May 27 |
Curcumin reduces COVID-19 risk: real-time meta-analysis of 28 studies (Version 44) | |
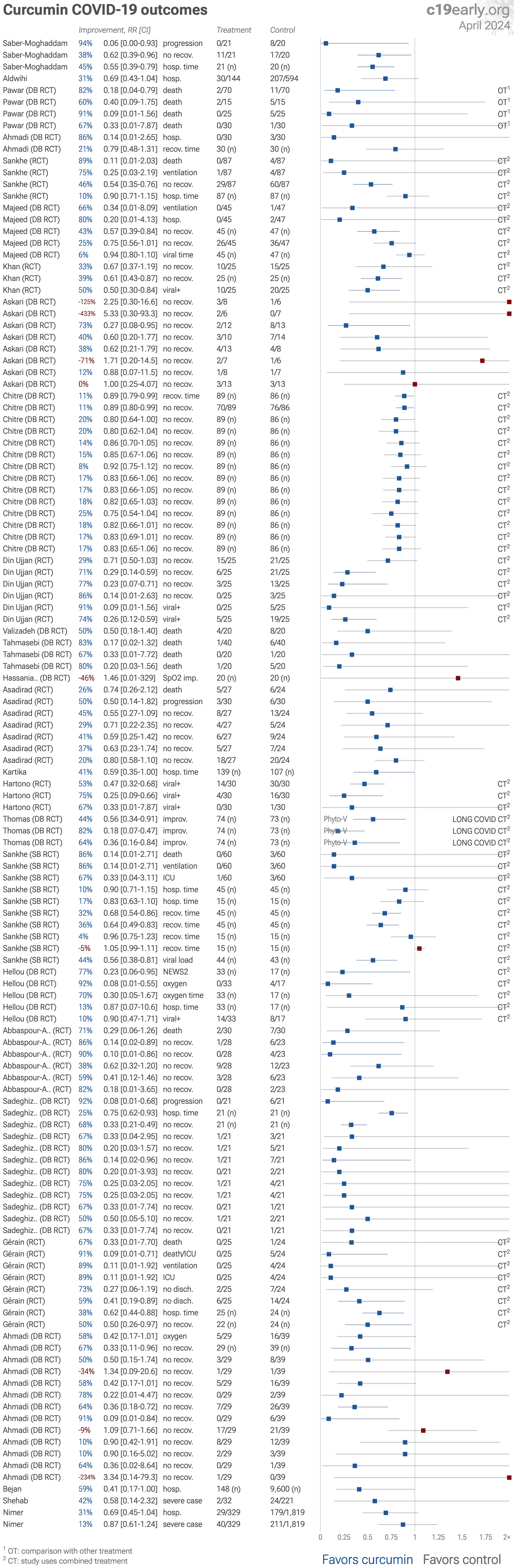
| Significantly lower risk is seen for mortality, ventilation, hospitalization, progression, recovery, and viral clearance. 19 studies from 17 independent teams in 9 countries show significant benefit. Meta-analysis using the most serious o.. | ||
Mar 23 |
et al., PLOS One, doi:10.1371/journal.pone.0345051 | Identification of polyphenols as novel neuropilin-1 cendR pocket inhibitors to block SARS-CoV-2 entry and enhance variant resistance |
| In silico study showing that polyphenols, particularly 6"-O-acetyldaidzin and phloretin, bind to the neuropilin-1 (NRP-1) b1 domain CendR pocket with higher affinity than the synthetic inhibitor EG01377, potentially blocking SARS-CoV.. | ||
Jan 22 |
et al., BMC Complementary Medicine and Therapies, doi:10.1186/s12906-026-05253-1 | Dose-dependent antiviral effects of glycyrrhizin, curcumin, and harmaline against clinical SARS-CoV-2 isolates, including D614G, Omicron BA.5, and Omicron XBB.1 |
| In vitro study in A549-AT cells showing that curcumin and glycyrrhizin effectively inhibit SARS-CoV-2 D614G, Omicron BA.5, and Omicron XBB.1 variants, while harmaline effectively inhibits only the Omicron variants, all at subtoxic concent.. | ||
Dec 17 2025 |
et al., BioMed Research International, doi:10.1155/bmri/9507417 | Targeting SIRT1: A Potential Strategy for Combating Severe COVID‐19 |
| Review showing that SIRT1 activation may benefit COVID-19 treatment through multiple anti-inflammatory and antiviral mechanisms. | ||
Dec 16 2025 |
et al., The Protein Journal, doi:10.1007/s10930-025-10309-1 | Halved but Potent: Exploring the Inhibitory Property of Curcumin Derivatives Against Evolving SARS-CoV-2 Strains |
| In silico study showing that curcumin, half-curcumin, catechin, and 8-hydroxydihydrosanguinarine (8-HDS) bind strongly to the spike (S) proteins of SARS-CoV-2 D614G and B.1.1.7 variants, potentially inhibiting viral entry via ACE2. | ||
Dec 3 2025 |
et al., BIO Web of Conferences, doi:10.1051/bioconf/202519804002 | Computational and Experimental Insights into the Antiviral Mechanism of Turmeric (Curcuma longa) against SARS-CoV-2 D614G |
| In silico and in vitro study showing that turmeric (Curcuma longa) extract inhibits SARS-CoV-2 D614G virus-like particle entry in Vero E6 cells. Authors identified 24 bioactive compounds from turmeric extract using LC-HRMS analysis, with.. | ||
Oct 31 2025 |
et al., Complementary Therapies in Medicine, doi:10.1016/j.ctim.2025.103295 | Effectiveness and Safety of Turmeric for the Treatment of COVID-19: An Updated Systematic Review and Meta-Analysis of Randomized Controlled Trials |
| 61% lower mortality (p=0.0006), 65% lower ventilation (p=0.004), 64% lower progression (p<0.0001), and 26% improved recovery (p=0.0001). Systematic review and meta-analysis of 23 RCTs showing significantly lower mortality, ventilation, and progression, and improved recovery with turmeric treatment. Nano-curcumin was most commonly used for improved bioavailability and showe.. | ||
Sep 16 2025 |
et al., Journal of Integrative and Complementary Medicine, doi:10.1177/27683605251379004 | Benefits of Nanocurcumin on Mortality in Patients with COVID-19: A Systematic Review and Meta-Analysis of Randomized Controlled Trials |
| 53% lower mortality (p=0.02). Systematic review and meta-analysis of six RCTs showing significantly lower COVID-19 mortality with nanocurcumin treatment. | ||
Aug 21 2025 |
et al., Scientific Reports, doi:10.1038/s41598-025-16092-4 | A Triple-blind randomized controlled trial on the effects of turmeric versus ginger on inflammatory biomarkers in patients with COVID-19 |
| RCT 144 COVID-19 outpatients showing a significant reduction in inflammatory markers (CRP and ESR) with both turmeric and ginger compared with placebo. There was no significant difference between groups for LDH or WBC. Baseline age differ.. | ||
Jul 16 2025 |
et al., Phytotherapy Research, doi:10.1002/ptr.70046 | Oral Administration of Water-Soluble Curcumin Complex Prevents ARDS With the Potential for COVID-19 Treatment |
| Mouse and in vitro study showing that orally administered water-soluble cyclodextrin-curcumin complex (CDC) protected C57BL/6 mice from Klebsiella or LPS-induced acute lung injury and down-regulated COVID-19 relevant pathways: treated ani.. | ||
Jun 4 2025 |
et al., Authorea Inc., doi:10.22541/au.174904567.74543434/v1 | Earlier clinical improvement in mild-moderate acute COVID patients treated with pharmacological-grade Curcumin |
| 37% improved recovery (p<0.0001). Prospective study of 131 mild-moderate COVID-19 patients showing significant reduction in time to symptom relief and complete recovery with pharmaceutical-grade curcumin. | ||
Jun 2 2025 |
et al., Phytochemistry Letters, doi:10.1016/j.phytol.2025.102999 | Utilizing natural compounds as ligands to disrupt the binding of SARS-CoV-2 receptor-binding domain to angiotensin-converting enzyme 2, impeding viral infection |
| In silico study showing that curcumin and arachidonic acid bind strongly to the SARS-CoV-2 receptor-binding domain (RBD), ACE2 receptor, and their complex interface, potentially disrupting viral infection. Curcumin demonstrated the highes.. | ||
May 26 2025 |
, H., Pharmaceuticals, doi:10.3390/ph18060798 | Structure-Based Design and In-Silico Evaluation of Computationally Proposed Curcumin Derivatives as Potential Inhibitors of the Coronaviral PLpro Enzymes |
| In silico study showing that novel curcumin derivatives strongly bind to papain-like protease (PLpro) enzymes from SARS-CoV, MERS-CoV, and SARS-CoV-2 with superior binding affinities compared to natural curcumin and reference antivirals. | ||
May 3 2025 |
et al., iScience, doi:10.1016/j.isci.2025.112575 | A pharmacokinetic study and critical reappraisal of curcumin formulations enhancing bioavailability |
| Crossover study of 9 healthy males comparing curcumin bioavailability from three commercial formulations (AOV, Longvida, NovaSOL). None of the three products tested contained the claimed amount of curcumin. Results showed plasma levels of.. | ||
Apr 30 2025 |
et al., Medical Forum Monthly, doi:10.60110/medforum.360407 | Assessment of the Anticoagulant Effect of Curcumin as Adjuvant Therapy to Enoxaparinin Covid-19 Iraqi Patients |
| Open-label RCT 58 COVID-19 patients in Iraq reporting lower D-dimer with curcumin. No clinical endpoints (mortality, ICU admission, mechanical ventilation, length of stay) were reported, despite the abstract’s claim of mortality reduction.. | ||
Apr 28 2025 |
et al., ChemistrySelect, doi:10.1002/slct.202406035 | Phytochemical Inhibitors of SARS‐CoV‐2 Entry: Targeting the ACE2‐RBD Interaction with l‐Tartaric Acid, l‐Ascorbic Acid, and Curcuma longa Extract |
| In vitro and in silico study showing that l-tartaric acid, l-ascorbic acid, and Curcuma longa extract (curcumin, demethoxycurcumin, bisdemethoxycurcumin) inhibit the SARS-CoV-2 spike RBD interaction with human ACE2. Authors demonstrate by.. | ||
