Poloxamer-Based Biomaterial as a Pharmaceutical Strategy to Improve the Ivermectin Performance
et al., Pharmaceutics, doi:10.3390/pharmaceutics17091101, Aug 2025
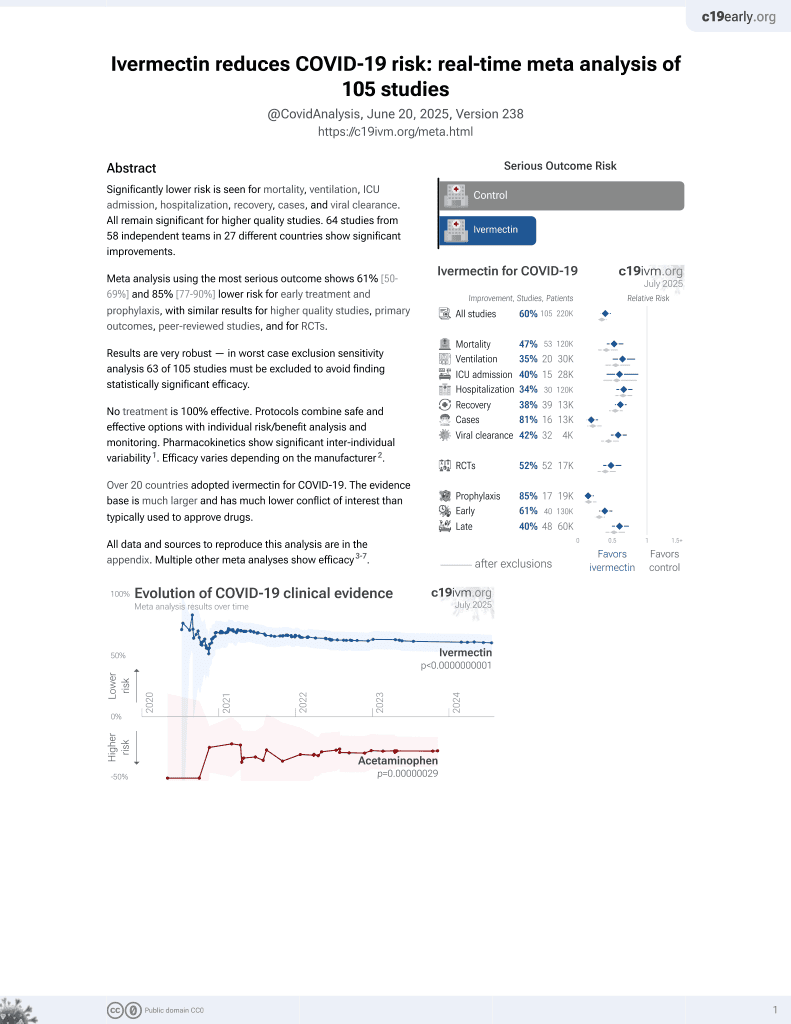
Ivermectin for COVID-19
4th treatment shown to reduce risk in
August 2020, now with p < 0.00000000001 from 106 studies, recognized in 24 countries.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
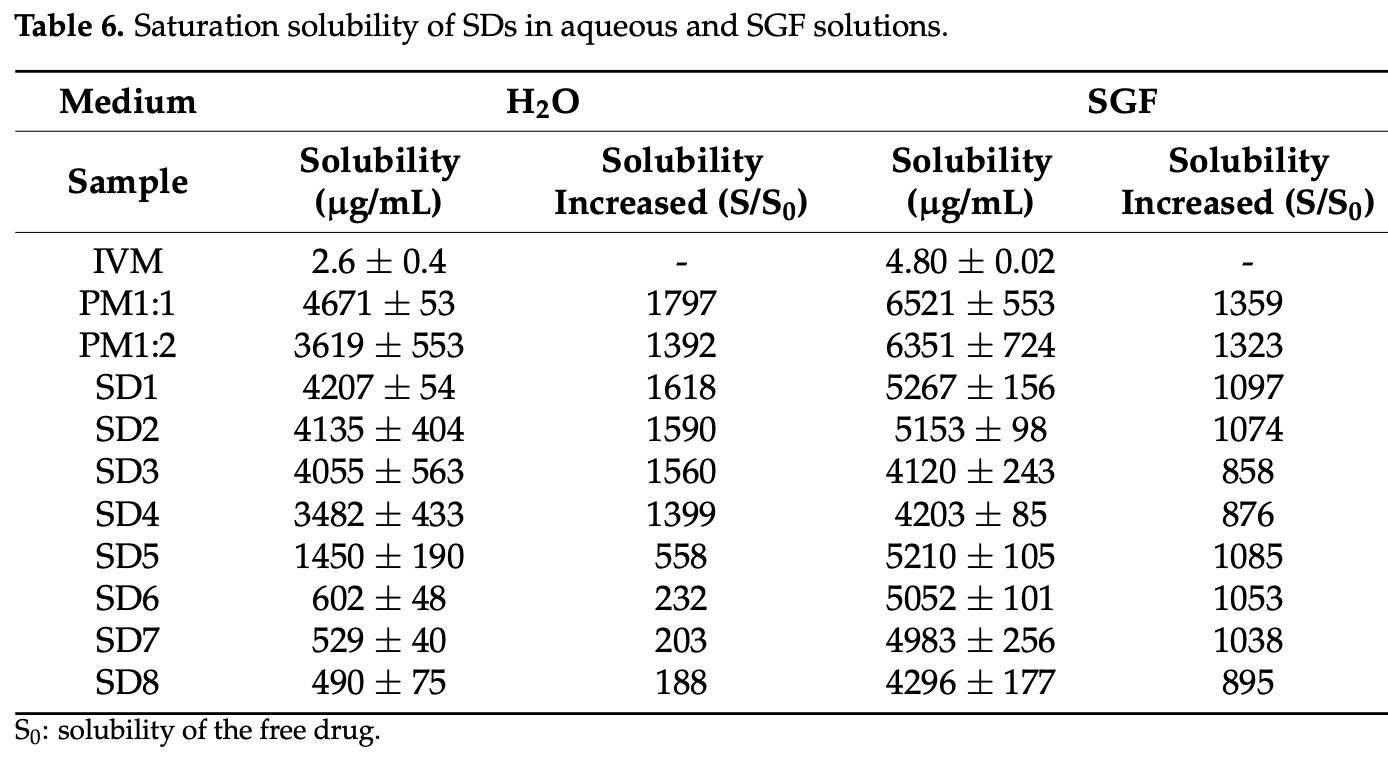
In vitro study showing that poloxamer 407 (P407)-based solid dispersions of ivermectin significantly improve aqueous solubility, dissolution, and drug release compared to pure ivermectin.
76 preclinical studies support the efficacy of ivermectin for COVID-19:
Ivermectin, better known for antiparasitic activity, is a broad spectrum antiviral with activity against many viruses including H7N773, Dengue38,74,75 , HIV-175, Simian virus 4076, Zika38,77,78 , West Nile78, Yellow Fever79,80, Japanese encephalitis79, Chikungunya80, Semliki Forest virus80, Human papillomavirus58, Epstein-Barr58, BK Polyomavirus81, and Sindbis virus80.
