Fluvoxamine: A Review of Its Mechanism of Action and Its Role in COVID-19
et al., Front. Pharmacol., doi:10.3389/fphar.2021.652688, Apr 2021
30th treatment shown to reduce risk in
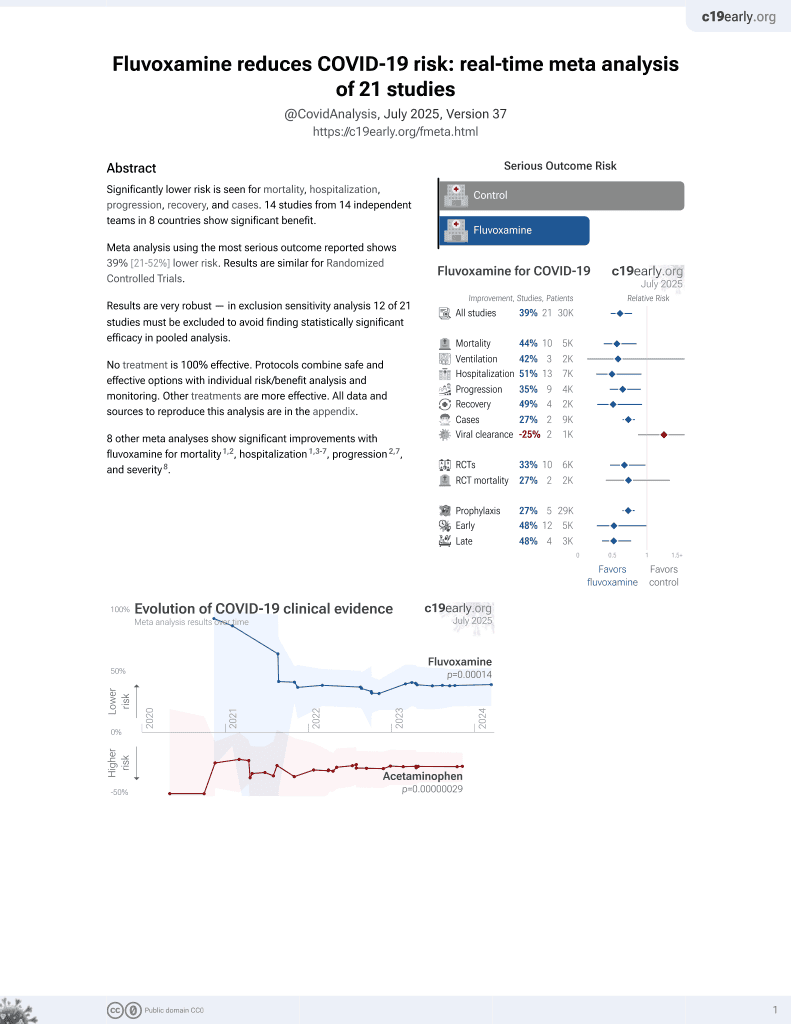
November 2021, now with p = 0.00014 from 21 studies, recognized in 2 countries.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
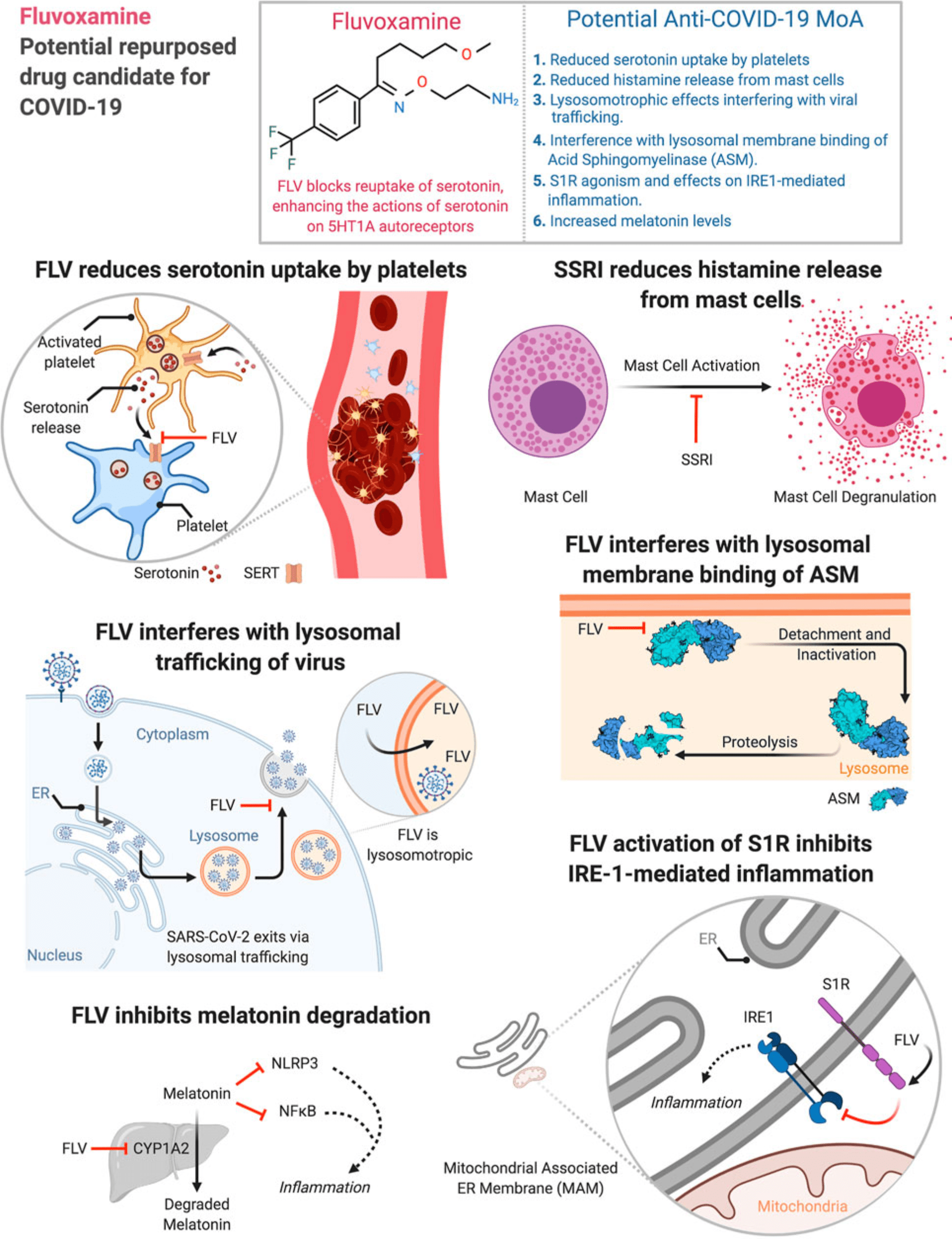
Review of mechanisms of action of fluvoxamine and other SSRIs that could be beneficial for COVID-19 treatment, including lower platelet aggregation, decreased mast cell degranulation, interference with endolysosomal viral trafficking, regulation of inositol-requiring enzyme 1α-driven inflammation, and increased melatonin levels.
1.
Scheim et al., Back to the Basics of SARS-CoV-2 Biochemistry: Microvascular Occlusive Glycan Bindings Govern Its Morbidities and Inform Therapeutic Responses, Viruses, doi:10.3390/v16040647.
2.
Hashimoto, K., Overview of the potential use of fluvoxamine for COVID-19 and long COVID, Discover Mental Health, doi:10.1007/s44192-023-00036-3.
3.
Hashimoto (B) et al., Mechanisms of action of fluvoxamine for COVID-19: a historical review, Molecular Psychiatry, doi:10.1038/s41380-021-01432-3.
4.
Hashimoto (C) et al., Old drug fluvoxamine, new hope for COVID-19, European Archives of Psychiatry and Clinical Neuroscience, doi:10.1007/s00406-021-01326-z.
5.
Hoertel et al., Repurposing antidepressants inhibiting the sphingomyelinase acid/ceramide system against COVID-19: current evidence and potential mechanisms, Molecular Psychiatry, doi:10.1038/s41380-021-01254-3.
Sukhatme et al., 20 Apr 2021, peer-reviewed, 4 authors.
Fluvoxamine: A Review of Its Mechanism of Action and Its Role in COVID-19
Frontiers in Pharmacology, doi:10.3389/fphar.2021.652688
Fluvoxamine is a well-tolerated, widely available, inexpensive selective serotonin reuptake inhibitor that has been shown in a small, double-blind, placebo-controlled, randomized study to prevent clinical deterioration of patients with mild coronavirus disease 2019 . Fluvoxamine is also an agonist for the sigma-1 receptor, through which it controls inflammation. We review here a body of literature that shows important mechanisms of action of fluvoxamine and other SSRIs that could play a role in COVID-19 treatment. These effects include: reduction in platelet aggregation, decreased mast cell degranulation, interference with endolysosomal viral trafficking, regulation of inositolrequiring enzyme 1α-driven inflammation and increased melatonin levels, which collectively have a direct antiviral effect, regulate coagulopathy or mitigate cytokine storm, which are known hallmarks of severe COVID-19.
