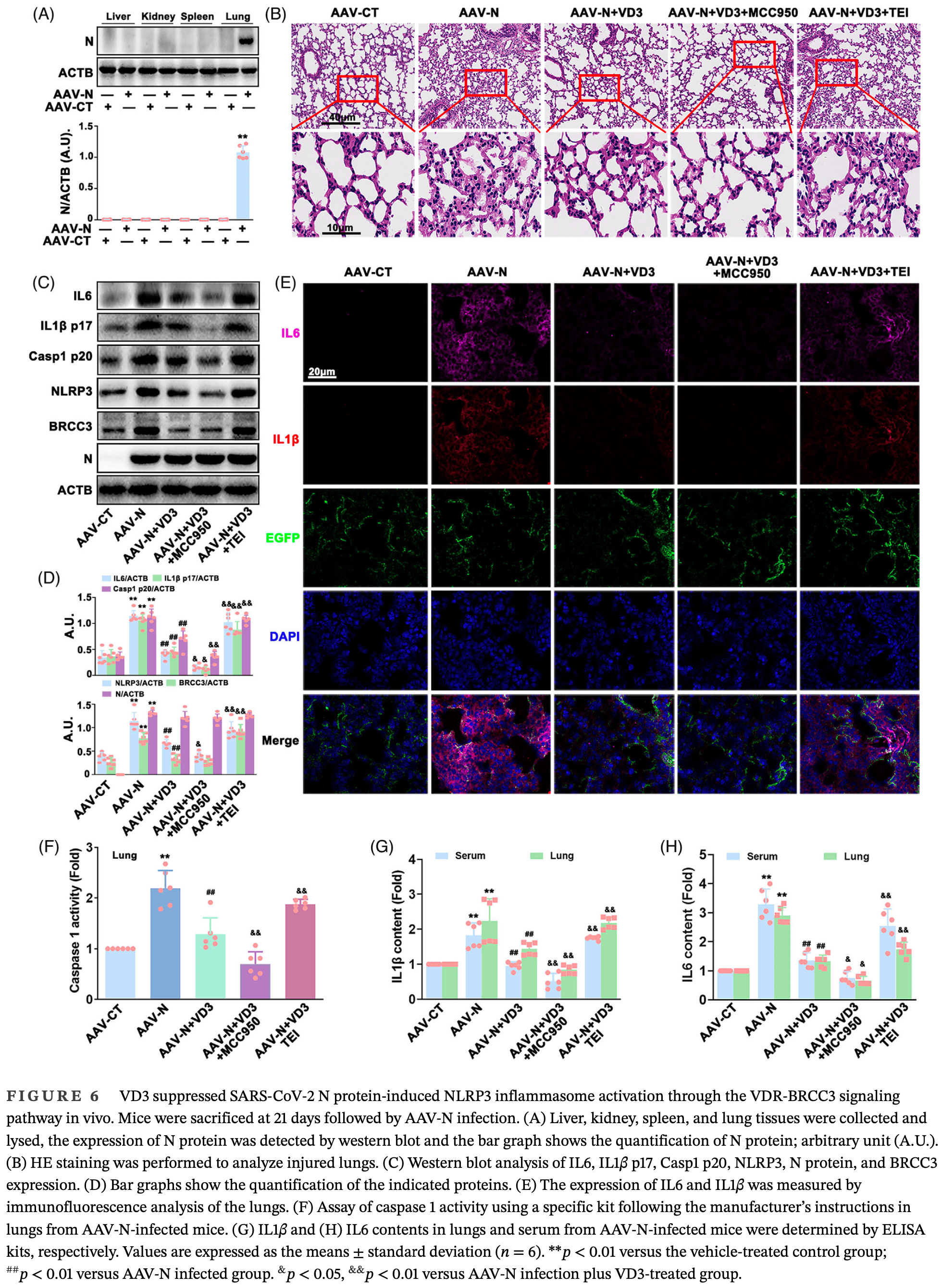
Vitamin D3 attenuates SARS‐CoV‐2 nucleocapsid protein‐caused hyperinflammation by inactivating the NLRP3 inflammasome through the VDR‐BRCC3 signaling pathway in vitro and in vivo
et al., MedComm, doi:10.1002/mco2.318, Jun 2023
Vitamin D for COVID-19
8th treatment shown to reduce risk in
October 2020, now with p < 0.00000000001 from 137 studies, recognized in 18 countries.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
In vitro and mouse study showing that vitamin D3 attenuates SARS-CoV-2 nucleocapsid protein-induced hyperinflammation by inactivating the NLRP3 inflammasome through the VDR-BRCC3 signaling pathway. In human bronchial epithelial cells overexpressing the N protein, vitamin D3 dose-dependently reduced IL-6 and IL-1β expression and secretion. Authors find that vitamin D3 inhibited N protein-triggered NLRP3 inflammasome activation, an effect enhanced by NLRP3 and caspase-1 inhibitors or siRNAs. Vitamin D3 increased NLRP3 ubiquitination and VDR-NLRP3 binding while decreasing BRCC3-NLRP3 association. In N protein-expressing mice, vitamin D3 improved lung injury and decreased NLRP3 inflammasome activation and hyperinflammation in a VDR-dependent manner.