Ivermectin inhibits importin-α/β-dependent nuclear import of viral proteins73,75,76,82 , shows spike-ACE2 disruption at 1nM with microfluidic diffusional sizing39, binds to glycan sites on the SARS-CoV-2 spike protein preventing interaction with blood and epithelial cells and inhibiting hemagglutination42,83, shows dose-dependent inhibition of wildtype and omicron variants37, exhibits dose-dependent inhibition of lung injury63,68, may inhibit SARS-CoV-2 via IMPase inhibition38, may inhibit SARS-CoV-2 induced formation of fibrin clots resistant to degradation10, inhibits SARS-CoV-2 3CLpro55, may inhibit SARS-CoV-2 RdRp activity1,29, may minimize viral myocarditis by inhibiting NF-κB/p65-mediated inflammation in macrophages62, may be beneficial for COVID-19 ARDS by blocking GSDMD and NET formation84, may interfere with SARS-CoV-2's immune evasion via ORF8 binding5, may inhibit SARS-CoV-2 by disrupting CD147 interaction85-88, may inhibit SARS-CoV-2 attachment to lipid rafts via spike NTD binding3, shows protection against inflammation, cytokine storm, and mortality in an LPS mouse model sharing key pathological features of severe COVID-1960,89, may be beneficial in severe COVID-19 by binding IGF1 to inhibit the promotion of inflammation, fibrosis, and cell proliferation that leads to lung damage9, significantly mitigates bleomycin-induced pulmonary fibrosis by reducing collagen accumulation and inflammatory cell infiltration61, improves oxidative stress markers while suppressing myofibroblast proliferation61, may minimize SARS-CoV-2 induced cardiac damage41,49, may counter immune evasion by inhibiting NSP15-TBK1/KPNA1 interaction and restoring IRF3 activation90, may disrupt SARS-CoV-2 N and ORF6 protein nuclear transport and their suppression of host interferon responses2, reduces TAZ/YAP nuclear import, relieving SARS-CoV-2-driven suppression of IRF3 and NF-κB antiviral pathways36, increases Bifidobacteria which play a key role in the immune system91, has immunomodulatory52 and anti-inflammatory72,92 properties, and has an extensive and very positive safety profile93.
1.
Li et al., Drug–Target Interaction Prediction via Dual-Interaction Fusion, Molecules, doi:10.3390/molecules31030498.
2.
Gayozo et al., Binding affinities analysis of ivermectin, nucleocapsid and ORF6 proteins of SARS-CoV-2 to human importins α isoforms: A computational approach, Biotecnia, doi:10.18633/biotecnia.v27.2485.
3.
Lefebvre et al., Characterization and Fluctuations of an Ivermectin Binding Site at the Lipid Raft Interface of the N-Terminal Domain (NTD) of the Spike Protein of SARS-CoV-2 Variants, Viruses, doi:10.3390/v16121836.
4.
Haque et al., Exploring potential therapeutic candidates against COVID-19: a molecular docking study, Discover Molecules, doi:10.1007/s44345-024-00005-5.
5.
Bagheri-Far et al., Non-spike protein inhibition of SARS-CoV-2 by natural products through the key mediator protein ORF8, Molecular Biology Research Communications, doi:10.22099/mbrc.2024.50245.2001.
6.
de Oliveira Só et al., In Silico Comparative Analysis of Ivermectin and Nirmatrelvir Inhibitors Interacting with the SARS-CoV-2 Main Protease, Preprints, doi:10.20944/preprints202404.1825.v1.
7.
Agamah et al., Network-based multi-omics-disease-drug associations reveal drug repurposing candidates for COVID-19 disease phases, ScienceOpen, doi:10.58647/DRUGARXIV.PR000010.v1.
8.
Oranu et al., Validation of the binding affinities and stabilities of ivermectin and moxidectin against SARS-CoV-2 receptors using molecular docking and molecular dynamics simulation, GSC Biological and Pharmaceutical Sciences, doi:10.30574/gscbps.2024.26.1.0030.
9.
Zhao et al., Identification of the shared gene signatures between pulmonary fibrosis and pulmonary hypertension using bioinformatics analysis, Frontiers in Immunology, doi:10.3389/fimmu.2023.1197752.
10.
Vottero et al., Computational Prediction of the Interaction of Ivermectin with Fibrinogen, Molecular Sciences, doi:10.3390/ijms241411449.
11.
Chellasamy et al., Docking and molecular dynamics studies of human ezrin protein with a modelled SARS-CoV-2 endodomain and their interaction with potential invasion inhibitors, Journal of King Saud University - Science, doi:10.1016/j.jksus.2022.102277.
12.
Umar et al., Inhibitory potentials of ivermectin, nafamostat, and camostat on spike protein and some nonstructural proteins of SARS-CoV-2: Virtual screening approach, Jurnal Teknologi Laboratorium, doi:10.29238/teknolabjournal.v11i1.344.
13.
Alvarado et al., Interaction of the New Inhibitor Paxlovid (PF-07321332) and Ivermectin With the Monomer of the Main Protease SARS-CoV-2: A Volumetric Study Based on Molecular Dynamics, Elastic Networks, Classical Thermodynamics and SPT, Computational Biology and Chemistry, doi:10.1016/j.compbiolchem.2022.107692.
14.
Aminpour et al., In Silico Analysis of the Multi-Targeted Mode of Action of Ivermectin and Related Compounds, Computation, doi:10.3390/computation10040051.
15.
Parvez et al., Insights from a computational analysis of the SARS-CoV-2 Omicron variant: Host–pathogen interaction, pathogenicity, and possible drug therapeutics, Immunity, Inflammation and Disease, doi:10.1002/iid3.639.
16.
Francés-Monerris et al., Microscopic interactions between ivermectin and key human and viral proteins involved in SARS-CoV-2 infection, Physical Chemistry Chemical Physics, doi:10.1039/D1CP02967C.
17.
González-Paz et al., Comparative study of the interaction of ivermectin with proteins of interest associated with SARS-CoV-2: A computational and biophysical approach, Biophysical Chemistry, doi:10.1016/j.bpc.2021.106677.
18.
González-Paz (B) et al., Structural Deformability Induced in Proteins of Potential Interest Associated with COVID-19 by binding of Homologues present in Ivermectin: Comparative Study Based in Elastic Networks Models, Journal of Molecular Liquids, doi:10.1016/j.molliq.2021.117284.
19.
Rana et al., Ivermectin and Doxycycline combination as a promising anti-viral drug candidate: an in-silico and DFT study, In Silico Pharmacology, doi:10.1007/s40203-026-00576-w.
20.
Muthusamy et al., Virtual Screening Reveals Potential Anti-Parasitic Drugs Inhibiting the Receptor Binding Domain of SARS-CoV-2 Spike protein, Journal of Virology & Antiviral Research, www.scitechnol.com/abstract/virtual-screening-reveals-potential-antiparasitic-drugs-inhibiting-the-receptor-binding-domain-of-sarscov2-spike-protein-16398.html.
21.
Qureshi et al., Mechanistic insights into the inhibitory activity of FDA approved ivermectin against SARS-CoV-2: old drug with new implications, Journal of Biomolecular Structure and Dynamics, doi:10.1080/07391102.2021.1906750.
22.
Schöning et al., Highly-transmissible Variants of SARS-CoV-2 May Be More Susceptible to Drug Therapy Than Wild Type Strains, Research Square, doi:10.21203/rs.3.rs-379291/v1.
23.
Bello et al., Elucidation of the inhibitory activity of ivermectin with host nuclear importin α and several SARS-CoV-2 targets, Journal of Biomolecular Structure and Dynamics, doi:10.1080/07391102.2021.1911857.
24.
Udofia et al., In silico studies of selected multi-drug targeting against 3CLpro and nsp12 RNA-dependent RNA-polymerase proteins of SARS-CoV-2 and SARS-CoV, Network Modeling Analysis in Health Informatics and Bioinformatics, doi:10.1007/s13721-021-00299-2.
25.
