TLR10 overexpression modulates immune response in A549 lung epithelial cells challenged with SARS-CoV-2 S and N proteins
et al., Frontiers in Immunology, doi:10.3389/fimmu.2024.1490478, Jan 2025
Vitamin D for COVID-19
8th treatment shown to reduce risk in
October 2020, now with p < 0.00000000001 from 137 studies, recognized in 18 countries.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
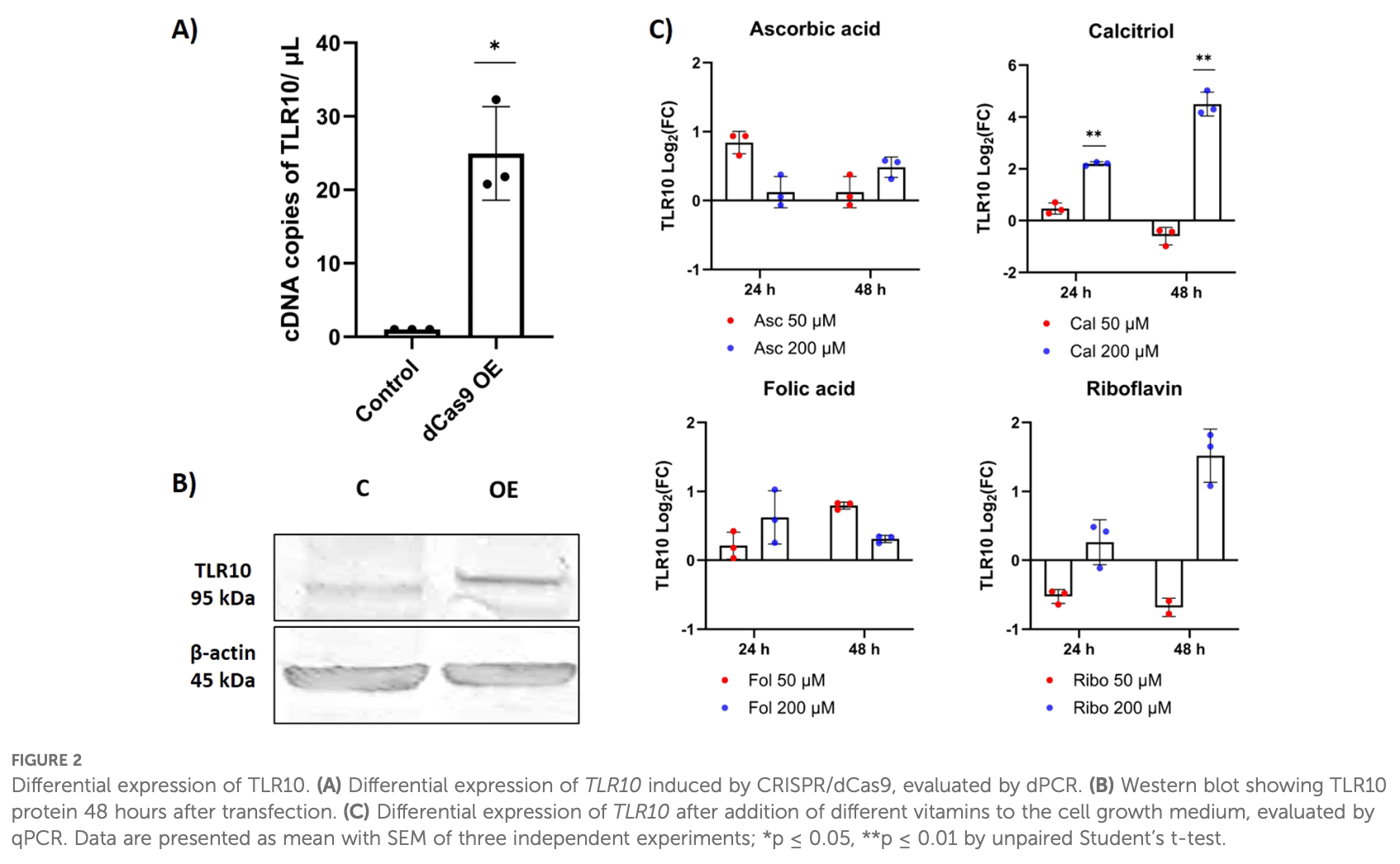
In vitro study showing that overexpression of TLR10 in A549 lung epithelial cells immunostimulated with SARS-CoV-2 S and N proteins mainly downregulated proinflammatory cytokines and interferons and affected gene expression in cocultured THP-1 monocytes. Authors suggest TLR10 could mediate the extent of SARS-CoV-2 infection by downregulating release of inflammatory cytokines and chemokines such as CXCL10, IL6, IL8, and IFNb. Supplementing A549 cell growth medium with calcitriol increased TLR10 expression 4-fold after 24 hours and 16-fold after 48 hours at 200 µM concentration. Modulating TLR10 expression may be beneficial for severe COVID-19 patients, in whom excessive inflammation leading to acute respiratory distress syndrome (ARDS) is a key feature.
29 preclinical studies support the efficacy of vitamin D for COVID-19:
Vitamin D has been identified by the European Food Safety Authority (EFSA) as having sufficient evidence for a causal relationship between intake and optimal immune system function27-30.
Vitamin D inhibits SARS-CoV-2 replication in vitro17,24, mitigates lung inflammation, damage, and lethality in mice with an MHV-3 model for β-CoV respiratory infections17,24, reduces SARS-CoV-2 replication in nasal epithelial cells via increased type I interferon expression20, downregulates proinflammatory cytokines IL-1β and TNF-α in SARS-CoV-2 spike protein-stimulated cells16, attenuates nucleocapsid protein-induced hyperinflammation by inactivating the NLRP3 inflammasome through the VDR-BRCC3 signaling pathway21, may be neuroprotective by protecting the blood-brain barrier, reducing neuroinflammation, and via immunomodulatory effects31, may mitigate hyperinflammation and cytokine storm by upregulating TLR10 expression which downregulates proinflammatory cytokines13, downregulates ACE2 and TMPRSS2 in human trophoblasts and minimizes spike protein-induced inflammation19, may minimize cytokine storm by dampening excessive cytokine production2, may suppress viral entry and replication via LL-37 induction11,12, and minimizes platelet aggregation mediated by SARS-CoV-2 spike protein via inhibiting integrin αIIbβ3 outside-in signaling15.
