Randomized controlled trial of convalescent plasma therapy against standard therapy in patients with severe COVID-19 disease
et al., Scientific Reports, doi:10.1038/s41598-021-89444-5, NCT04356534, Nov 2020 (preprint)
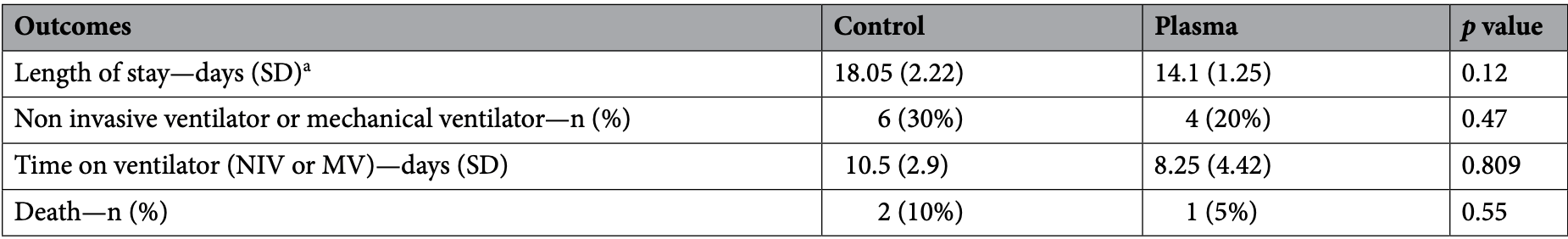
Small RCT with 40 hospitalized patients in Bahrain, 20 treated with convalescent plasma, not showing significant differences.
|
risk of death, 50.0% lower, RR 0.50, p = 0.55, treatment 1 of 20 (5.0%), control 2 of 20 (10.0%), NNT 20.
|
|
noninvasive or mechanical ventilation, 33.3% lower, RR 0.67, p = 0.47, treatment 4 of 20 (20.0%), control 6 of 20 (30.0%), NNT 10, primary outcome.
|
|
hospitalization time, 21.9% lower, relative time 0.78, p = 0.12, treatment 20, control 20.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
AlQahtani et al., 4 Nov 2020, Randomized Controlled Trial, Bahrain, peer-reviewed, 11 authors, study period 19 April, 2020 - 9 July, 2020, trial NCT04356534 (history).
Randomized controlled trial of convalescent plasma therapy against standard therapy in patients with severe COVID-19 disease
Scientific Reports, doi:10.1038/s41598-021-89444-5
Convalescent plasma (CP) therapy in COVID-19 disease may improve clinical outcome in severe disease. This pilot study was undertaken to inform feasibility and safety of further definitive studies. This was a prospective, interventional and randomized open label pilot trial in patients with severe COVID-19. Twenty COVID-19 patients received two 200 ml transfusions of convalescent patient CP over 24-h compared with 20 who received standard of care. The primary outcome was the requirement for ventilation (non-invasive or mechanical ventilation). The secondary outcomes were biochemical parameters and mortality at 28 days. The CP group were a higher risk group with higher ferritin levels (p < 0.05) though respiratory indices did not differ. The primary outcome measure was required in 6 controls and 4 patients on CP (risk ratio 0.67, 95% CI 0.22-2.0, p = 0.72); mean time on ventilation (NIV or MV) did not differ. There were no differences in secondary measures at the end of the study. Two patients died in the control and one patient in the CP arm. There were no significant differences in the primary or secondary outcome measures between CP and standard therapy, although a larger definitive study is needed for confirmation. However, the study did show that CP therapy appears to be safe in hospitalized COVID-19 patients with hypoxia. Clinical trials registration NCT04356534: 22/04/2020.