Choudhury et al., Exploring the binding efficacy of ivermectin against the key proteins of SARS-CoV-2 pathogenesis: an in silico approach, Future Medicine, doi:10.2217/fvl-2020-0342.
26.
Kern et al., Modeling of SARS-CoV-2 Treatment Effects for Informed Drug Repurposing, Frontiers in Pharmacology, doi:10.3389/fphar.2021.625678.
27.
Saha et al., The Binding mechanism of ivermectin and levosalbutamol with spike protein of SARS-CoV-2, Structural Chemistry, doi:10.1007/s11224-021-01776-0.
28.
Eweas et al., Molecular Docking Reveals Ivermectin and Remdesivir as Potential Repurposed Drugs Against SARS-CoV-2, Frontiers in Microbiology, doi:10.3389/fmicb.2020.592908.
29.
Parvez (B) et al., Prediction of potential inhibitors for RNA-dependent RNA polymerase of SARS-CoV-2 using comprehensive drug repurposing and molecular docking approach, International Journal of Biological Macromolecules, doi:10.1016/j.ijbiomac.2020.09.098.
30.
Francés-Monerris (B) et al., Has Ivermectin Virus-Directed Effects against SARS-CoV-2? Rationalizing the Action of a Potential Multitarget Antiviral Agent, ChemRxiv, doi:10.26434/chemrxiv.12782258.v1.
31.
Kalhor et al., Repurposing of the approved small molecule drugs in order to inhibit SARS-CoV-2 S protein and human ACE2 interaction through virtual screening approaches, Journal of Biomolecular Structure and Dynamics, doi:10.1080/07391102.2020.1824816.
32.
Swargiary, A., Ivermectin as a promising RNA-dependent RNA polymerase inhibitor and a therapeutic drug against SARS-CoV2: Evidence from in silico studies, Research Square, doi:10.21203/rs.3.rs-73308/v1.
33.
Maurya, D., A Combination of Ivermectin and Doxycycline Possibly Blocks the Viral Entry and Modulate the Innate Immune Response in COVID-19 Patients, American Chemical Society (ACS), doi:10.26434/chemrxiv.12630539.v1.
34.
Lehrer et al., Ivermectin Docks to the SARS-CoV-2 Spike Receptor-binding Domain Attached to ACE2, In Vivo, 34:5, 3023-3026, doi:10.21873/invivo.12134.
35.
Suravajhala et al., Comparative Docking Studies on Curcumin with COVID-19 Proteins, Preprints, doi:10.20944/preprints202005.0439.v3.
36.
Kofler et al., M-Motif, a potential non-conventional NLS in YAP/TAZ and other cellular and viral proteins that inhibits classic protein import, iScience, doi:10.1016/j.isci.2025.112105.
37.
Shahin et al., The selective effect of Ivermectin on different human coronaviruses; in-vitro study, Research Square, doi:10.21203/rs.3.rs-4180797/v1.
38.
Jitobaom et al., Identification of inositol monophosphatase as a broad‐spectrum antiviral target of ivermectin, Journal of Medical Virology, doi:10.1002/jmv.29552.
39.
Fauquet et al., Microfluidic Diffusion Sizing Applied to the Study of Natural Products and Extracts That Modulate the SARS-CoV-2 Spike RBD/ACE2 Interaction, Molecules, doi:10.3390/molecules28248072.
40.
García-Aguilar et al., In Vitro Analysis of SARS-CoV-2 Spike Protein and Ivermectin Interaction, International Journal of Molecular Sciences, doi:10.3390/ijms242216392.
41.
Liu et al., SARS-CoV-2 viral genes Nsp6, Nsp8, and M compromise cellular ATP levels to impair survival and function of human pluripotent stem cell-derived cardiomyocytes, Stem Cell Research & Therapy, doi:10.1186/s13287-023-03485-3.
42.
Boschi et al., SARS-CoV-2 Spike Protein Induces Hemagglutination: Implications for COVID-19 Morbidities and Therapeutics and for Vaccine Adverse Effects, bioRxiv, doi:10.1101/2022.11.24.517882.
43.
De Forni et al., Synergistic drug combinations designed to fully suppress SARS-CoV-2 in the lung of COVID-19 patients, PLoS ONE, doi:10.1371/journal.pone.0276751.
44.
Saha (B) et al., Manipulation of Spray-Drying Conditions to Develop an Inhalable Ivermectin Dry Powder, Pharmaceutics, doi:10.3390/pharmaceutics14071432.
45.
Jitobaom (B) et al., Synergistic anti-SARS-CoV-2 activity of repurposed anti-parasitic drug combinations, BMC Pharmacology and Toxicology, doi:10.1186/s40360-022-00580-8.
46.
Croci et al., Liposomal Systems as Nanocarriers for the Antiviral Agent Ivermectin, International Journal of Biomaterials, doi:10.1155/2016/8043983.
47.
Zheng et al., Red blood cell-hitchhiking mediated pulmonary delivery of ivermectin: Effects of nanoparticle properties, International Journal of Pharmaceutics, doi:10.1016/j.ijpharm.2022.121719.
48.
Delandre et al., Antiviral Activity of Repurposing Ivermectin against a Panel of 30 Clinical SARS-CoV-2 Strains Belonging to 14 Variants, Pharmaceuticals, doi:10.3390/ph15040445.
49.
Liu (B) et al., Genome-wide analyses reveal the detrimental impacts of SARS-CoV-2 viral gene Orf9c on human pluripotent stem cell-derived cardiomyocytes, Stem Cell Reports, doi:10.1016/j.stemcr.2022.01.014.
50.
Segatori et al., Effect of Ivermectin and Atorvastatin on Nuclear Localization of Importin Alpha and Drug Target Expression Profiling in Host Cells from Nasopharyngeal Swabs of SARS-CoV-2- Positive Patients, Viruses, doi:10.3390/v13102084.
51.
Jitobaom (C) et al., Favipiravir and Ivermectin Showed in Vitro Synergistic Antiviral Activity against SARS-CoV-2, Research Square, doi:10.21203/rs.3.rs-941811/v1.
52.
Munson et al., Niclosamide and ivermectin modulate caspase-1 activity and proinflammatory cytokine secretion in a monocytic cell line, British Society For Nanomedicine Early Career Researcher Summer Meeting, 2021, web.archive.org/web/20230401070026/https://michealmunson.github.io/COVID.pdf.
53.
Mountain Valley MD, Mountain Valley MD Receives Successful Results From BSL-4 COVID-19 Clearance Trial on Three Variants Tested With Ivectosol™, 5/18, www.globenewswire.com/en/news-release/2021/05/18/2231755/0/en/Mountain-Valley-MD-Receives-Successful-Results-From-BSL-4-COVID-19-Clearance-Trial-on-Three-Variants-Tested-With-Ivectosol.html.
54.
Yesilbag et al., Ivermectin also inhibits the replication of bovine respiratory viruses (BRSV, BPIV-3, BoHV-1, BCoV and BVDV) in vitro, Virus Research, doi:10.1016/j.virusres.2021.198384.
55.
Mody et al., Identification of 3-chymotrypsin like protease (3CLPro) inhibitors as potential anti-SARS-CoV-2 agents, Communications Biology, doi:10.1038/s42003-020-01577-x.
56.
Jeffreys et al., Remdesivir-ivermectin combination displays synergistic interaction with improved in vitro activity against SARS-CoV-2, International Journal of Antimicrobial Agents, doi:10.1016/j.ijantimicag.2022.106542.
57.
Surnar et al., Clinically Approved Antiviral Drug in an Orally Administrable Nanoparticle for COVID-19, ACS Pharmacol. Transl. Sci., doi:10.1021/acsptsci.0c00179.
58.
