Vitamin D and lumisterol novel metabolites can inhibit SARS-CoV-2 replication machinery enzymes
et al., Endocrinology and Metabolism, doi:10.1152/ajpendo.00174.2021, Jul 2021
Vitamin D for COVID-19
8th treatment shown to reduce risk in
October 2020, now with p < 0.00000000001 from 137 studies, recognized in 18 countries.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
In silico analysis showing that vitamin D and lumisterol metabolites may inhibit SARS-CoV-2 Mpro and RdRP.
29 preclinical studies support the efficacy of vitamin D for COVID-19:
Vitamin D has been identified by the European Food Safety Authority (EFSA) as having sufficient evidence for a causal relationship between intake and optimal immune system function27-30.
Vitamin D inhibits SARS-CoV-2 replication in vitro17,24, mitigates lung inflammation, damage, and lethality in mice with an MHV-3 model for β-CoV respiratory infections17,24, reduces SARS-CoV-2 replication in nasal epithelial cells via increased type I interferon expression20, downregulates proinflammatory cytokines IL-1β and TNF-α in SARS-CoV-2 spike protein-stimulated cells16, attenuates nucleocapsid protein-induced hyperinflammation by inactivating the NLRP3 inflammasome through the VDR-BRCC3 signaling pathway21, may be neuroprotective by protecting the blood-brain barrier, reducing neuroinflammation, and via immunomodulatory effects31, may mitigate hyperinflammation and cytokine storm by upregulating TLR10 expression which downregulates proinflammatory cytokines13, downregulates ACE2 and TMPRSS2 in human trophoblasts and minimizes spike protein-induced inflammation19, may minimize cytokine storm by dampening excessive cytokine production2, may suppress viral entry and replication via LL-37 induction11,12, and minimizes platelet aggregation mediated by SARS-CoV-2 spike protein via inhibiting integrin αIIbβ3 outside-in signaling15.
Cholecalciferol and calcifediol directly bind two allosteric pockets on the SARS-CoV-2 Spike RBD, bias the trimer toward a closed state, weaken ACE2 engagement, and reduce viral entry in cell models1.
Calcitriol may destabilize the Spike protein architecture and inhibit IL-17R dimerization, blocking viral entry and mitigating hyperinflammatory cytokine storm32.
Vitamin D improves regulatory immune cell levels and control of proinflammatory cytokines in severe COVID-1933.
Calcifediol inhibits SARS-CoV-2 papain-like protease (PLpro), a critical enzyme for viral replication14.
Symptomatic COVID-19 is associated with a lower frequency of natural killer (NK) cells and vitamin D has been shown to improve NK cell activity34,35.
1.
García-Marín et al., Exploring SARS-CoV-2 Spike RBD Pockets as Targets for Generic Drugs: A Combined Computational, Biophysical, and Biological Approach, ACS Omega, doi:10.1021/acsomega.5c05175.
2.
Alzahrani, A., A new investigation into the molecular mechanism of cholecalciferol towards reducing cytokines storm, Octahedron Drug Research, doi:10.21608/odr.2024.308273.1043.
3.
Haque et al., Exploring potential therapeutic candidates against COVID-19: a molecular docking study, Discover Molecules, doi:10.1007/s44345-024-00005-5.
4.
Morales-Bayuelo et al., New findings on ligand series used as SARS-CoV-2 virus inhibitors within the frameworks of molecular docking, molecular quantum similarity and chemical reactivity indices, F1000Research, doi:10.12688/f1000research.123550.3.
5.
Chellasamy et al., Docking and molecular dynamics studies of human ezrin protein with a modelled SARS-CoV-2 endodomain and their interaction with potential invasion inhibitors, Journal of King Saud University - Science, doi:10.1016/j.jksus.2022.102277.
6.
Pandya et al., Unravelling Vitamin B12 as a potential inhibitor against SARS-CoV-2: A computational approach, Informatics in Medicine Unlocked, doi:10.1016/j.imu.2022.100951.
7.
Mansouri et al., The impact of calcitriol and estradiol on the SARS-CoV-2 biological activity: a molecular modeling approach, Scientific Reports, doi:10.1038/s41598-022-04778-y.
8.
Song et al., Vitamin D3 and its hydroxyderivatives as promising drugs against COVID-19: a computational study, Journal of Biomolecular Structure and Dynamics, doi:10.1080/07391102.2021.1964601.