Coronavirus disease 2019 (COVID-19 ) is caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and has developed into a pandemic with serious global public health and economic sequelae. As of 25 February 2021, more than 113 million cases have been confirmed worldwide leading to over 2.5 million deaths 1 . There have been a number of reports of medications, such as remdesivir, with antiviral properties that have shown efficacy against SARS-CoV-2 with shorter time to recovery 2 . Whilst there are now several effective vaccines emerging and being used globally, the rate of COVID-19 disease and its complications currently remain high. Plasma therapy using Convalescent Plasma (CP) transfusion refers to a form of passive therapy, where neutralizing antibodies from a recovered donor are injected into the infected patient with the aim of altering the course of the disease 3 . This has been shown to be effective in severe acute respiratory syndrome 4 , Ebola virus infection 5 and in H1N1 influenza 6 . More recently, there has been a report of the use of CP in the treatment of five mechanically ventilated COVID-19 patients with the suggestion of expedited recovery as the patients improved one week after the transfusion 7 . A randomized trial with CP therapy in severely and critically ill COVID-19 patients undertaken in China was stopped early as recruitment slowed down due to the decrease in COVID-19 cases in China 8 . Similarly, an open label randomized trial in the Netherlands was stopped prematurely due to the detection of baseline neutralizing..
Author contributions A.A., A.A. and S.L.A. analyzed the data and wrote the manuscript. S.Y.A., A.Z., A.H., P.W. and M.A. performed data collection, S.O. interpreted data and edited the manuscript. R.C. performed the statistical analysis. M.A.Q. supervised data collection, data analysis and edited the manuscript. All authors reviewed and approved the final version of the manuscript. M.A. is the guarantor of this work.
Competing interests The authors declare no competing interests.
References
Agarwal, Convalescent plasma in the management of moderate covid-19 in adults in India: Open label phase II multicentre randomised controlled trial (PLACID Trial), BMJ, doi:10.1136/bmj.m3939
Beigel, Remdesivir for the treatment of Covid-19-Preliminary report, N. Engl. J. Med, doi:10.1056/NEJMoa2007764
Birkett, Day, Internal pilot studies for estimating sample size, Stat. Med
Cao, A trial of lopinavir-ritonavir in adults hospitalized with severe Covid-19, N. Engl. J. Med, doi:10.1056/NEJMoa2001282
Cavalcanti, Hydroxychloroquine with or without azithromycin in mild-to-moderate Covid-19, N. Engl. J. Med, doi:10.1056/NEJMoa2019014
Cheng, Ferritin in the coronavirus disease 2019 (COVID-19): A systematic review and meta-analysis, J. Clin. Lab. Anal, doi:10.1002/jcla.23618
Corman, Detection of 2019 novel coronavirus (2019-nCoV) by real-time RT-PCR, doi:10.2807/1560-7917.Es.2020.25.3.2000045
Gharbharan, Convalescent plasma for COVID-19. A randomized clinical trial
Gharbharan, Convalescent plasma for COVID-19. A randomized clinical trial, doi:10.1101/2020.07.01.20139857
Hariyanto, Inflammatory and hematologic markers as predictors of severe outcomes in COVID-19 infection: A systematic review and meta-analysis, Am. J. Emerg. Med, doi:10.1016/j.ajem.2020.12.076
Hung, Convalescent plasma treatment reduced mortality in patients with severe pandemic influenza A (H1N1) 2009 virus infection, Clin. Infect. Dis, doi:10.1093/cid/ciq106
Hung, Triple combination of interferon beta-1b, lopinavir-ritonavir, and ribavirin in the treatment of patients admitted to hospital with COVID-19: An open-label, randomised, phase 2 trial, Lancet, doi:10.1016/s0140-6736(20)31042-4
Joyner, Early safety indicators of COVID-19 convalescent plasma in 5000 patients, J. Clin. Investig, doi:10.1172/jci140200
