Effectiveness of COVID-19 treatment with nirmatrelvir-ritonavir or molnupiravir among U.S. Veterans: target trial emulation studies with one-month and six-month outcomes
et al., medRxiv, doi:10.1101/2022.12.05.22283134, Dec 2022
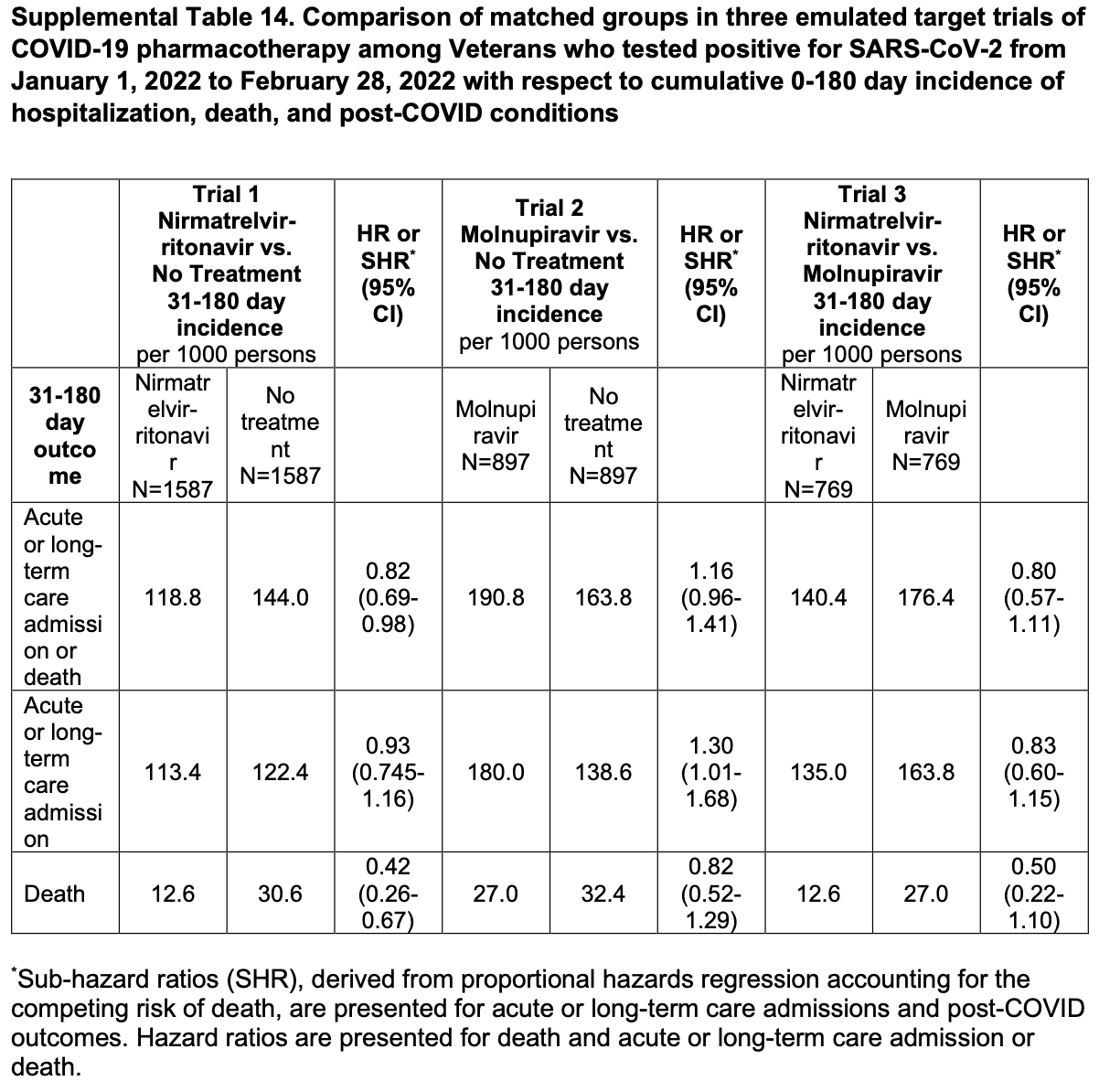
Retrospective 112,380 high-risk patients in the USA, showing lower mortality with paxlovid treatment. The title and headers of Table S14 are conflicting but the data appears to match the title.
