Inhibition of SARS-CoV-2 3CLpro by chemically modified tyrosinase from Agaricus bisporus
et al., RSC Medicinal Chemistry, doi:10.1039/D4MD00289J, Sep 2024
Quercetin for COVID-19
36th treatment shown to reduce risk in
January 2022, now with p = 0.0018 from 9 studies.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
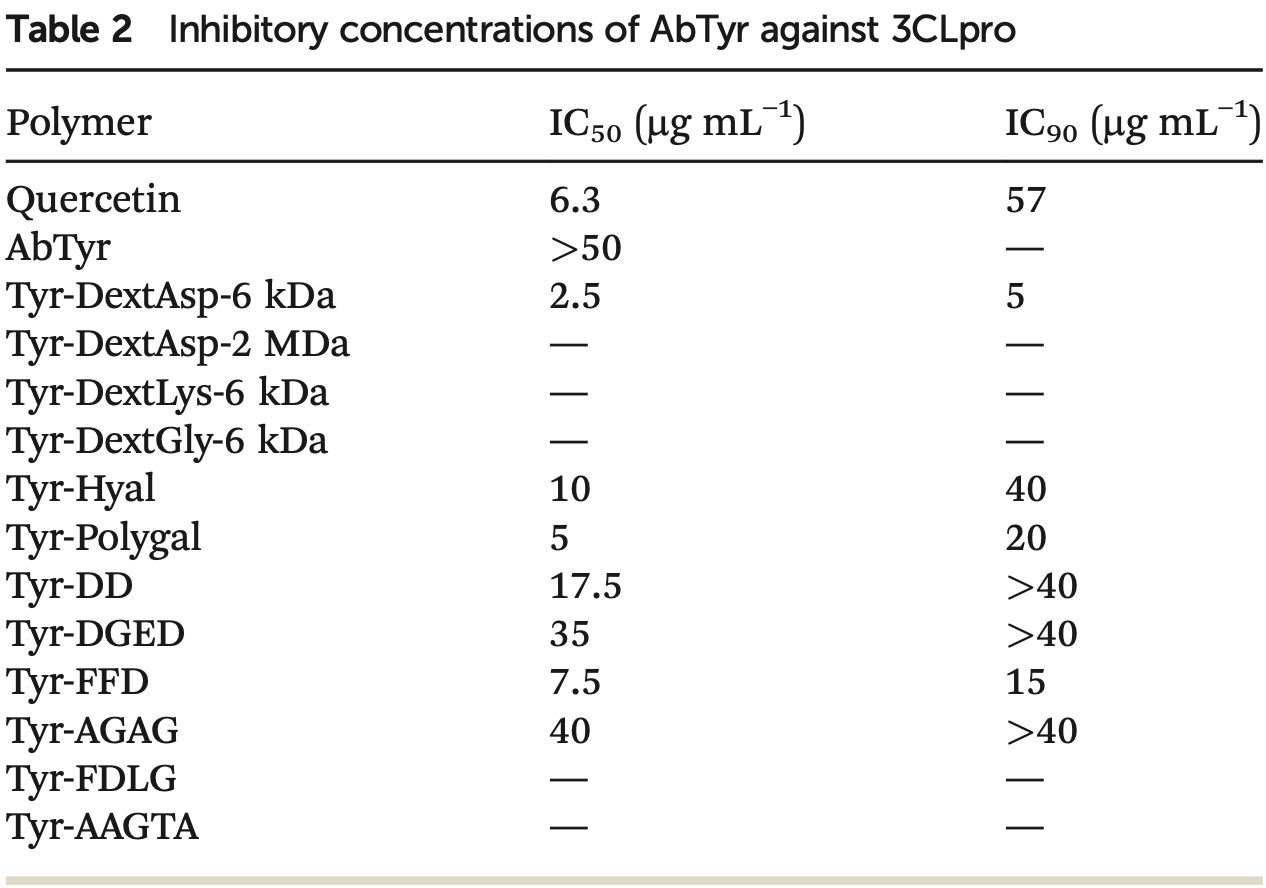
In vitro study showing that a purified tyrosinase enzyme from the mushroom Agaricus bisporus, chemically modified with polymers, inhibited SARS-CoV-2 3CLpro protease activity and viral replication in Vero E6 cells. The tyrosinase bioconjugate with dextran-aspartic acid (6 kDa) polymer showed the highest 3CLpro inhibition, with an IC50 of 2.5 μg/ml and IC90 of 5 μg/ml. This bioconjugate reduced SARS-CoV-2 viral load by 88% at 100 μg/mL after 48 hours in Vero E6 cells, with no observed cytotoxicity. Authors propose that these chemically modified tyrosinase enzymes could be promising antiviral protein therapeutics against SARS-CoV-2. Quercetin was used as a positive control and showed an IC50 of 6.3 μg/ml.
Bioavailability. Quercetin has low bioavailability and studies typically use advanced formulations to improve bioavailability which may be required to reach therapeutic concentrations.
92 preclinical studies support the efficacy of quercetin for COVID-19:
In silico studies predict inhibition of SARS-CoV-2, or minimization of side effects, with quercetin or metabolites via binding to the spikeA,12,13,19,20,33,35,36,38,41,49,50,52,53,76 (and specifically the receptor binding domainB,9), MproC,8,9,12,13,17,19,21,23,25,27,29,31,34,35,38,41,45,47-49,53-56,73 , RNA-dependent RNA polymeraseD,9,11-13,19,43 , PLproE,13,48,56 , ACE2F,28,33,34,38,39,48,52 , TMPRSS2G,33, nucleocapsidH,13, helicaseI,13,40,45 , endoribonucleaseJ,50, NSP16/10K,16, cathepsin LL,37, Wnt-3M,33, FZDN,33, LRP6O,33, ezrinP,51, ADRPQ,49, NRP1R,52, EP300S,26, PTGS2T,34, HSP90AA1U,26,34 , matrix metalloproteinase 9V,42, IL-6W,32,46 , IL-10X,32, VEGFAY,46, and RELAZ,46 proteins, and inhibition of spike-ACE2 interactionAA,10.
In vitro studies demonstrate inhibition of the MproC,25,59,64,72 protein, and inhibition of spike-ACE2 interactionAA,60.
In vitro studies demonstrate efficacy in Calu-3AB,63, A549AC,32, HEK293-ACE2+AD,71, Huh-7AE,36, Caco-2AF,62, Vero E6AG,30,53,62 , mTECAH,65, RAW264.7AI,65, and HLMECAJ,10 cells.
Animal studies demonstrate efficacy in K18-hACE2 miceAK,68, db/db miceAL,65,75 , BALB/c miceAM,74, and rats30.