Apr 16 2025 |
et al., Italian Journal of Pediatrics, doi:10.1186/s13052-025-01961-5 | Role of nutrient supplements in children with post-COVID condition: a retrospective preliminary observation and narrative review |
| Retrospective 1,243 children with COVID-19 showing lower risk of long COVID at 6 months when treated with a Multi-Element Product (MEP) containing antioxidants and anti-inflammatory compounds (magnesium 200 mg, quercetin 150 mg, curcumin .. | ||
Apr 4 2025 |
et al., Food Chemistry Advances, doi:10.1016/j.focha.2025.100969 | Novel dietary herbal preparations with inhibitory activities against multiple SARS-CoV-2 targets: A multidisciplinary investigation into antiviral activities |
| In vitro and in silico study showing potent inhibition of SARS-CoV-2 main protease (3CLpro) by novel polyherbal dietary preparations. Curcumin was used as a positive control and inhibited protease activity by >95% at 50 μM. | ||
Feb 20 2025 |
et al., Journal of Pharmacy and Pharmacology Research, doi:10.26502/fjppr.0105 | Exploring the Potential of Siddha Formulation MilagaiKudineer-Derived Phytotherapeutics Against SARS-CoV-2: An In-Silico Investigation for Antiviral Intervention |
| In silico computational docking study of phytochemicals from MilagaiKudineer, showing potential inhibitory activity against SARS-CoV-2 targets. Authors evaluated binding affinities for the spike glycoprotein receptor-binding domain, RNA-d.. | ||
Jan 16 2025 |
et al., Life, doi:10.3390/life15010113 | Immune-Boosting and Antiviral Effects of Antioxidants in COVID-19 Pneumonia: A Therapeutic Perspective |
| Review of immune-boosting and antiviral effects of antioxidants in COVID-19 pneumonia. Authors provide an overview of the literature on the use of antioxidants, including vitamins, trace elements, ozone, glutathione, L-carnitine, melatoni.. | ||
Dec 31 2024 |
et al., Trends in Food Science & Technology, doi:10.1016/j.tifs.2024.104864 | Bioactive compounds,quercetin, curcumin and β-glucan,regulate innate immunity via the gut-liver-brain axis |
| Review of how bioactive compounds quercetin, curcumin, and β-glucan regulate innate immunity through the gut-liver-brain axis, with focus on COVID-19. Authors describe how SARS-CoV-2 infection triggers excessive immune activation and cyto.. | ||
Nov 27 2024 |
et al., Bioresources and Bioprocessing, doi:10.1186/s40643-024-00822-z | Assessing multi-target antiviral and antioxidant activities of natural compounds against SARS-CoV-2: an integrated in vitro and in silico study |
| In vitro and in silico study showing that propolis, curcumin, quercetin, and ginseng compounds inhibit SARS-CoV-2 infection and bind to key viral proteins. In Vero CCL-81 cells, propolis and curcumin significantly reduced SARS-CoV-2 viral.. | ||
Nov 21 2024 |
et al., Discover Molecules, doi:10.1007/s44345-024-00005-5 | Exploring potential therapeutic candidates against COVID-19: a molecular docking study |
| In silico study showing potential inhibition of SARS-CoV-2 proteins by various compounds including dactinomycin, itraconazole, ivermectin, vitamin D, quercetin, curcumin, montelukast, bromhexine, hesperidin, EGCG and raloxifene. Authors p.. | ||
Sep 16 2024 |
et al., AMB Express, doi:10.1186/s13568-024-01739-8 | Establishment of in-house assay for screening of anti-SARS-CoV-2 protein inhibitors |
| In vitro study showing that curcumin, quercetin, gallic acid, and silymarin inhibit SARS-CoV-2 spike protein binding to the ACE2 receptor. Authors developed a novel immunofluorescent assay to screen potential inhibitors of the spike-ACE2 .. | ||
Aug 11 2024 |
et al., Cells, doi:10.3390/cells13161331 | Spike Protein of SARS-CoV-2 Activates Cardiac Fibrogenesis through NLRP3 Inflammasomes and NF-κB Signaling |
| In vitro study showing that the SARS-CoV-2 spike protein can activate cardiac fibroblasts through ACE2-dependent mechanisms, leading to cardiac fibrosis via the NLRP3 inflammasome and NF-κB signaling pathways. The results suggest that COV.. | ||
Jul 1 2024 |
et al., Animals and Zoonoses, doi:10.1016/j.azn.2024.06.001 | Thermal shift assay (TSA)-based drug screening strategy for rapid discovery of inhibitors against the Nsp13 helicase of SARS-CoV-2 |
| In vitro study showing that 11 compounds inhibited the SARS-CoV-2 helicase Nsp13, with 7 compounds inhibiting ATPase activity and 4 inhibiting RNA unwinding activity at micromolar concentrations. Authors screened 1,970 FDA-approved drugs .. | ||
Jun 24 2024 |
et al., Journal of Health, Population and Nutrition, doi:10.1186/s41043-024-00584-6 | Efficacy of highly bioavailable oral curcumin in asymptomatic or mild COVID-19 patients: a double-blind, randomized, placebo-controlled trial |
| 47% lower progression (p=0.48). RCT 138 COVID-19 outpatients in Japan showing lower progression to fever and hypoxemia with curcuRouge, a highly bioavailable oral curcumin formulation, compared to placebo. The curcuRouge group also had a greater reduction in body temper.. | ||
Jun 3 2024 |
et al., Emergent Materials, doi:10.1007/s42247-024-00754-6 | Nanoparticulate curcumin spray imparts prophylactic and therapeutic properties against SARS-CoV-2 |
| In vitro study showing that a water-dispersible nano-spray formulation of curcumin inhibits SARS-CoV-2 infection in Vero E6 cells. Authors found that pre-treatment effectively inhibited SARS-CoV-2 infection, and subsequent exposure to the.. | ||
May 31 2024 |
et al., Phytomedicine, doi:10.1016/j.phymed.2024.155784 | Computational Discovery of Mitochondrial Dysfunction Biomarkers in Severe SARS-CoV-2 Infection: Facilitating Pytomedicine Screening |
| In silico and in vitro study showing potential benefits of curcumin for severe COVID-19 by protecting mitochondrial function and reducing dysregulated metabolism and immune response. Authors identified five mitochondrial dysfunction bioma.. | ||
May 31 2024 |
et al., Biotechnology of Medicinal Plants with Antiallergy Properties, doi:10.1007/978-981-97-1467-4_4 | Antiallergic Implications of Curcumin During COVID-19: Current Status and Perspectives |
| Review of the potential of curcumin for allergic and inflammatory reactions in COVID-19. Authors highlight that curcumin has numerous health benefits, including antioxidative, anti-inflammatory, antidiabetic, and antimicrobial activities... | ||
May 30 2024 |
et al., Baku State University Journal of Chemistry and Material Sciences, 1:2 | In Silico investigation of the effects of curcuminoids on the spike protein of the omicron variant of SARS-CoV-2 |
| In silico study showing potential inhibition of SARS-CoV-2 omicron variant spike protein by curcuminoids. Authors performed molecular docking studies to examine interactions between curcumin, demethoxycurcumin, and bis-demethoxycurcumin w.. | ||
May 29 2024 |