Joyner, Effect of convalescent plasma on mortality among hospitalized patients with COVID-19: Initial three-month experience, doi:10.1101/2020.08.12.20169359
Joyner, Safety update: COVID-19 convalescent plasma in 20,000 hospitalized patients, Mayo Clin, doi:10.1016/j.mayocp.2020.06.028
Ledford, Coronavirus breakthrough: Dexamethasone is first drug shown to save lives, Nature, doi:10.1038/d41586-020-01824-5
Li, Effect of convalescent plasma therapy on time to clinical improvement in patients with severe and life-threatening COVID-19: A randomized clinical trial, JAMA, doi:10.1001/jama.2020.10044
Newson, Confidence intervals for rank statistics: Percentile slopes, differences, and ratios, Stand. Genom. Sci
Pathak, Convalescent plasma is ineffective for covid-19, BMJ, doi:10.1136/bmj.m4072
Sahr, Evaluation of convalescent whole blood for treating Ebola Virus Disease in Freetown, Sierra Leone, J. Infect, doi:10.1016/j.jinf.2016.11.009
Sahu, Jindal, Siddiqui, Cerny, Gerber, Convalescent plasma therapy: A passive therapy for an aggressive COVID-19, J. Med. Virol, doi:10.1002/jmv.26047
Sanfilippo, La Rosa, Oliveri, Astuto, Convalescent plasma for COVID-19: The risk of pulmonary embolism should not be underestimated!, Crit. Care
Shen, Treatment of 5 critically ill patients with COVID-19 with convalescent plasma, JAMA, doi:10.1001/jama.2020.4783
Simonovich, A randomized trial of convalescent plasma in Covid-19 severe pneumonia, N. Engl. J. Med, doi:10.1056/NEJMoa2031304
Soo, Retrospective comparison of convalescent plasma with continuing high-dose methylprednisolone treatment in SARS patients, Clin. Microbiol. Infect, doi:10.1111/j.1469-0691.2004.00956.x
DOI record:
{
"DOI": "10.1038/s41598-021-89444-5",
"ISSN": [
"2045-2322"
],
"URL": "http://dx.doi.org/10.1038/s41598-021-89444-5",
"abstract": "<jats:title>Abstract</jats:title><jats:p>Convalescent plasma (CP) therapy in COVID-19 disease may improve clinical outcome in severe disease. This pilot study was undertaken to inform feasibility and safety of further definitive studies. This was a prospective, interventional and randomized open label pilot trial in patients with severe COVID-19. Twenty COVID-19 patients received two 200 ml transfusions of convalescent patient CP over 24-h compared with 20 who received standard of care. The primary outcome was the requirement for ventilation (non-invasive or mechanical ventilation). The secondary outcomes were biochemical parameters and mortality at 28 days. The CP group were a higher risk group with higher ferritin levels (<jats:italic>p</jats:italic> < 0.05) though respiratory indices did not differ. The primary outcome measure was required in 6 controls and 4 patients on CP (risk ratio 0.67, 95% CI 0.22–2.0, <jats:italic>p</jats:italic> = 0.72); mean time on ventilation (NIV or MV) did not differ. There were no differences in secondary measures at the end of the study. Two patients died in the control and one patient in the CP arm. There were no significant differences in the primary or secondary outcome measures between CP and standard therapy, although a larger definitive study is needed for confirmation. However, the study did show that CP therapy appears to be safe in hospitalized COVID-19 patients with hypoxia.</jats:p><jats:p><jats:italic>Clinical trials registration</jats:italic> NCT04356534: 22/04/2020.</jats:p>",
"alternative-id": [
"89444"
],
"article-number": "9927",
"assertion": [
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "Received",
"name": "received",
"order": 1,
"value": "11 December 2020"
},
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "Accepted",
"name": "accepted",
"order": 2,
"value": "23 April 2021"
},
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "First Online",
"name": "first_online",
"order": 3,
"value": "11 May 2021"
},
{
"group": {
"label": "Competing interests",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 1,
"value": "The authors declare no competing interests."
