Overview of the potential use of fluvoxamine for COVID-19 and long COVID
, K., Discover Mental Health, doi:10.1007/s44192-023-00036-3, Mar 2023
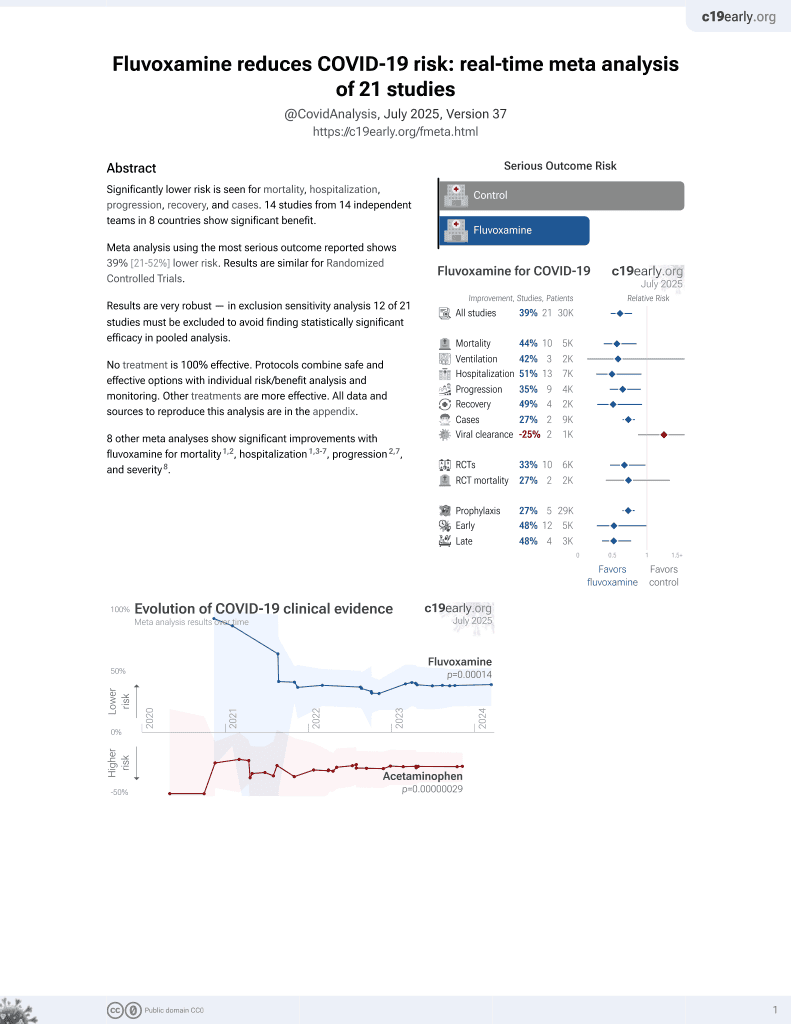
30th treatment shown to reduce risk in
November 2021, now with p = 0.00014 from 21 studies, recognized in 2 countries.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
Review of the potential benefits of fluvoxamine for COVID-19 treatment and minimizing long COVID symptoms.
1.
Scheim et al., Back to the Basics of SARS-CoV-2 Biochemistry: Microvascular Occlusive Glycan Bindings Govern Its Morbidities and Inform Therapeutic Responses, Viruses, doi:10.3390/v16040647.
2.
Hashimoto, K., Overview of the potential use of fluvoxamine for COVID-19 and long COVID, Discover Mental Health, doi:10.1007/s44192-023-00036-3.
3.
Hashimoto (B) et al., Mechanisms of action of fluvoxamine for COVID-19: a historical review, Molecular Psychiatry, doi:10.1038/s41380-021-01432-3.
4.
Hashimoto (C) et al., Old drug fluvoxamine, new hope for COVID-19, European Archives of Psychiatry and Clinical Neuroscience, doi:10.1007/s00406-021-01326-z.
5.
Hoertel et al., Repurposing antidepressants inhibiting the sphingomyelinase acid/ceramide system against COVID-19: current evidence and potential mechanisms, Molecular Psychiatry, doi:10.1038/s41380-021-01254-3.
Hashimoto et al., 21 Mar 2023, peer-reviewed, 1 author.
Contact: hashimoto@faculty.chiba-u.jp.
Overview of the potential use of fluvoxamine for COVID-19 and long COVID
Discover Mental Health, doi:10.1007/s44192-023-00036-3
Coronavirus disease 2019 (COVID-19) has presented a serious worldwide threat to public health since its emergence in late 2019. From a safety point of view, drug repurposing has received particular attention. Several clinical studies have demonstrated that the use of fluvoxamine, a selective serotonin reuptake inhibitor with potent sigma-1 receptor agonism, in the early-stage of infection might be associated with the prevention of clinical deterioration in individuals with SARS-CoV-2 infection, although several reports have shown that a low dose of fluvoxamine may be ineffective. There is increasing evidence that SARS-CoV-2 can cross the blood-brain barrier, resulting in a number of psychiatric and neurologic symptoms in COVID-19 survivors. Importantly, about half of COVID-19 survivors experience a variety of longterm sequelae, including psychiatric and neurologic symptoms, known as long COVID. In this priority review, the author presents an overview of the potential use of fluvoxamine in the treatment of COVID-19 and long COVID.
Author contributions KH did the reference search and wrote the manuscript. The author read and approved the final manuscript.
Declarations Competing interests Dr. Hashimoto is the inventor of filed patent applications on "The use of R-Ketamine in the treatment of psychiatric diseases", "(S)-norketamine and salt thereof as pharmaceutical", "R-Ketamine and derivative thereof as prophylactic or therapeutic agent for neurodegeneration disease or recognition function disorder", "Preventive or therapeutic agent and pharmaceutical composition for inflammatory diseases or bone diseases", and "R-Ketamine and its derivatives as a preventive or therapeutic agent for a neurodevelopmental disorder" by the Chiba University. Dr. Hashimoto has also received speakers' honoraria, consultant fee, or research support from Abbott, Boehringer-Ingelheim, Daiichi-Sankyo, Meiji Seika Pharma, Seikagaku Corporation, Dainippon-Sumitomo, Taisho, Otsuka, Murakami Farm and Perception Neuroscience. Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons..
References
Adnot, Houssaini, Abid, Amsellem, Serotonin transporter and serotonin receptors, Handb Exp Pharmacol, doi:10.1007/978-3-642-38664-0_15
Albayrak, Hashimoto, Sigma-1 receptor agonists and their clinical implication in neuropsychiatric disorders, Adv Exp Med Biol, doi:10.1007/978-3-319-50174-1_11
Aydillo, Rombauts, Stadlbauer, Aslam, Abelenda-Alonso et al., Immunological imprinting of the antibody response in COVID-19 patients, Nat Commun, doi:10.1038/s41467-021-23977-1
Beckmann, Becker, Ceramide and related molecules in viral infections, Int J Mol Sci, doi:10.3390/ijms22115676
Bhimraj, Gallagher, Lack of benefit of fluvoxamine for COVID-19, JAMA, doi:10.1001/jama.2022.23954
Bhuta, Khokher, Kesireddy, Iftikhar, Beran et al., Fluvoxamine in nonhospitalized patients with acute COVID-19 infection and the lack of efficacy in reducing rates of hospitalization, mechanical ventilation, and mortality in placebo-controlled trials: a systematic review and meta-analysis, Am J Ther, doi:10.1097/MJT.0000000000001496
Boldrini, Canoll, Klein, How COVID-19 affects the brain, JAMA Psychiat, doi:10.1001/jamapsychiatry.2021.0500
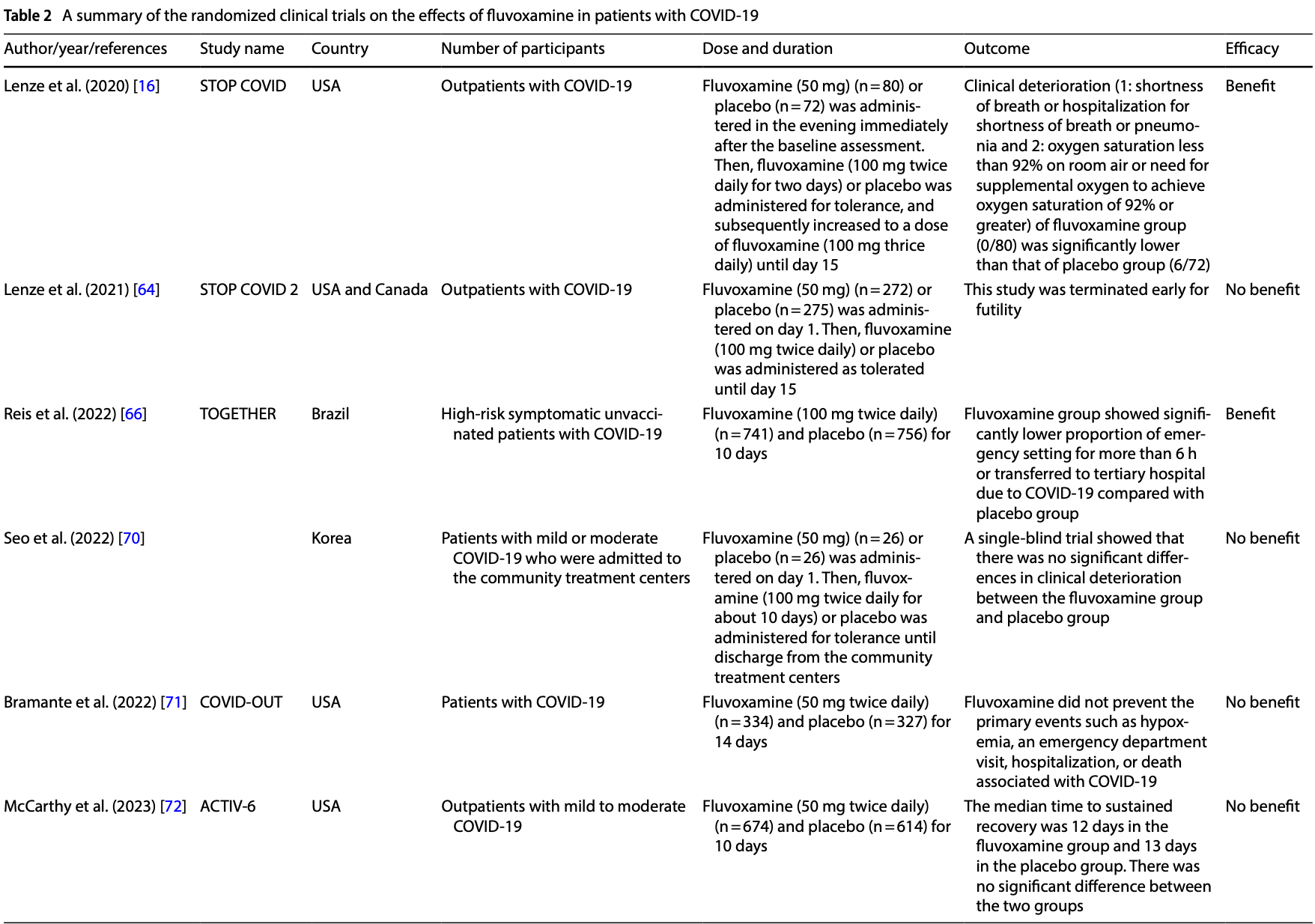
Bramante, Huling, Tignanelli, Buse, Liebovitz et al., Randomized trial of metformin, ivermectin, and fluvoxamine for Covid-19, N Engl J Med, doi:10.1056/NEJMoa2201662
Brazil, How your first brush with COVID warps your immunity, Nature, doi:10.1038/d41586-023-00086-1
Brimson, Prasanth, Malar, Brimson, Thitilertdecha et al., Drugs that offer the potential to reduce hospitalization and mortality from SARS-CoV-2 infection: the possible role of the sigma-1 receptor and autophagy, Expert Opin Ther Targets, doi:10.1080/14728222.2021.1952987
Calusic, Marcec, Luksa, Jurkovic, Kovac et al., Safety and efficacy of fluvoxamine in COVID-19 ICU patients: an open label, prospective cohort trial with matched controls, Bri J Clin Pharmacol, doi:10.1111/bcp.15126
Cheema, Jafar, Elrashedy, Shahid, Awan et al., Efficacy and safety of fluvoxamine for the treatment of COVID-19 patients, J Infect, doi:10.1016/j.jinf.2022.10.012
Davis, Mccorkell, Vogel, Topol, Long COVID: major findings, mechanisms and recommendation, Nat Rev Microbiol, doi:10.1038/s41579-022-00846-2
Deng, Rayner, Ramaraju, Abbas, Carcia et al., Efficacy and safety of selective serotonin reuptake inhibitors in COVID-19 management: a systematic review and meta-analysis, Clin Microbiol Infect, doi:10.1016/j.cmi.2023.01.010
Diaz, Baweja, Protective effect of fluvoxamine for COVID-19 in obsessive-compulsive disorder: a real-world case-control study, Prim Care Comp CNS Disord, doi:10.4088/PCC.22br03337
Dong, Zhang, Yao, Ren, Yang et al., Effects of escitalopram, R-citalopram, and reboxetine on serum levels of tumor necrosis factor-alpha, interleukin-10, and depression-like behavior in mice after lipopolysaccharide administration, Pharmacol Biochem Behav, doi:10.1016/j.pbb.2016.02.005
Douaud, Lee, Alfaro-Almagro, Arthofer, Wang et al., SARS-CoV-2 is associated with changes in brain structure in UK Biobank, Nature, doi:10.1038/s41586-022-04569-5
Erickson, Rhea, Knopp, Banks, Interactions of SARS-CoV-2 with the blood-brain barrier, Int J Mol Sci, doi:10.3390/ijms22052681
Fenton, Lee, Antidepressants with anti-inflammatory properties may be useful in long COVID depression, Drugs Ther Perspect, doi:10.1007/s40267-022-00975-x
Fico, Isayeva, Prisco, Oliva, Solè et al., Psychotropic drug repurposing for COVID-19: a systematic review and meta-analysis, Eur Neuropsychopharmacol, doi:10.1016/j.euroneuro.2022.10.004
Friesland, Mingorance, Chung, Chisari, Gastaminza, Sigma-1 receptor regulates early steps of viral RNA replication at the inset of hepatitis C virus infection, J Virol, doi:10.1128/JVI.03557-12
Gordon, Hiatt, Bouhaddou, Rezelj, Ulferts et al., Comparative host-coronavirus protein interaction networks reveal pan-viral disease mechanisms, Science, doi:10.1126/science.abe9403
Gordon, Jang, Bouhaddou, Xu, Obernier et al., A SARS-CoV-2 protein interaction map reveals targets for drug repurposing, Nature, doi:10.1038/s41586-020-2286-9
Gorman, Kane, Mcauley, Acute respiratory distress syndrome in adults: diagnosis, outcomes, long-term sequalae, and management, Lancet, doi:10.1016/S0140-6736(22)01439-8
Groff, Sun, Ssentongo, Ba, Parsons et al., Short-term and long-term rates of postacute sequelae of SARS-CoV-2 infection: a systematic review, JAMA Netw Open, doi:10.1001/jamanetworkopen.2021.28568
Gulbins, Palmada, Reichel, Lüth, Böhmer et al., Acid sphingomyelinase-ceramide system mediates effects of antidepressant drugs, Nat Med, doi:10.1038/nm.3214
Gulbins, Schumacher, Becker, Wilker, Soddemann et al., Antidepressants act by inducing autophagy controlled by sphingomyelin-ceramide, Mol Psychiatry, doi:10.1038/s41380-018-0090-9
Guo, Harari, Chernecki, Thorlund, Forrest, Fluvoxamine for the early treatment of COVID-19: a meta-analysis of randomized clinical trials, Am J Trop Med Hyg, doi:10.4269/ajtmh.21-1310