Conflict of Interest: Author VVS was employed by the company GlobalCures, Inc. Author AR has received research support for clinical trials of fluvoxamine for COVID-19 from the Taylor Family Institute for Innovative Psychiatric Treatment at Washington University, Fast Grants, and the COVID-19 Early Treatment Fund, and she is an inventor on a patent application filed by Washington University in St. Louis, which is relevant to methods of treating COVID-19, including fluvoxamine. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. Copyright © 2021 Sukhatme, Reiersen, Vayttaden and Sukhatme. This is an openaccess article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
References
Ackermann, Verleden, Kuehnel, Haverich, Welte et al., Pulmonary vascular endothelialitis, thrombosis, and angiogenesis in covid-19, N. Engl. J. Med, doi:10.1056/nejmoa2015432
Alidjinou, Bertin, Sane, Caloone, Engelmann et al., Emergence of fluoxetine-resistant variants during treatment of human pancreatic cell cultures persistently infected with coxsackievirus B4, Viruses, doi:10.3390/v11060486
Althaus, Marini, Zlamal, Pelzl, Singh et al., Antibody-induced procoagulant platelets in severe COVID-19 infection, Blood, doi:10.1182/blood.2020008762
Armocida, Palmieri, Frati, Santoro, Pesce, How SARS-Cov-2 can involve the central nervous system. A systematic analysis of literature of the department of human neurosciences of Sapienza University, Italy, J. Clin. Neurosci, doi:10.1016/j.jocn.2020.07.007
Baharav, Bar, Taler, Gil-Ad, Karp et al., Immunomodulatory effect of sertraline in a rat model of rheumatoid arthritis, Neuroimmunomodulation, doi:10.1159/000339109
Bale, Jr, Virus and immune-mediated encephalitides: epidemiology, diagnosis, treatment, and prevention, Pediatr. Neurol, doi:10.1016/j.pediatrneurol.2015.03.013
Bauer, Manganaro, Zonsics, Strating, El Kazzi et al., Fluoxetine inhibits enterovirus replication by targeting the viral 2C protein in a stereospecific manner, ACS Infect. Dis, doi:10.1021/acsinfecdis.9b00179
Berger, Gray, Roth, The expanded biology of serotonin, Annu. Rev. Med, doi:10.1146/annurev.med.60.042307.110802
Bernales, Mcdonald, Walter, Autophagy counterbalances endoplasmic reticulum expansion during the unfolded protein response, Plos Biol, doi:10.1371/journal.pbio.0040423
Breiden, Sandhoff, Emerging mechanisms of drug-induced phospholipidosis, Biol. Chem, doi:10.1515/hsz-2019-0270
Carneiro, Cook, Murphy, Blakely, Interactions between integrin αIIbβ3 and the serotonin transporter regulate serotonin transport and platelet aggregation in mice and humans, J. Clin. Invest, doi:10.1172/jci33374
Carpinteiro, Edwards, Hoffmann, Kochs, Gripp et al., Pharmacological inhibition of acid sphingomyelinase prevents uptake of SARS-CoV-2 by epithelial cells, Cel Rep. Med, doi:10.1016/j.xcrm.2020.100142
Castano-Rodriguez, Honrubia, Gutierrez-Alvarez, Dediego, Nieto-Torres et al., Role of severe acute respiratory syndrome coronavirus viroporins E, 3a, and 8a in replication and pathogenesis, mBio, doi:10.1128/mbio.02325-17
Celada, Dolera, Alvarez, Artigas, Effects of acute and chronic treatment with fluvoxamine on extracellular and platelet serotonin in the blood of major depressive patients. Relationship to clinical improvement, J. Affective Disord, doi:10.1016/0165-0327(92)90082-h
Chan, Siu, Chin, Yuen, Zheng, Modulation of the unfolded protein response by the severe acute respiratory syndrome coronavirus spike protein, Jvi, doi:10.1128/jvi.00659-06
Chen, Wu, Guo, Cao, Huang et al., Clinical and immunological features of severe and moderate coronavirus disease 2019, J. Clin. Invest, doi:10.1172/jci137244
Chen, Xiao, Chen, Luo, Wang et al., Effects of fluoxetine on mast cell morphology and protease-1 expression in gastric antrum in a rat model of depression, Wjg, doi:10.3748/wjg.14.6993
Chen, Zhou, Dong, Qu, Gong et al., Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study, The Lancet, doi:10.1016/s0140-6736(20)30211-7
Cheng, Yang, Xia, Qiu, Wang et al., The nonstructural protein 2C of a Picorna-like virus displays nucleic acid helix destabilizing activity that can be functionally separated from its ATPase activity, J. Virol, doi:10.1128/jvi.00245-13
Chu, Chan, Zhang, Wang, Yuan et al., Middle East respiratory syndrome coronavirus and bat coronavirus HKU9 both can utilize GRP78 for attachment onto host cells, J. Biol. Chem, doi:10.1074/jbc.ra118.001897
Cloutier, Allaeys, Marcoux, Machlus, Mailhot et al., Platelets release pathogenic serotonin and return to circulation after immune complex-mediated sequestration, Proc. Natl. Acad. Sci, doi:10.1073/pnas.1720553115
Cottam, Maier, Manifava, Vaux, Chandra-Schoenfelder et al., Coronavirus nsp6 proteins generate autophagosomes from the endoplasmic reticulum via an omegasome intermediate, Autophagy, doi:10.4161/auto.7.11.16642
Dediego, Nieto-Torres, Jimenez-Guardeno, Regla-Nava, Alvarez et al., Severe acute respiratory syndrome coronavirus envelope protein regulates cell stress response and apoptosis, Plos Pathog, doi:10.1371/journal.ppat.1002315
Dell'osso, Allen, Hollander, Fluvoxamine: a selective serotonin re-uptake inhibitor for the treatment of obsessive-compulsive disorder, Expert Opin. Pharmacother, doi:10.1517/14656566.6.15.2727
Drugbank, None, Fluvoxamine
Dube, Le Coupanec, Wong, Rini, Desforges et al., Axonal transport enables neuron-to-neuron propagation of human coronavirus OC43, J. Virol, doi:10.1128/jvi.00404-18
Duerschmied, Suidan, Demers, Herr, Carbo et al., Platelet serotonin promotes the recruitment of neutrophils to sites of acute inflammation in mice, Blood, doi:10.1182/blood-2012-06-437392
Fajgenbaum, Cytokine storm, N. Engl. J. Med, doi:10.1056/nejmra2026131
Fda, EUA for casirivimab and imdevimab for the treatment of mild to moderate COVID-19
Fda, Fluvoxamine maleate tablets label
Ferjan, Erjavec, Changes in histamine and serotonin secretion from rat peritoneal mast cells caused by antidepressants, Inflamm. Res, doi:10.1007/bf02265168
Friesland, Mingorance, Chung, Chisari, Gastaminza, Sigma-1 receptor regulates early steps of viral RNA replication at the onset of hepatitis C virus infection, J. Virol, doi:10.1128/jvi.03557-12
Fukuda, Ushio, Kawasaki, Niyonsaba, Takeuchi et al., Expression and functional characterization of retinoic acid-inducible gene-I-like receptors of mast cells in response to viral infection, J. Innate Immun, doi:10.1159/000343895
Fung, Liao, Liu, Regulation of stress responses and translational control by coronavirus, Viruses, doi:10.3390/v8070184
Fung, Liu, The ER stress sensor IRE1 and MAP kinase ERK modulate autophagy induction in cells infected with coronavirus infectious bronchitis virus, Virology, doi:10.1016/j.virol.2019.05.002
García, Volt, Venegas, Doerrier, Escames et al., Disruption of the NF-κB/NLRP3 connection by melatonin requires retinoid-related orphan receptor-α and blocks the septic response in mice, FASEB J, doi:10.1096/fj.15-273656
Geiser, Conrad, Imbierowicz, Meier, Liedtke et al., Coagulation activation and fibrinolysis impairment are reduced in patients with anxiety and depression when medicated with serotonergic antidepressants, Psychiatry Clin. Neurosci, doi:10.1111/j.1440-1819.2011.02241.x
Gekker, Hu, Sheng, Rock, Lokensgard et al., Cocaine-induced HIV-1 expression in microglia involves sigma-1 receptors and transforming growth factor-β1, Int. Immunopharmacology, doi:10.1016/j.intimp.2005.12.005
Ghareghani, Zibara, Sadeghi, Dokoohaki, Sadeghi et al., Fluvoxamine stimulates oligodendrogenesis of cultured neural stem cells and attenuates inflammation and demyelination in an animal model of multiple sclerosis, Sci. Rep, doi:10.1038/s41598-017-04968-z
Ghosh, Dellibovi-Ragheb, Kerviel, Pak, Qiu et al., β-Coronaviruses use lysosomes for egress instead of the biosynthetic secretory pathway, Cell, doi:10.1016/j.cell.2020.10.039
Gordon, Hiatt, Bouhaddou, Rezelj, Ulferts et al., Comparative host-coronavirus protein interaction networks reveal pan-viral disease mechanisms, JScience, doi:10.1126/science.abe9403
Grunwell, Giacalone, Stephenson, Margaroli, Dobosh et al., Neutrophil dysfunction in the airways of children with acute respiratory failure due to lower respiratory tract viral and bacterial coinfections, Sci. Rep, doi:10.1038/s41598-019-39726-w
Gulbins, Palmada, Reichel, Lüth, Böhmer et al., Acid sphingomyelinase-ceramide system mediates effects of antidepressant drugs, Nat. Med, doi:10.1038/nm.3214