Li (B) et al., Quantitative proteomics reveals a broad-spectrum antiviral property of ivermectin, benefiting for COVID-19 treatment, J. Cellular Physiology, doi:10.1002/jcp.30055.
59.
Caly et al., The FDA-approved drug ivermectin inhibits the replication of SARS-CoV-2 in vitro, Antiviral Research, doi:10.1016/j.antiviral.2020.104787.
60.
Zhang et al., Ivermectin inhibits LPS-induced production of inflammatory cytokines and improves LPS-induced survival in mice, Inflammation Research, doi:10.1007/s00011-008-8007-8.
61.
Zaher et al., Testing the Possible Protective Effect of Ivermectin on Bleomycin-Induced Pulmonary Fibrosis in Albino Rats: Histological and Immunohistochemical Study, Medicina, doi:10.3390/medicina62030560.
62.
Gao et al., Ivermectin ameliorates acute myocarditis via the inhibition of importin-mediated nuclear translocation of NF-κB/p65, International Immunopharmacology, doi:10.1016/j.intimp.2024.112073.
63.
Abd-Elmawla et al., Suppression of NLRP3 inflammasome by ivermectin ameliorates bleomycin-induced pulmonary fibrosis, Journal of Zhejiang University-SCIENCE B, doi:10.1631/jzus.B2200385.
64.
Uematsu et al., Prophylactic administration of ivermectin attenuates SARS-CoV-2 induced disease in a Syrian Hamster Model, The Journal of Antibiotics, doi:10.1038/s41429-023-00623-0.
65.
Albariqi et al., Pharmacokinetics and Safety of Inhaled Ivermectin in Mice as a Potential COVID-19 Treatment, International Journal of Pharmaceutics, doi:10.1016/j.ijpharm.2022.121688.
66.
Errecalde et al., Safety and Pharmacokinetic Assessments of a Novel Ivermectin Nasal Spray Formulation in a Pig Model, Journal of Pharmaceutical Sciences, doi:10.1016/j.xphs.2021.01.017.
67.
Madrid et al., Safety of oral administration of high doses of ivermectin by means of biocompatible polyelectrolytes formulation, Heliyon, doi:10.1016/j.heliyon.2020.e05820.
68.
Ma et al., Ivermectin contributes to attenuating the severity of acute lung injury in mice, Biomedicine & Pharmacotherapy, doi:10.1016/j.biopha.2022.113706.
69.
de Melo et al., Attenuation of clinical and immunological outcomes during SARS-CoV-2 infection by ivermectin, EMBO Mol. Med., doi:10.15252/emmm.202114122.
70.
Arévalo et al., Ivermectin reduces in vivo coronavirus infection in a mouse experimental model, Scientific Reports, doi:10.1038/s41598-021-86679-0.
71.
Chaccour et al., Nebulized ivermectin for COVID-19 and other respiratory diseases, a proof of concept, dose-ranging study in rats, Scientific Reports, doi:10.1038/s41598-020-74084-y.
72.
Yan et al., Anti-inflammatory effects of ivermectin in mouse model of allergic asthma, Inflammation Research, doi:10.1007/s00011-011-0307-8.
73.
Götz et al., Influenza A viruses escape from MxA restriction at the expense of efficient nuclear vRNP import, Scientific Reports, doi:10.1038/srep23138.
74.
Tay et al., Nuclear localization of dengue virus (DENV) 1–4 non-structural protein 5; protection against all 4 DENV serotypes by the inhibitor Ivermectin, Antiviral Research, doi:10.1016/j.antiviral.2013.06.002.
75.
Wagstaff et al., Ivermectin is a specific inhibitor of importin α/β-mediated nuclear import able to inhibit replication of HIV-1 and dengue virus, Biochemical Journal, doi:10.1042/BJ20120150.
76.
Wagstaff (B) et al., An AlphaScreen®-Based Assay for High-Throughput Screening for Specific Inhibitors of Nuclear Import, SLAS Discovery, doi:10.1177/1087057110390360.
77.
Barrows et al., A Screen of FDA-Approved Drugs for Inhibitors of Zika Virus Infection, Cell Host & Microbe, doi:10.1016/j.chom.2016.07.004.
78.
Yang et al., The broad spectrum antiviral ivermectin targets the host nuclear transport importin α/β1 heterodimer, Antiviral Research, doi:10.1016/j.antiviral.2020.104760.
79.
Mastrangelo et al., Ivermectin is a potent inhibitor of flavivirus replication specifically targeting NS3 helicase activity: new prospects for an old drug, Journal of Antimicrobial Chemotherapy, doi:10.1093/jac/dks147.
80.
Varghese et al., Discovery of berberine, abamectin and ivermectin as antivirals against chikungunya and other alphaviruses, Antiviral Research, doi:10.1016/j.antiviral.2015.12.012.
81.
Bennett et al., Role of a nuclear localization signal on the minor capsid Proteins VP2 and VP3 in BKPyV nuclear entry, Virology, doi:10.1016/j.virol.2014.10.013.
82.
Kosyna et al., The importin α/β-specific inhibitor Ivermectin affects HIF-dependent hypoxia response pathways, Biological Chemistry, doi:10.1515/hsz-2015-0171.
83.
Scheim et al., Sialylated Glycan Bindings from SARS-CoV-2 Spike Protein to Blood and Endothelial Cells Govern the Severe Morbidities of COVID-19, International Journal of Molecular Sciences, doi:10.3390/ijms242317039.
84.
Liu (C) et al., Crosstalk between neutrophil extracellular traps and immune regulation: insights into pathobiology and therapeutic implications of transfusion-related acute lung injury, Frontiers in Immunology, doi:10.3389/fimmu.2023.1324021.
85.
Shouman et al., SARS-CoV-2-associated lymphopenia: possible mechanisms and the role of CD147, Cell Communication and Signaling, doi:10.1186/s12964-024-01718-3.
86.
Scheim (B), D., Ivermectin for COVID-19 Treatment: Clinical Response at Quasi-Threshold Doses Via Hypothesized Alleviation of CD147-Mediated Vascular Occlusion, SSRN, doi:10.2139/ssrn.3636557.
87.
Scheim (C), D., From Cold to Killer: How SARS-CoV-2 Evolved without Hemagglutinin Esterase to Agglutinate and Then Clot Blood Cells, Center for Open Science, doi:10.31219/osf.io/sgdj2.
88.
Behl et al., CD147-spike protein interaction in COVID-19: Get the ball rolling with a novel receptor and therapeutic target, Science of The Total Environment, doi:10.1016/j.scitotenv.2021.152072.
89.
DiNicolantonio et al., Ivermectin may be a clinically useful anti-inflammatory agent for late-stage COVID-19, Open Heart, doi:10.1136/openhrt-2020-001350.
90.
Mothae et al., SARS-CoV-2 host-pathogen interactome: insights into more players during pathogenesis, Virology, doi:10.1016/j.virol.2025.110607.
91.
Hazan et al., Treatment with Ivermectin Increases the Population of Bifidobacterium in the Gut, ACG 2023, acg2023posters.eventscribe.net/posterspeakers.asp.
92.
DiNicolantonio (B) et al., Anti-inflammatory activity of ivermectin in late-stage COVID-19 may reflect activation of systemic glycine receptors, Open Heart, doi:10.1136/openhrt-2021-001655.
93.
Descotes, J., Medical Safety of Ivermectin, ImmunoSafe Consultance, web.archive.org/web/20240313025927/https://www.medincell.com/wp-content/uploads/2021/03/Clinical_Safety_of_Ivermectin-March_2021.pdf.
94.