29 preclinical studies support the efficacy of vitamin D for COVID-19:
Vitamin D has been identified by the European Food Safety Authority (EFSA) as having sufficient evidence for a causal relationship between intake and optimal immune system function27-30.
Vitamin D inhibits SARS-CoV-2 replication in vitro17,24, mitigates lung inflammation, damage, and lethality in mice with an MHV-3 model for β-CoV respiratory infections17,24, reduces SARS-CoV-2 replication in nasal epithelial cells via increased type I interferon expression20, downregulates proinflammatory cytokines IL-1β and TNF-α in SARS-CoV-2 spike protein-stimulated cells16, attenuates nucleocapsid protein-induced hyperinflammation by inactivating the NLRP3 inflammasome through the VDR-BRCC3 signaling pathway21, may be neuroprotective by protecting the blood-brain barrier, reducing neuroinflammation, and via immunomodulatory effects31, may mitigate hyperinflammation and cytokine storm by upregulating TLR10 expression which downregulates proinflammatory cytokines13, downregulates ACE2 and TMPRSS2 in human trophoblasts and minimizes spike protein-induced inflammation19, may minimize cytokine storm by dampening excessive cytokine production2, may suppress viral entry and replication via LL-37 induction11,12, and minimizes platelet aggregation mediated by SARS-CoV-2 spike protein via inhibiting integrin αIIbβ3 outside-in signaling15.
Cholecalciferol and calcifediol directly bind two allosteric pockets on the SARS-CoV-2 Spike RBD, bias the trimer toward a closed state, weaken ACE2 engagement, and reduce viral entry in cell models1.
Calcitriol may destabilize the Spike protein architecture and inhibit IL-17R dimerization, blocking viral entry and mitigating hyperinflammatory cytokine storm32.
Vitamin D improves regulatory immune cell levels and control of proinflammatory cytokines in severe COVID-1933.
Calcifediol inhibits SARS-CoV-2 papain-like protease (PLpro), a critical enzyme for viral replication14.
Symptomatic COVID-19 is associated with a lower frequency of natural killer (NK) cells and vitamin D has been shown to improve NK cell activity34,35.
1.
García-Marín et al., Exploring SARS-CoV-2 Spike RBD Pockets as Targets for Generic Drugs: A Combined Computational, Biophysical, and Biological Approach, ACS Omega, doi:10.1021/acsomega.5c05175.
2.
Alzahrani, A., A new investigation into the molecular mechanism of cholecalciferol towards reducing cytokines storm, Octahedron Drug Research, doi:10.21608/odr.2024.308273.1043.
3.
Haque et al., Exploring potential therapeutic candidates against COVID-19: a molecular docking study, Discover Molecules, doi:10.1007/s44345-024-00005-5.
4.
Morales-Bayuelo et al., New findings on ligand series used as SARS-CoV-2 virus inhibitors within the frameworks of molecular docking, molecular quantum similarity and chemical reactivity indices, F1000Research, doi:10.12688/f1000research.123550.3.
5.
Chellasamy et al., Docking and molecular dynamics studies of human ezrin protein with a modelled SARS-CoV-2 endodomain and their interaction with potential invasion inhibitors, Journal of King Saud University - Science, doi:10.1016/j.jksus.2022.102277.
6.
Pandya et al., Unravelling Vitamin B12 as a potential inhibitor against SARS-CoV-2: A computational approach, Informatics in Medicine Unlocked, doi:10.1016/j.imu.2022.100951.
7.
Mansouri et al., The impact of calcitriol and estradiol on the SARS-CoV-2 biological activity: a molecular modeling approach, Scientific Reports, doi:10.1038/s41598-022-04778-y.
8.
Song et al., Vitamin D3 and its hydroxyderivatives as promising drugs against COVID-19: a computational study, Journal of Biomolecular Structure and Dynamics, doi:10.1080/07391102.2021.1964601.
9.
Qayyum et al., Vitamin D and lumisterol novel metabolites can inhibit SARS-CoV-2 replication machinery enzymes, Endocrinology and Metabolism, doi:10.1152/ajpendo.00174.2021.
10.
Al-Mazaideh et al., Vitamin D is a New Promising Inhibitor to the Main Protease (Mpro) of COVID-19 by Molecular Docking, Journal of Pharmaceutical Research International, doi:10.9734/jpri/2021/v33i29B31603.
11.
Roth et al., Vitamin D-inducible antimicrobial peptide LL-37 binds SARS-CoV-2 Spike and accessory proteins ORF7a and ORF8, Frontiers in Cellular and Infection Microbiology, doi:10.3389/fcimb.2025.1671738.
12.