Quercetin reduced proinflammatory cytokines and protected lung and kidney tissue against LPS-induced damage in mice74, inhibits LPS-induced cytokine storm by modulating key inflammatory and antioxidant pathways in macrophages15, may block ACE2-spike interaction and NLRP3 inflammasome, limiting viral entry and inflammation6, upregulates the SIRT1/AMPK axis to inhibit oxidative injury and accelerate viral clearance77, inhibits SARS-CoV-2 ORF3a ion channel activity, which contributes to viral pathogenicity and cytotoxicity67, may alleviate COVID-19 ARDS via inhibition of EGFR and JAK2 inflammatory targets2, may destabilize the Spike protein, IL-6R, and integrins via conserved residues, blocking viral entry, hyperinflammation, and platelet aggregation78, and may reduce COVID-19 neuroinflammation and cognitive dysfunction through anti-inflammatory mechanisms and neuroprotective effects1.
1.
Xie et al., Identification of key genes modules linking brain aging signatures and COVID-19-associated cognitive impairment, Virus Research, doi:10.1016/j.virusres.2026.199707.
2.
Gupta et al., Harnessing phytoconstituents to treat COVID-19 triggered acute respiratory distress syndrome: Insights from network pharmacology, and molecular modeling, Phytochemistry Letters, doi:10.1016/j.phytol.2025.104105.
3.
Sun et al., Feasibility of the inhibitor development for SARS-CoV-2: a systematic approach for drug design, Journal of Molecular Modeling, doi:10.1007/s00894-025-06541-2.
4.
Torabfam et al., Improving quercetin solubility via structural modification enhances dual-target coronavirus entry: an integrated in-vitro and in-silico study, Scientific Reports, doi:10.1038/s41598-025-27374-2.
5.
Abdelhameed et al., Phytochemical and antiviral investigation of Cynanchum acutum L. extract and derived semi-synthetic analogs targeting SARS-CoV-2 main protease, Future Journal of Pharmaceutical Sciences, doi:10.1186/s43094-025-00907-2.
6.
Manikyam et al., INP-Guided Network Pharmacology Discloses Multi-Target Therapeutic Strategy Against Cytokine and IgE Storms in the SARS-CoV-2 NB.1.8.1 Variant, Research Square, doi:10.21203/rs.3.rs-6819274/v1.
7.
Makoana et al., Integration of metabolomics and chemometrics with in-silico and in-vitro approaches to unravel SARS-Cov-2 inhibitors from South African plants, PLOS ONE, doi:10.1371/journal.pone.0320415.
8.
Bano et al., Biochemical Screening of Phytochemicals and Identification of Scopoletin as a Potential Inhibitor of SARS-CoV-2 Mpro, Revealing Its Biophysical Impact on Structural Stability, Viruses, doi:10.3390/v17030402.
9.
Rajamanickam et al., Exploring the Potential of Siddha Formulation MilagaiKudineer-Derived Phytotherapeutics Against SARS-CoV-2: An In-Silico Investigation for Antiviral Intervention, Journal of Pharmacy and Pharmacology Research, doi:10.26502/fjppr.0105.
10.
Moharram et al., Secondary metabolites of Alternaria alternate appraisal of their SARS-CoV-2 inhibitory and anti-inflammatory potentials, PLOS ONE, doi:10.1371/journal.pone.0313616.
11.
Metwaly et al., Integrated study of Quercetin as a potent SARS-CoV-2 RdRp inhibitor: Binding interactions, MD simulations, and In vitro assays, PLOS ONE, doi:10.1371/journal.pone.0312866.
12.
Al balawi et al., Assessing multi-target antiviral and antioxidant activities of natural compounds against SARS-CoV-2: an integrated in vitro and in silico study, Bioresources and Bioprocessing, doi:10.1186/s40643-024-00822-z.
13.
Haque et al., Exploring potential therapeutic candidates against COVID-19: a molecular docking study, Discover Molecules, doi:10.1007/s44345-024-00005-5.
14.
Pan et al., Decoding the mechanism of Qingjie formula in the prevention of COVID-19 based on network pharmacology and molecular docking, Heliyon, doi:10.1016/j.heliyon.2024.e39167.
15.
Xu et al., Quercetin inhibited LPS-induced cytokine storm by interacting with the AKT1-FoxO1 and Keap1-Nrf2 signaling pathway in macrophages, Scientific Reports, doi:10.1038/s41598-024-71569-y.
16.
Tamil Selvan et al., Computational Investigations to Identify Potent Natural Flavonoid Inhibitors of the Nonstructural Protein (NSP) 16/10 Complex Against Coronavirus, Cureus, doi:10.7759/cureus.68098.
17.
Sunita et al., Characterization of Phytochemical Inhibitors of the COVID-19 Primary Protease Using Molecular Modelling Approach, Asian Journal of Microbiology and Biotechnology, doi:10.56557/ajmab/2024/v9i28800.
18.
Wu et al., Biomarkers Prediction and Immune Landscape in Covid-19 and “Brain Fog”, Elsevier BV, doi:10.2139/ssrn.4897774.
19.
Raman et al., Phytoconstituents of Citrus limon (Lemon) as Potential Inhibitors Against Multi Targets of SARS‐CoV‐2 by Use of Molecular Modelling and In Vitro Determination Approaches, ChemistryOpen, doi:10.1002/open.202300198.
20.
Asad et al., Exploring the antiviral activity of Adhatoda beddomei bioactive compounds in interaction with coronavirus spike protein, Archives of Medical Reports, 1:1, archmedrep.com/index.php/amr/article/view/3.
21.
Irfan et al., Phytoconstituents of Artemisia Annua as potential inhibitors of SARS CoV2 main protease: an in silico study, BMC Infectious Diseases, doi:10.1186/s12879-024-09387-w.
22.
Yuan et al., Network pharmacology and molecular docking reveal the mechanisms of action of Panax notoginseng against post-COVID-19 thromboembolism, Review of Clinical Pharmacology and Pharmacokinetics - International Edition, doi:10.61873/DTFA3974.
23.
Nalban et al., Targeting COVID-19 (SARS-CoV-2) main protease through phytochemicals of Albizia lebbeck: molecular docking, molecular dynamics simulation, MM–PBSA free energy calculations, and DFT analysis, Journal of Proteins and Proteomics, doi:10.1007/s42485-024-00136-w.
24.
Zhou et al., Bioinformatics and system biology approaches to determine the connection of SARS-CoV-2 infection and intrahepatic cholangiocarcinoma, PLOS ONE, doi:10.1371/journal.pone.0300441.
25.
Waqas et al., Discovery of Novel Natural Inhibitors Against SARS-CoV-2 Main Protease: A Rational Approach to Antiviral Therapeutics, Current Medicinal Chemistry, doi:10.2174/0109298673292839240329081008.
26.
Hasanah et al., Decoding the therapeutic potential of empon-empon: a bioinformatics expedition unraveling mechanisms against COVID-19 and atherosclerosis, International Journal of Applied Pharmaceutics, doi:10.22159/ijap.2024v16i2.50128.
27.
Shaik et al., Computational identification of selected bioactive compounds from Cedrus deodara as inhibitors against SARS-CoV-2 main protease: a pharmacoinformatics study, Indian Drugs, doi:10.53879/id.61.02.13859.
28.
Wang et al., Investigating the Mechanism of Qu Du Qiang Fei 1 Hao Fang Formula against Coronavirus Disease 2019 Based on Network Pharmacology Method, World Journal of Traditional Chinese Medicine, doi:10.4103/2311-8571.395061.