Wissel et al., Tolerability, Safety, and Pharmacokinetics of Ivermectin After Nasal Application in Healthy Adult Subjects, The Journal of Clinical Pharmacology, doi:10.1002/jcph.70137.
95.
Mohammed et al., A remodeled ivermectin polycaprolactone-based nanoparticles for inhalation as a promising treatment of pulmonary inflammatory diseases, European Journal of Pharmaceutical Sciences, doi:10.1016/j.ejps.2024.106714.
Mezzano et al., 23 Aug 2025, Argentina, peer-reviewed, 5 authors.
Contact: cgarnero@unc.edu.ar (corresponding author), belen.mezzano@unc.edu.ar, soledad.bueno@unc.edu.ar, mrlonghi@unc.edu.ar, vfuertes@unc.edu.ar.
In vitro studies are an important part of preclinical research, however results may be very different in vivo.
Abstract:
Article
Poloxamer-Based Biomaterial as a Pharmaceutical Strategy to Improve the Ivermectin Performance
, Maria Soledad Bueno 1
, Valeria Cintia Fuertes
, Marcela Raquel Longhi 1
- 1 Departamento de Ciencias Farmacéuticas, Facultad de Ciencias Químicas, Universidad Nacional de Córdoba, Unidad de Investigación y Desarrollo en Tecnología Farmacéutica (UNITEFA) CONICET-UNC, Córdoba X5000HUA, Argentina; belen.mezzano@unc.edu.ar (B.A.M.); soledad.bueno@unc.edu.ar (M.S.B.); mrlonghi@unc.edu.ar (M.R.L.)
- 2 Departamento de Fisicoquímica, Facultad de Ciencias Químicas, Universidad Nacional de Córdoba, Instituto de Investigaciones en Fisicoquímica de Córdoba (INFIQC) CONICET-UNC, Córdoba X5000HUA, Argentina; vfuertes@unc.edu.ar
* Correspondence: cgarnero@unc.edu.ar
Abstract
Background: Poloxamers are promising biomaterials for drug delivery applications due to their ability to enhance biopharmaceutical properties. Methods: This study focused on designing solid dispersions of ivermectin using poloxamer 407 by the fusion method and evaluating how variables of synthesis affect the polymer's behavior and the resulting biopharmaceutical properties of ivermectin. Poloxamer 407 was selected based on a solubility test of preformulation studies. Initially, eight formulations were developed using different synthesis conditions, including polymer proportion, cooling gradient, and final process temperature. These were assessed by several characterization studies. Finally, saturation solubility dissolution profiles and in vitro drug release were also evaluated. Results: Acombination of techniques confirmed the compatibility between poloxamer 407 and ivermectin in the solid dispersions. The rate of temperature in the cooling process of synthesis showed a significant impact on the polymer self-assembly, affecting their ability to entrap ivermectin. The optimized solid dispersion comprised ivermectin and poloxamer 407 in a 1:1 w / w ratio prepared by rapid cooling. This decrease in the crystallinity index and the nanometric size of particles of the solid dispersions could explain their ability to improve 1600-fold the aqueous solubility, as well as enhance the drug dissolution and in vitro drug release compared to pure ivermectin. Conclusions: Therefore, it follows that these poloxamer-based solid dispersions are promising alternatives to improve the bioavailability of ivermectin.
Keywords: polymer; solid dispersion; fusion method; solubility; dissolution; crystallinity
DOI record:
{
"DOI": "10.3390/pharmaceutics17091101",
"ISSN": [
"1999-4923"
],
"URL": "http://dx.doi.org/10.3390/pharmaceutics17091101",
"abstract": "<jats:p>Background: Poloxamers are promising biomaterials for drug delivery applications due to their ability to enhance biopharmaceutical properties. Methods: This study focused on designing solid dispersions of ivermectin using poloxamer 407 by the fusion method and evaluating how variables of synthesis affect the polymer’s behavior and the resulting biopharmaceutical properties of ivermectin. Poloxamer 407 was selected based on a solubility test of preformulation studies. Initially, eight formulations were developed using different synthesis conditions, including polymer proportion, cooling gradient, and final process temperature. These were assessed by several characterization studies. Finally, saturation solubility dissolution profiles and in vitro drug release were also evaluated. Results: A combination of techniques confirmed the compatibility between poloxamer 407 and ivermectin in the solid dispersions. The rate of temperature in the cooling process of synthesis showed a significant impact on the polymer self-assembly, affecting their ability to entrap ivermectin. The optimized solid dispersion comprised ivermectin and poloxamer 407 in a 1:1 w/w ratio prepared by rapid cooling. This decrease in the crystallinity index and the nanometric size of particles of the solid dispersions could explain their ability to improve 1600-fold the aqueous solubility, as well as enhance the drug dissolution and in vitro drug release compared to pure ivermectin. Conclusions: Therefore, it follows that these poloxamer-based solid dispersions are promising alternatives to improve the bioavailability of ivermectin.</jats:p>",
"alternative-id": [
"pharmaceutics17091101"
],
"author": [
{
"ORCID": "https://orcid.org/0000-0001-5607-0110",
"affiliation": [
{
"name": "Departamento de Ciencias Farmacéuticas, Facultad de Ciencias Químicas, Universidad Nacional de Córdoba, Unidad de Investigación y Desarrollo en Tecnología Farmacéutica (UNITEFA) CONICET-UNC, Córdoba X5000HUA, Argentina"
}
],
"authenticated-orcid": false,
"family": "Mezzano",
"given": "Belén Alejandra",
"sequence": "first"
},
{
"ORCID": "https://orcid.org/0000-0001-9511-0234",
"affiliation": [
{
"name": "Departamento de Ciencias Farmacéuticas, Facultad de Ciencias Químicas, Universidad Nacional de Córdoba, Unidad de Investigación y Desarrollo en Tecnología Farmacéutica (UNITEFA) CONICET-UNC, Córdoba X5000HUA, Argentina"
}
],
"authenticated-orcid": false,
"family": "Bueno",
"given": "Maria Soledad",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0002-5331-7293",
"affiliation": [
{
"name": "Departamento de Fisicoquímica, Facultad de Ciencias Químicas, Universidad Nacional de Córdoba, Instituto de Investigaciones en Fisicoquímica de Córdoba (INFIQC) CONICET-UNC, Córdoba X5000HUA, Argentina"
}
],
