Efficacy and Safety of Favipiravir in Moderate COVID-19 Pneumonia Patients without Oxygen Therapy: A Randomized, Phase III Clinical Trial
et al., Infectious Diseases and Therapy, doi:10.1007/s40121-021-00517-4, Aug 2021
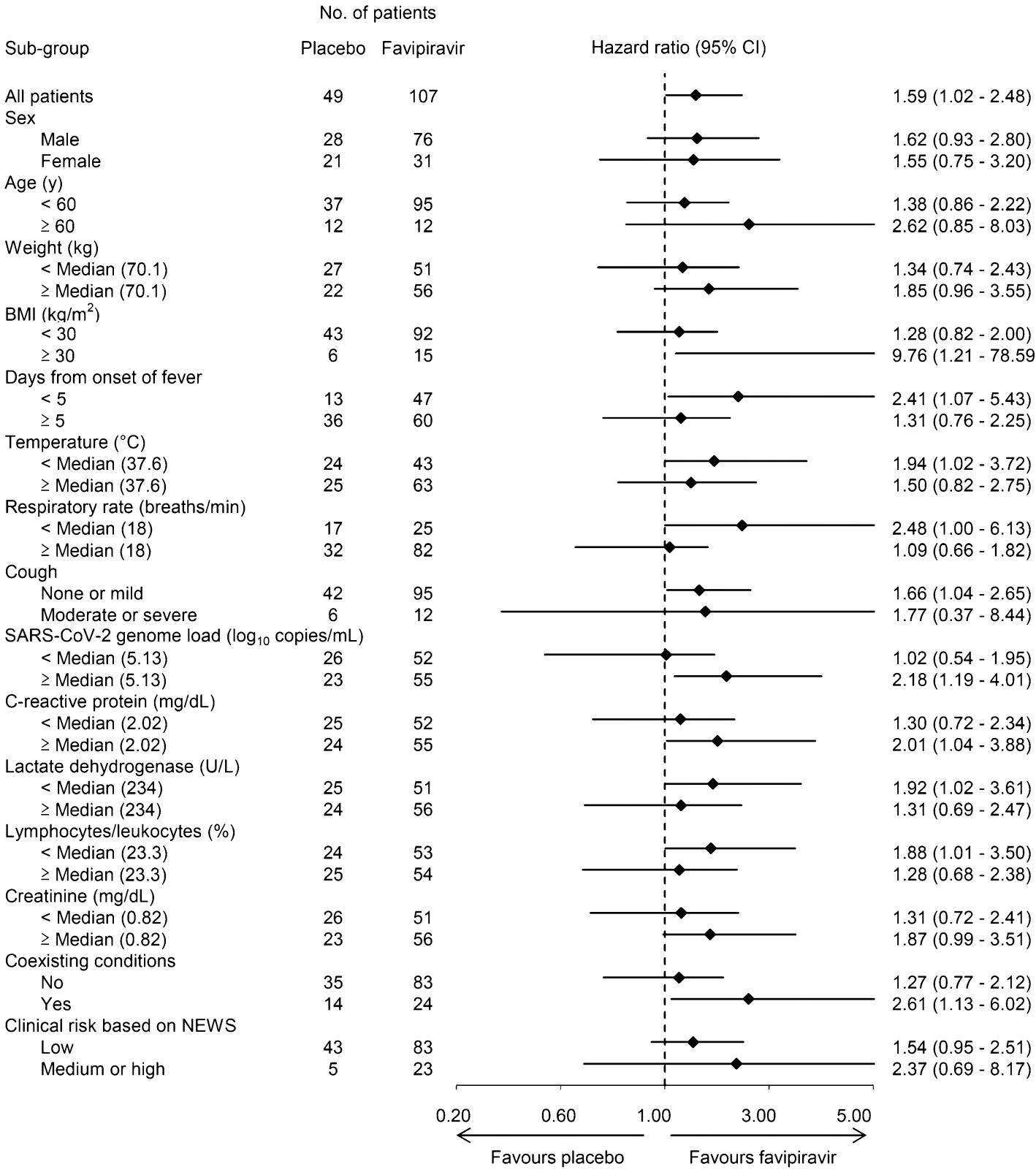
RCT 156 patients in Japan, 107 treated with favipiravir, showing significant improvement in a composite outcome defined as the time to improvement in temperature, SpO2, CT findings, and recovery to PCR-.
Potential risks of favipiravir include kidney injury1-3, liver injury2-5, cardiovascular events5,6, pulmonary toxicity6,7, and mutagenicity, carcinogenicity, teratogenicity, embryotoxicity, and the creation of dangerous variants8-14.
Standard of Care (SOC) for COVID-19 in the study country,
Japan, is very poor with very low average efficacy for approved treatments15.
