Various Combinations of Favipiravir, Lopinavir-Ritonavir, Darunavir-Ritonavir, High-Dose Oseltamivir, and Hydroxychloroquine for the Treatment of COVID-19: A Randomized Controlled Trial (FIGHT-COVID-19 Study)
et al., SSRN Electronic Journal, doi:10.2139/ssrn.3936499, FIGHT-COVID-19, NCT04303299, Oct 2021
RCT 200 moderate/severe patients in Thailand, showing significantly lower progression with favipiravir vs. oseltamivir.
This paper contains multiple data issues and the preprint has not been published in a journal.
The manuscript title and abstract describe the study as a "randomized controlled trial", however, the methodology states that 90 eligible mild COVID-19 patients were randomly assigned in a 1:1:1 ratio to the three active treatment arms, which were then "compared to thirty voluntary quarantine patients".
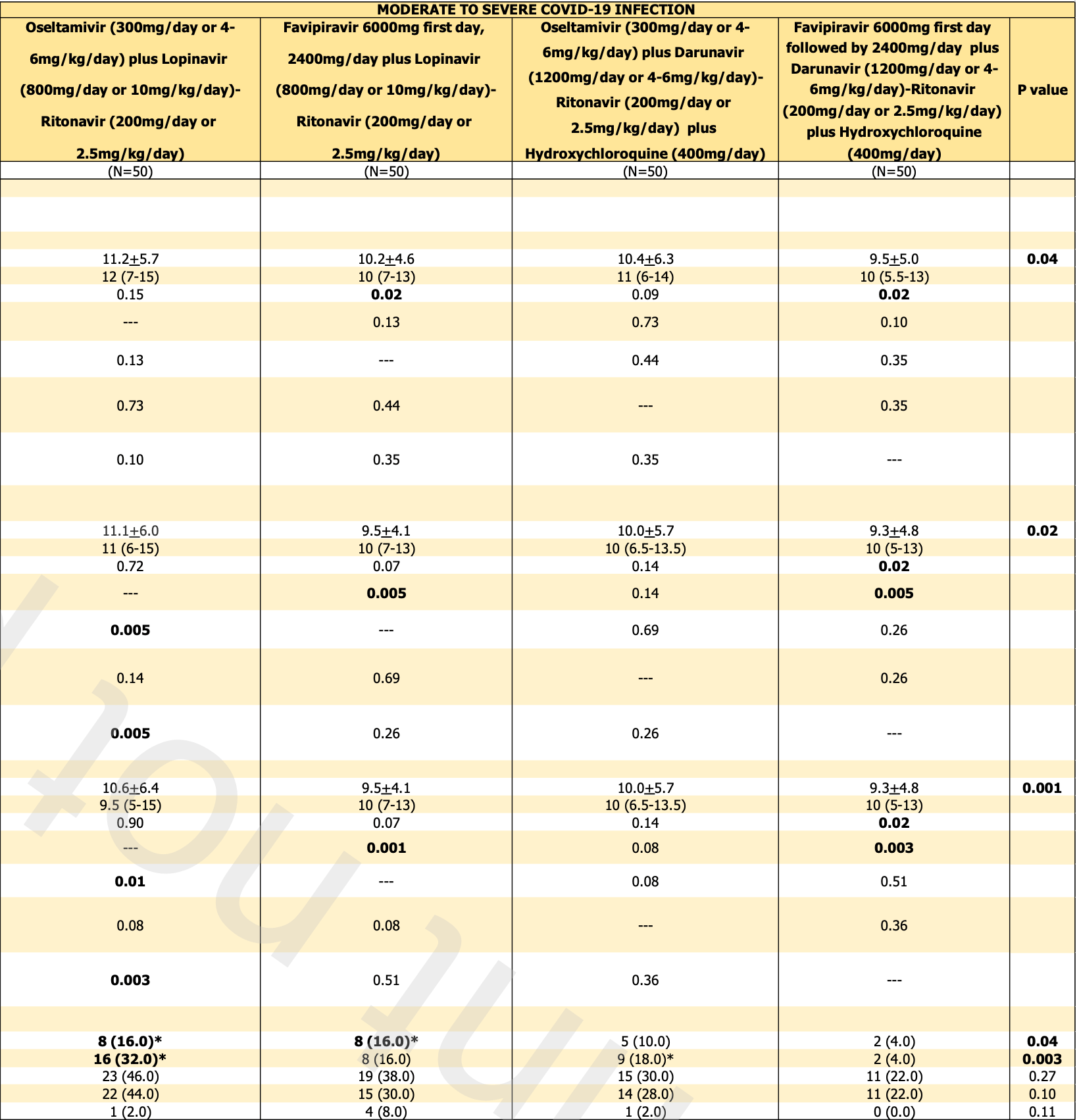
The ITT p-value for the primary comparison (favipiravir+darunavir-ritonavir+HCQ vs. oseltamivir+lopinavir-ritonavir) in moderate-to-severe COVID-19 is reported differently in the abstract (p = 0.15) and results tex (p = 0.10).