29.
Singh et al., Unlocking the potential of phytochemicals in inhibiting SARS-CoV-2 M Pro protein - An in-silico and cell-based approach, Research Square, doi:10.21203/rs.3.rs-3888947/v1.
30.
El-Megharbel et al., Chemical and spectroscopic characterization of (Artemisinin/Quercetin/ Zinc) novel mixed ligand complex with assessment of its potent high antiviral activity against SARS-CoV-2 and antioxidant capacity against toxicity induced by acrylamide in male rats, PeerJ, doi:10.7717/peerj.15638.
31.
Akinwumi et al., Evaluation of therapeutic potentials of some bioactive compounds in selected African plants targeting main protease (Mpro) in SARS-CoV-2: a molecular docking study, Egyptian Journal of Medical Human Genetics, doi:10.1186/s43042-023-00456-4.
32.
Yang et al., Active ingredient and mechanistic analysis of traditional Chinese medicine formulas for the prevention and treatment of COVID-19: Insights from bioinformatics and in vitro experiments, Medicine, doi:10.1097/MD.0000000000036238.
33.
Chandran et al., Molecular docking analysis of quercetin with known CoVid-19 targets, Bioinformation, doi:10.6026/973206300191081.
34.
Qin et al., Exploring the bioactive compounds of Feiduqing formula for the prevention and management of COVID-19 through network pharmacology and molecular docking, Medical Data Mining, doi:10.53388/MDM202407003.
35.
Moschovou et al., Exploring the Binding Effects of Natural Products and Antihypertensive Drugs on SARS-CoV-2: An In Silico Investigation of Main Protease and Spike Protein, International Journal of Molecular Sciences, doi:10.3390/ijms242115894.
36.
Pan (B) et al., Quercetin: A promising drug candidate against the potential SARS-CoV-2-Spike mutants with high viral infectivity, Computational and Structural Biotechnology Journal, doi:10.1016/j.csbj.2023.10.029.
37.
Ahmed et al., Evaluation of the Effect of Zinc, Quercetin, Bromelain and Vitamin C on COVID-19 Patients, International Journal of Diabetes Management, doi:10.61797/ijdm.v2i2.259.
38.
Thapa et al., In-silico Approach for Predicting the Inhibitory Effect of Home Remedies on Severe Acute Respiratory Syndrome Coronavirus-2, Makara Journal of Science, doi:10.7454/mss.v27i3.1609.
39.
Alkafaas et al., A study on the effect of natural products against the transmission of B.1.1.529 Omicron, Virology Journal, doi:10.1186/s12985-023-02160-6.
40.
Singh (B) et al., Flavonoids as Potent Inhibitor of SARS-CoV-2 Nsp13 Helicase: Grid Based Docking Approach, Middle East Research Journal of Pharmaceutical Sciences, doi:10.36348/merjps.2023.v03i04.001.
41.
Mandal et al., In silico anti-viral assessment of phytoconstituents in a traditional (Siddha Medicine) polyherbal formulation – Targeting Mpro and pan-coronavirus post-fusion Spike protein, Journal of Traditional and Complementary Medicine, doi:10.1016/j.jtcme.2023.07.004.
42.
Sai Ramesh et al., Computational analysis of the phytocompounds of Mimusops elengi against spike protein of SARS CoV2 – An Insilico model, International Journal of Biological Macromolecules, doi:10.1016/j.ijbiomac.2023.125553.
43.
Corbo et al., Inhibitory potential of phytochemicals on five SARS-CoV-2 proteins: in silico evaluation of endemic plants of Bosnia and Herzegovina, Biotechnology & Biotechnological Equipment, doi:10.1080/13102818.2023.2222196.
44.
Azmi et al., Utilization of quercetin flavonoid compounds in onion (Allium cepa L.) as an inhibitor of SARS-CoV-2 spike protein against ACE2 receptors, 11th International Seminar on New Paradigm and Innovation on Natural Sciences and its Application, doi:10.1063/5.0140285.
45.
Alanzi et al., Structure-based virtual identification of natural inhibitors of SARS-CoV-2 and its Delta and Omicron variant proteins, Future Virology, doi:10.2217/fvl-2022-0184.
46.
Yang (B) et al., In silico evidence implicating novel mechanisms of Prunella vulgaris L. as a potential botanical drug against COVID-19-associated acute kidney injury, Frontiers in Pharmacology, doi:10.3389/fphar.2023.1188086.
47.
Wang (B) et al., Computational Analysis of Lianhua Qingwen as an Adjuvant Treatment in Patients with COVID-19, Society of Toxicology Conference, 2023, www.researchgate.net/publication/370491709_Y_Wang_A_E_Tan_O_Chew_A_Hsueh_and_D_E_Johnson_2023_Computational_Analysis_of_Lianhua_Qingwen_as_an_Adjuvant_Treatment_in_Patients_with_COVID-19_Toxicologist_1921_507.
48.
Ibeh et al., Computational studies of potential antiviral compounds from some selected Nigerian medicinal plants against SARS-CoV-2 proteins, Informatics in Medicine Unlocked, doi:10.1016/j.imu.2023.101230.
49.
Nguyen et al., The Potential of Ameliorating COVID-19 and Sequelae From Andrographis paniculata via Bioinformatics, Bioinformatics and Biology Insights, doi:10.1177/11779322221149622.
50.
Alavi et al., Interaction of Epigallocatechin Gallate and Quercetin with Spike Glycoprotein (S-Glycoprotein) of SARS-CoV-2: In Silico Study, Biomedicines, doi:10.3390/biomedicines10123074.
51.
Chellasamy et al., Docking and molecular dynamics studies of human ezrin protein with a modelled SARS-CoV-2 endodomain and their interaction with potential invasion inhibitors, Journal of King Saud University - Science, doi:10.1016/j.jksus.2022.102277.
52.
Şimşek et al., In silico identification of SARS-CoV-2 cell entry inhibitors from selected natural antivirals, Journal of Molecular Graphics and Modelling, doi:10.1016/j.jmgm.2021.108038.
53.
Kandeil et al., Bioactive Polyphenolic Compounds Showing Strong Antiviral Activities against Severe Acute Respiratory Syndrome Coronavirus 2, Pathogens, doi:10.3390/pathogens10060758.
54.
Rehman et al., Natural Compounds as Inhibitors of SARS-CoV-2 Main Protease (3CLpro): A Molecular Docking and Simulation Approach to Combat COVID-19, Current Pharmaceutical Design, doi:10.2174/1381612826999201116195851.
55.
Sekiou et al., In-Silico Identification of Potent Inhibitors of COVID-19 Main Protease (Mpro) and Angiotensin Converting Enzyme 2 (ACE2) from Natural Products: Quercetin, Hispidulin, and Cirsimaritin Exhibited Better Potential Inhibition than Hydroxy-Chloroquine Against COVID-19 Main Protease Active Site and ACE2, ChemRxiv, doi:10.26434/chemrxiv.12181404.v1.
