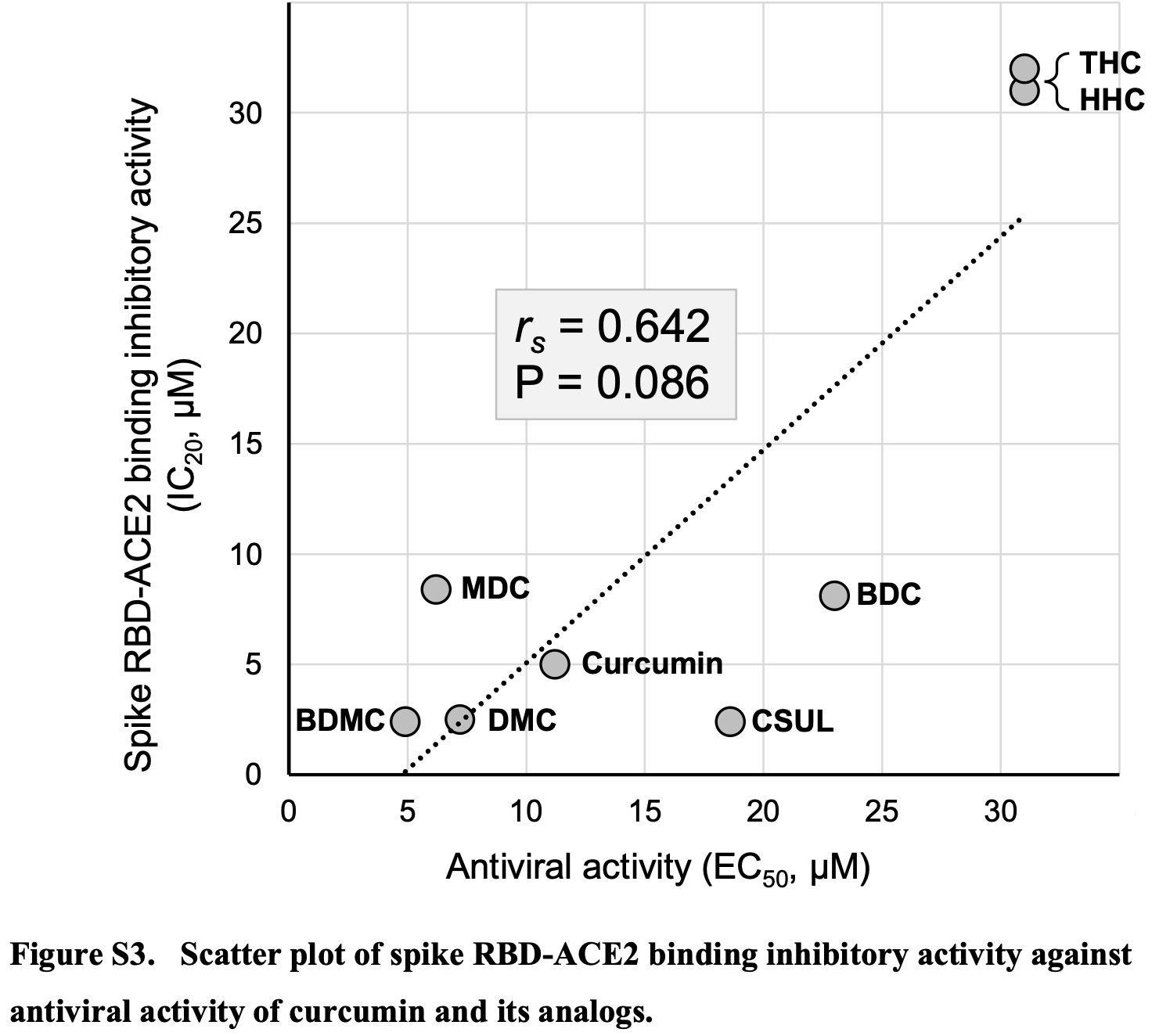
Antiviral activity of curcumin and its analogs selected by an artificial intelligence-supported activity prediction system in SARS-CoV-2-infected VeroE6 cells
et al., Natural Product Research, doi:10.1080/14786419.2023.2194647, Mar 2023
Curcumin for COVID-19
16th treatment shown to reduce risk in
February 2021, now with p = 0.0000000061 from 28 studies.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
In vitro study of curcumin and its analogs with 6 of 8 compounds showing SARS-CoV-2 antiviral activity with EC50 values <30μM and binding inhibitory activity with IC20 values <30μM.