Ha, Van Krieken, Carlos, Lee, The stress-inducible molecular chaperone GRP78 as potential therapeutic target for coronavirus infection, J. Infect, doi:10.1016/j.jinf.2020.06.017
Hallifax, Houston, Saturable uptake of lipophilic amine drugs into isolated hepatocytes: mechanisms and consequences for quantitative clearance prediction, Drug Metab. Dispos, doi:10.1124/dmd.107.015131
Hanner, Moebius, Flandorfer, Knaus, Striessnig et al., Purification, molecular cloning, and expression of the mammalian sigma1-binding site, Proc. Natl. Acad. Sci, doi:10.1073/pnas.93.15.8072
Hashimoto, Activation of sigma-1 receptor chaperone in the treatment of neuropsychiatric diseases and its clinical implication, J. Pharmacol. Sci, doi:10.1016/j.jphs.2014.11.010
Hashimoto, Repurposing of CNS drugs to treat COVID-19 infection: targeting the sigma-1 receptor, Eur. Arch. Psychiatry Clin. Neurosci, doi:10.1162/2e3983f5.c37587dd
Hayashi, Su, Sigma-1 receptor chaperones at the ERmitochondrion interface regulate Ca(2+) signaling and cell survival, Cell, doi:10.1016/j.cell.2007.08.036
Helms, Kremer, Merdji, Clere-Jehl, Schenck et al., Neurologic features in severe SARS-CoV-2 infection, N. Engl. J. Med, doi:10.1056/nejmc2008597
Helms, Kremer, Merdji, Schenck, Severac et al., Delirium and encephalopathy in severe COVID-19: a cohort analysis of ICU patients, Crit. Care, doi:10.1186/s13054-020-03200-1
Hinte, Van Anken, Tirosh, Brune, Repression of viral gene expression and replication by the unfolded protein response effector XBP1u, Elife, doi:10.7554/elife.51804
Hoertel, Sánchez-Rico, Vernet, Beeker, Jannot et al., Association between antidepressant use and reduced risk of intubation or death in hospitalized patients with COVID-19: results from an observational study, Mol. Psychiat, doi:10.1038/s41380-021-01021-4
Homolak, Kodvanj, Widely available lysosome targeting agents should be considered as potential therapy for COVID-19, Int. J. Antimicrob. Agents, doi:10.1016/j.ijantimicag.2020.106044
Huang, Wang, Li, Ren, Zhao et al., Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China, The Lancet, doi:10.1016/s0140-6736(20)30183-5
Härtter, Wang, Weigmann, Friedberg, Arand et al., Differential effects of fluvoxamine and other antidepressants on the biotransformation of melatonin, J. Clin. Psychopharmacol, doi:10.1097/00004714-200104000-00008
Irons, Fluvoxamine in the treatment of anxiety disorders, Neuropsychiatr. Dis. Treat
Ishima, Fujita, Hashimoto, Interaction of new antidepressants with sigma-1 receptor chaperones and their potentiation of neurite outgrowth in PC12 cells, Eur. J. Pharmacol, doi:10.1016/j.ejphar.2014.01.064
Javors, Houston, Tekell, Brannan, Frazer, Reduction of platelet serotonin content in depressed patients treated with either paroxetine or desipramine, Int. J. Neuropsychopharm, doi:10.1017/s146114570000198x
Joseph, Puttaswamy, Krovvidi, Non-respiratory functions of the lung, Continuing Educ. Anaesth. Crit. Care Pain, doi:10.1093/bjaceaccp/mks060
Kalkman, Feuerbach, Antidepressant therapies inhibit inflammation and microglial M1-polarization, Pharmacol. Ther, doi:10.1016/j.pharmthera.2016.04.001
Kazmi, Hensley, Pope, Funk, Loewen et al., Lysosomal sequestration (trapping) of lipophilic amine (cationic amphiphilic) drugs in immortalized human hepatocytes (Fa2N-4 cells)
Kim, Xu, Reed, Cell death and endoplasmic reticulum stress: disease relevance and therapeutic opportunities, Nat. Rev. Drug Discov, doi:10.1038/nrd2755
Kornhuber, Tripal, Reichel, Terfloth, Bleich et al., Identification of new functional inhibitors of acid sphingomyelinase using a Structure−Property−Activity relation model, J. Med. Chem, doi:10.1021/jm070524a
Lee, Kuo, Lin, Fu, Lin et al., Dengue virus-induced ER stress is required for autophagy activation, viral replication, and pathogenesis both in vitro and in vivo, Sci. Rep, doi:10.1038/s41598-017-18909-3
Lenze, Fluvoxamine for early treatment of covid-19 (stop covid 2)
Lenze, Mattar, Zorumski, Stevens, Schweiger et al., Fluvoxamine vs placebo and clinical deterioration in outpatients with symptomatic COVID-19, JAMA, doi:10.1001/jama.2020.22760
Leung, Shore, Fluvoxamine-associated bleeding, Can. J. Psychiatry, doi:10.1177/070674379604100919
Lu, Zheng, Xu, Schwarz, Du et al., Severe acute respiratory syndrome-associated coronavirus 3a protein forms an ion channel and modulates virus release, Proc. Natl. Acad. Sci, doi:10.1073/pnas.0605402103
Manganaro, Zonsics, Bauer, Lorenzo Lopez, Donselaar et al., Synthesis and antiviral effect of novel fluoxetine analogues as enterovirus 2C inhibitors, Antiviral Res, doi:10.1016/j.antiviral.2020.104781
Martin, Eades, Thompson, Huppler, Gilbert, The effects of morphine-and nalorphine-like drugs in the nondependent and morphine-dependent chronic spinal dog, J. Pharmacol. Exp. Ther
Martinon, Chen, Lee, Glimcher, TLR activation of the transcription factor XBP1 regulates innate immune responses in macrophages, Nat. Immunol, doi:10.1038/ni.1857
Mccloskey, Postolache, Vittone, Nghiem, Monsale et al., Selective serotonin reuptake inhibitors: measurement of effect on platelet function, Translational Res, doi:10.1016/j.trsl.2007.10.004
Motta Junior, Miggiolaro, Nagashima, De Paula, Baena et al., Mast cells in alveolar septa of COVID-19 patients: a pathogenic pathway that may link interstitial edema to immunothrombosis, Front. Immunol, doi:10.3389/fimmu.2020.574862
Naji Esfahani, Rafiee, Haghjooy Javanmard, Evaluation of the effect of antidepressant drug, fluvoxamine, on cyclooxygenase-2 protein expression in lipopolysaccharide-stimulated macrophages, Adv. Biomed. Res, doi:10.4103/abr.abr_141_18
Narayan, Cellar, Mallison, Price, Nelson et al., Serotonin transporter-blocking properties of nefazodone assessed by measurement of platelet serotonin, J. Clin. Psychopharmacol, doi:10.1097/00004714-199802000-00011
Narita, Hashimoto, Tomitaka, Minabe, Interactions of selective serotonin reuptake inhibitors with subtypes of σ receptors in rat brain, Eur. J. Pharmacol, doi:10.1016/0014-2999(96)00254-3
Nazimek, Strobel, Bryniarski, Kozlowski, Filipczak-Bryniarska et al., The role of macrophages in anti-inflammatory activity of antidepressant drugs, Immunobiology, doi:10.1016/j.imbio.2016.07.001
Nazy, Jevtic, Moore, Huynh, Smith et al., Platelet-activating immune complexes identified in critically ill COVID-19 patients suspected of heparin-induced thrombocytopenia, J. Thromb. Haemost, doi:10.1111/jth.15283
Nhlbi, NIH ACTIV trial of blood thinners pauses enrollment of critically ill COVID-19 patients
Ni, Watts, 5-hydroxytryptamine in the cardiovascular system: focus on the serotonin transporter (SERT), Clin. Exp. Pharmacol. Physiol, doi:10.1111/j.1440-1681.2006.04410.x
Nishimura, Ishima, Iyo, Hashimoto, Potentiation of nerve growth factor-induced neurite outgrowth by fluvoxamine: role of sigma-1 receptors, IP3 receptors and cellular signaling pathways, PLoS One, doi:10.1371/journal.pone.0002558
Ogata, Hino, Saito, Morikawa, Kondo et al., Autophagy is activated for cell survival after endoplasmic ReticulumStress, Mcb, doi:10.1128/mcb.01453-06
Prentice, Mcauliffe, Lu, Subbarao, Denison, Identification and characterization of severe acute respiratory syndrome coronavirus replicase proteins, Jvi, doi:10.1128/jvi.78.18.9977-9986.2004
Qiu, Zheng, Chang, Zhao, Tan et al., Tolllike receptor-mediated IRE1α activation as a therapeutic target for inflammatory arthritis, EMBO J, doi:10.1038/emboj.2013.183
Rafiee, Hajhashemi, Javanmard, Fluvoxamine inhibits some inflammatory genes expression in LPS/stimulated human endothelial cells, U937 macrophages, and carrageenan-induced paw edema in rat, Iran J. Basic Med. Sci, doi:10.22038/ijbms.2016.7598
Ramlall, Zucker, Tatonetti, Melatonin is significantly associated with survival of intubated COVID-19 patients, medRxiv, doi:10.1101/2020.10.15.20213546
Ratajczak, Kucia, SARS-CoV-2 infection and overactivation of Nlrp3 inflammasome as a trigger of cytokine "storm" and risk factor for damage of hematopoietic stem cells, Leukemia, doi:10.1038/s41375-020-0887-9
Rosen, Seki, Fernandez-Castaneda, Beiter, Eccles et al., Modulation of the sigma-1 receptor-IRE1 pathway is beneficial in preclinical models of inflammation and sepsis, Sci. Transl Med, doi:10.1126/scitranslmed.aau5266
