Early Outpatient Treatment for Covid-19 with Convalescent Plasma
et al., New England Journal of Medicine, doi:10.1056/NEJMoa2119657, CSSC-004, NCT04373460, Dec 2021 (preprint)
RCT 1,181 outpatients in the USA, mean 6 days from symptom onset, showing lower hospitalization with treatment.
Standard of Care (SOC) for COVID-19 in the study country,
the USA, is very poor with very low average efficacy for approved treatments1.
Only expensive, high-profit treatments were approved for early treatment. Low-cost treatments were excluded, reducing the probability of early treatment due to access and cost barriers, and eliminating complementary and synergistic benefits seen with many low-cost treatments.
|
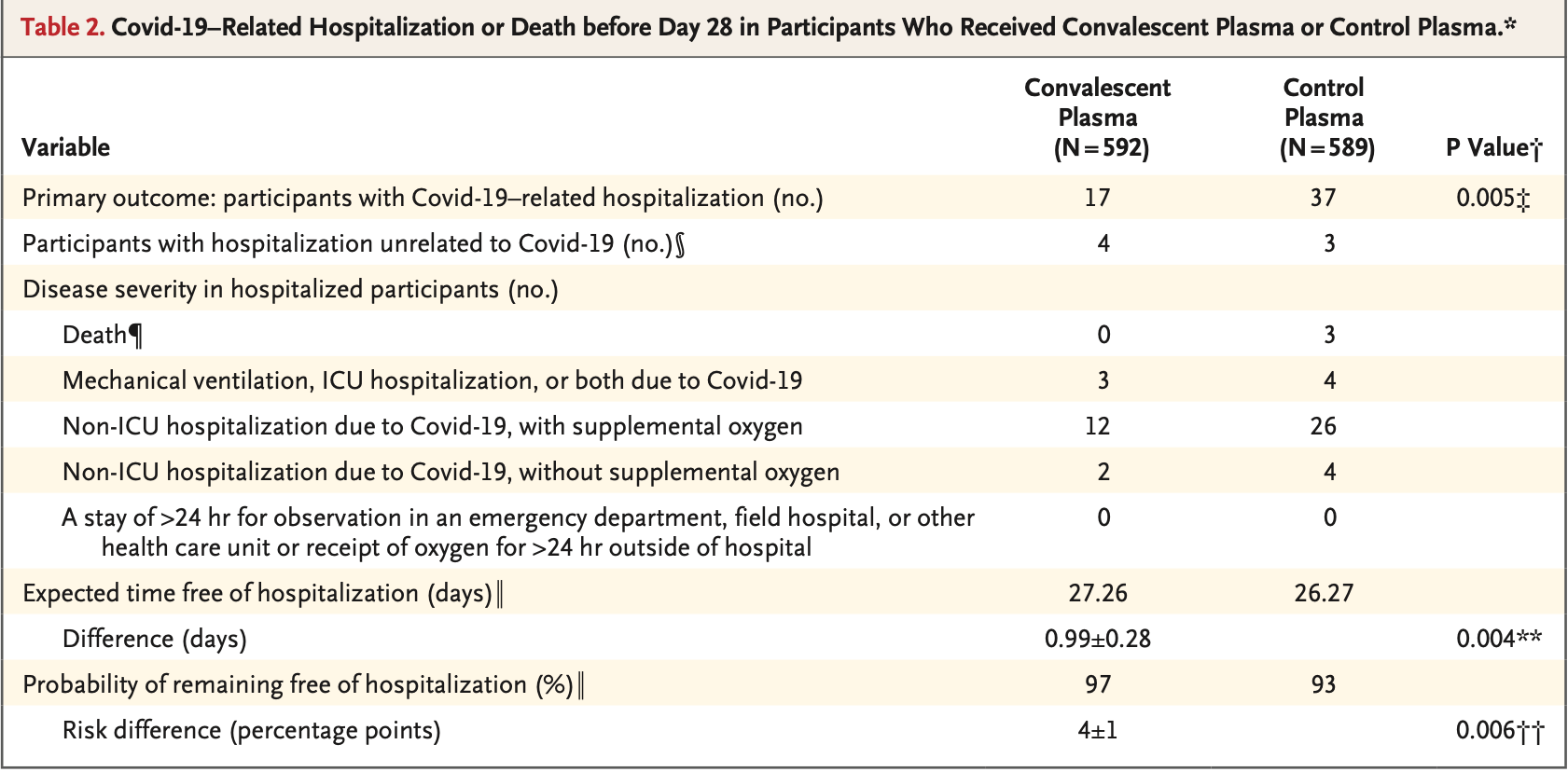
risk of death, 85.7% lower, RR 0.14, p = 0.12, treatment 0 of 592 (0.0%), control 3 of 589 (0.5%), NNT 196, relative risk is not 0 because of continuity correction due to zero events (with reciprocal of the contrasting arm).