64 preclinical studies support the efficacy of curcumin for COVID-19:
In silico studies predict inhibition of SARS-CoV-2 with curcumin or metabolites via binding to the spikeA,3,7,8,13,18,20,26,29 (and specifically the receptor binding domainB,2,4,6,16,19,22 ), MproC,6-8,13,15,17-19,21,22,24,27,29,30,32,50 , RNA-dependent RNA polymeraseD,6-8,19,28 , PLproE,8, ACE2F,4,20,21,23 , nucleocapsidG,14,31 , nsp10H,31, and helicaseI,38 proteins, and inhibition of spike-ACE2 interactionJ,2,5 .
In vitro studies demonstrate inhibition of the spikeA,43 (and specifically the receptor binding domainB,53), MproC,25,43,50,52 , ACE2F,53, and TMPRSS2K,53 proteins, and inhibition of spike-ACE2 interactionJ,5,36 .
In vitro studies demonstrate efficacy in Calu-3L,51, A549M,43, A549-ATN,33, 293TO,9, HEK293-hACE2P,25,41 , 293T/hACE2/TMPRSS2Q,42, Vero E6R,3,15,19,29,41,43,45,47,49,51 , and SH-SY5YS,40 cells.
Curcumin decreases pro-inflammatory cytokines induced by SARS-CoV-2 in peripheral blood mononuclear cells49, alleviates SARS-CoV-2 spike protein-induced mitochondrial membrane damage and oxidative stress9, may limit COVID-19 induced cardiac damage by inhibiting the NF-κB signaling pathway which mediates the profibrotic effects of the SARS-CoV-2 spike protein on cardiac fibroblasts37, is predicted to inhibit the interaction between the SARS-CoV-2 spike protein receptor binding domain and the human ACE2 receptor for the delta and omicron variants16, lowers ACE2 and STAT3, curbing lung inflammation and ARDS in preclinical COVID-19 models34, inhibits SARS-CoV-2 ORF3a ion channel activity, which contributes to viral pathogenicity and cytotoxicity44, has direct virucidal action by disrupting viral envelope integrity46, may inhibit viral replication and modulate inflammatory pathways like NF-κB via SIRT1 activation54, and can function as a photosensitizer in photodynamic therapy to generate reactive oxygen species that damage the virus46.
1.
Ataya et al., Identification of polyphenols as novel neuropilin-1 cendR pocket inhibitors to block SARS-CoV-2 entry and enhance variant resistance, PLOS One, doi:10.1371/journal.pone.0345051.
2.
Jena et al., Halved but Potent: Exploring the Inhibitory Property of Curcumin Derivatives Against Evolving SARS-CoV-2 Strains, The Protein Journal, doi:10.1007/s10930-025-10309-1.
3.
Marzouk et al., Computational and Experimental Insights into the Antiviral Mechanism of Turmeric (Curcuma longa) against SARS-CoV-2 D614G, BIO Web of Conferences, doi:10.1051/bioconf/202519804002.
4.
Wu et al., Utilizing natural compounds as ligands to disrupt the binding of SARS-CoV-2 receptor-binding domain to angiotensin-converting enzyme 2, impeding viral infection, Phytochemistry Letters, doi:10.1016/j.phytol.2025.102999.
5.
Najimi et al., Phytochemical Inhibitors of SARS‐CoV‐2 Entry: Targeting the ACE2‐RBD Interaction with l‐Tartaric Acid, l‐Ascorbic Acid, and Curcuma longa Extract, ChemistrySelect, doi:10.1002/slct.202406035.
6.
Rajamanickam et al., Exploring the Potential of Siddha Formulation MilagaiKudineer-Derived Phytotherapeutics Against SARS-CoV-2: An In-Silico Investigation for Antiviral Intervention, Journal of Pharmacy and Pharmacology Research, doi:10.26502/fjppr.0105.
7.
Al balawi et al., Assessing multi-target antiviral and antioxidant activities of natural compounds against SARS-CoV-2: an integrated in vitro and in silico study, Bioresources and Bioprocessing, doi:10.1186/s40643-024-00822-z.
8.
Haque et al., Exploring potential therapeutic candidates against COVID-19: a molecular docking study, Discover Molecules, doi:10.1007/s44345-024-00005-5.
9.
Zhang et al., Computational Discovery of Mitochondrial Dysfunction Biomarkers in Severe SARS-CoV-2 Infection: Facilitating Pytomedicine Screening, Phytomedicine, doi:10.1016/j.phymed.2024.155784.