Schloer, Brunotte, Goretzko, Mecate-Zambrano, Korthals et al., Targeting the endolysosomal host-SARS-CoV-2 interface by clinically licensed functional inhibitors of acid sphingomyelinase (FIASMA) including the antidepressant fluoxetine, Emerging Microbes & Infections, doi:10.1080/22221751.2020.1829082
Seftel, Boulware, Prospective cohort of fluvoxamine for early treatment of COVID-19, Open Forum Infect. Dis, doi:10.1093/ofid/ofab050
Sukhatme, Sukhatme, A call to action: immediate deployment of select repurposed drugs for COVID-19 outpatient treatment, doi:10.1377/hblog20210208.174907/full/
Sundstrom, Little, Villinger, Ellis, Ansari, Signaling through Toll-like receptors triggers HIV-1 replication in latently infected mast cells, J. Immunol, doi:10.4049/jimmunol.172.7.4391
Szabo, Kovacs, Frecska, Rajnavolgyi, Psychedelic N,Ndimethyltryptamine and 5-methoxy-N,N-dimethyltryptamine modulate innate and adaptive inflammatory responses through the sigma-1 receptor of human monocyte-derived dendritic cells, PLoS One, doi:10.1371/journal.pone.0106533
Taler, Gil-Ad, Korob, Weizman, The immunomodulatory effect of the antidepressant sertraline in an experimental autoimmune encephalomyelitis mouse model of multiple sclerosis, Neuroimmunomodulation, doi:10.1159/000321634
Taler, Gil-Ad, Lomnitski, Korov, Baharav et al., Immunomodulatory effect of selective serotonin reuptake inhibitors (SSRIs) on human T lymphocyte function and gene expression, Eur. Neuropsychopharmacol, doi:10.1016/j.euroneuro.2007.03.010
Tay, Poh, Rénia, Macary, Ng, The trinity of COVID-19: immunity, inflammation and intervention, Nat. Rev. Immunol, doi:10.1038/s41577-020-0311-8
Theoharides, COVID -19, pulmonary mast cells, cytokine storms, and beneficial actions of luteolin, Biofactors, doi:10.1002/biof.1633
Thomas, Vane, 5-hydroxytryptamine in the circulation of the dog, Nature, doi:10.1038/216335a0
Tynan, Weidenhofer, Hinwood, Cairns, Day et al., A comparative examination of the anti-inflammatory effects of SSRI and SNRI antidepressants on LPS stimulated microglia, Brain Behav. Immun, doi:10.1016/j.bbi.2011.12.011
Ulferts, Van Der Linden, Thibaut, Lanke, Leyssen et al., Selective serotonin reuptake inhibitor fluoxetine inhibits replication of human enteroviruses B and D by targeting viral protein 2C, Antimicrob. Agents Chemother, doi:10.1128/aac.02084-12
Van Den Berg, Velde, Severe COVID-19: NLRP3 inflammasome dysregulated, Front. Immunol, doi:10.3389/fimmu.2020.01580
Vanhoutte, Platelet-derived serotonin, the endothelium, and cardiovascular disease, J. Cardiovasc. Pharmacol, doi:10.1097/00005344-199100175-00003
Vela, Repurposing sigma-1 receptor ligands for COVID-19 therapy?, Front. Pharmacol, doi:10.3389/fphar.2020.582310
Versteeg, Van De Nes, Bredenbeek, Spaan, The coronavirus spike protein induces endoplasmic reticulum stress and upregulation of intracellular chemokine mRNA concentrations, J. Virol, doi:10.1128/jvi.01033-07
Wang, Lai, Bernard, Macleod, Cogen et al., Skin mast cells protect mice against vaccinia virus by triggering mast cell receptor S1PR2 and releasing antimicrobial peptides, J.I, doi:10.4049/jimmunol.1101703
Wishart, Feunang, Guo, Lo, Marcu et al., DrugBank 5.0: a major update to the DrugBank database for 2018, Nucleic Acids Res, doi:10.1093/nar/gkx1037
Xue, Fu, Ma, Zhang, Li et al., The PERK arm of the unfolded protein response negatively regulates transmissible gastroenteritis virus replication by suppressing protein translation and promoting type I interferon production, J. Virol, doi:10.1128/jvi.00431-18
Yang, Shen, Targeting the endocytic pathway and autophagy process as a novel therapeutic strategy in COVID-19, Int. J. Biol. Sci, doi:10.7150/ijbs.45498
Yesilkaya, Balcioglu, Sahin, Reissuing the sigma receptors for SARS-CoV-2, J. Clin. Neurosci, doi:10.1016/j.jocn.2020.08.014
Yue, Nabar, Shi, Kamenyeva, Xiao et al., SARS-Coronavirus Open Reading Frame-3a drives multimodal necrotic cell death, Cell Death Dis, doi:10.1038/s41419-018-0917-y
Zaid, Guessous, Puhm, Elhamdani, Chentoufi et al., Platelet reactivity to thrombin differs between patients with COVID-19 and those with ARDS unrelated to COVID-19, Blood Adv, doi:10.1182/bloodadvances.2020003513
Zuo, Quinn, Kye, Cooper, Damoiseaux et al., Fluoxetine is a potent inhibitor of coxsackievirus replication, Antimicrob. Agents Chemother, doi:10.1128/aac.00983-12
DOI record:
{
"DOI": "10.3389/fphar.2021.652688",
"ISSN": [
"1663-9812"
],
"URL": "http://dx.doi.org/10.3389/fphar.2021.652688",
"abstract": "<jats:p>Fluvoxamine is a well-tolerated, widely available, inexpensive selective serotonin reuptake inhibitor that has been shown in a small, double-blind, placebo-controlled, randomized study to prevent clinical deterioration of patients with mild coronavirus disease 2019 (COVID-19). Fluvoxamine is also an agonist for the sigma-1 receptor, through which it controls inflammation. We review here a body of literature that shows important mechanisms of action of fluvoxamine and other SSRIs that could play a role in COVID-19 treatment. These effects include: reduction in platelet aggregation, decreased mast cell degranulation, interference with endolysosomal viral trafficking, regulation of inositol-requiring enzyme 1α-driven inflammation and increased melatonin levels, which collectively have a direct antiviral effect, regulate coagulopathy or mitigate cytokine storm, which are known hallmarks of severe COVID-19.</jats:p>",
"alternative-id": [
"10.3389/fphar.2021.652688"
],
"author": [
{
"affiliation": [],
"family": "Sukhatme",
"given": "Vikas P.",
"sequence": "first"
},
{
"affiliation": [],
"family": "Reiersen",
"given": "Angela M.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Vayttaden",
"given": "Sharat J.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Sukhatme",
"given": "Vidula V.",
"sequence": "additional"
}
],
"container-title": "Frontiers in Pharmacology",
"container-title-short": "Front. Pharmacol.",
"content-domain": {
"crossmark-restriction": true,
"domain": [
"frontiersin.org"
]
},
"created": {
"date-parts": [
[
2021,
4,
20
]
],
"date-time": "2021-04-20T11:25:33Z",
"timestamp": 1618917933000
},
"deposited": {
"date-parts": [
[
2022,
12,
24
]
],
"date-time": "2022-12-24T20:39:11Z",
"timestamp": 1671914351000
},
"indexed": {
"date-parts": [
[
2024,
5,
1
]
],
"date-time": "2024-05-01T19:33:46Z",
"timestamp": 1714592026659
},
"is-referenced-by-count": 131,
"issued": {
"date-parts": [
[
2021,
4,
20
]
]
},
"license": [
{
"URL": "https://creativecommons.org/licenses/by/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
4,
20
]
],
"date-time": "2021-04-20T00:00:00Z",
"timestamp": 1618876800000
}
}
],
"link": [
{
"URL": "https://www.frontiersin.org/articles/10.3389/fphar.2021.652688/full",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "1965",
"original-title": [],
"prefix": "10.3389",
"published": {
"date-parts": [
[
2021,
4,
20
]
]
},
"published-online": {
"date-parts": [
[
2021,
4,
20
]
]
},
"publisher": "Frontiers Media SA",
"reference": [
{
"DOI": "10.1056/nejmoa2015432",
"article-title": "Pulmonary vascular endothelialitis, thrombosis, and angiogenesis in covid-19",
"author": "Ackermann",
"doi-asserted-by": "publisher",
"first-page": "120",
"journal-title": "N. Engl. J. Med.",
"key": "B1",
"volume": "383",
"year": "2020"
},
{
"DOI": "10.3390/v11060486",
"article-title": "Emergence of fluoxetine-resistant variants during treatment of human pancreatic cell cultures persistently infected with coxsackievirus B4",
"author": "Alidjinou",
"doi-asserted-by": "publisher",
"first-page": "486",
"journal-title": "Viruses",
"key": "B2",
"volume": "11",
"year": "2019"
},
{
"DOI": "10.1182/blood.2020008762",
"article-title": "Antibody-induced procoagulant platelets in severe COVID-19 infection",
"author": "Althaus",
"doi-asserted-by": "publisher",
"first-page": "1061",
"journal-title": "Blood",
"key": "B3",
"volume": "137",
"year": "2020"
},
{
"DOI": "10.1016/j.jocn.2020.07.007",
"article-title": "How SARS-Cov-2 can involve the central nervous system. A systematic analysis of literature of the department of human neurosciences of Sapienza University, Italy",
"author": "Armocida",
"doi-asserted-by": "publisher",
"first-page": "231",
"journal-title": "J. Clin. Neurosci.",
"key": "B4",
"volume": "79",
"year": "2020"
},
{
"DOI": "10.1159/000339109",
"article-title": "Immunomodulatory effect of sertraline in a rat model of rheumatoid arthritis",
"author": "Baharav",
"doi-asserted-by": "publisher",
"first-page": "309",