56.
Zhang et al., In silico screening of Chinese herbal medicines with the potential to directly inhibit 2019 novel coronavirus, Journal of Integrative Medicine, doi:10.1016/j.joim.2020.02.005.
57.
Sisti et al., Evaluation of respiratory virus transmissibility and resilience from fomites: the case of 11 SARS-CoV-2 clinical isolates, Applied and Environmental Microbiology, doi:10.1128/aem.00774-25.
58.
Spinelli et al., Amphibian‐Derived Peptides as Natural Inhibitors of SARS‐CoV‐2 Main Protease (Mpro): A Combined In Vitro and In Silico Approach, Chemistry & Biodiversity, doi:10.1002/cbdv.202403202.
59.
Aguilera-Rodriguez et al., Inhibition of SARS-CoV-2 3CLpro by chemically modified tyrosinase from Agaricus bisporus, RSC Medicinal Chemistry, doi:10.1039/D4MD00289J.
60.
Emam et al., Establishment of in-house assay for screening of anti-SARS-CoV-2 protein inhibitors, AMB Express, doi:10.1186/s13568-024-01739-8.
61.
Fang et al., Development of nanoparticles incorporated with quercetin and ACE2-membrane as a novel therapy for COVID-19, Journal of Nanobiotechnology, doi:10.1186/s12951-024-02435-2.
62.
Roy et al., Quercetin inhibits SARS-CoV-2 infection and prevents syncytium formation by cells co-expressing the viral spike protein and human ACE2, Virology Journal, doi:10.1186/s12985-024-02299-w.
63.
DiGuilio et al., Quercetin improves and protects Calu-3 airway epithelial barrier function, Frontiers in Cell and Developmental Biology, doi:10.3389/fcell.2023.1271201.
64.
Zhang (B) et al., Discovery of the covalent SARS‐CoV‐2 Mpro inhibitors from antiviral herbs via integrating target‐based high‐throughput screening and chemoproteomic approaches, Journal of Medical Virology, doi:10.1002/jmv.29208.
65.
Wu (B) et al., SARS-CoV-2 N protein induced acute kidney injury in diabetic db/db mice is associated with a Mincle-dependent M1 macrophage activation, Frontiers in Immunology, doi:10.3389/fimmu.2023.1264447.
66.
Xu (B) et al., Bioactive compounds from Huashi Baidu decoction possess both antiviral and anti-inflammatory effects against COVID-19, Proceedings of the National Academy of Sciences, doi:10.1073/pnas.2301775120.
67.
Fam et al., Channel activity of SARS-CoV-2 viroporin ORF3a inhibited by adamantanes and phenolic plant metabolites, Scientific Reports, doi:10.1038/s41598-023-31764-9.
68.
Aguado et al., Senolytic therapy alleviates physiological human brain aging and COVID-19 neuropathology, bioRxiv, doi:10.1101/2023.01.17.524329.
69.
Goc et al., Inhibitory effects of specific combination of natural compounds against SARS-CoV-2 and its Alpha, Beta, Gamma, Delta, Kappa, and Mu variants, European Journal of Microbiology and Immunology, doi:10.1556/1886.2021.00022.
70.
Munafò et al., Quercetin and Luteolin Are Single-digit Micromolar Inhibitors of the SARS-CoV-2 RNA-dependent RNA Polymerase, Research Square, doi:10.21203/rs.3.rs-1149846/v1.
71.
Singh (C) et al., The spike protein of SARS-CoV-2 virus induces heme oxygenase-1: Pathophysiologic implications, Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease, doi:10.1016/j.bbadis.2021.166322.
72.
Bahun et al., Inhibition of the SARS-CoV-2 3CLpro main protease by plant polyphenols, Food Chemistry, doi:10.1016/j.foodchem.2021.131594.
73.
Abian et al., Structural stability of SARS-CoV-2 3CLpro and identification of quercetin as an inhibitor by experimental screening, International Journal of Biological Macromolecules, doi:10.1016/j.ijbiomac.2020.07.235.
74.
Shaker et al., Anti-cytokine Storm Activity of Fraxin, Quercetin, and their Combination on Lipopolysaccharide-Induced Cytokine Storm in Mice: Implications in COVID-19, Iranian Journal of Medical Sciences, doi:10.30476/ijms.2023.98947.3102.
75.
Wu (C) et al., Treatment with Quercetin inhibits SARS-CoV-2 N protein-induced acute kidney injury by blocking Smad3-dependent G1 cell cycle arrest, Molecular Therapy, doi:10.1016/j.ymthe.2022.12.002.
76.
Azmi (B) et al., The role of vitamin D receptor and IL‐6 in COVID‐19, Molecular Genetics & Genomic Medicine, doi:10.1002/mgg3.2172.
a.
The trimeric spike (S) protein is a glycoprotein that mediates viral entry by binding to the host ACE2 receptor, is critical for SARS-CoV-2's ability to infect host cells, and is a target of neutralizing antibodies. Inhibition of the spike protein prevents viral attachment, halting infection at the earliest stage.
b.
The receptor binding domain is a specific region of the spike protein that binds ACE2 and is a major target of neutralizing antibodies. Focusing on the precise binding site allows highly specific disruption of viral attachment with reduced potential for off-target effects.
c.
The main protease or Mpro, also known as 3CLpro or nsp5, is a cysteine protease that cleaves viral polyproteins into functional units needed for replication. Inhibiting Mpro disrupts the SARS-CoV-2 lifecycle within the host cell, preventing the creation of new copies.
d.
RNA-dependent RNA polymerase (RdRp), also called nsp12, is the core enzyme of the viral replicase-transcriptase complex that copies the positive-sense viral RNA genome into negative-sense templates for progeny RNA synthesis. Inhibiting RdRp blocks viral genome replication and transcription.
e.
The papain-like protease (PLpro) has multiple functions including cleaving viral polyproteins and suppressing the host immune response by deubiquitination and deISGylation of host proteins. Inhibiting PLpro may block viral replication and help restore normal immune responses.
f.
The angiotensin converting enzyme 2 (ACE2) protein is a host cell transmembrane protein that serves as the cellular receptor for the SARS-CoV-2 spike protein. ACE2 is expressed on many cell types, including epithelial cells in the lungs, and allows the virus to enter and infect host cells. Inhibition may affect ACE2's physiological function in blood pressure control.
g.
Transmembrane protease serine 2 (TMPRSS2) is a host cell protease that primes the spike protein, facilitating cellular entry. TMPRSS2 activity helps enable cleavage of the spike protein required for membrane fusion and virus entry. Inhibition may especially protect respiratory epithelial cells, buy may have physiological effects.
h.
The nucleocapsid (N) protein binds and encapsulates the viral genome by coating the viral RNA. N enables formation and release of infectious virions and plays additional roles in viral replication and pathogenesis. N is also an immunodominant antigen used in diagnostic assays.
i.
The helicase, or nsp13, protein unwinds the double-stranded viral RNA, a crucial step in replication and transcription. Inhibition may prevent viral genome replication and the creation of new virus components.
j.