10.
Öztürkkan et al., In Silico investigation of the effects of curcuminoids on the spike protein of the omicron variant of SARS-CoV-2, Baku State University Journal of Chemistry and Material Sciences, 1:2, bsuj.bsu.edu.az/uploads/pdf/ec4204d62f7802de54e6092bf7860029.pdf.
11.
Yunze et al., Therapeutic effect and potential mechanism of curcumin, an active ingredient in Tongnao Decoction, on COVID-19 combined with stroke: a network pharmacology study and GEO database mining, Research Square, doi:10.21203/rs.3.rs-4329762/v1.
12.
Agamah et al., Network-based multi-omics-disease-drug associations reveal drug repurposing candidates for COVID-19 disease phases, ScienceOpen, doi:10.58647/DRUGARXIV.PR000010.v1.
13.
Boseila et al., Throat spray formulated with virucidal Pharmaceutical excipients as an effective early prophylactic or treatment strategy against pharyngitis post-exposure to SARS CoV-2, European Journal of Pharmaceutics and Biopharmaceutics, doi:10.1016/j.ejpb.2024.114279.
14.
Hidayah et al., Bioinformatics study of curcumin, demethoxycurcumin, bisdemethoxycurcumin and cyclocurcumin compounds in Curcuma longa as an antiviral agent via nucleocapsid on SARS-CoV-2 inhibition, International Conference on Organic and Applied Chemistry, doi:10.1063/5.0197724.
15.
Singh et al., Unlocking the potential of phytochemicals in inhibiting SARS-CoV-2 M Pro protein - An in-silico and cell-based approach, Research Square, doi:10.21203/rs.3.rs-3888947/v1.
16.
Kant et al., Structure-based drug discovery to identify SARS-CoV2 spike protein–ACE2 interaction inhibitors, Journal of Biomolecular Structure and Dynamics, doi:10.1080/07391102.2023.2300060.
17.
Naderi Beni et al., In silico studies of anti-oxidative and hot temperament-based phytochemicals as natural inhibitors of SARS-CoV-2 Mpro, PLOS ONE, doi:10.1371/journal.pone.0295014.
18.
Moschovou et al., Exploring the Binding Effects of Natural Products and Antihypertensive Drugs on SARS-CoV-2: An In Silico Investigation of Main Protease and Spike Protein, International Journal of Molecular Sciences, doi:10.3390/ijms242115894.
19.
Eleraky et al., Curcumin Transferosome-Loaded Thermosensitive Intranasal in situ Gel as Prospective Antiviral Therapy for SARS-Cov-2, International Journal of Nanomedicine, doi:10.2147/IJN.S423251.
20.
Singh (B) et al., Computational studies to analyze effect of curcumin inhibition on coronavirus D614G mutated spike protein, The Seybold Report, doi:10.17605/OSF.IO/TKEXJ.
21.
Thapa et al., In-silico Approach for Predicting the Inhibitory Effect of Home Remedies on Severe Acute Respiratory Syndrome Coronavirus-2, Makara Journal of Science, doi:10.7454/mss.v27i3.1609.
22.
Srivastava et al., Paradigm of Well-Orchestrated Pharmacokinetic Properties of Curcuminoids Relative to Conventional Drugs for the Inactivation of SARS-CoV-2 Receptors: An In Silico Approach, Stresses, doi:10.3390/stresses3030043.
23.
Alkafaas et al., A study on the effect of natural products against the transmission of B.1.1.529 Omicron, Virology Journal, doi:10.1186/s12985-023-02160-6.
24.
Winih Kinasih et al., Analisis in silico interaksi senyawa kurkuminoid terhadap enzim main protease 6LU7 dari SARS-CoV-2, Duta Pharma Journal, doi:10.47701/djp.v3i1.2904.
25.
Wu (B) et al., Potential Mechanism of Curcumin and Resveratrol against SARS-CoV-2, Research Square, doi:10.21203/rs.3.rs-2780614/v1.
26.
Nag et al., Curcumin inhibits spike protein of new SARS-CoV-2 variant of concern (VOC) Omicron, an in silico study, Computers in Biology and Medicine, doi:10.1016/j.compbiomed.2022.105552.
27.
Rampogu et al., Molecular Docking and Molecular Dynamics Simulations Discover Curcumin Analogue as a Plausible Dual Inhibitor for SARS-CoV-2, International Journal of Molecular Sciences, doi:10.3390/ijms23031771.