9.
Qayyum et al., Vitamin D and lumisterol novel metabolites can inhibit SARS-CoV-2 replication machinery enzymes, Endocrinology and Metabolism, doi:10.1152/ajpendo.00174.2021.
10.
Al-Mazaideh et al., Vitamin D is a New Promising Inhibitor to the Main Protease (Mpro) of COVID-19 by Molecular Docking, Journal of Pharmaceutical Research International, doi:10.9734/jpri/2021/v33i29B31603.
11.
Roth et al., Vitamin D-inducible antimicrobial peptide LL-37 binds SARS-CoV-2 Spike and accessory proteins ORF7a and ORF8, Frontiers in Cellular and Infection Microbiology, doi:10.3389/fcimb.2025.1671738.
12.
Vercellino et al., Influence of Sex and 1,25α Dihydroxyvitamin D3 on SARS-CoV-2 Infection and Viral Entry, Pathogens, doi:10.3390/pathogens14080765.
13.
Knez et al., TLR10 overexpression modulates immune response in A549 lung epithelial cells challenged with SARS-CoV-2 S and N proteins, Frontiers in Immunology, doi:10.3389/fimmu.2024.1490478.
14.
Chen et al., In Vitro Characterization of Inhibition Function of Calcifediol to the Protease Activity of SARS-COV-2 PLpro, Journal of Medical Virology, doi:10.1002/jmv.70085.
15.
Wang et al., 1,25‐Dihydroxyvitamin D3 attenuates platelet aggregation potentiated by SARS‐CoV‐2 spike protein via inhibiting integrin αIIbβ3 outside‐in signaling, Cell Biochemistry and Function, doi:10.1002/cbf.4039.
16.
Alcalá-Santiago et al., Disentangling the Immunomodulatory Effects of Vitamin D on the SARS-CoV-2 Virus by In Vitro Approaches, The 14th European Nutrition Conference FENS 2023, doi:10.3390/proceedings2023091415.
17.
Campolina-Silva et al., Dietary Vitamin D Mitigates Coronavirus-Induced Lung Inflammation and Damage in Mice, Viruses, doi:10.3390/v15122434.
18.
Moatasim et al., Potent Antiviral Activity of Vitamin B12 against Severe Acute Respiratory Syndrome Coronavirus 2, Middle East Respiratory Syndrome Coronavirus, and Human Coronavirus 229E, Microorganisms, doi:10.3390/microorganisms11112777.
19.
Vargas-Castro et al., Calcitriol prevents SARS-CoV spike-induced inflammation in human trophoblasts through downregulating ACE2 and TMPRSS2 expression, The Journal of Steroid Biochemistry and Molecular Biology, doi:10.1016/j.jsbmb.2024.106625.
20.
Sposito et al., Age differential CD13 and interferon expression in airway epithelia affect SARS-CoV-2 infection - effects of vitamin D, Mucosal Immunology, doi:10.1016/j.mucimm.2023.08.002.
21.
Chen (B) et al., Vitamin D3 attenuates SARS‐CoV‐2 nucleocapsid protein‐caused hyperinflammation by inactivating the NLRP3 inflammasome through the VDR‐BRCC3 signaling pathway in vitro and in vivo, MedComm, doi:10.1002/mco2.318.
22.
Rybakovsky et al., Calcitriol modifies tight junctions, improves barrier function, and reduces TNF‐α‐induced barrier leak in the human lung‐derived epithelial cell culture model, 16HBE 14o‐, Physiological Reports, doi:10.14814/phy2.15592.
23.
DiGuilio et al., The multiphasic TNF-α-induced compromise of Calu-3 airway epithelial barrier function, Experimental Lung Research, doi:10.1080/01902148.2023.2193637.
24.
Pickard et al., Discovery of re-purposed drugs that slow SARS-CoV-2 replication in human cells, PLOS Pathogens, doi:10.1371/journal.ppat.1009840.
25.
Mok et al., Calcitriol, the active form of vitamin D, is a promising candidate for COVID-19 prophylaxis, bioRxiv, doi:10.1101/2020.06.21.162396.
26.
Fernandes de Souza et al., Lung Inflammation Induced by Inactivated SARS-CoV-2 in C57BL/6 Female Mice Is Controlled by Intranasal Instillation of Vitamin D, Cells, doi:10.3390/cells12071092.
