SARS-CoV-2 infects human primary cytotrophoblasts mainly through a non-canonical entry route
et al., Molecular Human Reproduction, doi:10.1093/molehr/gaag015, Feb 2026
HCQ for COVID-19
1st treatment shown to reduce risk in
March 2020, now with p < 0.00000000001 from 424 studies, used in 59 countries.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
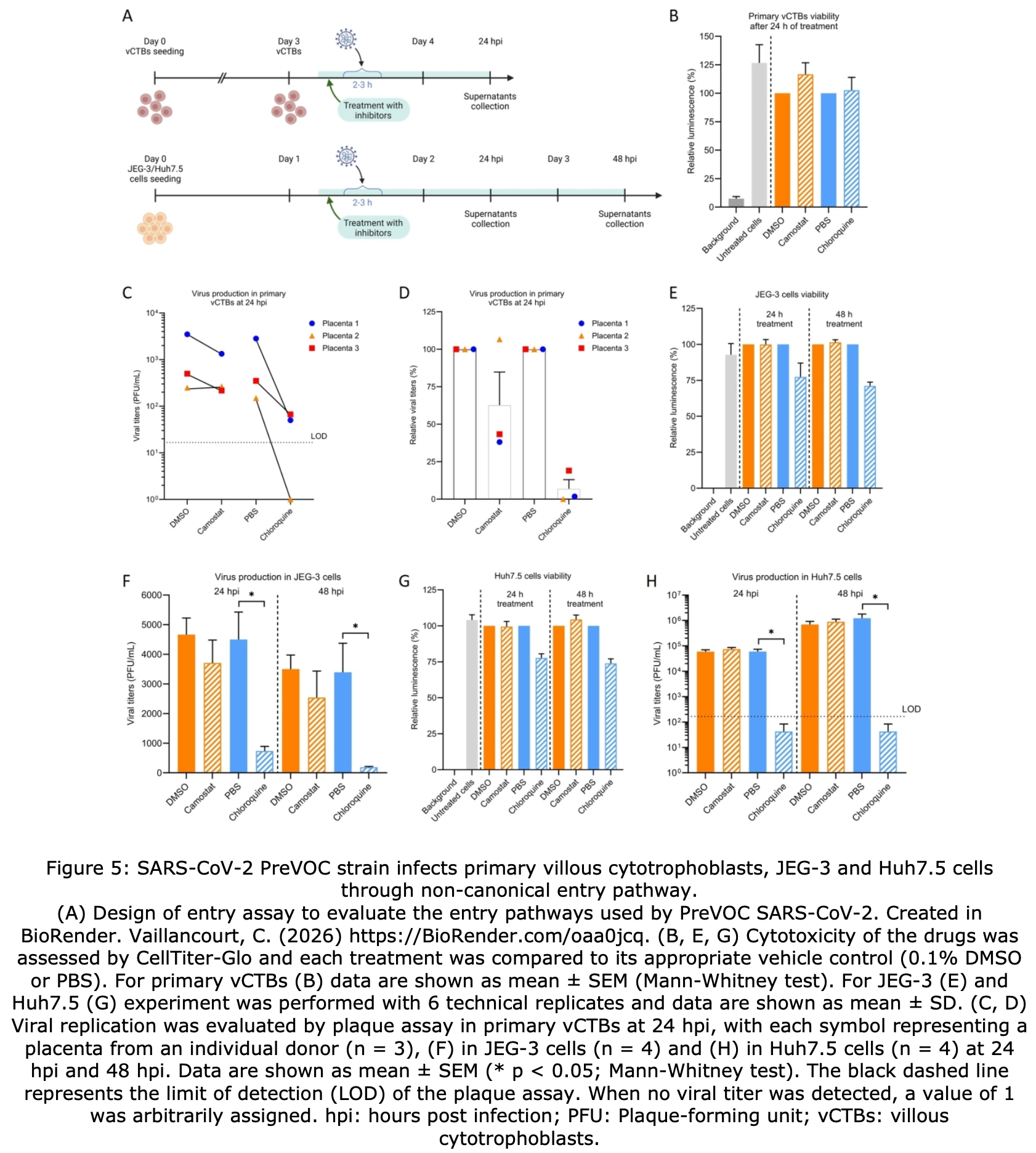
In vitro study showing that human primary cytotrophoblasts are permissive to SARS-CoV-2 infection, with potential implications for antiviral treatment during pregnancy. Authors tested primary villous cytotrophoblasts isolated from term placentas and found they support complete viral replication cycles for the ancestral SARS-CoV-2 strain, with viral entry occurring predominantly through a non-canonical endosomal pathway. Authors found that chloroquine phosphate, which blocks endosomal acidification, reduced viral production by 96-100% in primary cells, while camostat mesylate (TMPRSS2 inhibitor) had minimal effect, confirming predominant endosomal entry.