"journal-title": "Neuroimmunomodulation",
"key": "B5",
"volume": "19",
"year": "2012"
},
{
"DOI": "10.1016/j.pediatrneurol.2015.03.013",
"article-title": "Virus and immune-mediated encephalitides: epidemiology, diagnosis, treatment, and prevention",
"author": "Bale",
"doi-asserted-by": "publisher",
"first-page": "3",
"journal-title": "Pediatr. Neurol.",
"key": "B6",
"volume": "53",
"year": "2015"
},
{
"DOI": "10.1021/acsinfecdis.9b00179",
"article-title": "Fluoxetine inhibits enterovirus replication by targeting the viral 2C protein in a stereospecific manner",
"author": "Bauer",
"doi-asserted-by": "publisher",
"first-page": "1609",
"journal-title": "ACS Infect. Dis.",
"key": "B7",
"volume": "5",
"year": "2019"
},
{
"DOI": "10.1146/annurev.med.60.042307.110802",
"article-title": "The expanded biology of serotonin",
"author": "Berger",
"doi-asserted-by": "publisher",
"first-page": "355",
"journal-title": "Annu. Rev. Med.",
"key": "B8",
"volume": "60",
"year": "2009"
},
{
"DOI": "10.1371/journal.pbio.0040423",
"article-title": "Autophagy counterbalances endoplasmic reticulum expansion during the unfolded protein response",
"author": "Bernales",
"doi-asserted-by": "publisher",
"first-page": "e423",
"journal-title": "Plos Biol.",
"key": "B9",
"volume": "4",
"year": "2006"
},
{
"DOI": "10.1515/hsz-2019-0270",
"article-title": "Emerging mechanisms of drug-induced phospholipidosis",
"author": "Breiden",
"doi-asserted-by": "publisher",
"first-page": "31",
"journal-title": "Biol. Chem.",
"key": "B10",
"volume": "401",
"year": "2019"
},
{
"DOI": "10.1172/jci33374",
"article-title": "Interactions between integrin αIIbβ3 and the serotonin transporter regulate serotonin transport and platelet aggregation in mice and humans",
"author": "Carneiro",
"doi-asserted-by": "publisher",
"first-page": "1544",
"journal-title": "J. Clin. Invest.",
"key": "B11",
"volume": "118",
"year": "2008"
},
{
"DOI": "10.1016/j.xcrm.2020.100142",
"article-title": "Pharmacological inhibition of acid sphingomyelinase prevents uptake of SARS-CoV-2 by epithelial cells",
"author": "Carpinteiro",
"doi-asserted-by": "publisher",
"first-page": "100142",
"journal-title": "Cel Rep. Med.",
"key": "B12",
"volume": "1",
"year": "2020"
},
{
"DOI": "10.1128/mbio.02325-17",
"article-title": "Role of severe acute respiratory syndrome coronavirus viroporins E, 3a, and 8a in replication and pathogenesis",
"author": "Castano-Rodriguez",
"doi-asserted-by": "publisher",
"first-page": "e02325",
"journal-title": "mBio",
"key": "B13",
"volume": "9",
"year": "2018"
},
{
"DOI": "10.1016/0165-0327(92)90082-h",
"article-title": "Effects of acute and chronic treatment with fluvoxamine on extracellular and platelet serotonin in the blood of major depressive patients. Relationship to clinical improvement",
"author": "Celada",
"doi-asserted-by": "publisher",
"first-page": "243",
"journal-title": "J. Affective Disord.",
"key": "B14",
"volume": "25",
"year": "1992"
},
{
"DOI": "10.1128/jvi.00659-06",
"article-title": "Modulation of the unfolded protein response by the severe acute respiratory syndrome coronavirus spike protein",
"author": "Chan",
"doi-asserted-by": "publisher",
"first-page": "9279",
"journal-title": "Jvi",
"key": "B15",
"volume": "80",
"year": "2006"
},
{
"DOI": "10.1172/jci137244",
"article-title": "Clinical and immunological features of severe and moderate coronavirus disease 2019",
"author": "Chen",
"doi-asserted-by": "publisher",
"first-page": "2620",
"journal-title": "J. Clin. Invest.",
"key": "B16",
"volume": "130",
"year": ""
},
{
"DOI": "10.1016/s0140-6736(20)30211-7",
"article-title": "Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study",
"author": "Chen",
"doi-asserted-by": "publisher",
"first-page": "507",
"journal-title": "The Lancet",
"key": "B17",
"volume": "395",
"year": ""
},
{
"DOI": "10.3748/wjg.14.6993",
"article-title": "Effects of fluoxetine on mast cell morphology and protease-1 expression in gastric antrum in a rat model of depression",
"author": "Chen",
"doi-asserted-by": "publisher",
"first-page": "6993",
"journal-title": "Wjg",
"key": "B18",
"volume": "14",
"year": "2008"
},
{
"DOI": "10.1128/jvi.00245-13",
"article-title": "The nonstructural protein 2C of a Picorna-like virus displays nucleic acid helix destabilizing activity that can be functionally separated from its ATPase activity",
"author": "Cheng",
"doi-asserted-by": "publisher",
"first-page": "5205",
"journal-title": "J. Virol.",
"key": "B19",
"volume": "87",
"year": "2013"
},
{
"DOI": "10.1074/jbc.ra118.001897",
"article-title": "Middle East respiratory syndrome coronavirus and bat coronavirus HKU9 both can utilize GRP78 for attachment onto host cells",
"author": "Chu",
"doi-asserted-by": "publisher",
"first-page": "11709",
"journal-title": "J. Biol. Chem.",
"key": "B20",
"volume": "293",
"year": "2018"
},
{
"DOI": "10.1073/pnas.1720553115",
"article-title": "Platelets release pathogenic serotonin and return to circulation after immune complex-mediated sequestration",
"author": "Cloutier",
"doi-asserted-by": "publisher",
"first-page": "E1550",
"journal-title": "Proc. Natl. Acad. Sci. USA",
"key": "B21",
"volume": "115",
"year": "2018"
},
{
"DOI": "10.4161/auto.7.11.16642",
"article-title": "Coronavirus nsp6 proteins generate autophagosomes from the endoplasmic reticulum via an omegasome intermediate",
"author": "Cottam",
"doi-asserted-by": "publisher",
"first-page": "1335",
"journal-title": "Autophagy",
"key": "B22",
"volume": "7",
"year": "2011"
},
{
"DOI": "10.1371/journal.ppat.1002315",
"article-title": "Severe acute respiratory syndrome coronavirus envelope protein regulates cell stress response and apoptosis",
"author": "DeDiego",
"doi-asserted-by": "publisher",
"first-page": "e1002315",
"journal-title": "Plos Pathog.",
"key": "B23",
"volume": "7",
"year": "2011"
},
{
"DOI": "10.1517/14656566.6.15.2727",
"article-title": "Fluvoxamine: a selective serotonin re-uptake inhibitor for the treatment of obsessive-compulsive disorder",
"author": "Dell'Osso",
"doi-asserted-by": "publisher",
"first-page": "2727",
"journal-title": "Expert Opin. Pharmacother.",
"key": "B24",
"volume": "6",
"year": "2005"
},
{
"first-page": "2020",
"key": "B25",
"volume-title": "Fluvoxamine",
"year": "2005"
},
{
"DOI": "10.1128/jvi.00404-18",
"article-title": "Axonal transport enables neuron-to-neuron propagation of human coronavirus OC43",
"author": "Dube",
"doi-asserted-by": "publisher",
"first-page": "e00404",
"journal-title": "J. Virol.",
"key": "B26",
"volume": "92",
"year": "2018"
},
{
"DOI": "10.1182/blood-2012-06-437392",
"article-title": "Platelet serotonin promotes the recruitment of neutrophils to sites of acute inflammation in mice",
"author": "Duerschmied",
"doi-asserted-by": "publisher",
"first-page": "1008",
"journal-title": "Blood",
"key": "B27",
"volume": "121",
"year": "2013"
},
{
"DOI": "10.1056/nejmra2026131",
"article-title": "Cytokine storm",
"author": "Fajgenbaum",
"doi-asserted-by": "publisher",
"first-page": "2255",
"journal-title": "N. Engl. J. Med.",
"key": "B28",
"volume": "383",
"year": "2020"
},
{
"key": "B29",
"volume-title": "Fluvoxamine maleate tablets label",
"year": "2012"
},
{
"key": "B30",
"volume-title": "EUA for bamlanivimab for the treatment of mild to moderate COVID-19",
"year": ""
},
{
"key": "B31",
"volume-title": "EUA for casirivimab and imdevimab for the treatment of mild to moderate COVID-19",
"year": ""
},
{
"DOI": "10.1007/bf02265168",
"article-title": "Changes in histamine and serotonin secretion from rat peritoneal mast cells caused by antidepressants",
"author": "Ferjan",
"doi-asserted-by": "publisher",
"first-page": "141",
"journal-title": "Inflamm. Res.",
"key": "B32",
"volume": "45",
"year": "1996"
},
{
"DOI": "10.1128/jvi.03557-12",
"article-title": "Sigma-1 receptor regulates early steps of viral RNA replication at the onset of hepatitis C virus infection",
"author": "Friesland",
"doi-asserted-by": "publisher",
"first-page": "6377",
"journal-title": "J. Virol.",
"key": "B33",
"volume": "87",
"year": "2013"
},
{
"DOI": "10.1159/000343895",
"article-title": "Expression and functional characterization of retinoic acid-inducible gene-I-like receptors of mast cells in response to viral infection",
"author": "Fukuda",
"doi-asserted-by": "publisher",
"first-page": "163",
"journal-title": "J. Innate Immun.",
"key": "B34",
"volume": "5",
"year": "2013"
},
{
"DOI": "10.3390/v8070184",