27.
Galmés et al., Suboptimal Consumption of Relevant Immune System Micronutrients Is Associated with a Worse Impact of COVID-19 in Spanish Populations, Nutrients, doi:10.3390/nu14112254.
28.
Galmés (B) et al., Current State of Evidence: Influence of Nutritional and Nutrigenetic Factors on Immunity in the COVID-19 Pandemic Framework, Nutrients, doi:10.3390/nu12092738.
29.
EFSA, Scientific Opinion on the substantiation of a health claim related to vitamin D and contribution to the normal function of the immune system pursuant to Article 14 of Regulation (EC) No 1924/2006, EFSA Journal, doi:10.2903/j.efsa.2015.4096.
30.
EFSA (B), Scientific Opinion on the substantiation of health claims related to vitamin D and normal function of the immune system and inflammatory response (ID 154, 159), maintenance of normal muscle function (ID 155) and maintenance of normal cardiovascular function (ID 159) pursuant to Article 13(1) of Regulation (E, EFSA Journal, doi:10.2903/j.efsa.2010.1468.
31.
Gotelli et al., Understanding the immune-endocrine effects of vitamin D in SARS-CoV-2 infection: a role in protecting against neurodamage?, Neuroimmunomodulation, doi:10.1159/000533286.
32.
Fadel et al., Targeting asparagine and cysteine in SARS-CoV-2 variants and human pro-inflammatory mediators to alleviate COVID-19 severity; a cross-section and in-silico study, Scientific Reports, doi:10.1038/s41598-025-19359-y.
33.
Saheb Sharif-Askari et al., Increased blood immune regulatory cells in severe COVID-19 with autoantibodies to type I interferons, Scientific Reports, doi:10.1038/s41598-023-43675-w.
Qayyum et al., 27 Jul 2021, peer-reviewed, 7 authors.