In Cohort B, the mean age was significantly different across treatment arms (p = 0.008, Table 1), however no adjustment for age was done.
Figure 1 states that the moderate-to-severe arm receiving favipiravir + darunavir-ritonavir + hydroxychloroquine experienced "4 treatment failure due to serious adverse events". Table 2 contradicts this, reporting the "failure antiviral treatment" for this arm as 2.
The statistical analysis section states that deaths were analyzed using Pearson Chi-square tests. An uncorrected Pearson Chi-square test yields a p-value near 0.045, but because the expected cell counts are at the minimum threshold of 5, this test is mathematically inappropriate. An appropriate test (Fisher's exact test) yields a non-significant p-value of 0.09.
This preprint was posted on SSRN on October 5, 2021, but has not been published in a peer-reviewed journal. The corresponding author and co-authors have published other COVID-19 work in peer-reviewed journals, including a favipiravir real-world data analysis. This suggests they have the capability and motivation to publish peer-reviewed work, making the failure to publish this particular study more notable.
For the oseltamivir+HCQ arm in Cohort A (ITT), the median time to negative PCR is reported as 7.5 days with IQR 4 to 25. An IQR of 4 to 25 represents a very large spread, with the 75th percentile at 25 days. However, the LOCF analysis reports the same median of 7.5 days but with IQR 4.5 to 14, and per-protocol shows median 8 days with IQR 4 to 15. The dramatic shrinking of the upper IQR from 25 (ITT) to 14 (LOCF) to 15 (PP) suggests that the ITT analysis was heavily influenced by a few patients who were lost to follow-up or had very prolonged viral shedding, potentially being carried forward as non-negative in the ITT analysis. This variability in a group of only 30 patients suggests the median is highly unstable and may not be a reliable estimate.
In Table 2 (ITT, Cohort A, oseltamivir+HCQ), the reported mean time to negative PCR is 8.9 days with an IQR of 4 to 25 and a median of 7.5 days for n=30. It is mathematically impossible for a sample of 30 to have a 75th percentile of 25 and a mean of 8.9, as the sum of the top 25% of values alone would make the mean higher than reported.
Implausible variance: Table 2 reports the ITT mean time to negative PCR for the quarantine group as 15.7 days with a standard deviation of 16.7 days. A standard deviation larger than the mean in this context suggests either an extreme, potentially erroneous outlier or a highly skewed distribution that makes the reporting of means misleading.
In Table 1, the 'All' column reports numbers that mathematically contradict the sum of the individual study arms. For example, it reports 73 elderly patients, but the sum of the 8 arms is 59, and it reports 70 obese patients, but the sum is 79.
Potential risks of favipiravir include kidney injury1-3, liver injury2-5, cardiovascular events5,6, pulmonary toxicity6,7, and mutagenicity, carcinogenicity, teratogenicity, embryotoxicity, and the creation of dangerous variants8-14.
