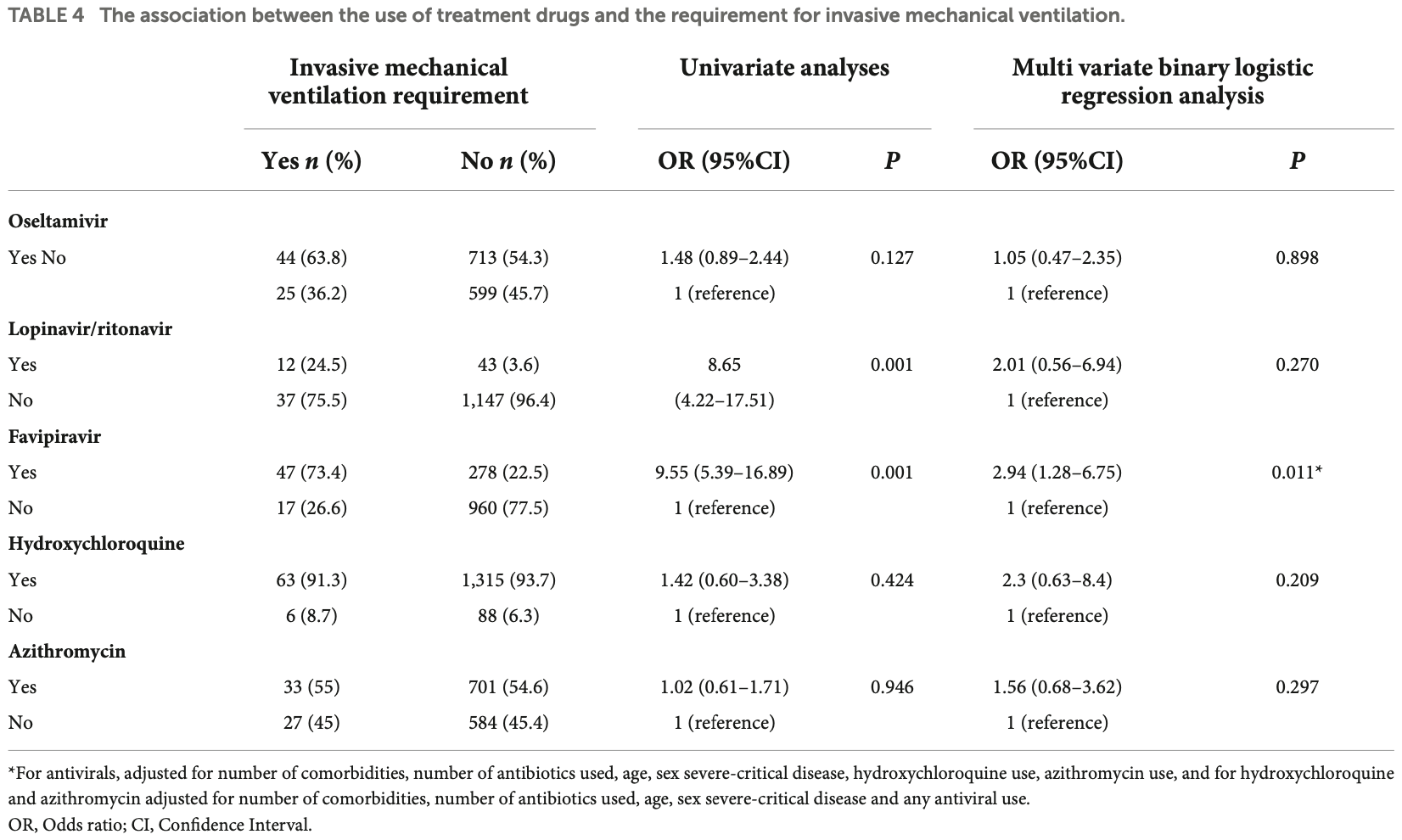
The association of antiviral drugs with COVID-19 morbidity: The retrospective analysis of a nationwide COVID-19 cohort
et al., Frontiers in Medicine, doi:10.3389/fmed.2022.894126, Aug 2022
Retrospective 1,472 hospitalized patients in Turkey, showing a higher ICU admission and ventilation with favipiravir. Results may be subject to confounding by indication.
Potential risks of favipiravir include kidney injury1-3, liver injury2-5, cardiovascular events5,6, pulmonary toxicity6,7, and mutagenicity, carcinogenicity, teratogenicity, embryotoxicity, and the creation of dangerous variants8-14.
This study is excluded in the after exclusion results of meta-analysis:
substantial unadjusted confounding by indication possible.
Study covers HCQ and favipiravir.
|
risk of mechanical ventilation, 184.4% higher, RR 2.84, p = 0.01, treatment 47 of 325 (14.5%), control 17 of 977 (1.7%), adjusted per study, odds ratio converted to relative risk, multivariable.
|
|
risk of ICU admission, 181.5% higher, RR 2.81, p = 0.001, treatment 75 of 325 (23.1%), control 35 of 969 (3.6%), adjusted per study, odds ratio converted to relative risk, multivariable.
|
|
hospitalization time, 100% higher, relative time 2.00, p = 0.001, treatment 265, control 746.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
1.
Abdulaziz et al., Clinical Features and Prognosis of Acute Kidney Injury in Hospital-Admitted Patients with COVID-19 in Egypt: A Single-Center Experience, Mansoura Medical Journal, doi:10.58775/2735-3990.1433.
2.