et al., Research Square, doi:10.21203/rs.3.rs-4329762/v1 | Therapeutic effect and potential mechanism of curcumin, an active ingredient in Tongnao Decoction, on COVID-19 combined with stroke: a network pharmacology study and GEO database mining |
| In silico study showing that curcumin, the active ingredient in Tongnao Decoction, may be beneficial for COVID-19 combined with stroke through the CCNA2, JAK2, MMP9, PPARG, PTGS2, and STAT3 target pathways, with MMP9 being the most import.. | ||
May 10 2024 |
et al., Scientific Reports, doi:10.1038/s41598-024-61662-7 | Antiviral, anti-inflammatory and antioxidant effects of curcumin and curcuminoids in SH-SY5Y cells infected by SARS-CoV-2 |
| In vitro study showing antiviral, anti-inflammatory, and antioxidant effects of curcumin and curcuminoids in SARS-CoV-2 infected SH-SY5Y human neuroblastoma cells. Authors found that the curcuminoid Me23 significantly decreased expression.. | ||
May 4 2024 |
et al., Global Journal of Medical, Pharmaceutical, and Biomedical Update, doi:10.25259/GJMPBU_78_2023 | Curcumin as a Promising Therapy for COVID-19: A Review |
| Review of curcumin for COVID-19. Authors discuss the molecular mechanisms of COVID-19 pathogenesis and the properties and mechanisms of action of curcumin. Curcumin exhibits antiviral activity against SARS-CoV-2 by interfering with viral .. | ||
Apr 16 2024 |
et al., ScienceOpen, doi:10.58647/DRUGARXIV.PR000010.v1 | Network-based multi-omics-disease-drug associations reveal drug repurposing candidates for COVID-19 disease phases |
| In silico study identifying potential drugs beneficial for COVID-19 by integrating transcriptomics, proteomics, metabolomics, lipidomics, and drug data. Authors explore interactions between drugs, molecular features, and disease severity... | ||
Apr 13 2024 |
et al., European Journal of Pharmaceutical Sciences, doi:10.1016/j.ejps.2024.106766 | Curcumin and quercetin co-encapsulated in nanoemulsions for nasal administration: a promising therapeutic and prophylactic treatment for viral respiratory infections |
| In vitro and ex vivo study showing that curcumin and quercetin co-encapsulated in nanoemulsions (NEs) for nasal administration inhibited murine β-coronavirus (MHV-3) infection. MHV-3 belongs to the same family as SARS-CoV-2. Authors found.. | ||
Apr 7 2024 |
et al., European Journal of Pharmaceutics and Biopharmaceutics, doi:10.1016/j.ejpb.2024.114279 | Throat spray formulated with virucidal Pharmaceutical excipients as an effective early prophylactic or treatment strategy against pharyngitis post-exposure to SARS CoV-2 |
| In silico, in vitro, and rat study showing potential prophylactic and therapeutic benefits of a curcumin-loaded self-nanoemulsifying drug delivery system throat spray against SARS-CoV-2 infection and pharyngitis. Molecular docking studies.. | ||
Mar 18 2024 |
et al., Pharmacological Reports, doi:10.1007/s43440-024-00585-6 | Effect of polyphenols against complications of COVID-19: current evidence and potential efficacy |
| Review of polyphenols for COVID-19. Authors note that polyphenols can inhibit key SARS-CoV-2 enzymes involved in viral replication and infection, and that many polyphenols have well-established safety profiles. Authors recommend additiona.. | ||
Feb 29 2024 |
et al., International Journal of Nanomedicine, doi:10.2147/ijn.s451206 | Natural Products-Based Inhaled Formulations for Treating Pulmonary Diseases |
| Review of nanoformulations for inhaled therapeutics for respiratory diseases including potential for COVID-19. Inhaled formulations deliver treatment directly to both the upper and lower respiratory tract, enabling higher local concentrat.. | ||
Feb 6 2024 |
et al., International Conference on Organic and Applied Chemistry, doi:10.1063/5.0197724 | Bioinformatics study of curcumin, demethoxycurcumin, bisdemethoxycurcumin and cyclocurcumin compounds in Curcuma longa as an antiviral agent via nucleocapsid on SARS-CoV-2 inhibition |
| In silico study showing binding of curcumin, demethoxycurcumin, bisdemethoxycurcumin and cyclocurcumin to the SARS-CoV-2 nucleocapsid protein. Authors find curcumin has the strongest predicted binding affinity. All compounds showed suitab.. | ||
Jan 30 2024 |
et al., Endocrine and Metabolic Science, doi:10.1016/j.endmts.2024.100163 | Exploring autophagy in treating SARS-CoV-2 spike protein-related pathology |
| Review of autophagy pathways for treating SARS-CoV-2 spike protein-related pathology. Authors note that the spike protein is implicated in long COVID and post-vaccination syndrome through mechanisms like inflammation, vascular damage, and.. | ||
Jan 29 2024 |
et al., Research Square, doi:10.21203/rs.3.rs-3888947/v1 | Unlocking the potential of phytochemicals in inhibiting SARS-CoV-2 M Pro protein - An in-silico and cell-based approach |
| In silico and in vitro study including quercetin and curcumin derivatives as potential SARS-CoV-2 main protease (Mpro) inhibitors. Molecular dynamics simulations and virtual screening identified quercetin and curcumin derivatives demethox.. | ||
Jan 23 2024 |
et al., Virology Journal, doi:10.1186/s12985-023-02282-x | A novel film spray containing curcumin inhibits SARS-CoV-2 and influenza virus infection and enhances mucosal immunity |
| In vitro study showing that a novel oro-nasal curcumin film spray inhibits SARS-CoV-2 and influenza virus infection of cells while enhancing mucosal innate immunity. The spray demonstrated potent dose-dependent antiviral activity against .. | ||
Jan 9 2024 |
et al., Biomedicine & Pharmacotherapy, doi:10.1016/j.biopha.2024.116131 | Immunoregulatory effects of nanocurcumin in inflammatory milieu: Focus on COVID-19 |
| Review of immunoregulatory effects of nanocurcumin in inflammatory conditions including COVID-19. Authors note that curcumin has shown therapeutic potential but has poor bioavailability, which nanoformulations can address. Studies suggest.. | ||
Jan 4 2024 |
et al., Journal of Biomolecular Structure and Dynamics, doi:10.1080/07391102.2023.2300060 | Structure-based drug discovery to identify SARS-CoV2 spike protein–ACE2 interaction inhibitors |
| In silico study showing that curcumin strongly inhibits the interaction between the SARS-CoV-2 spike protein receptor binding domain (RBD) and the human ACE2 receptor for the delta and omicron variants. Molecular docking and dynamics simu.. | ||
Dec 31 2023 |
et al., Curcumin and Neurodegenerative Diseases, doi:10.1007/978-981-99-7731-4_14 | Effect of Curcumin on the Process of Neuroinflammation Caused by COVID-19 |
| Review of the effect of curcumin on neuroinflammation caused by COVID-19. Authors note that curcumin has antioxidant, anti-inflammatory, antiviral, neuroprotective, and immune-modulating properties that may be helpful before and after SAR.. | ||
Dec 31 2023 |
et al., Pathogens, doi:10.3390/pathogens13010039 | The Role of the NRF2 Pathway in the Pathogenesis of Viral Respiratory Infections |
| Review discussing the role of the NRF2 antioxidant pathway in regulating viral replication and associated inflammation and cell damage pathways during respiratory viral infections. Authors summarize evidence showing that respiratory virus.. | ||