Vercellino et al., Influence of Sex and 1,25α Dihydroxyvitamin D3 on SARS-CoV-2 Infection and Viral Entry, Pathogens, doi:10.3390/pathogens14080765.
13.
Knez et al., TLR10 overexpression modulates immune response in A549 lung epithelial cells challenged with SARS-CoV-2 S and N proteins, Frontiers in Immunology, doi:10.3389/fimmu.2024.1490478.
14.
Chen et al., In Vitro Characterization of Inhibition Function of Calcifediol to the Protease Activity of SARS-COV-2 PLpro, Journal of Medical Virology, doi:10.1002/jmv.70085.
15.
Wang et al., 1,25‐Dihydroxyvitamin D3 attenuates platelet aggregation potentiated by SARS‐CoV‐2 spike protein via inhibiting integrin αIIbβ3 outside‐in signaling, Cell Biochemistry and Function, doi:10.1002/cbf.4039.
16.
Alcalá-Santiago et al., Disentangling the Immunomodulatory Effects of Vitamin D on the SARS-CoV-2 Virus by In Vitro Approaches, The 14th European Nutrition Conference FENS 2023, doi:10.3390/proceedings2023091415.
17.
Campolina-Silva et al., Dietary Vitamin D Mitigates Coronavirus-Induced Lung Inflammation and Damage in Mice, Viruses, doi:10.3390/v15122434.
18.
Moatasim et al., Potent Antiviral Activity of Vitamin B12 against Severe Acute Respiratory Syndrome Coronavirus 2, Middle East Respiratory Syndrome Coronavirus, and Human Coronavirus 229E, Microorganisms, doi:10.3390/microorganisms11112777.
19.
Vargas-Castro et al., Calcitriol prevents SARS-CoV spike-induced inflammation in human trophoblasts through downregulating ACE2 and TMPRSS2 expression, The Journal of Steroid Biochemistry and Molecular Biology, doi:10.1016/j.jsbmb.2024.106625.
20.
Sposito et al., Age differential CD13 and interferon expression in airway epithelia affect SARS-CoV-2 infection - effects of vitamin D, Mucosal Immunology, doi:10.1016/j.mucimm.2023.08.002.
21.
Chen (B) et al., Vitamin D3 attenuates SARS‐CoV‐2 nucleocapsid protein‐caused hyperinflammation by inactivating the NLRP3 inflammasome through the VDR‐BRCC3 signaling pathway in vitro and in vivo, MedComm, doi:10.1002/mco2.318.
22.
Rybakovsky et al., Calcitriol modifies tight junctions, improves barrier function, and reduces TNF‐α‐induced barrier leak in the human lung‐derived epithelial cell culture model, 16HBE 14o‐, Physiological Reports, doi:10.14814/phy2.15592.
23.
DiGuilio et al., The multiphasic TNF-α-induced compromise of Calu-3 airway epithelial barrier function, Experimental Lung Research, doi:10.1080/01902148.2023.2193637.
24.
Pickard et al., Discovery of re-purposed drugs that slow SARS-CoV-2 replication in human cells, PLOS Pathogens, doi:10.1371/journal.ppat.1009840.
25.
Mok et al., Calcitriol, the active form of vitamin D, is a promising candidate for COVID-19 prophylaxis, bioRxiv, doi:10.1101/2020.06.21.162396.
26.
Fernandes de Souza et al., Lung Inflammation Induced by Inactivated SARS-CoV-2 in C57BL/6 Female Mice Is Controlled by Intranasal Instillation of Vitamin D, Cells, doi:10.3390/cells12071092.
27.
Galmés et al., Suboptimal Consumption of Relevant Immune System Micronutrients Is Associated with a Worse Impact of COVID-19 in Spanish Populations, Nutrients, doi:10.3390/nu14112254.
28.
Galmés (B) et al., Current State of Evidence: Influence of Nutritional and Nutrigenetic Factors on Immunity in the COVID-19 Pandemic Framework, Nutrients, doi:10.3390/nu12092738.
29.
EFSA, Scientific Opinion on the substantiation of a health claim related to vitamin D and contribution to the normal function of the immune system pursuant to Article 14 of Regulation (EC) No 1924/2006, EFSA Journal, doi:10.2903/j.efsa.2015.4096.
30.
EFSA (B), Scientific Opinion on the substantiation of health claims related to vitamin D and normal function of the immune system and inflammatory response (ID 154, 159), maintenance of normal muscle function (ID 155) and maintenance of normal cardiovascular function (ID 159) pursuant to Article 13(1) of Regulation (E, EFSA Journal, doi:10.2903/j.efsa.2010.1468.
31.
Gotelli et al., Understanding the immune-endocrine effects of vitamin D in SARS-CoV-2 infection: a role in protecting against neurodamage?, Neuroimmunomodulation, doi:10.1159/000533286.
32.