Resistance. Variants may be resistant to paxlovid1-9. Use may promote the emergence of variants that weaken host immunity and potentially contribute to long COVID10. Confounding by contraindication. Hoertel et al. find that over 50% of patients that died had a contraindication for the use of Paxlovid11. Retrospective studies that do not exclude contraindicated patients may significantly overestimate efficacy. Black box warning. The FDA notes that severe, life-threatening, and/or fatal adverse reactions due to drug interactions have been reported in patients treated with paxlovid12. Kidney and liver injury. Studies show significantly increased risk of acute kidney injury13 and liver injury14,15. Viral rebound. Studies show significantly increased risk of replication-competent viral rebound16-18.
Standard of Care (SOC) for COVID-19 in the study country,
the USA, is very poor with very low average efficacy for approved treatments19.
Only expensive, high-profit treatments were approved for early treatment. Low-cost treatments were excluded, reducing the probability of early treatment due to access and cost barriers, and eliminating complementary and synergistic benefits seen with many low-cost treatments.
Study covers paxlovid and molnupiravir.
|
risk of death, 58.0% lower, HR 0.42, p < 0.001, treatment 20 of 1,587 (1.3%), control 48.6 of 1,587 (3.1%), NNT 55, cumulative 0-180 days, propensity score matching, day 180, Table S14.
|
|
risk of death, 78.8% lower, RR 0.21, p < 0.001, treatment 5 of 1,587 (0.3%), control 23.6 of 1,587 (1.5%), NNT 85, propensity score matching, day 30.
|
|
risk of mechanical ventilation, 48.3% lower, RR 0.52, p = 0.51, treatment 3 of 1,587 (0.2%), control 5.8 of 1,587 (0.4%), NNT 567, propensity score matching, day 30.
|
|
risk of ICU admission, 56.1% lower, RR 0.44, p = 0.03, treatment 10 of 1,587 (0.6%), control 22.8 of 1,587 (1.4%), NNT 124, propensity score matching, day 30.
|
|
risk of hospitalization, 7.0% lower, HR 0.93, p = 0.53, treatment 180 of 1,587 (11.3%), control 194.2 of 1,587 (12.2%), NNT 112, cumulative 0-180 days, propensity score matching, day 180, Table S14.
|
|
risk of hospitalization, 34.0% lower, RR 0.66, p = 0.03, treatment 43 of 1,587 (2.7%), control 65.2 of 1,587 (4.1%), NNT 71, propensity score matching, day 30.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
1.
Zhou et al., Nirmatrelvir-resistant SARS-CoV-2 variants with high fitness in an infectious cell culture system, Science Advances, doi:10.1126/sciadv.add7197.
2.
Moghadasi et al., Rapid resistance profiling of SARS-CoV-2 protease inhibitors, npj Antimicrobials and Resistance, doi:10.1038/s44259-023-00009-0.
3.
Jochmans et al., The Substitutions L50F, E166A, and L167F in SARS-CoV-2 3CLpro Are Selected by a Protease Inhibitor In Vitro and Confer Resistance To Nirmatrelvir, mBio, doi:10.1128/mbio.02815-22.
4.
Lopez et al., SARS-CoV-2 Resistance to Small Molecule Inhibitors, Current Clinical Microbiology Reports, doi:10.1007/s40588-024-00229-6.
5.
Zvornicanin et al., Molecular Mechanisms of Drug Resistance and Compensation in SARS-CoV-2 Main Protease: The Interplay Between E166 and L50, bioRxiv, doi:10.1101/2025.01.24.634813.
6.
Vukovikj et al., Impact of SARS-CoV-2 variant mutations on susceptibility to monoclonal antibodies and antiviral drugs: a non-systematic review, April 2022 to October 2024, Eurosurveillance, doi:10.2807/1560-7917.ES.2025.30.10.2400252.