28.
Singh (C) et al., Potential of turmeric-derived compounds against RNA-dependent RNA polymerase of SARS-CoV-2: An in-silico approach, Computers in Biology and Medicine, doi:10.1016/j.compbiomed.2021.104965.
29.
Kandeil et al., Bioactive Polyphenolic Compounds Showing Strong Antiviral Activities against Severe Acute Respiratory Syndrome Coronavirus 2, Pathogens, doi:10.3390/pathogens10060758.
30.
Rajagopal et al., Activity of phytochemical constituents of Curcuma longa (turmeric) and Andrographis paniculata against coronavirus (COVID-19): an in silico approach, Future Journal of Pharmaceutical Sciences, doi:10.1186/s43094-020-00126-x.
31.
Suravajhala et al., Comparative Docking Studies on Curcumin with COVID-19 Proteins, Preprints, doi:10.20944/preprints202005.0439.v3.
32.
Sekiou et al., In-Silico Identification of Potent Inhibitors of COVID-19 Main Protease (Mpro) and Angiotensin Converting Enzyme 2 (ACE2) from Natural Products: Quercetin, Hispidulin, and Cirsimaritin Exhibited Better Potential Inhibition than Hydroxy-Chloroquine Against COVID-19 Main Protease Active Site and ACE2, ChemRxiv, doi:10.26434/chemrxiv.12181404.v1.
33.
Grüneberg et al., Dose-dependent antiviral effects of glycyrrhizin, curcumin, and harmaline against clinical SARS-CoV-2 isolates, including D614G, Omicron BA.5, and Omicron XBB.1, BMC Complementary Medicine and Therapies, doi:10.1186/s12906-026-05253-1.
34.
Aktay et al., Oral Administration of Water-Soluble Curcumin Complex Prevents ARDS With the Potential for COVID-19 Treatment, Phytotherapy Research, doi:10.1002/ptr.70046.
35.
Olubiyi et al., Novel dietary herbal preparations with inhibitory activities against multiple SARS-CoV-2 targets: A multidisciplinary investigation into antiviral activities, Food Chemistry Advances, doi:10.1016/j.focha.2025.100969.
36.
Emam et al., Establishment of in-house assay for screening of anti-SARS-CoV-2 protein inhibitors, AMB Express, doi:10.1186/s13568-024-01739-8.
37.
Van Tin et al., Spike Protein of SARS-CoV-2 Activates Cardiac Fibrogenesis through NLRP3 Inflammasomes and NF-κB Signaling, Cells, doi:10.3390/cells13161331.
38.
Li et al., Thermal shift assay (TSA)-based drug screening strategy for rapid discovery of inhibitors against the Nsp13 helicase of SARS-CoV-2, Animals and Zoonoses, doi:10.1016/j.azn.2024.06.001.
39.
Kamble et al., Nanoparticulate curcumin spray imparts prophylactic and therapeutic properties against SARS-CoV-2, Emergent Materials, doi:10.1007/s42247-024-00754-6.
40.
Nicoliche et al., Antiviral, anti-inflammatory and antioxidant effects of curcumin and curcuminoids in SH-SY5Y cells infected by SARS-CoV-2, Scientific Reports, doi:10.1038/s41598-024-61662-7.
41.
Nittayananta et al., A novel film spray containing curcumin inhibits SARS-CoV-2 and influenza virus infection and enhances mucosal immunity, Virology Journal, doi:10.1186/s12985-023-02282-x.
42.
Septisetyani et al., Curcumin and turmeric extract inhibited SARS-CoV-2 pseudovirus cell entry and Spike mediated cell fusion, bioRxiv, doi:10.1101/2023.09.28.560070.
43.
Mohd Abd Razak et al., In Vitro Anti-SARS-CoV-2 Activities of Curcumin and Selected Phenolic Compounds, Natural Product Communications, doi:10.1177/1934578X231188861.
44.
Fam et al., Channel activity of SARS-CoV-2 viroporin ORF3a inhibited by adamantanes and phenolic plant metabolites, Scientific Reports, doi:10.1038/s41598-023-31764-9.
45.
Teshima et al., Antiviral activity of curcumin and its analogs selected by an artificial intelligence-supported activity prediction system in SARS-CoV-2-infected VeroE6 cells, Natural Product Research, doi:10.1080/14786419.2023.2194647.