The endoribonuclease, also known as NendoU or nsp15, cleaves specific sequences in viral RNA which may help the virus evade detection by the host immune system. Inhibition may hinder the virus's ability to mask itself from the immune system, facilitating a stronger immune response.
k.
The NSP16/10 complex consists of non-structural proteins 16 and 10, forming a 2'-O-methyltransferase that modifies the viral RNA cap structure. This modification helps the virus evade host immune detection by mimicking host mRNA, making NSP16/10 a promising antiviral target.
l.
Cathepsin L is a host lysosomal cysteine protease that can prime the spike protein through an alternative pathway when TMPRSS2 is unavailable. Dual targeting of cathepsin L and TMPRSS2 may maximize disruption of alternative pathways for virus entry.
m.
Wingless-related integration site (Wnt) ligand 3 is a host signaling molecule that activates the Wnt signaling pathway, which is important in development, cell growth, and tissue repair. Some studies suggest that SARS-CoV-2 infection may interfere with the Wnt signaling pathway, and that Wnt3a is involved in SARS-CoV-2 entry.
n.
The frizzled (FZD) receptor is a host transmembrane receptor that binds Wnt ligands, initiating the Wnt signaling cascade. FZD serves as a co-receptor, along with ACE2, in some proposed mechanisms of SARS-CoV-2 infection. The virus may take advantage of this pathway as an alternative entry route.
o.
Low-density lipoprotein receptor-related protein 6 is a cell surface co-receptor essential for Wnt signaling. LRP6 acts in tandem with FZD for signal transduction and has been discussed as a potential co-receptor for SARS-CoV-2 entry.
p.
The ezrin protein links the cell membrane to the cytoskeleton (the cell's internal support structure) and plays a role in cell shape, movement, adhesion, and signaling. Drugs that occupy the same spot on ezrin where the viral spike protein would bind may hindering viral attachment, and drug binding could further stabilize ezrin, strengthening its potential natural capacity to impede viral fusion and entry.
q.
The Adipocyte Differentiation-Related Protein (ADRP, also known as Perilipin 2 or PLIN2) is a lipid droplet protein regulating the storage and breakdown of fats in cells. SARS-CoV-2 may hijack the lipid handling machinery of host cells and ADRP may play a role in this process. Disrupting ADRP's interaction with the virus may hinder the virus's ability to use lipids for replication and assembly.
r.
Neuropilin-1 (NRP1) is a cell surface receptor with roles in blood vessel development, nerve cell guidance, and immune responses. NRP1 may function as a co-receptor for SARS-CoV-2, facilitating viral entry into cells. Blocking NRP1 may disrupt an alternative route of viral entry.
s.
EP300 (E1A Binding Protein P300) is a transcriptional coactivator involved in several cellular processes, including growth, differentiation, and apoptosis, through its acetyltransferase activity that modifies histones and non-histone proteins. EP300 facilitates viral entry into cells and upregulates inflammatory cytokine production.
t.
Prostaglandin G/H synthase 2 (PTGS2, also known as COX-2) is an enzyme crucial for the production of inflammatory molecules called prostaglandins. PTGS2 plays a role in the inflammatory response that can become severe in COVID-19 and inhibitors (like some NSAIDs) may have benefits in dampening harmful inflammation, but note that prostaglandins have diverse physiological functions.
u.
Heat Shock Protein 90 Alpha Family Class A Member 1 (HSP90AA1) is a chaperone protein that helps other proteins fold correctly and maintains their stability. HSP90AA1 plays roles in cell signaling, survival, and immune responses. HSP90AA1 may interact with numerous viral proteins, but note that it has diverse physiological functions.
v.
Matrix metalloproteinase 9 (MMP9), also called gelatinase B, is a zinc-dependent enzyme that breaks down collagen and other components of the extracellular matrix. MMP9 levels increase in severe COVID-19. Overactive MMP9 can damage lung tissue and worsen inflammation. Inhibition of MMP9 may prevent excessive tissue damage and help regulate the inflammatory response.
w.
The interleukin-6 (IL-6) pro-inflammatory cytokine (signaling molecule) has a complex role in the immune response and may trigger and perpetuate inflammation. Elevated IL-6 levels are associated with severe COVID-19 cases and cytokine storm. Anti-IL-6 therapies may be beneficial in reducing excessive inflammation in severe COVID-19 cases.
x.
The interleukin-10 (IL-10) anti-inflammatory cytokine helps regulate and dampen immune responses, preventing excessive inflammation. IL-10 levels can also be elevated in severe COVID-19. IL-10 could either help control harmful inflammation or potentially contribute to immune suppression.
y.
Vascular Endothelial Growth Factor A (VEGFA) promotes the growth of new blood vessels (angiogenesis) and has roles in inflammation and immune responses. VEGFA may contribute to blood vessel leakiness and excessive inflammation associated with severe COVID-19.
z.
RELA is a transcription factor subunit of NF-kB and is a key regulator of inflammation, driving pro-inflammatory gene expression. SARS-CoV-2 may hijack and modulate NF-kB pathways.
aa.
The interaction between the SARS-CoV-2 spike protein and the human ACE2 receptor is a primary method of viral entry, inhibiting this interaction can prevent the virus from attaching to and entering host cells, halting infection at an early stage.
ab.
Calu-3 is a human lung adenocarcinoma cell line with moderate ACE2 and TMPRSS2 expression and SARS-CoV-2 susceptibility. It provides a model of the human respiratory epithelium, but many not be ideal for modeling early stages of infection due to the moderate expression levels of ACE2 and TMPRSS2.
ac.
A549 is a human lung carcinoma cell line with low ACE2 expression and SARS-CoV-2 susceptibility. Viral entry/replication can be studied but the cells may not replicate all aspects of lung infection.
ad.
HEK293-ACE2+ is a human embryonic kidney cell line engineered for high ACE2 expression and SARS-CoV-2 susceptibility.
ae.
Huh-7 cells were derived from a liver tumor (hepatoma).
af.
Caco-2 cells come from a colorectal adenocarcinoma (cancer). They are valued for their ability to form a polarized cell layer with properties similar to the intestinal lining.
ag.
Vero E6 is an African green monkey kidney cell line with low/no ACE2 expression and high SARS-CoV-2 susceptibility. The cell line is easy to maintain and supports robust viral replication, however the monkey origin may not accurately represent human responses.
ah.
mTEC is a mouse tubular epithelial cell line.
ai.
RAW264.7 is a mouse macrophage cell line.
aj.
HLMEC (Human Lung Microvascular Endothelial Cells) are primary endothelial cells derived from the lung microvasculature. They are used to study endothelial function, inflammation, and viral interactions, particularly in the context of lung infections such as SARS-CoV-2. HLMEC express ACE2 and are susceptible to SARS-CoV-2 infection, making them a relevant model for studying viral entry and endothelial responses in the lung.
ak.
A mouse model expressing the human ACE2 receptor under the control of the K18 promoter.
al.
A mouse model of obesity and severe insulin resistance leading to type 2 diabetes due to a mutation in the leptin receptor gene that impairs satiety signaling.
am.