Dec 25 2023 |
et al., Pharmaceuticals, doi:10.3390/ph17010034 | Photodynamic Action of Curcumin and Methylene Blue against Bacteria and SARS-CoV-2—A Review |
| Review of the use of curcumin and methylene blue as photosensitizers in photodynamic therapy (PDT) against bacteria and SARS-CoV-2. Authors explains the principles behind PDT and the mechanisms of curcumin and methylene blue as photosensi.. | ||
Dec 20 2023 |
et al., Functional Foods in Health and Disease, doi:10.31989/ffhd.v13i12.1207 | Safety evaluation of high bioavailability curcumin in Healthy Japanese Adults: A Randomized, placebo-controlled, double-blind, parallel-group comparison study |
| Safety RCTs for highly bioavailable curcumin supplements CR-033P and TS-P1 in healthy Japanese adults, testing 150 mg/day curcumin equivalent of CR-033P or TS-P1 for 12 weeks, and 750 mg/day curcumin equivalent of TS-P1 for 4 weeks. Both .. | ||
Dec 15 2023 |
, A., MDPI AG, doi:10.20944/preprints202312.1178.v1 | Neglected Effective Early Therapies against COVID-19: Focus on Functional Foods and Related Active Substances. A Review |
| Review of a group of compounds with efficacy shown in RCTs, safety, favorable opportunity costs, biological plausibility, and accessibility without major conflicts of interest. The compounds include curcumin, nigella sativa (black seed), .. | ||
Nov 30 2023 |
et al., PLOS ONE, doi:10.1371/journal.pone.0295014 | In silico studies of anti-oxidative and hot temperament-based phytochemicals as natural inhibitors of SARS-CoV-2 Mpro |
| In silico study showing potential benefits of the phytochemicals curcumin, savinin, and betulinic acid as an inhibitor of the SARS-CoV-2 main protease (Mpro). Molecular docking analysis revealed these hot temperament, antioxidant compound.. | ||
Nov 21 2023 |
et al., Cureus, doi:10.7759/cureus.49204 | Clinical Approach to Post-acute Sequelae After COVID-19 Infection and Vaccination |
| Review of evidence that the SARS-CoV-2 spike protein can damage cardiovascular, hematological, neurological, respiratory, gastrointestinal, and immunological systems, and may be a primary cause of long COVID symptoms. Authors introduce a .. | ||
Nov 15 2023 |
et al., Journal of Ayurveda and Integrative Medicine, doi:10.1016/j.jaim.2023.100778 | Effectiveness of ayurvedic formulation, NAOQ19 along with standard care in the treatment of mild-moderate COVID-19 patients: A double blind, randomized, placebo-controlled, multicentric trial |
| 13% shorter hospitalization (p=0.05) and 12% faster viral clearance (p=0.02). RCT 150 mild-moderate COVID-19 patients showing faster viral clearance and shorter hospitalization with NAOQ19, a polyherbal formulation containing 13 antiviral/antinflammatory compounds. NAOQ19 ingredients include curcumin, andrographis,.. | ||
Nov 2 2023 |
et al., International Journal of Molecular Sciences, doi:10.3390/ijms242115894 | Exploring the Binding Effects of Natural Products and Antihypertensive Drugs on SARS-CoV-2: An In Silico Investigation of Main Protease and Spike Protein |
| In silico molecular docking and molecular dynamics analysis identifying curcumin, quercetin, rosmarinic acid, and salvianolic acid B as having favorable binding to Mpro and three distinct sites on the S protein. Molecular dynamics simulat.. | ||
Oct 16 2023 |
et al., International Journal of Nanomedicine, doi:10.2147/IJN.S423251 | Curcumin Transferosome-Loaded Thermosensitive Intranasal in situ Gel as Prospective Antiviral Therapy for SARS-Cov-2 |
| In silico, in vitro, and in vivo study showing antiviral efficacy of a curcumin nanoformulation against SARS-CoV-2. Molecular docking revealed potential binding and inhibition of curcumin with viral proteins involved in entry and replicat.. | ||
Sep 29 2023 |
et al., bioRxiv, doi:10.1101/2023.09.28.560070 | Curcumin and turmeric extract inhibited SARS-CoV-2 pseudovirus cell entry and Spike mediated cell fusion |
| In vitro study showing that curcumin inhibits SARS-CoV-2 pseudovirus cell entry and syncytia formation at 1-10μM in cells overexpressing ACE2 and TMPRSS2. Authors find that both curcumin and turmeric extract are potential inhibitors of SA.. | ||
Sep 21 2023 |
et al., The Seybold Report, doi:10.17605/OSF.IO/TKEXJ | Computational studies to analyze effect of curcumin inhibition on coronavirus D614G mutated spike protein |
| In silico study showing that curcumin binds strongly to both the SARS-CoV-2 spike protein and ACE2 receptor. Curcumin's binding energy was similar for the wildtype spike protein and a mutated D614G variant. | ||
Sep 19 2023 |
et al., Makara Journal of Science, doi:10.7454/mss.v27i3.1609 | In-silico Approach for Predicting the Inhibitory Effect of Home Remedies on Severe Acute Respiratory Syndrome Coronavirus-2 |
| In silico analysis showing that curcumin and quercetin may be beneficial for COVID-19 by binding to the main protease (Mpro), spike protein, and ACE2 receptor. Both compounds had suitable ADME properties and minimal predicted toxicity. | ||
Sep 16 2023 |
et al., Natural Product Communications, doi:10.1177/1934578X231188861 | In Vitro Anti-SARS-CoV-2 Activities of Curcumin and Selected Phenolic Compounds |
| In vitro study showing that of 9 phenolic compounds tested, only curcumin inhibited SARS-CoV-2 cytopathic effects in infected monkey kidney Vero E6 cells. Curcumin showed antiviral activity against wildtype, alpha, delta, and omicron vari.. | ||
Aug 30 2023 |
et al., Stresses, doi:10.3390/stresses3030043 | Paradigm of Well-Orchestrated Pharmacokinetic Properties of Curcuminoids Relative to Conventional Drugs for the Inactivation of SARS-CoV-2 Receptors: An In Silico Approach |
| In silico analysis of curcuminoids showing similar drug-like properties and strong binding to the main protease and spike receptor binding domain proteins. Curcuminoid binding was comparable or better than existing drugs favipiravir, remd.. | ||
Aug 25 2023 |
et al., Virology Journal, doi:10.1186/s12985-023-02160-6 | A study on the effect of natural products against the transmission of B.1.1.529 Omicron |
| Review of natural products for SARS-CoV-2 omicron including an in silico study showing quercetin, curcumin, ascorbic acid, nigellidine, and chloroquine among many compounds docked to the ACE2 metallopeptidase domain. Quercetin, curcumin, .. | ||
Aug 3 2023 |
et al., Frontiers in Nutrition, doi:10.3389/fnut.2023.1241016 | Association of dietary intake of polyphenols, lignans, and phytosterols with immune-stimulating microbiota and COVID-19 risk in a group of Polish men and women |
| Dietary analysis of 95 adults in Poland, showing lower risk of COVID-19 with higher intake of polyphenols, lignans, and phytosterols. Results were statistically significant for total phytosterols, secoisolariciresinol, β-sitosterol, matai.. | ||
Jul 28 2023 |