7.
Deschenes et al., Functional and structural characterization of treatment-emergent nirmatrelvir resistance mutations at low frequencies in the main protease (Mpro) reveals a unique evolutionary route for SARS-CoV-2 to gain resistance, The Journal of Infectious Diseases, doi:10.1093/infdis/jiaf294.
8.
Zhou (B) et al., SARS-CoV-2 Mpro inhibitor ensitrelvir: asymmetrical cross-resistance with nirmatrelvir and emerging resistance hotspots, Emerging Microbes & Infections, doi:10.1080/22221751.2025.2552716.
9.
Chen et al., SARS-CoV-2 3CLpro mutations T21I and E166A confer differential resistance to simnotrelvir, bofutrelvir, and ensitrelvir, Journal of Virology, doi:10.1128/jvi.02223-25.
10.
Thomas et al., Nirmatrelvir-Resistant Mutations in SARS-CoV-2 Mpro Enhance Host Immune Evasion via Cleavage of NF-κB Essential Modulator, bioRxiv, doi:10.1101/2024.10.18.619137.
11.
Hoertel et al., Prevalence of Contraindications to Nirmatrelvir-Ritonavir Among Hospitalized Patients With COVID-19 at Risk for Progression to Severe Disease, JAMA Network Open, doi:10.1001/jamanetworkopen.2022.42140.
12.
FDA, Fact sheet for healthcare providers: emergency use authorization for paxlovid, www.fda.gov/media/155050/download.
13.
Kamo et al., Association of Antiviral Drugs for the Treatment of COVID-19 With Acute Renal Failure, In Vivo, doi:10.21873/invivo.13637.
14.
Wang et al., Development and validation of a nomogram to assess the occurrence of liver dysfunction in patients with COVID-19 pneumonia in the ICU, BMC Infectious Diseases, doi:10.1186/s12879-025-10684-1.
15.
Siby et al., Temporal Trends in Serious Adverse Events Associated with Oral Antivirals During the COVID-19 Pandemic: Insights from the FAERS Database (2020–2023), Open Forum Infectious Diseases, doi:10.1093/ofid/ofaf695.1825.
16.
Edelstein et al., SARS-CoV-2 virologic rebound with nirmatrelvir-ritonavir therapy, medRxiv, doi:10.1101/2023.06.23.23288598.
17.
Shah et al., SARS-CoV-2 infectious shedding and rebound among adults with and without oral antiviral use: two case-ascertained prospective household studies, The Lancet Microbe, doi:10.1016/j.lanmic.2025.101227.
Bajema et al., 6 Dec 2022, retrospective, USA, preprint, median age 67.0, 18 authors, study period 1 January, 2022 - 28 February, 2022.
Effectiveness of COVID-19 treatment with nirmatrelvir-ritonavir or molnupiravir among U.S. Veterans: target trial emulation studies with one-month and six-month outcomes
doi:10.1101/2022.12.05.22283134
Background: Information about the effectiveness of oral antivirals in preventing short-and longterm COVID-19-related outcomes during the Omicron surge is limited. We sought to determine the effectiveness of nirmatrelvir-ritonavir and molnupiravir for the outpatient treatment of COVID-19. Methods: We conducted three retrospective target trial emulation studies comparing matched patient cohorts who received nirmatrelvir-ritonavir versus no treatment, molnupiravir versus no treatment, and nirmatrelvir-ritonavir versus molnupiravir in the Veterans Health Administration (VHA). Participants were Veterans in VHA care at risk for severe COVID-19 who tested positive for SARS-CoV-2 in the outpatient setting during January and February 2022. Primary outcomes included all-cause 30-day hospitalization or death and 31-180-day incidence of acute or longterm care admission, death, or post-COVID-19 conditions. For 30-day outcomes, we calculated unadjusted risk rates, risk differences, and risk ratios. For 31-180-day outcomes, we used unadjusted time-to-event analyses. Results: Participants were 90% male with median age 67 years and 26% unvaccinated. Compared to matched untreated controls, nirmatrelvir-ritonavir-treated participants (N=1,587) had a lower 30-day risk of hospitalization (27.10/1000 versus 41.06/1000, risk difference [RD] -13.97, 95% CI -23.85 to -4.09) and death (3.15/1000 versus 14.86/1000, ). Among persons who were alive at day 31, further significant reductions in 31-180-day incidence of hospitalization (sub-hazard ratio 1.07, 95% CI 0.83 to 1.37) or death (hazard ratio 0.61, 95% CI 0.35 to 1.08) were not observed. Molnupiravir-treated participants aged ≥65 years (n=543) had a lower combined 30-day risk of hospitalization or death (55.25/1000 versus 82.35/1000, . A statistically significant difference in 30-day or 31-180-day risk of hospitalization or death was not observed between matched nirmatrelvir-or molnupiravir-treated participants. Incidence of most post-COVID conditions was similar across comparison groups. Conclusions: Nirmatrelvir-ritonavir was highly effective in preventing 30-day hospitalization and death. Short-term benefit from molnupiravir was observed in older groups. Significant reductions in adverse outcomes from 31-180 days were not observed with either antiviral. for use under a CC0 license.
References
Adjei, Hong, Molinari, Bull-Otterson, Ajani et al., Mortality Risk Among Patients Hospitalized Primarily for COVID-19 During the Omicron and Delta Variant Pandemic Periods -United States, April 2020, MMWR Morb Mortal Wkly Rep
Aggarwal, Molina, Beaty, Bennett, Carlson et al., Real-world Use of Nirmatrelvir-Ritonavir in COVID-19 Outpatients During the Emergence of Omicron Variants BA, BA2
Al-Aly, Xie, Bowe, High-dimensional characterization of post-acute sequelae of COVID-19, Nature
Arbel, Sagy, Hoshen, Battat, Lavie et al., Nirmatrelvir Use and Severe Covid-19 Outcomes during the Omicron Surge, N Engl J Med
Austin, Cafri, Variance estimation when using propensity-score matching with replacement with survival or time-to-event outcomes, Stat Med
Bajema, Wang, Hynes, Rowneki, Hickok et al., Early Adoption of Anti-SARS-CoV-2 Pharmacotherapies Among US Veterans With Mild to Moderate COVID-19, January and February, JAMA Network Open
Bernal, Da Silva, Musungaie, Kovalchuk, Gonzalez et al., Molnupiravir for Oral Treatment of Covid-19 in Nonhospitalized Patients, N Engl J Med
Bernal, Da Silva, Musungaie, Kovalchuk, Gonzalez et al., Molnupiravir for Oral Treatment of Covid-19 in Nonhospitalized Patients, N Engl J Med
Butler, None
Dickerman, Gerlovin, Madenci, Kurgansky, Ferolito et al., Comparative Effectiveness of BNT162b2 and mRNA-1273 Vaccines in U.S. Veterans, N Engl J Med
Dryden-Peterson, Kim, Kim, Caniglia, Lennes et al., Nirmatrelvir plus ritonavir for early COVID-19 and hospitalization in a large US health system
Hammond, Leister-Tebbe, Gardner, Abreu, Wisemandle, Oral Nirmatrelvir for High-Risk, Nonhospitalized Adults with Covid-19, N Engl J Med
Hernán, Robins, Using Big Data to Emulate a Target Trial When a Randomized Trial Is Not Available, Am J Epidemiol
Ioannou, Locke, Green, Berry, Comparison of Moderna versus Pfizer-BioNTech COVID-19 vaccine outcomes: A target trial emulation study in the U.S. Veterans Affairs healthcare system, EClinicalMedicine
Ioannou, Locke, Green, Berry, Hare et al., Risk Factors for Hospitalization, Mechanical Ventilation, or Death Among 10 131 US Veterans With SARS-CoV-2 Infection, JAMA Netw Open
Ioannou, Locke, Hare, Bohnert, Boyko et al., COVID-19 Vaccination Effectiveness Against Infection or Death in a National U.S. Health Care System : A Target Trial Emulation Study, Ann Intern Med
Labrecque, Swanson, Target trial emulation: teaching epidemiology and beyond, Eur J Epidemiol
Lance, Csp-Cerc, VA Connecticut Healthcare System
Lewnard, Malden, Hong, Puzniak, Kim et al., Effectiveness of nirmatrelvir-ritonavir against hospital admission: a matched cohort study in a large US healthcare system