Fadel et al., Targeting asparagine and cysteine in SARS-CoV-2 variants and human pro-inflammatory mediators to alleviate COVID-19 severity; a cross-section and in-silico study, Scientific Reports, doi:10.1038/s41598-025-19359-y.
33.
Saheb Sharif-Askari et al., Increased blood immune regulatory cells in severe COVID-19 with autoantibodies to type I interferons, Scientific Reports, doi:10.1038/s41598-023-43675-w.
Chen et al., 21 Jun 2023, peer-reviewed, 11 authors.
Contact: hongbinsong@263.net, bianxiuwu@263.net.
Vitamin D3 attenuates SARS‐CoV‐2 nucleocapsid protein‐caused hyperinflammation by inactivating the NLRP3 inflammasome through the VDR‐BRCC3 signaling pathway in vitro and in vivo
MedComm, doi:10.1002/mco2.318
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection-caused coronavirus disease 2019 (COVID-19) is a global crisis with no satisfactory therapies. Vitamin D3 (VD3) is considered a potential candidate for COVID-19 treatment; however, little information is available regarding the exact effects of VD3 on SARS-CoV-2 infection and the underlying mechanism. Herein, we confirmed that VD3 reduced SARS-CoV-2 nucleocapsid (N) protein-caused hyperinflammation in human bronchial epithelial (HBE) cells. Meanwhile, VD3 inhibited the NOD-like receptor family pyrin domain containing 3 (NLRP3) inflammasome activation in N protein-overexpressed HBE (HBE-N) cells. Notably, the inhibitors of caspase-1, NLRP3, and NLRP3 or caspase-1 small interference RNA (siRNA) enhanced VD3-induced NLRP3 inflammasome inactivation, with subsequent suppression of interleukin-6 (IL6) and IL1β release in HBE-N cells, which were abolished by the NLRP3 agonist. Moreover, VD3 increased NLRP3 ubiquitination (Ub-NLRP3) expression and the binding of the VDR with NLRP3, with decreased BRCA1/BRCA2-containing complex subunit 3 (BRCC3) expression and NLRP3-BRCC3 association. VD3-induced Ub-NLRP3 expression, NLRP3 inflammasome inactivation, and hyperinflammation inhibition were improved by the BRCC3
AUTHOR CONTRIBUTIONS
A C K N O W L E D G M E N T S We are particularly grateful to Figdraw (https://www. figdraw.com/static/index.html) for providing a useful tool to draw the diagram of the mechanism. This work was supported by the National Natural Science Foundation of China (grant numbers: 82073544 and 82202189) and the Beijing Municipal Natural Science Foundation of China (grant number: 7232229).
C O N F L I C T O F I N T E R E S T S TAT E M E N T The authors declare no conflict of interest.
D ATA AVA I L A B I L I T Y S TAT E M E N T All data and materials are available to the researchers once published.
E T H I C S S TAT E M E N T All animal experiments were carried out in strict accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals by the National Institutes of Health and were approved by the Animal Care and Use Committee of the Army Medical University (AMUWEC20212432, Chongqing, China).
O R C I D Xiaofeng Hu https://orcid.org/0000-0003-0569-8535
R E F E R E N C E S S U P P O R T I N G I N F O R M AT I O N Additional supporting information can be found online in the Supporting Information section at the end of this article.
References
Akther, Haque, Park, Kang, Lee, NLRP3 Ubiquitination-A new approach to target NLRP3 inflammasome activation, Int J Mol Sci
Cantorna, Zhu, Froicu, Wittke, Vitamin D status, 1,25dihydroxyvitamin D3, and the immune system, Am J Clin Nutr
Chen, Moriyama, Chang, Ichinohe, Severe acute respiratory syndrome coronavirus Viroporin 3a activates the NLRP3 inflammasome, Front Microbiol
Chen, Zhu, Ran, Lang, Yi et al., Trimethylamine-N-Oxide induces vascular inflammation by activating the NLRP3 inflammasome through the SIRT3-SOD2-mtROS signaling pathway, J Am Heart Assoc
Dong, He, Ye, Vitamin D3 ameliorates nitrogen mustard-induced cutaneous inflammation by inactivating the NLRP3 inflammasome through the SIRT3-SOD2-mtROS signaling pathway, Clin Transl Med
Ebadi, Montano-Loza, Perspective: improving vitamin D status in the management of COVID-19, Eur J Clin Nutr
Freeman, Swartz Targeting the NLRP3 inflammasome in severe COVID-19, Front Immunol
Guo, Cao, Hong, The origin, transmission and clinical therapies on coronavirus disease 2019 (COVID-19) outbreak-an update on the status, Mil Med Res
