Early High-Titer Plasma Therapy to Prevent Severe Covid-19 in Older Adults
et al., NEJM, doi:10.1056/NEJMoa2033700, INFANT-COVID-19, NCT04479163, Jan 2021
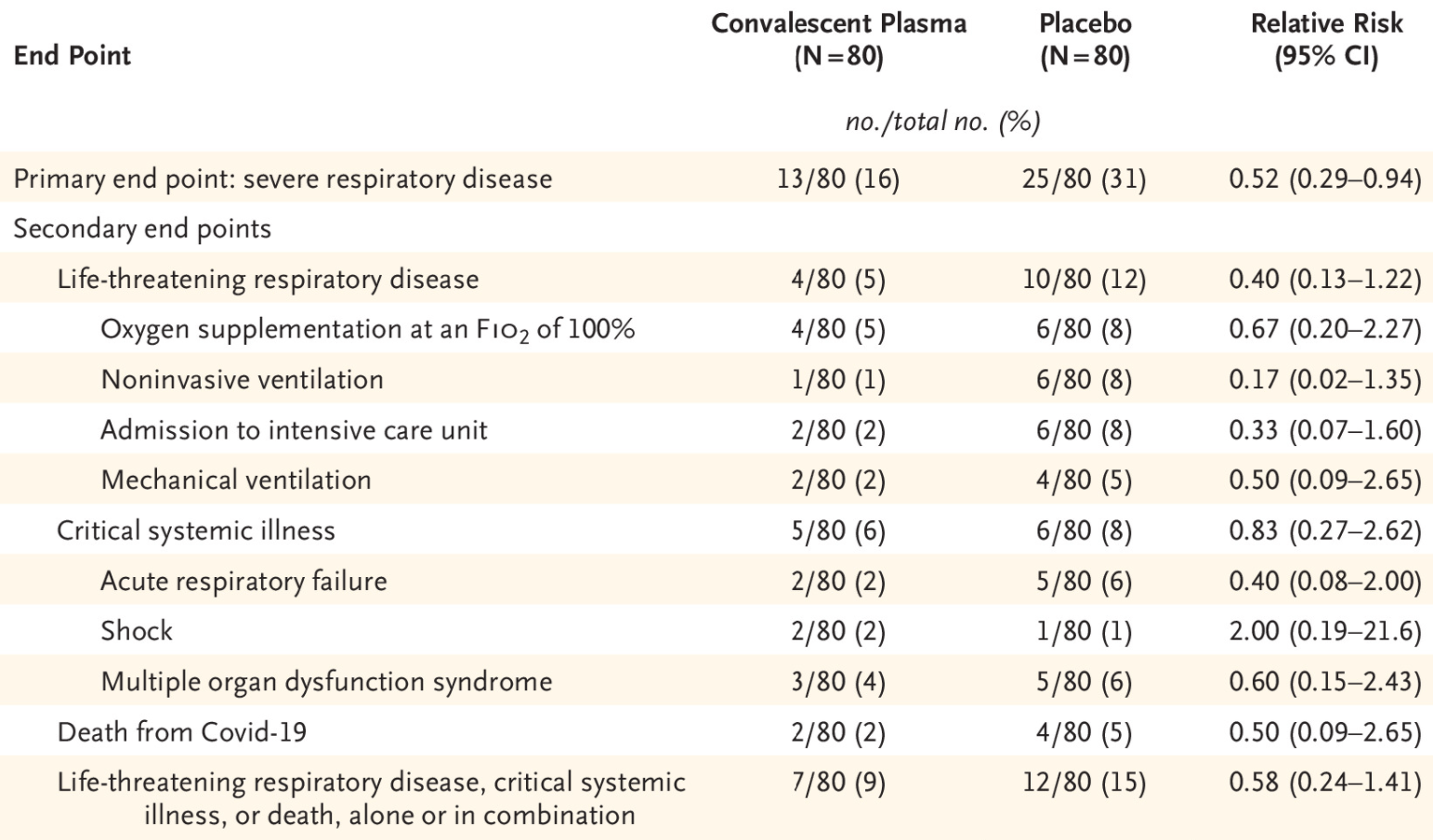
RCT 160 patients ≥65 with symptom onset <72 hours, 80 treated with convalescent plasma, showing lower progression to severe disease with treatment.