Hanner, Moebius, Flandorfer, Knaus, Striessnig et al., Purification, molecular cloning, and expression of the mammalian sigma 1 -binding site, Proc Natl Acad Sci, doi:10.1073/pnas.93.15.8072
Hanson, Abbafati, Aerts, Al-Aly, Ashbaugh et al., Estimated global proportions of individuals with persistent fatigue, cognitive, and respiratory symptom clusters following symptomatic COVID-19 in 2020 and 2021, JAMA, doi:10.1001/jama.2022.18931
Hashimoto, Activation of sigma-1 receptor chaperone in the treatment of neuropsychiatric diseases and its clinical implication, J Pharmacol Sci, doi:10.1016/j.jphs.2014.11.010
Hashimoto, Fujita, Iyo, Phencyclidine-induced cognitive deficits in mice are improved by subsequent subchronic administration of fluvoxamine: role of sigma-1 receptors, Neuropsychopharmacology, doi:10.1038/sj.npp.1301047
Hashimoto, Goromaru, High-affinity [ 3 H]6-nitroquipazine binding to the 5-hydroxytryptamine transport system in rat lung, Biochem Pharmacol, doi:10.1016/0006-2952(91)90169-6
Hashimoto, Goromaru, High-affinity binding of [ 3 H]6-nitroquipazine to 5-hydroxytryptamine transporter in human platelets, Eur J Pharmacol, doi:10.1016/0014-2999(90)90356-b
Hashimoto, Inflammatory biomarkers as differential predictors of antidepressant response, Int J Mol Sci, doi:10.3390/ijms16047796
Hashimoto, Repurposing of CNS drugs to treat COVID-19 infection: targeting the sigma-1 receptor, Eur Arch Psychiatry Clin Neurosci, doi:10.1007/s00406-020-01231-x
Hashimoto, Sigma-1 receptor chaperone and brain-derived neurotrophic factor: emerging links between cardiovascular disease and depression, Prog Neurobiol, doi:10.1016/j.pneurobio.2012.09.001
Hashimoto, Suzuki, Hashimoto, Comments to "Fluvoxamine and long COVID-19: a new role for sigma-1 receptor (S1R) agonists" by Khani and Entezari-Maleki, Mol Psychiatry, doi:10.1038/s41380-022-01546-2
Hashimoto, Suzuki, Hashimoto, Mechanisms of action of fluvoxamine for COVID-19: a historical review, Mol Psychiatry, doi:10.1038/s41380-021-01432-3
Hashimoto, Suzuki, Hashimoto, Old drug fluvoxamine, new hope for COVID-19, Eur Arch Psychiatry Clin Neurosci, doi:10.1007/s00406-021-01326-z
Hayashi, Su, Sigma-1 receptor chaperones at the ER-mitochondrion interface regulate Ca 2+ signaling and cell survival, Cell, doi:10.1016/j.cell.2007.08.036
Hindmarch, Hashimoto, Cognition and depression: the effects of fluvoxamine, a sigma-1 receptor agonist, reconsidered, Hum Psychopharmacol, doi:10.1002/hup.1106
Hoertel, Sánchez-Rico, Gulbins, Kornhuber, Carpinteiro et al., Association between FIASMAs and reduced risk of intubation or death in individuals hospitalized for severe COVID-19: an observational multicenter study, Clin Pharmacol Ther, doi:10.1002/cpt.2317
Hoertel, Sánchez-Rico, Vernet, Beeker, Jannot et al., Association between antidepressant use and reduced risk of intubation or death in hospitalized patients with COVID-19: results from an observational study, Mol Psychiatry, doi:10.1038/s41380-021-01021-4
Ishikawa, Ishiwata, Ishii, Kimura, Sakata et al., High occupancy of sigma-1 receptors in the human brain after single oral administration of fluvoxamine: a positron emission tomography study using [ 11 C]SA4503, Biol Psychiatry, doi:10.1016/j.biopsych.2007.04.001
Ishima, Fujita, Hashimoto, Interactions of new antidepressants with sigma-1 receptor chaperons and their potentiation of neurite outgrowth in PC12 cells, Eur J Pharmacol, doi:10.1016/j.ejphar.2014.01.064
Ishima, Fujita, Kohno, Kunitachi, Hotio et al., Improvement of phencyclidine-induced cognitive deficits in mice by subsequent subchronic administration of fluvoxamine, but not sertraline, Open Clin Chem J, doi:10.2174/1874241600902010007
Karlstad, Hovi, Husby, Härkänen, Selmer et al., SARS-CoV-2 vaccination and mycarditis in a Nordic cohort study of 23 million residents, JAMA Cardiol, doi:10.1001/jamacardio.2022.0583
Khani, Entezari-Maleki, Fluvoxamine and long COVID-19: a new role for sigma-1 receptor (S1R) agonists, Mol Psychiatry, doi:10.1038/s41380-022-01545-3
Kim, Huh, Park, Peck, Jung, Association between vaccination and acute myocardial infarction and ischemic stroke after COVID-19 infection, JAMA, doi:10.1001/jama.2022.12992
Kirenga, Mugenyi, Sánchez-Rico, Kyobe, Muttamba et al., Association of fluvoxamine with mortality and symptom resolution among inpatients with COVID-19 in Uganda: a prospective interventional open-label cohort study, Mol Psychiatry, doi:10.1038/s41380-023-02004-3
Koc, Xiao, Liu, Li, Chen, Long COVID and its management, Int J Biol Sci, doi:10.7150/ijbs.75056
Kondratiuk, Pillay, Kon, Lalvani, A conceptual framework to accelerate the clinical impact of evolving research into long COVID, Lancet Infect Dis, doi:10.1016/S1473-3099(21)00136-5
Kornhuber, Hoertel, Gulbins, The acid sphingomyelinase/ceramide system in COVID-19, Mol Psychiatry, doi:10.1038/s41380-021-01309-5
Kornhuber, Muehlbacher, Trapp, Pechmann, Friedl et al., Identification of novel functional inhibitors of acid sphingomyelinase, PLoS ONE, doi:10.1371/journal.pone.0023852
Kornhuber, Tripal, Reichel, Mühle, Rhein et al., Functional inhibitors of acid sphingomyelinase (FIASMAs): a novel pharmacological group of drugs with broad clinical applications, Cell Physiol Biochem, doi:10.1159/000315101
Lee, Vigod, Bortolussi-Courval, Hanula, Boulware et al., Fluvoxamine for outpatients management of COVID-19 to prevent hospitalization: a systematic review and meta-analysis, JAMA Netw Open, doi:10.1001/jamanetworkopen.2022.6269
Lenze, Fluvoxamine for early treatment of Covid-19 (Stop Covid 2
Lenze, Mattar, Zorumski, Zorumski, Stevens et al., Fluvoxamine vs placebo and clinical deterioration in outpatients with symptomatic COVID-19. A randomized clinical trial, JAMA, doi:10.1001/jama.2020.22760
Li, Wang, Wu, Gulbins, Grassmé et al., Acid sphingomyelinase-ceramide system in bacterial infections, Cell Physiol Biochem, doi:10.33594/000000021
Lu, Chao, Chang, Lan, Lai, Effect of fluvoxamine on outcomes of nonhospitalized patients with COVID-19: a systematic review and meta-analysis, J Infect Public Health, doi:10.1016/j.jiph.2022.10.010
Mahdi, Hermán, Réthelyi, Bálint, Potential role of the antidepressants fluoxetine and fluvoxamine in the treatment of COVID-19, Int J Mol Sci, doi:10.3390/ijms23073812
Marcec, Dodig, Likic, A meta-analysis regarding fluvoxamine and hospitalization risk of COVID-19 patients: TOGETHER making a difference, J Infect, doi:10.1016/j.jinf.2022.11.011
Maurer-Spurej, Pittendreigh, Solomons, The influence of selective serotonin reuptake inhibitors on human platelet serotonin, Thromb Haemost, doi:10.1160/TH03-05-0330
Mccarthy, Naggie, Boulware, Lindsell, Stewart et al., Effect of fluvoxamine vs placebo on time to sustained recovery in outpatients with mild to moderate COVID-19: a randomized clinical trial, JAMA, doi:10.1001/jama.2022.24100
Mercado, Killic, Molecular mechanisms of SERT in platelets: regulation of plasma serotonin levels, Mol Interv, doi:10.1124/mi.10.4.6
Mills, Reis, Wilson, Thorlund, Forrest et al., Early treatment with fluvoxamine among patients with COVID-19: a costconsequence model, Am J Trop Med Hyg, doi:10.4269/ajtmh.22-0106
Narita, Hashimoto, Tomitaka, Minabe, Interaction of selective serotonin reuptake inhibitors with subtypes of sigma receptors in rat brain, Eur J Pharmacol, doi:10.1016/0014-2999(96)00254-3
Ng, Salim, Chu, Drug repurposing for COVID-19: approaches, challenges and promising candidates, Pharmacol Ther, doi:10.1016/j.pharmthera.2021.107930
Nishimura, Ishima, Iyo, Hashimoto, Potentiation of nerve growth factor-induced neurite outgrowth by fluvoxamine: role of sigma-1 receptors, IP 3 receptors and cellular signaling pathways, PLoS ONE, doi:10.1371/journal.pone.0002558
Ohgi, Futamura, Kikuchi, Hashimoto, Effects of antidepressants on alterations in serum cytokines and depressive-like behavior in mice after lipopolysaccharide administration, Pharmacol Biochem Behav, doi:10.1016/j.pbb.2012.12.003
Oldfield, Hibberd, Bridle, How does severe acute respiratory syndrome-coronavirus-2 affect the brain and its implications for the vaccines currently in use, Vaccines, doi:10.3390/vaccines10010001
Oskotsky, Maric, Tang, Oskotsky, Wong et al., Mortality risk among patients with COVID-19 prescribed selective serotonin reuptake inhibitor antidepressants, JAMA Netw Open, doi:10.1001/jamanetworkopen.2021.33090
Oster, Shay, Su, Gee, Creech et al., Myocarditis cases reported after mRN-based COVID-19 vaccination in the US from December 2020 to August 2021, JAMA, doi:10.1001/jama.2021.24110