40 preclinical studies support the efficacy of HCQ for COVID-19:
1.
Shang et al., Identification of Cathepsin L as the molecular target of hydroxychloroquine with chemical proteomics, Molecular & Cellular Proteomics, doi:10.1016/j.mcpro.2025.101314.
2.
González-Paz et al., Biophysical Analysis of Potential Inhibitors of SARS-CoV-2 Cell Recognition and Their Effect on Viral Dynamics in Different Cell Types: A Computational Prediction from In Vitro Experimental Data, ACS Omega, doi:10.1021/acsomega.3c06968.
3.
Alkafaas et al., A study on the effect of natural products against the transmission of B.1.1.529 Omicron, Virology Journal, doi:10.1186/s12985-023-02160-6.
4.
Guimarães Silva et al., Are Non-Structural Proteins From SARS-CoV-2 the Target of Hydroxychloroquine? An in Silico Study, ACTA MEDICA IRANICA, doi:10.18502/acta.v61i2.12533.
5.
Nguyen et al., The Potential of Ameliorating COVID-19 and Sequelae From Andrographis paniculata via Bioinformatics, Bioinformatics and Biology Insights, doi:10.1177/11779322221149622.
7.
Yadav et al., Repurposing the Combination Drug of Favipiravir, Hydroxychloroquine and Oseltamivir as a Potential Inhibitor Against SARS-CoV-2: A Computational Study, Research Square, doi:10.21203/rs.3.rs-628277/v1.
8.
Hussein et al., Molecular Docking Identification for the efficacy of Some Zinc Complexes with Chloroquine and Hydroxychloroquine against Main Protease of COVID-19, Journal of Molecular Structure, doi:10.1016/j.molstruc.2021.129979.
9.
Baildya et al., Inhibitory capacity of Chloroquine against SARS-COV-2 by effective binding with Angiotensin converting enzyme-2 receptor: An insight from molecular docking and MD-simulation studies, Journal of Molecular Structure, doi:10.1016/j.molstruc.2021.129891.
10.
Noureddine et al., Quantum chemical studies on molecular structure, AIM, ELF, RDG and antiviral activities of hybrid hydroxychloroquine in the treatment of COVID-19: molecular docking and DFT calculations, Journal of King Saud University - Science, doi:10.1016/j.jksus.2020.101334.
11.
Tarek et al., Pharmacokinetic Basis of the Hydroxychloroquine Response in COVID-19: Implications for Therapy and Prevention, European Journal of Drug Metabolism and Pharmacokinetics, doi:10.1007/s13318-020-00640-6.
12.
Rowland Yeo et al., Impact of Disease on Plasma and Lung Exposure of Chloroquine, Hydroxychloroquine and Azithromycin: Application of PBPK Modeling, Clinical Pharmacology & Therapeutics, doi:10.1002/cpt.1955.
13.
Pinatel et al., SARS-CoV-2 infects human primary cytotrophoblasts mainly through a non-canonical entry route, Molecular Human Reproduction, doi:10.1093/molehr/gaag015.
14.
Hitti et al., Hydroxychloroquine attenuates double-stranded RNA-stimulated hyper-phosphorylation of tristetraprolin/ZFP36 and AU-rich mRNA stabilization, Immunology, doi:10.1111/imm.13835.
15.
Yan et al., Super-resolution imaging reveals the mechanism of endosomal acidification inhibitors against SARS-CoV-2 infection, ChemBioChem, doi:10.1002/cbic.202400404.
16.
Mohd Abd Razak et al., In Vitro Anti-SARS-CoV-2 Activities of Curcumin and Selected Phenolic Compounds, Natural Product Communications, doi:10.1177/1934578X231188861.
17.
Alsmadi et al., The In Vitro, In Vivo, and PBPK Evaluation of a Novel Lung-Targeted Cardiac-Safe Hydroxychloroquine Inhalation Aerogel, AAPS PharmSciTech, doi:10.1208/s12249-023-02627-3.
18.
Wen et al., Cholinergic α7 nAChR signaling suppresses SARS-CoV-2 infection and inflammation in lung epithelial cells, Journal of Molecular Cell Biology, doi:10.1093/jmcb/mjad048.