Maffucci, Va, Nicholas, Smith, Mph, VA Connecticut Healthcare System
Najjar-Debbiny, Gronich, Weber, Khoury, Amar et al., Effectiveness of Paxlovid in Reducing Severe COVID-19 and Mortality in High Risk Patients, Clin Infect Dis
Osborne, Veigulis, Arreola, Röösli, Curtin, Automated EHR score to predict COVID-19 outcomes at US Department of Veterans Affairs, PLoS One
Randall, Stern, Su, Pageshttps
Wong, Au, Lau, Lau, Cowling et al., Real-world effectiveness of molnupiravir and nirmatrelvir plus ritonavir against mortality, hospitalisation, and in-hospital outcomes among community-dwelling, ambulatory patients with confirmed SARS-CoV-2 infection during the omicron wave in Hong Kong: an observational study, Lancet
Xie, Bowe, Al-Aly, Burdens of post-acute sequelae of COVID-19 by severity of acute infection, demographics and health status, Nat Commun
Xie, Choi, Al-Aly, Nirmatrelvir and the Risk of Post-Acute Sequelae of COVID-19, medRxiv
Xie, Xu, Al-Aly, Risks of mental health outcomes in people with covid-19: cohort study, BMJ
Xie, Xu, Bowe, Al-Aly, Long-term cardiovascular outcomes of COVID-19, Nat Med
Yip, Lui, Lai, Wong, Tse et al., Impact of the use of oral antiviral agents on the risk of hospitalization in community COVID-19 patients, Clin Infect Dis
DOI record:
{
"DOI": "10.1101/2022.12.05.22283134",
"URL": "http://dx.doi.org/10.1101/2022.12.05.22283134",
"abstract": "<jats:p>Background: Information about the effectiveness of oral antivirals in preventing short- and long-term COVID-19-related outcomes during the Omicron surge is limited. We sought to determine the effectiveness of nirmatrelvir-ritonavir and molnupiravir for the outpatient treatment of COVID-19. Methods: We conducted three retrospective target trial emulation studies comparing matched patient cohorts who received nirmatrelvir-ritonavir versus no treatment, molnupiravir versus no treatment, and nirmatrelvir-ritonavir versus molnupiravir in the Veterans Health Administration (VHA). Participants were Veterans in VHA care at risk for severe COVID-19 who tested positive for SARS-CoV-2 in the outpatient setting during January and February 2022. Primary outcomes included all-cause 30-day hospitalization or death and 31-180-day incidence of acute or long-term care admission, death, or post-COVID-19 conditions. For 30-day outcomes, we calculated unadjusted risk rates, risk differences, and risk ratios. For 31-180-day outcomes, we used unadjusted time-to-event analyses. Results: Participants were 90% male with median age 67 years and 26% unvaccinated. Compared to matched untreated controls, nirmatrelvir-ritonavir-treated participants (N=1,587) had a lower 30-day risk of hospitalization (27.10/1000 versus 41.06/1000, risk difference [RD] -13.97, 95% CI -23.85 to -4.09) and death (3.15/1000 versus 14.86/1000, RD -11.71, 95% CI -16.07 to -7.35). Among persons who were alive at day 31, further significant reductions in 31-180-day incidence of hospitalization (sub-hazard ratio 1.07, 95% CI 0.83 to 1.37) or death (hazard ratio 0.61, 95% CI 0.35 to 1.08) were not observed. Molnupiravir-treated participants aged ≥65 years (n=543) had a lower combined 30-day risk of hospitalization or death (55.25/1000 versus 82.35/1000, RD -27.10, 95% CI -50.63 to -3.58). A statistically significant difference in 30-day or 31-180-day risk of hospitalization or death was not observed between matched nirmatrelvir- or molnupiravir-treated participants. Incidence of most post-COVID conditions was similar across comparison groups. Conclusions: Nirmatrelvir-ritonavir was highly effective in preventing 30-day hospitalization and death. Short-term benefit from molnupiravir was observed in older groups. Significant reductions in adverse outcomes from 31-180 days were not observed with either antiviral.</jats:p>",