Ülger et al., Experimental evaluation of favipiravir (T-705)-induced liver and kidney toxicity in rats, Food and Chemical Toxicology, doi:10.1016/j.fct.2025.115472.
3.
El-Fetouh et al., Experimental Studies on Some Drugs Used in Covid-19 Treatment (Favipiravir and Dexamethasone) in Albino Rats, Journal of Advanced Veterinary Research, 13:10, www.advetresearch.com/index.php/AVR/article/view/1635.
4.
Almutairi et al., Liver Injury in Favipiravir-Treated COVID-19 Patients: Retrospective Single-Center Cohort Study, Tropical Medicine and Infectious Disease, doi:10.3390/tropicalmed8020129.
5.
Siby et al., Temporal Trends in Serious Adverse Events Associated with Oral Antivirals During the COVID-19 Pandemic: Insights from the FAERS Database (2020–2023), Open Forum Infectious Diseases, doi:10.1093/ofid/ofaf695.1825.
6.
Ozhan et al., Evaluation of the cardiopulmonary effects of repurposed COVID-19 therapeutics in healthy rats, Scientific Reports, doi:10.1038/s41598-025-31048-4.
7.
Ülger (B) et al., Evaluation of the effects of favipiravir (T-705) on the lung tissue of healty rats: An experimental study, Food and Chemical Toxicology, doi:10.1016/j.fct.2025.115235.
8.
Zhirnov et al., Favipiravir: the hidden threat of mutagenic action, Journal of microbiology, epidemiology and immunobiology, doi:10.36233/0372-9311-114.
9.
Waters et al., Human genetic risk of treatment with antiviral nucleoside analog drugs that induce lethal mutagenesis: the special case of molnupiravir, Environmental and Molecular Mutagenesis, doi:10.1002/em.22471.
10.
Hadj Hassine et al., Lethal Mutagenesis of RNA Viruses and Approved Drugs with Antiviral Mutagenic Activity, Viruses, doi:10.3390/v14040841.
11.
Shum, C., An investigational study into the drug-associated mutational signature in SARS-CoV-2 viruses, The University of Hong Kong, PhD Thesis, hub.hku.hk/handle/10722/344396.
12.
Shiraki et al., Convenient screening of the reproductive toxicity of favipiravir and antiviral drugs in Caenorhabditis elegans, Heliyon, doi:10.1016/j.heliyon.2024.e35331.
Babayigit et al., 31 Aug 2022, retrospective, Turkey, peer-reviewed, mean age 51.9, 68 authors, study period 11 March, 2020 - 18 July, 2020.
Contact: habayram@ku.edu.tr.
The association of antiviral drugs with COVID-19 morbidity: The retrospective analysis of a nationwide COVID-19 cohort
Frontiers in Medicine, doi:10.3389/fmed.2022.894126
Furthermore, favipiravir was associated with increased risks of ICU admission (OR [95% CI]: 3.02 [1.70-5.35]; p = 0.001) and invasive mechanical ventilation requirement (OR [95% CI]: 2.94 [1.28-6.75]; p = 0.011).
Conclusion: Our findings demonstrated that antiviral drugs including lopinavir, ritonavir, and favipiravir were associated with negative clinical outcomes such as increased risks for lengthy hospital stay, ICU admission, and invasive mechanical ventilation requirement. Therefore, repurposing such agents without proven clinical evidence might not be the best approach for COVID-19 treatment.
Ethics statement The studies involving human participants were reviewed and approved by Institutional Review Board of Gazi University Faculty of Medicine, Ankara, Turkey (356/22.05.2020). Written informed consent for participation was not required for this study in accordance with the national legislation and the institutional requirements.
Author contributions CB, NKk, HBa, OI, FO, AS, ASC, BD, and AK designed the study. CB, NKk, PC, SAB, OKa, PA, II, AAk, YS, OB, EA, SM, CC, AD, BK, BB, HO, GO, ZT, BE, VA, OKi, ME, TU, OAl, EN, OAt, AAy, DC, YG, FF, FD, NKs, MT, GG, SA, TeT, TiT, OO, OD, PY, IB, HBo, IO, SB, BG, FB, OE, IH, HK, GS, OU, MA, GA, BD, EC, EG, OG, YH, MO, and ASC collected the data. SK and SAN analyzed the data. CB, NKk, SK, and HBa searched the literature and wrote the manuscript. CB, NKk, SK, SAB, PC, and HBa edited and revised manuscript according to journal's instructions. CB, NKk, SK, PC, SAB, and HBa edited and controlled the final version of the manuscript. All authors approved the final version of the manuscript.
Conflict of interest The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The reviewer BH declared a shared affiliation with the authors, II and AK to the handling editor at the time of review.
Publisher's note All claims expressed in this article are solely those of the authors and do not necessarily represent those of..