He, Dong, Zhu, Xu, Chen et al., Ultrasoundtriggered microbubble destruction enhances the radiosensitivity of glioblastoma by inhibiting PGRMC1-mediated autophagy in vitro and in vivo, Mil Med Res
Hu, Dong, Wang, Jing, Chen, Vitamin D(3)-vitamin D receptor axis suppresses pulmonary emphysema by maintaining alveolar macrophage homeostasis and function, EBioMedicine
Huang, Ju, Role of vitamin D/VDR nuclear translocation in down-regulation of NF-κB/NLRP3/caspase-1 axis in lupus nephritis, Int Immunopharmacol
Ishizuka, Kurihara, Miura, Vitamin D antagonist, TEI-9647, inhibits osteoclast formation induced by 1alpha,25dihydroxyvitamin D3 from pagetic bone marrow cells, J Steroid Biochem Mol Biol
Jin, Cai, Cheng, A rapid advice guideline for the diagnosis and treatment of 2019 novel coronavirus (2019-nCoV) infected pneumonia (standard version), Military Medical Research
Jin, Zhan, Peng, Chemoprophylaxis, diagnosis, treatments, and discharge management of COVID-19: An evidence-based clinical practice guideline (updated version), Mil Med Res
Jolliffe, Camargo, Sluyter, Vitamin D supplementation to prevent acute respiratory infections: a systematic review and meta-analysis of aggregate data from randomised controlled trials, Lancet Diabetes Endocrinol
Juliana, Fernandes-Alnemri, Kang, Farias, Qin et al., Non-transcriptional priming and deubiquitination regulate NLRP3 inflammasome activation, J Biol Chem
Lara, Macías-Verde, Burgos-Burgos, Age-induced NLRP3 inflammasome over-activation increases lethality of SARS-CoV-2 pneumonia in elderly patients, Aging Dis
Li, Zhong, Li, Wang, Effects of 1,25-dihydroxyvitamin D3 on experimental periodontitis and AhR/NF-κB/NLRP3 inflammasome pathway in a mouse model, J Appl Oral Sci
Liu, Qian, Cao, Post-translational modification control of innate immunity, Immunity
Liu, Shi, Cai, Pathological changes in the lungs and lymphatic organs of 12 COVID-19 autopsy cases, Natl Sci Rev
Lucchesi, Silimbani, Musuraca, Clinical and biological data on the use of hydroxychloroquine against SARS-CoV-2 could support the role of the NLRP3 inflammasome in the pathogenesis of respiratory disease, J Med Virol
Ma, Zhu, Zhao, SARS-CoV-2 nucleocapsid suppresses host pyroptosis by blocking Gasdermin D cleavage, EMBO J
Manchanda, Bid, Vitamin D receptor and type 2 diabetes mellitus: Growing therapeutic opportunities, Indian J Hum Genet
Mangan, Olhava, Roush, Seidel, Glick et al., Targeting the NLRP3 inflammasome in inflammatory diseases, Nat Rev Drug Discov
Martineau, Jolliffe, Hooper, Vitamin D supplementation to prevent acute respiratory tract infections: Systematic review and meta-analysis of individual participant data, BMJ
Nieto-Torres, Verdiá-Báguena, Jimenez-Guardeño, Severe acute respiratory syndrome coronavirus E protein transports calcium ions and activates the NLRP3 inflammasome, Virology
Pan, Shen, Yu, SARS-CoV-2 N protein promotes NLRP3 inflammasome activation to induce hyperinflammation, Nat Commun
Py, Kim, Vakifahmetoglu-Norberg, Yuan, Deubiquitination of NLRP3 by BRCC3 critically regulates inflammasome activity, Mol Cell
Rao, Chen, Wu, Vitamin D receptor inhibits NLRP3 activation by impeding its BRCC3-mediated deubiquitination, Front Immunol
Ren, Zhang, Xiao, ABRO1 promotes NLRP3 inflammasome activation through regulation of NLRP3 deubiquitination, EMBO J
Rodrigues, De Sá, Ishimoto, Inflammasomes are activated in response to SARS-CoV-2 infection and are associated with COVID-19 severity in patients, J Exp Med
Sassi, Tamone, Amelio, Vitamin D: Nutrient, hormone, and immunomodulator, Nutrients
Sowa, Bennett, Gygi, Harper, Defining the human deubiquitinating enzyme interaction landscape, Cell
Tang, Tu, Lin, Sequential ubiquitination of NLRP3 by RNF125 and Cbl-b limits inflammasome activation and endotoxemia, J Exp Med
Teshome, Adane, Girma, Mekonnen, The impact of vitamin D level on COVID-19 infection: Systematic review and meta-analysis, Front Public Health
Wei, Christakos, Mechanisms underlying the regulation of innate and adaptive immunity by vitamin D, Nutrients
White, Reeves, Pharmacological properties of foods and nutrients, J Ethn Subst Abuse
Wu, Wang, Cai, Effects of different therapeutic schedules on patients with COVID-19: A prospective casecontrol study in China, Advanced Gut & Microbiome Research