Cholecalciferol and calcifediol directly bind two allosteric pockets on the SARS-CoV-2 Spike RBD, bias the trimer toward a closed state, weaken ACE2 engagement, and reduce viral entry in cell models1.
Calcitriol may destabilize the Spike protein architecture and inhibit IL-17R dimerization, blocking viral entry and mitigating hyperinflammatory cytokine storm32.
Vitamin D improves regulatory immune cell levels and control of proinflammatory cytokines in severe COVID-1933.
Calcifediol inhibits SARS-CoV-2 papain-like protease (PLpro), a critical enzyme for viral replication14.
Symptomatic COVID-19 is associated with a lower frequency of natural killer (NK) cells and vitamin D has been shown to improve NK cell activity34,35.
1.
García-Marín et al., Exploring SARS-CoV-2 Spike RBD Pockets as Targets for Generic Drugs: A Combined Computational, Biophysical, and Biological Approach, ACS Omega, doi:10.1021/acsomega.5c05175.
2.
Alzahrani, A., A new investigation into the molecular mechanism of cholecalciferol towards reducing cytokines storm, Octahedron Drug Research, doi:10.21608/odr.2024.308273.1043.
3.
Haque et al., Exploring potential therapeutic candidates against COVID-19: a molecular docking study, Discover Molecules, doi:10.1007/s44345-024-00005-5.
4.
Morales-Bayuelo et al., New findings on ligand series used as SARS-CoV-2 virus inhibitors within the frameworks of molecular docking, molecular quantum similarity and chemical reactivity indices, F1000Research, doi:10.12688/f1000research.123550.3.
5.
Chellasamy et al., Docking and molecular dynamics studies of human ezrin protein with a modelled SARS-CoV-2 endodomain and their interaction with potential invasion inhibitors, Journal of King Saud University - Science, doi:10.1016/j.jksus.2022.102277.
6.
Pandya et al., Unravelling Vitamin B12 as a potential inhibitor against SARS-CoV-2: A computational approach, Informatics in Medicine Unlocked, doi:10.1016/j.imu.2022.100951.
7.
Mansouri et al., The impact of calcitriol and estradiol on the SARS-CoV-2 biological activity: a molecular modeling approach, Scientific Reports, doi:10.1038/s41598-022-04778-y.
8.
Song et al., Vitamin D3 and its hydroxyderivatives as promising drugs against COVID-19: a computational study, Journal of Biomolecular Structure and Dynamics, doi:10.1080/07391102.2021.1964601.
9.
Qayyum et al., Vitamin D and lumisterol novel metabolites can inhibit SARS-CoV-2 replication machinery enzymes, Endocrinology and Metabolism, doi:10.1152/ajpendo.00174.2021.
10.
Al-Mazaideh et al., Vitamin D is a New Promising Inhibitor to the Main Protease (Mpro) of COVID-19 by Molecular Docking, Journal of Pharmaceutical Research International, doi:10.9734/jpri/2021/v33i29B31603.
11.
Roth et al., Vitamin D-inducible antimicrobial peptide LL-37 binds SARS-CoV-2 Spike and accessory proteins ORF7a and ORF8, Frontiers in Cellular and Infection Microbiology, doi:10.3389/fcimb.2025.1671738.
12.
Vercellino et al., Influence of Sex and 1,25α Dihydroxyvitamin D3 on SARS-CoV-2 Infection and Viral Entry, Pathogens, doi:10.3390/pathogens14080765.
13.
Knez et al., TLR10 overexpression modulates immune response in A549 lung epithelial cells challenged with SARS-CoV-2 S and N proteins, Frontiers in Immunology, doi:10.3389/fimmu.2024.1490478.
14.
Chen et al., In Vitro Characterization of Inhibition Function of Calcifediol to the Protease Activity of SARS-COV-2 PLpro, Journal of Medical Virology, doi:10.1002/jmv.70085.
15.
Wang et al., 1,25‐Dihydroxyvitamin D3 attenuates platelet aggregation potentiated by SARS‐CoV‐2 spike protein via inhibiting integrin αIIbβ3 outside‐in signaling, Cell Biochemistry and Function, doi:10.1002/cbf.4039.
16.
Alcalá-Santiago et al., Disentangling the Immunomodulatory Effects of Vitamin D on the SARS-CoV-2 Virus by In Vitro Approaches, The 14th European Nutrition Conference FENS 2023, doi:10.3390/proceedings2023091415.
17.
Campolina-Silva et al., Dietary Vitamin D Mitigates Coronavirus-Induced Lung Inflammation and Damage in Mice, Viruses, doi:10.3390/v15122434.
18.
Moatasim et al., Potent Antiviral Activity of Vitamin B12 against Severe Acute Respiratory Syndrome Coronavirus 2, Middle East Respiratory Syndrome Coronavirus, and Human Coronavirus 229E, Microorganisms, doi:10.3390/microorganisms11112777.
19.
Vargas-Castro et al., Calcitriol prevents SARS-CoV spike-induced inflammation in human trophoblasts through downregulating ACE2 and TMPRSS2 expression, The Journal of Steroid Biochemistry and Molecular Biology, doi:10.1016/j.jsbmb.2024.106625.
20.
Sposito et al., Age differential CD13 and interferon expression in airway epithelia affect SARS-CoV-2 infection - effects of vitamin D, Mucosal Immunology, doi:10.1016/j.mucimm.2023.08.002.