Owens, Knight, Nemeroff, Second-generation SSRIs: human monoamine transporter binding profile of escitalopram and R-fluoxetine, Biol Psychiatry, doi:10.1016/s0006-3223(01)01145-3
Pavord, Hunt, Horner, Bewley, Karpusheff et al., Vaccine induced immune thrombocytopenia and thrombosis: summary of NICE guidance, BMJ, doi:10.1136/bmj.n2195
Perry, Tamborska, Singh, Craven, Marigold et al., Cerebral venous thrombosis after vaccination against, Lancet, doi:10.1016/S0140-6736(21)01608-1
Petrovszki, Walter, Vigh, Kocsis, Valkai et al., Penetration of the SARS-CoV-2 spike protein across the blood-brain barrier, as revealed by a combination of a human cell culture model system and optical biosensing, Biomedicines, doi:10.3390/biomedicines10010188
Pineda, Singh, Pineda, Umanzor, Baires et al., Impact of fluvoxamine on outpatient treatment of COVID-19 in Honduras in a prospective observational real-world study, Front Pharmacol, doi:10.3389/fphar.2022.1054644
Puelles, Lütgehetmann, Lindenmeyer, Sperhake, Wong et al., Multiorgan and renal tropism of SARS-CoV-2, N Engl J Med, doi:10.1056/NEJMc2011400
Reis, Silva, Silva, Thabane, Milagres et al., Effect of early treatment with fluvoxamine on risk of emergency care and hospitalization among patients with COVID-19: the TOGETHER randomized, platform clinical trial, Lancet Glob Health, doi:10.1016/S2214-109X(21)00448-4
Rhea, Logsdon, Hansen, Williams, Reed et al., The S1 protein of SARS-CoV-2 crosses the blood-brain barrier in mice, Nat Neurosci, doi:10.1038/s41593-020-00771-8
Rosen, Seki, Fernández-Castañeda, Beiter, Eccles et al., Modulation of the sigma-1 receptor-IRE1 pathway is beneficial in preclinical models of inflammation and sepsis, Sci Transl Med, doi:10.1126/scitranslmed.aau5266
Seftel, Boulware, Prospective cohort of fluvoxamine for early treatment of coronavirus disease 19, Open Forum Infect Dis, doi:10.1093/ofid/ofab050
Seo, Kim, Bae, Park, Chung et al., Fluvoxamine treatment of patients with symptomatic COVID-19 in a community treatment center: a preliminary result of randomized controlled trial, Infect Chemother, doi:10.3947/ic.2021.0142
Shaffer, Lots of long COVID treatment leads, but few are proven, Proc Natl Acad Sci, doi:10.1073/pnas.2213524119
Sidky, Sahner, Girvin, Hotaling, Michael et al., Assessing the effect of selective serotonin reuptake inhibitors in the prevention of post-acute sequelae of COVID-19, medRxiv, doi:10.1101/2022.11.09.22282142
Sollmann, Beer, Kirchhoff, SARS-CoV-2 infection and the brain: direct evidence for brain changes in milder cases, Signal Transduct Target Ther, doi:10.1038/s41392-022-01072-1
Solomon, Normandin, Bhattacharyya, Mukerji, Keller et al., Neurological features of Covid-19, N Engl J Med, doi:10.1056/NEJMc2019373
Song, Zhang, Israelow, Lu-Culligan, Prado et al., Neuroinvasion of SARS-CoV-2 in human and mouse brain, J Exp Med, doi:10.1084/jem.20202135
Soriano, Murthy, Marshall, Relan, Diaz, WHO Clinical Case Definition Working Group on Post-COVID-19 Condition. A clinical case definition of post-COVID-19 condition by a Delphi consensus, Lancet Infect Dis, doi:10.1016/S1473-3099(21)00703-9
Stefanou, Palaiodimou, De Sousa, Theodorou, Bakola et al., Acute arterial ischemic stroke following COVID-19 vaccination: a systematic review and meta-analysis, Neurology, doi:10.1212/WNL.0000000000200996
Stein, Ramelli, Grazioli, Chung, Singh et al., SARS-CoV-2 infection and persistence in the human body and brain at autopsy, Nature, doi:10.1038/s41586-022-05542-y
Sukhatme, Reiersen, Vayttaden, Sykhatme, Fluvoxamine: a review of its mechanisms of actions and its role in COVID-19, Front Pharmacol, doi:10.3389/fphar.2021.652688
Takano, Suhara, Sudo, Inoue, Hashimoto et al., Comparative evaluation of two serotonin transporter ligands in the human brain: [ 11 C](+)McN5652 and [ 11 C]cyanoimipramine, Eur J Nucl Med, doi:10.1007/s00259-002-0884-4
Theoharides, Could SARS-CoV-2 spike protein be responsible for long-COVID syndrome?, Mol Neurobiol, doi:10.1007/s12035-021-02696-0
Trkulja, There is no reasonable evidence to support efficacy of fluvoxamine in prevention of disease deterioration in COVID-19 outpatients: a comment on two recent meta-analyses advocating its use, J Infect, doi:10.1016/j.jinf.2022.11.016
Trkulja, Why we should not recommend or offer fluvoxamine to COVID-19 patients?, Eur J Clin Pharmacol, doi:10.1007/s00228-022-03447-3
Törnquist, Asghar, Srinivasan, Korhonen, Lindholm, Sphingolipids as modulators of SARS-CoV-2 infection, Front Cell Dev Biol, doi:10.3389/fcell.2021.689854
Vasallo, Gastaminza, Cellular stress response in hepatitis C virus infection: mastering a two-edged sword, Virus Res, doi:10.1016/j.virusres.2015.03.013
Vela, Repurposing sigma-1 receptor ligands for COVID-19 therapy?, Front Pharmacol, doi:10.3389/fphar.2020.582310
Venkatesan, Repurposing drugs for treatment of COVID-19, Lancet Respir Med, doi:10.1016/S2213-2600(21)00270-8
Wang, Wang, Liu, Qiao, Baldwin et al., Effects of SSRIs on peripheral inflammatory markers in patients with major depressive disorder: a systematic review and meta-analysis, Brain Behav Immun, doi:10.1016/j.bbi.2019.02.021
Wu, Xu, Chen, Duan, Hashimoto et al., Nervous system involvement after infection with COVID-19 and other coronaviruses, Brain Behav Immun, doi:10.1016/j.bbi.2020.03.031
Zeng, Zhao, Li, Lu, Liu, A systematic review and meta-analysis of long-term physical and mental sequelae of COVID-19 pandemic: call for research priority and action, Mol Psychiatry, doi:10.1038/s41380-022-01614-7
Zhang, Xie, Hashimoto, Current status of potential therapeutic candidates for the COVID-19 crisis, Brain Behav Immun, doi:10.1016/j.bbi.2020.04.046
Zhang, Zhou, Bao, Liu, Zhu et al., SAES-CoV-2 crosses the blood-brain barrier accompanied with basement membrane disruption without tight junctions alteration, Signal Transduct Target, doi:10.1038/s41392-021-00719-9
DOI record:
{
"DOI": "10.1007/s44192-023-00036-3",
"ISSN": [
"2731-4383"
],
"URL": "http://dx.doi.org/10.1007/s44192-023-00036-3",
"abstract": "<jats:title>Abstract</jats:title><jats:p>Coronavirus disease 2019 (COVID-19) has presented a serious worldwide threat to public health since its emergence in late 2019. From a safety point of view, drug repurposing has received particular attention. Several clinical studies have demonstrated that the use of fluvoxamine, a selective serotonin reuptake inhibitor with potent sigma-1 receptor agonism, in the early-stage of infection might be associated with the prevention of clinical deterioration in individuals with SARS-CoV-2 infection, although several reports have shown that a low dose of fluvoxamine may be ineffective. There is increasing evidence that SARS-CoV-2 can cross the blood–brain barrier, resulting in a number of psychiatric and neurologic symptoms in COVID-19 survivors. Importantly, about half of COVID-19 survivors experience a variety of long-term sequelae, including psychiatric and neurologic symptoms, known as long COVID. In this priority review, the author presents an overview of the potential use of fluvoxamine in the treatment of COVID-19 and long COVID.</jats:p>",
"alternative-id": [
"36"
],
"article-number": "9",
"assertion": [
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "Received",
"name": "received",
"order": 1,
"value": "25 January 2023"
},
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "Accepted",
"name": "accepted",
"order": 2,
"value": "2 March 2023"
},
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "First Online",
"name": "first_online",
"order": 3,
"value": "21 March 2023"
},
{
"group": {
"label": "Declarations",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 1
},
{
"group": {
"label": "Competing interests",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 2,
"value": "Dr. Hashimoto is the inventor of filed patent applications on “The use of <i>R</i>-Ketamine in the treatment of psychiatric diseases”, “(<i>S</i>)-norketamine and salt thereof as pharmaceutical”, “<i>R</i>-Ketamine and derivative thereof as prophylactic or therapeutic agent for neurodegeneration disease or recognition function disorder”, “Preventive or therapeutic agent and pharmaceutical composition for inflammatory diseases or bone diseases”, and “<i>R</i>-Ketamine and its derivatives as a preventive or therapeutic agent for a neurodevelopmental disorder” by the Chiba University. Dr. Hashimoto has also received speakers’ honoraria, consultant fee, or research support from Abbott, Boehringer-Ingelheim, Daiichi-Sankyo, Meiji Seika Pharma, Seikagaku Corporation, Dainippon-Sumitomo, Taisho, Otsuka, Murakami Farm and Perception Neuroscience."