This study is excluded in meta-analysis:
multiple data issues - pending author response.
Study covers favipiravir and HCQ.
|
risk of death, 23.1% lower, RR 0.77, p = 0.66, treatment 10 of 100 (10.0%), control 13 of 100 (13.0%), NNT 33, favipiravir arms vs. oseltamivir arms.
|
|
risk of progression, 60.0% lower, RR 0.40, p = 0.009, treatment 10 of 100 (10.0%), control 25 of 100 (25.0%), NNT 6.7, favipiravir arms vs. oseltamivir arms.
|
|
time to viral-, 8.7% lower, relative time 0.91, p = 0.43, treatment mean 9.5 (±5.0) n=50, control mean 10.4 (±6.3) n=50, HCQ arms, primary outcome.
|
|
time to viral-, 8.9% lower, relative time 0.91, p = 0.34, treatment mean 10.2 (±4.6) n=50, control mean 11.2 (±5.7) n=50, non-HCQ arms, primary outcome.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
1.
Abdulaziz et al., Clinical Features and Prognosis of Acute Kidney Injury in Hospital-Admitted Patients with COVID-19 in Egypt: A Single-Center Experience, Mansoura Medical Journal, doi:10.58775/2735-3990.1433.
2.
Ülger et al., Experimental evaluation of favipiravir (T-705)-induced liver and kidney toxicity in rats, Food and Chemical Toxicology, doi:10.1016/j.fct.2025.115472.
3.
El-Fetouh et al., Experimental Studies on Some Drugs Used in Covid-19 Treatment (Favipiravir and Dexamethasone) in Albino Rats, Journal of Advanced Veterinary Research, 13:10, www.advetresearch.com/index.php/AVR/article/view/1635.
4.
Almutairi et al., Liver Injury in Favipiravir-Treated COVID-19 Patients: Retrospective Single-Center Cohort Study, Tropical Medicine and Infectious Disease, doi:10.3390/tropicalmed8020129.
5.
Siby et al., Temporal Trends in Serious Adverse Events Associated with Oral Antivirals During the COVID-19 Pandemic: Insights from the FAERS Database (2020–2023), Open Forum Infectious Diseases, doi:10.1093/ofid/ofaf695.1825.
6.
Ozhan et al., Evaluation of the cardiopulmonary effects of repurposed COVID-19 therapeutics in healthy rats, Scientific Reports, doi:10.1038/s41598-025-31048-4.
7.
Ülger (B) et al., Evaluation of the effects of favipiravir (T-705) on the lung tissue of healty rats: An experimental study, Food and Chemical Toxicology, doi:10.1016/j.fct.2025.115235.
8.
Zhirnov et al., Favipiravir: the hidden threat of mutagenic action, Journal of microbiology, epidemiology and immunobiology, doi:10.36233/0372-9311-114.
9.
Waters et al., Human genetic risk of treatment with antiviral nucleoside analog drugs that induce lethal mutagenesis: the special case of molnupiravir, Environmental and Molecular Mutagenesis, doi:10.1002/em.22471.
10.
Hadj Hassine et al., Lethal Mutagenesis of RNA Viruses and Approved Drugs with Antiviral Mutagenic Activity, Viruses, doi:10.3390/v14040841.
11.
Shum, C., An investigational study into the drug-associated mutational signature in SARS-CoV-2 viruses, The University of Hong Kong, PhD Thesis, hub.hku.hk/handle/10722/344396.
12.
Shiraki et al., Convenient screening of the reproductive toxicity of favipiravir and antiviral drugs in Caenorhabditis elegans, Heliyon, doi:10.1016/j.heliyon.2024.e35331.
Atipornwanich et al., 5 Oct 2021, Randomized Controlled Trial, Thailand, peer-reviewed, 16 authors, study period 19 August, 2020 - 28 August, 2021, this trial compares with another treatment - results may be better when compared to placebo, this trial uses multiple treatments in the treatment arm (combined with lopinavir/ritonavir or duranivir/ritonavir/HCQ) - results of individual treatments may vary, trial NCT04303299 (history) (FIGHT-COVID-19).