In silico studies are an important part of preclinical research, however results may be very different in vivo.
Vitamin D and lumisterol novel metabolites can inhibit SARS-CoV-2 replication machinery enzymes
American Journal of Physiology-Endocrinology and Metabolism, doi:10.1152/ajpendo.00174.2021
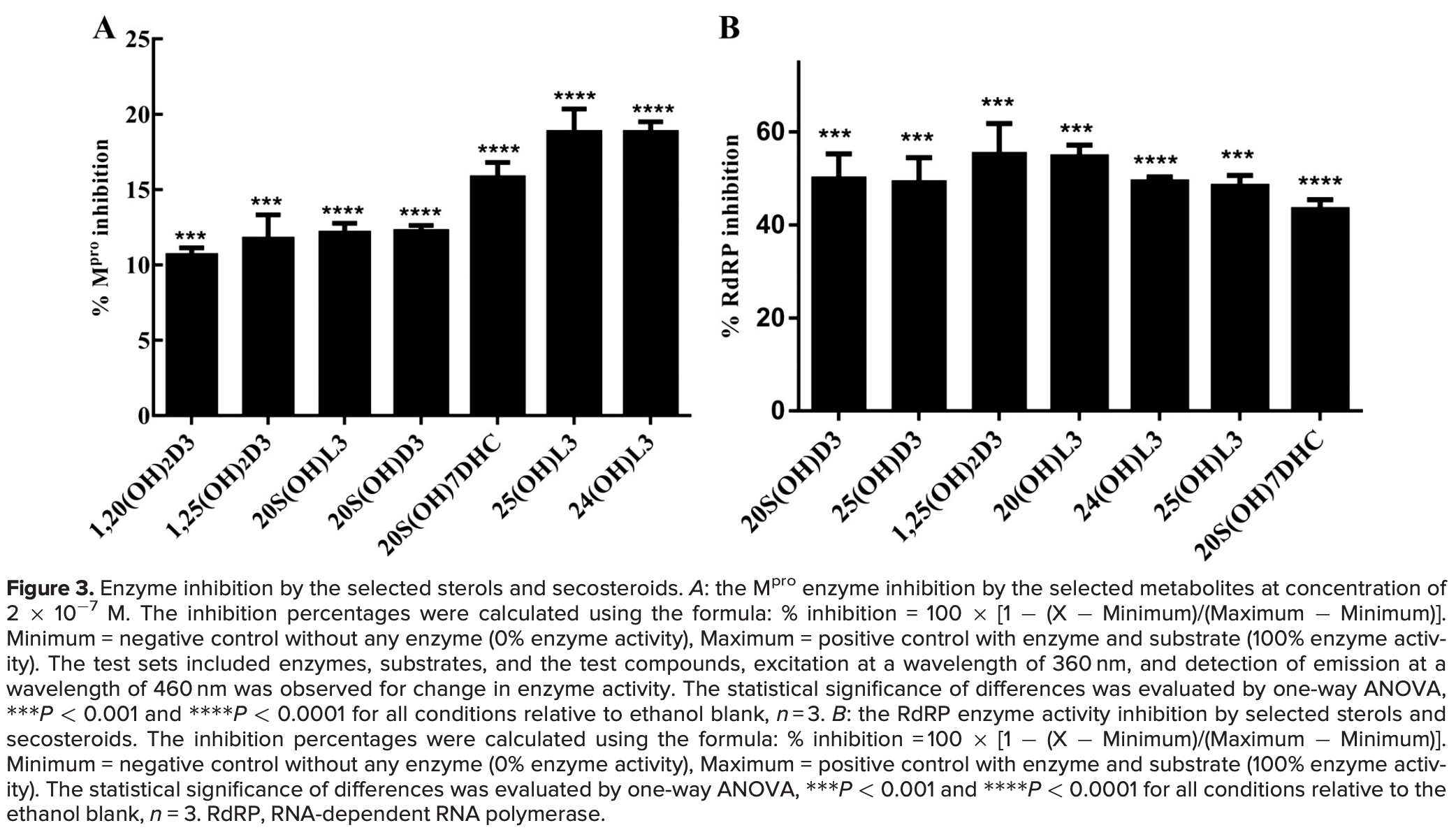
Vitamin D deficiency significantly correlates with the severity of SARS-CoV-2 infection. Molecular docking-based virtual screening studies predict that novel vitamin D and related lumisterol hydroxymetabolites are able to bind to the active sites of two SARS-CoV-2 transcription machinery enzymes with high affinity. These enzymes are the main protease (M pro ) and RNA-dependent RNA polymerase (RdRP), which play important roles in viral replication and establishing infection. Based on predicted binding affinities and specific interactions, we identified 10 vitamin D3 (D3) and lumisterol (L3) analogs as likely binding partners of SARS-CoV-2 M pro and RdRP and, therefore, tested their ability to inhibit these enzymes. Activity measurements demonstrated that 25 (OH)L3, 24(OH)L3, and 20(OH)7DHC are the most effective of the hydroxymetabolites tested at inhibiting the activity of SARS-CoV-2 M pro causing 10%-19% inhibition. These same derivatives as well as other hydroxylumisterols and hydroxyvitamin D3 metabolites inhibited RdRP by 50%-60%. Thus, inhibition of these enzymes by vitamin D and lumisterol metabolites may provide a novel approach to hindering the SARS-CoV-2 infection. NEW & NOTEWORTHY Active forms of vitamin D and lumisterol can inhibit SARS-CoV-2 replication machinery enzymes, which indicates that novel vitamin D and lumisterol metabolites are candidates for antiviral drug research.