}
],
"author": [
{
"affiliation": [],
"family": "Hashimoto",
"given": "Kenji",
"sequence": "first"
}
],
"container-title": "Discover Mental Health",
"container-title-short": "Discov Ment Health",
"content-domain": {
"crossmark-restriction": false,
"domain": [
"link.springer.com"
]
},
"created": {
"date-parts": [
[
2023,
3,
21
]
],
"date-time": "2023-03-21T14:05:01Z",
"timestamp": 1679407501000
},
"deposited": {
"date-parts": [
[
2023,
3,
21
]
],
"date-time": "2023-03-21T14:09:07Z",
"timestamp": 1679407747000
},
"indexed": {
"date-parts": [
[
2023,
3,
22
]
],
"date-time": "2023-03-22T05:00:01Z",
"timestamp": 1679461201237
},
"is-referenced-by-count": 0,
"issue": "1",
"issued": {
"date-parts": [
[
2023,
3,
21
]
]
},
"journal-issue": {
"issue": "1",
"published-online": {
"date-parts": [
[
2023,
12
]
]
}
},
"language": "en",
"license": [
{
"URL": "https://creativecommons.org/licenses/by/4.0",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2023,
3,
21
]
],
"date-time": "2023-03-21T00:00:00Z",
"timestamp": 1679356800000
}
},
{
"URL": "https://creativecommons.org/licenses/by/4.0",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2023,
3,
21
]
],
"date-time": "2023-03-21T00:00:00Z",
"timestamp": 1679356800000
}
}
],
"link": [
{
"URL": "https://link.springer.com/content/pdf/10.1007/s44192-023-00036-3.pdf",
"content-type": "application/pdf",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://link.springer.com/article/10.1007/s44192-023-00036-3/fulltext.html",
"content-type": "text/html",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://link.springer.com/content/pdf/10.1007/s44192-023-00036-3.pdf",
"content-type": "application/pdf",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "297",
"original-title": [],
"prefix": "10.1007",
"published": {
"date-parts": [
[
2023,
3,
21
]
]
},
"published-online": {
"date-parts": [
[
2023,
3,
21
]
]
},
"publisher": "Springer Science and Business Media LLC",
"reference": [
{
"DOI": "10.1001/jama.2021.24110",
"author": "ME Oster",
"doi-asserted-by": "publisher",
"first-page": "331",
"issue": "4",
"journal-title": "JAMA",
"key": "36_CR1",
"unstructured": "Oster ME, Shay DK, Su JR, Gee J, Creech CB, Broder KR, et al. Myocarditis cases reported after mRN-based COVID-19 vaccination in the US from December 2020 to August 2021. JAMA. 2022;327(4):331–40. https://doi.org/10.1001/jama.2021.24110.",
"volume": "327",
"year": "2022"
},
{
"DOI": "10.1001/jamacardio.2022.0583",
"author": "Ø Karlstad",
"doi-asserted-by": "publisher",
"first-page": "600",
"issue": "6",
"journal-title": "JAMA Cardiol",
"key": "36_CR2",
"unstructured": "Karlstad Ø, Hovi P, Husby A, Härkänen T, Selmer RM, Pihlström N, et al. SARS-CoV-2 vaccination and mycarditis in a Nordic cohort study of 23 million residents. JAMA Cardiol. 2022;7(6):600–12. https://doi.org/10.1001/jamacardio.2022.0583.",
"volume": "7",
"year": "2022"
},
{
"DOI": "10.1016/S0140-6736(21)01608-1",
"author": "RJ Perry",
"doi-asserted-by": "publisher",
"first-page": "1147",
"issue": "10306",
"journal-title": "Lancet",
"key": "36_CR3",
"unstructured": "Perry RJ, Tamborska A, Singh B, Craven B, Marigold R, Arthur-Farraj P, et al. Cerebral venous thrombosis after vaccination against. Lancet. 2021;398(10306):1147–56. https://doi.org/10.1016/S0140-6736(21)01608-1.",
"volume": "398",
"year": "2021"
},
{
"DOI": "10.1136/bmj.n2195",
"author": "S Pavord",
"doi-asserted-by": "publisher",
"journal-title": "BMJ",
"key": "36_CR4",
"unstructured": "Pavord S, Hunt BJ, Horner D, Bewley S, Karpusheff J, Guideline Committee. Vaccine induced immune thrombocytopenia and thrombosis: summary of NICE guidance. BMJ. 2021;375: n2195. https://doi.org/10.1136/bmj.n2195.",
"volume": "375",
"year": "2021"
},
{
"DOI": "10.1001/jama.2022.12992",
"author": "YE Kim",
"doi-asserted-by": "publisher",
"first-page": "887",
"issue": "9",
"journal-title": "JAMA",
"key": "36_CR5",
"unstructured": "Kim YE, Huh K, Park YJ, Peck KR, Jung J. Association between vaccination and acute myocardial infarction and ischemic stroke after COVID-19 infection. JAMA. 2022;328(9):887–9. https://doi.org/10.1001/jama.2022.12992.",
"volume": "328",
"year": "2022"
},
{
"DOI": "10.1212/WNL.0000000000200996",
"author": "MI Stefanou",
"doi-asserted-by": "publisher",
"journal-title": "Neurology",
"key": "36_CR6",
"unstructured": "Stefanou MI, Palaiodimou L, Aguiar de Sousa D, Theodorou A, Bakola E, Katsaros DE, et al. Acute arterial ischemic stroke following COVID-19 vaccination: a systematic review and meta-analysis. Neurology. 2022. https://doi.org/10.1212/WNL.0000000000200996.",
"year": "2022"
},
{
"DOI": "10.1038/d41586-023-00086-1",
"author": "R Brazil",
"doi-asserted-by": "publisher",
"first-page": "428",
"issue": "7944",
"journal-title": "Nature",
"key": "36_CR7",
"unstructured": "Brazil R. How your first brush with COVID warps your immunity. Nature. 2023;613(7944):428–30. https://doi.org/10.1038/d41586-023-00086-1.",
"volume": "613",
"year": "2023"
},
{
"DOI": "10.1038/s41467-021-23977-1",
"author": "T Aydillo",
"doi-asserted-by": "publisher",
"first-page": "3781",
"issue": "1",
"journal-title": "Nat Commun",
"key": "36_CR8",
"unstructured": "Aydillo T, Rombauts A, Stadlbauer D, Aslam S, Abelenda-Alonso G, Escalera A, et al. Immunological imprinting of the antibody response in COVID-19 patients. Nat Commun. 2021;12(1):3781. https://doi.org/10.1038/s41467-021-23977-1.",
"volume": "12",
"year": "2021"
},
{
"DOI": "10.1007/s00406-020-01231-x",
"author": "K Hashimoto",
"doi-asserted-by": "publisher",
"first-page": "249",
"issue": "1",
"journal-title": "Eur Arch Psychiatry Clin Neurosci",
"key": "36_CR9",
"unstructured": "Hashimoto K. Repurposing of CNS drugs to treat COVID-19 infection: targeting the sigma-1 receptor. Eur Arch Psychiatry Clin Neurosci. 2021;271(1):249–58. https://doi.org/10.1007/s00406-020-01231-x.",
"volume": "271",
"year": "2021"
},
{
"DOI": "10.1007/s00406-021-01326-z",
"author": "Y Hashimoto",
"doi-asserted-by": "publisher",
"first-page": "161",
"issue": "1",
"journal-title": "Eur Arch Psychiatry Clin Neurosci",
"key": "36_CR10",
"unstructured": "Hashimoto Y, Suzuki T, Hashimoto K. Old drug fluvoxamine, new hope for COVID-19. Eur Arch Psychiatry Clin Neurosci. 2022;272(1):161–3. https://doi.org/10.1007/s00406-021-01326-z.",
"volume": "272",
"year": "2022"
},
{
"DOI": "10.1038/s41380-021-01432-3",
"author": "Y Hashimoto",
"doi-asserted-by": "publisher",
"first-page": "1898",
"issue": "4",
"journal-title": "Mol Psychiatry",
"key": "36_CR11",
"unstructured": "Hashimoto Y, Suzuki T, Hashimoto K. Mechanisms of action of fluvoxamine for COVID-19: a historical review. Mol Psychiatry. 2022;27(4):1898–907. https://doi.org/10.1038/s41380-021-01432-3.",
"volume": "27",
"year": "2022"
},
{
"DOI": "10.1016/j.bbi.2020.04.046",
"author": "J Zhang",
"doi-asserted-by": "publisher",
"first-page": "59",
"journal-title": "Brain Behav Immun",
"key": "36_CR12",
"unstructured": "Zhang J, Xie B, Hashimoto K. Current status of potential therapeutic candidates for the COVID-19 crisis. Brain Behav Immun. 2020;87:59–73. https://doi.org/10.1016/j.bbi.2020.04.046.",
"volume": "87",
"year": "2020"
},
{
"DOI": "10.1016/j.pharmthera.2021.107930",
"author": "YL Ng",
"doi-asserted-by": "publisher",
"journal-title": "Pharmacol Ther",
"key": "36_CR13",
"unstructured": "Ng YL, Salim CK, Chu JJH. Drug repurposing for COVID-19: approaches, challenges and promising candidates. Pharmacol Ther. 2021;228: 107930. https://doi.org/10.1016/j.pharmthera.2021.107930.",
"volume": "228",
"year": "2021"
},
{
"DOI": "10.1016/S2213-2600(21)00270-8",
"author": "P Venkatesan",
"doi-asserted-by": "publisher",
"first-page": "e63",
"issue": "7",
"journal-title": "Lancet Respir Med",
"key": "36_CR14",
"unstructured": "Venkatesan P. Repurposing drugs for treatment of COVID-19. Lancet Respir Med. 2021;9(7):e63. https://doi.org/10.1016/S2213-2600(21)00270-8.",
"volume": "9",
"year": "2021"
},
{
"DOI": "10.1016/j.euroneuro.2022.10.004",
"author": "G Fico",
"doi-asserted-by": "publisher",
"first-page": "30",
"journal-title": "Eur Neuropsychopharmacol",
"key": "36_CR15",
"unstructured": "Fico G, Isayeva U, De Prisco M, Oliva V, Solè B, Montejo L, et al. Psychotropic drug repurposing for COVID-19: a systematic review and meta-analysis. Eur Neuropsychopharmacol. 2023;66:30–44. https://doi.org/10.1016/j.euroneuro.2022.10.004.",
"volume": "66",
"year": "2023"
},
{
"DOI": "10.1001/jama.2020.22760",
"author": "E Lenze",
"doi-asserted-by": "publisher",
"first-page": "2292",
"issue": "22",
"journal-title": "JAMA",
"key": "36_CR16",
"unstructured": "Lenze E, Mattar C, Zorumski CF, Zorumski CF, Stevens A, Schweiger J, et al. Fluvoxamine vs placebo and clinical deterioration in outpatients with symptomatic COVID-19. A randomized clinical trial. JAMA. 2020;324(22):2292–300. https://doi.org/10.1001/jama.2020.22760.",
"volume": "324",
"year": "2020"
},
{
"DOI": "10.1016/S1473-3099(21)00136-5",
"author": "AL Kondratiuk",
"doi-asserted-by": "publisher",
"first-page": "756",
"issue": "6",
"journal-title": "Lancet Infect Dis",
"key": "36_CR17",
"unstructured": "Kondratiuk AL, Pillay TD, Kon OM, Lalvani A. A conceptual framework to accelerate the clinical impact of evolving research into long COVID. Lancet Infect Dis. 2021;21(6):756–7. https://doi.org/10.1016/S1473-3099(21)00136-5.",
"volume": "21",
"year": "2021"
},
{
"DOI": "10.1001/jama.2022.18931",
"author": "S Wulf Hanson",
"doi-asserted-by": "publisher",
"first-page": "1604",
"issue": "16",
"journal-title": "JAMA",
"key": "36_CR18",
"unstructured": "Wulf Hanson S, Abbafati C, Aerts JG, Al-Aly Z, Ashbaugh C, Ballouz T, et al. Estimated global proportions of individuals with persistent fatigue, cognitive, and respiratory symptom clusters following symptomatic COVID-19 in 2020 and 2021. JAMA. 2022;328(16):1604–15. https://doi.org/10.1001/jama.2022.18931.",
"volume": "328",
"year": "2022"
},
{
"DOI": "10.1016/S0140-6736(22)01439-8",
"author": "EA Gorman",
"doi-asserted-by": "publisher",
"first-page": "1157",
"issue": "10358",
"journal-title": "Lancet",
"key": "36_CR19",
"unstructured": "Gorman EA, O’Kane CM, McAuley DF. Acute respiratory distress syndrome in adults: diagnosis, outcomes, long-term sequalae, and management. Lancet. 2022;400(10358):1157–70. https://doi.org/10.1016/S0140-6736(22)01439-8.",