References
Antoniou, Dilaveris, Manolakou, Galanakos, Magkas et al., QT prolongation and malignant arrhythmia: How serious a problem?, Eur Cardiol, doi:10.15420/ecr.2017:16:1
Arshad, Kilgore, Chaudhry, Jacobsen, Wang et al., Treatment with hydroxychloroquine, azithromycin, and combination in patients hospitalized with COVID-19, Int J Infect Dis, doi:10.1016/j.ijid.2020.06.099
Beigel, Tomashek, Dodd, Mehta, Zingman et al., Remdesivir for the treatment of Covid-19-final report, N Engl J Med, doi:10.1056/NEJMoa2007764
Bernal, Da Silva, Musungaie, Kovalchuk, Gonzalez et al., Molnupiravir for oral treatment of Covid-19 in nonhospitalized patients, N Engl J Med, doi:10.1056/NEJMoa2116044
Catteau, Dauby, Montourcy, Bottieau, Hautekiet et al., Low-dose hydroxychloroquine therapy and mortality in hospitalised patients with COVID-19: A nationwide observational study of 8075 participants, Int J Antimicrob Agents
Chan, Lai, Chu, Tsui, Tam et al., Treatment of severe acute respiratory syndrome with lopinavir/ritonavir: A multicentre retrospective matched cohort study, Hong Kong Med J
Chinello, Petrosillo, Pittalis, Biava, Ippolito et al., Cardiac safety of off-label COVID-19 drug therapy: A review and proposed monitoring protocol, Eur Heart J Acute Cardiovasc Care, doi:10.1177/2048872620922784
Choy, Wong, Kaewpreedee, Sia, Chen et al., None
Covid, 19 Risk and Treatments (Corist) Collaboration. Use of hydroxychloroquine in hospitalised COVID-19 patients is associated with reduced mortality: Findings from the observational multicentre Italian CORIST study, Eur J Intern Med
Croxtall, Perry, Lopinavir/Ritonavir: A review of its use in the management of HIV-1 infection, Drugs, doi:10.2165/11204950-000000000-00000
Doi, Hibino, Hase, Yamamoto, Kasamatsu et al., A prospective, randomized, open-label trial of early versus late favipiravir therapy in hospitalized patients with COVID-19, Antimicrob Agents Chemother, doi:10.1128/AAC.01897-20
Fiolet, Guihur, Rebeaud, Mulot, Peiffer-Smadja et al., Azithromycin in addition to standard of care versus standard of care alone in the treatment of patients admitted to the hospital with severe COVID-19 in Brazil (COALITION II): A randomised clinical trial, Clin Microbiol Infect, doi:10.1016/S0140-6736(20)31862-6
Food, Coronavirus (COVID-19) update: FDA Authorizes additional oral antiviral for treatment of COVID-19 in certain adults
Gao, Tian, Yang, Wei, Breakthrough: Chloroquine phosphate has shown apparent efficacy in treatment of COVID-19 associated pneumonia in clinical studies, Biosci Trends, doi:10.1097/CM9.0000000000000819
Geleris, Sun, Platt, Zucker, Baldwin et al., Observational study of hydroxychloroquine in hospitalized patients with Covid-19, N Engl J Med, doi:10.1056/NEJMoa2012410
Ghafar, Efficacy of favipiravir in COVID-19 treatment: A multi-center randomized study, Arch Virol, doi:10.1007/s00705-021-04956-9
Guner, Hasanoglu, Kayaaslan, Aypak, Akinci et al., Comparing ICU admission rates of mild/moderate COVID-19 patients treated with hydroxychloroquine, favipiravir, and hydroxychloroquine plus favipiravir, J Infect Public Health, doi:10.14744/nci.2021.60420
Ivashchenko, Dmitriev, Vostokova, Azarova, Blinow et al., AVIFAVIR for treatment of patients with moderate coronavirus disease 2019 (COVID-19): Interim results of a phase II/III multicenter randomized clinical trial, Clin Infect Dis, doi:10.1093/cid/ciaa1176
Joshi, Parkar, Ansari, Vora, Talwar et al., Role of favipiravir in the treatment of COVID-19, Int J Infect Dis, doi:10.1016/j.ijid.2020.10.069
Karim, Repurposed antiviral drugs for covid-19 -interim WHO solidarity trial results, N Engl J Med, doi:10.1056/NEJMx200021
Kokturk, Babayigit, Kul, Cetinkaya, Nayci et al., The predictors of COVID-19 mortality in a nationwide cohort of Turkish patients, Respir Med, doi:10.1016/j.rmed.2021.106433
Manabe, Kambayashi, Akatsu, Kudo, Ghanei et al., Favipiravir for the treatment of patients with COVID-19: A systematic review and meta-analysis, Br MC Infect Dis, doi:10.1186/s12879-021-06164-x
Mitjà, Corbacho-Monné, Ubals, Tebé, Peñafiel et al., Hydroxychloroquine for early treatment of adults with mild Covid-19: A randomized-controlled trial, Clin Infect Dis, doi:10.1093/cid/ciaa1009
Pan, Peto, Henao-Restrepo, Preziosi, Sathiyamoorthy, None
Pilkington, Pepperrell, Hill, A review of the safety of favipiravira potential treatment in the COVID-19 pandemic?, J Virus Erad, doi:10.1016/S2055-6640(20)30016-9
Ranieri, Rubenfeld, Thompson, Ferguson, classification criteria for macrophage activation syndrome complicating systemic juvenile idiopathic arthritis: A European league against rheumatism/American College of rheumatology/paediatric rheumatology international trials organisation collaborative initiative, Ann Rheum Dis, doi:10.1378/chest.101.6.1644
Remdesivir, Cao, Wang, Wen, Liu et al., A trial of lopinavirritonavir in adults hospitalized with severe covid-19, Antiviral Res, doi:10.1056/NEJMc2008043
Rice, Janj, Kokturk, Kul, Cetinkaya et al., In defence of evidence-based medicine for the treatment of COVID-19 acute respiratory distress syndrome, doi:10.1513/AnnalsATS.202004-325IP
Rosenberg, Dufort, Udo, Wilberschied, Kumar et al., Association of treatment with hydroxychloroquine or azithromycin with inhospital mortality in patients with COVID-19 in New York state, JAMA, doi:10.1001/jama.2020.8630