Only expensive, high-profit treatments were approved for early treatment. Low-cost treatments were excluded, reducing the probability of early treatment due to access and cost barriers, and eliminating complementary and synergistic benefits seen with many low-cost treatments.
|
time to improvement, 37.1% lower, HR 0.63, p = 0.01, treatment 107, control 49, adjusted per study, inverted to make HR<1 favor treatment, Cox proportional hazards, composite time to improvement in temperature, SpO2, CT findings, and recovery to PCR-.
|
|
time to improvement, 58.5% lower, HR 0.41, p = 0.01, treatment 47, control 13, adjusted per study, inverted to make HR<1 favor treatment, <5 days from onset of fever, Cox proportional hazards, composite time to improvement in temperature, SpO2, CT findings, and recovery to PCR-.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
1.
Abdulaziz et al., Clinical Features and Prognosis of Acute Kidney Injury in Hospital-Admitted Patients with COVID-19 in Egypt: A Single-Center Experience, Mansoura Medical Journal, doi:10.58775/2735-3990.1433.
2.
Ülger et al., Experimental evaluation of favipiravir (T-705)-induced liver and kidney toxicity in rats, Food and Chemical Toxicology, doi:10.1016/j.fct.2025.115472.
3.
El-Fetouh et al., Experimental Studies on Some Drugs Used in Covid-19 Treatment (Favipiravir and Dexamethasone) in Albino Rats, Journal of Advanced Veterinary Research, 13:10, www.advetresearch.com/index.php/AVR/article/view/1635.
4.
Almutairi et al., Liver Injury in Favipiravir-Treated COVID-19 Patients: Retrospective Single-Center Cohort Study, Tropical Medicine and Infectious Disease, doi:10.3390/tropicalmed8020129.
5.
Siby et al., Temporal Trends in Serious Adverse Events Associated with Oral Antivirals During the COVID-19 Pandemic: Insights from the FAERS Database (2020–2023), Open Forum Infectious Diseases, doi:10.1093/ofid/ofaf695.1825.
6.
Ozhan et al., Evaluation of the cardiopulmonary effects of repurposed COVID-19 therapeutics in healthy rats, Scientific Reports, doi:10.1038/s41598-025-31048-4.
7.
Ülger (B) et al., Evaluation of the effects of favipiravir (T-705) on the lung tissue of healty rats: An experimental study, Food and Chemical Toxicology, doi:10.1016/j.fct.2025.115235.
8.
Zhirnov et al., Favipiravir: the hidden threat of mutagenic action, Journal of microbiology, epidemiology and immunobiology, doi:10.36233/0372-9311-114.
9.
Waters et al., Human genetic risk of treatment with antiviral nucleoside analog drugs that induce lethal mutagenesis: the special case of molnupiravir, Environmental and Molecular Mutagenesis, doi:10.1002/em.22471.
10.
Hadj Hassine et al., Lethal Mutagenesis of RNA Viruses and Approved Drugs with Antiviral Mutagenic Activity, Viruses, doi:10.3390/v14040841.
11.
Shum, C., An investigational study into the drug-associated mutational signature in SARS-CoV-2 viruses, The University of Hong Kong, PhD Thesis, hub.hku.hk/handle/10722/344396.
12.
Shiraki et al., Convenient screening of the reproductive toxicity of favipiravir and antiviral drugs in Caenorhabditis elegans, Heliyon, doi:10.1016/j.heliyon.2024.e35331.
13.
Cenikli et al., Does Favipiravir interact with DNA? Design of electrochemical DNA nanobiosensor to investigate the interaction between DNA and Favipiravir used in the treatment of COVID-19, Talanta, doi:10.1016/j.talanta.2025.128084.
Shinkai et al., 27 Aug 2021, Single Blind Randomized Controlled Trial, Japan, peer-reviewed, 39 authors, average treatment delay 4.8 days.