"volume": "400",
"year": "2022"
},
{
"DOI": "10.1016/S1473-3099(21)00703-9",
"author": "JB Soriano",
"doi-asserted-by": "publisher",
"first-page": "e102",
"issue": "4",
"journal-title": "Lancet Infect Dis",
"key": "36_CR20",
"unstructured": "Soriano JB, Murthy S, Marshall JC, Relan P, Diaz JV, WHO Clinical Case Definition Working Group on Post-COVID-19 Condition. A clinical case definition of post-COVID-19 condition by a Delphi consensus. Lancet Infect Dis. 2022;22(4):e102–7. https://doi.org/10.1016/S1473-3099(21)00703-9.",
"volume": "22",
"year": "2022"
},
{
"DOI": "10.1038/s41380-022-01614-7",
"author": "N Zeng",
"doi-asserted-by": "publisher",
"first-page": "423",
"issue": "1",
"journal-title": "Mol Psychiatry",
"key": "36_CR21",
"unstructured": "Zeng N, Zhao YM, Yan W, Li C, Lu QD, Liu L, et al. A systematic review and meta-analysis of long-term physical and mental sequelae of COVID-19 pandemic: call for research priority and action. Mol Psychiatry. 2023;28(1):423–33. https://doi.org/10.1038/s41380-022-01614-7.",
"volume": "28",
"year": "2023"
},
{
"DOI": "10.1073/pnas.2213524119",
"author": "L Shaffer",
"doi-asserted-by": "publisher",
"issue": "36",
"journal-title": "Proc Natl Acad Sci USA",
"key": "36_CR22",
"unstructured": "Shaffer L. Lots of long COVID treatment leads, but few are proven. Proc Natl Acad Sci USA. 2022;119(36): e2213524119. https://doi.org/10.1073/pnas.2213524119.",
"volume": "119",
"year": "2022"
},
{
"DOI": "10.1007/978-3-642-38664-0_15",
"author": "S Adnot",
"doi-asserted-by": "publisher",
"first-page": "365",
"journal-title": "Handb Exp Pharmacol",
"key": "36_CR23",
"unstructured": "Adnot S, Houssaini A, Abid S, Marcos E, Amsellem V. Serotonin transporter and serotonin receptors. Handb Exp Pharmacol. 2013;218:365–80. https://doi.org/10.1007/978-3-642-38664-0_15.",
"volume": "218",
"year": "2013"
},
{
"DOI": "10.1016/0006-2952(91)90169-6",
"author": "K Hashimoto",
"doi-asserted-by": "publisher",
"first-page": "1679",
"issue": "11",
"journal-title": "Biochem Pharmacol",
"key": "36_CR24",
"unstructured": "Hashimoto K, Goromaru T. High-affinity [3H]6-nitroquipazine binding to the 5-hydroxytryptamine transport system in rat lung. Biochem Pharmacol. 1991;41(11):1679–82. https://doi.org/10.1016/0006-2952(91)90169-6.",
"volume": "41",
"year": "1991"
},
{
"DOI": "10.1007/s00259-002-0884-4",
"author": "A Takano",
"doi-asserted-by": "publisher",
"first-page": "1289",
"issue": "10",
"journal-title": "Eur J Nucl Med",
"key": "36_CR25",
"unstructured": "Takano A, Suhara T, Sudo Y, Inoue M, Hashimoto K, Zhang MR, et al. Comparative evaluation of two serotonin transporter ligands in the human brain: [11C](+)McN5652 and [11C]cyanoimipramine. Eur J Nucl Med. 2002;29(10):1289–97. https://doi.org/10.1007/s00259-002-0884-4.",
"volume": "29",
"year": "2002"
},
{
"DOI": "10.1016/0014-2999(90)90356-b",
"author": "K Hashimoto",
"doi-asserted-by": "publisher",
"first-page": "295",
"issue": "3",
"journal-title": "Eur J Pharmacol",
"key": "36_CR26",
"unstructured": "Hashimoto K, Goromaru T. High-affinity binding of [3H]6-nitroquipazine to 5-hydroxytryptamine transporter in human platelets. Eur J Pharmacol. 1990;187(3):295–302. https://doi.org/10.1016/0014-2999(90)90356-b.",
"volume": "187",
"year": "1990"
},
{
"DOI": "10.1160/TH03-05-0330",
"author": "E Maurer-Spurej",
"doi-asserted-by": "publisher",
"first-page": "119",
"issue": "1",
"journal-title": "Thromb Haemost",
"key": "36_CR27",
"unstructured": "Maurer-Spurej E, Pittendreigh C, Solomons K. The influence of selective serotonin reuptake inhibitors on human platelet serotonin. Thromb Haemost. 2004;91(1):119–28. https://doi.org/10.1160/TH03-05-0330.",
"volume": "91",
"year": "2004"
},
{
"DOI": "10.1124/mi.10.4.6",
"author": "CP Mercado",
"doi-asserted-by": "publisher",
"first-page": "231",
"issue": "4",
"journal-title": "Mol Interv",
"key": "36_CR28",
"unstructured": "Mercado CP, Killic F. Molecular mechanisms of SERT in platelets: regulation of plasma serotonin levels. Mol Interv. 2010;10(4):231–41. https://doi.org/10.1124/mi.10.4.6.",
"volume": "10",
"year": "2010"
},
{
"DOI": "10.1016/s0006-3223(01)01145-3",
"author": "MJ Owens",
"doi-asserted-by": "publisher",
"first-page": "345",
"issue": "5",
"journal-title": "Biol Psychiatry",
"key": "36_CR29",
"unstructured": "Owens MJ, Knight DL, Nemeroff CB. Second-generation SSRIs: human monoamine transporter binding profile of escitalopram and R-fluoxetine. Biol Psychiatry. 2001;50(5):345–50. https://doi.org/10.1016/s0006-3223(01)01145-3.",
"volume": "50",
"year": "2001"
},
{
"DOI": "10.1016/j.pbb.2012.12.003",
"author": "Y Ohgi",
"doi-asserted-by": "publisher",
"first-page": "853",
"issue": "4",
"journal-title": "Pharmacol Biochem Behav",
"key": "36_CR30",
"unstructured": "Ohgi Y, Futamura T, Kikuchi T, Hashimoto K. Effects of antidepressants on alterations in serum cytokines and depressive-like behavior in mice after lipopolysaccharide administration. Pharmacol Biochem Behav. 2013;103(4):853–9. https://doi.org/10.1016/j.pbb.2012.12.003.",
"volume": "103",
"year": "2013"
},
{
"DOI": "10.1016/j.pbb.2016.02.005",
"author": "C Dong",
"doi-asserted-by": "publisher",
"first-page": "7",
"journal-title": "Pharmacol Biochem Behav",
"key": "36_CR31",
"unstructured": "Dong C, Zhang JC, Yao W, Ren Q, Yang C, Ma M, et al. Effects of escitalopram, R-citalopram, and reboxetine on serum levels of tumor necrosis factor-alpha, interleukin-10, and depression-like behavior in mice after lipopolysaccharide administration. Pharmacol Biochem Behav. 2016;144:7–12. https://doi.org/10.1016/j.pbb.2016.02.005.",
"volume": "144",
"year": "2016"
},
{
"DOI": "10.3390/ijms16047796",
"author": "K Hashimoto",
"doi-asserted-by": "publisher",
"first-page": "7796",
"issue": "4",
"journal-title": "Int J Mol Sci",
"key": "36_CR32",
"unstructured": "Hashimoto K. Inflammatory biomarkers as differential predictors of antidepressant response. Int J Mol Sci. 2015;16(4):7796–801. https://doi.org/10.3390/ijms16047796.",
"volume": "16",
"year": "2015"
},
{
"DOI": "10.1016/j.bbi.2019.02.021",
"author": "L Wang",
"doi-asserted-by": "publisher",
"first-page": "24",
"journal-title": "Brain Behav Immun",
"key": "36_CR33",
"unstructured": "Wang L, Wang R, Liu L, Qiao D, Baldwin DS, Hou R. Effects of SSRIs on peripheral inflammatory markers in patients with major depressive disorder: a systematic review and meta-analysis. Brain Behav Immun. 2019;79:24–38. https://doi.org/10.1016/j.bbi.2019.02.021.",
"volume": "79",
"year": "2019"
},
{
"DOI": "10.1038/s41380-021-01021-4",
"author": "N Hoertel",
"doi-asserted-by": "publisher",
"first-page": "5199",
"issue": "9",
"journal-title": "Mol Psychiatry",
"key": "36_CR34",
"unstructured": "Hoertel N, Sánchez-Rico M, Vernet R, Beeker N, Jannot AS, Neuraz A, et al. Association between antidepressant use and reduced risk of intubation or death in hospitalized patients with COVID-19: results from an observational study. Mol Psychiatry. 2021;26(9):5199–212. https://doi.org/10.1038/s41380-021-01021-4.",
"volume": "26",
"year": "2021"
},
{
"DOI": "10.1073/pnas.93.15.8072",
"author": "M Hanner",
"doi-asserted-by": "publisher",
"first-page": "8072",
"issue": "15",
"journal-title": "Proc Natl Acad Sci USA",
"key": "36_CR35",
"unstructured": "Hanner M, Moebius FF, Flandorfer A, Knaus HG, Striessnig J, Kempner E, et al. Purification, molecular cloning, and expression of the mammalian sigma1-binding site. Proc Natl Acad Sci USA. 1996;93(15):8072–7. https://doi.org/10.1073/pnas.93.15.8072.",
"volume": "93",
"year": "1996"
},
{
"DOI": "10.1016/j.cell.2007.08.036",
"author": "T Hayashi",
"doi-asserted-by": "publisher",
"first-page": "596",
"issue": "3",
"journal-title": "Cell",
"key": "36_CR36",
"unstructured": "Hayashi T, Su TP. Sigma-1 receptor chaperones at the ER-mitochondrion interface regulate Ca2+ signaling and cell survival. Cell. 2007;131(3):596–610. https://doi.org/10.1016/j.cell.2007.08.036.",
"volume": "131",
"year": "2007"
},
{
"DOI": "10.1016/0014-2999(96)00254-3",
"author": "N Narita",
"doi-asserted-by": "publisher",
"first-page": "117",
"issue": "1",
"journal-title": "Eur J Pharmacol",
"key": "36_CR37",
"unstructured": "Narita N, Hashimoto K, Tomitaka S, Minabe Y. Interaction of selective serotonin reuptake inhibitors with subtypes of sigma receptors in rat brain. Eur J Pharmacol. 1996;307(1):117–9. https://doi.org/10.1016/0014-2999(96)00254-3.",
"volume": "307",
"year": "1996"
},
{
"DOI": "10.1038/sj.npp.1301047",
"author": "K Hashimoto",
"doi-asserted-by": "publisher",
"first-page": "514",
"issue": "3",
"journal-title": "Neuropsychopharmacology",
"key": "36_CR38",
"unstructured": "Hashimoto K, Fujita Y, Iyo M. Phencyclidine-induced cognitive deficits in mice are improved by subsequent subchronic administration of fluvoxamine: role of sigma-1 receptors. Neuropsychopharmacology. 2007;32(3):514–21. https://doi.org/10.1038/sj.npp.1301047.",
"volume": "32",
"year": "2007"
},
{
"DOI": "10.2174/1874241600902010007",
"author": "T Ishima",
"doi-asserted-by": "publisher",
"first-page": "7",
"journal-title": "Open Clin Chem J",
"key": "36_CR39",
"unstructured": "Ishima T, Fujita Y, Kohno M, Kunitachi S, Hotio M, Takatsu Y, et al. Improvement of phencyclidine-induced cognitive deficits in mice by subsequent subchronic administration of fluvoxamine, but not sertraline. Open Clin Chem J. 2009;2:7–11. https://doi.org/10.2174/1874241600902010007.",
"volume": "2",
"year": "2009"
},
{
"DOI": "10.1371/journal.pone.0002558",
"author": "T Nishimura",
"doi-asserted-by": "publisher",
"issue": "7",
"journal-title": "PLoS ONE",
"key": "36_CR40",
"unstructured": "Nishimura T, Ishima T, Iyo M, Hashimoto K. Potentiation of nerve growth factor-induced neurite outgrowth by fluvoxamine: role of sigma-1 receptors, IP3 receptors and cellular signaling pathways. PLoS ONE. 2008;3(7): e2558. https://doi.org/10.1371/journal.pone.0002558.",
"volume": "3",
"year": "2008"
},
{
"DOI": "10.1016/j.ejphar.2014.01.064",
"author": "T Ishima",
"doi-asserted-by": "publisher",
"first-page": "167",
"journal-title": "Eur J Pharmacol",
"key": "36_CR41",
