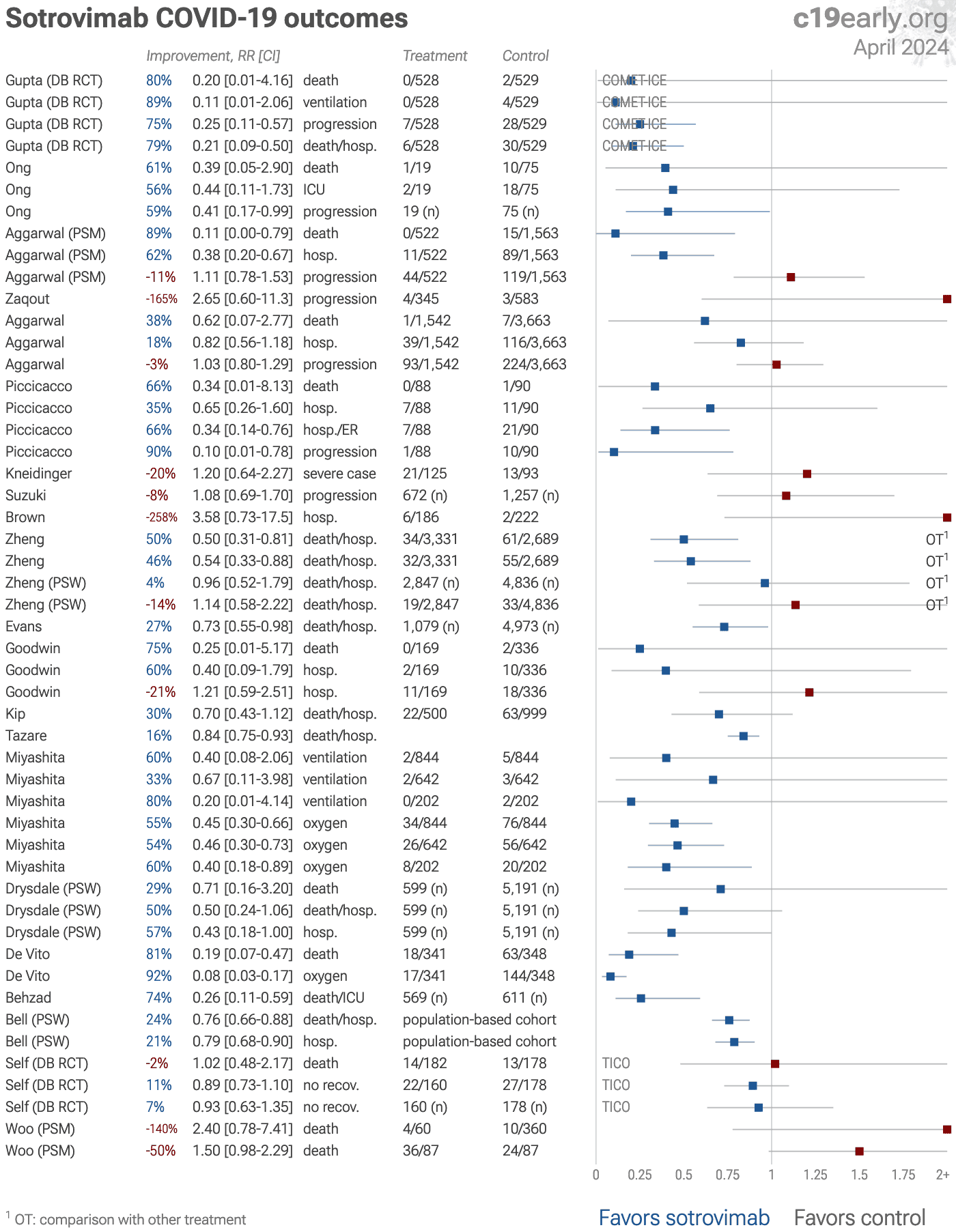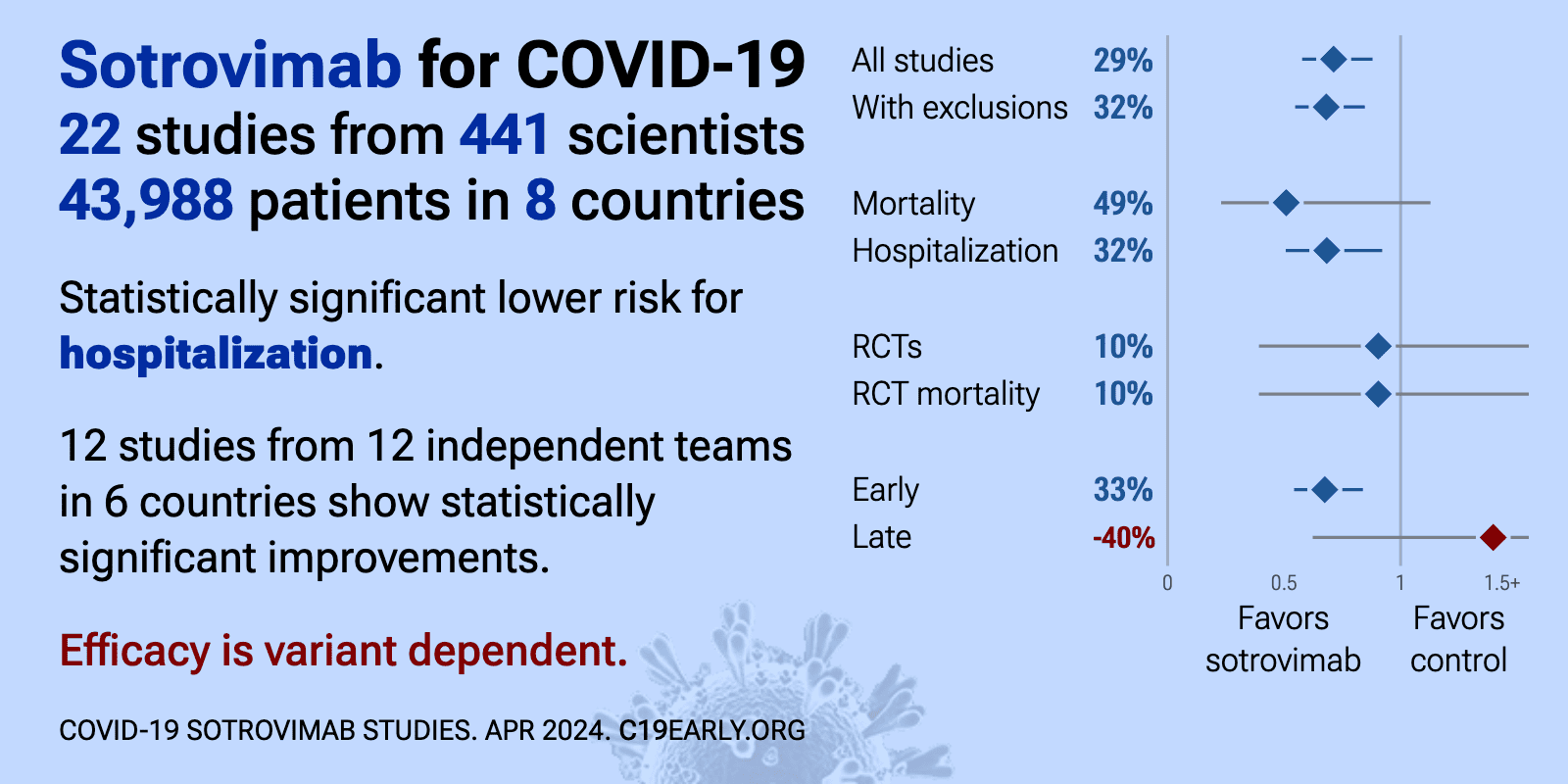
Efficacy is variant dependent. In Vitro studies suggest lower efficacy for omicron BA.11-3, BA.4, BA.54, XBB.1.9.3, XBB.1.5.24, XBB.2.9, CH.1.15, and no efficacy for BA.26, XBB, XBB.1.5, XBB.1.9.17, XBB.1.16, BQ.1.1.45, and CL.15. US EUA has been revoked. mAb use may create new variants that spread globally8-10, and may be associated with increased risk of autoimmune disease11, prolonged viral loads, clinical deterioration, and immune escape9,12-16 .
Sotrovimab was adopted
in 42 countries.
Recent:Ashoor Ngiam Martin-Blondel Chen-Xu.
Jun 7 |
Sotrovimab reduced COVID-19 risk: real-time meta-analysis of 29 studies (Version 49) | |
| Significantly lower risk is seen for mortality, ICU admission, and hospitalization. 17 studies from 15 independent teams in 6 countries show significant benefit. Meta-analysis using the most serious outcome reported shows 22% [10R.. | ||
Jan 31 |
et al., International Journal of Infectious Diseases, doi:10.1016/j.ijid.2026.108435 | Early administration of neutralising monoclonal antibodies and post-acute sequelae of COVID-19 |
| Retrospective 19,689 hospitalized COVID-19 patients in Singapore showing no significant reduction in overall post-acute sequelae of COVID-19 (PASC) with early monoclonal antibody treatment, but increased risk of autoimmune complications. .. | ||
Jan 11 |
et al., Open Forum Infectious Diseases, doi:10.1093/ofid/ofaf695.340 | Sotrovimab for Pre-exposure Prophylaxis against SARS-CoV2 in a Vulnerable Patient Population: Results from the PROTECT-V trial |
| 66% lower mortality (p=1), 66% lower hospitalization (p=0.62), and 20% fewer symptomatic cases (p=0.51). RCT 619 vulnerable patients showing no significant difference in symptomatic COVID-19 infections at 12 weeks with sotrovimab pre-exposure prophylaxis. | ||
Dec 31 2025 |
et al., iScience, doi:10.1016/j.isci.2025.114226 | Viral clearance and escape during therapy of COVID-19 outpatients: A prospective cohort study |
| Prospective cohort study of 114 high-risk COVID-19 outpatients showing that monoclonal antibody treatments with suboptimal neutralizing activity against infecting variants resulted in higher viral loads and increased emergence of escape m.. | ||
Oct 1 2025 |
et al., Infection and Drug Resistance, doi:10.2147/IDR.S540928 | The Emergence of Escape Mutations in COVID-19 Following Anti-Spike Monoclonal Antibody Treatment: How Do We Tackle It? |
| Review of treatment-emergent resistance to anti-spike monoclonal antibodies in COVID-19 patients. Monotherapies like bamlanivimab and sotrovimab showed very high resistance rates, while combination approaches had lower risk. The rapid evo.. | ||
Sep 2 2025 |
et al., medRxiv, doi:10.1101/2025.08.29.25334732 | Long-term follow-up of treatment comparisons in RECOVERY: a randomised, open-label, platform trial for patients hospitalised with COVID-19 |
| 6-month followup of RECOVERY patients. Results are reported within the respective trials for each treatment. | ||
May 19 2025 |
et al., Journal of Infection, doi:10.1016/j.jinf.2025.106510 | Monitoring the emergence of resistance with sotrovimab in immunocompromised patients with COVID-19: LUNAR study |
| Prospective study of 217 mostly immunocompromised COVID-19 outpatients in the UK showing viral clearance in 84% by day 28 after sotrovimab treatment, and treatment-emergent substitutions in the sotrovimab epitope in 30% of patients with p.. | ||
Mar 22 2025 |
et al., Infection, doi:10.1007/s15010-025-02505-z | Impact of treatment of COVID-19 with sotrovimab on post-acute sequelae of COVID-19 (PASC): an analysis of National COVID Cohort Collaborative (N3C) data |
| 4% lower long COVID (p=0.002). N3C retrospective 9,504 sotrovimab-treated high-risk COVID-19 patients versus 619,668 untreated high-risk controls showing reduced risk of post-acute sequelae of COVID-19 (PASC) with treatment. ATT weighting failed to adjust for "hea.. | ||
Mar 18 2025 |
et al., PNAS Nexus, doi:10.1093/pnasnexus/pgaf085 | Clinical and molecular landscape of prolonged SARS-CoV-2 infection with resistance to remdesivir in immunocompromised patients |