Xin, Zhai, 1,25-Dihydroxy Vitamin D(3) attenuates the oxidative stress-mediated inflammation induced by PM(2.5)via the p38/NF-κB/NLRP3 pathway, Inflammation
Xu, Ong, Wang, Role of adjunctive treatment strategies in COVID-19 and a review of international and national clinical guidelines, Mil Med Res
Zemb, Bergman, Camargo, Vitamin D deficiency and the COVID-19 pandemic, J Glob Antimicrob Resist
Zeng, Xie, Feng, Specific inhibition of the NLRP3 inflammasome suppresses immune overactivation and alleviates COVID-19 like pathology in mice, EBioMedicine
Zhao, Di, Xu, The NLRP3 inflammasome and COVID-19: Activation, pathogenesis and therapeutic strategies, Cytokine Growth Factor Rev
DOI record:
{
"DOI": "10.1002/mco2.318",
"ISSN": [
"2688-2663",
"2688-2663"
],
"URL": "http://dx.doi.org/10.1002/mco2.318",
"abstract": "<jats:title>Abstract</jats:title><jats:p>Severe acute respiratory syndrome coronavirus 2 (SARS‐CoV‐2) infection‐caused coronavirus disease 2019 (COVID‐19) is a global crisis with no satisfactory therapies. Vitamin D3 (VD3) is considered a potential candidate for COVID‐19 treatment; however, little information is available regarding the exact effects of VD3 on SARS‐CoV‐2 infection and the underlying mechanism. Herein, we confirmed that VD3 reduced SARS‐CoV‐2 nucleocapsid (N) protein‐caused hyperinflammation in human bronchial epithelial (HBE) cells. Meanwhile, VD3 inhibited the NOD‐like receptor family pyrin domain containing 3 (NLRP3) inflammasome activation in N protein‐overexpressed HBE (HBE‐N) cells. Notably, the inhibitors of caspase‐1, NLRP3, and <jats:italic>NLRP3</jats:italic> or <jats:italic>caspase‐1</jats:italic> small interference RNA (siRNA) enhanced VD3‐induced NLRP3 inflammasome inactivation, with subsequent suppression of interleukin‐6 (IL6) and IL1β release in HBE‐N cells, which were abolished by the NLRP3 agonist. Moreover, VD3 increased NLRP3 ubiquitination (Ub‐NLRP3) expression and the binding of the VDR with NLRP3, with decreased BRCA1/BRCA2‐containing complex subunit 3 (BRCC3) expression and NLRP3‐BRCC3 association. VD3‐induced Ub‐NLRP3 expression, NLRP3 inflammasome inactivation, and hyperinflammation inhibition were improved by the BRCC3 inhibitor or <jats:italic>BRCC3</jats:italic> siRNA, which were attenuated by the vitamin D receptor (VDR) antagonist or <jats:italic>VDR</jats:italic> siRNA in HBE‐N cells. Finally, the results of the in vivo study in AAV‐Lung‐enhanced green fluorescent protein‐N‐infected lungs were consistent with the findings of the in vitro experiment. In conclusion, VD3 attenuated N protein‐caused hyperinflammation by inactivating the NLRP3 inflammasome partially through the VDR‐BRCC3 signaling pathway.</jats:p>",
"alternative-id": [
"10.1002/mco2.318"
],
"author": [
{
"affiliation": [
{
"name": "Institute of Pathology and Southwest Cancer Centre Southwest Hospital Army Medical University Chongqing China"
},
{
"name": "Institute of Toxicology School of Military Preventive Medicine Army Medical University Chongqing China"
}
],
"family": "Chen",
"given": "Mingliang",
"sequence": "first"
},
{
"affiliation": [
{
"name": "Department of Ultrasound Xinqiao Hospital Army Medical University Chongqing China"
}
],
"family": "He",
"given": "Ying",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0003-0569-8535",
"affiliation": [
{
"name": "Department of Health Supervision and Surveillance Chinese PLA Center for Disease Control and Prevention Beijing China"
}
],
"authenticated-orcid": false,
"family": "Hu",
"given": "Xiaofeng",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Institute of Toxicology School of Military Preventive Medicine Army Medical University Chongqing China"
}
],
"family": "Dong",
"given": "Xunhu",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Institute of Pathology and Southwest Cancer Centre Southwest Hospital Army Medical University Chongqing China"
}
],
"family": "Yan",
"given": "Zexuan",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Institute of Pathology and Southwest Cancer Centre Southwest Hospital Army Medical University Chongqing China"
}
],
"family": "Zhao",
"given": "Qingning",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Institute of Pathology and Southwest Cancer Centre Southwest Hospital Army Medical University Chongqing China"
}