"authenticated-orcid": false,
"family": "Fuertes",
"given": "Valeria Cintia",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0002-3189-9886",
"affiliation": [
{
"name": "Departamento de Ciencias Farmacéuticas, Facultad de Ciencias Químicas, Universidad Nacional de Córdoba, Unidad de Investigación y Desarrollo en Tecnología Farmacéutica (UNITEFA) CONICET-UNC, Córdoba X5000HUA, Argentina"
}
],
"authenticated-orcid": false,
"family": "Longhi",
"given": "Marcela Raquel",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0002-3347-9842",
"affiliation": [
{
"name": "Departamento de Ciencias Farmacéuticas, Facultad de Ciencias Químicas, Universidad Nacional de Córdoba, Unidad de Investigación y Desarrollo en Tecnología Farmacéutica (UNITEFA) CONICET-UNC, Córdoba X5000HUA, Argentina"
}
],
"authenticated-orcid": false,
"family": "Garnero",
"given": "Claudia",
"sequence": "additional"
}
],
"container-title": "Pharmaceutics",
"container-title-short": "Pharmaceutics",
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2025,
8,
25
]
],
"date-time": "2025-08-25T00:09:32Z",
"timestamp": 1756080572000
},
"deposited": {
"date-parts": [
[
2025,
10,
9
]
],
"date-time": "2025-10-09T18:34:55Z",
"timestamp": 1760034895000
},
"funder": [
{
"award": [
"11220200102912CO"
],
"award-info": [
{
"award-number": [
"11220200102912CO"
]
}
],
"name": "Consejo Nacional de Investigaciones Científicas y Técnicas"
},
{
"award": [
"PICT 2020-0297"
],
"award-info": [
{
"award-number": [
"PICT 2020-0297"
]
}
],
"name": "Consejo Nacional de Investigaciones Científicas y Técnicas"
},
{
"award": [
"33620230100359CB"
],
"award-info": [
{
"award-number": [
"33620230100359CB"
]
}
],
"name": "Consejo Nacional de Investigaciones Científicas y Técnicas"
},
{
"award": [
"11220200102912CO"
],
"award-info": [
{
"award-number": [
"11220200102912CO"
]
}
],
"name": "the Fondo para la Investigación Científica y Tecnológica"
},
{
"award": [
"PICT 2020-0297"
],
"award-info": [
{
"award-number": [
"PICT 2020-0297"
]
}
],
"name": "the Fondo para la Investigación Científica y Tecnológica"
},
{
"award": [
"33620230100359CB"
],
"award-info": [
{
"award-number": [
"33620230100359CB"
]
}
],
"name": "the Fondo para la Investigación Científica y Tecnológica"
},
{
"award": [
"11220200102912CO"
],
"award-info": [
{
"award-number": [
"11220200102912CO"
]
}
],
"name": "the Secretaría de Ciencia y Técnica de la Universidad Nacional de Córdoba (SECyT-UNC)"
},
{
"award": [
"PICT 2020-0297"
],
"award-info": [
{
"award-number": [
"PICT 2020-0297"
]
}
],
"name": "the Secretaría de Ciencia y Técnica de la Universidad Nacional de Córdoba (SECyT-UNC)"
},
{
"award": [
"33620230100359CB"
],
"award-info": [
{
"award-number": [
"33620230100359CB"
]
}
],
"name": "the Secretaría de Ciencia y Técnica de la Universidad Nacional de Córdoba (SECyT-UNC)"
}
],
"indexed": {
"date-parts": [
[
2026,
1,
23
]
],
"date-time": "2026-01-23T12:04:58Z",
"timestamp": 1769169898827,
"version": "3.49.0"
},
"is-referenced-by-count": 1,
"issue": "9",
"issued": {
"date-parts": [
[
2025,
8,
23
]
]
},
"journal-issue": {
"issue": "9",
"published-online": {
"date-parts": [
[
2025,
9
]
]
}
},
"language": "en",
"license": [
{
"URL": "https://creativecommons.org/licenses/by/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2025,
8,
23
]
],
"date-time": "2025-08-23T00:00:00Z",
"timestamp": 1755907200000
}
}
],
"link": [
{
"URL": "https://www.mdpi.com/1999-4923/17/9/1101/pdf",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "1968",
"original-title": [],
"page": "1101",
"prefix": "10.3390",
"published": {
"date-parts": [
[
2025,
8,
23
]
]
},
"published-online": {
"date-parts": [
[
2025,
8,
23
]
]
},
"publisher": "MDPI AG",
"reference": [
{
"key": "ref_1",
"unstructured": "Organización Panamericana de la Salud, and Organización Mundial de la Salud (2016). Enfermedades Infecciosas Desatendidas en las Américas: Historias de Éxito e Innovación para Llegar a los Más Necesitados, Organización Panamericana de la Salud."
},
{
"DOI": "10.1016/j.ijantimicag.2019.05.003",
"article-title": "Ivermectin: From theory to clinical application",
"author": "Ashour",
"doi-asserted-by": "crossref",
"first-page": "134",
"journal-title": "Int. J. Antimicrob. Agents",
"key": "ref_2",
"volume": "54",
"year": "2019"
},
{
"key": "ref_3",
"unstructured": "Philip, W. (2014). Avermectin. Encyclopedia of Toxicology, Elsevier Inc.. [3rd ed.]."
},
{
"article-title": "Ivermectina: Sus Múltiples Usos, Seguridad y Toxicidad",
"author": "Victoria",
"first-page": "358",
"journal-title": "Rev. Chil. Dermatol.",
"key": "ref_4",
"volume": "26",
"year": "2010"
},
{
"DOI": "10.3109/03639045.2012.660952",
"article-title": "Injectable PLA-based in situ forming implants for controlled release of Ivermectin a BCS Class II drug: Solvent selection based on physicochemical characterization",
"author": "Camargo",
"doi-asserted-by": "crossref",
"first-page": "146",
"journal-title": "Drug Dev. Ind. Pharm.",
"key": "ref_5",
"volume": "39",
"year": "2013"
},
{
"article-title": "Formulation and evaluation of ivermectin solid dispersion",
"author": "Verma",
"first-page": "15",
"journal-title": "J. Drug Deliv. Ther.",
"key": "ref_6",
"volume": "7",
"year": "2017"
},
{
"DOI": "10.1080/21691401.2017.1307207",
"article-title": "Ivermection-loaded solid lipid nanoparticles: Preparation, characterisation, stability and transdermal behaviour",
"author": "Guo",
"doi-asserted-by": "crossref",
"first-page": "255",
"journal-title": "Artif. Cells Nanomed. Biotechnol.",
"key": "ref_7",
"volume": "46",
"year": "2018"
},
{
"DOI": "10.3390/medicines6010007",
"doi-asserted-by": "crossref",
"key": "ref_8",
"unstructured": "Giuliano, E., Paolino, D., Fresta, M., and Cosco, D. (2018). Drug-Loaded Biocompatible Nanocarriers Embedded in Poloxamer 407 Hydrogels as Therapeutic Formulations. Medicines, 6."
},
{
"DOI": "10.3390/IECP2020-08686",
"doi-asserted-by": "crossref",
"key": "ref_9",
"unstructured": "Campos, S.N., Cid, A.G., Romer, A.I., Villegas, M., Briones Nieva, C.A., Gonzo, E.E., and Bermúdez, J.M. (2021). Solid dispersions as a technological strategy to improve the bio-performance of antiparasitic drugs with limited solubility. Proceedings, 78."