| Clinical and virological study of 3 immunocompromised B-cell lymphoma patients with prolonged SARS-CoV-2 infection showing development of remdesivir and sotrovimab resistance. Through serial viral genome sequencing, authors identified NSP.. | ||
Mar 13 2025 |
et al., Eurosurveillance, doi:10.2807/1560-7917.ES.2025.30.10.2400252 | Impact of SARS-CoV-2 variant mutations on susceptibility to monoclonal antibodies and antiviral drugs: a non-systematic review, April 2022 to October 2024 |
| Review of SARS-CoV-2 variant mutations and their impact on susceptibility to monoclonal antibodies and antivirals. Mutations in the ORF1ab region led to decreased susceptibility to paxlovid, while nsp12 mutations reduced efficacy for remd.. | ||
Feb 6 2025 |
et al., Clinical Microbiology and Infection, doi:10.1016/j.cmi.2025.02.002 | Long-term outcomes of passive immunotherapy for COVID-19: a pooled analysis of a large multinational platform randomized clinical trial |
| 34% higher mortality (p=0.39) and 12% lower combined mortality/hospitalization (p=0.5). Analysis of 2,311 hospitalized COVID-19 patients from four randomized controlled trials (ACTIV-3/TICO) showing no significant difference in long-term mortality with sotrovimab, amubarvimab/romlusevimab, tixagevimab-cilgavimab, or ensovibep. | ||
Jan 29 2025 |
et al., Open Forum Infectious Diseases, doi:10.1093/ofid/ofae631.2172 | Evaluating the use of Monoclonal Antibodies - Sotrovimab, Casirivimab/Imedvimab (REGEN-COV) and Tixagevimab/Cilgavimab (EVUSHELD) for COVID-19 Treatment in Singapore |
| PSM retrospective 366 hospitalized COVID-19 patients in Singapore showing no statistically significant reduction in severe outcomes with monoclonal antibodies (mAbs), except for lower oxygen use in patients treated with sotrovimab during .. | ||
Jan 27 2025 |
et al., Lancet Infectious Diseases, doi:10.1016/S1473-3099(25)00361-5 (date from preprint) | Sotrovimab versus usual care in patients admitted to hospital with COVID-19 (RECOVERY): a randomised, controlled, open-label, platform trial |
| 6% lower mortality (p=0.47), 4% lower hospital discharge (p=0.51), and 2% lower combined mortality/ICU admission (p=0.82). RCT 1,723 hospitalized COVID-19 patients showing lower 28-day mortality with sotrovimab in patients with high serum nucleocapsid antigen levels, but no significant benefit in the overall population. The trial used a higher dose of sotrovi.. | ||
Oct 10 2024 |
et al., JMIR Bioinformatics and Biotechnology, doi:10.2196/58018 | Comparison of the Neutralization Power of Sotrovimab Against SARS-CoV-2 Variants: Development of a Rapid Computational Method |
| In silico computational method developed to predict the neutralization power of monoclonal antibodies against new SARS-CoV-2 variants. The method evaluates binding affinity of antibodies based on molecular interactions and Gibbs free ener.. | ||
Oct 4 2024 |
et al., European Journal of Medical Research, doi:10.1186/s40001-024-02062-5 | Does early combination vs. Monotherapy improve clinical outcomes of clinically extremely vulnerable patients with COVID-19? Results from a retrospective propensity-weighted analysis |
| 72% lower mortality (p=0.15) and 77% lower progression (p=0.03). Retrospective 81 severely immunocompromised COVID-19 outpatients in Italy showing improved composite outcome of death, hospitalization, and emergency department encounters with early combination therapy of an antiviral plus sotrovimab com.. | ||
Sep 19 2024 |
et al., Antiviral Research, doi:10.1016/j.antiviral.2024.106006 | Identification of antibody-resistant SARS-CoV-2 mutants via N4-Hydroxycytidine mutagenesis |
| In vitro selection study identifying SARS-CoV-2 spike mutations that confer resistance to therapeutic antibodies. Using a Wuhan-like strain and omicron B.1.1.529, the authors used sequential mutagenesis with the molnupiravir active compou.. | ||
Aug 19 2024 |
, D., MDPI AG, doi:10.20944/preprints202408.1340.v1 | In Silico Evaluation of Ten Monoclonal Antibodies Neutralization Power of SARS-CoV-2 Variants EG.5, BA.2.86 and JN.1 |
| In silico study evaluating the neutralization power of 10 monoclonal antibodies (9 previously EUA-granted, 1 under clinical investigation) against SARS-CoV-2 variants EG.5, BA.2.86, and JN.1. Most available neutralizing antibodies have si.. | ||
Aug 12 2024 |