"accepted": {
"date-parts": [
[
2022,
12,
6
]
]
},
"author": [
{
"ORCID": "http://orcid.org/0000-0002-3229-5590",
"affiliation": [],
"authenticated-orcid": false,
"family": "Bajema",
"given": "Kristina L.",
"sequence": "first"
},
{
"affiliation": [],
"family": "Berry",
"given": "Kristin",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-1261-0098",
"affiliation": [],
"authenticated-orcid": false,
"family": "Streja",
"given": "Elani",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Rajeevan",
"given": "Nallakkandi",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Li",
"given": "Yuli",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Yan",
"given": "Lei",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-7924-8372",
"affiliation": [],
"authenticated-orcid": false,
"family": "Cunningham",
"given": "Francesca",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-6436-7157",
"affiliation": [],
"authenticated-orcid": false,
"family": "Hynes",
"given": "Denise M.",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-9213-5134",
"affiliation": [],
"authenticated-orcid": false,
"family": "Rowneki",
"given": "Mazhgan",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Bohnert",
"given": "Amy",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-3695-192X",
"affiliation": [],
"authenticated-orcid": false,
"family": "Boyko",
"given": "Edward J.",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-4226-9310",
"affiliation": [],
"authenticated-orcid": false,
"family": "Iwashyna",
"given": "Theodore J.",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0003-1765-5938",
"affiliation": [],
"authenticated-orcid": false,
"family": "Maciejewski",
"given": "Matthew L.",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-8896-2487",
"affiliation": [],
"authenticated-orcid": false,
"family": "Osborne",
"given": "Thomas F.",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-7439-6322",
"affiliation": [],
"authenticated-orcid": false,
"family": "Viglianti",
"given": "Elizabeth M.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Aslan",
"given": "Mihaela",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-1217-0002",
"affiliation": [],
"authenticated-orcid": false,
"family": "Huang",
"given": "Grant D.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ioannou",
"given": "George N.",
"sequence": "additional"
}
],
"container-title": [],
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2022,
12,
7
]
],
"date-time": "2022-12-07T05:15:22Z",
"timestamp": 1670390122000
},
"deposited": {
"date-parts": [
[
2022,
12,
7
]
],
"date-time": "2022-12-07T05:15:22Z",
"timestamp": 1670390122000
},
"group-title": "Infectious Diseases (except HIV/AIDS)",
"indexed": {
"date-parts": [
[
2022,
12,
7
]
],
"date-time": "2022-12-07T06:15:11Z",
"timestamp": 1670393711776
},
"institution": [
{
"name": "medRxiv"
}
],
"is-referenced-by-count": 0,
"issued": {
"date-parts": [
[
2022,
12,
6
]
]
},
"link": [
{
"URL": "https://syndication.highwire.org/content/doi/10.1101/2022.12.05.22283134",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "246",
"original-title": [],
"posted": {
"date-parts": [
[
2022,
12,
6
]
]
},
"prefix": "10.1101",
"published": {
"date-parts": [
[
2022,
12,
6
]
]
},
"publisher": "Cold Spring Harbor Laboratory",
"reference-count": 0,
"references-count": 0,
"relation": {},
"resource": {
"primary": {
"URL": "http://medrxiv.org/lookup/doi/10.1101/2022.12.05.22283134"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subtitle": [],
"subtype": "preprint",
"title": "Effectiveness of COVID-19 treatment with nirmatrelvir-ritonavir or molnupiravir among U.S. Veterans: target trial emulation studies with one-month and six-month outcomes",
"type": "posted-content"
}