46.
Zupin et al., Optimization of Anti-SARS-CoV-2 Treatments Based on Curcumin, Used Alone or Employed as a Photosensitizer, Viruses, doi:10.3390/v14102132.
47.
Leka et al., In vitro antiviral activity against SARS-CoV-2 of common herbal medicinal extracts and their bioactive compounds, Phytotherapy Research, doi:10.1002/ptr.7463.
48.
Goc et al., Inhibitory effects of specific combination of natural compounds against SARS-CoV-2 and its Alpha, Beta, Gamma, Delta, Kappa, and Mu variants, European Journal of Microbiology and Immunology, doi:10.1556/1886.2021.00022.
49.
Marín-Palma et al., Curcumin Inhibits In Vitro SARS-CoV-2 Infection In Vero E6 Cells through Multiple Antiviral Mechanisms, Molecules, doi:10.3390/molecules26226900.
50.
Bahun et al., Inhibition of the SARS-CoV-2 3CLpro main protease by plant polyphenols, Food Chemistry, doi:10.1016/j.foodchem.2021.131594.
51.
Bormann et al., Turmeric Root and Its Bioactive Ingredient Curcumin Effectively Neutralize SARS-CoV-2 In Vitro, Viruses, doi:10.3390/v13101914.
52.
Guijarro-Real et al., Potential In Vitro Inhibition of Selected Plant Extracts against SARS-CoV-2 Chymotripsin-Like Protease (3CLPro) Activity, Foods, doi:10.3390/foods10071503.
a.
The trimeric spike (S) protein is a glycoprotein that mediates viral entry by binding to the host ACE2 receptor, is critical for SARS-CoV-2's ability to infect host cells, and is a target of neutralizing antibodies. Inhibition of the spike protein prevents viral attachment, halting infection at the earliest stage.
b.
The receptor binding domain is a specific region of the spike protein that binds ACE2 and is a major target of neutralizing antibodies. Focusing on the precise binding site allows highly specific disruption of viral attachment with reduced potential for off-target effects.
c.
The main protease or Mpro, also known as 3CLpro or nsp5, is a cysteine protease that cleaves viral polyproteins into functional units needed for replication. Inhibiting Mpro disrupts the SARS-CoV-2 lifecycle within the host cell, preventing the creation of new copies.
d.
RNA-dependent RNA polymerase (RdRp), also called nsp12, is the core enzyme of the viral replicase-transcriptase complex that copies the positive-sense viral RNA genome into negative-sense templates for progeny RNA synthesis. Inhibiting RdRp blocks viral genome replication and transcription.
e.
The papain-like protease (PLpro) has multiple functions including cleaving viral polyproteins and suppressing the host immune response by deubiquitination and deISGylation of host proteins. Inhibiting PLpro may block viral replication and help restore normal immune responses.
f.
The angiotensin converting enzyme 2 (ACE2) protein is a host cell transmembrane protein that serves as the cellular receptor for the SARS-CoV-2 spike protein. ACE2 is expressed on many cell types, including epithelial cells in the lungs, and allows the virus to enter and infect host cells. Inhibition may affect ACE2's physiological function in blood pressure control.
g.
The nucleocapsid (N) protein binds and encapsulates the viral genome by coating the viral RNA. N enables formation and release of infectious virions and plays additional roles in viral replication and pathogenesis. N is also an immunodominant antigen used in diagnostic assays.
h.
Non-structural protein 10 (nsp10) serves as an RNA chaperone and stabilizes conformations of nsp12 and nsp14 in the replicase-transcriptase complex, which synthesizes new viral RNAs. Nsp10 disruption may destabilize replicase-transcriptase complex activity.
i.
The helicase, or nsp13, protein unwinds the double-stranded viral RNA, a crucial step in replication and transcription. Inhibition may prevent viral genome replication and the creation of new virus components.
j.
The interaction between the SARS-CoV-2 spike protein and the human ACE2 receptor is a primary method of viral entry, inhibiting this interaction can prevent the virus from attaching to and entering host cells, halting infection at an early stage.
k.
Transmembrane protease serine 2 (TMPRSS2) is a host cell protease that primes the spike protein, facilitating cellular entry. TMPRSS2 activity helps enable cleavage of the spike protein required for membrane fusion and virus entry. Inhibition may especially protect respiratory epithelial cells, buy may have physiological effects.
l.