et al., Journal of Infection and Public Health, doi:10.1016/j.jiph.2024.102512 | Sotrovimab lost neutralization efficacy against SARS-CoV-2 subvariants but remained clinically effective: Were monoclonal antibodies against COVID-19 rejected too early? |
| Retrospective 14 outpatients treated with sotrovimab showing that while sotrovimab lost in vitro neutralization efficacy against omicron subvariants BA.1 and BA.2, it remained clinically effective in reducing viral load in patients who di.. | ||
Aug 11 2024 |
, D., Current Topics in Microbiology and Immunology, doi:10.1007/82_2024_268 | Monoclonal Antibody Therapies Against SARS-CoV-2: Promises and Realities |
| Review of monoclonal antibodies for SARS-CoV-2. Author notes that the omicron variant has reset achievements to date. | ||
Aug 8 2024 |
et al., Clinical Infectious Diseases, doi:10.1093/cid/ciae408 | Single monoclonal antibodies should not be used for COVID-19 therapy: a call for antiviral stewardship |
| Review arguing against use of single monoclonal antibodies for COVID-19 treatment, particularly in immunosuppressed patients, due to the risk of rapidly selecting for resistant viral variants. Authors suggest that while monoclonal antibod.. | ||
Aug 5 2024 |
et al., Viral Immunology, doi:10.1089/vim.2024.0034 | Effective Treatment of COVID-19 Infection with Repurposed Drugs: Case Reports |
| Review of the successful treatment of COVID-19 using existing medications including HCQ, AZ, ivermectin, famotidine, monoclonal antibodies, and others. Authors note that the typical treatment of severe viral infections with multiple thera.. | ||
Jul 16 2024 |
et al., PLOS ONE, doi:10.1371/journal.pone.0304822 | Real-world effectiveness of sotrovimab in preventing hospitalization and mortality in high-risk patients with COVID-19 in the United States: A cohort study from the Mayo Clinic electronic health records |
| 50% lower mortality (p=0.2), 12% lower combined mortality/hospitalization (p=0.7), 74% lower ICU admission (p=0.006), and 59% lower need for oxygen therapy (p<0.0001). Retrospective 35,485 high-risk COVID-19 outpatients showing lower ICU admission and respiratory support with sotrovimab. There was no significant difference for hospitalization. | ||
Jun 19 2024 |
et al., International Journal of Infectious Diseases, doi:10.1016/j.ijid.2024.107136 | Real-world evidence of sotrovimab effectiveness for preventing severe outcomes in patients with COVID-19: A quality improvement propensity matched retrospective cohort study of a pan-provincial program in Alberta, Canada |
| 20% higher combined mortality/hospitalization (p=0.2). Retrospective propensity-matched study of 22,289 high-risk COVID-19 outpatients in Canada, showing no significant difference in combined hospitalization/mortality with sotrovimab treatment. In a subgroup analysis of patients with no comor.. | ||
Apr 30 2024 |
et al., European Review for Medical and Pharmacological Sciences, doi:10.26355/eurrev_202404_35925 | The safety signal detection and analysis of monoclonal antibodies against SARS-CoV-2 based on real-world evidence – the suitable selectivity for different populations |
| FDA adverse event reporting analysis for bebtelovimab, tixagevimab/cilgavimab, and sotrovimab, showing distinct safety profiles with concerning adverse events for each drug. Bebtelovimab showed the highest associations with gastrointestin.. | ||
Apr 23 2024 |
et al., Immunity, Inflammation and Disease, doi:10.1002/iid3.1262 | Comparison of effectiveness and safety of molnupiravir versus sotrovimab for COVID‐19: A systematic review and meta‐analysis |
| Meta analysis of 13 studies involving 16,166 patients showing higher mortality and higher incidence of adverse events with molnupiravir compared with sotrovimab. | ||
Mar 30 2024 |
et al., Infection, doi:10.1007/s15010-024-02223-y | SARS-CoV-2 journey: from alpha variant to omicron and its sub-variants |
| Review of SARS-CoV-2 variants showing increased transmissibility, disease severity, and immune escape with mutations in the spike protein receptor binding domain. Authors cover variants from the initial D614G mutation through omicron sub-.. | ||
Feb 20 2024 |
et al., Clinical Drug Investigation, doi:10.1007/s40261-024-01344-4 | Real-World Effectiveness of Sotrovimab for the Early Treatment of COVID-19: Evidence from the US National COVID Cohort Collaborative (N3C) |