"article-title": "Regulation of stress responses and translational control by coronavirus",
"author": "Fung",
"doi-asserted-by": "publisher",
"first-page": "184",
"journal-title": "Viruses",
"key": "B35",
"volume": "8",
"year": "2016"
},
{
"DOI": "10.1016/j.virol.2019.05.002",
"article-title": "The ER stress sensor IRE1 and MAP kinase ERK modulate autophagy induction in cells infected with coronavirus infectious bronchitis virus",
"author": "Fung",
"doi-asserted-by": "publisher",
"first-page": "34",
"journal-title": "Virology",
"key": "B36",
"volume": "533",
"year": "2019"
},
{
"DOI": "10.1096/fj.15-273656",
"article-title": "Disruption of the NF-κB/NLRP3 connection by melatonin requires retinoid-related orphan receptor-α and blocks the septic response in mice",
"author": "García",
"doi-asserted-by": "publisher",
"first-page": "3863",
"journal-title": "FASEB J.",
"key": "B37",
"volume": "29",
"year": "2015"
},
{
"DOI": "10.1111/j.1440-1819.2011.02241.x",
"article-title": "Coagulation activation and fibrinolysis impairment are reduced in patients with anxiety and depression when medicated with serotonergic antidepressants",
"author": "Geiser",
"doi-asserted-by": "publisher",
"first-page": "518",
"journal-title": "Psychiatry Clin. Neurosci.",
"key": "B38",
"volume": "65",
"year": "2011"
},
{
"DOI": "10.1016/j.intimp.2005.12.005",
"article-title": "Cocaine-induced HIV-1 expression in microglia involves sigma-1 receptors and transforming growth factor-β1",
"author": "Gekker",
"doi-asserted-by": "publisher",
"first-page": "1029",
"journal-title": "Int. Immunopharmacology",
"key": "B39",
"volume": "6",
"year": "2006"
},
{
"DOI": "10.1038/s41598-017-04968-z",
"article-title": "Fluvoxamine stimulates oligodendrogenesis of cultured neural stem cells and attenuates inflammation and demyelination in an animal model of multiple sclerosis",
"author": "Ghareghani",
"doi-asserted-by": "publisher",
"first-page": "4923",
"journal-title": "Sci. Rep.",
"key": "B40",
"volume": "7",
"year": "2017"
},
{
"DOI": "10.1016/j.cell.2020.10.039",
"article-title": "β-Coronaviruses use lysosomes for egress instead of the biosynthetic secretory pathway",
"author": "Ghosh",
"doi-asserted-by": "publisher",
"first-page": "1520",
"journal-title": "Cell",
"key": "B41",
"volume": "183",
"year": "2020"
},
{
"DOI": "10.1126/science.abe9403",
"article-title": "Comparative host-coronavirus protein interaction networks reveal pan-viral disease mechanisms",
"author": "Gordon",
"doi-asserted-by": "publisher",
"first-page": "eabe9403",
"journal-title": "JScience",
"key": "B42",
"volume": "370",
"year": "2020"
},
{
"DOI": "10.1038/s41598-019-39726-w",
"article-title": "Neutrophil dysfunction in the airways of children with acute respiratory failure due to lower respiratory tract viral and bacterial coinfections",
"author": "Grunwell",
"doi-asserted-by": "publisher",
"first-page": "2874",
"journal-title": "Sci. Rep.",
"key": "B43",
"volume": "9",
"year": "2019"
},
{
"DOI": "10.1038/nm.3214",
"article-title": "Acid sphingomyelinase-ceramide system mediates effects of antidepressant drugs",
"author": "Gulbins",
"doi-asserted-by": "publisher",
"first-page": "934",
"journal-title": "Nat. Med.",
"key": "B44",
"volume": "19",
"year": "2013"
},
{
"DOI": "10.1016/j.jinf.2020.06.017",
"article-title": "The stress-inducible molecular chaperone GRP78 as potential therapeutic target for coronavirus infection",
"author": "Ha",
"doi-asserted-by": "publisher",
"first-page": "452",
"journal-title": "J. Infect.",
"key": "B45",
"volume": "81",
"year": "2020"
},
{
"DOI": "10.1124/dmd.107.015131",
"article-title": "Saturable uptake of lipophilic amine drugs into isolated hepatocytes: mechanisms and consequences for quantitative clearance prediction",
"author": "Hallifax",
"doi-asserted-by": "publisher",
"first-page": "1325",
"journal-title": "Drug Metab. Dispos",
"key": "B46",
"volume": "35",
"year": "2007"
},
{
"DOI": "10.1073/pnas.93.15.8072",
"article-title": "Purification, molecular cloning, and expression of the mammalian sigma1-binding site",
"author": "Hanner",
"doi-asserted-by": "publisher",
"first-page": "8072",
"journal-title": "Proc. Natl. Acad. Sci.",
"key": "B47",
"volume": "93",
"year": "1996"
},
{
"DOI": "10.1097/00004714-200104000-00008",
"article-title": "Differential effects of fluvoxamine and other antidepressants on the biotransformation of melatonin",
"author": "Härtter",
"doi-asserted-by": "publisher",
"first-page": "167",
"journal-title": "J. Clin. Psychopharmacol.",
"key": "B48",
"volume": "21",
"year": "2001"
},
{
"DOI": "10.1016/j.jphs.2014.11.010",
"article-title": "Activation of sigma-1 receptor chaperone in the treatment of neuropsychiatric diseases and its clinical implication",
"author": "Hashimoto",
"doi-asserted-by": "publisher",
"first-page": "6",
"journal-title": "J. Pharmacol. Sci.",
"key": "B49",
"volume": "127",
"year": "2015"
},
{
"DOI": "10.1162/2e3983f5.c37587dd",
"article-title": "Repurposing of CNS drugs to treat COVID-19 infection: targeting the sigma-1 receptor",
"author": "Hashimoto",
"doi-asserted-by": "publisher",
"first-page": "249",
"journal-title": "Eur. Arch. Psychiatry Clin. Neurosci.",
"key": "B50",
"volume": "271",
"year": "2021"
},
{
"DOI": "10.1016/j.cell.2007.08.036",
"article-title": "Sigma-1 receptor chaperones at the ER- mitochondrion interface regulate Ca(2+) signaling and cell survival",
"author": "Hayashi",
"doi-asserted-by": "publisher",
"first-page": "596",
"journal-title": "Cell",
"key": "B51",
"volume": "131",
"year": "2007"
},
{
"DOI": "10.1056/nejmc2008597",
"article-title": "Neurologic features in severe SARS-CoV-2 infection",
"author": "Helms",
"doi-asserted-by": "publisher",
"first-page": "2268",
"journal-title": "N. Engl. J. Med.",
"key": "B52",
"volume": "382",
"year": ""
},
{
"DOI": "10.1186/s13054-020-03200-1",
"article-title": "Delirium and encephalopathy in severe COVID-19: a cohort analysis of ICU patients",
"author": "Helms",
"doi-asserted-by": "publisher",
"first-page": "491",
"journal-title": "Crit. Care",
"key": "B53",
"volume": "24",
"year": ""
},
{
"DOI": "10.7554/elife.51804",
"article-title": "Repression of viral gene expression and replication by the unfolded protein response effector XBP1u",
"author": "Hinte",
"doi-asserted-by": "publisher",
"first-page": "e51804",
"journal-title": "Elife",
"key": "B54",
"volume": "9",
"year": "2020"
},
{
"DOI": "10.1038/s41380-021-01021-4",
"article-title": "Association between antidepressant use and reduced risk of intubation or death in hospitalized patients with COVID-19: results from an observational study",
"author": "Hoertel",
"doi-asserted-by": "crossref",
"key": "B55",
"volume-title": "Mol. Psychiat.",
"year": "2021"
},
{
"DOI": "10.1016/j.ijantimicag.2020.106044",
"article-title": "Widely available lysosome targeting agents should be considered as potential therapy for COVID-19",
"author": "Homolak",
"doi-asserted-by": "publisher",
"first-page": "106044",
"journal-title": "Int. J. Antimicrob. Agents",
"key": "B56",
"volume": "56",
"year": "2020"
},
{
"DOI": "10.1016/s0140-6736(20)30183-5",
"article-title": "Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China",
"author": "Huang",
"doi-asserted-by": "publisher",
"first-page": "497",
"journal-title": "The Lancet",
"key": "B57",
"volume": "395",
"year": "2020"
},
{
"article-title": "Fluvoxamine in the treatment of anxiety disorders",
"author": "Irons",
"first-page": "289",
"journal-title": "Neuropsychiatr. Dis. Treat.",
"key": "B58",
"volume": "1",
"year": "2005"
},
{
"DOI": "10.1016/j.ejphar.2014.01.064",
"article-title": "Interaction of new antidepressants with sigma-1 receptor chaperones and their potentiation of neurite outgrowth in PC12 cells",
"author": "Ishima",
"doi-asserted-by": "publisher",
"first-page": "167",
"journal-title": "Eur. J. Pharmacol.",
"key": "B59",
"volume": "727",
"year": "2014"
},
{
"DOI": "10.1017/s146114570000198x",
"article-title": "Reduction of platelet serotonin content in depressed patients treated with either paroxetine or desipramine",
"author": "Javors",
"doi-asserted-by": "publisher",
"first-page": "229",
"journal-title": "Int. J. Neuropsychopharm.",
"key": "B60",
"volume": "3",
"year": "2000"
},
{
"DOI": "10.1093/bjaceaccp/mks060",
"article-title": "Non-respiratory functions of the lung",
"author": "Joseph",
"doi-asserted-by": "publisher",
"first-page": "98",
"journal-title": "Continuing Educ. Anaesth. Crit. Care Pain",
"key": "B61",