19.
Kamga Kapchoup et al., In vitro effect of hydroxychloroquine on pluripotent stem cells and their cardiomyocytes derivatives, Frontiers in Pharmacology, doi:10.3389/fphar.2023.1128382.
20.
Milan Bonotto et al., Cathepsin inhibitors nitroxoline and its derivatives inhibit SARS-CoV-2 infection, Antiviral Research, doi:10.1016/j.antiviral.2023.105655.
21.
Miao et al., SIM imaging resolves endocytosis of SARS-CoV-2 spike RBD in living cells, Cell Chemical Biology, doi:10.1016/j.chembiol.2023.02.001.
22.
Yuan et al., Hydroxychloroquine blocks SARS-CoV-2 entry into the endocytic pathway in mammalian cell culture, Communications Biology, doi:10.1038/s42003-022-03841-8.
23.
Faísca et al., Enhanced In Vitro Antiviral Activity of Hydroxychloroquine Ionic Liquids against SARS-CoV-2, Pharmaceutics, doi:10.3390/pharmaceutics14040877.
24.
Delandre et al., Antiviral Activity of Repurposing Ivermectin against a Panel of 30 Clinical SARS-CoV-2 Strains Belonging to 14 Variants, Pharmaceuticals, doi:10.3390/ph15040445.
25.
Purwati et al., An in vitro study of dual drug combinations of anti-viral agents, antibiotics, and/or hydroxychloroquine against the SARS-CoV-2 virus isolated from hospitalized patients in Surabaya, Indonesia, PLOS One, doi:10.1371/journal.pone.0252302.
26.
Zhang et al., SARS-CoV-2 spike protein dictates syncytium-mediated lymphocyte elimination, Cell Death & Differentiation, doi:10.1038/s41418-021-00782-3.
27.
Dang et al., Structural basis of anti-SARS-CoV-2 activity of hydroxychloroquine: specific binding to NTD/CTD and disruption of LLPS of N protein, bioRxiv, doi:10.1101/2021.03.16.435741.
28.
Shang (B) et al., Inhibitors of endosomal acidification suppress SARS-CoV-2 replication and relieve viral pneumonia in hACE2 transgenic mice, Virology Journal, doi:10.1186/s12985-021-01515-1.
29.
Wang et al., Chloroquine and hydroxychloroquine as ACE2 blockers to inhibit viropexis of 2019-nCoV Spike pseudotyped virus, Phytomedicine, doi:10.1016/j.phymed.2020.153333.
30.
Sheaff, R., A New Model of SARS-CoV-2 Infection Based on (Hydroxy)Chloroquine Activity, bioRxiv, doi:10.1101/2020.08.02.232892.
31.
Ou et al., Hydroxychloroquine-mediated inhibition of SARS-CoV-2 entry is attenuated by TMPRSS2, PLOS Pathogens, doi:10.1371/journal.ppat.1009212.
32.
Andreani et al., In vitro testing of combined hydroxychloroquine and azithromycin on SARS-CoV-2 shows synergistic effect, Microbial Pathogenesis, doi:10.1016/j.micpath.2020.104228.
33.
Clementi et al., Combined Prophylactic and Therapeutic Use Maximizes Hydroxychloroquine Anti-SARS-CoV-2 Effects in vitro, Front. Microbiol., 10 July 2020, doi:10.3389/fmicb.2020.01704.
34.
Liu et al., Hydroxychloroquine, a less toxic derivative of chloroquine, is effective in inhibiting SARS-CoV-2 infection in vitro, Cell Discovery 6, 16 (2020), doi:10.1038/s41421-020-0156-0.
35.
Yao et al., In Vitro Antiviral Activity and Projection of Optimized Dosing Design of Hydroxychloroquine for the Treatment of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2), Clin. Infect. Dis., 2020 Mar 9, doi:10.1093/cid/ciaa237.
Pinatel et al., 26 Feb 2026, Canada, peer-reviewed, 9 authors.
Contact: cathy.vaillancourt@inrs.ca, laurent.chatel-chaix@inrs.ca.
In vitro studies are an important part of preclinical research, however results may be very different in vivo.