| 24% lower combined mortality/hospitalization (p=0.0001) and 21% lower hospitalization (p=0.001). N3C retrospective 4,992 high-risk outpatients with mild-to-moderate COVID-19 showing reduced risk of hospitalization or death with sotrovimab treatment compared to 541,325 untreated controls during periods of Delta and Omicron BA.2 varian.. | ||
Jan 26 2024 |
et al., JAMA Health Forum, doi:10.1001/jamahealthforum.2023.5044 | Clinical Risk and Outpatient Therapy Utilization for COVID-19 in the Medicare Population |
| Analysis of Medicare beneficiaries in 2022 showing that outpatient COVID-19 treatments like antivirals and monoclonal antibodies were disproportionately used by patients at lower risk of severe infection and outcomes. Retrospective studie.. | ||
Dec 4 2023 |
et al., Journal of Infection and Public Health, doi:10.1016/j.jiph.2023.11.029 | Real world Effectiveness of Sotrovimab in Preventing COVID-19–related Hospitalisation or Death in Patients Infected with Omicron BA.2 |
| 74% lower combined mortality/ICU admission (p=0.001). Analysis of 1,180 high-risk COVID-19 outpatients infected with Omicron BA.2 showing lower risk of death or ICU admission with sotrovimab treatment. | ||
Nov 30 2023 |
et al., iScience, doi:10.1016/j.isci.2023.108147 | Antiviral efficacy against and replicative fitness of an XBB.1.9.1 clinical isolate |
| In vitro and animal study showing that the SARS-CoV-2 omicron subvariant XBB.1.9.1 has similar antigenicity, antiviral susceptibility, and replicative ability compared to XBB.1.5. Casirivimab, imdevimab, tixagevimab, cilgavimab, sotrovima.. | ||
Nov 27 2023 |
et al., Open Forum Infectious Diseases, doi:10.1093/ofid/ofad500.579 | Real-world Effectiveness of Sotrovimab for COVID-19: Evidence from United States (US) Administrative Claims Data |
| 80% lower mortality (p<0.0001), 30% lower ICU admission (p=0.0002), and 16% lower hospitalization (p<0.0001). Retrospective 34,160 COVID-19 patients in the USA treated with sotrovimab matched to 68,320 untreated patients from May 2021-April 2022, during periods when Delta and early Omicron were predominant. Sotrovimab-treated patients had signifi.. | ||
Nov 23 2023 |
et al., The Journal of Infectious Diseases, doi:10.1093/infdis/jiad523 | Spike protein genetic evolution in patients at high-risk of severe COVID-19 treated by monoclonal antibodies |
| Prospective study of 264 high-risk COVID-19 patients treated with monoclonal antibodies. Tixagevimab/cilgavimab was associated with 5 times higher risk of emergence of mutations. Treatment with sotrovimab was linked to mutations associate.. | ||
Sep 28 2023 |
et al., Vaccines, doi:10.3390/vaccines11101533 | In Vitro Efficacy of Antivirals and Monoclonal Antibodies against SARS-CoV-2 Omicron Lineages XBB.1.9.1, XBB.1.9.3, XBB.1.5, XBB.1.16, XBB.2.4, BQ.1.1.45, CH.1.1, and CL.1 |
| In vitro study showing sharply reduced neutralization of SARS-CoV-2 variants XBB.1.9.1, XBB.1.9.3, XBB.1.5, XBB.1.16, XBB.2.4, BQ.1.1.45, CH.1.1, and CL.1 with monoclonal antibodies cilgavimab, tixagevimab, imdevimab, etsevimab, casirivim.. | ||
Sep 27 2023 |
et al., Microorganisms, doi:10.3390/microorganisms11102417 | Prevalence of SARS-CoV-2 Omicron Sublineages and Spike Protein Mutations Conferring Resistance against Monoclonal Antibodies in a Swedish Cohort during 2022–2023 |
| Analysis of 7,950 SARS-CoV-2 samples from central Sweden collected between March 2022 and May 2023 tracking the prevalence of omicron sublineages and mutations in the spike protein conferring resistance to monoclonal antibodies over time... | ||
Aug 17 2023 |
et al., Viruses, doi:10.3390/v15081757 | What Is the Efficacy of Sotrovimab in Reducing Disease Progression and Death in People with COVID-19 during the Omicron Era? Answers from a Real-Life Study |
| 81% lower mortality (p=0.0005) and 92% lower need for oxygen therapy (p<0.0001). Retrospective 689 COVID-19 patients in Italy, showing lower mortality with sotrovimab treatment. | ||
Aug 10 2023 |
et al., Drug Resistance Updates, doi:10.1016/j.drup.2023.100991 | Analysis of SARS-CoV-2 mutations associated with resistance to therapeutic monoclonal antibodies that emerge after treatment |