|
|
risk of ICU admission, 25.4% lower, RR 0.75, p = 0.73, treatment 3 of 592 (0.5%), control 4 of 589 (0.7%), NNT 580.
|
|
risk of hospitalization, 54.3% lower, RR 0.46, p = 0.005, treatment 17 of 592 (2.9%), control 37 of 589 (6.3%), NNT 29.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Sullivan et al., 21 Dec 2021, Double Blind Randomized Controlled Trial, USA, peer-reviewed, 58 authors, study period 3 June, 2020 - 1 October, 2021, average treatment delay 6.0 days, trial NCT04373460 (history) (CSSC-004).
Early Outpatient Treatment for Covid-19 with Convalescent Plasma
New England Journal of Medicine, doi:10.1056/nejmoa2119657
BACKGROUND Polyclonal convalescent plasma may be obtained from donors who have recovered from coronavirus disease 2019 (Covid-19). The efficacy of this plasma in preventing serious complications in outpatients with recent-onset Covid-19 is uncertain.
METHODS In this multicenter, double-blind, randomized, controlled trial, we evaluated the efficacy and safety of Covid-19 convalescent plasma, as compared with control plasma, in symptomatic adults (≥18 years of age) who had tested positive for severe acute respiratory syndrome coronavirus 2, regardless of their risk factors for disease progression or vaccination status. Participants were enrolled within 8 days after symptom onset and received a transfusion within 1 day after randomization. The primary outcome was Covid-19-related hospitalization within 28 days after transfusion.
RESULTS Participants were enrolled from June 3, 2020, through October 1, 2021. A total of 1225 participants underwent randomization, and 1181 received a transfusion. In the prespecified modified intention-to-treat analysis that included only participants who received a transfusion, the primary outcome occurred in 17 of 592 participants (2.9%) who received convalescent plasma and 37 of 589 participants (6.3%) who received control plasma (absolute risk reduction, 3.4 percentage points; 95% confidence interval, 1.0 to 5.8; P = 0.005), which corresponded to a relative risk reduction of 54%. Evidence of efficacy in vaccinated participants cannot be inferred from these data because 53 of the 54 participants with Covid-19 who were hospitalized were unvaccinated and 1 participant was partially vaccinated. A total of 16 grade 3 or 4 adverse events (7 in the convalescent-plasma group and 9 in the controlplasma group) occurred in participants who were not hospitalized.
CONCLUSIONS In participants with Covid-19, most of whom were unvaccinated, the administration of convalescent plasma within 9 days after the onset of symptoms reduced the risk of disease progression leading to hospitalization. (Funded by the Department of Defense and others; CSSC-004 ClinicalTrials.gov number, NCT04373460.
n engl j med nejm.org 11 Convalescent Plasma for Early SARS-CoV-2 Infection the Moriah Fund; Octapharma; the Healthnetwork Foundation; and the Shear Family Foundation. Disclosure forms provided by the authors are available with the full text of this article at NEJM.org. A data sharing statement provided by the authors is available with the full text of this article at NEJM.org. We thank the trial participants who generously gave of their time and donated biologic specimens.
Appendix The authors' full names and academic degrees are as follows: David J. Sullivan, M.D., Kelly A. Gebo, M.D., M.P.H., Shmuel Shoham, M.D., Evan M. Bloch
References
Agarwal, Mukherjee, Kumar, Convalescent plasma in the management of moderate covid-19 in adults in India: open label phase II multicentre randomised controlled trial (PLACID Trial), BMJ
Bloch, Goel, Montemayor, Cohn, Tobian, Promoting access to COVID-19 convalescent plasma in lowand middle-income countries, Transfus Apher Sci
Bloch, Shoham, Casadevall, Deployment of convalescent plasma for the prevention and treatment of COVID-19, J Clin Invest
Bégin, Callum, Jamula, Convalescent plasma for hospitalized patients with COVID-19: an open-label, randomized controlled trial, Nat Med
Casadevall, Henderson, Joyner, Pirofski, SARS-CoV-2 variants and convalescent plasma: reality, fallacies, and opportunities, J Clin Invest
Casadevall, Pirofski, Joyner, The principles of antibody therapy for infectious diseases with relevance for COVID-19, mBio
Dougan, Nirula, Azizad, Bamlanivimab plus etesevimab in mild or moderate Covid-19, N Engl J Med
Díaz, Colantuoni, Hanley, Rosenblum, Improved precision in the analysis of randomized trials with survival outcomes, without assuming proportional hazards, Lifetime Data Anal
Gupta, Gonzalez-Rojas, Juarez, Early treatment for Covid-19 with SARS-CoV-2 neutralizing antibody sotrovimab, N Engl J Med
Joyner, Bruno, Klassen, Safety update: COVID-19 convalescent plasma in 20,000 hospitalized patients, Mayo Clin Proc
Joyner, Wright, Fairweather, Early safety indicators of COVID-19 convalescent plasma in 5000 patients, J Clin Invest
Klein, Pekosz, Park, Sex, age, and hospitalization drive antibody responses in a COVID-19 convalescent plasma donor population, J Clin Invest
Korley, Durkalski-Mauldin, Yeatts, Early convalescent plasma for high-risk outpatients with Covid-19, N Engl J Med
Kunze, Johnson, Van Helmond, Mortality in individuals treated with COVID-19 convalescent plasma varies with the geographic provenance of donors, Nat Commun
Li, Zhang, Hu, Effect of convalescent plasma therapy on time to clinical improvement in patients with severe and life-threatening COVID-19: a randomized clinical trial, JAMA
Libster, Marc, Wappner, Early high-titer plasma therapy to prevent severe Covid-19 in older adults, N Engl J Med
Liu, Lin, Baine, Convalescent plasma treatment of severe COVID-19: a propensity score-matched control study, Nat Med
Natarajan, Crowley, Butler, Markers of polyfunctional SARS-CoV-2 antibodies in convalescent plasma, mBio
O'donnell, Grinsztejn, Cummings, A randomized doubleblind controlled trial of convalescent plasma in adults with severe COVID-19, J Clin Invest
Piechotta, Iannizzi, Chai, Convalescent plasma or hyperimmune immunoglobulin for people with COVID-19: a living systematic review, Cochrane Database Syst Rev
Pommeret, Colomba, Bigenwald, Bamlanivimab + etesevimab therapy induces SARS-CoV-2 immune escape mutations and secondary clinical deterioration in COVID-19 patients with B-cell malignancies, Ann Oncol
