30-day mortality with high antibody level convalescent plasma compared to low antibody level plasma. The effect was only seen in patients not on mechanical venti.. | ||
Mar 3 2021 |
et al., PLOS Medicine, doi:10.1371/journal.pmed.1003415 | Early versus deferred anti-SARS-CoV-2 convalescent plasma in patients admitted for COVID-19: A randomized phase II clinical trial |
| 247% higher mortality (p=0.17), 163% higher ventilation (p=0.22), and 23% higher progression (p=0.51). Small RCT with 28 early and 30 deferred (treated according to prespecified deterioration criteria) convalescent plasma patients, not showing significant differences. "Early" is relative, with a median of 5 days from symptom onse.. | ||
Feb 4 2021 |
et al., NCT04547127 | A Multicenter, Randomized, Open-label, Parallel Group Pilot Study to Evaluate Safety and Efficacy of Convalescent Methylene Blue Treated (MBT) Plasma From Donors Recovered From Coronavirus Disease 2019 (COVID-19) With Standard Medical Treatment (SMT) Versus SMT Alone in Subjects With COVID-19 Requiring Admission to the Intensive Care Unit (ICU) |
| 200 patient convalescent plasma late treatment RCT with results not reported over 5 years after completion. | ||
Feb 1 2021 |
et al., NCT04425837 | Effectiveness and Safety of Convalescent Plasma in Patients With High-risk COVID-19: A Randomized, Controlled Study CRI-CP (Coronavirus Investigation - Convalescent Plasma) |
| Estimated 236 patient convalescent plasma late treatment RCT with results not reported over 5 years after estimated completion. | ||
Jan 15 2021 |
, The Lancet, doi:10.1016/S0140-6736(21)00897-7 (press release 1/15/2021) | Convalescent plasma in patients admitted to hospital with COVID-19 (RECOVERY): a randomised controlled, open-label, platform trial |
| 1% higher mortality (p=0.78) and 1% lower hospital discharge (p=0.57). RCT 16,287 hospitalized patients in the UK, showing no significant differences with convalescent plasma treatment. Subgroup analysis shows better results for those treated ≤7 days from symptom onset. 6-month results are from [Horby] | ||
Jan 6 2021 |
et al., NEJM, doi:10.1056/NEJMoa2033700 | Early High-Titer Plasma Therapy to Prevent Severe Covid-19 in Older Adults |
| 67% lower ICU admission (p=0.17) and 48% lower progression (p=0.03). RCT 160 patients ≥65 with symptom onset <72 hours, 80 treated with convalescent plasma, showing lower progression to severe disease with treatment. | ||
Dec 31 2020 |
et al., NCT04388410 | Phase 2b/3 Trial to Evaluate the Safety and Efficacy of Plasma Transfusion From Convalescent Patients With SARS-CoV-2 Infection on Severity and Mortality of COVID-19 in Hospitalized Patients |
| Estimated 410 patient convalescent plasma late treatment RCT with results not reported over 5 years after estimated completion. | ||
Dec 30 2020 |
et al., NCT04385186 | Inactivated Convalescent Plasma as a Therapeutic Alternative in Hospitalized Patients CoViD-19 |
| Estimated 60 patient convalescent plasma late treatment RCT with results not reported over 5 years after estimated completion. | ||
Dec 30 2020 |
et al., NCT04366245 | Phase I / II Multicentre, Randomized and Controlled Clinical Trial to Evaluate the Efficacy of Treatment With Hyperimmune Plasma Obtained From Convalescent Antibodies of COVID-19 Infection |
| 72 patient convalescent plasma late treatment RCT with results not reported over 5 years after completion. | ||
Nov 29 2020 |
et al., Nature Communications, doi:10.1038/s41467-022-28064-7 (date from preprint) | A phase 2 single center open label randomised control trial for convalescent plasma therapy in patients with severe COVID-19 |
| 33% lower mortality (p=0.34). RCT 80 severe COVID-19 patients in India showing no significant difference in 30-day mortality with convalescent plasma therapy (CPT). Patients receiving CPT had greater reduction in inflammatory cytokines, but this did not translate to c.. | ||
Nov 24 2020 |
et al., NEJM, doi:10.1056/NEJMoa2031304 | A Randomized Trial of Convalescent Plasma in Covid-19 Severe Pneumonia |