|
risk of death, 50.0% lower, RR 0.50, p = 0.43, treatment 2 of 80 (2.5%), control 4 of 80 (5.0%), NNT 40.
|
|
risk of mechanical ventilation, 50.0% lower, RR 0.50, p = 0.43, treatment 2 of 80 (2.5%), control 4 of 80 (5.0%), NNT 40.
|
|
risk of ICU admission, 67.0% lower, RR 0.33, p = 0.17, treatment 2 of 80 (2.5%), control 6 of 80 (7.5%), NNT 20.
|
|
risk of progression, 48.0% lower, RR 0.52, p = 0.03, treatment 13 of 80 (16.2%), control 25 of 80 (31.2%), NNT 6.7.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Libster et al., 6 Jan 2021, Double Blind Randomized Controlled Trial, Argentina, peer-reviewed, 56 authors, study period 4 June, 2020 - 25 October, 2020, average treatment delay 1.825 days, trial NCT04479163 (history) (INFANT-COVID-19).
Early High-Titer Plasma Therapy to Prevent Severe Covid-19 in Older Adults
New England Journal of Medicine, doi:10.1056/nejmoa2033700
which is the author's version after external peer review and before publication in the Journal, is available under a CC BY license at PMC7793608.
Appendix The authors' full names and academic degrees are as follows: Romina Libster, M.D., Gonzalo Pérez Marc, M.D., Diego Wappner, M.D., Silvina Coviello, M.S., Alejandra Bianchi, Virginia Braem, Ignacio Esteban, M.D., Mauricio T. Caballero, M.D., Cristian Wood, M.D., Mabel Berrueta, M.D., Aníbal Rondan, M.D., Gabriela Lescano, M.D., Pablo Cruz, M.D., Yvonne Ritou, M.D., Valeria Fernández Viña, M.D., Damián Álvarez Paggi, Ph.D., Sebastián Esperante, Ph.D., Adrián Ferreti, Gastón Ofman, M.D., Álvaro Ciganda, B.I.T., Rocío Rodriguez, Jorge Lantos, M.D., Ricardo Valentini, M.D., Nicolás Itcovici, M.D., Alejandra Hintze, M.D., M. Laura Oyarvide, M.D., Candela Etchegaray, M.D., Alejandra Neira, M.D., Ivonne Name, M.D., Julieta Alfonso, M.D., Rocío López Castelo, M.D., Gisela Caruso, M.D., Sofía Rapelius, M.S., Fernando Alvez, M.D., Federico Etchenique, M.D., Federico Dimase, M.D., Darío Alvarez, M.S., Sofía S. Aranda, M.D., Clara Sánchez Yanotti, Julián De Luca, Sofía Jares Baglivo, Sofía Laudanno, Florencia Nowogrodzki, Ramiro Larrea, M.
References
Agarwal, Mukherjee, Kumar, Chatterjee, Bhatnagar et al., Convalescent plasma in the management of moderate covid-19 in adults in India: open label phase II multicentre randomised controlled trial (PLACID Trial), BMJ
Arciuolo, Jablonski, Zucker, Rosen, Effectiveness of measles vaccination and immune globulin post-exposure prophylaxis in an outbreak setting -New York City, 2013, Clin Infect Dis
Avendaño-Sola, Ramos-Martinez, Munez-Rubio, Convalescent plasma for COVID-19: a multicenter, randomized clinical trial, doi:10.1101/2020.08.26.20182444v1
Cao, Wang, Wen, A trial of lopinavir-ritonavir in adults hospitalized with severe Covid-19, N Engl J Med
Chen, Nirula, Heller, SARS-CoV-2 neutralizing antibody LY-CoV555 in outpatients with Covid-19, doi:10.1056/NEJMoa2029849.19
Gold, Rossen, Ahmad, Race, ethnicity, and age trends in persons who died from COVID-19 -United States, May-August 2020, MMWR Morb Mortal Wkly Rep
Joyner, Senefeld, Klassen, Effect of convalescent plasma on mortality among hospitalized patients with COVID-19: initial three-month experience, doi:10.1101/2020.08.12.20169359v1
Joyner, Wright, Fairweather, Early safety indicators of COVID-19 convalescent plasma in 5000 patients, J Clin Invest
Li, Zhang, Hu, Effect of convalescent plasma therapy on time to clinical improvement in patients with severe and life-threatening COVID-19: a randomized clinical trial, JAMA
Liu, Lin, Baine, Convalescent plasma treatment of severe COVID-19: a propensity score-matched control study, Nat Med
Maiztegui, Fernandez, De Damilano, Efficacy of immune plasma in treatment of Argentine haemorrhagic fever and association between treatment and a late neurological syndrome, Lancet
Salazar, Christensen, Graviss, Treatment of coronavirus disease 2019 patients with convalescent plasma reveals a signal of significantly decreased mortality, Am J Pathol
Simonovich, Pratx, Scibona, A randomized trial of convalescent plasma in Covid-19 severe pneumonia, N Engl J Med
Stokes, Zambrano, Anderson, Coronavirus disease 2019 case surveillance -United States, January 22, MMWR Morb Mortal Wkly Rep
Stone, Frigault, Boyd, Efficacy of tocilizumab in patients hospitalized with Covid-19, N Engl J Med
The, Group, Effect of hydroxychloroquine in hospitalized patients with Covid-19, N Engl J Med
Williamson, Walker, Bhaskaran, Factors associated with COVID-19-related death using OpenSAFELY, Nature
DOI record:
{
"DOI": "10.1056/nejmoa2033700",
"ISSN": [
"0028-4793",
"1533-4406"
],
"URL": "http://dx.doi.org/10.1056/NEJMoa2033700",
"alternative-id": [
"10.1056/NEJMoa2033700"
],
"author": [
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Libster",
"given": "Romina",
"sequence": "first"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Pérez Marc",
"given": "Gonzalo",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Wappner",