Rasheed, Fatak, Hashim, The therapeutic potential of convalescent plasma therapy on treating critically-ill COVID-19 patients residing in respiratory care units in hospitals in Baghdad, Iraq, Infez Med
Tobian, Cohn, Shaz, COVID-19 convalescent plasma, Blood
Verdun, EUA 26382 -emergency use authorization of COVID-19 convalescent plasma, Section 564 of the Federal Food, Drug, and Cosmetic Act (FD&C Act
Weinreich, Sivapalasingam, Norton, REGN-COV2, a neutralizing antibody cocktail, in outpatients with Covid-19, N Engl J Med
DOI record:
{
"DOI": "10.1056/nejmoa2119657",
"ISSN": [
"0028-4793",
"1533-4406"
],
"URL": "http://dx.doi.org/10.1056/nejmoa2119657",
"alternative-id": [
"10.1056/NEJMoa2119657"
],
"author": [
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Sullivan",
"given": "David J.",
"sequence": "first"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Gebo",
"given": "Kelly A.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Shoham",
"given": "Shmuel",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-8181-9517",
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"authenticated-orcid": false,
"family": "Bloch",
"given": "Evan M.",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-2355-612X",
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"authenticated-orcid": false,
"family": "Lau",
"given": "Bryan",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Shenoy",
"given": "Aarthi G.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Mosnaim",
"given": "Giselle S.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Gniadek",
"given": "Thomas J.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Fukuta",
"given": "Yuriko",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Patel",
"given": "Bela",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Heath",
"given": "Sonya L.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Levine",
"given": "Adam C.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Meisenberg",
"given": "Barry R.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Spivak",
"given": "Emily S.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Anjan",
"given": "Shweta",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Huaman",
"given": "Moises A.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Blair",
"given": "Janis E.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Currier",
"given": "Judith S.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Paxton",
"given": "James H.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Gerber",
"given": "Jonathan M.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Petrini",
"given": "Joann R.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Broderick",
"given": "Patrick B.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Rausch",
"given": "William",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Cordisco",
"given": "Marie-Elena",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Hammel",
"given": "Jean",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Greenblatt",
"given": "Benjamin",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Cluzet",
"given": "Valerie C.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Cruser",
"given": "Daniel",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Oei",
"given": "Kevin",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Abinante",
"given": "Matthew",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Hammitt",
"given": "Laura L.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Sutcliffe",
"given": "Catherine G.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Forthal",
"given": "Donald N.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Zand",
"given": "Martin S.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Cachay",
"given": "Edward R.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Raval",
"given": "Jay S.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Kassaye",
"given": "Seble G.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Foster",
"given": "E. Colin",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Roth",
"given": "Michael",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Marshall",
"given": "Christi E.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Yarava",
"given": "Anusha",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Lane",
"given": "Karen",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "McBee",
"given": "Nichol A.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Gawad",
"given": "Amy L.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Karlen",
"given": "Nicky",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Singh",
"given": "Atika",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Ford",
"given": "Daniel E.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Jabs",
"given": "Douglas A.",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-0673-6823",
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"authenticated-orcid": false,
"family": "Appel",
"given": "Lawrence J.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Shade",
"given": "David M.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Ehrhardt",
"given": "Stephan",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Baksh",
"given": "Sheriza N.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Laeyendecker",
"given": "Oliver",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0003-3248-1761",
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"authenticated-orcid": false,
"family": "Pekosz",
"given": "Andrew",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Klein",
"given": "Sabra L.",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-9402-9167",
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"authenticated-orcid": false,
"family": "Casadevall",
"given": "Arturo",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Tobian",
"given": "Aaron A.R.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From the Department of Medicine, Division of Infectious Diseases (K.A.G., S.S.), the Department of Pathology (E.M.B., C.E.M., A.A.R.T.), the Department of Neurology, Brain Injury Outcomes Division (A.Y., K.L., N.A.M., A.L.G., N.K., D.F.H.), the Department of Ophthalmology (D.A.J.), the Welch Center for Prevention, Epidemiology and Clinical Research (L.J.A.), and the Institute for Clinical and Translational Research (D.E.F.), Johns Hopkins School of Medicine, and the Departments of Molecular Microbiology..."