SARS-CoV-2 infects human primary cytotrophoblasts mainly through a noncanonical entry route
doi:10.1093/molehr/gaag015/8500690
Since the beginning of the COVID-19 pandemic, vulnerable populations such as pregnant persons have been at higher risk of severe symptoms and poor outcomes. Although reports of SARS-CoV-2 vertical transmission remain rare, several studies showed that maternal infection during pregnancy can induce histomorphological and inflammatory alterations in the placenta. However, the permissiveness of human trophoblasts to various variants of the virus remains poorly characterized. In this study, human primary villous cytotrophoblasts isolated from term placentas, along with trophoblastic cell lines BeWo, JEG-3, and HIPEC-65 were infected with the ancestral SARS-CoV-2 strain, which disseminated worldwide in early 2020. Permissiveness was assessed with quantitative RT-PCR, immunostaining of viral protein Nucleocapsid, and plaque assays. To investigate viral entry routes, cells were treated with Camostat mesylate (an inhibitor of the co-entry factor TMPRSS2) or chloroquine phosphate (an endosomal entry inhibitor) and viral fitness was assessed by plaque assays. Primary villous cytotrophoblasts and JEG-3 cells were also tested for infection with three pre-omicron SARS-CoV-2 variants of concern. Our results show that primary villous cytotrophoblasts are permissive to all tested SARS-CoV-2 strains in vitro. Infection with the ancestral SARS-CoV-2 strain relies mainly on a non-canonical endosomal entry pathway. Notably, JEG-3 cells represent an appropriate and convenient model for studying trophoblast infection by SARS-CoV-2, as they exhibit high permissiveness to the ancestral strain, and the SARS-CoV-2 entry pathway is similar to that in villous cytotrophoblasts. Overall, this study reveals that the cytotrophoblastic permissiveness to SARS-CoV-2 depends on the viral genetic nature and provides new insights into its entry route in human trophoblasts.
Authors' roles
Conflict of interest The authors declare no conflict of interests.
References
Agarwal, Hunt, Stegemann, Rochwerg, Lamontagne et al., A living WHO guideline on drugs for covid-19, BMJ
Al-Nasiry, Spitz, Hanssens, Luyten, Pijnenborg, Differential effects of inducers of syncytialization and apoptosis on BeWo and JEG-3 choriocarcinoma cells, Human Reproduction
Allotey, Stallings, Bonet, Yap, Chatterjee et al., Clinical manifestations, risk factors, and maternal and perinatal outcomes of coronavirus disease 2019 in pregnancy: living systematic review and meta-analysis, BMJ
Andrews, FastQC: a quality control tool for high throughput sequence data
Argueta, Lacko, Bram, Tada, Carrau et al., Inflammatory responses in the placenta upon SARS-CoV-2 infection late in pregnancy, iScience
Benlarbi, Laroche, Fink, Fu, Mulloy et al., Identification and differential usage of a host metalloproteinase entry pathway by SARS-CoV-2 Delta and Omicron, iScience
Bloise, Zhang, Nakpu, Hamada, Dunk et al., Expression of severe acute respiratory syndrome coronavirus 2 cell entry genes, angiotensin-converting enzyme 2 and transmembrane protease serine 2, in the placenta across gestation and at the maternalfetal interface in pregnancies complicated by preterm birth or preeclampsia, American Journal of Obstetrics and Gynecology
Borges, Bose, Frank, Kaufmann, Pötgens, A two-colour fluorescence assay for the measurement of syncytial fusion between trophoblast-derived cell lines, Placenta
Bray, Pimentel, Melsted, Pachter, Near-optimal probabilistic RNA-seq quantification, Nat Biotechnol
Brien, Bernier, Clavel, Kebiche, Jacob et al., Mediators of inflammation at the maternal-fetal interface are altered by SARS-CoV-2 infection and pandemic stress, Am J Reprod Immunol
Brien, Soglio, Soglio, Couture, Boucoiran et al., Pandemic stress and SARS-CoV-2 infection are associated with pathological changes at the maternal-fetal interface, Placenta
Cantuti-Castelvetri, Ojha, Pedro, Djannatian, Franz et al., Neuropilin-1 facilitates SARS-CoV-2 cell entry and infectivity, Science