et al., International Journal of Clinical Practice, doi:10.1155/2023/5734675 | Efficacy of Nanocurcumin as an Add-On Treatment for Patients Hospitalized with COVID-19: A Double-Blind, Randomized Clinical Trial |
| 58% lower need for oxygen therapy (p=0.06) and 67% improved recovery (p=0.04). RCT 76 hospitalized patients, showing improved recovery with nanocurcumin. Authors note that pure curcumin is limited due to rapid metabolism, low bio-availability, weak aqueous solubility, and systemic deletion, and that the nanocurcumin.. | ||
Jun 30 2023 |
et al., Duta Pharma Journal, doi:10.47701/djp.v3i1.2904 | Analisis in silico interaksi senyawa kurkuminoid terhadap enzim main protease 6LU7 dari SARS-CoV-2 |
| In silico study of the interaction between curcumin and its derivatives with the main protease enzyme from SARS-CoV-2. The compounds tested were curcumin, bisdemethoxycurcumin, and demethoxycurcumin. All compounds met the requirements of .. | ||
Jun 22 2023 |
et al., Frontiers in Nutrition, doi:10.3389/fnut.2023.1137407 | NASAFYTOL® supplementation in adults hospitalized with COVID-19 infection: results from an exploratory open-label randomized controlled trial |
| 91% lower combined mortality/ICU admission (p=0.02), 89% lower ventilation (p=0.05), 89% lower ICU admission (p=0.05), and 73% higher hospital discharge (p=0.07). RCT 49 hospitalized COVID-19 patients, 25 treated with curcumin and quercetin, shower lower mortality/ICU admission and improved recovery with treatment. All patients received vitamin D. 336mg curcumin, 520mg quercetin, and 18μg vitamin D.. | ||
Apr 29 2023 |
et al., Phytotherapy Research, doi:10.1002/ptr.7844 | Promising clinical outcomes of nano‐curcumin treatment as an adjunct therapy in hospitalized COVID‐19 patients: A randomized, double‐blinded, placebo‐controlled trial |
| 92% lower progression (p=0.02), 25% shorter hospitalization (p=0.007), and 68% improved recovery (p<0.0001). RCT 42 hospitalized moderate/severe COVID-19 patients in Iran, showing lower progression and improved recovery with nano-curcumin. Nano-curcumin 70mg bid for 14 days. | ||
Apr 13 2023 |
et al., Research Square, doi:10.21203/rs.3.rs-2780614/v1 | Potential Mechanism of Curcumin and Resveratrol against SARS-CoV-2 |
| In vitro study showing that curcumin and resveratrol inhibit SARS-CoV-2 infection through multiple mechanisms. Curcumin and resveratrol inhibit SARS-CoV-2 pseudovirus cell entry in HEK293-ACE2 cells with IC50 values of 18.02 μM and 8.76 μ.. | ||
Apr 13 2023 |
et al., Indian Journal of Medical Research, doi:10.4103/ijmr.ijmr_1701_22 | Indian food habit & food ingredients may have a role in lowering the severity & high death rate from COVID-19 in Indians: findings from the first nutrigenomic analysis |
| Nutrigenomics study of blood transcriptomes from Indian and western COVID-19 patients, showing Indian dietary habits and food ingredients including higher zinc, iron, curcumin, fiber, catechins, and EGCG may be associated with reduced sev.. | ||
Apr 1 2023 |
et al., Scientific Reports, doi:10.1038/s41598-023-31764-9 | Channel activity of SARS-CoV-2 viroporin ORF3a inhibited by adamantanes and phenolic plant metabolites |
| In vitro study showing that adamantane derivatives and six out of ten tested polyphenols including curcumin and quercetin inhibited the SARS-CoV-2 viroporin ORF3a, which contributes to viral pathogenicity and cytotoxicity. Authors used ce.. | ||
Mar 28 2023 |
et al., Natural Product Research, doi:10.1080/14786419.2023.2194647 | Antiviral activity of curcumin and its analogs selected by an artificial intelligence-supported activity prediction system in SARS-CoV-2-infected VeroE6 cells |
| In vitro study of curcumin and its analogs with 6 of 8 compounds showing SARS-CoV-2 antiviral activity with EC50 values <30μM and binding inhibitory activity with IC20 values <30μM. | ||
Mar 13 2023 |
et al., ACS Omega, doi:10.1021/acsomega.2c07326 | Curcumin Formulations for Better Bioavailability: What We Learned from Clinical Trials Thus Far? |
| Review of clinical studies on curcumin formulations for improved bioavailability. Authors note that bioavailability has been improved by a factor of >50,000. | ||
Feb 21 2023 |
et al., AYU (An International Quarterly Journal of Research in Ayurveda), doi:10.4103/ayu.ayu_92_22 | Safety and efficacy of COROPROTECT kit as an add-on therapy in the management of mild-to-moderate COVID-19: A randomized, placebo-controlled trial |
| 86% improved recovery (p<0.0001) and 53% improved viral clearance (p<0.0001). RCT with 300 mild to moderate hospitalized COVID-19 patients, showing faster recovery, faster viral clearance, and a reduction in inflammatory markers with COROPROTECT, which includes curcumin, andrographis, and several additional treatme.. | ||
Feb 17 2023 |
et al., Phytotherapy Research, doi:10.1002/ptr.7778 | The effectiveness of nano‐curcumin on patients with COVID‐19: A systematic review of clinical trials |
| Meta analysis of 8 nano-curcumin COVID-19 studies, showing significantly lower mortality and shorter hospitalization with treatment. | ||
Jan 18 2023 |
et al., Archives of Clinical and Medical Case Reports, 7:1 | The Efficacy of a Plant Based Formulation in the Symptomatic Management of Mild COVID-19 Cases: A Double Blind, Randomized Controlled Trial |
| 98% improved recovery (p<0.0001) and 76% improved viral clearance (p<0.0001). RCT 50 patients in India testing NAOQ19 which includes curcumin along with many other ingredients, showing improved recovery and viral clearance. SOC included vitamin C and zinc. NAOQ19 is a plant based formulation with 19 ingredients fro.. | ||
Jan 18 2023 |
et al., Frontiers in Nutrition, doi:10.3389/fnut.2022.1023997 | The possible therapeutic role of curcumin and quercetin in the early-stage of COVID-19—Results from a pragmatic randomized clinical trial |
| 29% improved recovery (p=0.11) and 91% improved viral clearance (p=0.05). Small RCT with 50 outpatients, 25 treated with curcumin, quercetin, and vitamin D, showing improved recovery and viral clearance with treatment. 168mg curcumin, 260mg, 360IU vitamin D3 daily for 14 days. Unadjusted baseline differences: t.. | ||
Jan 14 2023 |
et al., Phytotherapy Research, doi:10.1002/ptr.7724 | Curcumin for the treatment of COVID-19 patients: A meta-analysis of randomized controlled trials |
| 62% lower mortality (p=0.003). Meta analysis of 13 curcumin RCTs showing lower mortality with treatment. Authors note that subgroup analysis suggested improved efficacy with early treatment and with combined treatment. | ||
Nov 23 2022 |
et al., Phytotherapy Research, doi:10.1002/ptr.7683 | Phase III randomized clinical trial of BV-4051, an Ayurvedic polyherbal formulation in moderate SARS-CoV-2 infections and its impact on inflammatory biomarkers |
| 11% faster recovery (p=0.04). RCT 208 moderate COVID-19 patients in India, 103 treated with a combination of turmeric, ashwagandha, boswellia, and ginger, showing improved recovery with treatment. The dose of curcumin is unknown and bioavailability may be poor. | ||
Sep 27 2022 |