Saadat, Safety and efficacy of favipiravir in moderate to severe SARS-CoV-2 pneumonia, Int Immunopharmacol, doi:10.1016/j.intimp.2021.107522
Soltane, Elmaaty, Tantawy, Antar, Yahya, Coronavirus disease (COVID-19) control between drug repurpising and vaccination: A comprehensive overview, Vaccines, doi:10.3390/vaccines9111317
Spinner, Gottlieb, Criner, López, Cattelan et al., FDA approves first treatment for COVID-19
Sun, Deng, Chen, Huang, Huang et al., Incidence of adverse drug reactions in COVID-19 patients in China: An active monitoring study by hospital pharmacovigilance system, Clin Pharmacol Ther, doi:10.1002/cpt.1866
Viladomiu, Effect of remdesivir vs standard care on clinical status at 11 days in patients with moderate COVID-19: A Randomized clinical trial, Ann Intern Med, doi:10.7326/M20-5752
Wang, Cao, Zhang, Xinglou, Jia et al., Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro, Cell Res, doi:10.1038/s41422-020-0282-0
Wu, Chen, Cai, Xia, Zhou et al., Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China, JAMA Intern Med, doi:10.1001/jamainternmed.2020.0994
Yousefi, Valizadeh, Ghaffari, Vahedi, Karbalaei et al., A global treatment for coronaviruses including COVID-19, J Cell Physiol, doi:10.1002/jcp.29785
Özlüşen, Kozan, Akcan, Kalender, Yaprak et al., Effectiveness of favipiravir in COVID-19: A live systematic review, Eur J Clin Microbiol Infect Dis, doi:10.1007/s10096-021-04307-1
DOI record:
{
"DOI": "10.3389/fmed.2022.894126",
"ISSN": [
"2296-858X"
],
"URL": "http://dx.doi.org/10.3389/fmed.2022.894126",
"abstract": "<jats:sec><jats:title>Background and objectives</jats:title><jats:p>Although several repurposed antiviral drugs have been used for the treatment of COVID-19, only a few such as remdesivir and molnupiravir have shown promising effects. The objectives of our study were to investigate the association of repurposed antiviral drugs with COVID-19 morbidity.</jats:p></jats:sec><jats:sec><jats:title>Methods</jats:title><jats:p>Patients admitted to 26 different hospitals located in 16 different provinces between March 11–July 18, 2020, were enrolled. Case definition was based on WHO criteria. Patients were managed according to the guidelines by Scientific Board of Ministry of Health of Turkey. Primary outcomes were length of hospitalization, intensive care unit (ICU) requirement, and intubation.</jats:p></jats:sec><jats:sec><jats:title>Results</jats:title><jats:p>We retrospectively evaluated 1,472 COVID-19 adult patients; 57.1% were men (mean age = 51.9 ± 17.7years). A total of 210 (14.3%) had severe pneumonia, 115 (7.8%) were admitted to ICUs, and 69 (4.7%) were intubated during hospitalization. The median (interquartile range) of duration of hospitalization, including ICU admission, was 7 (5–12) days. Favipiravir (<jats:italic>n</jats:italic> = 328), lopinavir/ritonavir (<jats:italic>n</jats:italic> = 55), and oseltamivir (<jats:italic>n</jats:italic> = 761) were administered as antiviral agents, and hydroxychloroquine (HCQ, <jats:italic>n</jats:italic> = 1,382) and azithromycin (<jats:italic>n</jats:italic> = 738) were used for their immunomodulatory activity. Lopinavir/ritonavir (β [95% CI]: 4.71 [2.31–7.11]; <jats:italic>p</jats:italic> = 0.001), favipiravir (β [95% CI]: 3.55 [2.56–4.55]; <jats:italic>p</jats:italic> = 0.001) and HCQ (β [95% CI]: 0.84 [0.02–1.67]; <jats:italic>p</jats:italic> = 0.046) were associated with increased risk of lengthy hospital stays. Furthermore, favipiravir was associated with increased risks of ICU admission (OR [95% CI]: 3.02 [1.70–5.35]; <jats:italic>p</jats:italic> = 0.001) and invasive mechanical ventilation requirement (OR [95% CI]: 2.94 [1.28–6.75]; <jats:italic>p</jats:italic> = 0.011).</jats:p></jats:sec><jats:sec><jats:title>Conclusion</jats:title><jats:p>Our findings demonstrated that antiviral drugs including lopinavir, ritonavir, and favipiravir were associated with negative clinical outcomes such as increased risks for lengthy hospital stay, ICU admission, and invasive mechanical ventilation requirement. Therefore, repurposing such agents without proven clinical evidence might not be the best approach for COVID-19 treatment.</jats:p></jats:sec>",
"alternative-id": [
"10.3389/fmed.2022.894126"
],
"author": [
{
"affiliation": [],
"family": "Babayigit",
"given": "Cenk",
"sequence": "first"
},
{
"affiliation": [],
"family": "Kokturk",
"given": "Nurdan",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Kul",
"given": "Seval",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Cetinkaya",
"given": "Pelin Duru",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Atis Nayci",
"given": "Sibel",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Argun Baris",
"given": "Serap",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Karcioglu",
"given": "Oguz",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Aysert",
"given": "Pinar",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Irmak",
"given": "Ilim",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Akbas Yuksel",
"given": "Aycan",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Sekibag",
"given": "Yonca",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Baydar Toprak",