21.
Chen (B) et al., Vitamin D3 attenuates SARS‐CoV‐2 nucleocapsid protein‐caused hyperinflammation by inactivating the NLRP3 inflammasome through the VDR‐BRCC3 signaling pathway in vitro and in vivo, MedComm, doi:10.1002/mco2.318.
22.
Rybakovsky et al., Calcitriol modifies tight junctions, improves barrier function, and reduces TNF‐α‐induced barrier leak in the human lung‐derived epithelial cell culture model, 16HBE 14o‐, Physiological Reports, doi:10.14814/phy2.15592.
23.
DiGuilio et al., The multiphasic TNF-α-induced compromise of Calu-3 airway epithelial barrier function, Experimental Lung Research, doi:10.1080/01902148.2023.2193637.
24.
Pickard et al., Discovery of re-purposed drugs that slow SARS-CoV-2 replication in human cells, PLOS Pathogens, doi:10.1371/journal.ppat.1009840.
25.
Mok et al., Calcitriol, the active form of vitamin D, is a promising candidate for COVID-19 prophylaxis, bioRxiv, doi:10.1101/2020.06.21.162396.
26.
Fernandes de Souza et al., Lung Inflammation Induced by Inactivated SARS-CoV-2 in C57BL/6 Female Mice Is Controlled by Intranasal Instillation of Vitamin D, Cells, doi:10.3390/cells12071092.
27.
Galmés et al., Suboptimal Consumption of Relevant Immune System Micronutrients Is Associated with a Worse Impact of COVID-19 in Spanish Populations, Nutrients, doi:10.3390/nu14112254.
28.
Galmés (B) et al., Current State of Evidence: Influence of Nutritional and Nutrigenetic Factors on Immunity in the COVID-19 Pandemic Framework, Nutrients, doi:10.3390/nu12092738.
29.
EFSA, Scientific Opinion on the substantiation of a health claim related to vitamin D and contribution to the normal function of the immune system pursuant to Article 14 of Regulation (EC) No 1924/2006, EFSA Journal, doi:10.2903/j.efsa.2015.4096.
30.
EFSA (B), Scientific Opinion on the substantiation of health claims related to vitamin D and normal function of the immune system and inflammatory response (ID 154, 159), maintenance of normal muscle function (ID 155) and maintenance of normal cardiovascular function (ID 159) pursuant to Article 13(1) of Regulation (E, EFSA Journal, doi:10.2903/j.efsa.2010.1468.
31.
Gotelli et al., Understanding the immune-endocrine effects of vitamin D in SARS-CoV-2 infection: a role in protecting against neurodamage?, Neuroimmunomodulation, doi:10.1159/000533286.
32.
Fadel et al., Targeting asparagine and cysteine in SARS-CoV-2 variants and human pro-inflammatory mediators to alleviate COVID-19 severity; a cross-section and in-silico study, Scientific Reports, doi:10.1038/s41598-025-19359-y.
33.
Saheb Sharif-Askari et al., Increased blood immune regulatory cells in severe COVID-19 with autoantibodies to type I interferons, Scientific Reports, doi:10.1038/s41598-023-43675-w.
Knez et al., 20 Jan 2025, peer-reviewed, 3 authors.
Contact: jernej.ogorevc@bf.uni-lj.si.
In vitro studies are an important part of preclinical research, however results may be very different in vivo.
TLR10 overexpression modulates immune response in A549 lung epithelial cells challenged with SARS-CoV-2 S and N proteins
Frontiers in Immunology, doi:10.3389/fimmu.2024.1490478
Toll-like receptors (TLRs) play an important role in the recognition of viral particles and activation of the innate immune system, but their role in SARS-CoV-2 infection is still poorly characterized. In the present study, we investigated the role of Toll-like receptor 10 (TLR10) in modulating the immune response during SARS-CoV-2 infection. The results showed that overexpression of TLR10 in A549 lung epithelial cells, immunostimulated with SARS-CoV-2 proteins S and N mainly downregulated proinflammatory cytokines and interferons and affected gene expression in the cocultured THP-1 monocytes. Our results suggest that TLR10 could mediate the extent of SARS-CoV-2 infection by downregulating the release of inflammatory cytokines and chemokines such as CXCL10, IL6, IL8, and IFNb. Modulation of TLR10 expression could have implications for the treatment of patients with severe COVID-19, in whom excessive inflammation leading to the development of acute respiratory distress syndrome (ARDS) is a key feature. However, further research is needed to fully understand the impact of modulating TLR10 expression on the antiviral response and the overall balance of the immune response during SARS-CoV-2 infection.