Carlson, Simeone, Ellington, Galang, Desisto et al., Pre-Delta, Delta, and Omicron Periods of the Coronavirus Disease 2019 (COVID-19) Pandemic and Health Outcomes During Delivery Hospitalization, Obstet Gynecol
Cdc, Centers for Disease Control and Prevention : Similarities and differences between flu and COVID-19
Chen, Neil, Tan, Rudraraju, Mohenska et al., A placental model of SARS-CoV-2 infection reveals ACE2-dependent susceptibility and differentiation impairment in syncytiotrophoblasts, Nat Cell Biol
Chen, Zhou, Chen, Gu, fastp: an ultra-fast all-in-one FASTQ preprocessor, Bioinformatics
Clabault, Cohen, Vaillancourt, Sanderson, Effects of selective serotonin-reuptake inhibitors (SSRIs) in JEG-3 and HIPEC cell models of the extravillous trophoblast, Placenta
Corman, Landt, Kaiser, Molenkamp, Meijer et al., Detection of 2019 novel coronavirus (2019-nCoV) by real-time RT-PCR, Euro Surveill
Correia, Marçal, Vieira, Santos, Novais et al., Congenital SARS-CoV-2 Infection in a Neonate With Severe Acute Respiratory Syndrome, Pediatr Infect Dis J
Daly, Simonetti, Klein, Chen, Williamson et al., Neuropilin-1 is a host factor for SARS-CoV-2 infection, Science
Desisto, Wb, Simeone, Risk for Stillbirth Among Women With and Without COVID-19 at Delivery Hospitalization -United States
Dittmar, Lee, Whig, Segrist, Li et al., Drug repurposing screens reveal cell-type-specific entry pathways and FDA-approved drugs active against SARS-Cov-2, Cell Rep
Dubey, Choudhary, Kumar, Tomar, Emerging SARS-CoV-2 Variants: Genetic Variability and Clinical Implications, Curr Microbiol
Ellington, Strid, Tong, Woodworth, Galang et al., Characteristics of Women of Reproductive Age with Laboratory-Confirmed SARS-CoV-2 Infection by Pregnancy Status -United States, January 22-June 7, 2020, MMWR Morb Mortal Wkly Rep
Ewels, Magnusson, Lundin, Käller, MultiQC: summarize analysis results for multiple tools and samples in a single report, Bioinformatics
Fahmi, Brügger, Démoulins, Zumkehr, Esteves et al., SARS-CoV-2 can infect and propagate in human placenta explants, Cell Reports Medicine
Flores-Vega, Monroy-Molina, Jiménez-Hernández, Torres, Santos-Preciado et al., SARS-CoV-2: Evolution and Emergence of New Viral Variants, Viruses
Harvey, Carabelli, Jackson, Gupta, Thomson et al., SARS-CoV-2 variants, spike mutations and immune escape, Nat Rev Microbiol
Hertz, Choriocarcinoma of women maintained in serial passage in hamster and rat, Proc Soc Exp Biol Med
Hoffmann, Kleine-Weber, Schroeder, Krüger, Herrler et al., SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor, Cell
Holmes, The Emergence and Evolution of SARS-CoV-2, Annu Rev Virol
Huang, Li, Ma, Transforming growth factor β1 promotes invasion of human JEG-3 trophoblast cells via TGF-β/Smad3 signaling pathway, Oncotarget
Kallol, Martin-Sancho, Morey, Aisagbonhi, Pizzo et al., Activation of the interferon pathway in trophoblast cells productively infected with SARS-CoV-2, Stem Cells Dev
Karmakar, Dhar, Das, Inhibition of cytotrophoblastic (JEG-3) cell invasion by interleukin 12 involves an interferon gamma-mediated pathway, J Biol Chem
Keyaerts, Vijgen, Maes, Neyts, Van Ranst, In vitro inhibition of severe acute respiratory syndrome coronavirus by chloroquine, Biochem Biophys Res Commun
Kliman, Nestler, Sermasi, Sanger, Strauss, Purification, characterization, and in vitro differentiation of cytotrophoblasts from human term placentae, Endocrinology
Koch, Uckeley, Doldan, Stanifer, Boulant et al., TMPRSS2 expression dictates the entry route used by SARS-CoV-2 to infect host cells, Embo j
Kohler, Bridson, Isolation of hormone-producing clonal lines of human choriocarcinoma, J Clin Endocrinol Metab
Korber, Fischer, Gnanakaran, Yoon, Theiler et al., Tracking Changes in SARS-CoV-2 Spike: Evidence that D614G Increases Infectivity of the COVID-19 Virus, Cell
Kotlyar, Grechukhina, Chen, Popkhadze, Grimshaw et al., Vertical transmission of coronavirus disease 2019: a systematic review and meta-analysis, Am J Obstet Gynecol