"given": "Oya",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Azak",
"given": "Emel",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Mulamahmutoglu",
"given": "Sait",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Cuhadaroglu",
"given": "Caglar",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Demirel",
"given": "Aslihan",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Kerget",
"given": "Bugra",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Baran Ketencioglu",
"given": "Burcu",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ozger",
"given": "Hasan Selcuk",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ozkan",
"given": "Gulcihan",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ture",
"given": "Zeynep",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ergan",
"given": "Begum",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Avkan Oguz",
"given": "Vildan",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Kilinc",
"given": "Oguz",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ercelik",
"given": "Merve",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ulukavak Ciftci",
"given": "Tansu",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Alici",
"given": "Ozlem",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Nurlu Temel",
"given": "Esra",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ataoglu",
"given": "Ozlem",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Aydin",
"given": "Asena",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Cetiner Bahcetepe",
"given": "Dilek",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Gullu",
"given": "Yusuf Taha",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Fakili",
"given": "Fusun",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Deveci",
"given": "Figen",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Kose",
"given": "Neslihan",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Tor",
"given": "Muge Meltem",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Gunluoglu",
"given": "Gulsah",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Altin",
"given": "Sedat",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Turgut",
"given": "Teyfik",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Tuna",
"given": "Tibel",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ozturk",
"given": "Onder",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Dikensoy",
"given": "Oner",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Yildiz Gulhan",
"given": "Pinar",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Basyigit",
"given": "Ilknur",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Boyaci",
"given": "Hasim",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Oguzulgen",
"given": "Ipek Kivilcim",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Borekci",
"given": "Sermin",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Gemicioglu",
"given": "Bilun",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Bayraktar",
"given": "Firat",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Elbek",
"given": "Osman",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Hanta",
"given": "Ismail",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Kuzu Okur",
"given": "Hacer",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Sagcan",
"given": "Gulseren",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Uzun",
"given": "Oguz",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Akgun",
"given": "Metin",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Altinisik",
"given": "Goksel",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Dursun",
"given": "Berna",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Cakir Edis",
"given": "Ebru",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Gulhan",
"given": "Erkmen",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Oner Eyuboglu",
"given": "Fusun",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Gultekin",
"given": "Okkes",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Havlucu",
"given": "Yavuz",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ozkan",
"given": "Metin",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Sakar Coskun",
"given": "Aysin",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Sayiner",
"given": "Abdullah",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Kalyoncu",
"given": "A. Fuat",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Itil",
"given": "Oya",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Bayram",
"given": "Hasan",
"sequence": "additional"
}
],
"container-title": "Frontiers in Medicine",
"container-title-short": "Front. Med.",
"content-domain": {
"crossmark-restriction": true,
"domain": [
"frontiersin.org"
]
},
"created": {
"date-parts": [
[
2022,
8,
31
]
],
"date-time": "2022-08-31T06:59:23Z",
"timestamp": 1661929163000
},
"deposited": {
"date-parts": [
[
2022,
8,
31
]
],
"date-time": "2022-08-31T06:59:31Z",
"timestamp": 1661929171000
},
"indexed": {
"date-parts": [
[
2022,
8,
31
]
],
"date-time": "2022-08-31T07:42:07Z",
"timestamp": 1661931727547
},
"is-referenced-by-count": 0,
"issued": {
"date-parts": [
[
2022,
8,
31
]
]
},
"license": [
{
"URL": "https://creativecommons.org/licenses/by/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2022,