References
Bikle, Vitamin D: newer concepts of its metabolism and function at the basic and clinical level, J Endocr Soc, doi:10.1210/jendso/bvz038
Coussens, Naude, Goliath, Chaplin, Wilkinson et al., High-dose vitamin D3 reduces deficiency caused by low UVB exposure and limits HIV-1 replication in urban Southern Africans, Proc Natl Acad Sci, doi:10.1073/pnas.1500909112
Dai, Zhang, Jiang, Su, Li et al., Structure-based design of antiviral drug candidates targeting the SARS-CoV-2 main protease, Science, doi:10.1126/science.abb4489
Grant, Anouti, Moukayed, Targeted 25-hydroxyvitamin D concentration measurements and vitamin D3 supplementation can have important patient and public health benefits, Eur J Clin Nutr, doi:10.1038/s41430-020-0564-0
Haussler, Jurutka, Mizwicki, Norman, Vitamin D receptor (VDR)-mediated actions of 1a, 25 (OH) 2vitamin D3: genomic and non-genomic mechanisms, Best Pract Res Clin Endocrinol Metab, doi:10.1016/j.beem.2011.05.010
Holick, Vitamin D: a millenium perspective, J Cell Biochem, doi:10.1002/jcb.10338
Lung, Lin, Yang, Chou, Shu et al., The potential chemical structure of anti-SARS-CoV-2 RNA-dependent RNA polymerase, Erratum in J Med Virol, doi:10.1002/jmv.25761
Maghbooli, Sahraian, Ebrahimi, Pazoki, Kafan et al., Vitamin D sufficiency, a serum 25-hydroxyvitamin D at least 30 ng/ mL reduced risk for adverse clinical outcomes in patients with COVID-19 infection, PLoS One, doi:10.1371/journal.pone.0239799
Mohammad, Shamsi, Anwar, Umair, Hussain et al., Identification of high-affinity inhibitors of SARS-CoV-2 main protease: towards the development of effective COVID-19 therapy, Virus Res, doi:10.1016/j.virusres.2020.198102
Mok, Ng, Ahidjo, Lee, Loe et al., Calcitriol, the active form of vitamin D, is a promising candidate for COVID-19 prophylaxis, bioRxiv, doi:10.1101/2020.06.21.16239
Naqvi, Fatima, Mohammad, Fatima, Singh et al., Insights into SARS-CoV-2 genome, structure, evolution, pathogenesis and therapies: structural genomics approach, Biochim Biophys Acta Mol Basis Dis, doi:10.1016/j.bbadis.2020.165878
Quesada-Gomez, Entrenas-Castillo, Bouillon, Vitamin D receptor stimulation to reduce acute respiratory distress syndrome (ARDS) in patients with coronavirus SARS-CoV-2 infections: revised Ms SBMB 2020_166, J Steroid Biochem Mol Biol, doi:10.1016/j.jsbmb.2020.105719
Rhodes, Subramanian, Laird, Griffin, Kenny, Perspective: vitamin D deficiency and COVID-19 severity-plausibly linked by latitude, ethnicity, impacts on cytokines, ACE2 and thrombosis, J Intern Med, doi:10.1111/joim.13149
Shirvani, Kalajian, Song, Holick, Disassociation of vitamin D's calcemic activity and non-calcemic genomic activity and individual responsiveness: a randomized controlled double-blind clinical trial, Sci Rep, doi:10.1038/s41598-019-53864-1
Slominski, Chaiprasongsuk, Janjetovic, Kim, Stefan et al., Photoprotective properties of vitamin D and lumisterol hydroxyderivatives, Cell Biochem Biophys, doi:10.1007/s12013-020-00913-6
Slominski, Kim, Hobrath, Janjetovic, Oak et al., Characterization of a new pathway that activates lumisterol in vivo to biologically active hydroxylumisterols, Sci Rep, doi:10.1038/s41598-017-10202-7
Slominski, Li, Kim, Semak, Wang et al., Novel activities of CYP11A1 and their potential physiological significance, J Steroid Biochem Mol Biol, doi:10.1016/j.jsbmb.2014.11.010
Slominski, Stefan, Athar, Holick, Jetten et al., COVID-19 and Vitamin D: a lesson from the skin, Exp Dermatol, doi:10.1111/exd.14170
Teymoori-Rad, Shokri, Salimi, Marashi, The interplay between vitamin D and viral infections, Rev Med Virol, doi:10.1002/rmv.2032
Tuckey, Cheng, Slominski, The serum vitamin D metabolome: what we know and what is still to discover, J Steroid Biochem Mol Biol, doi:10.1016/j.jsbmb.2018.09.003