Lanoix, Beghdadi, Lafond, Vaillancourt, Human placental trophoblasts synthesize melatonin and express its receptors, J Pineal Res
Lapehn, Nair, Firsick, Macdonald, Thoreson et al., A transcriptomic comparison of in vitro models of the human placenta, Placenta
Letko, Marzi, Munster, Functional assessment of cell entry and receptor usage for SARS-CoV-2 and other lineage B betacoronaviruses, Nat Microbiol
Li, Chen, Zhang, Xiong, Li, The SARS-CoV-2 receptor ACE2 expression of maternal-fetal interface and fetal organs by single-cell transcriptome study, PLoS One
Lu-Culligan, Chavan, Vijayakumar, Irshaid, Courchaine et al., Maternal respiratory SARS-CoV-2 infection in pregnancy is associated with a robust inflammatory response at the maternal-fetal interface, Med (N Y)
Mantzourani, Vasilakaki, Gerogianni, Kokotos, The discovery and development of transmembrane serine protease 2 (TMPRSS2) inhibitors as candidate drugs for the treatment of COVID-19, Expert Opin Drug Discov
Mcclymont, Albert, Alton, Boucoiran, Castillo et al., Association of SARS-CoV-2 Infection During Pregnancy With Maternal and Perinatal Outcomes, Jama
Morales, Cortés, Hernández, Guiot, Torres et al., Vertical transmission: evidence of COVID-19 in a twin pregnancy, JBRA Assist Reprod
Orendi, Gauster, Moser, Meiri, Huppertz, The choriocarcinoma cell line BeWo: syncytial fusion and expression of syncytium-specific proteins, REPRODUCTION
Ou, Liu, Lei, Li, Mi et al., Characterization of spike glycoprotein of SARS-CoV-2 on virus entry and its immune cross-reactivity with SARS-CoV, Nat Commun
Ouyang, Bagalkot, Fitzgerald, Sadovsky, Chu et al., Term Human Placental Trophoblasts Express SARS-CoV-2 Entry Factors ACE2, TMPRSS2, and Furin, mSphere
Owen, Allerton, Anderson, Aschenbrenner, Avery et al., An oral SARS-CoV-2 M(pro) inhibitor clinical candidate for the treatment of COVID-19, Science
Pastuschek, Nonn, Gutiérrez-Samudio, Murrieta-Coxca, Müller et al., Molecular characteristics of established trophoblast-derived cell lines, Placenta
Pattillo, Gey, The Establishment of a Cell Line of Human Hormone-synthesizing Trophoblastic Cells in Vitro1, Cancer Research
Pavan, Tarrade, Hermouet, Delouis, Titeux et al., Human invasive trophoblasts transformed with simian virus 40 provide a new tool to study the role of PPARgamma in cell invasion process, Carcinogenesis
Pringle, Tadros, Callister, Lumbers, The expression and localization of the human placental prorenin/renin-angiotensin system throughout pregnancy: Roles in trophoblast invasion and angiogenesis?, Placenta
Ramirez, Fernandez-Antunez, Galli, Underwood, Pham et al., Overcoming Culture Restriction for SARS-CoV-2 in Human Cells Facilitates the Screening of Compounds Inhibiting Viral Replication, Antimicrob Agents Chemother
Raschetti, Vivanti, Vauloup-Fellous, Loi, Benachi et al., Synthesis and systematic review of reported neonatal SARS-CoV-2 infections, Nat Commun
Sagrillo-Fagundes, Clabault, Laurent, Hudon-Thibeault, Salustiano et al., Human Primary Trophoblast Cell Culture Model to Study the Protective Effects of Melatonin Against Hypoxia/reoxygenation-induced Disruption
Saravolatz, Depcinski, Sharma, Molnupiravir and Nirmatrelvir-Ritonavir: Oral COVID Antiviral Drugs, Clin Infect Dis
Savarino, Boelaert, Cassone, Majori, Cauda, Effects of chloroquine on viral infections: an old drug against today's diseases?, Lancet Infect Dis
Schindelin, Arganda-Carreras, Frise, Kaynig, Longair et al., Fiji: an open-source platform for biological-image analysis, Nature Methods
Schwartz, Baldewijns, Benachi, Bugatti, Bulfamante et al., Hofbauer Cells and COVID-19 in Pregnancy, Arch Pathol Lab Med
Tallarek, Urbschat, Brito, Stanelle-Bertram, Krasemann et al., Inefficient Placental Virus Replication and Absence of Neonatal Cell-Specific Immunity Upon Sars-CoV-2 Infection During Pregnancy, Front Immunol
Team, R: A language and environment for statistical computing, MSOR connections