et al., Viruses, doi:10.3390/v14102132 | Optimization of Anti-SARS-CoV-2 Treatments Based on Curcumin, Used Alone or Employed as a Photosensitizer |
| In vitro study showing that curcumin inhibits SARS-CoV-2 replication and can be used as a photosensitizer in photodynamic therapy. Authors found that 10 μM curcumin directly applied to the virus before cell inoculation inhibited SARS-CoV-.. | ||
Sep 17 2022 |
et al., European Journal of Pharmacology, doi:10.1016/j.ejphar.2022.175267 | Immunomodulatory role of Nanocurcumin in COVID-19 patients with dropped natural killer cells frequency and function |
| 71% lower mortality (p=0.15) and 86% improved recovery (p=0.04). RCT with 30 nanocurcumin and 30 control patients in Iran, showing lower mortality and improved recovery, without statistical significance, and improved NK cell function. 160mg nanocurcumin for 21 days. | ||
Jun 30 2022 |
et al., Frontiers in Virology, doi:10.3389/fviro.2022.923018 | Dimethoxycurcumin Acidifies Endolysosomes and Inhibits SARS-CoV-2 Entry |
| In vitro study showing that dimethoxycurcumin (DiMC), a methylated analog of curcumin, blocked the entry of SARS-CoV-2 pseudovirus into human lung Calu-3 cells and monkey kidney Vero E6 cells at 2-4μM concentrations. DiMC acidified endoly.. | ||
Jun 10 2022 |
et al., F1000Research, doi:10.12688/f1000research.121933.1 | Effect of natural products use prior to infection with COVID-19 on disease severity and hospitalization: A self-reported cross-sectional survey study |
| 31% lower hospitalization (p=0.08) and 13% lower severe cases (p=0.47). Survey 2,148 COVID-19 recovered patients in Jordan, showing lower hospitalization with turmeric prophylaxis, not reaching statistical significance. | ||
Jun 6 2022 |
et al., Trials, doi:10.1186/s13063-022-06375-w | The efficacy of curcumin-piperine co-supplementation on clinical symptoms, duration, severity, and inflammatory factors in COVID-19 outpatients: a randomized double-blind, placebo-controlled trial |
| 26% improved recovery (p=0.26). Small RCT 46 outpatients in Iran, 23 treated with curcimin-piperine, showing no significant difference in recovery. 1000mg curcumin and 10mg piperine/day for 14 days. | ||
May 19 2022 |
et al., Journal of Cellular and Molecular Medicine, doi:10.1111/jcmm.17337 | Effect of ArtemiC in patients with COVID-19: A Phase II prospective study |
| 77% improved recovery (p=0.04), 92% lower need for oxygen therapy (p=0.01), 13% shorter hospitalization (p=0.92), and 10% improved viral clearance (p=0.77). RCT 50 hospitalized patients in Israel, 33 treated with curcumin, vitamin C, artemisinin, and frankincense oral spray, showing improved recovery with treatment. | ||
May 1 2022 |
et al., Computers in Biology and Medicine, doi:10.1016/j.compbiomed.2022.105552 | Curcumin inhibits spike protein of new SARS-CoV-2 variant of concern (VOC) Omicron, an in silico study |
| In silico study showing showing significant inhibitory potential of curcumin for omicron. | ||
May 1 2022 |
et al., Frontiers in Pharmacology, doi:10.3389/fphar.2022.898062 | Oral Co-Supplementation of Curcumin, Quercetin, and Vitamin D3 as an Adjuvant Therapy for Mild to Moderate Symptoms of COVID-19—Results From a Pilot Open-Label, Randomized Controlled Trial |
| 33% improved recovery (p=0.15) and 50% improved viral clearance (p=0.009). RCT 50 COVID+ outpatients in Pakistan, 25 treated with curcumin, quercetin, and vitamin D, showing significantly faster viral clearance, significantly improved CRP, and faster resolution of acute symptoms (p=0.154). 168mg curcumin, 260mg .. | ||
Apr 28 2022 |
et al., Phytotherapy Research, doi:10.1002/ptr.7468 | The effect of curcumin on the risk of mortality in patients with COVID-19: A systematic review and meta-analysis of randomized trials |
| 77% lower mortality (p=0.002). Meta analysis of 3 curcumin RCTs showing lower mortality with treatment. Author notes the small sample sizes of the included trials. | ||
Apr 9 2022 |
et al., Phytotherapy Research, doi:10.1002/ptr.7463 | In vitro antiviral activity against SARS-CoV-2 of common herbal medicinal extracts and their bioactive compounds |
| In vitro study showing antiviral activity of curcumin longa, with protection against SARS-CoV-2-induced cytopathic effects at a concentration of 3.125μg/ml. | ||
Mar 25 2022 |
et al., Complementary Therapies in Medicine, doi:10.1016/j.ctim.2022.102824 | A Randomized, Controlled, Blinded, Parallel Group, Clinical Trial to study the role of Ayurcov (AyurCoro3), one day regimen as an adjuvant therapy for COVID-19 disease management, at dedicated Covid Hospital (DCH) in India |
| 86% lower mortality (p=0.24), 86% lower ventilation (p=0.24), 10% shorter hospitalization (p=0.4), and 32% faster recovery (p=0.001). RCT with 60 hospitalized patients treated with Ayurcov and 60 control patients in India, showing improved viral clearance and faster symptom resolution in the mild/moderate group, but no significant differences in the severe group. Ayurco.. | ||
Mar 22 2022 |
et al., COVID, doi:10.3390/covid2040031 | A Randomised, Double-Blind, Placebo-Controlled Trial Evaluating Concentrated Phytochemical-Rich Nutritional Capsule in Addition to a Probiotic Capsule on Clinical Outcomes among Individuals with COVID-19—The UK Phyto-V Study |
| 44% greater improvement (p=0.02). RCT 147 long COVID patients in the UK, 56 treated with a phytochemical-rich concentrated food capsule, showing improved recovery with treatment. Treatment included curcumin, bioflavonoids, chamomile, ellagic acid, and resveratrol. | ||
Feb 28 2022 |
et al., Tropical Journal of Pharmaceutical Research, doi:10.4314/tjpr.v21i2.13 | Immune-boosting effect of natural remedies and supplements on progress of, and recovery from COVID-19 infection |
| 42% lower severe cases (p=0.55). Retrospective survey-based analysis of 349 COVID-19 patients, showing a lower risk of severe cases with vitamin D, zinc, turmeric, and honey prophylaxis in unadjusted analysis, without statistical significance. REC/UG/2020/03. | ||
Feb 22 2022 |
et al., Pharmacognosy Journal, doi:10.5530/pj.2022.14.27 | The Effect of Curcumin and Virgin Coconut Oil Towards Cytokines Levels in COVID-19 Patients at Universitas Sebelas Maret Hospital, Surakarta, Indonesia |
| 53% improved viral clearance (p<0.0001). RCT with 30 patients treated with curcumin and virgin coconut oil (VCO), and 30 SOC patients in Indonesia, showing faster viral clearance with treatment. Treatment also reduced IL-1β, IL-2, IL-6, IL-18, and IFN-β levels. VCO improves the.. | ||
Feb 4 2022 |
et al., International Journal of Molecular Sciences, doi:10.3390/ijms23031771 | Molecular Docking and Molecular Dynamics Simulations Discover Curcumin Analogue as a Plausible Dual Inhibitor for SARS-CoV-2 |
| In silico molecular dynamics simulation study finding a curcumin analogue (curA) as a promising dual inhibitor for SARS-CoV-2. | ||
Jan 28 2022 |
et al., ICE on IMERI, 2021 | Curcumin as adjuvant Therapy in Mild - Moderate Covid 19 |