}
],
"author": [
{
"affiliation": [],
"family": "AlQahtani",
"given": "Manaf",
"sequence": "first"
},
{
"affiliation": [],
"family": "Abdulrahman",
"given": "Abdulkarim",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Almadani",
"given": "Abdulrahman",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Alali",
"given": "Salman Yousif",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Al Zamrooni",
"given": "Alaa Mahmood",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Hejab",
"given": "Amal Hamza",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Conroy",
"given": "Ronán M.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Wasif",
"given": "Pearl",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Otoom",
"given": "Sameer",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Atkin",
"given": "Stephen L.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Abduljalil",
"given": "Manal",
"sequence": "additional"
}
],
"container-title": "Scientific Reports",
"container-title-short": "Sci Rep",
"content-domain": {
"crossmark-restriction": false,
"domain": [
"link.springer.com"
]
},
"created": {
"date-parts": [
[
2021,
5,
11
]
],
"date-time": "2021-05-11T10:09:20Z",
"timestamp": 1620727760000
},
"deposited": {
"date-parts": [
[
2022,
12,
3
]
],
"date-time": "2022-12-03T08:01:55Z",
"timestamp": 1670054515000
},
"indexed": {
"date-parts": [
[
2024,
3,
29
]
],
"date-time": "2024-03-29T06:39:12Z",
"timestamp": 1711694352867
},
"is-referenced-by-count": 71,
"issue": "1",
"issued": {
"date-parts": [
[
2021,
5,
11
]
]
},
"journal-issue": {
"issue": "1",
"published-online": {
"date-parts": [
[
2021,
12
]
]
}
},
"language": "en",
"license": [
{
"URL": "https://creativecommons.org/licenses/by/4.0",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
5,
11
]
],
"date-time": "2021-05-11T00:00:00Z",
"timestamp": 1620691200000
}
},
{
"URL": "https://creativecommons.org/licenses/by/4.0",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
5,
11
]
],
"date-time": "2021-05-11T00:00:00Z",
"timestamp": 1620691200000
}
}
],
"link": [
{
"URL": "https://www.nature.com/articles/s41598-021-89444-5.pdf",
"content-type": "application/pdf",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://www.nature.com/articles/s41598-021-89444-5",
"content-type": "text/html",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://www.nature.com/articles/s41598-021-89444-5.pdf",
"content-type": "application/pdf",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "297",
"original-title": [],
"prefix": "10.1038",
"published": {
"date-parts": [
[
2021,
5,
11
]
]
},
"published-online": {
"date-parts": [
[
2021,
5,
11
]
]
},
"publisher": "Springer Science and Business Media LLC",
"reference": [
{
"key": "89444_CR1",
"unstructured": "Coronavirus Update (Live): 46,432,714 Cases and 1,200,927 Deaths from COVID-19 Virus Pandemic - Worldometer. Worldometers.info. https://www.worldometers.info/coronavirus/."
},
{
"DOI": "10.1056/NEJMoa2007764",
"author": "JH Beigel",
"doi-asserted-by": "publisher",
"journal-title": "N. Engl. J. Med.",
"key": "89444_CR2",
"unstructured": "Beigel, J. H. et al. Remdesivir for the treatment of Covid-19—Preliminary report. N. Engl. J. Med. https://doi.org/10.1056/NEJMoa2007764 (2020).",
"year": "2020"
},
{
"DOI": "10.1002/jmv.26047",
"author": "KK Sahu",
"doi-asserted-by": "publisher",
"first-page": "2251",
"issue": "11",
"journal-title": "J. Med. Virol.",
"key": "89444_CR3",
"unstructured": "Sahu, K. K., Jindal, V., Siddiqui, A. D., Cerny, J. & Gerber, J. M. Convalescent plasma therapy: A passive therapy for an aggressive COVID-19. J. Med. Virol. 92(11), 2251–2253. https://doi.org/10.1002/jmv.26047 (2020).",
"volume": "92",
"year": "2020"
},
{
"DOI": "10.1111/j.1469-0691.2004.00956.x",
"author": "YO Soo",
"doi-asserted-by": "publisher",
"first-page": "676",
"issue": "7",
"journal-title": "Clin. Microbiol. Infect.",