A mouse model commonly used in infectious disease and cancer research due to higher immune response and susceptibility to infection.
Aguilera-Rodriguez et al., 16 Sep 2024, peer-reviewed, 8 authors.
Contact: josempalomo@icp.csic.es.
In vitro studies are an important part of preclinical research, however results may be very different in vivo.
Inhibition of SARS-CoV-2 3CLpro by chemically modified tyrosinase from Agaricus bisporus
RSC Medicinal Chemistry, doi:10.1039/d4md00289j
Antiviral compounds are crucial to controlling the SARS-CoV-2 pandemic. Approved drugs have been tested for their efficacy against COVID-19, and new pharmaceuticals are being developed as a complementary tool to vaccines. In this work, a cheap and fast purification method for natural tyrosinase from Agaricus bisporus (AbTyr) fresh mushrooms was developed to evaluate the potential of this enzyme as a therapeutic protein via the inhibition of SARS-CoV-2 3CLpro protease activity in vitro. AbTyr showed a mild inhibition of 3CLpro. Thus, different variants of this protein were synthesized through chemical modifications, covalently binding different tailor-made glycans and peptides to the amino terminal groups of the protein. These new tyrosinase conjugates were purified and characterized through circular dichroism and fluorescence spectroscopy analyses, and their stability was evaluated under different conditions. Subsequently, all these tyrosinase conjugates were tested for 3CLpro protease inhibition. From them, the conjugate between tyrosinase and a dextran-aspartic acid (6 kDa) polymer showed the highest inhibition, with an IC 50 of 2.5 μg ml -1 and IC 90 of 5 μg ml -1 , with no cytotoxicity activity by polymer insertion. Finally, SARS-CoV-2 virus infection was studied. It was found that this new AbTyr-Dext6000 protein showed an 80% decrease in viral load. These results show the capacity of these tyrosinase bioconjugates as potential therapeutic proteins, opening the possibility of extension and applicability against other different viruses.
Conflicts of interest There is no conflict of interest to declare.
References
Anand, Palm, Mesters, Siddell, Ziebuhr et al., Structure of coronavirus main proteinase reveals combination of a chymotrypsin fold with an extra α-helical domain, EMBO J
Anjani, Kumar, Rathi, Poonam, Recent updates on the biological efficacy of approved drugs and potent synthetic compounds against SARS-CoV-2, RSC Adv
Biemans, Jäkel, De Waal, Bea, Kuiperij et al., Limitations of the hCMEC/D3 cell line as a model for Aβ clearance by the human blood-brain barrier, J. Neurosci. Res
Boutureira, Bernardes, Advances in chemical protein modification, Chem. Rev
Chen, Gao, Liu, Li, Chen et al., Advances in research on 3C-like protease (3CLpro) inhibitors against SARS-CoV-2 since 2020, RSC Med. Chem
Cho, Daniel, Buechler, Litzinger, Maio et al., Optimized clinical performance of growth hormone with an expanded genetic code, Proc. Natl. Acad. Sci. U. S. A
Delft, Hall, Kwong, Purcell, Singh et al., Accelerating antiviral drug discovery: lessons from COVID-19, Nat. Rev. Drug Discovery
Eastman, Roth, Brimacombe, Simeonov, Shen et al., Remdesivir: A Review of Its Discovery and Development Leading to Emergency Use Authorization for Treatment of COVID-19, ACS Cent. Sci
Farcet, Kosma, Determination of modification degree of polysialylated therapeutic proteins using 1H-NMR spectroscopy, Int. J. Biol. Macromol
Grifagni, Lenci, De Santis, Orsetti, Barracchia et al., Development of a GC-376 Based Peptidomimetic PROTAC as a Degrader of 3-Chymotrypsin-like Protease of SARS-CoV-2, ACS Med. Chem. Lett
Hashemian, Sheida, Taghizadieh, Memar, Hamblin et al., Paxlovid (Nirmatrelvir/Ritonavir): A new approach to Covid-19 therapy?, Biomed. Pharmacother
Hegyi, Ziebuhr, Conservation of substrate specificities among coronavirus main proteases, J. Gen. Virol
Hou, Shuai, Zhang, Xie, Tang et al., Development of Highly Potent Noncovalent Inhibitors of SARS-CoV-2 3CLpro, ACS Cent. Sci
Hou, Shuai, Zhang, Xie, Tang et al., None, ACS Cent. Sci
Hu, Guo, Zhou, Shi, Characteristics of SARS-CoV-2 and COVID-19, Nat. Rev. Microbiol
Ismaya, Rozeboom, Weijn, Mes, Fusetti et al., Crystal structure of agaricus bisporus mushroom tyrosinase: Identity of the tetramer subunits and interaction with tropolone, Biochemistry
Jin, Du, Xu, Deng, Liu et al., Structure of Mpro from SARS-CoV-2 and discovery of its inhibitors, Nature
Khiali, Khani, Rouy, Entezari-Maleki, Comprehensive Review on Molnupiravir in COVID-19: A Novel Promising Antiviral to Combat the Pandemic, Future Microbiol
Lopez-Tejedor, Clavería-Gimeno, Velazquez-Campoy, Abian, Palomo, In Vitro Antiviral Activity of Tyrosinase from Mushroom Agaricus bisporus against Hepatitis C Virus, Pharmaceuticals
Lopez-Tejedor, Palomo, Efficient purification of a highly active H-subunit of tyrosinase from Agaricus bisporus, Protein Expression Purif
Pretzler, Bijelic, Rompel, Heterologous expression and characterization of functional mushroom tyrosinase, Sci. Rep
Romero, Rivero, Guisan, Palomo, Novel enzyme-polymer conjugates for biotechnological applications, PeerJ
Stephanopoulos, Francis, Choosing an effective protein bioconjugation strategy, Nat. Chem. Biol
Tedesco, Calugi, Lenci, Trabocchi, Peptidomimetic Small-Molecule Inhibitors of 3CLPro Activity and Spike-ACE2 Interaction: Toward Dual-Action Molecules against Coronavirus Infections, J. Org. Chem
Tian, Qiang, Yang, Gao, Zhai et al., Development of de-novo coronavirus 3-chymotrypsin-like protease (3CLpro) inhibitors since COVID-19 outbreak: A strategy to tackle challenges of persistent virus infection, Eur. J. Med. Chem
Wichers, Gerritsen, Chapelon, Tyrosinase isoforms from the fruitbodies of Agaricus bisporus, Phytochemistry
