Jun 7 |
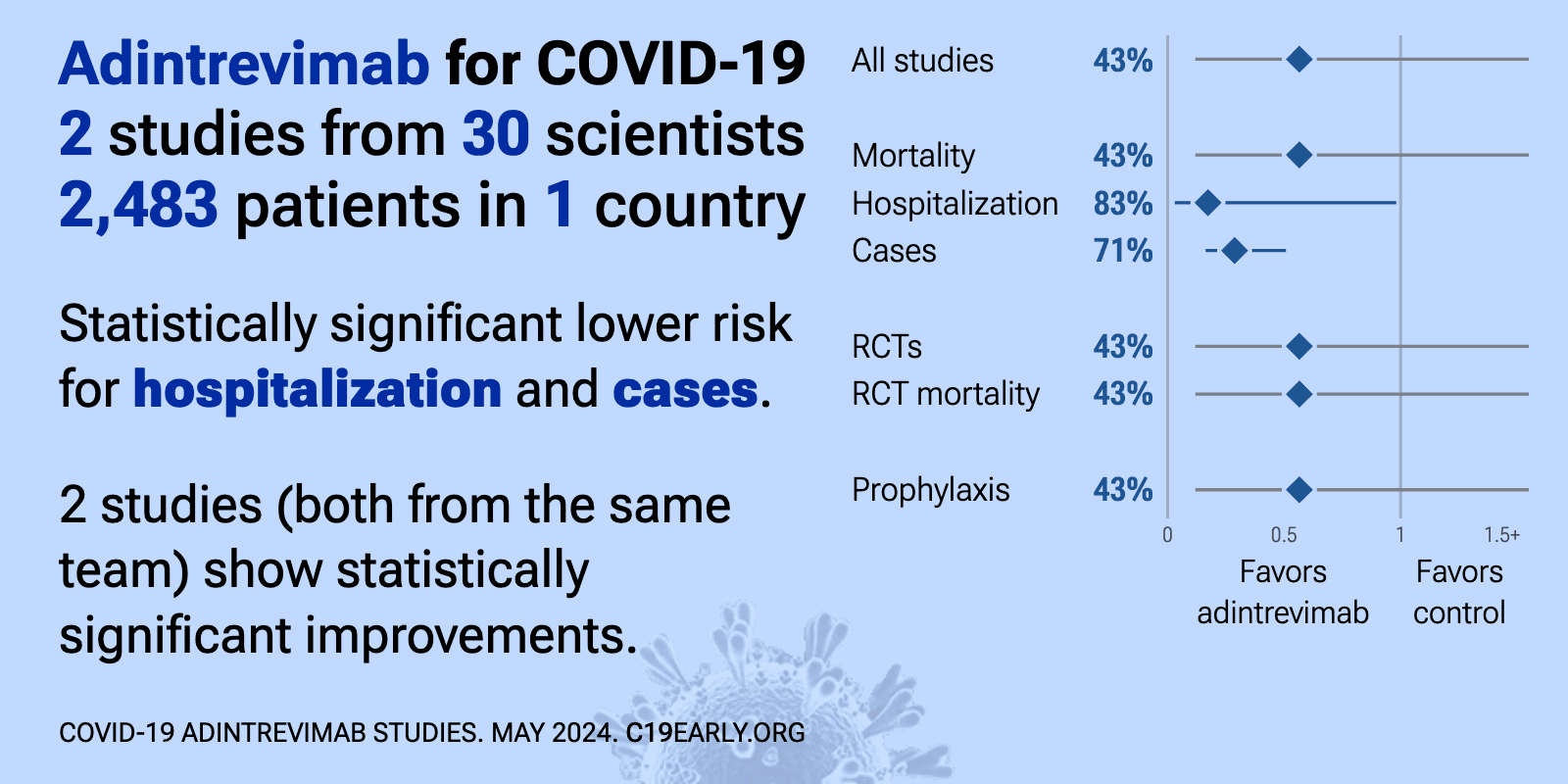
Adintrevimab for COVID-19: real-time meta-analysis of 2 studies | |
| Significantly lower risk is seen for hospitalization and cases. 2 studies (both from the same team/sponsor) show significant benefit. Meta-analysis using the most serious outcome reported shows 43% [-169‑88%] lower risk, withou.. | ||
Aug 11 2024 |
, D., Current Topics in Microbiology and Immunology, doi:10.1007/82_2024_268 | Monoclonal Antibody Therapies Against SARS-CoV-2: Promises and Realities |
| Review of monoclonal antibodies for SARS-CoV-2. Author notes that the omicron variant has reset achievements to date. | ||
Aug 8 2024 |
et al., Clinical Infectious Diseases, doi:10.1093/cid/ciae408 | Single monoclonal antibodies should not be used for COVID-19 therapy: a call for antiviral stewardship |
| Review arguing against use of single monoclonal antibodies for COVID-19 treatment, particularly in immunosuppressed patients, due to the risk of rapidly selecting for resistant viral variants. Authors suggest that while monoclonal antibod.. | ||
Jun 13 2023 |
et al., Open Forum Infectious Diseases, doi:10.1093/ofid/ofad314 | Prevention of COVID-19 Following a Single Intramuscular Administration of Adintrevimab: Results From a Phase 2/3 Randomized, Double-Blind, Placebo-Controlled Trial (EVADE) |
| 75% fewer symptomatic cases (p=0.03). PEP RCT 351 patients, showing lower COVID-19 cases with adintrevimab. The PrEP trial is listed separately [Ison]. | ||
Jun 13 2023 |
et al., Open Forum Infectious Diseases, doi:10.1093/ofid/ofad314 | Prevention of COVID-19 Following a Single Intramuscular Administration of Adintrevimab: Results From a Phase 2/3 Randomized, Double-Blind, Placebo-Controlled Trial (EVADE) |