Tregoning, Flight, Higham, Wang, Pierce, Progress of the COVID-19 vaccine effort: viruses, vaccines and variants versus efficacy, effectiveness and escape, Nature Reviews Immunology
Vargas, Moreau, Bellego, Lafond, Barbeau, Induction of trophoblast cell fusion by a protein tyrosine phosphatase inhibitor, Placenta
Vivanti, Vauloup-Fellous, Prevot, Zupan, Suffee et al., Transplacental transmission of SARS-CoV-2 infection, Nature Communications
Walls, Park, Tortorici, Wall, Mcguire et al., Structure, Function, and Antigenicity of the SARS-CoV-2 Spike Glycoprotein, Cell
Wang, Cao, Zhang, Yang, Liu et al., Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro, Cell Res
Wang, Iketani, Li, Liu, Guo et al., Alarming antibody evasion properties of rising SARS-CoV-2 BQ and XBB subvariants, Cell
Warren, Jordan, Lo, Ray, Mackman et al., Therapeutic efficacy of the small molecule GS-5734 against Ebola virus in rhesus monkeys, Nature
Wei, Wan, Yan, Wang, Zhang et al., HDL-scavenger receptor B type 1 facilitates SARS-CoV-2 entry, Nat Metab
Wice, Menton, Geuze, Schwartz, Modulators of cyclic AMP metabolism induce syncytiotrophoblast formation in vitro, Exp Cell Res
Wong, Khong, Tan, The Effects of COVID-19 on Placenta and Pregnancy: What Do We Know So Far? Diagnostics
Zhou, Choi, Liu, Zhang, Tian et al., Is SARS-CoV-2 Infection a Risk Factor for Early Pregnancy Loss? ACE2 and TMPRSS2 Coexpression and Persistent Replicative Infection in Primitive Trophoblast, J Infect Dis
Zhou, Yang, Wang, Hu, Zhang et al., vCTBs, villous cytotrophoblasts. Figure 6: SARS-CoV-2 variant Beta is more effective to infect JEG-3 cells. JEG-3 cells were infected with SARS-CoV-2 Alpha (B.1.1.7) (n = 4)
DOI record:
{
"DOI": "10.1093/molehr/gaag015",
"ISSN": [
"1360-9947",
"1460-2407"
],
"URL": "http://dx.doi.org/10.1093/molehr/gaag015",
"abstract": "<jats:title>Abstract</jats:title>\n <jats:p>Since the beginning of the COVID-19 pandemic, vulnerable populations such as pregnant persons have been at higher risk of severe symptoms and poor outcomes. Although reports of SARS-CoV-2 vertical transmission remain rare, several studies showed that maternal infection during pregnancy can induce histomorphological and inflammatory alterations in the placenta. However, the permissiveness of human trophoblasts to various variants of the virus remains poorly characterized. In this study, human primary villous cytotrophoblasts isolated from term placentas, along with trophoblastic cell lines BeWo, JEG-3, and HIPEC-65 were infected with the ancestral SARS-CoV-2 strain, which disseminated worldwide in early 2020. Permissiveness was assessed with quantitative RT-PCR, immunostaining of viral protein Nucleocapsid, and plaque assays. To investigate viral entry routes, cells were treated with Camostat mesylate (an inhibitor of the co-entry factor TMPRSS2) or chloroquine phosphate (an endosomal entry inhibitor) and viral fitness was assessed by plaque assays. Primary villous cytotrophoblasts and JEG-3 cells were also tested for infection with three pre-omicron SARS-CoV-2 variants of concern. Our results show that primary villous cytotrophoblasts are permissive to all tested SARS-CoV-2 strains in vitro. Infection with the ancestral SARS-CoV-2 strain relies mainly on a non-canonical endosomal entry pathway. Notably, JEG-3 cells represent an appropriate and convenient model for studying trophoblast infection by SARS-CoV-2, as they exhibit high permissiveness to the ancestral strain, and the SARS-CoV-2 entry pathway is similar to that in villous cytotrophoblasts. Overall, this study reveals that the cytotrophoblastic permissiveness to SARS-CoV-2 depends on the viral genetic nature and provides new insights into its entry route in human trophoblasts.</jats:p>",
"article-number": "gaag015",
"author": [
{
"affiliation": [
{
"name": "Institut national de la recherche scientifique Centre Armand-Frappier Santé Biotechnologie, , Laval, QC,",
"place": [
"Canada"
]
},
{
"name": "CIUSSS-Nord-de-l’île-de-Montréal (Hôpital Sacré-Cœur) , Montréal, QC,",
"place": [
"Canada"
]
}
],
"family": "Pinatel",
"given": "Hélène",