"given": "Diego",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Coviello",
"given": "Silvina",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Bianchi",
"given": "Alejandra",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Braem",
"given": "Virginia",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Esteban",
"given": "Ignacio",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Caballero",
"given": "Mauricio T.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Wood",
"given": "Cristian",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Berrueta",
"given": "Mabel",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Rondan",
"given": "Aníbal",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Lescano",
"given": "Gabriela",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Cruz",
"given": "Pablo",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Ritou",
"given": "Yvonne",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Fernández Viña",
"given": "Valeria",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Álvarez Paggi",
"given": "Damián",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Esperante",
"given": "Sebastián",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Ferreti",
"given": "Adrián",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Ofman",
"given": "Gastón",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Ciganda",
"given": "Álvaro",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Rodriguez",
"given": "Rocío",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Lantos",
"given": "Jorge",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Valentini",
"given": "Ricardo",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Itcovici",
"given": "Nicolás",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Hintze",
"given": "Alejandra",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Oyarvide",
"given": "M. Laura",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Etchegaray",
"given": "Candela",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Neira",
"given": "Alejandra",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Name",
"given": "Ivonne",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Alfonso",
"given": "Julieta",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "López Castelo",
"given": "Rocío",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Caruso",
"given": "Gisela",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Rapelius",
"given": "Sofía",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Alvez",
"given": "Fernando",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Etchenique",
"given": "Federico",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Dimase",
"given": "Federico",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Alvarez",
"given": "Darío",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Aranda",
"given": "Sofía S.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Sánchez Yanotti",
"given": "Clara",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "De Luca",
"given": "Julián",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Jares Baglivo",
"given": "Sofía",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Laudanno",
"given": "Sofía",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Nowogrodzki",
"given": "Florencia",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Larrea",
"given": "Ramiro",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Silveyra",
"given": "María",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Leberzstein",
"given": "Gabriel",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Debonis",
"given": "Alejandra",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Molinos",
"given": "Juan",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "González",
"given": "Miguel",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Perez",
"given": "Eduardo",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Kreplak",
"given": "Nicolás",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Pastor Argüello",
"given": "Susana",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Gibbons",
"given": "Luz",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Althabe",
"given": "Fernando",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Bergel",
"given": "Eduardo",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Fundación INFANT (R. Libster, S.C., A.B., I.E., M.T.C., C.W., D.A.P., S.E., A.F., G.O., S.S.A., C.S.Y., J.D.L., S.J.B., S.L., F.N., F.P.P.), iTrials (R. Libster, G.P.M., D.W., S.C., A.B., V.B., S.S.A., F.P.P.), Swiss Medical Group (D.W., J.L., F.E., J.M.), National Scientific and Technical Research Council (M.T.C., D.A.P., S.E.), Hospital Dr. Carlos Bocalandro (A.R., G. Lescano), Centro Gallego (P.C.), Instituto de Efectividad Clínica y Sanitaria (M.B., A.C., R.R., L.G., E.B.), Hospital Simplemente..."