"unstructured": "Ishima T, Fujita Y, Hashimoto K. Interactions of new antidepressants with sigma-1 receptor chaperons and their potentiation of neurite outgrowth in PC12 cells. Eur J Pharmacol. 2014;727:167–73. https://doi.org/10.1016/j.ejphar.2014.01.064.",
"volume": "727",
"year": "2014"
},
{
"DOI": "10.1002/hup.1106",
"author": "I Hindmarch",
"doi-asserted-by": "publisher",
"first-page": "193",
"issue": "3",
"journal-title": "Hum Psychopharmacol",
"key": "36_CR42",
"unstructured": "Hindmarch I, Hashimoto K. Cognition and depression: the effects of fluvoxamine, a sigma-1 receptor agonist, reconsidered. Hum Psychopharmacol. 2010;25(3):193–200. https://doi.org/10.1002/hup.1106.",
"volume": "25",
"year": "2010"
},
{
"DOI": "10.1016/j.pneurobio.2012.09.001",
"author": "K Hashimoto",
"doi-asserted-by": "publisher",
"first-page": "15",
"journal-title": "Prog Neurobiol",
"key": "36_CR43",
"unstructured": "Hashimoto K. Sigma-1 receptor chaperone and brain-derived neurotrophic factor: emerging links between cardiovascular disease and depression. Prog Neurobiol. 2013;100:15–29. https://doi.org/10.1016/j.pneurobio.2012.09.001.",
"volume": "100",
"year": "2013"
},
{
"DOI": "10.1016/j.jphs.2014.11.010",
"author": "K Hashimoto",
"doi-asserted-by": "publisher",
"first-page": "6",
"issue": "1",
"journal-title": "J Pharmacol Sci",
"key": "36_CR44",
"unstructured": "Hashimoto K. Activation of sigma-1 receptor chaperone in the treatment of neuropsychiatric diseases and its clinical implication. J Pharmacol Sci. 2015;127(1):6–9. https://doi.org/10.1016/j.jphs.2014.11.010.",
"volume": "127",
"year": "2015"
},
{
"DOI": "10.1007/978-3-319-50174-1_11",
"author": "Y Albayrak",
"doi-asserted-by": "publisher",
"first-page": "153",
"journal-title": "Adv Exp Med Biol",
"key": "36_CR45",
"unstructured": "Albayrak Y, Hashimoto K. Sigma-1 receptor agonists and their clinical implication in neuropsychiatric disorders. Adv Exp Med Biol. 2017;964:153–61. https://doi.org/10.1007/978-3-319-50174-1_11.",
"volume": "964",
"year": "2017"
},
{
"DOI": "10.1126/scitranslmed.aau5266",
"author": "DA Rosen",
"doi-asserted-by": "publisher",
"issue": "478",
"journal-title": "Sci Transl Med",
"key": "36_CR46",
"unstructured": "Rosen DA, Seki SM, Fernández-Castañeda A, Beiter RM, Eccles JD, Woodfolk JA, et al. Modulation of the sigma-1 receptor-IRE1 pathway is beneficial in preclinical models of inflammation and sepsis. Sci Transl Med. 2019;11(478): eaau5266. https://doi.org/10.1126/scitranslmed.aau5266.",
"volume": "11",
"year": "2019"
},
{
"DOI": "10.1128/JVI.03557-12",
"author": "M Friesland",
"doi-asserted-by": "publisher",
"first-page": "6377",
"issue": "11",
"journal-title": "J Virol",
"key": "36_CR47",
"unstructured": "Friesland M, Mingorance L, Chung J, Chisari FV, Gastaminza P. Sigma-1 receptor regulates early steps of viral RNA replication at the inset of hepatitis C virus infection. J Virol. 2013;87(11):6377–90. https://doi.org/10.1128/JVI.03557-12.",
"volume": "87",
"year": "2013"
},
{
"DOI": "10.1016/j.virusres.2015.03.013",
"author": "C Vasallo",
"doi-asserted-by": "publisher",
"first-page": "100",
"journal-title": "Virus Res",
"key": "36_CR48",
"unstructured": "Vasallo C, Gastaminza P. Cellular stress response in hepatitis C virus infection: mastering a two-edged sword. Virus Res. 2015;209:100–17. https://doi.org/10.1016/j.virusres.2015.03.013.",
"volume": "209",
"year": "2015"
},
{
"DOI": "10.3389/fphar.2020.582310",
"author": "JM Vela",
"doi-asserted-by": "publisher",
"journal-title": "Front Pharmacol",
"key": "36_CR49",
"unstructured": "Vela JM. Repurposing sigma-1 receptor ligands for COVID-19 therapy? Front Pharmacol. 2020;11: 582310. https://doi.org/10.3389/fphar.2020.582310.",
"volume": "11",
"year": "2020"
},
{
"DOI": "10.1038/s41586-020-2286-9",
"author": "DE Gordon",
"doi-asserted-by": "publisher",
"first-page": "459",
"issue": "7816",
"journal-title": "Nature",
"key": "36_CR50",
"unstructured": "Gordon DE, Jang GM, Bouhaddou M, Xu J, Obernier K, White KM, et al. A SARS-CoV-2 protein interaction map reveals targets for drug repurposing. Nature. 2020;583(7816):459–68. https://doi.org/10.1038/s41586-020-2286-9.",
"volume": "583",
"year": "2020"
},
{
"DOI": "10.1126/science.abe9403",
"author": "DE Gordon",
"doi-asserted-by": "publisher",
"issue": "6521",
"journal-title": "Science",
"key": "36_CR51",
"unstructured": "Gordon DE, Hiatt J, Bouhaddou M, Rezelj VV, Ulferts S, Braberg H, et al. Comparative host-coronavirus protein interaction networks reveal pan-viral disease mechanisms. Science. 2020;370(6521): eabe9403. https://doi.org/10.1126/science.abe9403.",
"volume": "370",
"year": "2020"
},
{
"DOI": "10.3389/fphar.2021.652688",
"author": "VP Sukhatme",
"doi-asserted-by": "publisher",
"journal-title": "Front Pharmacol",
"key": "36_CR52",
"unstructured": "Sukhatme VP, Reiersen AM, Vayttaden SJ, Sykhatme W. Fluvoxamine: a review of its mechanisms of actions and its role in COVID-19. Front Pharmacol. 2021;12: 652688. https://doi.org/10.3389/fphar.2021.652688.",
"volume": "12",
"year": "2021"
},
{
"DOI": "10.1080/14728222.2021.1952987",
"author": "JM Brimson",
"doi-asserted-by": "publisher",
"first-page": "435",
"issue": "6",
"journal-title": "Expert Opin Ther Targets",
"key": "36_CR53",
"unstructured": "Brimson JM, Prasanth MI, Malar DS, Brimson S, Thitilertdecha P, Tencomnao T. Drugs that offer the potential to reduce hospitalization and mortality from SARS-CoV-2 infection: the possible role of the sigma-1 receptor and autophagy. Expert Opin Ther Targets. 2021;25(6):435–49. https://doi.org/10.1080/14728222.2021.1952987.",
"volume": "25",
"year": "2021"
},
{
"DOI": "10.33594/000000021",
"author": "C Li",
"doi-asserted-by": "publisher",
"first-page": "280",
"issue": "2",
"journal-title": "Cell Physiol Biochem",
"key": "36_CR54",
"unstructured": "Li C, Wang A, Wu Y, Gulbins E, Grassmé H, Zhao Z. Acid sphingomyelinase-ceramide system in bacterial infections. Cell Physiol Biochem. 2019;52(2):280–301. https://doi.org/10.33594/000000021.",
"volume": "52",
"year": "2019"
},
{
"DOI": "10.3390/ijms22115676",
"author": "N Beckmann",
"doi-asserted-by": "publisher",
"first-page": "5676",
"issue": "11",
"journal-title": "Int J Mol Sci",
"key": "36_CR55",
"unstructured": "Beckmann N, Becker KA. Ceramide and related molecules in viral infections. Int J Mol Sci. 2021;22(11):5676. https://doi.org/10.3390/ijms22115676.",
"volume": "22",
"year": "2021"
},
{
"DOI": "10.3389/fcell.2021.689854",
"author": "K Törnquist",
"doi-asserted-by": "publisher",
"journal-title": "Front Cell Dev Biol",
"key": "36_CR56",
"unstructured": "Törnquist K, Asghar MY, Srinivasan V, Korhonen L, Lindholm D. Sphingolipids as modulators of SARS-CoV-2 infection. Front Cell Dev Biol. 2021;9: 689854. https://doi.org/10.3389/fcell.2021.689854.",
"volume": "9",
"year": "2021"
},
{
"DOI": "10.1159/000315101",
"author": "J Kornhuber",
"doi-asserted-by": "publisher",
"first-page": "9",
"issue": "1",
"journal-title": "Cell Physiol Biochem",
"key": "36_CR57",
"unstructured": "Kornhuber J, Tripal P, Reichel M, Mühle C, Rhein C, Muehlbacher M, et al. Functional inhibitors of acid sphingomyelinase (FIASMAs): a novel pharmacological group of drugs with broad clinical applications. Cell Physiol Biochem. 2010;26(1):9–20. https://doi.org/10.1159/000315101.",
"volume": "26",
"year": "2010"
},
{
"DOI": "10.1038/s41380-021-01309-5",
"author": "J Kornhuber",
"doi-asserted-by": "publisher",
"first-page": "307",
"issue": "1",
"journal-title": "Mol Psychiatry",
"key": "36_CR58",
"unstructured": "Kornhuber J, Hoertel N, Gulbins E. The acid sphingomyelinase/ceramide system in COVID-19. Mol Psychiatry. 2022;27(1):307–14. https://doi.org/10.1038/s41380-021-01309-5.",
"volume": "27",
"year": "2022"
},
{
"DOI": "10.1371/journal.pone.0023852",
"author": "J Kornhuber",
"doi-asserted-by": "publisher",
"issue": "8",
"journal-title": "PLoS ONE",
"key": "36_CR59",
"unstructured": "Kornhuber J, Muehlbacher M, Trapp S, Pechmann S, Friedl A, Reichel M, et al. Identification of novel functional inhibitors of acid sphingomyelinase. PLoS ONE. 2011;6(8): e23852. https://doi.org/10.1371/journal.pone.0023852.",
"volume": "6",
"year": "2011"
},
{
"DOI": "10.1038/nm.3214",
"author": "E Gulbins",
"doi-asserted-by": "publisher",
"first-page": "934",
"issue": "7",
"journal-title": "Nat Med",
"key": "36_CR60",
"unstructured": "Gulbins E, Palmada M, Reichel M, Lüth A, Böhmer C, Amato D, et al. Acid sphingomyelinase-ceramide system mediates effects of antidepressant drugs. Nat Med. 2013;19(7):934–8. https://doi.org/10.1038/nm.3214.",
"volume": "19",
"year": "2013"
},
{
"DOI": "10.1038/s41380-018-0090-9",
"author": "A Gulbins",
"doi-asserted-by": "publisher",
"first-page": "2324",
"issue": "12",
"journal-title": "Mol Psychiatry",
"key": "36_CR61",
"unstructured": "Gulbins A, Schumacher F, Becker KA, Wilker B, Soddemann M, Boldrin F, et al. Antidepressants act by inducing autophagy controlled by sphingomyelin-ceramide. Mol Psychiatry. 2018;23(12):2324–46. https://doi.org/10.1038/s41380-018-0090-9.",
"volume": "23",
"year": "2018"
},
{
"DOI": "10.1002/cpt.2317",
"author": "N Hoertel",
"doi-asserted-by": "publisher",
"first-page": "1498",
"issue": "6",
"journal-title": "Clin Pharmacol Ther",
"key": "36_CR62",
"unstructured": "Hoertel N, Sánchez-Rico M, Gulbins E, Kornhuber J, Carpinteiro A, Lenze EJ, et al. Association between FIASMAs and reduced risk of intubation or death in individuals hospitalized for severe COVID-19: an observational multicenter study. Clin Pharmacol Ther. 2021;110(6):1498–511. https://doi.org/10.1002/cpt.2317.",
"volume": "110",
"year": "2021"
},
{
"DOI": "10.1016/j.biopsych.2007.04.001",
"author": "M Ishikawa",
"doi-asserted-by": "publisher",
"first-page": "878",
"issue": "8",
"journal-title": "Biol Psychiatry",
"key": "36_CR63",
"unstructured": "Ishikawa M, Ishiwata K, Ishii K, Kimura Y, Sakata M, Naganawa M, et al. High occupancy of sigma-1 receptors in the human brain after single oral administration of fluvoxamine: a positron emission tomography study using [11C]SA4503. Biol Psychiatry. 2007;62(8):878–83. https://doi.org/10.1016/j.biopsych.2007.04.001.",
"volume": "62",
"year": "2007"
},
{
"key": "36_CR64",
"unstructured": "Lenze E. Fluvoxamine for early treatment of Covid-19 (Stop Covid 2). ClinicalTrials.gov Identifier: NCT04668950."