| Review of reports of treatment-emergent resistance to COVID-19 monoclonal antibodies (mAbs), showing that some post-mAb treatment mutations appeared to spread globally soon after the mAb was introduced, raising concerns about transmission.. | ||
Jul 27 2023 |
et al., BMJ Open Respiratory Research, doi:10.1136/bmjresp-2023-002238 | Comparative effectiveness of sotrovimab versus no treatment in non-hospitalised high-risk COVID-19 patients in north west London: a retrospective cohort study |
| 29% lower mortality (p=0.65), 50% lower combined mortality/hospitalization (p=0.07), and 57% lower hospitalization (p=0.05). Retrospective 599 high-risk sotrovimab patients and 5,191 untreated controls, showing lower hospitalization/mortality with treatment, without statistical significance in the overall cohort. Efficacy was better for those ≥65, and efficacy .. | ||
May 31 2023 |
et al., Viruses, doi:10.3390/v15061300 | Clinical Efficacy of the Neutralizing Antibody Therapy Sotrovimab in Patients with SARS-CoV-2 Omicron BA.1 and BA.2 Subvariant Infections |
| 55% lower need for oxygen therapy (p<0.0001). Retrospective 844 patients treated with sotrovimab and matched controls in Japan, showing lower risk of oxygen therapy with treatment. | ||
May 16 2023 |
et al., medRxiv, doi:10.1101/2023.05.12.23289914 | Effectiveness of Sotrovimab and Molnupiravir in community settings in England across the Omicron BA.1 and BA.2 sublineages: emulated target trials using the OpenSAFELY platform |
| 16% lower combined mortality/hospitalization (p=0.002). OpenSAFELY retrospective 75,048 outpatients in the UK, using the clone-censor-weight approach to address immortal time bias, showing lower combined mortality/hospitalization with sotrovimab treatment. | ||
Apr 4 2023 |
et al., Annals of Internal Medicine, doi:10.7326/M22-1286 | Evolving Real-World Effectiveness of Monoclonal Antibodies for Treatment of COVID-19 |
| 30% lower combined mortality/hospitalization (p=0.14). Retrospective 2,571 patients treated with mAbs in the USA, and 5,135 control patients, showing lower combined mortality/hospitalization for bamlanivimab, bamlanivimab/etesevimab, casirivimab/imdevimab, sotrovimab, and bebtelovimab, with s.. | ||
Mar 24 2023 |
et al., medRxiv, doi:10.1101/2023.03.21.23287410 | Intramuscular Versus Intravenous SARS-CoV-2 Neutralizing Antibody Sotrovimab for Treatment of COVID-19 (COMET-TAIL): A Randomized Non-inferiority Clinical Trial |
| RCT 982 high risk outpatients in the USA reporting that intramuscular sotrovimab was non-inferior to intravenous administration. Death and hospitalization was more frequent with intramuscular administration, without statistical significan.. | ||
Mar 15 2023 |
et al., PLOS ONE, doi:10.1371/journal.pone.0281915 | Evaluation of outpatient treatment for non-hospitalised patients with COVID-19: The experience of a regional centre in the UK |
| 75% lower mortality (p=0.55) and 60% lower hospitalization (p=0.35). Retrospective 604 outpatients in the UK, showing lower risk of hospitalization with sotrovimab treatment, without statistical significance due to the small number of hospitalizations. | ||
Jan 25 2023 |
et al., Journal of Infection, doi:10.1016/j.jinf.2023.02.012 | Real-world effectiveness of molnupiravir, nirmatrelvir-ritonavir, and sotrovimab on preventing hospital admission among higher-risk patients with COVID-19 in Wales: A retrospective cohort study |
| 27% lower combined mortality/hospitalization (p=0.03). Retrospective high risk outpatients in the UK, showing lower hospitalization/death with sotrovimab treatment. Residual confounding is likely with adjustments having no detail on specific comorbidities. | ||
Jan 22 2023 |
et al., medRxiv, doi:10.1101/2023.01.20.23284849 | Comparative effectiveness of Paxlovid versus sotrovimab and molnupiravir for preventing severe COVID-19 outcomes in non-hospitalised patients: observational cohort study using the OpenSAFELY platform |
| 4% lower combined mortality/hospitalization (p=0.91). OpenSAFELY retrospective 7,683 outpatients in the UK, showing no significant difference in hospitalization/death between paxlovid and sotrovimab. | ||
Dec 8 2022 |
et al., Microbiology Spectrum, doi:10.1128/spectrum.04103-22 | Sotrovimab in Hospitalized Patients with SARS-CoV-2 Omicron Variant Infection: a Propensity Score-Matched Retrospective Cohort Study |