Calu-3 is a human lung adenocarcinoma cell line with moderate ACE2 and TMPRSS2 expression and SARS-CoV-2 susceptibility. It provides a model of the human respiratory epithelium, but many not be ideal for modeling early stages of infection due to the moderate expression levels of ACE2 and TMPRSS2.
m.
A549 is a human lung carcinoma cell line with low ACE2 expression and SARS-CoV-2 susceptibility. Viral entry/replication can be studied but the cells may not replicate all aspects of lung infection.
n.
A549-AT is a human lung carcinoma cell line stably transfected with ACE2 and TMPRSS2 receptors. Unlike the parental line, this overexpression ensures stable infection and enhanced viral entry, allowing for the evaluation of antiviral efficacy against various SARS-CoV-2 variants.
o.
293T is a human embryonic kidney cell line that can be engineered for high ACE2 expression and SARS-CoV-2 susceptibility. 293T cells are easily transfected and support high protein expression.
p.
HEK293-hACE2 is a human embryonic kidney cell line with high ACE2 expression and SARS-CoV-2 susceptibility. Cells have been transfected with a plasmid to express the human ACE2 (hACE2) protein.
q.
293T/hACE2/TMPRSS2 is a human embryonic kidney cell line engineered for high ACE2 and TMPRSS2 expression, which mimics key aspects of human infection. 293T/hACE2/TMPRSS2 cells are very susceptible to SARS-CoV-2 infection.
r.
Vero E6 is an African green monkey kidney cell line with low/no ACE2 expression and high SARS-CoV-2 susceptibility. The cell line is easy to maintain and supports robust viral replication, however the monkey origin may not accurately represent human responses.
s.
SH-SY5Y is a human neuroblastoma cell line that exhibits neuronal phenotypes. It is commonly used as an in vitro model for studying neurotoxicity, neurodegenerative diseases, and neuronal differentiation.
Teshima et al., 28 Mar 2023, peer-reviewed, 10 authors.
In vitro studies are an important part of preclinical research, however results may be very different in vivo.
Supplemental online material Antiviral activity of curcumin and its analogs selected by an artificial intelligencesupported activity prediction system in SARS-CoV-2-infected VeroE6 cells
Curcumin has been reported to exert its anti-SARS-CoV-2 activity by inhibiting the binding of spike receptor-binding domain (RBD) to angiotensin-converting enzyme-2 (ACE2). To identify more potent compounds, we evaluated the antiviral activities of curcumin and its analogs in SARS-CoV-2-infected cells. An artificial intelligence-supported activity prediction system was used to select the compounds, and 116 of the 334 curcumin analogs were proposed to have spike RBD-ACE2 binding inhibitory activity. These compounds were narrowed down to eight compounds for confirmatory studies. Six out of the eight compounds showed antiviral activity with EC50 values of less than 30 µM and binding inhibitory activity with IC20 values of less than 30 µM. Structure-activity relationship analyses revealed that the double bonds in the carbon chain connecting the two phenolic groups were essential for both activities. X-ray co-crystallography studies are needed to clarify the true binding pose and design more potent derivatives.
Cells African green monkey kidney cell line VeroE6 and VeroE6 expressing the transmembrane serine protease TMPRSS2 (VeroE6/TMPRSS2, Matsuyama et al. 2020) were purchased from the National Institutes of Biomedical Innovation, Health and Nutrition, Osaka, Japan. VeroE6 cells were cultured in minimal essential medium (MEM; Sigma-Aldrich, MO, USA) supplemented with 10% fetal bovine serum (FBS) and antibiotics. VeroE6/TMPRSS2 cells were cultured in Dulbecco's modified Eagle's medium (DMEM; Thermo Fisher Scientific, MA, USA) supplemented with 5% FBS and 1 mg/mL G418 (Nacalai, Kyoto, Japan). The cells were grown at 37°C in a CO2 incubator. Virus SARS-CoV-2 viral strain JPN/TY/WK/521 was provided by the National Institute of Infectious Diseases, Tokyo, Japan. Viruses were propagated in monolayers of VeroE6/TMPRSS2 cells in DMEM supplemented with 2% FBS and 1 mg/mL G418 at a multiplicity of infection of 0.01. Virus titration Infectivity was titrated by a focus-forming assay as described previously (Yasugi et al. 2013 ) with slight modifications. Viruses were serially diluted 10-fold in DMEM supplemented with 2% FBS and infected with confluent VeroE6 cells for 8 h. The cells were fixed with 4% paraformaldehyde for 15 min and washed with phosphate-buffered saline (PBS) three times. After the cells were permeabilized with 0.1% Triton ® X-100 for 15 min and washed with PBS three times, they were incubated with rabbit anti-nucleocapsid monoclonal antibody (Thermo Fisher Scientific) at a..