| 84% lower hospitalization (p=0.07) and 70% fewer symptomatic cases (p<0.0001). PrEP RCT 1480 patients, showing lower COVID-19 cases with adintrevimab. The PEP trial is listed separately [Ison]. | ||
Jan 24 2023 |
et al., Antimicrobial Agents and Chemotherapy, doi:10.1128/aac.01353-22 | Prophylactic Administration of the Monoclonal Antibody Adintrevimab Protects against SARS-CoV-2 in Hamster and Non-Human Primate Models of COVID-19 |
| Hamster and monkey study showing dose-dependent protection against SARS-CoV-2 infection with prophylactic administration of a single dose of adintrevimab. | ||
References
Focosi et al., Analysis of SARS-CoV-2 mutations associated with resistance to therapeutic monoclonal antibodies that emerge after treatment, Drug Resistance Updates, doi:10.1016/j.drup.2023.100991.
Leducq et al., Spike protein genetic evolution in patients at high-risk of severe COVID-19 treated by monoclonal antibodies, The Journal of Infectious Diseases, doi:10.1093/infdis/jiad523.
Bruhn et al., Somatic hypermutation shapes the viral escape profile of SARS-CoV-2 neutralising antibodies, eBioMedicine, doi:10.1016/j.ebiom.2025.105770.
Ngiam et al., Early administration of neutralising monoclonal antibodies and post-acute sequelae of COVID-19, International Journal of Infectious Diseases, doi:10.1016/j.ijid.2026.108435.
Choudhary et al., Emergence of SARS-CoV-2 Resistance with Monoclonal Antibody Therapy, medRxiv, doi:10.1101/2021.09.03.21263105.
Günther et al., Variant-specific humoral immune response to SARS-CoV-2 escape mutants arising in clinically severe, prolonged infection, medRxiv, doi:10.1101/2024.01.06.24300890.
Casadevall et al., Single monoclonal antibodies should not be used for COVID-19 therapy: a call for antiviral stewardship, Clinical Infectious Diseases, doi:10.1093/cid/ciae408.