Ethics statement Ethical approval was not required for the studies on humans in accordance with the local legislation and institutional requirements because only commercially available established cell lines were used. Ethical approval was not required for the
Conflict of interest The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher's note All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fimmu.2024.1490478/ full#supplementary-material
References
Aboudounya, Heads, COVID-19 and toll-like receptor 4 (TLR4): SARS-coV-2 may bind and activate TLR4 to increase ACE2 expression, facilitating entry and causing hyperinflammation, Mediators of Inflammation, doi:10.1155/2021/8874339
Astuti, Ysrafil, Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2): An overview of viral structure and host response, Diabetes Metab Syndrome: Clin Res Rev, doi:10.1016/j.dsx.2020.04.020
Bals, Hiemstra, Innate immunity in the lung: How epithelial cells fight against respiratory pathogens, Eur Respir J, doi:10.1183/09031936.03.00098803
Chavez, Wiegand, Ter-Ovanesyan, Braff, Davidsohn, Highlyefficient Cas9-mediated transcriptional programming, Nature, doi:10.1038/nmeth.3312
Chow, Chang, Chan, Inhalation delivery technology for genomeediting of respiratory diseases, Advanced Drug Delivery Rev, doi:10.1016/j.addr.2020.06.001
Coperchini, Chiovato, Croce, Magri, Rotondi, The cytokine storm in COVID-19: An overview of the involvement of the chemokine/chemokine-receptor system, Cytokine Growth Factor Rev, doi:10.1016/j.cytogfr.2020.05.003
Coperchini, Chiovato, Croce, The cytokine storm in COVID-19: An overview of the involvement of the chemokine/chemokine-receptor system, Frontiers in Immunology, doi:10.3389/fimmu.2021.668507
Duell, Cripps, Schembri, Ulett, Epithelial cell coculture models for studying infectious diseases: Benefits and limitations, J Biomed Biotechnol, doi:10.1155/2011/852419
Fore, Indriputri, Mamutse, Nugraha, TLR10 and its unique antiinflammatory properties and potential use as a target in therapeutics, Immune Network, doi:10.4110/in.2020.20.e21
Frank, Wallach, Raden, Hinkelmann, Brehm et al., Distinct SARS-CoV-2 RNA fragments activate Toll-like receptors 7 and 8 and induce cytokine release from human macrophages and microglia, Frontiers Immunol, doi:10.3389/fimmu.2022.1066456
Gedeon, Bokes, Delayed protein synthesis reduces the correlation between mRNA and protein fluctuations, Biophysical Journal
Gschwandtner, Derler, Midwood, More than just attractive: how CCL2 influences myeloid cell behavior beyond chemotaxis, Front Immunol, doi:10.3389/fimmu.2019.02759
Guan, Ranoa, Jiang, Mutha, Li et al., Human TLRs 10 and 1 share common mechanisms of innate immune sensing but not signaling, J Immunol, doi:10.4049/jimmunol.0901888
Hadjadj, Yatim, Barnabei, Corneau, Boussier, Impaired type I interferon activity and inflammatory responses in severe COVID-19 patients, Science, doi:10.1126/science.abc6027
Hasan, Chaffois, Gaillard, Saulnier, Merck et al., Human TLR10 is a functional receptor, expressed by B cells, J Immunol, doi:10.4049/jimmunol.174.5.2942
Hasan, Dollet, Vlach, Differential induction of gene promoter constructs by constitutively active human TLRs, Biochem Biophys Res Commun, doi:10.1016/j.bbrc.2004.06.134
Haystead, Lee, Cho, Gullickson, Hughes et al., Investigation of SARS-CoV-2 individual proteins reveals the in vitro and in vivo immunogenicity of membrane protein, Sci Rep, doi:10.1038/s41598-023-49077-2
Henrick, Yao, Zahoor, Abimiku, Osawe et al., TLR10 senses HIV-1 proteins and significantly enhances HIV-1 infection, Front Immunol, doi:10.3389/fimmu.2019.00482
Hou, Xiao, Tang, Xie, Diversity of macrophages in lung homeostasis and diseases, Front Immunol, doi:10.3389/fimmu.2021.753940
Huang, Wang, Li, Ren, Zhao et al., Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China, The Lancet, doi:10.1016/S0140-6736(20)30183-5
Josićk, Loṕez, Ott, Shiau, Bennett, Stochastic delay accelerates signaling in gene networks, PloS Computational Biology, doi:10.1371/journal.pcbi.1002264
Karki, Sharma, Tuladhar, Williams, Zalduondo et al., Synergism of TNF-a and IFN-g Triggers inflammatory cell death, tissue damage, and mortality in SARS-coV-2 infection and cytokine shock syndromes, Cell, doi:10.1016/j.cell.2020.11.025
Khan, Shafiei, Longoria, Schoggins, Savani et al., SARS-CoV-2 spike protein induces inflammation via TLR2-dependent activation of the NF-kB pathway, ELife, doi:10.7554/eLife.68563
Knez, Narat, Ogorevc, Differential gene expression induced by different TLR agonists in A549 lung epithelial cells is modulated by CRISPR activation of TLR10, Biomolecules, doi:10.3390/biom13010019
Kümmel, Krumbein, Fragkou, Hünerbein, Reiter et al., Vitamin D supplementation for the treatment of COVID-19: A systematic review and meta-analysis of randomized controlled trials, Front Immunol, doi:10.3389/fimmu.2022.1023903
Lee, Kok, Jaume, Cheung, Yip et al., Toll-like receptor 10 is involved in induction of innate immune responses to influenza virus infection, Proc Natl Acad Sci, doi:10.1073/pnas.1324266111
Lee, Lee, Kong, A survey of genetic variants in SARS-CoV-2 interacting domains of ACE2, TMPRSS2 and TLR3/7/8 across populations, Infection, Genetics and Evolution, doi:10.1016/j.meegid.2020.104507
Lee, Yip, Yan, Jin, Wei et al., Recognition of doublestranded RNA and regulation of interferon pathway by toll-like receptor 10, Front Immunol, doi:10.3389/fimmu.2018.00516