8,
31
]
],
"date-time": "2022-08-31T00:00:00Z",
"timestamp": 1661904000000
}
}
],
"link": [
{
"URL": "https://www.frontiersin.org/articles/10.3389/fmed.2022.894126/full",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "1965",
"original-title": [],
"prefix": "10.3389",
"published": {
"date-parts": [
[
2022,
8,
31
]
]
},
"published-online": {
"date-parts": [
[
2022,
8,
31
]
]
},
"publisher": "Frontiers Media SA",
"reference": [
{
"journal-title": "WHO coronavirus (covid-19) dashboard.",
"key": "B1",
"year": "2021"
},
{
"DOI": "10.1016/j.ijid.2020.06.099",
"article-title": "Treatment with hydroxychloroquine, azithromycin, and combination in patients hospitalized with COVID-19.",
"author": "Arshad",
"doi-asserted-by": "publisher",
"first-page": "396",
"journal-title": "Int J Infect Dis.",
"key": "B2",
"volume": "97",
"year": "2020"
},
{
"DOI": "10.1016/j.ijantimicag.2020.106144",
"article-title": "Low-dose hydroxychloroquine therapy and mortality in hospitalised patients with COVID-19: A nationwide observational study of 8075 participants.",
"author": "Catteau",
"doi-asserted-by": "crossref",
"journal-title": "Int J Antimicrob Agents.",
"key": "B3",
"volume": "56",
"year": "2020"
},
{
"DOI": "10.1016/j.ejim.2020.08.019",
"article-title": "Use of hydroxychloroquine in hospitalised COVID-19 patients is associated with reduced mortality: Findings from the observational multicentre Italian CORIST study.",
"doi-asserted-by": "crossref",
"first-page": "38",
"journal-title": "Eur J Intern Med.",
"key": "B4",
"volume": "82",
"year": "2020"
},
{
"DOI": "10.1128/AAC.01897-20",
"article-title": "A prospective, randomized, open-label trial of early versus late favipiravir therapy in hospitalized patients with COVID-19.",
"author": "Doi",
"doi-asserted-by": "publisher",
"first-page": "e1897",
"journal-title": "Antimicrob Agents Chemother.",
"key": "B5",
"volume": "64",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa2012410",
"article-title": "Observational study of hydroxychloroquine in hospitalized patients with Covid-19.",
"author": "Geleris",
"doi-asserted-by": "publisher",
"first-page": "2411",
"journal-title": "N Engl J Med.",
"key": "B6",
"volume": "382",
"year": "2020"
},
{
"DOI": "10.1093/cid/ciaa1176",
"article-title": "AVIFAVIR for treatment of patients with moderate coronavirus disease 2019 (COVID-19): Interim results of a phase II/III multicenter randomized clinical trial.",
"author": "Ivashchenko",
"doi-asserted-by": "publisher",
"first-page": "531",
"journal-title": "Clin Infect Dis.",
"key": "B7",
"volume": "73",
"year": "2021"
},
{
"DOI": "10.1093/cid/ciaa1009",
"article-title": "Hydroxychloroquine for early treatment of adults with mild Covid-19: A randomized-controlled trial.",
"author": "Mitjà",
"doi-asserted-by": "publisher",
"journal-title": "Clin Infect Dis.",
"key": "B8",
"volume": "73",
"year": "2021"
},
{
"DOI": "10.1001/jama.2020.8630",
"article-title": "Association of treatment with hydroxychloroquine or azithromycin with in-hospital mortality in patients with COVID-19 in New York state.",
"author": "Rosenberg",
"doi-asserted-by": "publisher",
"first-page": "2493",
"journal-title": "JAMA.",
"key": "B9",
"volume": "323",
"year": "2020"
},
{
"journal-title": "Guideline for treatment of adult patients with covid-19.",
"key": "B10",
"year": "2022"
},
{
"DOI": "10.1016/j.rmed.2021.106433",
"article-title": "The predictors of COVID-19 mortality in a nationwide cohort of Turkish patients.",
"author": "Kokturk",
"doi-asserted-by": "publisher",
"journal-title": "Respir Med.",
"key": "B11",
"volume": "183",
"year": "2021"
},
{
"journal-title": "World health organization Covid-19: Case definitions. Case definitions updated in public health surveillance for covid-19.",
"key": "B12",
"year": "2020"
},
{
"journal-title": "Clinical management of covid-19: Interim guidance.",
"key": "B13",
"year": "2020"
},
{
"DOI": "10.1001/jama.2012.5669",
"article-title": "Acute respiratory distress syndrome: The Berlin definition.",
"author": "Ranieri",
"doi-asserted-by": "publisher",
"first-page": "2526",
"journal-title": "JAMA.",
"key": "B14",
"volume": "307",
"year": "2012"
},
{
"DOI": "10.1378/chest.101.6.1644",
"article-title": "Definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis.",
"author": "Bone",
"doi-asserted-by": "publisher",
"first-page": "1644",
"journal-title": "Chest.",
"key": "B15",
"volume": "101",
"year": "1992"
},
{
"DOI": "10.1136/annrheumdis-2015-208982",
"article-title": "2016 classification criteria for macrophage activation syndrome complicating systemic juvenile idiopathic arthritis: A European league against rheumatism/American College of rheumatology/paediatric rheumatology international trials organisation collaborative initiative.",
"author": "Ravelli",
"doi-asserted-by": "crossref",
"first-page": "481",
"journal-title": "Ann Rheum Dis.",
"key": "B16",
"volume": "75",
"year": "2016"
},
{
"DOI": "10.15420/ecr.2017:16:1",
"article-title": "QT prolongation and malignant arrhythmia: How serious a problem?",
"author": "Antoniou",
"doi-asserted-by": "publisher",
"first-page": "112",
"journal-title": "Eur Cardiol.",
"key": "B17",
"volume": "12",
"year": "2017"
},
{
"DOI": "10.1038/s41422-020-0282-0",