| 4% lower mortality (p=1) and 19% improved 7-point scale results (p=0.4). RCT 333 hospitalized patients in Argentina, 228 treated with convalescent plasma, showing no significant differences in clinical status or mortality. | ||
Nov 4 2020 |
et al., Scientific Reports, doi:10.1038/s41598-021-89444-5 (date from preprint) | Randomized controlled trial of convalescent plasma therapy against standard therapy in patients with severe COVID-19 disease |
| 33% lower ventilation (p=0.47) and 22% shorter hospitalization (p=0.12). Small RCT with 40 hospitalized patients in Bahrain, 20 treated with convalescent plasma, not showing significant differences. | ||
Oct 31 2020 |
et al., NCT04380935 | Effectiveness and Safety of Convalescent Plasma Therapy on COVID-19 Patients With Acute Respiratory Distress Syndrome in Referral Hospitals in Indonesia |
| Estimated 60 patient convalescent plasma late treatment RCT with results not reported over 5 years after estimated completion. | ||
Oct 30 2020 |
et al., NCT04403477 | Convalescent Plasma Transfusion Therapy in Severe COVID-19 Patients- a Tolerability, Efficacy and Dose-response Phase II RCT |
| Estimated 60 patient convalescent plasma late treatment RCT with results not reported over 5 years after estimated completion. | ||
Oct 27 2020 |
et al., Anais da Academia Brasileira de Ciências, doi:10.1590/0001-3765202220210202 | Comparison of safety and efficacy of convalescent plasma with fresh frozen plasma in severe covid-19 patients |
| 323% higher mortality (p=0.22), 221% higher ventilation (p=0.33), 25% shorter hospitalization (p=0.08), and 33% improved viral clearance (p=0.11). RCT 29 severe COVID-19 patients showing no significant differences with convalescent plasma compared to fresh frozen plasma. | ||
Oct 22 2020 |
et al., BMJ, doi:10.1136/bmj.m3939 | Convalescent plasma in the management of moderate covid-19 in adults in India: open label phase II multicentre randomised controlled trial (PLACID Trial) |
| 7% higher mortality (p=0.74), 7% higher progression (p=0.74), 1% lower ventilation (p=0.98), and 28% improved viral clearance (p=0.02). RCT 464 hospitalized patients in India, 235 treated with convalescent plasma, showing no improvement in combined death at 28 days or progression to severe disease. | ||
Sep 30 2020 |
et al., NCT04542967 | Convalescent Plasma as a Treatment for Patients With Severe COVID-19 Disease |
| 150 patient convalescent plasma late treatment RCT with results not reported over 5 years after completion. | ||
Sep 29 2020 |
et al., medRxiv, doi:10.1101/2020.08.26.20182444 | Convalescent Plasma for COVID-19: A multicenter, randomized clinical trial |
| 88% lower mortality (p=0.12) and 93% lower progression (p=0.01). Early terminated RCT with 81 hospitalized patients, 38 treated with convalescent plasma, showing lower progression with treatment. | ||
Sep 23 2020 |
et al., NCT04392414 | Randomized, Open Label, Prospective Study of the Safety and Efficacy of Hyperimmune Convalescent Plasma in Moderate and Severe COVID-19 Disease |
| 60 patient convalescent plasma late treatment RCT with results not reported over 5 years after completion. | ||
Jun 17 2020 |
et al., NCT04442958 | Effectiveness of Convalescent Immune Plasma Therapy in Severe COVID-19 Patients With Acute Respiratory Distress Syndrome |
| 60 patient convalescent plasma late treatment RCT with results not reported over 5 years after completion. | ||
Jun 3 2020 |
et al., JAMA, doi:10.1001/jama.2020.10044 | Effect of Convalescent Plasma Therapy on Time to Clinical Improvement in Patients With Severe and Life-threatening COVID-19: A Randomized Clinical Trial |
| 35% lower mortality (p=0.3), 15% greater improvement (p=0.37), and 76% improved viral clearance (p=0.01). Small RCT 103 severe condition patients, 52 treated with convalescent plasma, showing improved viral clearance but no statistically significant improvements in mortality or clinical improvement. ChiCTR2000029757. | ||