"volume": "13",
"year": "2013"
},
{
"DOI": "10.1016/j.pharmthera.2016.04.001",
"article-title": "Antidepressant therapies inhibit inflammation and microglial M1-polarization",
"author": "Kalkman",
"doi-asserted-by": "publisher",
"first-page": "82",
"journal-title": "Pharmacol. Ther.",
"key": "B62",
"volume": "163",
"year": "2016"
},
{
"DOI": "10.1124/dmd.112.050054",
"article-title": "Lysosomal sequestration (trapping) of lipophilic amine (cationic amphiphilic) drugs in immortalized human hepatocytes (Fa2N-4 cells)",
"author": "Kazmi",
"doi-asserted-by": "publisher",
"first-page": "897",
"journal-title": "Drug Metab. Dispos",
"key": "B63",
"volume": "41",
"year": "2013"
},
{
"DOI": "10.1038/nrd2755",
"article-title": "Cell death and endoplasmic reticulum stress: disease relevance and therapeutic opportunities",
"author": "Kim",
"doi-asserted-by": "publisher",
"first-page": "1013",
"journal-title": "Nat. Rev. Drug Discov.",
"key": "B64",
"volume": "7",
"year": "2008"
},
{
"DOI": "10.1021/jm070524a",
"article-title": "Identification of new functional inhibitors of acid sphingomyelinase using a Structure−Property−Activity relation model",
"author": "Kornhuber",
"doi-asserted-by": "publisher",
"first-page": "219",
"journal-title": "J. Med. Chem.",
"key": "B65",
"volume": "51",
"year": "2008"
},
{
"DOI": "10.1038/s41598-017-18909-3",
"article-title": "Dengue virus-induced ER stress is required for autophagy activation, viral replication, and pathogenesis both in vitro and in vivo",
"author": "Lee",
"doi-asserted-by": "publisher",
"first-page": "489",
"journal-title": "Sci. Rep.",
"key": "B66",
"volume": "8",
"year": "2018"
},
{
"author": "Lenze",
"key": "B67",
"volume-title": "Fluvoxamine for early treatment of covid-19 (stop covid 2)",
"year": "2020"
},
{
"DOI": "10.1001/jama.2020.22760",
"article-title": "Fluvoxamine vs placebo and clinical deterioration in outpatients with symptomatic COVID-19",
"author": "Lenze",
"doi-asserted-by": "publisher",
"first-page": "2292",
"journal-title": "JAMA",
"key": "B68",
"volume": "324",
"year": "2020"
},
{
"DOI": "10.1177/070674379604100919",
"article-title": "Fluvoxamine-associated bleeding",
"author": "Leung",
"doi-asserted-by": "publisher",
"first-page": "604",
"journal-title": "Can. J. Psychiatry",
"key": "B69",
"volume": "41",
"year": "1996"
},
{
"DOI": "10.1073/pnas.0605402103",
"article-title": "Severe acute respiratory syndrome-associated coronavirus 3a protein forms an ion channel and modulates virus release",
"author": "Lu",
"doi-asserted-by": "publisher",
"first-page": "12540",
"journal-title": "Proc. Natl. Acad. Sci.",
"key": "B70",
"volume": "103",
"year": "2006"
},
{
"DOI": "10.1016/j.antiviral.2020.104781",
"article-title": "Synthesis and antiviral effect of novel fluoxetine analogues as enterovirus 2C inhibitors",
"author": "Manganaro",
"doi-asserted-by": "publisher",
"first-page": "104781",
"journal-title": "Antiviral Res.",
"key": "B71",
"volume": "178",
"year": "2020"
},
{
"article-title": "The effects of morphine- and nalorphine- like drugs in the nondependent and morphine-dependent chronic spinal dog",
"author": "Martin",
"first-page": "517",
"journal-title": "J. Pharmacol. Exp. Ther.",
"key": "B72",
"volume": "197",
"year": "1976"
},
{
"DOI": "10.1038/ni.1857",
"article-title": "TLR activation of the transcription factor XBP1 regulates innate immune responses in macrophages",
"author": "Martinon",
"doi-asserted-by": "publisher",
"first-page": "411",
"journal-title": "Nat. Immunol.",
"key": "B73",
"volume": "11",
"year": "2010"
},
{
"DOI": "10.1016/j.trsl.2007.10.004",
"article-title": "Selective serotonin reuptake inhibitors: measurement of effect on platelet function",
"author": "McCloskey",
"doi-asserted-by": "publisher",
"first-page": "168",
"journal-title": "Translational Res.",
"key": "B74",
"volume": "151",
"year": "2008"
},
{
"DOI": "10.3389/fimmu.2020.574862",
"article-title": "Mast cells in alveolar septa of COVID-19 patients: a pathogenic pathway that may link interstitial edema to immunothrombosis",
"author": "Motta Junior",
"doi-asserted-by": "publisher",
"first-page": "574862",
"journal-title": "Front. Immunol.",
"key": "B75",
"volume": "11",
"year": "2020"
},
{
"DOI": "10.4103/abr.abr_141_18",
"article-title": "Evaluation of the effect of antidepressant drug, fluvoxamine, on cyclooxygenase-2 protein expression in lipopolysaccharide-stimulated macrophages",
"author": "Naji Esfahani",
"doi-asserted-by": "publisher",
"first-page": "5",
"journal-title": "Adv. Biomed. Res.",
"key": "B76",
"volume": "8",
"year": "2019"
},
{
"DOI": "10.1097/00004714-199802000-00011",
"article-title": "Serotonin transporter-blocking properties of nefazodone assessed by measurement of platelet serotonin",
"author": "Narayan",
"doi-asserted-by": "publisher",
"first-page": "67",
"journal-title": "J. Clin. Psychopharmacol.",
"key": "B77",
"volume": "18",
"year": "1998"
},
{
"DOI": "10.1016/0014-2999(96)00254-3",
"article-title": "Interactions of selective serotonin reuptake inhibitors with subtypes of σ receptors in rat brain",
"author": "Narita",
"doi-asserted-by": "publisher",
"first-page": "117",
"journal-title": "Eur. J. Pharmacol.",
"key": "B78",
"volume": "307",
"year": "1996"
},
{
"DOI": "10.1016/j.imbio.2016.07.001",
"article-title": "The role of macrophages in anti-inflammatory activity of antidepressant drugs",
"author": "Nazimek",
"doi-asserted-by": "publisher",
"first-page": "823",
"journal-title": "Immunobiology",
"key": "B79",
"volume": "222",
"year": "2017"
},
{
"DOI": "10.1111/jth.15283",
"article-title": "Platelet-activating immune complexes identified in critically ill COVID-19 patients suspected of heparin-induced thrombocytopenia",
"author": "Nazy",
"doi-asserted-by": "crossref",
"key": "B80",
"volume-title": "J. Thromb. Haemost.",
"year": "2021"
},
{
"key": "B81",
"volume-title": "NIH ACTIV trial of blood thinners pauses enrollment of critically ill COVID-19 patients",
"year": "2020"
},
{
"DOI": "10.1111/j.1440-1681.2006.04410.x",
"article-title": "5-hydroxytryptamine in the cardiovascular system: focus on the serotonin transporter (SERT)",
"author": "Ni",
"doi-asserted-by": "publisher",
"first-page": "575",
"journal-title": "Clin. Exp. Pharmacol. Physiol.",
"key": "B82",
"volume": "33",
"year": "2006"
},
{
"DOI": "10.1371/journal.pone.0002558",
"article-title": "Potentiation of nerve growth factor-induced neurite outgrowth by fluvoxamine: role of sigma-1 receptors, IP3 receptors and cellular signaling pathways",
"author": "Nishimura",
"doi-asserted-by": "publisher",
"first-page": "e2558",
"journal-title": "PLoS One",
"key": "B83",
"volume": "3",
"year": "2008"
},
{
"DOI": "10.1128/mcb.01453-06",
"article-title": "Autophagy is activated for cell survival after endoplasmic ReticulumStress",
"author": "Ogata",
"doi-asserted-by": "publisher",
"first-page": "9220",
"journal-title": "Mcb",
"key": "B84",
"volume": "26",
"year": "2006"
},
{
"DOI": "10.1128/jvi.78.18.9977-9986.2004",
"article-title": "Identification and characterization of severe acute respiratory syndrome coronavirus replicase proteins",
"author": "Prentice",
"doi-asserted-by": "publisher",
"first-page": "9977",
"journal-title": "Jvi",
"key": "B85",
"volume": "78",
"year": "2004"
},
{
"DOI": "10.1038/emboj.2013.183",
"article-title": "Toll-like receptor-mediated IRE1α activation as a therapeutic target for inflammatory arthritis",
"author": "Qiu",
"doi-asserted-by": "publisher",
"first-page": "2477",
"journal-title": "EMBO J.",
"key": "B86",
"volume": "32",
"year": "2013"
},
{
"DOI": "10.22038/ijbms.2016.7598",
"article-title": "Fluvoxamine inhibits some inflammatory genes expression in LPS/stimulated human endothelial cells, U937 macrophages, and carrageenan-induced paw edema in rat",
"author": "Rafiee",
"doi-asserted-by": "publisher",
"first-page": "977",
"journal-title": "Iran J. Basic Med. Sci.",
"key": "B87",
"volume": "19",
"year": "2016"
},
{
"DOI": "10.1101/2020.10.15.20213546",
"article-title": "Melatonin is significantly associated with survival of intubated COVID-19 patients",
"author": "Ramlall",
"doi-asserted-by": "crossref",
"key": "B88",
"volume-title": "medRxiv",
"year": "2020"
},
{
"DOI": "10.1038/s41375-020-0887-9",
"article-title": "SARS-CoV-2 infection and overactivation of Nlrp3 inflammasome as a trigger of cytokine \"storm\" and risk factor for damage of hematopoietic stem cells",
"author": "Ratajczak",
"doi-asserted-by": "publisher",