| 140% higher mortality (p=0.12). PSM retrospective 1,254 hospitalized patients in Germany, 147 treated with sotrovimab, showing higher mortality with sotrovimab, without statistical significance. | ||
Nov 16 2022 |
et al., BMJ, doi:10.1136/bmj-2022-071932 | Comparative effectiveness of sotrovimab and molnupiravir for prevention of severe covid-19 outcomes in patients in the community: observational cohort study with the OpenSAFELY platform |
| 50% lower combined mortality/hospitalization (p=0.005). Retrospective 3,331 sotrovimab and 2,689 molnupiravir patients in the UK, showing lower risk of combined hospitalization/death with sotrovimab. | ||
Oct 7 2022 |
et al., Journal of Medical Virology, doi:10.1002/jmv.28186 | Viral load decrease in SARS‐CoV‐2 BA.1 and BA.2 Omicron sublineages infection after treatment with monoclonal antibodies and direct antiviral agents |
| Observational study of 521 outpatients with mild-to-moderate COVID-19 showing paxlovid had stronger viral load reduction against Omicron BA.1 and BA.2 variants compared to sotrovimab, molnupiravir, and remdesivir, with the exception of co.. | ||
Oct 6 2022 |
et al., Open Forum Infectious Diseases, doi:10.1093/ofid/ofac527 | Demographics and outcomes of initial phase of COVID-19 Medicines Delivery Units across 4 UK centres during peak B1.1.529 omicron epidemic: a service evaluation |
| 258% higher hospitalization (p=0.15). Retrospective 186 patients in the UK treated with sotrovimab, and 222 eligible but declining treatment, showing no significant difference in hospitalization. No group details are provided and the results are subject to confounding by indi.. | ||
Oct 5 2022 |
et al., Research Square, doi:10.21203/rs.3.rs-2118653/v1 | Real-world clinical outcomes of treatment with molnupiravir for patients with mild- to-moderate coronavirus disease 2019 during the Omicron variant pandemic |
| 8% higher progression (p=0.73). Retrospective 1,921 patients in Japan, showing no significant difference in progression with sotrovimab use. | ||
Oct 3 2022 |
et al., Clinical Infectious Diseases, doi:10.1093/cid/ciac802 | Rapid Selection of Sotrovimab Escape Variants in Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) Omicron-Infected Immunocompromised Patients |
| Prospective analysis of 57 COVID-19 patients receiving sotrovimab, showing rapid creation of escape mutations within immunodeficient patients. Combined treatment with remdesivir reduced the creation of escape variants. | ||
Sep 16 2022 |
et al., Scientific Reports, doi:10.1038/s41598-022-19993-w | Reevaluation of antibody-dependent enhancement of infection in anti-SARS-CoV-2 therapeutic antibodies and mRNA-vaccine antisera using FcR- and ACE2-positive cells |
| In vitro study showing no antibody-dependent enhancement (ADE) with sotrovimab. ADE was seen for casirivimab/imdevimab within a specific concentration range. | ||
Sep 11 2022 |
et al., medRxiv, doi:10.1101/2022.09.07.22279497 | Real-world Effectiveness of Sotrovimab for the Early Treatment of COVID-19 During SARS-CoV-2 Delta and Omicron Waves in the United States |
| 88% lower mortality (p<0.0001) and 61% lower hospitalization (p<0.0001). Retrospective 1,530,501 high-risk patients in the USA, 15,633 treated with sotrovimab, showing significantly lower mortality and hospitalization with treatment. Sotrovimab maintained efficacy throughout the period analyzed - September 202.. | ||
Sep 9 2022 |
et al., Infection, doi:10.1007/s15010-022-01914-8 | Outcome of lung transplant recipients infected with SARS-CoV-2/Omicron/B.1.1.529: a Nationwide German study |
| 20% higher severe cases (p=0.79). Retrospective 218 COVID+ lung transplant patients in Germany, showing no significant difference in severe cases with early sotrovimab use. | ||
Aug 1 2022 |
et al., Journal of Antimicrobial Chemotherapy, doi:10.1093/jac/dkac256 | Real-world effectiveness of early remdesivir and sotrovimab in the highest-risk COVID-19 outpatients during the Omicron surge |
| 35% lower hospitalization (p=0.46), 66% fewer combined hospitalization/ER visits (p=0.01), and 90% lower progression (p=0.009). Retrospective high-risk outpatients in the USA, 82 treated with remdesivir, 88 with sotrovimab, and 90 control patients, showing significantly lower combined hospitalization/ER visits with both treatments in unadjusted results. The domina.. | ||