Li, Liao, Wang, Tan, Luo et al., The ORF6, ORF8 and nucleocapsid proteins of SARS-CoV-2 inhibit type I interferon signaling pathway, Virus Res, doi:10.1016/j.virusres.2020.198074
Liu, Yin, Chen, Wu, Zheng et al., LPS-induced proinflammatory cytokine expression in human airway epithelial cells and macrophages via NF-kB, STAT3 or AP-1 activation, Mol Med Rep, doi:10.3892/mmr.2018.8542
Livak, Schmittgen, Analysis of gene expression data using PCR and the 2-DDCT method, Methods, doi:10.1006/meth.2001.1262
Mabrey, Morrell, Wurfel, TLRs in COVID-19: How they drive immunopathology and the rationale for modulation, Innate Immun, doi:10.1177/17534259211051364
Majidi, Rabbani, Gholami, Gholamalizadeh, Bourbour et al., The effect of vitamin C on pathological parameters and survival duration of critically ill coronavirus disease 2019 patients: A randomized clinical trial, Front Immunol, doi:10.3389/fimmu.2021.717816
Onofrio, Caraglia, Facchini, Margherita, Placido et al., Toll-like receptors and COVID-19: A two-faced story with an exciting ending, Future Sci OA, doi:10.1073/pnas.1410293111
Oosting, Cheng, Bolscher, Vestering-Stenger, Plantinga et al., Human TLR10 is an anti-inflammatory pattern-recognition receptor, Proceedings of the National of Sciences of the United States of America, doi:10.1073/pnas.1410293111
Paolini, Borella, Biasi, Neroni, Mattioli et al., Cell death in coronavirus infections: uncovering its role during COVID-19, Cells, doi:10.3390/cells10071585
Poulas, Farsalinos, Zanidis, Activation of TLR7 and innate immunity as an efficient method against COVID-19 pandemic: imiquimod as a potential therapy, Nat Rev Immunol, doi:10.1038/nri3547
Regan, Nally, Carmody, Houston, Shanahan et al., Identification of TLR10 as a key mediator of the inflammatory response to listeria monocytogenes in intestinal epithelial cells and macrophages, J Immunol, doi:10.4049/jimmunol.1203245
Ropa, Cooper, Capitano, Van't Hof, Broxmeyer, Human hematopoietic stem, progenitor, and immune cells respond ex vivo to SARS-coV-2 spike protein, Stem Cell Rev Rep, doi:10.1007/s12015-020-10056-z
Sha, Truong-Tran, Plitt, Beck, Schleimer, Activation of airway epithelial cells by toll-like receptor agonists, Am J Respir Cell Mol Biol, doi:10.1165/rcmb.2003-0388OC
Sinopoli, Sciurti, Isonne, Santoro, Baccolini, The efficacy of multivitamin, vitamin A, vitamin B, vitamin C, and vitamin D supplements in the prevention and management of COVID-19 and long-COVID: an updated systematic review and meta-analysis of randomized clinical trials, Nutrients, doi:10.3390/nu16091345
Soy, Keser, Atagündüz, Tabak, Atagündüz et al., Cytokine storm in COVID-19: pathogenesis and overview of anti-inflammatory agents used in treatment, Clin Rheumatol, doi:10.1007/s10067-020-05190-5
Stappers, Oosting, Ioana, Reimnitz, Mouton et al., Genetic variation in TLR10, an inhibitory toll-like receptor, influences susceptibility to complicated skin and skin structure infections, J Infect Dis, doi:10.1093/infdis/jiv229
Verma, Jung, Kim, 1,25-Dihydroxyvitamin D3 up-regulates TLR10 while down-regulating TLR2, 4, and 5 in human monocyte THP-1, J Steroid Biochem Mol Biol, doi:10.1016/j.jsbmb.2013.12.012
Verma, Kim, 1,25-dihydroxyvitamin D3 facilitates M2 polarization and upregulates TLR10 expression on human microglial cells, Neuroimmunomodulation
Wang, Jiang, Chen, Montaner, Cytokine storm and leukocyte changes in mild versus severe SARS-CoV-2 infection: Review of 3939 COVID-19 patients in China and emerging pathogenesis and therapy concepts, J Leukoc Biol, doi:10.1002/JLB.3COVR0520-272R
Zheng, Karki, Williams, Yang, Fitzpatrick et al., TLR2 senses the SARS-CoV-2 envelope protein to produce inflammatory cytokines, Nat Immunol, doi:10.1038/s41590-021-00937-x
DOI record:
{
"DOI": "10.3389/fimmu.2024.1490478",
"ISSN": [
"1664-3224"
],
"URL": "http://dx.doi.org/10.3389/fimmu.2024.1490478",
"abstract": "<jats:p>Toll-like receptors (TLRs) play an important role in the recognition of viral particles and activation of the innate immune system, but their role in SARS-CoV-2 infection is still poorly characterized. In the present study, we investigated the role of Toll-like receptor 10 (TLR10) in modulating the immune response during SARS-CoV-2 infection. The results showed that overexpression of <jats:italic>TLR10</jats:italic> in A549 lung epithelial cells, immunostimulated with SARS-CoV-2 proteins S and N mainly downregulated proinflammatory cytokines and interferons and affected gene expression in the cocultured THP-1 monocytes. Our results suggest that TLR10 could mediate the extent of SARS-CoV-2 infection by downregulating the release of inflammatory cytokines and chemokines such as <jats:italic>CXCL10</jats:italic>, <jats:italic>IL6</jats:italic>, <jats:italic>IL8</jats:italic>, and <jats:italic>IFNβ</jats:italic>. Modulation of <jats:italic>TLR10</jats:italic> expression could have implications for the treatment of patients with severe COVID-19, in whom excessive inflammation leading to the development of acute respiratory distress syndrome (ARDS) is a key feature. However, further research is needed to fully understand the impact of modulating <jats:italic>TLR10</jats:italic> expression on the antiviral response and the overall balance of the immune response during SARS-CoV-2 infection.</jats:p>",