"first-page": "1726",
"journal-title": "Leukemia",
"key": "B89",
"volume": "34",
"year": "2020"
},
{
"DOI": "10.1126/scitranslmed.aau5266",
"article-title": "Modulation of the sigma-1 receptor-IRE1 pathway is beneficial in preclinical models of inflammation and sepsis",
"author": "Rosen",
"doi-asserted-by": "publisher",
"first-page": "eaau5266",
"journal-title": "Sci. Transl Med.",
"key": "B90",
"volume": "11",
"year": "2019"
},
{
"DOI": "10.1080/22221751.2020.1829082",
"article-title": "Targeting the endolysosomal host-SARS-CoV-2 interface by clinically licensed functional inhibitors of acid sphingomyelinase (FIASMA) including the antidepressant fluoxetine",
"author": "Schloer",
"doi-asserted-by": "publisher",
"first-page": "2245",
"journal-title": "Emerging Microbes & Infections",
"key": "B91",
"volume": "9",
"year": "2020"
},
{
"DOI": "10.1093/ofid/ofab050",
"article-title": "Prospective cohort of fluvoxamine for early treatment of COVID-19",
"author": "Seftel",
"doi-asserted-by": "publisher",
"first-page": "ofab050",
"journal-title": "Open Forum Infect. Dis.",
"key": "B92",
"volume": "8",
"year": "2021"
},
{
"article-title": "A call to action: immediate deployment of select repurposed drugs for COVID-19 outpatient treatment",
"author": "Sukhatme",
"first-page": "2021",
"journal-title": "February",
"key": "B93",
"volume": "12",
"year": "2021"
},
{
"DOI": "10.4049/jimmunol.172.7.4391",
"article-title": "Signaling through Toll-like receptors triggers HIV-1 replication in latently infected mast cells",
"author": "Sundstrom",
"doi-asserted-by": "publisher",
"first-page": "4391",
"journal-title": "J. Immunol.",
"key": "B94",
"volume": "172",
"year": "2004"
},
{
"DOI": "10.1371/journal.pone.0106533",
"article-title": "Psychedelic N,N-dimethyltryptamine and 5-methoxy-N,N-dimethyltryptamine modulate innate and adaptive inflammatory responses through the sigma-1 receptor of human monocyte-derived dendritic cells",
"author": "Szabo",
"doi-asserted-by": "publisher",
"first-page": "e106533",
"journal-title": "PLoS One",
"key": "B95",
"volume": "9",
"year": "2014"
},
{
"DOI": "10.1159/000321634",
"article-title": "The immunomodulatory effect of the antidepressant sertraline in an experimental autoimmune encephalomyelitis mouse model of multiple sclerosis",
"author": "Taler",
"doi-asserted-by": "publisher",
"first-page": "117",
"journal-title": "Neuroimmunomodulation",
"key": "B96",
"volume": "18",
"year": "2011"
},
{
"DOI": "10.1016/j.euroneuro.2007.03.010",
"article-title": "Immunomodulatory effect of selective serotonin reuptake inhibitors (SSRIs) on human T lymphocyte function and gene expression",
"author": "Taler",
"doi-asserted-by": "publisher",
"first-page": "774",
"journal-title": "Eur. Neuropsychopharmacol.",
"key": "B97",
"volume": "17",
"year": "2007"
},
{
"DOI": "10.1038/s41577-020-0311-8",
"article-title": "The trinity of COVID-19: immunity, inflammation and intervention",
"author": "Tay",
"doi-asserted-by": "publisher",
"first-page": "363",
"journal-title": "Nat. Rev. Immunol.",
"key": "B98",
"volume": "20",
"year": "2020"
},
{
"DOI": "10.1002/biof.1633",
"article-title": "COVID ‐19, pulmonary mast cells, cytokine storms, and beneficial actions of luteolin",
"author": "Theoharides",
"doi-asserted-by": "publisher",
"first-page": "306",
"journal-title": "Biofactors",
"key": "B99",
"volume": "46",
"year": "2020"
},
{
"DOI": "10.1038/216335a0",
"article-title": "5-hydroxytryptamine in the circulation of the dog",
"author": "Thomas",
"doi-asserted-by": "publisher",
"first-page": "335",
"journal-title": "Nature",
"key": "B100",
"volume": "216",
"year": "1967"
},
{
"DOI": "10.1016/j.bbi.2011.12.011",
"article-title": "A comparative examination of the anti-inflammatory effects of SSRI and SNRI antidepressants on LPS stimulated microglia",
"author": "Tynan",
"doi-asserted-by": "publisher",
"first-page": "469",
"journal-title": "Brain Behav. Immun.",
"key": "B101",
"volume": "26",
"year": "2012"
},
{
"DOI": "10.1128/aac.02084-12",
"article-title": "Selective serotonin reuptake inhibitor fluoxetine inhibits replication of human enteroviruses B and D by targeting viral protein 2C",
"author": "Ulferts",
"doi-asserted-by": "publisher",
"first-page": "1952",
"journal-title": "Antimicrob. Agents Chemother.",
"key": "B102",
"volume": "57",
"year": "2013"
},
{
"DOI": "10.3389/fimmu.2020.01580",
"article-title": "Severe COVID-19: NLRP3 inflammasome dysregulated",
"author": "van den Berg",
"doi-asserted-by": "publisher",
"first-page": "1580",
"journal-title": "Front. Immunol.",
"key": "B103",
"volume": "11",
"year": "2020"
},
{
"DOI": "10.1097/00005344-199100175-00003",
"article-title": "Platelet-derived serotonin, the endothelium, and cardiovascular disease",
"author": "Vanhoutte",
"doi-asserted-by": "publisher",
"first-page": "S13",
"journal-title": "J. Cardiovasc. Pharmacol.",
"key": "B104",
"volume": "17",
"year": "1991"
},
{
"DOI": "10.3389/fphar.2020.582310",
"article-title": "Repurposing sigma-1 receptor ligands for COVID-19 therapy?",
"author": "Vela",
"doi-asserted-by": "publisher",
"first-page": "582310",
"journal-title": "Front. Pharmacol.",
"key": "B105",
"volume": "11",
"year": "2020"
},
{
"DOI": "10.1128/jvi.01033-07",
"article-title": "The coronavirus spike protein induces endoplasmic reticulum stress and upregulation of intracellular chemokine mRNA concentrations",
"author": "Versteeg",
"doi-asserted-by": "publisher",
"first-page": "10981",
"journal-title": "J. Virol.",
"key": "B106",
"volume": "81",
"year": "2007"
},
{
"DOI": "10.4049/jimmunol.1101703",
"article-title": "Skin mast cells protect mice against vaccinia virus by triggering mast cell receptor S1PR2 and releasing antimicrobial peptides",
"author": "Wang",
"doi-asserted-by": "publisher",
"first-page": "345",
"journal-title": "J.I.",
"key": "B107",
"volume": "188",
"year": "2012"
},
{
"DOI": "10.1093/nar/gkx1037",
"article-title": "DrugBank 5.0: a major update to the DrugBank database for 2018",
"author": "Wishart",
"doi-asserted-by": "publisher",
"first-page": "D1074",
"journal-title": "Nucleic Acids Res.",
"key": "B108",
"volume": "46",
"year": "2018"
},
{
"DOI": "10.1128/jvi.00431-18",
"article-title": "The PERK arm of the unfolded protein response negatively regulates transmissible gastroenteritis virus replication by suppressing protein translation and promoting type I interferon production",
"author": "Xue",
"doi-asserted-by": "publisher",
"first-page": "e00431",
"journal-title": "J. Virol.",
"key": "B109",
"volume": "92",
"year": "2018"
},
{
"DOI": "10.7150/ijbs.45498",
"article-title": "Targeting the endocytic pathway and autophagy process as a novel therapeutic strategy in COVID-19",
"author": "Yang",
"doi-asserted-by": "publisher",
"first-page": "1724",
"journal-title": "Int. J. Biol. Sci.",
"key": "B110",
"volume": "16",
"year": "2020"
},
{
"DOI": "10.1016/j.jocn.2020.08.014",
"article-title": "Reissuing the sigma receptors for SARS-CoV-2",
"author": "Yesilkaya",
"doi-asserted-by": "publisher",
"first-page": "72",
"journal-title": "J. Clin. Neurosci.",
"key": "B111",
"volume": "80",
"year": "2020"
},
{
"DOI": "10.1038/s41419-018-0917-y",
"article-title": "SARS-Coronavirus Open Reading Frame-3a drives multimodal necrotic cell death",
"author": "Yue",
"doi-asserted-by": "publisher",
"first-page": "904",
"journal-title": "Cell Death Dis",
"key": "B112",
"volume": "9",
"year": "2018"
},
{
"DOI": "10.1182/bloodadvances.2020003513",
"article-title": "Platelet reactivity to thrombin differs between patients with COVID-19 and those with ARDS unrelated to COVID-19",
"author": "Zaid",
"doi-asserted-by": "publisher",
"first-page": "635",
"journal-title": "Blood Adv.",
"key": "B113",
"volume": "5",
"year": "2021"
},
{
"DOI": "10.1128/aac.00983-12",
"article-title": "Fluoxetine is a potent inhibitor of coxsackievirus replication",
"author": "Zuo",
"doi-asserted-by": "publisher",
"first-page": "4838",
"journal-title": "Antimicrob. Agents Chemother.",
"key": "B114",
"volume": "56",
"year": "2012"
}
],
"reference-count": 114,
"references-count": 114,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.frontiersin.org/articles/10.3389/fphar.2021.652688/full"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "Fluvoxamine: A Review of Its Mechanism of Action and Its Role in COVID-19",
"type": "journal-article",
"update-policy": "http://dx.doi.org/10.3389/crossmark-policy",
"volume": "12"
}