Jul 28 2022 |
et al., Clinical Kidney Journal, doi:10.1093/ckj/sfac177 | Treatment with sotrovimab for SARS-CoV-2 infection in a cohort of high-risk kidney transplant recipients |
| Retrospective 82 kidney transplant recipients treated with sotrovimab, showing lower risk of serious COVID-19 outcomes with early treatment. | ||
Jun 18 2022 |
et al., International Journal of Infectious Diseases, doi:10.1016/j.ijid.2022.10.002 (date from preprint) | Change in Effectiveness of Sotrovimab for Preventing Hospitalization and Mortality for At-risk COVID-19 Outpatients During an Omicron BA.1 and BA.1.1-Predominant Phase |
| 38% lower mortality (p=0.62), 18% lower hospitalization (p=0.32), and 3% higher progression (p=0.83). Retrospective 30,247 outpatients in the USA, showing no significant differences with sotrovimab with omicron BA.1. | ||
May 3 2022 |
et al., medRxiv, doi:10.1101/2022.05.03.22274524 | Kidney Transplant Recipients and Omicron: Outcomes, effect of vaccines and the efficacy and safety of novel treatments |
| 90% lower hospitalization (p=0.008). Retrospective 116 kidney transplant recipients diagnosed with COVID-19 in the community during the Omicron era in the UK, showing lower hospitalization with sotrovimab, but no significant difference with molnupiravir. Two molnupiravir-tre.. | ||
Apr 21 2022 |
et al., International Journal of Infectious Diseases, doi:10.1016/j.ijid.2022.09.023 (date from preprint) | Effectiveness of the neutralizing antibody sotrovimab among high-risk patients with mild-to-moderate SARS-CoV-2 in Qatar |
| 165% higher progression (p=0.19). Retrospective 345 sotrovimab treated patients in Qatar matched with 583 patients that opted not to receive treatment, showing higher progression with treatment, without statistical significance. | ||
Apr 5 2022 |
Update, 4/5 | FDA updates Sotrovimab emergency use authorization |
| FDA has revoked sotrovimab's authorization in all regions due to predicted low efficacy with BA.2, and BA.2 exceeding 50% prevalence in all regions. | ||
Apr 5 2022 |
et al., The Journal of Infectious Diseases, doi:10.1093/infdis/jiac206 (date from preprint) | Real-World Evidence of the Neutralizing Monoclonal Antibody Sotrovimab for Preventing Hospitalization and Mortality in COVID-19 Outpatients |
| 89% lower mortality (p=0.05), 62% lower hospitalization (p=0.002), and 11% higher progression (p=0.55). PSM retrospective 10,036 outpatients, 522 treated with sotrovimab, showing lower mortality and hospitalization with treatment. | ||
Mar 5 2022 |
et al., Antibiotics, doi:10.3390/antibiotics11030345 | Real-World Use of Sotrovimab for Pre-Emptive Treatment in High-Risk Hospitalized COVID-19 Patients: An Observational Cross-Sectional Study |
| 56% lower ICU admission (p=0.35) and 59% lower progression (p=0.05). Retrospective 19 sotrovimab patients and 75 controls is Singapore, showing lower progression with treatment. | ||
Feb 16 2022 |
et al., bioRxiv, doi:10.1101/2022.02.15.480166 | SARS-CoV-2 Omicron BA.2 Variant Evades Neutralization by Therapeutic Monoclonal Antibodies |
| In vitro study showing that omicron BA.2 evades all monoclonal antibodies tested, including sotrovimab and tixagevimab/cilgavimab which retained activity for omicron BA.1. | ||
Dec 23 2021 |
et al., The Lancet Infectious Diseases, doi:10.1016/S1473-3099(21)00751-9 | Efficacy and safety of two neutralising monoclonal antibody therapies, sotrovimab and BRII-196 plus BRII-198, for adults hospitalised with COVID-19 (TICO): a randomised controlled trial |
| 2% higher mortality (p=0.96) and 11% improved recovery (p=0.29). RCT with 182 sotrovimab patients, 176 BRII-196+BRII-198 patients, and 178 control patients, median 8 days from symptom onset, showing no significant differences and terminated early due to futility. Long-term results are reported in [Mou.. | ||
Dec 21 2021 |
et al., medRxiv, doi:10.1101/2021.12.18.21267628 | Resistance conferring mutations in SARS-CoV-2 delta following sotrovimab infusion |
| Retrospective 100 sotrovimab patients in Australia, 23 PCR+ more than 10 days post-infusion (68 with status unknown), showing rapid development of spike gene mutations that have been shown to confer high level resistance to sotrovimab in .. | ||