References
Kanda, Investigation of the freely available easy-to-use software 'EZR' for medical statistics, Bone Marrow Transplant
Komatsu, Ikeda, Tanaka, Matsuzaki, Development of practical artificial intelligence system for drug discovery and its application to activity prediction of small molecule protein-protein interaction modulators, J Biol Macromol
Komatsu, Tanaka, Matsuzaki, Efficient drug discovery approach for PPI and ubiquitin-proteasome targets with unique in silico screening and AI-based activity prediction technologies, Medical Science Digest
Komatsu, Tanaka, Ye, Ikeda, Matsuzaki et al., Identification of SARS-CoV-2 main protease inhibitors from FDA-approved drugs by artificial intelligence-supported activity prediction system, J Biomol Struct Dyn
Matsuyama, Nao, Shirato, Kawase, Saito et al., Enhanced isolation of SARS-CoV-2 by TMPRSS2-expressing cells, Proc Natl Acad Sci
Sato, Kashiwakura, Yamaguchi, Yoshino, Tanaka et al., Discovery of a novel small-molecule interleukin-6 inhibitor through virtual screening using artificial intelligence, Med Chem
Yasugi, Kubota-Koketsu, Yamashita, Kawashita, Du et al., Human monoclonal antibodies broadly neutralizing against influenza B virus, PLoS Pathog, doi:10.1371/journal.ppat.1003150
DOI record:
{
"DOI": "10.1080/14786419.2023.2194647",
"ISSN": [
"1478-6419",
"1478-6427"
],
"URL": "http://dx.doi.org/10.1080/14786419.2023.2194647",
"alternative-id": [
"10.1080/14786419.2023.2194647"
],
"assertion": [
{
"label": "Peer Review Statement",
"name": "peerreview_statement",
"order": 1,
"value": "The publishing and review policy for this title is described in its Aims & Scope."
},
{
"URL": "http://www.tandfonline.com/action/journalInformation?show=aimsScope&journalCode=gnpl20",
"label": "Aim & Scope",
"name": "aims_and_scope_url",
"order": 2,
"value": "http://www.tandfonline.com/action/journalInformation?show=aimsScope&journalCode=gnpl20"
},
{
"group": {
"label": "Publication History",
"name": "publication_history"
},
"label": "Received",
"name": "received",
"order": 0,
"value": "2022-11-28"
},
{
"group": {
"label": "Publication History",
"name": "publication_history"
},
"label": "Revised",
"name": "revised",
"order": 1,
"value": "2023-02-26"
},
{
"group": {
"label": "Publication History",
"name": "publication_history"
},
"label": "Accepted",
"name": "accepted",
"order": 2,
"value": "2023-03-17"
},
{
"group": {
"label": "Publication History",
"name": "publication_history"
},
"label": "Published",
"name": "published",
"order": 3,
"value": "2023-03-28"
}
],
"author": [
{
"ORCID": "http://orcid.org/0000-0003-3524-100X",
"affiliation": [
{
"name": "Research & Development Headquarters, Lequio Pharma Co., Ltd, Naha, Japan"
}
],
"authenticated-orcid": false,
"family": "Teshima",
"given": "Koji",
"sequence": "first"
},
{
"ORCID": "http://orcid.org/0000-0001-9505-9934",
"affiliation": [
{
"name": "Interprotein Corporation, Osaka, Japan"
}
],
"authenticated-orcid": false,
"family": "Tanaka",
"given": "Takeshi",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Interprotein Corporation, Osaka, Japan"
}
],
"family": "Ye",
"given": "Zhengmao",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Interprotein Corporation, Osaka, Japan"
}
],
"family": "Ikeda",
"given": "Ken",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Interprotein Corporation, Osaka, Japan"
}
],
"family": "Matsuzaki",
"given": "Takao",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Research & Development Headquarters, Lequio Pharma Co., Ltd, Naha, Japan"
}
],
"family": "Shiroma",
"given": "Tamotsu",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Interprotein Corporation, Osaka, Japan"
}
],
"family": "Muroya",