"key": "89444_CR4",
"unstructured": "Soo, Y. O. et al. Retrospective comparison of convalescent plasma with continuing high-dose methylprednisolone treatment in SARS patients. Clin. Microbiol. Infect. 10(7), 676–678. https://doi.org/10.1111/j.1469-0691.2004.00956.x (2004).",
"volume": "10",
"year": "2004"
},
{
"DOI": "10.1016/j.jinf.2016.11.009",
"author": "F Sahr",
"doi-asserted-by": "publisher",
"first-page": "302",
"issue": "3",
"journal-title": "J. Infect.",
"key": "89444_CR5",
"unstructured": "Sahr, F. et al. Evaluation of convalescent whole blood for treating Ebola Virus Disease in Freetown, Sierra Leone. J. Infect. 74(3), 302–309. https://doi.org/10.1016/j.jinf.2016.11.009 (2017).",
"volume": "74",
"year": "2017"
},
{
"DOI": "10.1093/cid/ciq106",
"author": "IF Hung",
"doi-asserted-by": "publisher",
"first-page": "447",
"issue": "4",
"journal-title": "Clin. Infect. Dis.",
"key": "89444_CR6",
"unstructured": "Hung, I. F. et al. Convalescent plasma treatment reduced mortality in patients with severe pandemic influenza A (H1N1) 2009 virus infection. Clin. Infect. Dis. 52(4), 447–456. https://doi.org/10.1093/cid/ciq106 (2011).",
"volume": "52",
"year": "2011"
},
{
"DOI": "10.1001/jama.2020.4783",
"author": "C Shen",
"doi-asserted-by": "publisher",
"journal-title": "JAMA",
"key": "89444_CR7",
"unstructured": "Shen, C. et al. Treatment of 5 critically ill patients with COVID-19 with convalescent plasma. JAMA https://doi.org/10.1001/jama.2020.4783 (2020).",
"year": "2020"
},
{
"DOI": "10.1001/jama.2020.10044",
"author": "L Li",
"doi-asserted-by": "publisher",
"first-page": "1",
"issue": "5",
"journal-title": "JAMA",
"key": "89444_CR8",
"unstructured": "Li, L. et al. Effect of convalescent plasma therapy on time to clinical improvement in patients with severe and life-threatening COVID-19: A randomized clinical trial. JAMA 324(5), 1–11. https://doi.org/10.1001/jama.2020.10044 (2020).",
"volume": "324",
"year": "2020"
},
{
"key": "89444_CR9",
"unstructured": "Gharbharan, A., et al. Convalescent plasma for COVID-19. A randomized clinical trial. medRxiv. 2020."
},
{
"DOI": "10.1101/2020.08.12.20169359",
"author": "MJ Joyner",
"doi-asserted-by": "publisher",
"journal-title": "medRxiv",
"key": "89444_CR10",
"unstructured": "Joyner, M. J. et al. Effect of convalescent plasma on mortality among hospitalized patients with COVID-19: Initial three-month experience. medRxiv https://doi.org/10.1101/2020.08.12.20169359 (2020).",
"year": "2020"
},
{
"DOI": "10.2807/1560-7917.Es.2020.25.3.2000045",
"author": "VM Corman",
"doi-asserted-by": "publisher",
"journal-title": "Eurosurveillance",
"key": "89444_CR11",
"unstructured": "Corman, V. M. et al. Detection of 2019 novel coronavirus (2019-nCoV) by real-time RT-PCR. Eurosurveillance https://doi.org/10.2807/1560-7917.Es.2020.25.3.2000045 (2020).",
"year": "2020"
},
{
"DOI": "10.1002/sim.4780132309",
"author": "MA Birkett",
"doi-asserted-by": "publisher",
"first-page": "2455",
"issue": "23–24",
"journal-title": "Stat. Med.",
"key": "89444_CR12",
"unstructured": "Birkett, M. A. & Day, S. J. Internal pilot studies for estimating sample size. Stat. Med. 13(23–24), 2455–2463 (1994).",
"volume": "13",
"year": "1994"
},
{
"author": "R Newson",
"first-page": "497",
"issue": "4",
"journal-title": "Stand. Genom. Sci.",
"key": "89444_CR13",
"unstructured": "Newson, R. Confidence intervals for rank statistics: Percentile slopes, differences, and ratios. Stand. Genom. Sci. 6(4), 497–520 (2006).",
"volume": "6",
"year": "2006"
},
{
"DOI": "10.15585/mmwr.mm6913e2",
"doi-asserted-by": "publisher",
"key": "89444_CR14",
"unstructured": "Preliminary estimates of the prevalence of selected underlying health conditions among patients with coronavirus disease 2019—United States, February 12-March 28, 2020. MMWR. 2020;69(13):382–386. https://doi.org/10.15585/mmwr.mm6913e2."