Dec 20 2021 |
et al., bioRxiv, doi:10.1101/2021.12.19.473354 | Variable loss of antibody potency against SARS-CoV-2 B.1.1.529 (Omicron) |
| In vitro study showing that omicron is substantially resistant to neutralization by monoclonal antibodies REGN10933, REGN10987, Ly-CoV016 and Ly-CoV555. S309 (the parent of Sotrovimab) had only 2-fold loss in potency. | ||
Dec 17 2021 |
et al., bioRxiv, doi:10.1101/2021.12.15.472828 | An infectious SARS-CoV-2 B.1.1.529 Omicron virus escapes neutralization by several therapeutic monoclonal antibodies |
| In vitro study (Vero-TMPRSS2 and Vero-hACE2-TMPRSS2) showing complete loss of inhibitory activity for B.1.1.529 omicron with LY-CoV555, LY-CoV016, REGN10933, REGN10987, and CT-P59, ~12-fold decrease for COV2-2196/COV2-2130, and minimal ch.. | ||
Dec 15 2021 |
et al., bioRxiv, doi:10.1101/2021.12.14.472719 | Striking Antibody Evasion Manifested by the Omicron Variant of SARS-CoV-2 |
| In vitro study (Vero-E6-TMPRSS2) showing 18 of 19 monoclonal antibodies were no longer effective or significantly impaired with B.1.1.529 omicron. | ||
Dec 4 2021 |
et al., JAMA, doi:10.1001/jama.2022.2832 (results published 12/4/21) | Effect of Sotrovimab on Hospitalization or Death Among High-risk Patients With Mild to Moderate COVID-19 |
| 89% lower ventilation (p=0.12), 75% lower progression (p=0.0004), and 79% lower combined mortality/hospitalization (p=0.0004). RCT 1,057 outpatients, 529 treated with sotrovimab, showing significantly lower hospitalization >24h or mortality with treatment. | ||
May 26 2021 |
et al., NEJM, doi:10.1056/NEJMoa2107934 (news release 5/26/2021) | Early Treatment for Covid-19 with SARS-CoV-2 Neutralizing Antibody Sotrovimab |
| 91% lower ICU admission (p=0.06) and 81% lower hospitalization (p=0.0007). Interim results from the COMET-ICE trial showing significantly lower hospitalization with treatment. NCT04545060. | ||
References
Liu et al., Striking Antibody Evasion Manifested by the Omicron Variant of SARS-CoV-2, bioRxiv, doi:10.1101/2021.12.14.472719.
Sheward et al., Variable loss of antibody potency against SARS-CoV-2 B.1.1.529 (Omicron), bioRxiv, doi:10.1101/2021.12.19.473354.
VanBlargan et al., An infectious SARS-CoV-2 B.1.1.529 Omicron virus escapes neutralization by several therapeutic monoclonal antibodies, bioRxiv, doi:10.1101/2021.12.15.472828.
Haars et al., Prevalence of SARS-CoV-2 Omicron Sublineages and Spike Protein Mutations Conferring Resistance against Monoclonal Antibodies in a Swedish Cohort during 2022–2023, Microorganisms, doi:10.3390/microorganisms11102417.
Pochtovyi et al., In Vitro Efficacy of Antivirals and Monoclonal Antibodies against SARS-CoV-2 Omicron Lineages XBB.1.9.1, XBB.1.9.3, XBB.1.5, XBB.1.16, XBB.2.4, BQ.1.1.45, CH.1.1, and CL.1, Vaccines, doi:10.3390/vaccines11101533.
Zhou et al., SARS-CoV-2 Omicron BA.2 Variant Evades Neutralization by Therapeutic Monoclonal Antibodies, bioRxiv, doi:10.1101/2022.02.15.480166.
Uraki et al., Antiviral efficacy against and replicative fitness of an XBB.1.9.1 clinical isolate, iScience, doi:10.1016/j.isci.2023.108147.
Focosi et al., Analysis of SARS-CoV-2 mutations associated with resistance to therapeutic monoclonal antibodies that emerge after treatment, Drug Resistance Updates, doi:10.1016/j.drup.2023.100991.
Leducq et al., Spike protein genetic evolution in patients at high-risk of severe COVID-19 treated by monoclonal antibodies, The Journal of Infectious Diseases, doi:10.1093/infdis/jiad523.
Bruhn et al., Somatic hypermutation shapes the viral escape profile of SARS-CoV-2 neutralising antibodies, eBioMedicine, doi:10.1016/j.ebiom.2025.105770.
Ngiam et al., Early administration of neutralising monoclonal antibodies and post-acute sequelae of COVID-19, International Journal of Infectious Diseases, doi:10.1016/j.ijid.2026.108435.
Choudhary et al., Emergence of SARS-CoV-2 Resistance with Monoclonal Antibody Therapy, medRxiv, doi:10.1101/2021.09.03.21263105.
Günther et al., Variant-specific humoral immune response to SARS-CoV-2 escape mutants arising in clinically severe, prolonged infection, medRxiv, doi:10.1101/2024.01.06.24300890.
Casadevall et al., Single monoclonal antibodies should not be used for COVID-19 therapy: a call for antiviral stewardship, Clinical Infectious Diseases, doi:10.1093/cid/ciae408.