"given": "Ayumu",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Interprotein Corporation, Osaka, Japan"
}
],
"family": "Hosoda",
"given": "Masato",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-9688-2006",
"affiliation": [
{
"name": "Graduate School of Veterinary Science, Osaka Metropolitan University, Osaka, Japan"
},
{
"name": "Asian Health Science Institute, Osaka Metropolitan University, Osaka, Japan"
},
{
"name": "Osaka International Research Center for Infectious Diseases, Osaka Metropolitan University, Izumisano, Japan"
}
],
"authenticated-orcid": false,
"family": "Yasugi",
"given": "Mayo",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-4206-2416",
"affiliation": [
{
"name": "Interprotein Corporation, Osaka, Japan"
}
],
"authenticated-orcid": false,
"family": "Komatsu",
"given": "Hirotsugu",
"sequence": "additional"
}
],
"container-title": "Natural Product Research",
"container-title-short": "Natural Product Research",
"content-domain": {
"crossmark-restriction": true,
"domain": [
"www.tandfonline.com"
]
},
"created": {
"date-parts": [
[
2023,
3,
29
]
],
"date-time": "2023-03-29T06:50:52Z",
"timestamp": 1680072652000
},
"deposited": {
"date-parts": [
[
2023,
4,
2
]
],
"date-time": "2023-04-02T23:50:17Z",
"timestamp": 1680479417000
},
"indexed": {
"date-parts": [
[
2023,
4,
3
]
],
"date-time": "2023-04-03T04:41:33Z",
"timestamp": 1680496893849
},
"is-referenced-by-count": 0,
"issued": {
"date-parts": [
[
2023,
3,
28
]
]
},
"language": "en",
"link": [
{
"URL": "https://www.tandfonline.com/doi/pdf/10.1080/14786419.2023.2194647",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "301",
"original-title": [],
"page": "1-6",
"prefix": "10.1080",
"published": {
"date-parts": [
[
2023,
3,
28
]
]
},
"published-online": {
"date-parts": [
[
2023,
3,
28
]
]
},
"publisher": "Informa UK Limited",
"reference": [
{
"DOI": "10.3390/ph14100954",
"doi-asserted-by": "publisher",
"key": "CIT0001"
},
{
"DOI": "10.1080/14786419.2022.2057492",
"doi-asserted-by": "publisher",
"key": "CIT0002"
},
{
"DOI": "10.1016/j.metop.2021.100141",
"doi-asserted-by": "publisher",
"key": "CIT0003"
},
{
"DOI": "10.1371/journal.pone.0253489",
"doi-asserted-by": "publisher",
"key": "CIT0004"
},
{
"DOI": "10.1038/s41598-021-88218-3",
"doi-asserted-by": "publisher",
"key": "CIT0005"
},
{
"DOI": "10.1080/07391102.2021.2024260",
"doi-asserted-by": "publisher",
"key": "CIT0006"
},
{
"author": "Malabadi RB",
"first-page": "135",
"issue": "2",
"journal-title": "Int J Res Sci Innov",
"key": "CIT0007",
"volume": "8",
"year": "2021"
},
{
"DOI": "10.3390/molecules26226900",
"doi-asserted-by": "publisher",
"key": "CIT0008"
},
{
"DOI": "10.1016/j.compbiomed.2021.104818",
"doi-asserted-by": "publisher",
"key": "CIT0009"
},
{
"DOI": "10.1080/07391102.2020.1868338",
"doi-asserted-by": "publisher",
"key": "CIT0010"
},
{
"DOI": "10.3389/fphar.2021.675287",
"doi-asserted-by": "publisher",
"key": "CIT0012"
},
{
"DOI": "10.1080/14786419.2021.1925894",
"doi-asserted-by": "publisher",
"key": "CIT0013"
},
{
"DOI": "10.1039/D0RA03167D",
"doi-asserted-by": "publisher",
"key": "CIT0014"
},
{
"DOI": "10.3390/nu14020256",
"doi-asserted-by": "publisher",
"key": "CIT0015"
}
],
"reference-count": 14,
"references-count": 14,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.tandfonline.com/doi/full/10.1080/14786419.2023.2194647"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"Organic Chemistry",
"Plant Science",
"Biochemistry",
"Analytical Chemistry"
],
"subtitle": [],
"title": "Antiviral activity of curcumin and its analogs selected by an artificial intelligence-supported activity prediction system in SARS-CoV-2-infected VeroE6 cells",
"type": "journal-article",
"update-policy": "http://dx.doi.org/10.1080/tandf_crossmark_01"
}