Yang, Yang, Ding, Liu, Lou et al., The crystal structures of severe acute respiratory syndrome virus main protease and its complex with an inhibitor, Proc Natl Acad Sci, doi:10.1073/pnas.1835675100
Yin, Mao, Luan, Shen, Shen et al., Structural basis for inhibition of the RNA-dependent RNA polymerase from SARS-CoV-2 by remdesivir, Science, doi:10.1126/science.abc1560
Zhang, Lin, Sun, Curth, Drosten et al., Crystal structure of SARS-CoV-2 main protease provides a basis for design of improved a-ketoamide inhibitors, Science, doi:10.1126/science.abb3405
Zu, Deng, Zhou, Li, Li et al., 25-Hydroxycholesterol is a potent SARS-CoV-2 inhibitor, Cell Res, doi:10.1038/s41422-020-00398-1
DOI record:
{
"DOI": "10.1152/ajpendo.00174.2021",
"ISSN": [
"0193-1849",
"1522-1555"
],
"URL": "http://dx.doi.org/10.1152/ajpendo.00174.2021",
"abstract": "<jats:p> Active forms of vitamin D and lumisterol can inhibit SARS-CoV-2 replication machinery enzymes, which indicates that novel vitamin D and lumisterol metabolites are candidates for antiviral drug research. </jats:p>",
"alternative-id": [
"10.1152/ajpendo.00174.2021"
],
"assertion": [
{
"group": {
"label": "Publication History",
"name": "publication_history"
},
"label": "Received",
"name": "received",
"order": 0,
"value": "2021-05-10"
},
{
"group": {
"label": "Publication History",
"name": "publication_history"
},
"label": "Accepted",
"name": "accepted",
"order": 2,
"value": "2021-06-17"
},
{
"group": {
"label": "Publication History",
"name": "publication_history"
},
"label": "Published",
"name": "published",
"order": 3,
"value": "2021-07-27"
}
],
"author": [
{
"affiliation": [
{
"name": "Department of Dermatology, University of Alabama at Birmingham, Birmingham, Alabama"
}
],
"family": "Qayyum",
"given": "Shariq",
"sequence": "first"
},
{
"ORCID": "http://orcid.org/0000-0002-0399-4835",
"affiliation": [
{
"name": "Centre for Interdisciplinary Research in Basic Sciences, Jamia Millia Islamia, New Delhi, India"
}
],
"authenticated-orcid": true,
"family": "Mohammad",
"given": "Taj",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Dermatology, University of Alabama at Birmingham, Birmingham, Alabama"
}
],
"family": "Slominski",
"given": "Radomir M.",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-3663-4940",
"affiliation": [
{
"name": "Centre for Interdisciplinary Research in Basic Sciences, Jamia Millia Islamia, New Delhi, India"
}
],
"authenticated-orcid": true,
"family": "Hassan",
"given": "Md Imtaiyaz",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "School of Molecular Sciences, The University of Western Australia, Perth, Western Australia, Australia"
}
],
"family": "Tuckey",
"given": "Robert C.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Dermatology, University of Alabama at Birmingham, Birmingham, Alabama"
}
],
"family": "Raman",
"given": "Chander",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-8963-3995",
"affiliation": [
{
"name": "Department of Dermatology, University of Alabama at Birmingham, Birmingham, Alabama"
},
{
"name": "Pathology and Laboratory Medicine Service, VA Medical Center, Birmingham, Alabama"
}
],
"authenticated-orcid": true,
"family": "Slominski",
"given": "Andrzej T.",
"sequence": "additional"
}
],
"container-title": "American Journal of Physiology-Endocrinology and Metabolism",
"container-title-short": "American Journal of Physiology-Endocrinology and Metabolism",
"content-domain": {
"crossmark-restriction": true,
"domain": [
"journals.physiology.org"
]
},
"created": {
"date-parts": [
[
2021,
6,
28
]
],
"date-time": "2021-06-28T17:53:10Z",
"timestamp": 1624902790000
},
"deposited": {
"date-parts": [
[
2021,
7,
27
]
],
"date-time": "2021-07-27T15:32:57Z",
"timestamp": 1627399977000
},
"funder": [
{
"DOI": "10.13039/100000738",
"award": [
"1I01BX004293-01A1"
],
"doi-asserted-by": "publisher",