},
{
"DOI": "10.1016/j.actbio.2020.04.028",
"article-title": "Poloxamer: A versatile tri-block copolymer for biomedical applications",
"author": "Zarrintaj",
"doi-asserted-by": "crossref",
"first-page": "37",
"journal-title": "Acta Biomater.",
"key": "ref_10",
"volume": "110",
"year": "2020"
},
{
"DOI": "10.1590/s2175-97902022e21125",
"article-title": "Mini-Review of Poloxamer as a Biocompatible Polymer for Advanced Drug Delivery",
"author": "Nugraha",
"doi-asserted-by": "crossref",
"first-page": "e21125",
"journal-title": "Braz. J. Pharm. Sci.",
"key": "ref_11",
"volume": "58",
"year": "2022"
},
{
"article-title": "Characterization of solid dispersion: A review",
"author": "Deshmane",
"first-page": "584",
"journal-title": "Int. J. Pharm. Allied Res.",
"key": "ref_12",
"volume": "4",
"year": "2014"
},
{
"DOI": "10.1016/j.xphs.2018.06.027",
"article-title": "Preparation and Characterization of Poloxamer 407 Solid Dispersions as an Alternative Strategy to Improve Benznidazole Bioperformance",
"author": "Simonazzi",
"doi-asserted-by": "crossref",
"first-page": "2829",
"journal-title": "J. Pharm. Sci.",
"key": "ref_13",
"volume": "107",
"year": "2018"
},
{
"DOI": "10.4155/tde-2019-0007",
"article-title": "Solid dispersión technology as a strategy to improve the bioavailability of poorly soluble drugs",
"author": "Cid",
"doi-asserted-by": "crossref",
"first-page": "363",
"journal-title": "Ther. Deliv.",
"key": "ref_14",
"volume": "10",
"year": "2019"
},
{
"article-title": "Current Trends on Solid Dispersions: Past, Present, and Future",
"author": "Malkawi",
"first-page": "5916013",
"journal-title": "Adv. Pharmacol. Pharm. Sci.",
"key": "ref_15",
"volume": "2022",
"year": "2022"
},
{
"DOI": "10.1590/s2175-97902023e21217",
"article-title": "PVP solid dispersions containing Poloxamer 407 or TPGS for the improvement of ursolic acid release",
"author": "Pironi",
"doi-asserted-by": "crossref",
"first-page": "e21217",
"journal-title": "Braz. J. Pharm. Sci.",
"key": "ref_16",
"volume": "59",
"year": "2023"
},
{
"DOI": "10.1080/10837450.2016.1200078",
"article-title": "Design and in vitro characterization of ivermectin nanocrystals liquid formulation based on a top–down approach",
"author": "Starkloff",
"doi-asserted-by": "crossref",
"first-page": "809",
"journal-title": "Pharm. Dev. Technol.",
"key": "ref_17",
"volume": "22",
"year": "2016"
},
{
"article-title": "Formulation and optimization of pH-sensitive nanocrystals for improved oral delivery",
"author": "Parodi",
"first-page": "1301",
"journal-title": "Drug. Deliv. Transl. Res.",
"key": "ref_18",
"volume": "14",
"year": "2023"
},
{
"DOI": "10.3390/pharmaceutics16081061",
"doi-asserted-by": "crossref",
"key": "ref_19",
"unstructured": "Kassaee, S.N., Ayoko, G.A., Richard, D., Wang, T., and Islam, N. (2024). Inhaled Ivermectin-Loaded Lipid Polymer Hybrid Nanoparticles: Development and Characterization. Pharmaceutics, 16."
},
{
"DOI": "10.1016/j.cyto.2023.156143",
"article-title": "Exploring drug repositioning for leishmaniasis treatment: Ivermectin plus polymeric micelles induce immunological response and protection against tegumentary leishmaniasis",
"author": "Freitas",
"doi-asserted-by": "crossref",
"first-page": "156143",
"journal-title": "Cytokine",
"key": "ref_20",
"volume": "164",
"year": "2023"
},
{
"DOI": "10.1016/j.exppara.2020.108059",
"article-title": "Ivermectin presents effective and selective antileishmanial activity in vitro and in vivo against Leishmania infantum and is therapeutic against visceral leishmaniasis",
"author": "Reis",
"doi-asserted-by": "crossref",
"first-page": "108059",
"journal-title": "Exp. Parasitol.",
"key": "ref_21",
"volume": "221",
"year": "2021"
},
{
"DOI": "10.1039/D2CS00569G",
"article-title": "Comparison and assessment of methods for cellulose crystallinity determination",
"author": "Khandoker",
"doi-asserted-by": "crossref",
"first-page": "6417",
"journal-title": "Chem. Soc. Rev.",
"key": "ref_22",
"volume": "52",
"year": "2023"
},
{
"DOI": "10.1016/j.intimp.2021.108004",
"article-title": "Safety of inhaled ivermectin as a repurposed direct drug for treatment of COVID-19: A preclinical tolerance study",
"author": "Mansour",
"doi-asserted-by": "crossref",
"first-page": "108004",
"journal-title": "Int. Immunopharmacol.",
"key": "ref_23",
"volume": "99",
"year": "2021"
},
{
"DOI": "10.1107/S0021889878012844",
"article-title": "Scherrer after Sixty Years: A Survey and Some New Results in the Determination of Crystallite Size",
"author": "Langford",
"doi-asserted-by": "crossref",
"first-page": "102",
"journal-title": "J. Appl. Cryst.",
"key": "ref_24",
"volume": "11",
"year": "1978"
},
{
"key": "ref_25",
"unstructured": "(2017). The United States Pharmacopeia (Standard No. USP 40-NF 35)."
},
{
"DOI": "10.3390/pharmaceutics13101703",
"doi-asserted-by": "crossref",
"key": "ref_26",
"unstructured": "Muselík, J., Komersová, A., Kubová, K., Matzick, K., and Skalická, B. (2021). A Critical Overview of FDA and EMA Statistical Methods to Compare In Vitro Drug Dissolution Profiles of Pharmaceutical Products. Pharmaceutics, 13."
},
{
"DOI": "10.1016/S0928-0987(01)00095-1",
"article-title": "Modeling and comparison of dissolution profiles",
"author": "Costa",
"doi-asserted-by": "crossref",
"first-page": "123",
"journal-title": "Eur. J. Pharm. Sci.",
"key": "ref_27",
"volume": "13",
"year": "2001"
},
{
"DOI": "10.3329/dujps.v23i2.78570",
"article-title": "Preparation, Characterization and In vitro Evaluation of Solid Dispersion Formulation of Clopidogrel Using Hydrophilic Polymers",
"author": "Koly",
"doi-asserted-by": "crossref",
"first-page": "123",
"journal-title": "Dhaka Univ. J. Pharm. Sci.",
"key": "ref_28",
"volume": "23",
"year": "2024"
},
{
"DOI": "10.3390/pharmaceutics12121214",
"doi-asserted-by": "crossref",
"key": "ref_29",
"unstructured": "Russo, J., Fiegel, J., and Brogden, N.K. (2020). Rheological and Drug Delivery Characteristics of Poloxamer-Based Diclofenac Sodium Formulations for Chronic Wound Site Analgesia. Pharmaceutics, 12."
},
{
"DOI": "10.1208/s12248-010-9185-1",
"article-title": "DDSolver: An Add-In Program for Modeling and Comparison of Drug Dissolution Profiles",
"author": "Zhang",
"doi-asserted-by": "crossref",
"first-page": "263",
"journal-title": "AAPS J.",
"key": "ref_30",
"volume": "12",
"year": "2010"
},
{
"article-title": "High-performance liquid chromatographic method for the determination of ivermectin in plasma",
"author": "Banmairuroi",
"first-page": "848",
"journal-title": "Southeast. Asian J. Trop. Med. Public Health",
"key": "ref_31",
"volume": "37",
"year": "2006"
},
{
"key": "ref_32",
"unstructured": "(2025, June 05). HPLC Method for Ivermectin Determination. Available online: https://www.dikmatech.com/Application/show/id/805."
},
{
"key": "ref_33",
"unstructured": "International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (2025, July 08). ICH Harmonised Guideline: Validation of Analytical Procedures: Text and Methodology Q2(R2) 2023. Available online: https://www.ich.org/page/quality-guidelines."
},
{
"DOI": "10.1016/j.apsb.2021.05.014",
"article-title": "Pharmaceutical amorphous solid dispersion: A review of manufacturing strategies",
"author": "Bhujbal",
"doi-asserted-by": "crossref",
"first-page": "2505",
"journal-title": "Acta Pharm. Sin. B",
"key": "ref_34",
"volume": "11",
"year": "2021"
},
{
"DOI": "10.1080/10717544.2017.1284945",
"article-title": "Sustained release ivermectin-loaded solid lipid dispersion for subcutaneous delivery: In vitro and in vivo evaluation",
"author": "Lu",
"doi-asserted-by": "crossref",
"first-page": "622",
"journal-title": "Drug Deliv.",
"key": "ref_35",
"volume": "24",
"year": "2017"
},
{
"key": "ref_36",
"unstructured": "Magellan Laboratories Incorporated (2001). Purification Process for Anti-Parasitic Fermentation Product. (No. US6265571B1), U.S. Patent, Available online: https://patents.google.com/patent/US6265571B1/."