Wichers, Recourt, Hendriks, Ebbelaar, Biancone et al., Cloning, expression and characterisation of two tyrosinase cDNAs from Agaricus bisporus, Appl. Microbiol. Biotechnol
Yevsieieva, Lohachova, Kyrychenko, Kovalenko, Ivanov et al., Main and papainlike proteases as prospectiveBtargets for pharmacological treatment of coronavirus SARS-CoV-2, RSC Adv
Zhang, Tang, Overview of Targets and Potential Drugs of SARS-CoV-2 According to the Viral Replication, J. Proteome Res
DOI record:
{
"DOI": "10.1039/d4md00289j",
"ISSN": [
"2632-8682"
],
"URL": "http://dx.doi.org/10.1039/D4MD00289J",
"abstract": "<jats:p>A novel antiviral and non-cytotoxic bioconjugate of tyrosinase from <jats:italic>Agaricus bisporus</jats:italic> (AbTyr)-dextran-aspartic acid (6 kDa) polymer is developed.</jats:p>",
"author": [
{
"affiliation": [
{
"name": "Instituto de Catálisis y Petroleoquímica (ICP), CSIC, C/Marie Curie 2, 28049 Madrid, Spain"
}
],
"family": "Aguilera-Rodriguez",
"given": "David",
"sequence": "first"
},
{
"affiliation": [
{
"name": "Instituto de Investigación Sanitaria Aragón (IIS Aragón), 50009, Zaragoza, Spain"
},
{
"name": "Centro de Investigación Biomédica en Red en el Área Temática de Enfermedades Hepáticas y Digestivas (CIBERehd), 28029, Madrid, Spain"
},
{
"name": "Institute for Biocomputation and Physics of Complex Systems, University of Zaragoza, Spain"
}
],
"family": "Ortega-Alarcon",
"given": "David",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Centro de Biología Molecular Severo Ochoa, Consejo Superior de Investigaciones Científicas (CSIC)-Universidad Autónoma de Madrid (UAM), 28049, Madrid, Spain"
}
],
"family": "Vazquez-Calvo",
"given": "Angela",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Instituto de Catálisis y Petroleoquímica (ICP), CSIC, C/Marie Curie 2, 28049 Madrid, Spain"
}
],
"family": "Ricci",
"given": "Veronica",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Instituto de Investigación Sanitaria Aragón (IIS Aragón), 50009, Zaragoza, Spain"
},
{
"name": "Centro de Investigación Biomédica en Red en el Área Temática de Enfermedades Hepáticas y Digestivas (CIBERehd), 28029, Madrid, Spain"
},
{
"name": "Institute for Biocomputation and Physics of Complex Systems, University of Zaragoza, Spain"
},
{
"name": "Department of Biochemistry and Molecular and Cell Biology, University of Zaragoza, Spain"
}
],
"family": "Abian",
"given": "Olga",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Instituto de Investigación Sanitaria Aragón (IIS Aragón), 50009, Zaragoza, Spain"
},
{
"name": "Centro de Investigación Biomédica en Red en el Área Temática de Enfermedades Hepáticas y Digestivas (CIBERehd), 28029, Madrid, Spain"
},
{
"name": "Institute for Biocomputation and Physics of Complex Systems, University of Zaragoza, Spain"
},
{
"name": "Department of Biochemistry and Molecular and Cell Biology, University of Zaragoza, Spain"
}
],
"family": "Velazquez-Campoy",
"given": "Adrian",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Centro de Biología Molecular Severo Ochoa, Consejo Superior de Investigaciones Científicas (CSIC)-Universidad Autónoma de Madrid (UAM), 28049, Madrid, Spain"
}
],
"family": "Alcami",
"given": "Antonio",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-6464-1216",
"affiliation": [
{
"name": "Instituto de Catálisis y Petroleoquímica (ICP), CSIC, C/Marie Curie 2, 28049 Madrid, Spain"
}
],
"authenticated-orcid": false,
"family": "Palomo",
"given": "Jose M.",
"sequence": "additional"
}
],
"container-title": "RSC Medicinal Chemistry",
"container-title-short": "RSC Med. Chem.",
"content-domain": {
"crossmark-restriction": true,
"domain": [
"rsc.org"
]
},
"created": {
"date-parts": [
[
2024,
9,
16
]
],
"date-time": "2024-09-16T08:20:45Z",
"timestamp": 1726474845000
},
"deposited": {
"date-parts": [
[
2024,
10,
4
]
],
"date-time": "2024-10-04T06:59:17Z",
"timestamp": 1728025157000
},
"funder": [
{
"DOI": "10.13039/501100003339",
"award": [
"SGL2103036",
"202480E088"
],
"doi-asserted-by": "publisher",
"id": [
{
"asserted-by": "publisher",
"id": "10.13039/501100003339",
"id-type": "DOI"
}
],
"name": "Consejo Superior de Investigaciones Científicas"
}
],
"indexed": {
"date-parts": [
[
2024,
10,
4
]
],
"date-time": "2024-10-04T07:40:45Z",
"timestamp": 1728027645179
},
"is-referenced-by-count": 0,
"issued": {
"date-parts": [
[
2024
]
]
},
"language": "en",
"license": [
{
"URL": "http://creativecommons.org/licenses/by/3.0/",
"content-version": "vor",
"delay-in-days": 259,
"start": {
"date-parts": [
[
2024,
9,
16
]
],
"date-time": "2024-09-16T00:00:00Z",
"timestamp": 1726444800000
}
}
],
"link": [
{
"URL": "http://pubs.rsc.org/en/content/articlepdf/2024/MD/D4MD00289J",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "292",
"original-title": [],
"prefix": "10.1039",
"published": {
"date-parts": [
[
2024
]
]
},
"published-online": {
"date-parts": [
[
2024
]
]
},
"publisher": "Royal Society of Chemistry (RSC)",
"reference": [
{
"DOI": "10.1038/s41579-020-00459-7",
"author": "Hu",
"doi-asserted-by": "crossref",
"first-page": "141",
"journal-title": "Nat. Rev. Microbiol.",
"key": "D4MD00289J/cit1/1",
"volume": "19",
"year": "2021"
},
{
"DOI": "10.1039/D2RA06834F",
"author": "Anjani",
"doi-asserted-by": "crossref",
"first-page": "3677",
"journal-title": "RSC Adv.",
"key": "D4MD00289J/cit2/1",
"volume": "13",
"year": "2023"
},
{
"DOI": "10.1021/acscentsci.2c01359",
"author": "Hou",
"doi-asserted-by": "crossref",
"first-page": "217",
"journal-title": "ACS Cent. Sci.",
"key": "D4MD00289J/cit3/1",
"volume": "9",
"year": "2023"
},
{
"DOI": "10.1038/s41573-023-00692-8",
"author": "von Delft",
"doi-asserted-by": "crossref",
"first-page": "585",