"name": "U.S. Department of Veterans Affairs"
},
{
"DOI": "10.13039/100000069",
"award": [
"1R01AR073004-01A1"
],
"doi-asserted-by": "publisher",
"name": "HHS | NIH | National Institute of Arthritis and Musculoskeletal and Skin Diseases"
},
{
"DOI": "10.13039/100000069",
"award": [
"R01AR071189-01A1"
],
"doi-asserted-by": "publisher",
"name": "HHS | NIH | National Institute of Arthritis and Musculoskeletal and Skin Diseases"
},
{
"DOI": "10.13039/100000060",
"award": [
"R21 AI149267-01A1"
],
"doi-asserted-by": "publisher",
"name": "HHS | NIH | National Institute of Allergy and Infectious Diseases"
},
{
"DOI": "10.13039/501100001411",
"award": [
"ISRM/12(22)/2020"
],
"doi-asserted-by": "publisher",
"name": "Indian Council of Medical Research"
}
],
"indexed": {
"date-parts": [
[
2024,
4,
13
]
],
"date-time": "2024-04-13T20:10:44Z",
"timestamp": 1713039044923
},
"is-referenced-by-count": 37,
"issue": "2",
"issued": {
"date-parts": [
[
2021,
8,
1
]
]
},
"journal-issue": {
"issue": "2",
"published-print": {
"date-parts": [
[
2021,
8,
1
]
]
}
},
"language": "en",
"link": [
{
"URL": "https://journals.physiology.org/doi/pdf/10.1152/ajpendo.00174.2021",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "24",
"original-title": [],
"page": "E246-E251",
"prefix": "10.1152",
"published": {
"date-parts": [
[
2021,
8,
1
]
]
},
"published-print": {
"date-parts": [
[
2021,
8,
1
]
]
},
"publisher": "American Physiological Society",
"reference": [
{
"DOI": "10.1371/journal.pone.0239799",
"doi-asserted-by": "publisher",
"key": "B1"
},
{
"DOI": "10.1016/j.beem.2011.05.010",
"doi-asserted-by": "publisher",
"key": "B2"
},
{
"DOI": "10.1038/s41430-020-0564-0",
"doi-asserted-by": "publisher",
"key": "B3"
},
{
"DOI": "10.1038/s41598-019-53864-1",
"doi-asserted-by": "publisher",
"key": "B4"
},
{
"DOI": "10.1002/rmv.2032",
"doi-asserted-by": "publisher",
"key": "B5"
},
{
"DOI": "10.1210/jendso/bvz038",
"doi-asserted-by": "publisher",
"key": "B6"
},
{
"DOI": "10.1016/j.jsbmb.2020.105719",
"doi-asserted-by": "publisher",
"key": "B7"
},
{
"DOI": "10.1111/exd.14170",
"doi-asserted-by": "publisher",
"key": "B8"
},
{
"DOI": "10.1111/joim.13149",
"doi-asserted-by": "publisher",
"key": "B9"
},
{
"DOI": "10.1073/pnas.1500909112",
"doi-asserted-by": "publisher",
"key": "B10"
},
{
"DOI": "10.1002/jcb.10338",
"doi-asserted-by": "publisher",
"key": "B12"
},
{
"DOI": "10.1038/s41422-020-00398-1",
"doi-asserted-by": "publisher",
"key": "B13"
},
{
"DOI": "10.1007/s12013-020-00913-6",
"doi-asserted-by": "publisher",
"key": "B14"
},
{
"DOI": "10.1016/j.jsbmb.2018.09.003",
"doi-asserted-by": "publisher",
"key": "B15"
},
{
"DOI": "10.1016/j.jsbmb.2014.11.010",
"doi-asserted-by": "publisher",
"key": "B16"
},
{
"DOI": "10.1038/s41598-017-10202-7",
"doi-asserted-by": "publisher",
"key": "B17"
},
{
"DOI": "10.1126/science.abb3405",
"doi-asserted-by": "publisher",
"key": "B18"
},
{
"DOI": "10.1126/science.abb4489",
"doi-asserted-by": "publisher",
"key": "B19"
},
{
"DOI": "10.1016/j.bbadis.2020.165878",
"doi-asserted-by": "publisher",
"key": "B20"
},
{
"DOI": "10.1073/pnas.1835675100",
"doi-asserted-by": "publisher",
"key": "B21"
},
{
"DOI": "10.1002/jmv.25761",
"doi-asserted-by": "publisher",
"key": "B22"
},
{
"DOI": "10.1126/science.abc1560",
"doi-asserted-by": "publisher",
"key": "B23"
},
{
"DOI": "10.1016/j.virusres.2020.198102",
"doi-asserted-by": "publisher",
"key": "B24"
}
],
"reference-count": 23,
"references-count": 23,
"relation": {},
"resource": {
"primary": {
"URL": "https://journals.physiology.org/doi/10.1152/ajpendo.00174.2021"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "Vitamin D and lumisterol novel metabolites can inhibit SARS-CoV-2 replication machinery enzymes",
"type": "journal-article",
"update-policy": "http://dx.doi.org/10.1152/aps-crossmark-policy",
"volume": "321"
}