},
{
"DOI": "10.1016/j.ajem.2020.12.076",
"author": "TI Hariyanto",
"doi-asserted-by": "publisher",
"first-page": "110",
"journal-title": "Am. J. Emerg. Med.",
"key": "89444_CR15",
"unstructured": "Hariyanto, T. I. et al. Inflammatory and hematologic markers as predictors of severe outcomes in COVID-19 infection: A systematic review and meta-analysis. Am. J. Emerg. Med. 41, 110–119. https://doi.org/10.1016/j.ajem.2020.12.076 (2020).",
"volume": "41",
"year": "2020"
},
{
"DOI": "10.1002/jcla.23618",
"author": "L Cheng",
"doi-asserted-by": "publisher",
"first-page": "e23618",
"issue": "10",
"journal-title": "J. Clin. Lab. Anal.",
"key": "89444_CR16",
"unstructured": "Cheng, L. et al. Ferritin in the coronavirus disease 2019 (COVID-19): A systematic review and meta-analysis. J. Clin. Lab. Anal. 34(10), e23618. https://doi.org/10.1002/jcla.23618 (2020).",
"volume": "34",
"year": "2020"
},
{
"DOI": "10.1038/d41586-020-01824-5",
"author": "H Ledford",
"doi-asserted-by": "publisher",
"first-page": "469",
"issue": "7813",
"journal-title": "Nature",
"key": "89444_CR17",
"unstructured": "Ledford, H. Coronavirus breakthrough: Dexamethasone is first drug shown to save lives. Nature 582(7813), 469. https://doi.org/10.1038/d41586-020-01824-5 (2020).",
"volume": "582",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa2001282",
"author": "B Cao",
"doi-asserted-by": "publisher",
"first-page": "1787",
"issue": "19",
"journal-title": "N. Engl. J. Med.",
"key": "89444_CR18",
"unstructured": "Cao, B. et al. A trial of lopinavir-ritonavir in adults hospitalized with severe Covid-19. N. Engl. J. Med. 382(19), 1787–1799. https://doi.org/10.1056/NEJMoa2001282 (2020).",
"volume": "382",
"year": "2020"
},
{
"DOI": "10.1016/s0140-6736(20)31042-4",
"author": "IF Hung",
"doi-asserted-by": "publisher",
"first-page": "1695",
"issue": "10238",
"journal-title": "Lancet",
"key": "89444_CR19",
"unstructured": "Hung, I. F. et al. Triple combination of interferon beta-1b, lopinavir-ritonavir, and ribavirin in the treatment of patients admitted to hospital with COVID-19: An open-label, randomised, phase 2 trial. Lancet 395(10238), 1695–1704. https://doi.org/10.1016/s0140-6736(20)31042-4 (2020).",
"volume": "395",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa2019014",
"author": "AB Cavalcanti",
"doi-asserted-by": "publisher",
"journal-title": "N. Engl. J. Med.",
"key": "89444_CR20",
"unstructured": "Cavalcanti, A. B. et al. Hydroxychloroquine with or without azithromycin in mild-to-moderate Covid-19. N. Engl. J. Med. https://doi.org/10.1056/NEJMoa2019014 (2020).",
"year": "2020"
},
{
"DOI": "10.1172/jci140200",
"author": "MJ Joyner",
"doi-asserted-by": "publisher",
"journal-title": "J. Clin. Investig.",
"key": "89444_CR21",
"unstructured": "Joyner, M. J. et al. Early safety indicators of COVID-19 convalescent plasma in 5000 patients. J. Clin. Investig. https://doi.org/10.1172/jci140200 (2020).",
"year": "2020"
},
{
"DOI": "10.1016/j.mayocp.2020.06.028",
"author": "MJ Joyner",
"doi-asserted-by": "publisher",
"first-page": "1888",
"issue": "9",
"journal-title": "Mayo Clin.",