},
{
"DOI": "10.1007/s10965-024-04248-1",
"article-title": "Optimization of the poloxamer 407-conjugated gelatin to synthesize pH-sensitive nanocarriers for controlled paclitaxel delivery",
"author": "Ly",
"doi-asserted-by": "crossref",
"first-page": "38",
"journal-title": "J. Polym. Res.",
"key": "ref_37",
"volume": "32",
"year": "2025"
},
{
"DOI": "10.3390/polym13183038",
"doi-asserted-by": "crossref",
"key": "ref_38",
"unstructured": "Ponnusamy, C., Sugumaran, A., Krishnaswami, V., Palanichamy, R., Velayutham, R., and Natesan, S. (2021). Development and Evaluation of Polyvinylpyrrolidone K90 and Poloxamer 407 Self-Assembled Nanomicelles: Enhanced Topical Ocular Delivery of Artemisinin. Polymers, 13."
},
{
"DOI": "10.1007/s10973-014-3691-9",
"article-title": "Preformulation study of ivermectin raw material",
"author": "Rolim",
"doi-asserted-by": "crossref",
"first-page": "807",
"journal-title": "J. Therm. Anal. Calorim.",
"key": "ref_39",
"volume": "120",
"year": "2013"
},
{
"DOI": "10.1208/s12249-019-1551-4",
"article-title": "Improving the Dissolution of Triclabendazole from Stable Crystalline Solid Dispersions Formulated for Oral Delivery",
"author": "Real",
"doi-asserted-by": "crossref",
"first-page": "16",
"journal-title": "AAPS Pharm. Sci. Tech.",
"key": "ref_40",
"volume": "21",
"year": "2019"
},
{
"DOI": "10.1208/s12249-009-9316-0",
"article-title": "Kneading Technique for Preparation of Binary Solid Dispersion of Meloxicam with Poloxamer 188",
"author": "Ghareeb",
"doi-asserted-by": "crossref",
"first-page": "1206",
"journal-title": "AAPS Pharm. Sci. Tech.",
"key": "ref_41",
"volume": "10",
"year": "2009"
},
{
"DOI": "10.3390/pharmaceutics16030325",
"doi-asserted-by": "crossref",
"key": "ref_42",
"unstructured": "Velho, M.C., Funk, N.L., Deon, M., Benvenutti, E.V., Buchner, S., Hinrichs, R., Pilger, D.A., and Beck, R.C.R. (2024). Ivermectin-Loaded Mesoporous Silica and Polymeric Nanocapsules: Impact on Drug Loading, In Vitro Solubility Enhancement, and Release Performance. Pharmaceutics, 16."
},
{
"DOI": "10.1007/s10973-015-4579-z",
"article-title": "Investigation on the Decomposition Kinetics of Ivermectin",
"author": "Li",
"doi-asserted-by": "crossref",
"first-page": "797",
"journal-title": "J. Therm. Anal. Calorim.",
"key": "ref_43",
"volume": "121",
"year": "2015"
},
{
"DOI": "10.3390/pharmaceutics15082164",
"doi-asserted-by": "crossref",
"key": "ref_44",
"unstructured": "Zhang, J., Yang, Z., Luo, L., Li, K., Zi, T., Ren, J., Pan, L., Wang, Z.Y., Wang, Z.H., and Liu, M. (2023). Impact of Poloxamer on Crystal Nucleation and Growth of Amorphous Clotrimazole. Pharmaceutics, 15."
},
{
"DOI": "10.4155/tde-2018-0037",
"article-title": "Development and in vitro evaluation of solid dispersions as strategy to improve albendazole biopharmaceutical behavior",
"author": "Simonazzi",
"doi-asserted-by": "crossref",
"first-page": "623",
"journal-title": "Ther. Deliv.",
"key": "ref_45",
"volume": "9",
"year": "2018"
},
{
"article-title": "Performance of poloxamer 407 as hydrophilic carrier on the binary mixtures with nimesulide",
"author": "Hassan",
"first-page": "1137",
"journal-title": "Farmacia",
"key": "ref_46",
"volume": "61",
"year": "2013"
},
{
"article-title": "Dissolution rate enhancement and physicochemical characterization of carbamazepine-poloxamer solid dispersions",
"author": "Kachrimanis",
"first-page": "268",
"journal-title": "Pharm. Dev. Technol.",
"key": "ref_47",
"volume": "21",
"year": "2015"
},
{
"article-title": "Development and characterization of solid dispersion of piroxicam for improvement of dissolution rate using hydrophilic carriers",
"author": "Ghanbarzadeh",
"first-page": "141",
"journal-title": "BioImpacts",
"key": "ref_48",
"volume": "4",
"year": "2014"
},
{
"DOI": "10.3390/polym15102249",
"doi-asserted-by": "crossref",
"key": "ref_49",
"unstructured": "Gutiérrez-Saucedo, R.A., Gómez-López, J.C., Villanueva-Briseño, A.A., Topete, A., Soltero-Martínez, J.F.A., Mendizábal, E., Jasso-Gastinel, C.F., Taboada, P., and Figueroa-Ochoa, E.B. (2023). Pluronic F127 and P104 Polymeric Micelles as Efficient Nanocarriers for Loading and Release of Single and Dual Antineoplastic Drugs. Polymers, 15."
},
{
"DOI": "10.2147/IJN.S261855",
"article-title": "Nanosized Cubosomal Thermogelling Dispersion Loaded with Saquinavir Mesylate to Improve Its Bioavailability: Preparation, Optimization, in vitro and in vivo Evaluation",
"author": "Hosny",
"doi-asserted-by": "crossref",
"first-page": "5113",
"journal-title": "Int. J. Nanomed.",
"key": "ref_50",
"volume": "16",
"year": "2020"
},
{
"DOI": "10.1016/j.jconrel.2016.06.017",
"article-title": "DLS and zeta potential—What they are and what they are not?",
"author": "Bhattacharjee",
"doi-asserted-by": "crossref",
"first-page": "337",
"journal-title": "J. Control. Release",
"key": "ref_51",
"volume": "325",
"year": "2016"
},
{
"DOI": "10.1590/S1984-82502016000100002",
"article-title": "Preparation and evaluation of azithromycin binary solid dispersions using various polyethylene glycols for the improvement of the drug solubility and dissolution rate",
"author": "Adeli",
"doi-asserted-by": "crossref",
"first-page": "1",
"journal-title": "Braz. J. Pharm. Sci.",
"key": "ref_52",
"volume": "52",
"year": "2016"
},
{
"DOI": "10.3390/pr10061094",
"doi-asserted-by": "crossref",
"key": "ref_53",
"unstructured": "Trucillo, P. (2022). Drug Carriers: A Review on the Most Used Mathematical Models for Drug Release. Processes, 10."
},
{
"article-title": "Pharmaceutical and Biomedical Applications of Polymers",
"author": "Tekade",
"first-page": "203",
"journal-title": "Advances in Pharmaceutical Product Development and Research, Basic Fundamentals of Drug Delivery",
"key": "ref_54",
"volume": "Volume 6",
"year": "2019"
}
],
"reference-count": 54,
"references-count": 54,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.mdpi.com/1999-4923/17/9/1101"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "Poloxamer-Based Biomaterial as a Pharmaceutical Strategy to Improve the Ivermectin Performance",
"type": "journal-article",
"update-policy": "https://doi.org/10.3390/mdpi_crossmark_policy",
"volume": "17"
}