"article-title": "Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro.",
"author": "Wang",
"doi-asserted-by": "publisher",
"first-page": "269",
"journal-title": "Cell Res.",
"key": "B18",
"volume": "30",
"year": "2020"
},
{
"DOI": "10.5582/bst.2020.01047",
"article-title": "Breakthrough: Chloroquine phosphate has shown apparent efficacy in treatment of COVID-19 associated pneumonia in clinical studies.",
"author": "Gao",
"doi-asserted-by": "publisher",
"first-page": "72",
"journal-title": "Biosci Trends.",
"key": "B19",
"volume": "14",
"year": "2020"
},
{
"DOI": "10.1097/CM9.0000000000000819",
"article-title": "Diagnosis and treatment protocol for novel coronavirus pneumonia (trial version 7).",
"author": "Wei",
"doi-asserted-by": "publisher",
"first-page": "1087",
"journal-title": "Chin Med J.",
"key": "B20",
"volume": "133",
"year": "2020"
},
{
"journal-title": "Coronavirus (covid-19) update: FDA revokes emergency use authorization for chloroquine and hydroxychloroquine.",
"key": "B21",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa2023184",
"article-title": "Repurposed antiviral drugs for covid-19 – interim WHO solidarity trial results.",
"author": "Pan",
"doi-asserted-by": "publisher",
"first-page": "497",
"journal-title": "N Engl J Med.",
"key": "B22",
"volume": "384",
"year": "2021"
},
{
"DOI": "10.1056/NEJMx200021",
"article-title": "Hydroxychloroquine with or without azithromycin in mild-to-moderate covid-19.",
"author": "Cavalcanti",
"doi-asserted-by": "publisher",
"first-page": "2041",
"journal-title": "N Engl J Med.",
"key": "B23",
"volume": "383",
"year": "2020"
},
{
"DOI": "10.1016/j.cmi.2020.08.022",
"article-title": "Effect of hydroxychloroquine with or without azithromycin on the mortality of coronavirus disease 2019 (COVID-19) patients: A systematic review and meta-analysis.",
"author": "Fiolet",
"doi-asserted-by": "publisher",
"first-page": "19",
"journal-title": "Clin Microbiol Infect.",
"key": "B24",
"volume": "27",
"year": "2021"
},
{
"DOI": "10.1016/S0140-6736(20)31862-6",
"article-title": "Azithromycin in addition to standard of care versus standard of care alone in the treatment of patients admitted to the hospital with severe COVID-19 in Brazil (COALITION II): A randomised clinical trial.",
"author": "Furtado",
"doi-asserted-by": "publisher",
"first-page": "959",
"journal-title": "Lancet.",
"key": "B25",
"volume": "396",
"year": "2020"
},
{
"DOI": "10.2165/11204950-000000000-00000",
"article-title": "Lopinavir/Ritonavir: A review of its use in the management of HIV-1 infection.",
"author": "Croxtall",
"doi-asserted-by": "publisher",
"first-page": "1885",
"journal-title": "Drugs.",
"key": "B26",
"volume": "70",
"year": "2010"
},
{
"DOI": "10.1002/jcp.29785",
"article-title": "A global treatment for coronaviruses including COVID-19.",
"author": "Yousefi",
"doi-asserted-by": "publisher",
"first-page": "9133",
"journal-title": "J Cell Physiol.",
"key": "B27",
"volume": "235",
"year": "2020"
},
{
"article-title": "Treatment of severe acute respiratory syndrome with lopinavir/ritonavir: A multicentre retrospective matched cohort study.",
"author": "Chan",
"first-page": "399",
"journal-title": "Hong Kong Med J.",
"key": "B28",
"volume": "9",
"year": "2003"
},
{
"DOI": "10.1016/j.antiviral.2020.104786",
"article-title": "Remdesivir, lopinavir, emetine, and homoharringtonine inhibit SARS-CoV-2 replication in vitro.",
"author": "Choy",
"doi-asserted-by": "publisher",
"journal-title": "Antiviral Res.",
"key": "B29",
"volume": "178",
"year": "2020"
},
{
"DOI": "10.1056/NEJMc2008043",
"article-title": "A trial of lopinavir-ritonavir in adults hospitalized with severe covid-19.",
"author": "Cao",
"doi-asserted-by": "publisher",
"first-page": "1787",
"journal-title": "N Engl J Med.",
"key": "B30",
"volume": "382",
"year": "2020"
},
{
"DOI": "10.1001/jamainternmed.2020.0994",
"article-title": "Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China.",
"author": "Wu",
"doi-asserted-by": "publisher",
"first-page": "934",
"journal-title": "JAMA Intern Med.",
"key": "B31",
"volume": "180",
"year": "2020"
},
{
"DOI": "10.1016/S0140-6736(20)32013-4",
"article-title": "Lopinavir-ritonavir in patients admitted to hospital with COVID-19 (RECOVERY): A randomised, controlled, open-label, platform trial.",
"doi-asserted-by": "crossref",
"first-page": "1345",
"journal-title": "Lancet.",
"key": "B32",
"volume": "396",
"year": "2020"
},
{
"journal-title": "Interim clinical guidance for management of patients with confirmed coronavirus disease (covid-19).",
"key": "B33",
"year": "2020"
},
{
"DOI": "10.1186/s12879-021-06164-x",
"article-title": "Favipiravir for the treatment of patients with COVID-19: A systematic review and meta-analysis.",
"author": "Manabe",
"doi-asserted-by": "publisher",
"journal-title": "Br MC Infect Dis.",
"key": "B34",
"volume": "21",
"year": "2021"
},
{
"DOI": "10.1016/j.intimp.2021.107522",
"article-title": "Safety and efficacy of favipiravir in moderate to severe SARS-CoV-2 pneumonia.",
"author": "Solaymani-Dodaran",
"doi-asserted-by": "publisher",
"journal-title": "Int Immunopharmacol.",
"key": "B35",
"volume": "95",
"year": "2021"
},
{
"DOI": "10.1007/s00705-021-04956-9",
"article-title": "Efficacy of favipiravir in COVID-19 treatment: A multi-center randomized study.",
"author": "Dabbous",