"alternative-id": [
"10.3389/fimmu.2024.1490478"
],
"author": [
{
"affiliation": [],
"family": "Knez",
"given": "Špela",
"sequence": "first"
},
{
"affiliation": [],
"family": "Narat",
"given": "Mojca",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ogorevc",
"given": "Jernej",
"sequence": "additional"
}
],
"container-title": "Frontiers in Immunology",
"container-title-short": "Front. Immunol.",
"content-domain": {
"crossmark-restriction": true,
"domain": [
"frontiersin.org"
]
},
"created": {
"date-parts": [
[
2025,
1,
20
]
],
"date-time": "2025-01-20T05:24:27Z",
"timestamp": 1737350667000
},
"deposited": {
"date-parts": [
[
2025,
1,
20
]
],
"date-time": "2025-01-20T05:24:29Z",
"timestamp": 1737350669000
},
"indexed": {
"date-parts": [
[
2025,
1,
21
]
],
"date-time": "2025-01-21T05:03:09Z",
"timestamp": 1737435789399,
"version": "3.33.0"
},
"is-referenced-by-count": 0,
"issued": {
"date-parts": [
[
2025,
1,
20
]
]
},
"license": [
{
"URL": "https://creativecommons.org/licenses/by/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2025,
1,
20
]
],
"date-time": "2025-01-20T00:00:00Z",
"timestamp": 1737331200000
}
}
],
"link": [
{
"URL": "https://www.frontiersin.org/articles/10.3389/fimmu.2024.1490478/full",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "1965",
"original-title": [],
"prefix": "10.3389",
"published": {
"date-parts": [
[
2025,
1,
20
]
]
},
"published-online": {
"date-parts": [
[
2025,
1,
20
]
]
},
"publisher": "Frontiers Media SA",
"reference": [
{
"DOI": "10.1016/j.dsx.2020.04.020",
"article-title": "Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2): An overview of viral structure and host response",
"author": "Astuti",
"doi-asserted-by": "publisher",
"journal-title": "Diabetes Metab Syndrome: Clin Res Rev",
"key": "B1",
"volume": "14",
"year": "2020"
},
{
"DOI": "10.1002/JLB.3COVR0520-272R",
"article-title": "Cytokine storm and leukocyte changes in mild versus severe SARS-CoV-2 infection: Review of 3939 COVID-19 patients in China and emerging pathogenesis and therapy concepts",
"author": "Wang",
"doi-asserted-by": "publisher",
"journal-title": "J Leukoc Biol",
"key": "B2",
"volume": "108",
"year": "2020"
},
{
"DOI": "10.1126/science.abc6027",
"article-title": "Impaired type I interferon activity and inflammatory responses in severe COVID-19 patients",
"author": "Hadjadj",
"doi-asserted-by": "publisher",
"journal-title": "Science",
"key": "B3",
"volume": "724",
"year": "2020"
},
{
"DOI": "10.3892/mmr.2018.8542",
"article-title": "LPS-induced proinflammatory cytokine expression in human airway epithelial cells and macrophages via NF-κB, STAT3 or AP-1 activation",
"author": "Liu",
"doi-asserted-by": "publisher",
"journal-title": "Mol Med Rep",
"key": "B4",
"volume": "17",
"year": "2018"
},
{
"DOI": "10.1016/S0140-6736(20)30183-5",
"article-title": "Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China",
"author": "Huang",
"doi-asserted-by": "publisher",
"first-page": "497",
"journal-title": "The Lancet",
"key": "B5",
"volume": "395",
"year": "2020"
},
{
"DOI": "10.3389/fimmu.2021.668507",
"article-title": "The cytokine storm in COVID-19: An overview of the involvement of the chemokine/chemokine-receptor system",
"author": "Coperchini",
"doi-asserted-by": "publisher",
"journal-title": "Frontiers in Immunology",
"key": "B6",
"volume": "12",
"year": "2021"
},
{
"DOI": "10.1183/09031936.03.00098803",
"article-title": "Innate immunity in the lung: How epithelial cells fight against respiratory pathogens",
"author": "Bals",
"doi-asserted-by": "publisher",
"journal-title": "Eur Respir J",
"key": "B7",
"volume": "23",
"year": "2004"
},
{
"DOI": "10.1073/pnas.1410293111",
"article-title": "Toll-like receptors and COVID-19: A two-faced story with an exciting ending",
"author": "Onofrio",
"doi-asserted-by": "publisher",
"journal-title": "Future Sci OA",
"key": "B8",
"volume": "6",
"year": "2020"
},
{
"DOI": "10.1038/nri3547",
"article-title": "Activation of TLR7 and innate immunity as an efficient method against COVID-19 pandemic: imiquimod as a potential therapy",
"author": "Poulas",
"doi-asserted-by": "publisher",
"journal-title": "Nat Rev Immunol",
"key": "B9",
"volume": "13",
"year": "2020"
},
{
"DOI": "10.3389/fimmu.2022.1066456",
"article-title": "Distinct SARS-CoV-2 RNA fragments activate Toll-like receptors 7 and 8 and induce cytokine release from human macrophages and microglia",
"author": "Frank",
"doi-asserted-by": "publisher",
"first-page": "1",
"journal-title": "Frontiers Immunol",
"key": "B10",
"volume": "13",
"year": "2023"
},
{
"DOI": "10.1016/j.meegid.2020.104507",
"article-title": "A survey of genetic variants in SARS-CoV-2 interacting domains of ACE2, TMPRSS2 and TLR3/7/8 across populations",
"author": "Lee",
"doi-asserted-by": "publisher",
"journal-title": "Infection, Genetics and Evolution",
"key": "B11",
"volume": "85",
"year": "2020"
},
{
"DOI": "10.7554/eLife.68563",
"article-title": "SARS-CoV-2 spike protein induces inflammation via TLR2-dependent activation of the NF-κB pathway",
"author": "Khan",
"doi-asserted-by": "publisher",
"first-page": "1",
"journal-title": "ELife",
"key": "B12",
"volume": "10",
"year": "2021"
},
{
"DOI": "10.1155/2021/8874339",
"article-title": "COVID-19 and toll-like receptor 4 (TLR4): SARS-coV-2 may bind and activate TLR4 to increase ACE2 expression, facilitating entry and causing hyperinflammation",