"journal-title": "Nat. Rev. Drug Discovery",
"key": "D4MD00289J/cit4/1",
"volume": "22",
"year": "2023"
},
{
"DOI": "10.1016/j.ejmech.2023.115979",
"author": "Tian",
"doi-asserted-by": "crossref",
"first-page": "115979",
"journal-title": "Eur. J. Med. Chem.",
"key": "D4MD00289J/cit5/1",
"volume": "264",
"year": "2024"
},
{
"DOI": "10.1039/D3RA06479D",
"author": "Yevsieieva",
"doi-asserted-by": "crossref",
"first-page": "35500",
"journal-title": "RSC Adv.",
"key": "D4MD00289J/cit6/1",
"volume": "13",
"year": "2023"
},
{
"DOI": "10.1021/acs.jproteome.0c00526",
"author": "Zhang",
"doi-asserted-by": "crossref",
"first-page": "49",
"journal-title": "J. Proteome Res.",
"key": "D4MD00289J/cit7/1",
"volume": "20",
"year": "2021"
},
{
"DOI": "10.1093/emboj/cdf327",
"author": "Anand",
"doi-asserted-by": "crossref",
"first-page": "3213",
"journal-title": "EMBO J.",
"key": "D4MD00289J/cit8/1",
"volume": "21",
"year": "2002"
},
{
"DOI": "10.1099/0022-1317-83-3-595",
"author": "Hegyi",
"doi-asserted-by": "crossref",
"first-page": "595",
"journal-title": "J. Gen. Virol.",
"key": "D4MD00289J/cit9/1",
"volume": "83",
"year": "2002"
},
{
"DOI": "10.1038/s41586-020-2223-y",
"author": "Jin",
"doi-asserted-by": "crossref",
"first-page": "289",
"journal-title": "Nature",
"key": "D4MD00289J/cit10/1",
"volume": "582",
"year": "2020"
},
{
"DOI": "10.1021/acscentsci.0c00489",
"author": "Eastman",
"doi-asserted-by": "crossref",
"first-page": "672",
"journal-title": "ACS Cent. Sci.",
"key": "D4MD00289J/cit11/1",
"volume": "6",
"year": "2020"
},
{
"DOI": "10.1016/j.biopha.2023.114367",
"author": "Hashemian",
"doi-asserted-by": "crossref",
"first-page": "114367",
"journal-title": "Biomed. Pharmacother.",
"key": "D4MD00289J/cit12/1",
"volume": "162",
"year": "2023"
},
{
"DOI": "10.2217/fmb-2021-0252",
"author": "Khiali",
"doi-asserted-by": "crossref",
"first-page": "377",
"journal-title": "Future\nMicrobiol.",
"key": "D4MD00289J/cit13/1",
"volume": "17",
"year": "2022"
},
{
"DOI": "10.1021/bi200395t",
"author": "Ismaya",
"doi-asserted-by": "crossref",
"first-page": "5477",
"journal-title": "Biochemistry",
"key": "D4MD00289J/cit14/1",
"volume": "50",
"year": "2011"
},
{
"DOI": "10.1038/s41598-017-01813-1",
"author": "Pretzler",
"doi-asserted-by": "crossref",
"first-page": "1",
"journal-title": "Sci. Rep.",
"key": "D4MD00289J/cit15/1",
"volume": "7",
"year": "2017"
},
{
"DOI": "10.1007/s00253-002-1194-2",
"author": "Wichers",
"doi-asserted-by": "crossref",
"first-page": "336",
"journal-title": "Appl. Microbiol. Biotechnol.",
"key": "D4MD00289J/cit16/1",
"volume": "61",
"year": "2003"
},
{
"DOI": "10.1016/0031-9422(96)00309-3",
"author": "Wichers",
"doi-asserted-by": "crossref",
"first-page": "333",
"journal-title": "Phytochemistry",
"key": "D4MD00289J/cit17/1",
"volume": "43",
"year": "1996"
},
{
"DOI": "10.3390/ph14080759",
"author": "Lopez-Tejedor",
"doi-asserted-by": "crossref",
"first-page": "759",
"journal-title": "Pharmaceuticals",
"key": "D4MD00289J/cit18/1",
"volume": "14",
"year": "2021"
},
{
"DOI": "10.1021/acscentsci.2c01359",
"author": "Hou",
"doi-asserted-by": "crossref",
"first-page": "217",
"journal-title": "ACS Cent. Sci.",
"key": "D4MD00289J/cit19/1",
"volume": "9",
"year": "2023"
},
{
"DOI": "10.1021/cr500399p",
"author": "Boutureira",
"doi-asserted-by": "crossref",
"first-page": "2174",
"journal-title": "Chem. Rev.",
"key": "D4MD00289J/cit20/1",
"volume": "115",
"year": "2015"
},
{
"DOI": "10.1038/nchembio.720",
"author": "Stephanopoulos",
"doi-asserted-by": "crossref",
"first-page": "876",
"journal-title": "Nat. Chem. Biol.",
"key": "D4MD00289J/cit21/1",
"volume": "7",
"year": "2011"
},
{
"DOI": "10.1073/pnas.1100387108",
"author": "Cho",
"doi-asserted-by": "crossref",
"first-page": "9060",
"journal-title": "Proc. Natl. Acad. Sci. U. S. A.",
"key": "D4MD00289J/cit22/1",
"volume": "108",
"year": "2011"
},
{
"DOI": "10.1016/j.ijbiomac.2021.06.165",
"author": "Farcet",
"doi-asserted-by": "crossref",
"first-page": "1015",
"issue": "31",
"journal-title": "Int. J. Biol. Macromol.",
"key": "D4MD00289J/cit23/1",
"volume": "185",
"year": "2021"
},
{
"DOI": "10.1002/jnr.23964",
"author": "Biemans",
"doi-asserted-by": "crossref",
"first-page": "1513",
"journal-title": "J. Neurosci. Res.",
"key": "D4MD00289J/cit24/1",
"volume": "95",
"year": "2017"
},
{
"DOI": "10.7717/peerj.27",
"author": "Romero",
"doi-asserted-by": "crossref",
"first-page": "e27",
"journal-title": "PeerJ",
"key": "D4MD00289J/cit25/1",
"volume": "2013",
"year": "2013"
},
{
"DOI": "10.1016/j.pep.2018.01.001",
"author": "Lopez-Tejedor",
"doi-asserted-by": "crossref",
"first-page": "64",
"journal-title": "Protein Expression Purif.",
"key": "D4MD00289J/cit26/1",
"volume": "145",
"year": "2018"
},
{
"DOI": "10.1021/acs.joc.2c01047",
"author": "Tedesco",
"doi-asserted-by": "crossref",
"first-page": "12041",
"journal-title": "J. Org. Chem.",
"key": "D4MD00289J/cit27/1",
"volume": "87",
"year": "2022"
},
{
"DOI": "10.1039/D2MD00344A",
"author": "Chen",
"doi-asserted-by": "crossref",
"first-page": "9",
"journal-title": "RSC Med. Chem.",
"key": "D4MD00289J/cit28/1",
"volume": "14",
"year": "2023"
},
{
"DOI": "10.1021/acsmedchemlett.3c00498",
"author": "Grifagni",
"doi-asserted-by": "crossref",
"first-page": "250",
"journal-title": "ACS Med. Chem. Lett.",
"key": "D4MD00289J/cit29/1",
"volume": "15",
"year": "2024"
}
],
"reference-count": 29,
"references-count": 29,
"relation": {},
"resource": {
"primary": {
"URL": "https://xlink.rsc.org/?DOI=D4MD00289J"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "Inhibition of SARS-CoV-2 3CLpro by chemically modified tyrosinase from <i>Agaricus bisporus</i>",
"type": "journal-article",
"update-policy": "http://dx.doi.org/10.1039/rsc_crossmark_policy"
}