"doi-asserted-by": "publisher",
"first-page": "949",
"journal-title": "Arch Virol.",
"key": "B36",
"volume": "166",
"year": "2021"
},
{
"DOI": "10.1007/s10096-021-04307-1",
"article-title": "Effectiveness of favipiravir in COVID-19: A live systematic review.",
"author": "Özlüşen",
"doi-asserted-by": "publisher",
"first-page": "2575",
"journal-title": "Eur J Clin Microbiol Infect Dis.",
"key": "B37",
"volume": "40",
"year": "2021"
},
{
"DOI": "10.1016/j.jiph.2020.12.017",
"article-title": "Comparing ICU admission rates of mild/moderate COVID-19 patients treated with hydroxychloroquine, favipiravir, and hydroxychloroquine plus favipiravir.",
"author": "Guner",
"doi-asserted-by": "publisher",
"first-page": "365",
"journal-title": "J Infect Public Health.",
"key": "B38",
"volume": "14",
"year": "2021"
},
{
"DOI": "10.14744/nci.2021.60420",
"article-title": "ICU admission rates in Istanbul following the addition of favipiravir to the national COVID-19 treatment protocol.",
"author": "Guner",
"doi-asserted-by": "publisher",
"first-page": "119",
"journal-title": "North Clin Istanb.",
"key": "B39",
"volume": "8",
"year": "2021"
},
{
"DOI": "10.1016/S2055-6640(20)30016-9",
"article-title": "A review of the safety of favipiravir – a potential treatment in the COVID-19 pandemic?",
"author": "Pilkington",
"doi-asserted-by": "publisher",
"first-page": "45",
"journal-title": "J Virus Erad.",
"key": "B40",
"volume": "6",
"year": "2020"
},
{
"DOI": "10.1016/j.ijid.2020.10.069",
"article-title": "Role of favipiravir in the treatment of COVID-19.",
"author": "Joshi",
"doi-asserted-by": "publisher",
"first-page": "501",
"journal-title": "Int J Infect Dis.",
"key": "B41",
"volume": "102",
"year": "2021"
},
{
"DOI": "10.1371/journal.pntd.0006034",
"article-title": "Qtc interval prolongation during favipiravir therapy in an ebolavirus-infected patient.",
"author": "Chinello",
"doi-asserted-by": "publisher",
"journal-title": "PLoS Negl Trop Dis.",
"key": "B42",
"volume": "11",
"year": "2017"
},
{
"DOI": "10.1177/2048872620922784",
"article-title": "Cardiac safety of off-label COVID-19 drug therapy: A review and proposed monitoring protocol.",
"author": "Naksuk",
"doi-asserted-by": "publisher",
"first-page": "215",
"journal-title": "Eur Heart J Acute Cardiovasc Care.",
"key": "B43",
"volume": "9",
"year": "2020"
},
{
"DOI": "10.1002/cpt.1866",
"article-title": "Incidence of adverse drug reactions in COVID-19 patients in China: An active monitoring study by hospital pharmacovigilance system.",
"author": "Sun",
"doi-asserted-by": "publisher",
"first-page": "791",
"journal-title": "Clin Pharmacol Ther.",
"key": "B44",
"volume": "108",
"year": "2020"
},
{
"journal-title": "FDA approves first treatment for COVID-19.",
"key": "B45",
"year": "2020"
},
{
"DOI": "10.1001/jama.2020.16349",
"article-title": "Effect of remdesivir vs standard care on clinical status at 11 days in patients with moderate COVID-19: A Randomized clinical trial.",
"author": "Spinner",
"doi-asserted-by": "publisher",
"first-page": "1048",
"journal-title": "JAMA.",
"key": "B46",
"volume": "324",
"year": "2020"
},
{
"DOI": "10.7326/M20-5752",
"article-title": "Remdesivir for adults with COVID-19: A living systematic review for an American college of physicians practice points.",
"author": "Wit",
"doi-asserted-by": "publisher",
"first-page": "209",
"journal-title": "Ann Intern Med.",
"key": "B47",
"volume": "174",
"year": "2021"
},
{
"DOI": "10.1056/NEJMoa2007764",
"article-title": "Remdesivir for the treatment of Covid-19-final report.",
"author": "Beigel",
"doi-asserted-by": "publisher",
"first-page": "1813",
"journal-title": "N Engl J Med.",
"key": "B48",
"volume": "383",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa2116044",
"article-title": "Molnupiravir for oral treatment of Covid-19 in nonhospitalized patients.",
"author": "Jayk Bernal",
"doi-asserted-by": "publisher",
"first-page": "509",
"journal-title": "N Engl J Med.",
"key": "B49",
"volume": "386",
"year": "2022"
},
{
"journal-title": "Coronavirus (COVID-19) update: FDA Authorizes additional oral antiviral for treatment of COVID-19 in certain adults.",
"key": "B50",
"year": "2021"
},
{
"DOI": "10.3390/vaccines9111317",
"article-title": "Coronavirus disease (COVID-19) control between drug repurpising and vaccination: A comprehensive overview.",
"author": "Al-Karmalawy",
"doi-asserted-by": "publisher",
"journal-title": "Vaccines.",
"key": "B51",
"volume": "9",
"year": "2021"
},
{
"DOI": "10.1513/AnnalsATS.202004-325IP",
"article-title": "In defence of evidence-based medicine for the treatment of COVID-19 acute respiratory distress syndrome.",
"author": "Rice",
"doi-asserted-by": "publisher",
"first-page": "787",
"journal-title": "Ann Am Thorac Soc.",
"key": "B52",
"volume": "17",
"year": "2020"
}
],
"reference-count": 52,
"references-count": 52,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.frontiersin.org/articles/10.3389/fmed.2022.894126/full"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"General Medicine"
],
"subtitle": [],
"title": "The association of antiviral drugs with COVID-19 morbidity: The retrospective analysis of a nationwide COVID-19 cohort",
"type": "journal-article",
"update-policy": "http://dx.doi.org/10.3389/crossmark-policy",
"volume": "9"
}