Various combinations of Favipiravir, Lopinavir-Ritonavir, Darunavir-Ritonavir, high-dose Oseltamivir, and Hydroxychloroquine for the treatment of Covid-19: A randomized controlled trial. (FIGHT-COVID-19 Study)
References
Hata, Koseki, Yamaguchi, Limited inhibitory effects of Oseltamivir and zanamivir on human sialidases, Antimicrob Agents Chemother, doi:10.1128/AAC.00344-08
Hung, Lung, Tso, Triple combination of interferon beta-1b, lopinavirritonavir, and ribavirin in the treatment of patients admitted to hospital with Covid -19: an open-label, randomised, phase 2 trial, Lancet, doi:10.1016/S0140-6736(20)31042-4
Kriangsak, Akksilp, Sawanpanyalert, Srisubat, Thanasithichai et al., Various Combination of Antiviral Treatment of Covid -19 Pneumonia
Shinkai, Tsushima, Tanaka, A Randomized, Phase III Clinical Trial, Infect Dis Ther, doi:10.1007/s40121-021-00517-4
Wang, Zhang, Du, Remdesivir in adults with severe Covid -19: a randomised, doubleblind, placebo-controlled, multicentre trial [published correction appears in, Lancet
DOI record:
{
"DOI": "10.2139/ssrn.3936499",
"ISSN": [
"1556-5068"
],
"URL": "http://dx.doi.org/10.2139/ssrn.3936499",
"author": [
{
"affiliation": [],
"family": "Atipornwanich",
"given": "Kriangsak",
"sequence": "first"
},
{
"affiliation": [],
"family": "Kongsaengdao",
"given": "Subsai",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Harnsomburana",
"given": "Piyathida",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Nanna",
"given": "Rienthong",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Chtuparisute",
"given": "Chatchawan",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Saengsayan",
"given": "Piamlarp",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Bangpattanasiri",
"given": "Kittima",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Manosuthi",
"given": "Weerawat",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Sawanpanyalert",
"given": "Narumol",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Srisubat",
"given": "Attasit",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Thanasithichai",
"given": "Somchai",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Maneeton",
"given": "Benchalak",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Maneeton",
"given": "Narong",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Suthisisang",
"given": "Chuthamanee",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Pratuangdejkul",
"given": "Jaturong",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Akksilp",
"given": "Somsak",
"sequence": "additional"
}
],
"container-title": [
"SSRN Electronic Journal"
],
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2021,
10,
10
]
],
"date-time": "2021-10-10T12:55:10Z",
"timestamp": 1633870510000
},
"deposited": {
"date-parts": [
[
2021,
10,
10
]
],
"date-time": "2021-10-10T12:55:13Z",
"timestamp": 1633870513000
},
"indexed": {
"date-parts": [
[
2022,
1,
10
]
],
"date-time": "2022-01-10T12:29:11Z",
"timestamp": 1641817751857
},
"is-referenced-by-count": 1,
"issn-type": [
{
"type": "electronic",
"value": "1556-5068"
}
],
"issued": {
"date-parts": [
[
2021
]
]
},
"language": "en",
"member": "78",
"original-title": [],
"prefix": "10.2139",
"published": {
"date-parts": [
[
2021
]
]
},
"published-other": {
"date-parts": [
[
2021
]
]
},
"publisher": "Elsevier BV",
"reference-count": 0,
"references-count": 0,
"relation": {},
"score": 1,
"short-container-title": [
"SSRN Journal"
],
"short-title": [],
"source": "Crossref",
"subtitle": [],
"title": [
"Various Combinations of Favipiravir, Lopinavir-Ritonavir, Darunavir-Ritonavir, High-Dose Oseltamivir, and Hydroxychloroquine for the Treatment of COVID-19: A Randomized Controlled Trial (FIGHT-COVID-19 Study)"
],
"type": "journal-article"
}
atipornwanich