"author": "Aboudounya",
"doi-asserted-by": "publisher",
"journal-title": "Mediators of Inflammation",
"key": "B13",
"volume": "14",
"year": "2021"
},
{
"DOI": "10.1038/s41590-021-00937-x",
"article-title": "TLR2 senses the SARS-CoV-2 envelope protein to produce inflammatory cytokines",
"author": "Zheng",
"doi-asserted-by": "publisher",
"journal-title": "Nat Immunol",
"key": "B14",
"volume": "22",
"year": "2021"
},
{
"DOI": "10.1038/s41598-023-49077-2",
"article-title": "Investigation of SARS-CoV-2 individual proteins reveals the in vitro and in vivo immunogenicity of membrane protein",
"author": "Haystead",
"doi-asserted-by": "publisher",
"first-page": "1",
"journal-title": "Sci Rep",
"key": "B15",
"volume": "13",
"year": "2023"
},
{
"DOI": "10.4110/in.2020.20.e21",
"article-title": "TLR10 and its unique anti-inflammatory properties and potential use as a target in therapeutics",
"author": "Fore",
"doi-asserted-by": "publisher",
"first-page": "1",
"journal-title": "Immune Network",
"key": "B16",
"volume": "20",
"year": "2020"
},
{
"DOI": "10.1165/rcmb.2003-0388OC",
"article-title": "Activation of airway epithelial cells by toll-like receptor agonists",
"author": "Sha",
"doi-asserted-by": "publisher",
"journal-title": "Am J Respir Cell Mol Biol",
"key": "B17",
"volume": "31",
"year": "2004"
},
{
"DOI": "10.3390/biom13010019",
"article-title": "Differential gene expression induced by different TLR agonists in A549 lung epithelial cells is modulated by CRISPR activation of TLR10",
"author": "Knez",
"doi-asserted-by": "publisher",
"first-page": "19",
"journal-title": "Biomolecules",
"key": "B18",
"volume": "13",
"year": "2023"
},
{
"DOI": "10.1159/000444300",
"article-title": "1,25-dihydroxyvitamin D3 facilitates M2 polarization and upregulates TLR10 expression on human microglial cells",
"author": "Verma",
"doi-asserted-by": "crossref",
"journal-title": "Neuroimmunomodulation",
"key": "B19",
"volume": "23",
"year": "2016"
},
{
"DOI": "10.3390/nu16091345",
"article-title": "The efficacy of multivitamin, vitamin A, vitamin B, vitamin C, and vitamin D supplements in the prevention and management of COVID-19 and long-COVID: an updated systematic review and meta-analysis of randomized clinical trials",
"author": "Sinopoli",
"doi-asserted-by": "publisher",
"journal-title": "Nutrients",
"key": "B20",
"volume": "16",
"year": "2024"
},
{
"DOI": "10.3389/fimmu.2021.717816",
"article-title": "The effect of vitamin C on pathological parameters and survival duration of critically ill coronavirus disease 2019 patients: A randomized clinical trial",
"author": "Majidi",
"doi-asserted-by": "publisher",
"journal-title": "Front Immunol",
"key": "B21",
"volume": "12",
"year": "2021"
},
{
"DOI": "10.3389/fimmu.2022.1023903",
"article-title": "Vitamin D supplementation for the treatment of COVID-19: A systematic review and meta-analysis of randomized controlled trials",
"author": "Kümmel",
"doi-asserted-by": "publisher",
"journal-title": "Front Immunol",
"key": "B22",
"volume": "13",
"year": "2022"
},
{
"DOI": "10.1038/nmeth.3312",
"article-title": "Highly-efficient Cas9-mediated transcriptional programming",
"author": "Chavez",
"doi-asserted-by": "publisher",
"journal-title": "Nature",
"key": "B23",
"volume": "12",
"year": "2015"
},
{
"DOI": "10.1006/meth.2001.1262",
"article-title": "Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCT method",
"author": "Livak",
"doi-asserted-by": "publisher",
"journal-title": "Methods",
"key": "B24",
"volume": "25",
"year": "2001"
},
{
"DOI": "10.4049/jimmunol.0901888",
"article-title": "Human TLRs 10 and 1 share common mechanisms of innate immune sensing but not signaling",
"author": "Guan",
"doi-asserted-by": "publisher",
"journal-title": "J Immunol",
"key": "B25",
"volume": "184",
"year": "2010"
},
{
"DOI": "10.4049/jimmunol.1203245",
"article-title": "Identification of TLR10 as a key mediator of the inflammatory response to listeria monocytogenes in intestinal epithelial cells and macrophages",
"author": "Regan",
"doi-asserted-by": "publisher",
"journal-title": "J Immunol",
"key": "B26",
"volume": "191",
"year": "2013"
},
{
"DOI": "10.4049/jimmunol.174.5.2942",
"article-title": "Human TLR10 is a functional receptor, expressed by B cells",
"author": "Hasan",
"doi-asserted-by": "publisher",
"journal-title": "J Immunol",
"key": "B27",
"volume": "174",
"year": "2005"
},
{
"DOI": "10.1073/pnas.1410293111",
"article-title": "Human TLR10 is an anti-inflammatory pattern-recognition receptor",
"author": "Oosting",
"doi-asserted-by": "publisher",
"journal-title": "Proceedings of the National Academy of Sciences of the United States of America",
"key": "B28",
"volume": "111",
"year": "2014"
},
{
"DOI": "10.1093/infdis/jiv229",
"article-title": "Genetic variation in TLR10, an inhibitory toll-like receptor, influences susceptibility to complicated skin and skin structure infections",
"author": "Stappers",
"doi-asserted-by": "publisher",
"journal-title": "J Infect Dis",
"key": "B29",
"volume": "212",
"year": "2015"
},
{
"DOI": "10.1016/j.bbrc.2004.06.134",
"article-title": "Differential induction of gene promoter constructs by constitutively active human TLRs",
"author": "Hasan",
"doi-asserted-by": "publisher",
"journal-title": "Biochem Biophys Res Commun",
"key": "B30",
"volume": "321",
"year": "2004"
},
{