| 41% shorter hospitalization (p=0.05). Retrospective 246 hospitalized patients in Indonesia, 136 treated with curcumin, showing shorter hospitalization time with treatment. All patients received vitamin C, D, and zinc. | ||
Jan 21 2022 |
et al., European Journal of Microbiology and Immunology, doi:10.1556/1886.2021.00022 | Inhibitory effects of specific combination of natural compounds against SARS-CoV-2 and its Alpha, Beta, Gamma, Delta, Kappa, and Mu variants |
| In vitro study testing combinations of plant extracts and micronutrients with several variants of SARS-CoV-2. A combination of vitamin C, N-acetylcysteine, curcumin, quercetin, resveratrol, theaflavin, naringenin, baicalin, and broccoli e.. | ||
Jan 17 2022 |
et al., Phytotherapy Research, doi:10.1002/ptr.7375 | Antiinflammatory potential of nano-curcumin as an alternative therapeutic agent for the treatment of mild-to-moderate hospitalized COVID-19 patients in a placebo-controlled clinical trial |
| 26% lower mortality (p=0.74), 50% lower progression (p=0.47), and 45% improved recovery (p=0.09). RCT 60 hospitalized patients in Iran, 30 treated with nano-curcumin, showing significant improvements in inflammatory cytokines, and improvements in clinical outcomes without statistical significance. 240 mg/day nano-curcumin for 7 days. | ||
Jan 12 2022 |
et al., Phytotherapy Research, doi:10.1002/ptr.7374 | Effect of nanocurcumin supplementation on the severity of symptoms and length of hospital stay in patients with COVID-19: A randomized double-blind placebo-controlled trial |
| 29% shorter hospitalization (p=0.22) and 9% improved recovery. RCT 48 hospitalized patients in Iran, 24 treated with nanocurcumin, showing lower hospitalization time with treatment. The number of patients shown in Table 3 (31 and 27 for each arm) is inconsistent with the number reported as randomized.. | ||
Jan 7 2022 |
et al., Nutrients, doi:10.3390/nu14020256 | Effectiveness of Curcumin on Outcomes of Hospitalized COVID-19 Patients: A Systematic Review of Clinical Trials |
| Review of 6 COVID-19 curcumin studies showing that treatment resulted in significant improvement in symptoms, duration of hospitalization, and mortality, and a significant decrease in proinflammatory cytokines and increase in anti-inflamm.. | ||
Dec 27 2021 |
et al., Research Square, doi:10.21203/rs.3.rs-1165680/v1 | An integrative approach to clinical recovery for COVID-19 patients using an Ayurvedic formulation: A multicentric double-blind randomized control trial |
| 89% improved recovery (p=0.05) and 24% improved viral clearance (p=0.47). Small RCT with 39 patients treated with NOQ19 and 37 placebo patients, showing improved recovery, without statistical significance. NOQ19 has multiple ingredients including curcumin, andrographis, and antiandrogen glycyrrhiza glabra. | ||
Nov 16 2021 |
et al., Molecules, doi:10.3390/molecules26226900 | Curcumin Inhibits In Vitro SARS-CoV-2 Infection In Vero E6 Cells through Multiple Antiviral Mechanisms |
| In vitro study showing antiviral and anti-inflammatory effects of curcumin during SARS-CoV-2 infection. Inhibition was seen with Vero E6 cells pre-infection and post-infection, and with D614G and the delta variant. The anti-inflammatory e.. | ||
Nov 14 2021 |
et al., Food Chemistry, doi:10.1016/j.foodchem.2021.131594 | Inhibition of the SARS-CoV-2 3CLpro main protease by plant polyphenols |
| In silico and in vitro study of plant polyphenols identifying quercetin, curcumin, ellagic acid, epigallocatechin gallate, and resveratrol as SARS-CoV-2 3CLpro inhibitors with IC50 between 11.8µM and 23.4µM. Real-time binding was analyzed.. | ||
Oct 22 2021 |
et al., Computers in Biology and Medicine, doi:10.1016/j.compbiomed.2021.104965 | Potential of turmeric-derived compounds against RNA-dependent RNA polymerase of SARS-CoV-2: An in-silico approach |
| In silico study showing strong binding affinity of curcumin and diacetylcurcumin with SARS-CoV-2 RNA-dependent RNA polymerase. Comparison with remdesivir and favipiravir suggested greater potential of these compounds as an RdRp inhibitor. | ||
Oct 11 2021 |
et al., Evidence-Based Complementary and Alternative Medicine, doi:10.1155/2021/8447545 | A Randomized, Double-Blind, Placebo-Controlled Study to Assess the Efficacy and Safety of a Nutritional Supplement (ImmuActive) for COVID-19 Patients |
| 43% improved recovery (p=0.004) and 6% faster viral clearance (p=0.47). RCT 100 patients in India, 50 treated with ImmuActive (curcumin, andrographolides, resveratrol, zinc, selenium, and piperine), showing improved recovery with treatment. | ||
Sep 23 2021 |
et al., Viruses, doi:10.3390/v13101914 | Turmeric Root and Its Bioactive Ingredient Curcumin Effectively Neutralize SARS-CoV-2 In Vitro |
| In vitro study showing curcumin neutralizes SARS-CoV2 in vitro with low subtoxic concentrations. Authors note that the clinical use of curcumin is hindered by poor bioavailability, and recommend using methods to increase bioavailability s.. | ||
Sep 19 2021 |
et al., Phytotherapy Research, doi:10.1002/ptr.7294 | A triple-blind, placebo-controlled, randomized clinical trial to evaluate the effect of curcumin-containing nanomicelles on cellular immune responses subtypes and clinical outcome in COVID-19 patients |
| 46% worse recovery (p=0.9). Small RCT with 40 low risk patients in Iran, 20 treated with nano-curcumin, showing no significant difference in outcomes with treatment. Authors note that treatment can improve peripheral blood inflammatory indices and modulate immune re.. | ||
Sep 8 2021 |
et al., Cureus, doi:10.7759/cureus.17829 | Turmeric as a Possible Treatment for COVID-19-Induced Anosmia and Ageusia |
| Small case study of 2 patients, showing significant improvement in taste and smell shortly after one dose of a turmeric supplement. Authors note that the risk of one dose is low in healthy individuals not on medications metabolized by cyt.. | ||
Aug 10 2021 |
et al., Journal of Ayurveda and Integrated Medical Sciences, doi:10.21760/jaims.6.4.6 | A prospective, multi center, single blind, randomized controlled study evaluating “AyurCoro3” as an adjuvant in the treatment of mild to moderate COVID |
| 89% lower mortality (p=0.12), 75% lower ventilation (p=0.37), 46% improved recovery (p=0.002), and 10% shorter hospitalization (p=0.4). RCT 174 patients in India, 87 treated with AyurCoro-3 (turmeric, gomutra, potassium alum, khadisakhar, bos indicus milk, ghee), showing faster recovery with treatment. EC/NEW/INST/2019/245. | ||
Jul 9 2021 |
et al., Medicine, doi:10.1097/MD.0000000000026601 | The enhanced bioavailability of free curcumin and bioactive-metabolite tetrahydrocurcumin from a dispersible, oleoresin-based turmeric formulation |
| Bioavailability RCT comparing CURCUGEN, a 50% curcuminoids-concentrated turmeric extract, with curcuminoids 95% standardized extract (C-95), showing significant improvements in bioavailability. | ||
Jun 29 2021 |