}
],
"family": "Hanley",
"given": "Daniel F.",
"sequence": "additional"
}
],
"container-title": [
"New England Journal of Medicine"
],
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2022,
3,
30
]
],
"date-time": "2022-03-30T21:01:13Z",
"timestamp": 1648674073000
},
"deposited": {
"date-parts": [
[
2022,
3,
30
]
],
"date-time": "2022-03-30T21:01:24Z",
"timestamp": 1648674084000
},
"funder": [
{
"name": "HealthNetwork Foundation"
},
{
"DOI": "10.13039/100016645",
"doi-asserted-by": "publisher",
"name": "Shear Family Foundation"
},
{
"DOI": "10.13039/100009898",
"award": [
"W911QY2090012"
],
"doi-asserted-by": "publisher",
"name": "Defense Health Agency"
},
{
"DOI": "10.13039/100007500",
"doi-asserted-by": "publisher",
"name": "Bloomberg Family Foundation"
},
{
"DOI": "10.13039/100017027",
"doi-asserted-by": "publisher",
"name": "State of Maryland"
},
{
"DOI": "10.13039/100000060",
"award": [
"3R01AI152078-01S1"
],
"doi-asserted-by": "publisher",
"name": "National Institute of Allergy and Infectious Diseases"
},
{
"DOI": "10.13039/100006108",
"award": [
"U24TR001609-S3"
],
"doi-asserted-by": "publisher",
"name": "National Center for Advancing Translational Sciences"
},
{
"DOI": "10.13039/100006492",
"doi-asserted-by": "publisher",
"name": "Division of Intramural Research, National Institute of Allergy and Infectious Diseases"
},
{
"name": "Mental Wellness Foundation"
},
{
"name": "Moriah Fund"
},
{
"DOI": "10.13039/501100016300",
"doi-asserted-by": "publisher",
"name": "Octapharma"
}
],
"indexed": {
"date-parts": [
[
2022,
3,
30
]
],
"date-time": "2022-03-30T21:44:02Z",
"timestamp": 1648676642175
},
"is-referenced-by-count": 0,
"issn-type": [
{
"type": "print",
"value": "0028-4793"
},
{
"type": "electronic",
"value": "1533-4406"
}
],
"issued": {
"date-parts": [
[
2022,
3,
30
]
]
},
"language": "en",
"license": [
{
"URL": "http://www.nejmgroup.org/legal/terms-of-use.htm",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2022,
3,
30
]
],
"date-time": "2022-03-30T00:00:00Z",
"timestamp": 1648598400000
}
}
],
"link": [
{
"URL": "http://www.nejm.org/doi/pdf/10.1056/NEJMoa2119657",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "150",
"original-title": [],
"prefix": "10.1056",
"published": {
"date-parts": [
[
2022,
3,
30
]
]
},
"published-online": {
"date-parts": [
[
2022,
3,
30
]
]
},
"publisher": "Massachusetts Medical Society",
"reference-count": 25,
"references-count": 25,
"relation": {},
"resource": {
"primary": {
"URL": "http://www.nejm.org/doi/10.1056/NEJMoa2119657"
}
},
"score": 1,
"short-container-title": [
"N Engl J Med"
],
"short-title": [],
"source": "Crossref",
"subject": [
"General Medicine"
],
"subtitle": [],
"title": [
"Early Outpatient Treatment for Covid-19 with Convalescent Plasma"
],
"type": "journal-article"
}