}
],
"family": "Polack",
"given": "Fernando P.",
"sequence": "additional"
}
],
"container-title": "New England Journal of Medicine",
"container-title-short": "N Engl J Med",
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2021,
1,
6
]
],
"date-time": "2021-01-06T22:01:50Z",
"timestamp": 1609970510000
},
"deposited": {
"date-parts": [
[
2021,
2,
17
]
],
"date-time": "2021-02-17T22:03:25Z",
"timestamp": 1613599405000
},
"funder": [
{
"DOI": "10.13039/100000865",
"doi-asserted-by": "publisher",
"name": "Bill and Melinda Gates Foundation"
},
{
"name": "The Fundacion INFANT Pandemic Fund"
}
],
"indexed": {
"date-parts": [
[
2024,
4,
9
]
],
"date-time": "2024-04-09T19:53:48Z",
"timestamp": 1712692428389
},
"is-referenced-by-count": 681,
"issue": "7",
"issued": {
"date-parts": [
[
2021,
2,
18
]
]
},
"journal-issue": {
"issue": "7",
"published-print": {
"date-parts": [
[
2021,
2,
18
]
]
}
},
"language": "en",
"license": [
{
"URL": "http://www.nejmgroup.org/legal/terms-of-use.htm",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
2,
18
]
],
"date-time": "2021-02-18T00:00:00Z",
"timestamp": 1613606400000
}
}
],
"link": [
{
"URL": "http://www.nejm.org/doi/pdf/10.1056/NEJMoa2033700",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "150",
"original-title": [],
"page": "610-618",
"prefix": "10.1056",
"published": {
"date-parts": [
[
2021,
2,
18
]
]
},
"published-print": {
"date-parts": [
[
2021,
2,
18
]
]
},
"publisher": "Massachusetts Medical Society",
"reference": [
{
"DOI": "10.15585/mmwr.mm6924e2",
"doi-asserted-by": "publisher",
"key": "r1"
},
{
"DOI": "10.1038/s41586-020-2521-4",
"doi-asserted-by": "publisher",
"key": "r2"
},
{
"DOI": "10.15585/mmwr.mm6942e1",
"doi-asserted-by": "publisher",
"key": "r3"
},
{
"DOI": "10.1056/NEJMoa2001282",
"doi-asserted-by": "publisher",
"key": "r4"
},
{
"DOI": "10.1056/NEJMoa2022926",
"doi-asserted-by": "publisher",
"key": "r5"
},
{
"DOI": "10.1056/NEJMoa2028836",
"doi-asserted-by": "publisher",
"key": "r6"
},
{
"DOI": "10.1001/jama.2020.10044",
"doi-asserted-by": "publisher",
"key": "r7"
},
{
"DOI": "10.1016/j.ajpath.2020.08.001",
"doi-asserted-by": "publisher",
"key": "r8"
},
{
"DOI": "10.1136/bmj.m3939",
"doi-asserted-by": "publisher",
"key": "r9"
},
{
"DOI": "10.1056/NEJMoa2031304",
"author": "Simonovich VA",
"doi-asserted-by": "crossref",
"first-page": "619",
"journal-title": "N Engl J Med",
"key": "r10",
"volume": "384",
"year": "2021"
},
{
"DOI": "10.1172/JCI140200",
"doi-asserted-by": "publisher",
"key": "r13"
},
{
"DOI": "10.1093/cid/cix639",
"doi-asserted-by": "publisher",
"key": "r14"
},
{
"DOI": "10.1016/S0140-6736(79)92335-3",
"doi-asserted-by": "publisher",
"key": "r16"
},
{
"DOI": "10.1056/NEJMoa2029849",
"doi-asserted-by": "publisher",
"key": "r18"
},
{
"DOI": "10.1038/s41591-020-1088-9",
"doi-asserted-by": "publisher",
"key": "r20"
}
],
"reference-count": 15,
"references-count": 15,
"relation": {},
"resource": {
"primary": {
"URL": "http://www.nejm.org/doi/10.1056/NEJMoa2033700"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"General Medicine"
],
"subtitle": [],
"title": "Early High-Titer Plasma Therapy to Prevent Severe Covid-19 in Older Adults",
"type": "journal-article",
"volume": "384"
}