et al., Foods, doi:10.3390/foods10071503 | Potential In Vitro Inhibition of Selected Plant Extracts against SARS-CoV-2 Chymotripsin-Like Protease (3CLPro) Activity |
| In vitro study of several plant extracts, showing strong inhibition of SARS-CoV-2 3CLpro activity by turmeric rhizomes. Commercial curcumin also inhibited 3CLpro activity, but did not fully account for the inhibitory effect of turmeric rh.. | ||
Jun 19 2021 |
et al., Food Science and Nutrition, doi:10.1002/fsn3.2226 | Oral nano-curcumin formulation efficacy in the management of mild to moderate outpatient COVID-19: A randomized triple-blind placebo-controlled clinical trial |
| 86% lower hospitalization (p=0.24) and 21% faster recovery (p=0.37). RCT 60 outpatients in Iran, 30 treated with nano-curcumin showing lower hospitalization and faster recovery with treatment. | ||
Jun 17 2021 |
et al., PLOS ONE, doi:10.1371/journal.pone.0253489 | Phenolic compounds disrupt spike-mediated receptor-binding and entry of SARS-CoV-2 pseudo-virions |
| In vitro study of 56 polyphenols showing that curcumin has high binding affinity to the RBD of the SARS-CoV-2 spike protein, inhibits ACE2 at non-toxic concentrations, and decreases activity of TMPRSS2. Promising results were also seen fo.. | ||
Jun 15 2021 |
et al., Pathogens, doi:10.3390/pathogens10060758 | Bioactive Polyphenolic Compounds Showing Strong Antiviral Activities against Severe Acute Respiratory Syndrome Coronavirus 2 |
| Vero E6 in vitro study showing curcumin, hesperidin, and quercetin significantly inhibited SARS-CoV-2 replication, and in silico analysis with promising Mpro and spike docking results. | ||
May 28 2021 |
et al., Frontiers in Pharmacology, doi:10.3389/fphar.2021.669362 | Oral Curcumin With Piperine as Adjuvant Therapy for the Treatment of COVID-19: A Randomized Clinical Trial |
| 82% lower mortality (p=0.02). RCT 140 patients, 70 treated with curcumin and piperine (for absorption), and 70 treated with probiotics, showing faster recovery, lower progression, and lower mortality with curcumin. | ||
May 11 2021 |
et al., International Journal of Environmental Research and Public Health, doi:10.3390/ijerph18105086 | Patients’ Behavior Regarding Dietary or Herbal Supplements before and during COVID-19 in Saudi Arabia |
| 31% lower hospitalization (p=0.1). Retrospective survey-based analysis of 738 COVID-19 patients in Saudi Arabia, showing lower hospitalization with vitamin C, turmeric, zinc, and nigella sativa, and higher hospitalization with vitamin D. For vitamin D, most patients contin.. | ||
May 7 2021 |
et al., Frontiers in Pharmacology, doi:10.3389/fphar.2021.675287 | Curcumin as a Potential Treatment for COVID-19 |
| Review of curcumin for COVID-19 including potential antiviral, anti-inflammatory, anticoagulant, antiplatelet, and cytoprotective effects. | ||
Mar 28 2021 |
et al., Life Sciences, doi:10.1016/j.lfs.2021.119437 | Nanocurcumin improves Treg cell responses in patients with mild and severe SARS-CoV2 |
| 83% lower mortality (p=0.11). RCT 40 hospitalized, 40 ICU, and 40 control patients in Iran, showing lower mortality and improved regulatory T cell responses with nanocurcumin treatment (SinaCurcumin). | ||
Feb 28 2021 |
et al., Clinical Pharmacology & Therapeutics, doi:10.1002/cpt.2376 (date from preprint) | DrugWAS: Drug‐wide Association Studies for COVID‐19 Drug Repurposing |
| 59% lower hospitalization (p=0.05). Retrospective 9,748 COVID-19 patients in the USA showing lower hospitalization with turmeric extract. | ||
Jan 3 2021 |
et al., Phytotherapy Research, doi:10.1002/ptr.7004 | Oral nano-curcumin formulation efficacy in management of mild to moderate hospitalized coronavirus disease-19 patients: An open label nonrandomized clinical trial |
| 94% lower progression (p=0.001), 38% improved recovery (p=0.04), and 45% shorter hospitalization (p=0.001). Small prospective nonrandomized trial with 41 patients, 21 treated with curcumin, showing lower disease progression and faster recovery with treatment. IRCT20200408046990N1. | ||
Nov 16 2020 |
et al., Journal of Clinical Trials, S3:004 | A Randomized, Comparative Clinical Study to Evaluate the Activity of CurvicTM Formulation for Management of SARS-COV-2 Infection (COVID-19) |
| 75% improved recovery (p=0.001) and 100% improved 7-point scale results (p=0.0001). RCT 200 COVID-19 positive patients in India, 100 treated with curcumin, vitamin C, vitamin K2-7, and selenomethionine, showing faster recovery with treatment. | ||
Nov 13 2020 |
et al., Metabolism Open, doi:10.1016/j.metop.2020.100066 | The combination of bromelain and curcumin as an immune-boosting nutraceutical in the prevention of severe COVID-19 |
| Review of the potential benefits of bromelain and curcumin for COVID-19, and potential synergistic effects of the combination. | ||
Oct 20 2020 |
et al., Int. Immunopharmacol., doi:10.1016/j.intimp.2020.107088 | Nano-curcumin therapy, a promising method in modulating inflammatory cytokines in COVID-19 patients |
| 50% lower mortality (p=0.3). Small RCT with 40 nano-curcumin patients and 40 control patients showing lower mortality with treatment. Authors conclude that nano-curcumin may be able to modulate the increased rate of inflammatory cytokines especially IL-1β and IL-6 mR.. | ||
Oct 16 2020 |
et al., Future Journal of Pharmaceutical Sciences, doi:10.1186/s43094-020-00126-x | Activity of phytochemical constituents of Curcuma longa (turmeric) and Andrographis paniculata against coronavirus (COVID-19): an in silico approach |
| In silico study of several phytochemical compounds from Curcuma longa (turmeric) and Andrographis paniculata for their potential activity against COVID-19 by targeting the SARS-CoV-2 main protease. Molecular docking analysis found the tur.. | ||
Jun 7 2020 |
et al., Preprints, doi:10.20944/preprints202005.0439.v3 | Comparative Docking Studies on Curcumin with COVID-19 Proteins |
| In silico study showing high binding affinity for the SARS-CoV-2 nucleocapsid and nsp10 proteins with curcumin. Authors propose curcumin for COVID-19 drug development. | ||
Jun 4 2020 |
, P., Cell Stress and Chaperones, doi:10.1007/s12192-020-01126-9 | COVID-19 and heme oxygenase: novel insight into the disease and potential therapies |
| Proposal that COVID-19 risk is related to low intracellular heme oxygenase (HO-1), and that therapies that raise HO-1 may be beneficial, which includes fluvoxamine, certain anesthetics (sevoflurane or isoflurane), hemin, estrogen, statins.. | ||
Apr 24 2020 |
et al., ChemRxiv, doi:10.26434/chemrxiv.12181404.v1 | In-Silico Identification of Potent Inhibitors of COVID-19 Main Protease (Mpro) and Angiotensin Converting Enzyme 2 (ACE2) from Natural Products: Quercetin, Hispidulin, and Cirsimaritin Exhibited Better Potential Inhibition than Hydroxy-Chloroquine Against COVID-19 Main Protease Active Site and ACE2 |
| In silico study of natural compounds identifying quercetin, curcumin, hispidulin, cirsimaritin, sulfasalazine, and artemisin as potential compounds that inhibit SARS-CoV-2. | ||