},
{
"DOI": "10.1093/ofid/ofab050",
"author": "D Seftel",
"doi-asserted-by": "publisher",
"issue": "2",
"journal-title": "Open Forum Infect Dis",
"key": "36_CR65",
"unstructured": "Seftel D, Boulware DR. Prospective cohort of fluvoxamine for early treatment of coronavirus disease 19. Open Forum Infect Dis. 2021;8(2): ofab050. https://doi.org/10.1093/ofid/ofab050.",
"volume": "8",
"year": "2021"
},
{
"DOI": "10.1016/S2214-109X(21)00448-4",
"author": "G Reis",
"doi-asserted-by": "publisher",
"first-page": "e42",
"issue": "1",
"journal-title": "Lancet Glob Health",
"key": "36_CR66",
"unstructured": "Reis G, Moreira Silva EADS, Medeiros Silva DC, Thabane L, Milagres AC, Ferreira TS, et al. Effect of early treatment with fluvoxamine on risk of emergency care and hospitalization among patients with COVID-19: the TOGETHER randomized, platform clinical trial. Lancet Glob Health. 2022;10(1):e42–51. https://doi.org/10.1016/S2214-109X(21)00448-4.",
"volume": "10",
"year": "2022"
},
{
"DOI": "10.1111/bcp.15126",
"author": "M Calusic",
"doi-asserted-by": "publisher",
"first-page": "2065",
"issue": "5",
"journal-title": "Bri J Clin Pharmacol",
"key": "36_CR67",
"unstructured": "Calusic M, Marcec R, Luksa L, Jurkovic I, Kovac N, Mihaljevic S, et al. Safety and efficacy of fluvoxamine in COVID-19 ICU patients: an open label, prospective cohort trial with matched controls. Bri J Clin Pharmacol. 2022;88(5):2065–73. https://doi.org/10.1111/bcp.15126.",
"volume": "88",
"year": "2022"
},
{
"DOI": "10.1001/jamanetworkopen.2021.33090",
"author": "T Oskotsky",
"doi-asserted-by": "publisher",
"issue": "11",
"journal-title": "JAMA Netw Open",
"key": "36_CR68",
"unstructured": "Oskotsky T, Maric I, Tang A, Oskotsky B, Wong RJ, Aghaeepour N, et al. Mortality risk among patients with COVID-19 prescribed selective serotonin reuptake inhibitor antidepressants. JAMA Netw Open. 2021;4(11): e2133090. https://doi.org/10.1001/jamanetworkopen.2021.33090.",
"volume": "4",
"year": "2021"
},
{
"DOI": "10.3389/fphar.2022.1054644",
"author": "E Pineda",
"doi-asserted-by": "publisher",
"first-page": "1054644",
"journal-title": "Front Pharmacol",
"key": "36_CR69",
"unstructured": "Pineda E, Singh J, Pineda MV, Umanzor JG, Baires F, Benitez LG, et al. Impact of fluvoxamine on outpatient treatment of COVID-19 in Honduras in a prospective observational real-world study. Front Pharmacol. 2022;13:1054644. https://doi.org/10.3389/fphar.2022.1054644.",
"volume": "13",
"year": "2022"
},
{
"DOI": "10.3947/ic.2021.0142",
"author": "H Seo",
"doi-asserted-by": "publisher",
"first-page": "102",
"issue": "1",
"journal-title": "Infect Chemother",
"key": "36_CR70",
"unstructured": "Seo H, Kim H, Bae S, Park S, Chung H, Sung HS, et al. Fluvoxamine treatment of patients with symptomatic COVID-19 in a community treatment center: a preliminary result of randomized controlled trial. Infect Chemother. 2022;54(1):102–13. https://doi.org/10.3947/ic.2021.0142.",
"volume": "54",
"year": "2022"
},
{
"DOI": "10.1056/NEJMoa2201662",
"author": "CT Bramante",
"doi-asserted-by": "publisher",
"first-page": "599",
"issue": "7",
"journal-title": "N Engl J Med",
"key": "36_CR71",
"unstructured": "Bramante CT, Huling JD, Tignanelli CJ, Buse JB, Liebovitz DM, Nicklas JM, et al. Randomized trial of metformin, ivermectin, and fluvoxamine for Covid-19. N Engl J Med. 2022;387(7):599–610. https://doi.org/10.1056/NEJMoa2201662.",
"volume": "387",
"year": "2022"
},
{
"DOI": "10.1001/jama.2022.24100",
"author": "MW McCarthy",
"doi-asserted-by": "publisher",
"first-page": "296",
"issue": "4",
"journal-title": "JAMA",
"key": "36_CR72",
"unstructured": "McCarthy MW, Naggie S, Boulware DR, Lindsell CJ, Stewart TG, Felker GM, et al. Effect of fluvoxamine vs placebo on time to sustained recovery in outpatients with mild to moderate COVID-19: a randomized clinical trial. JAMA. 2023;329(4):296–305. https://doi.org/10.1001/jama.2022.24100.",
"volume": "329",
"year": "2023"
},
{
"DOI": "10.1001/jama.2022.23954",
"author": "A Bhimraj",
"doi-asserted-by": "publisher",
"first-page": "291",
"issue": "4",
"journal-title": "JAMA",
"key": "36_CR73",
"unstructured": "Bhimraj A, Gallagher JC. Lack of benefit of fluvoxamine for COVID-19. JAMA. 2023;329(4):291–2. https://doi.org/10.1001/jama.2022.23954.",
"volume": "329",
"year": "2023"
},
{
"DOI": "10.4088/PCC.22br03337",
"author": "AD Diaz",
"doi-asserted-by": "publisher",
"first-page": "22br03337",
"issue": "5",
"journal-title": "Prim Care Comp CNS Disord",
"key": "36_CR74",
"unstructured": "Diaz AD, Baweja R. Protective effect of fluvoxamine for COVID-19 in obsessive–compulsive disorder: a real-world case-control study. Prim Care Comp CNS Disord. 2022;24(5):22br03337. https://doi.org/10.4088/PCC.22br03337.",
"volume": "24",
"year": "2022"
},
{
"DOI": "10.1056/NEJMc2011400",
"author": "VG Puelles",
"doi-asserted-by": "publisher",
"first-page": "590",
"issue": "6",
"journal-title": "N Engl J Med",
"key": "36_CR75",
"unstructured": "Puelles VG, Lütgehetmann M, Lindenmeyer MT, Sperhake JP, Wong MN, Allweiss L, et al. Multiorgan and renal tropism of SARS-CoV-2. N Engl J Med. 2020;383(6):590–2. https://doi.org/10.1056/NEJMc2011400.",
"volume": "383",
"year": "2020"
},
{
"DOI": "10.1056/NEJMc2019373",
"author": "IH Solomon",
"doi-asserted-by": "publisher",
"first-page": "989",
"issue": "10",
"journal-title": "N Engl J Med",
"key": "36_CR76",
"unstructured": "Solomon IH, Normandin E, Bhattacharyya S, Mukerji SS, Keller K, Ali AS, et al. Neurological features of Covid-19. N Engl J Med. 2020;383(10):989–92. https://doi.org/10.1056/NEJMc2019373.",
"volume": "383",
"year": "2020"
},
{
"DOI": "10.1038/s41593-020-00771-8",
"author": "EM Rhea",
"doi-asserted-by": "publisher",
"first-page": "368",
"issue": "3",
"journal-title": "Nat Neurosci",
"key": "36_CR77",
"unstructured": "Rhea EM, Logsdon AF, Hansen KM, Williams LM, Reed MJ, Baumann KK, et al. The S1 protein of SARS-CoV-2 crosses the blood-brain barrier in mice. Nat Neurosci. 2021;24(3):368–78. https://doi.org/10.1038/s41593-020-00771-8.",
"volume": "24",
"year": "2021"
},
{
"DOI": "10.1038/s41392-021-00719-9",
"author": "L Zhang",
"doi-asserted-by": "publisher",
"first-page": "337",
"issue": "1",
"journal-title": "Signal Transduct Target",
"key": "36_CR78",
"unstructured": "Zhang L, Zhou L, Bao L, Liu J, Zhu H, Lv Q, et al. SAES-CoV-2 crosses the blood–brain barrier accompanied with basement membrane disruption without tight junctions alteration. Signal Transduct Target. 2021;6(1):337. https://doi.org/10.1038/s41392-021-00719-9.",
"volume": "6",
"year": "2021"
},
{
"DOI": "10.3390/biomedicines10010188",
"author": "D Petrovszki",
"doi-asserted-by": "publisher",
"first-page": "188",
"issue": "1",
"journal-title": "Biomedicines",
"key": "36_CR79",
"unstructured": "Petrovszki D, Walter FR, Vigh JP, Kocsis A, Valkai S, Deli MA, Dér A. Penetration of the SARS-CoV-2 spike protein across the blood-brain barrier, as revealed by a combination of a human cell culture model system and optical biosensing. Biomedicines. 2022;10(1):188. https://doi.org/10.3390/biomedicines10010188.",
"volume": "10",
"year": "2022"
},
{
"DOI": "10.1038/s41586-022-04569-5",
"author": "G Douaud",
"doi-asserted-by": "publisher",
"first-page": "697",
"issue": "7907",
"journal-title": "Nature",
"key": "36_CR80",
"unstructured": "Douaud G, Lee S, Alfaro-Almagro F, Arthofer C, Wang C, McCarthy P, et al. SARS-CoV-2 is associated with changes in brain structure in UK Biobank. Nature. 2022;604(7907):697–707. https://doi.org/10.1038/s41586-022-04569-5.",
"volume": "604",
"year": "2022"
},
{
"DOI": "10.1038/s41392-022-01072-1",
"author": "N Sollmann",
"doi-asserted-by": "publisher",
"first-page": "230",
"issue": "1",
"journal-title": "Signal Transduct Target Ther",
"key": "36_CR81",
"unstructured": "Sollmann N, Beer AJ, Kirchhoff F. SARS-CoV-2 infection and the brain: direct evidence for brain changes in milder cases. Signal Transduct Target Ther. 2022;7(1):230. https://doi.org/10.1038/s41392-022-01072-1.",
"volume": "7",
"year": "2022"
},
{
"DOI": "10.1084/jem.20202135",
"author": "E Song",
"doi-asserted-by": "publisher",
"issue": "3",
"journal-title": "J Exp Med",
"key": "36_CR82",
"unstructured": "Song E, Zhang C, Israelow B, Lu-Culligan A, Prado AV, Skriabine S, et al. Neuroinvasion of SARS-CoV-2 in human and mouse brain. J Exp Med. 2021;218(3): e20202135. https://doi.org/10.1084/jem.20202135.",
"volume": "218",
"year": "2021"
},
{
"DOI": "10.1038/s41586-022-05542-y",
"author": "SR Stein",
"doi-asserted-by": "publisher",
"first-page": "758",
"issue": "7941",
"journal-title": "Nature",
"key": "36_CR83",
"unstructured": "Stein SR, Ramelli SC, Grazioli A, Chung JY, Singh M, Yinda CK, et al. SARS-CoV-2 infection and persistence in the human body and brain at autopsy. Nature. 2022;612(7941):758–63. https://doi.org/10.1038/s41586-022-05542-y.",
"volume": "612",
"year": "2022"
},
{
"DOI": "10.1016/j.bbi.2020.03.031",
"author": "Y Wu",
"doi-asserted-by": "publisher",
"first-page": "18",
"journal-title": "Brain Behav Immun",
"key": "36_CR84",
"unstructured": "Wu Y, Xu X, Chen Z, Duan J, Hashimoto K, Yang L, et al. Nervous system involvement after infection with COVID-19 and other coronaviruses. Brain Behav Immun. 2020;87:18–22. https://doi.org/10.1016/j.bbi.2020.03.031.",
"volume": "87",
"year": "2020"
},
{
"DOI": "10.3390/ijms22052681",
"author": "MA Erickson",
"doi-asserted-by": "publisher",
"first-page": "2681",
"issue": "5",
"journal-title": "Int J Mol Sci",
"key": "36_CR85",
"unstructured": "Erickson MA, Rhea EM, Knopp RC, Banks WA. Interactions of SARS-CoV-2 with the blood–brain barrier. Int J Mol Sci. 2021;22(5):2681. https://doi.org/10.3390/ijms22052681.",
"volume": "22",
"year": "2021"
},
{
"DOI": "10.3390/vaccines10010001",
"author": "PR Oldfield",
"doi-asserted-by": "publisher",
"first-page": "1",
"issue": "1",
"journal-title": "Vaccines (Basel)",
"key": "36_CR86",