"key": "89444_CR22",
"unstructured": "Joyner, M. J. et al. Safety update: COVID-19 convalescent plasma in 20,000 hospitalized patients. Mayo Clin. 95(9), 1888–1897. https://doi.org/10.1016/j.mayocp.2020.06.028 (2020).",
"volume": "95",
"year": "2020"
},
{
"DOI": "10.1186/s13054-019-2683-3",
"author": "F Sanfilippo",
"doi-asserted-by": "publisher",
"first-page": "1",
"journal-title": "Crit. Care",
"key": "89444_CR23",
"unstructured": "Sanfilippo, F., La Rosa, V., Oliveri, F. & Astuto, M. Convalescent plasma for COVID-19: The risk of pulmonary embolism should not be underestimated!. Crit. Care 24, 1–2 (2020).",
"volume": "24",
"year": "2020"
},
{
"DOI": "10.1136/bmj.m4072",
"author": "EB Pathak",
"doi-asserted-by": "publisher",
"first-page": "m4072",
"journal-title": "BMJ",
"key": "89444_CR24",
"unstructured": "Pathak, E. B. Convalescent plasma is ineffective for covid-19. BMJ 371, m4072. https://doi.org/10.1136/bmj.m4072 (2020).",
"volume": "371",
"year": "2020"
},
{
"key": "89444_CR25",
"unstructured": "Recommendations for Investigational COVID-19 Convalescent Plasma. Food and Drug Administration. Accessed September 2, 2020, https://www.fda.gov/vaccines-blood-biologics/investigational-new-drug-ind-or-device-exemption-ide-process-cber/recommendations-investigational-covid-19-convalescent-plasma."
},
{
"DOI": "10.1136/bmj.m3939",
"author": "A Agarwal",
"doi-asserted-by": "publisher",
"first-page": "m3939",
"journal-title": "BMJ",
"key": "89444_CR26",
"unstructured": "Agarwal, A. et al. Convalescent plasma in the management of moderate covid-19 in adults in India: Open label phase II multicentre randomised controlled trial (PLACID Trial). BMJ 371, m3939. https://doi.org/10.1136/bmj.m3939 (2020).",
"volume": "371",
"year": "2020"
},
{
"DOI": "10.1101/2020.07.01.20139857",
"author": "A Gharbharan",
"doi-asserted-by": "publisher",
"journal-title": "medRxiv",
"key": "89444_CR27",
"unstructured": "Gharbharan, A. et al. Convalescent plasma for COVID-19. A randomized clinical trial. medRxiv https://doi.org/10.1101/2020.07.01.20139857 (2020).",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa2031304",
"author": "VA Simonovich",
"doi-asserted-by": "publisher",
"first-page": "619",
"issue": "7",
"journal-title": "N. Engl. J. Med.",
"key": "89444_CR28",
"unstructured": "Simonovich, V. A. et al. A randomized trial of convalescent plasma in Covid-19 severe pneumonia. N. Engl. J. Med. 384(7), 619–629. https://doi.org/10.1056/NEJMoa2031304 (2021).",
"volume": "384",
"year": "2021"
}
],
"reference-count": 28,
"references-count": 28,
"relation": {
"has-preprint": [
{
"asserted-by": "object",
"id": "10.1101/2020.11.02.20224303",
"id-type": "doi"
}
]
},
"resource": {
"primary": {
"URL": "https://www.nature.com/articles/s41598-021-89444-5"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"Multidisciplinary"
],
"subtitle": [],
"title": "Randomized controlled trial of convalescent plasma therapy against standard therapy in patients with severe COVID-19 disease",
"type": "journal-article",
"update-policy": "http://dx.doi.org/10.1007/springer_crossmark_policy",
"volume": "11"
}