],
"family": "Li",
"given": "Jingyuan",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Institute of Pathology and Southwest Cancer Centre Southwest Hospital Army Medical University Chongqing China"
}
],
"family": "Xiang",
"given": "Dongfang",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Institute of Pathology and Southwest Cancer Centre Southwest Hospital Army Medical University Chongqing China"
}
],
"family": "Lin",
"given": "Yong",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Health Supervision and Surveillance Chinese PLA Center for Disease Control and Prevention Beijing China"
}
],
"family": "Song",
"given": "Hongbin",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Institute of Pathology and Southwest Cancer Centre Southwest Hospital Army Medical University Chongqing China"
}
],
"family": "Bian",
"given": "Xiuwu",
"sequence": "additional"
}
],
"container-title": "MedComm",
"container-title-short": "MedComm",
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2023,
6,
24
]
],
"date-time": "2023-06-24T07:17:35Z",
"timestamp": 1687591055000
},
"deposited": {
"date-parts": [
[
2023,
9,
6
]
],
"date-time": "2023-09-06T16:28:59Z",
"timestamp": 1694017739000
},
"funder": [
{
"DOI": "10.13039/501100001809",
"award": [
"82073544",
"82202189"
],
"doi-asserted-by": "publisher",
"name": "National Natural Science Foundation of China"
}
],
"indexed": {
"date-parts": [
[
2023,
12,
26
]
],
"date-time": "2023-12-26T08:13:55Z",
"timestamp": 1703578435394
},
"is-referenced-by-count": 1,
"issue": "4",
"issued": {
"date-parts": [
[
2023,
6,
21
]
]
},
"journal-issue": {
"issue": "4",
"published-print": {
"date-parts": [
[
2023,
8
]
]
}
},
"language": "en",
"license": [
{
"URL": "http://creativecommons.org/licenses/by/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2023,
6,
21
]
],
"date-time": "2023-06-21T00:00:00Z",
"timestamp": 1687305600000
}
}
],
"link": [
{
"URL": "https://onlinelibrary.wiley.com/doi/pdf/10.1002/mco2.318",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "311",
"original-title": [],
"prefix": "10.1002",
"published": {
"date-parts": [
[
2023,
6,
21
]
]
},
"published-online": {
"date-parts": [
[
2023,
6,
21
]
]
},
"published-print": {
"date-parts": [
[
2023,
8
]
]
},
"publisher": "Wiley",
"reference": [
{
"DOI": "10.1186/s40779-020-00240-0",
"doi-asserted-by": "publisher",
"key": "e_1_2_10_2_1"
},
{
"DOI": "10.1093/nsr/nwaa247",
"doi-asserted-by": "publisher",
"key": "e_1_2_10_3_1"
},
{
"article-title": "Pharmacological properties of foods and nutrients",
"author": "White JV",
"first-page": "1",
"issue": "1",
"journal-title": "J Ethn Subst Abuse",
"key": "e_1_2_10_4_1",
"volume": "15",
"year": "2008"
},
{
"article-title": "Role of adjunctive treatment strategies in COVID‐19 and a review of international and national clinical guidelines",
"author": "Xu X",
"first-page": "22",
"issue": "1",
"journal-title": "Mil Med Res",
"key": "e_1_2_10_5_1",
"volume": "7",
"year": "2020"
},
{
"DOI": "10.1093/ajcn/80.6.1717S",
"doi-asserted-by": "publisher",
"key": "e_1_2_10_6_1"
},
{
"DOI": "10.1136/bmj.i6583",
"doi-asserted-by": "publisher",
"key": "e_1_2_10_7_1"
},
{
"DOI": "10.1016/j.jgar.2020.05.006",
"doi-asserted-by": "publisher",
"key": "e_1_2_10_8_1"
},
{
"DOI": "10.3389/fpubh.2021.624559",
"doi-asserted-by": "publisher",
"key": "e_1_2_10_9_1"
},
{
"DOI": "10.1038/nrd.2018.97",
"doi-asserted-by": "publisher",
"key": "e_1_2_10_10_1"
},
{
"DOI": "10.1016/j.cytogfr.2021.06.002",
"doi-asserted-by": "publisher",
"key": "e_1_2_10_11_1"
},
{
"DOI": "10.1002/jmv.26217",
"doi-asserted-by": "publisher",
"key": "e_1_2_10_12_1"
},
{
"DOI": "10.1084/jem.20201707",
"doi-asserted-by": "publisher",
"key": "e_1_2_10_13_1"
},
{
"DOI": "10.14336/AD.2020.0601",
"doi-asserted-by": "publisher",
"key": "e_1_2_10_14_1"
},
{
"DOI": "10.1038/s41467-021-25015-6",
"doi-asserted-by": "publisher",
"key": "e_1_2_10_15_1"
},
{
"DOI": "10.1016/j.ebiom.2021.103803",
"doi-asserted-by": "publisher",
"key": "e_1_2_10_16_1"
},
{
"DOI": "10.1007/s10753-018-0928-y",
"doi-asserted-by": "crossref",
"key": "e_1_2_10_17_1",
"unstructured": "XinL CheB ZhaiB et al.1 25‐Dihydroxy Vitamin D(3) attenuates the oxidative stress‐mediated inflammation induced by PM(2.5)via the p38/NF‐κB/NLRP3 pathway.Inflammation.2019;42(2):702‐713."