"unstructured": "Oldfield PR, Hibberd J, Bridle BW. How does severe acute respiratory syndrome-coronavirus-2 affect the brain and its implications for the vaccines currently in use. Vaccines (Basel). 2021;10(1):1. https://doi.org/10.3390/vaccines10010001.",
"volume": "10",
"year": "2021"
},
{
"DOI": "10.1001/jamapsychiatry.2021.0500",
"author": "M Boldrini",
"doi-asserted-by": "publisher",
"first-page": "682",
"issue": "6",
"journal-title": "JAMA Psychiat",
"key": "36_CR87",
"unstructured": "Boldrini M, Canoll PD, Klein RS. How COVID-19 affects the brain. JAMA Psychiat. 2021;78(6):682–3. https://doi.org/10.1001/jamapsychiatry.2021.0500.",
"volume": "78",
"year": "2021"
},
{
"DOI": "10.1007/s12035-021-02696-0",
"author": "TC Theoharides",
"doi-asserted-by": "publisher",
"first-page": "1850",
"issue": "3",
"journal-title": "Mol Neurobiol",
"key": "36_CR88",
"unstructured": "Theoharides TC. Could SARS-CoV-2 spike protein be responsible for long-COVID syndrome? Mol Neurobiol. 2022;59(3):1850–61. https://doi.org/10.1007/s12035-021-02696-0.",
"volume": "59",
"year": "2022"
},
{
"DOI": "10.1001/jamanetworkopen.2021.28568",
"author": "D Groff",
"doi-asserted-by": "publisher",
"issue": "10",
"journal-title": "JAMA Netw Open",
"key": "36_CR89",
"unstructured": "Groff D, Sun A, Ssentongo AE, Ba DM, Parsons N, Poudel GR, et al. Short-term and long-term rates of postacute sequelae of SARS-CoV-2 infection: a systematic review. JAMA Netw Open. 2021;4(10): e2128568. https://doi.org/10.1001/jamanetworkopen.2021.28568.",
"volume": "4",
"year": "2021"
},
{
"DOI": "10.1038/s41579-022-00846-2",
"author": "HE Davis",
"doi-asserted-by": "publisher",
"journal-title": "Nat Rev Microbiol",
"key": "36_CR90",
"unstructured": "Davis HE, McCorkell L, Vogel JM, Topol EJ. Long COVID: major findings, mechanisms and recommendation. Nat Rev Microbiol. 2023. https://doi.org/10.1038/s41579-022-00846-2.",
"year": "2023"
},
{
"DOI": "10.7150/ijbs.75056",
"author": "HC Koc",
"doi-asserted-by": "publisher",
"first-page": "4768",
"issue": "12",
"journal-title": "Int J Biol Sci",
"key": "36_CR91",
"unstructured": "Koc HC, Xiao J, Liu W, Li Y, Chen G. Long COVID and its management. Int J Biol Sci. 2022;18(12):4768–80. https://doi.org/10.7150/ijbs.75056.",
"volume": "18",
"year": "2022"
},
{
"DOI": "10.1101/2022.11.09.22282142",
"author": "H Sidky",
"doi-asserted-by": "publisher",
"journal-title": "medRxiv",
"key": "36_CR92",
"unstructured": "Sidky H, Sahner DK, Girvin AT, Hotaling N, Michael SG, Kurilla MG, et al. Assessing the effect of selective serotonin reuptake inhibitors in the prevention of post-acute sequelae of COVID-19. medRxiv. 2022. https://doi.org/10.1101/2022.11.09.22282142.",
"year": "2022"
},
{
"DOI": "10.1007/s40267-022-00975-x",
"author": "C Fenton",
"doi-asserted-by": "publisher",
"first-page": "65",
"issue": "2",
"journal-title": "Drugs Ther Perspect",
"key": "36_CR93",
"unstructured": "Fenton C, Lee A. Antidepressants with anti-inflammatory properties may be useful in long COVID depression. Drugs Ther Perspect. 2023;39(2):65–70. https://doi.org/10.1007/s40267-022-00975-x.",
"volume": "39",
"year": "2023"
},
{
"DOI": "10.1038/s41380-022-01545-3",
"author": "E Khani",
"doi-asserted-by": "publisher",
"first-page": "3562",
"issue": "9",
"journal-title": "Mol Psychiatry",
"key": "36_CR94",
"unstructured": "Khani E, Entezari-Maleki T. Fluvoxamine and long COVID-19: a new role for sigma-1 receptor (S1R) agonists. Mol Psychiatry. 2022;27(9):3562. https://doi.org/10.1038/s41380-022-01545-3.",
"volume": "27",
"year": "2022"
},
{
"DOI": "10.1038/s41380-022-01546-2",
"author": "Y Hashimoto",
"doi-asserted-by": "publisher",
"first-page": "3563",
"issue": "9",
"journal-title": "Mol Psychiatry",
"key": "36_CR95",
"unstructured": "Hashimoto Y, Suzuki T, Hashimoto K. Comments to “Fluvoxamine and long COVID-19: a new role for sigma-1 receptor (S1R) agonists” by Khani and Entezari-Maleki. Mol Psychiatry. 2022;27(9):3563–4. https://doi.org/10.1038/s41380-022-01546-2.",
"volume": "27",
"year": "2022"
},
{
"DOI": "10.4269/ajtmh.21-1310",
"author": "CM Guo",
"doi-asserted-by": "publisher",
"first-page": "1315",
"issue": "5",
"journal-title": "Am J Trop Med Hyg",
"key": "36_CR96",
"unstructured": "Guo CM, Harari O, Chernecki C, Thorlund K, Forrest JI. Fluvoxamine for the early treatment of COVID-19: a meta-analysis of randomized clinical trials. Am J Trop Med Hyg. 2022;106(5):1315–20. https://doi.org/10.4269/ajtmh.21-1310.",
"volume": "106",
"year": "2022"
},
{
"DOI": "10.1097/MJT.0000000000001496",
"author": "S Bhuta",
"doi-asserted-by": "publisher",
"first-page": "e298",
"issue": "3",
"journal-title": "Am J Ther",
"key": "36_CR97",
"unstructured": "Bhuta S, Khokher W, Kesireddy N, Iftikhar S, Beran A, Mhanna M, et al. Fluvoxamine in nonhospitalized patients with acute COVID-19 infection and the lack of efficacy in reducing rates of hospitalization, mechanical ventilation, and mortality in placebo-controlled trials: a systematic review and meta-analysis. Am J Ther. 2022;29(3):e298–304. https://doi.org/10.1097/MJT.0000000000001496.",
"volume": "29",
"year": "2022"
},
{
"DOI": "10.1001/jamanetworkopen.2022.6269",
"author": "TC Lee",
"doi-asserted-by": "publisher",
"issue": "4",
"journal-title": "JAMA Netw Open",
"key": "36_CR98",
"unstructured": "Lee TC, Vigod S, Bortolussi-Courval É, Hanula R, Boulware DR, Lenze EJ, et al. Fluvoxamine for outpatients management of COVID-19 to prevent hospitalization: a systematic review and meta-analysis. JAMA Netw Open. 2022;5(4): e226269. https://doi.org/10.1001/jamanetworkopen.2022.6269.",
"volume": "5",
"year": "2022"
},
{
"DOI": "10.1016/j.jinf.2022.10.012",
"author": "HA Cheema",
"doi-asserted-by": "publisher",
"first-page": "702",
"issue": "6",
"journal-title": "J Infect",
"key": "36_CR99",
"unstructured": "Cheema HA, Jafar U, Elrashedy AA, Shahid A, Awan RU, Ehsan M, et al. Efficacy and safety of fluvoxamine for the treatment of COVID-19 patients. J Infect. 2022;85(6):702–69. https://doi.org/10.1016/j.jinf.2022.10.012.",
"volume": "85",
"year": "2022"
},
{
"DOI": "10.1016/j.jiph.2022.10.010",
"author": "LC Lu",
"doi-asserted-by": "publisher",
"first-page": "1259",
"issue": "11",
"journal-title": "J Infect Public Health",
"key": "36_CR100",
"unstructured": "Lu LC, Chao CM, Chang SP, Lan SH, Lai CC. Effect of fluvoxamine on outcomes of nonhospitalized patients with COVID-19: a systematic review and meta-analysis. J Infect Public Health. 2022;15(11):1259–64. https://doi.org/10.1016/j.jiph.2022.10.010.",
"volume": "15",
"year": "2022"
},
{
"DOI": "10.1016/j.jinf.2022.11.011",
"author": "R Marcec",
"doi-asserted-by": "publisher",
"first-page": "154",
"issue": "2",
"journal-title": "J Infect",
"key": "36_CR101",
"unstructured": "Marcec R, Dodig VM, Likic R. A meta-analysis regarding fluvoxamine and hospitalization risk of COVID-19 patients: TOGETHER making a difference. J Infect. 2023;86(2):154–225. https://doi.org/10.1016/j.jinf.2022.11.011.",
"volume": "86",
"year": "2023"
},
{
"DOI": "10.1016/j.jinf.2022.11.016",
"author": "V Trkulja",
"doi-asserted-by": "publisher",
"first-page": "154",
"issue": "2",
"journal-title": "J Infect",
"key": "36_CR102",
"unstructured": "Trkulja V. There is no reasonable evidence to support efficacy of fluvoxamine in prevention of disease deterioration in COVID-19 outpatients: a comment on two recent meta-analyses advocating its use. J Infect. 2023;86(2):154–225. https://doi.org/10.1016/j.jinf.2022.11.016.",
"volume": "86",
"year": "2023"
},
{
"DOI": "10.1007/s00228-022-03447-3",
"author": "V Trkulja",
"doi-asserted-by": "publisher",
"first-page": "321",
"issue": "2",
"journal-title": "Eur J Clin Pharmacol",
"key": "36_CR103",
"unstructured": "Trkulja V. Why we should not recommend or offer fluvoxamine to COVID-19 patients? Eur J Clin Pharmacol. 2023;79(2):321–2. https://doi.org/10.1007/s00228-022-03447-3.",
"volume": "79",
"year": "2023"
},
{
"DOI": "10.1016/j.cmi.2023.01.010",
"author": "J Deng",
"doi-asserted-by": "publisher",
"journal-title": "Clin Microbiol Infect",
"key": "36_CR104",
"unstructured": "Deng J, Rayner D, Ba Ramaraju H, Abbas U, Carcia C, Heybati K, et al. Efficacy and safety of selective serotonin reuptake inhibitors in COVID-19 management: a systematic review and meta-analysis. Clin Microbiol Infect. 2023. https://doi.org/10.1016/j.cmi.2023.01.010.",
"year": "2023"
},
{
"DOI": "10.1038/s41380-023-02004-3",
"doi-asserted-by": "publisher",
"key": "36_CR105",
"unstructured": "Kirenga BJ, Mugenyi L, Sánchez-Rico M, Kyobe H, Muttamba W, Mugume R, et al. Association of fluvoxamine with mortality and symptom resolution among inpatients with COVID-19 in Uganda: a prospective interventional open-label cohort study. Mol Psychiatry. 2023;1–8. https://doi.org/10.1038/s41380-023-02004-3."
},
{
"DOI": "10.3390/ijms23073812",
"author": "M Mahdi",
"doi-asserted-by": "publisher",
"first-page": "3812",
"issue": "7",
"journal-title": "Int J Mol Sci",
"key": "36_CR106",
"unstructured": "Mahdi M, Hermán L, Réthelyi JM, Bálint BL. Potential role of the antidepressants fluoxetine and fluvoxamine in the treatment of COVID-19. Int J Mol Sci. 2022;23(7):3812. https://doi.org/10.3390/ijms23073812.",
"volume": "23",
"year": "2022"
},
{
"DOI": "10.4269/ajtmh.22-0106",
"author": "FP Mills",
"doi-asserted-by": "publisher",
"first-page": "101",
"issue": "1",
"journal-title": "Am J Trop Med Hyg",
"key": "36_CR107",
"unstructured": "Mills FP, Reis G, Wilson LA, Thorlund K, Forrest JI, Guo CM, et al. Early treatment with fluvoxamine among patients with COVID-19: a cost-consequence model. Am J Trop Med Hyg. 2022;108(1):101–6. https://doi.org/10.4269/ajtmh.22-0106.",
"volume": "108",
"year": "2022"
}
],
"reference-count": 107,
"references-count": 107,
"relation": {},
"resource": {
"primary": {
"URL": "https://link.springer.com/10.1007/s44192-023-00036-3"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subtitle": [],
"title": "Overview of the potential use of fluvoxamine for COVID-19 and long COVID",
"type": "journal-article",
"update-policy": "http://dx.doi.org/10.1007/springer_crossmark_policy",
"volume": "3"
}