"DOI": "10.3389/fimmu.2018.00516",
"article-title": "Recognition of double-stranded RNA and regulation of interferon pathway by toll-like receptor 10",
"author": "Lee",
"doi-asserted-by": "publisher",
"journal-title": "Front Immunol",
"key": "B31",
"volume": "9",
"year": "2018"
},
{
"DOI": "10.3389/fimmu.2019.00482",
"article-title": "TLR10 senses HIV-1 proteins and significantly enhances HIV-1 infection",
"author": "Henrick",
"doi-asserted-by": "publisher",
"journal-title": "Front Immunol",
"key": "B32",
"volume": "10",
"year": "2019"
},
{
"DOI": "10.1073/pnas.1324266111",
"article-title": "Toll-like receptor 10 is involved in induction of innate immune responses to influenza virus infection",
"author": "Lee",
"doi-asserted-by": "publisher",
"journal-title": "Proc Natl Acad Sci United States America",
"key": "B33",
"volume": "111",
"year": "2014"
},
{
"DOI": "10.1016/j.jsbmb.2013.12.012",
"article-title": "1,25-Dihydroxyvitamin D3 up-regulates TLR10 while down-regulating TLR2, 4, and 5 in human monocyte THP-1",
"author": "Verma",
"doi-asserted-by": "publisher",
"first-page": "1",
"journal-title": "J Steroid Biochem Mol Biol",
"key": "B34",
"volume": "141",
"year": "2014"
},
{
"DOI": "10.1016/j.cytogfr.2020.05.003",
"article-title": "The cytokine storm in COVID-19: An overview of the involvement of the chemokine/chemokine-receptor system",
"author": "Coperchini",
"doi-asserted-by": "publisher",
"first-page": "25",
"journal-title": "Cytokine Growth Factor Rev",
"key": "B35",
"volume": "53",
"year": "2020"
},
{
"DOI": "10.1177/17534259211051364",
"article-title": "TLRs in COVID-19: How they drive immunopathology and the rationale for modulation",
"author": "Mabrey",
"doi-asserted-by": "publisher",
"journal-title": "Innate Immun",
"key": "B36",
"volume": "27",
"year": "2021"
},
{
"DOI": "10.1016/j.cell.2020.11.025",
"article-title": "Synergism of TNF-α and IFN-γ Triggers inflammatory cell death, tissue damage, and mortality in SARS-coV-2 infection and cytokine shock syndromes",
"author": "Karki",
"doi-asserted-by": "publisher",
"first-page": "149",
"journal-title": "Cell",
"key": "B37",
"volume": "184",
"year": "2021"
},
{
"DOI": "10.1007/s10067-020-05190-5",
"article-title": "Cytokine storm in COVID-19: pathogenesis and overview of anti-inflammatory agents used in treatment",
"author": "Soy",
"doi-asserted-by": "publisher",
"journal-title": "Clin Rheumatol",
"key": "B38",
"volume": "39",
"year": "2020"
},
{
"DOI": "10.3389/fimmu.2021.753940",
"article-title": "Diversity of macrophages in lung homeostasis and diseases",
"author": "Hou",
"doi-asserted-by": "publisher",
"journal-title": "Front Immunol",
"key": "B39",
"volume": "12",
"year": "2021"
},
{
"DOI": "10.1007/s12015-020-10056-z",
"article-title": "Human hematopoietic stem, progenitor, and immune cells respond ex vivo to SARS-coV-2 spike protein",
"author": "Ropa",
"doi-asserted-by": "publisher",
"journal-title": "Stem Cell Rev Rep",
"key": "B40",
"volume": "17",
"year": "2021"
},
{
"DOI": "10.3390/cells10071585",
"article-title": "Cell death in coronavirus infections: uncovering its role during COVID-19",
"author": "Paolini",
"doi-asserted-by": "publisher",
"journal-title": "Cells",
"key": "B41",
"volume": "10",
"year": "2021"
},
{
"DOI": "10.3389/fimmu.2019.02759",
"article-title": "More than just attractive: how CCL2 influences myeloid cell behavior beyond chemotaxis",
"author": "Gschwandtner",
"doi-asserted-by": "publisher",
"journal-title": "Front Immunol",
"key": "B42",
"volume": "10",
"year": "2019"
},
{
"DOI": "10.1155/2011/852419",
"article-title": "Epithelial cell coculture models for studying infectious diseases: Benefits and limitations",
"author": "Duell",
"doi-asserted-by": "publisher",
"journal-title": "J Biomed Biotechnol",
"key": "B43",
"volume": "852419",
"year": "2011"
},
{
"DOI": "10.1371/journal.pcbi.1002264",
"article-title": "Stochastic delay accelerates signaling in gene networks",
"author": "Josić",
"doi-asserted-by": "publisher",
"journal-title": "PloS Computational Biology",
"key": "B44",
"volume": "7",
"year": "2011"
},
{
"DOI": "10.1016/j.bpj.2012.06.025",
"article-title": "Delayed protein synthesis reduces the correlation between mRNA and protein fluctuations",
"author": "Gedeon",
"doi-asserted-by": "crossref",
"journal-title": "Biophysical Journal",
"key": "B45",
"volume": "103",
"year": "2012"
},
{
"DOI": "10.1016/j.virusres.2020.198074",
"article-title": "The ORF6, ORF8 and nucleocapsid proteins of SARS-CoV-2 inhibit type I interferon signaling pathway",
"author": "Li",
"doi-asserted-by": "publisher",
"journal-title": "Virus Res",
"key": "B46",
"volume": "286",
"year": "2020"
},
{
"DOI": "10.1016/j.addr.2020.06.001",
"article-title": "Inhalation delivery technology for genome-editing of respiratory diseases",
"author": "Chow",
"doi-asserted-by": "publisher",
"journal-title": "Advanced Drug Delivery Rev",
"key": "B47",
"volume": "168",
"year": "2021"
}
],
"reference-count": 47,
"references-count": 47,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.frontiersin.org/articles/10.3389/fimmu.2024.1490478/full"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "TLR10 overexpression modulates immune response in A549 lung epithelial cells challenged with SARS-CoV-2 S and N proteins",
"type": "journal-article",
"update-policy": "https://doi.org/10.3389/crossmark-policy",
"volume": "15"
}