},
{
"DOI": "10.1590/1678-7757-2018-0713",
"doi-asserted-by": "publisher",
"key": "e_1_2_10_18_1"
},
{
"DOI": "10.1002/ctm2.312",
"doi-asserted-by": "publisher",
"key": "e_1_2_10_19_1"
},
{
"DOI": "10.3390/ijms22168780",
"doi-asserted-by": "publisher",
"key": "e_1_2_10_20_1"
},
{
"DOI": "10.1016/j.molcel.2012.11.009",
"doi-asserted-by": "publisher",
"key": "e_1_2_10_21_1"
},
{
"DOI": "10.3389/fimmu.2019.02783",
"doi-asserted-by": "publisher",
"key": "e_1_2_10_22_1"
},
{
"DOI": "10.4103/0971-6866.107975",
"doi-asserted-by": "publisher",
"key": "e_1_2_10_23_1"
},
{
"DOI": "10.3390/nu10111656",
"doi-asserted-by": "publisher",
"key": "e_1_2_10_24_1"
},
{
"DOI": "10.1016/j.intimp.2021.108131",
"doi-asserted-by": "publisher",
"key": "e_1_2_10_25_1"
},
{
"DOI": "10.1016/j.ebiom.2019.06.039",
"doi-asserted-by": "publisher",
"key": "e_1_2_10_26_1"
},
{
"DOI": "10.1016/j.immuni.2016.06.020",
"doi-asserted-by": "publisher",
"key": "e_1_2_10_27_1"
},
{
"DOI": "10.1084/jem.20182091",
"doi-asserted-by": "publisher",
"key": "e_1_2_10_28_1"
},
{
"DOI": "10.1186/s40779-020-0233-6",
"doi-asserted-by": "publisher",
"key": "e_1_2_10_29_1"
},
{
"article-title": "Chemoprophylaxis, diagnosis, treatments, and discharge management of COVID‐19: An evidence‐based clinical practice guideline (updated version)",
"author": "Jin YH",
"first-page": "41",
"issue": "1",
"journal-title": "Mil Med Res",
"key": "e_1_2_10_30_1",
"volume": "7",
"year": "2020"
},
{
"DOI": "10.1038/s41430-020-0661-0",
"doi-asserted-by": "publisher",
"key": "e_1_2_10_31_1"
},
{
"DOI": "10.3390/nu7105392",
"doi-asserted-by": "publisher",
"key": "e_1_2_10_32_1"
},
{
"DOI": "10.1016/S2213-8587(21)00051-6",
"doi-asserted-by": "publisher",
"key": "e_1_2_10_33_1"
},
{
"DOI": "10.1155/2022/7009876",
"doi-asserted-by": "publisher",
"key": "e_1_2_10_34_1"
},
{
"DOI": "10.3389/fimmu.2020.01518",
"doi-asserted-by": "publisher",
"key": "e_1_2_10_35_1"
},
{
"DOI": "10.1016/j.virol.2015.08.010",
"doi-asserted-by": "publisher",
"key": "e_1_2_10_36_1"
},
{
"DOI": "10.3389/fmicb.2019.00050",
"doi-asserted-by": "publisher",
"key": "e_1_2_10_37_1"
},
{
"DOI": "10.15252/embj.2021108249",
"doi-asserted-by": "publisher",
"key": "e_1_2_10_38_1"
},
{
"DOI": "10.1016/j.cell.2009.04.042",
"doi-asserted-by": "publisher",
"key": "e_1_2_10_39_1"
},
{
"DOI": "10.15252/embj.2018100376",
"doi-asserted-by": "publisher",
"key": "e_1_2_10_40_1"
},
{
"DOI": "10.1161/JAHA.117.006347",
"doi-asserted-by": "publisher",
"key": "e_1_2_10_41_1"
},
{
"DOI": "10.1016/j.jsbmb.2004.03.025",
"doi-asserted-by": "publisher",
"key": "e_1_2_10_42_1"
},
{
"article-title": "Ultrasound‐triggered microbubble destruction enhances the radiosensitivity of glioblastoma by inhibiting PGRMC1‐mediated autophagy in vitro and in vivo",
"author": "He Y",
"first-page": "9",
"issue": "1",
"journal-title": "Mil Med Res",
"key": "e_1_2_10_43_1",
"volume": "9",
"year": "2022"
},
{
"DOI": "10.1074/jbc.M112.407130",
"doi-asserted-by": "publisher",
"key": "e_1_2_10_44_1"
}
],
"reference-count": 43,
"references-count": 43,
"relation": {},
"resource": {
"primary": {
"URL": "https://onlinelibrary.wiley.com/doi/10.1002/mco2.318"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "Vitamin D3 attenuates SARS‐CoV‐2 nucleocapsid protein‐caused hyperinflammation by inactivating the NLRP3 inflammasome through the VDR‐BRCC3 signaling pathway in vitro and in vivo",
"type": "journal-article",
"volume": "4"
}