"sequence": "first"
},
{
"affiliation": [
{
"name": "Sainte-Justine Hospital Research Center , Montréal, QC,",
"place": [
"Canada"
]
}
],
"family": "Brien",
"given": "Marie-Eve",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Institut national de la recherche scientifique Centre Armand-Frappier Santé Biotechnologie, , Laval, QC,",
"place": [
"Canada"
]
}
],
"family": "Broquière",
"given": "Mathilde",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Université Laval Département de médecine moléculaire, , Québec, QC,",
"place": [
"Canada"
]
},
{
"name": "Centre de recherche du CHU de Québec-Université Laval Axe Endo-Nephro, , Québec, QC,",
"place": [
"Canada"
]
}
],
"family": "Scott-Boyer",
"given": "Marie-Pier",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Université Laval Département de médecine moléculaire, , Québec, QC,",
"place": [
"Canada"
]
},
{
"name": "Centre de recherche du CHU de Québec-Université Laval Axe Endo-Nephro, , Québec, QC,",
"place": [
"Canada"
]
}
],
"family": "Droit",
"given": "Arnaud",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Sainte-Justine Hospital Research Center , Montréal, QC,",
"place": [
"Canada"
]
},
{
"name": "Mayo Clinic Department of Obstetrics and Gynecology; Department of Immunology, , Rochester, MN,",
"place": [
"USA"
]
}
],
"family": "Girard",
"given": "Sylvie",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Institut national de la recherche scientifique Centre Armand-Frappier Santé Biotechnologie, , Laval, QC,",
"place": [
"Canada"
]
}
],
"family": "Delbès",
"given": "Géraldine",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0002-7390-8250",
"affiliation": [
{
"name": "Institut national de la recherche scientifique Centre Armand-Frappier Santé Biotechnologie, , Laval, QC,",
"place": [
"Canada"
]
}
],
"authenticated-orcid": false,
"family": "Chatel-Chaix",
"given": "Laurent",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0003-0543-6244",
"affiliation": [
{
"name": "Institut national de la recherche scientifique Centre Armand-Frappier Santé Biotechnologie, , Laval, QC,",
"place": [
"Canada"
]
},
{
"name": "CIUSSS-Nord-de-l’île-de-Montréal (Hôpital Sacré-Cœur) , Montréal, QC,",
"place": [
"Canada"
]
}
],
"authenticated-orcid": false,
"family": "Vaillancourt",
"given": "Cathy",
"sequence": "additional"
}
],
"container-title": "Molecular Human Reproduction",
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2026,
2,
25
]
],
"date-time": "2026-02-25T12:48:52Z",
"timestamp": 1772023732000
},
"deposited": {
"date-parts": [
[
2026,
2,
27
]
],
"date-time": "2026-02-27T08:19:34Z",
"timestamp": 1772180374000
},
"indexed": {
"date-parts": [
[
2026,
2,
27
]
],
"date-time": "2026-02-27T09:03:33Z",
"timestamp": 1772183013289,
"version": "3.50.1"
},
"is-referenced-by-count": 0,
"issued": {
"date-parts": [
[
2026,
2,
26
]
]
},
"language": "en",
"license": [
{
"URL": "https://creativecommons.org/licenses/by/4.0/",
"content-version": "am",
"delay-in-days": 1,
"start": {
"date-parts": [
[
2026,
2,
27
]
],
"date-time": "2026-02-27T00:00:00Z",
"timestamp": 1772150400000
}
}
],
"link": [
{
"URL": "https://academic.oup.com/molehr/advance-article-pdf/doi/10.1093/molehr/gaag015/67149205/gaag015.pdf",
"content-type": "application/pdf",
"content-version": "am",
"intended-application": "syndication"
},
{
"URL": "https://academic.oup.com/molehr/advance-article-pdf/doi/10.1093/molehr/gaag015/67149205/gaag015.pdf",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "286",
"original-title": [],
"prefix": "10.1093",
"published": {
"date-parts": [
[
2026,
2,
26
]
]
},
"published-online": {
"date-parts": [
[
2026,
2,
26
]
]
},
"publisher": "Oxford University Press (OUP)",
"reference-count": 0,
"references-count": 0,
"relation": {},
"resource": {
"primary": {
"URL": "https://academic.oup.com/molehr/advance-article/doi/10.1093/molehr/gaag015/8500690"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "SARS-CoV-2 infects human primary cytotrophoblasts mainly through a non-canonical entry route",
"type": "journal-article"
}