Efficacy and Safety of Favipiravir in Moderate COVID-19 Pneumonia Patients without Oxygen Therapy: A Randomized, Phase III Clinical Trial
Infectious Diseases and Therapy, doi:10.1007/s40121-021-00517-4
Introduction: Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the cause of coronavirus disease 2019 , is an enveloped, single-stranded RNA virus. Favipi-ravir is an orally administrable antiviral drug whose mechanism of action is to selectively inhibit RNA-dependent RNA polymerase. A preliminary trial in COVID-19 patients reported significant improvements across a multitude of clinical parameters, but these findings have not been confirmed in an adequate well-controlled trial. We conducted a randomized, single-blind, placebo-controlled Phase III trial assessing the efficacy and safety of favipiravir in patients with Supplementary Information The online version contains supplementary material available at https://
Authorship. All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published. Authorship Contributions. Conceptualization: Satoshi Iwata, Tsutomu Sakurai; Methodology: Satoshi Iwata, Osamu Kobayashi, Hiroyuki Kunishima, Masaharu Shinkai; Formal analysis: Yoshihiko Matsumoto, Tsutomu Sakurai; Investigation: Masaharu Shinkai, Kenji Tsushima, Shingo Tanaka, Eri Hagiwara, Norihito Tarumoto, Ichiro Kawada, Yuji Hirai, Sho Fujiwara, Yuko Komase, Takeshi Saraya, Hidefumi Koh, Naho Kagiyama, Megumi Shimada, Daiki Kanou, Shinichi Antoku, Yujiro Uchida, Yutaka Tokue, Mikio Takamori, Yasuhiro Gon, Kenya Ie, Yoshitaka Yamazaki, Kazumasa Harada, Naoki Miyao, Takashi Naka, Mitsunaga Iwata, Atsushi Nakagawa, Kazutoshi Hiyama, Yoshihiko Ogawa, Masahiro Shinoda, Shinichiro Ota, Takatomo Hirouchi, Jiro Terada, Shuichi Kawano and Takashi Ogura; Supervision: Satoshi Iwata; Writing -original draft preparation; Masaharu Shinkai. All authors commented on previous versions of the manuscript. All authors read and approved the submitted version. Disclosures. Tsutomu Sakurai and Yoshihiko Matsumoto are employees of the sponsor. Masaharu Shinkai, Kenji Tsushima, Shingo Tanaka, Eri Hagiwara, Norihito Tarumoto, Ichiro Kawada, Yuji Hirai, Sho Fujiwara, Yuko Komase, Takeshi Saraya, Hidefumi Koh, Naho Kagiyama, Megumi..
References
Cai, Yang, Liu, Experimental treatment with favipiravir for COVID-19: An open-label control study, Eng
Cele, Gazy, Jackson, Escape of SARS-CoV-2 501Y.V2 from neutralization by convalescent plasma, Nature
Cui, Hung, Wang, Modification of sample size in group sequential clinical trials, Biometrics
Doi, Hibino, Hase, A prospective, randomized, open-label trial of early versus late favipiravir therapy in hospitalized patients with COVID-19, Antimicrob Agents Chemother
Fujii, Ibe, Ishigo, Early favipiravir treatment was associated with early defervescence in non-severe COVID-19 patients, J Infect Chemother
Galloway, Paul, Maccannell, Emergence of SARS-CoV-2 B.1.1.7 Lineage-United States, MMWR Morb Mortal Wkly Rep
Gordon, Tchesnokov, Woolner, Remdesivir is a direct-acting antiviral that inhibits RNA-dependent RNA polymerase from severe acute respiratory syndrome coronavirus 2 with high potency, J Biol Chem
Gowen, Wong, Jung, In vitro and in vivo activities of T-705 against arenavirus and bunyavirus infections, Antimicrob Agents Chemother
Ivashchenko, Dmitriev, Vostokova, AVIFAVIR for treatment of patients with moderate COVID-19: interim results of a phase II/III multicenter randomized clinical trial, Clin Infect Dis
Kaptein, Jacobs, Langendries, Favipiravir at high doses has potent antiviral activity in SARS-CoV-2-infected hamsters, whereas hydroxychloroquine lacks activity, Proc Natl Acad Sci
Li, Wu, Nie, The impact of mutations in SARS-CoV-2 spike on viral infectivity and antigenicity, Cell
Mendenhall, Russell, Smee, Effective oral favipiravir (T-705) therapy initiated after the onset of clinical disease in a model of arenavirus hemorrhagic fever, PLoS Negl Trop Dis
Morikawa, Shinoda, Ota, Clinical features of 154 COVID-19 patients and parameters for effective detection of pneumonia at the time of initial diagnosis in Japan, Intern Med
Oxley, Mocco, Majidi, Large-vessel stroke as a presenting feature of Covid-19 in the young, N Engl J Med
Shanon, Selisko, Le, Rapid incorporation of favipiravir by the fast and permissive viral RNA polymerase complex results in SARS-CoV-2 lethal mutagenesis, Nat Commun
Sidwell, Barnard, Day, Efficacy of orally administered T-705 on lethal avian influenza A (H5N1) virus infections in mice, Antimicrob Agents Chemother
Suemori, Saijo, Yamanaka, A multicenter non-randomized, uncontrolled single arm trial for evaluation of the efficacy and the safety of the treatment with favipiravir for patients with severe fever with thrombocytopenia syndrome, PLoS Negl Trop Dis
Udwadia, Singh, Barkate, Efficacy and safety of favipiravir, an oral RNA-dependent RNA polymerase inhibitor, in mild-to-moderate COVID-19: a randomized, comparative, open-label, multicenter, phase 3 clinical trial, Int J Infect Dis
Wang, Cao, Zhang, Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro, Cell Res
Wang, Chen, Tissue distributions of antiviral drugs affect their capabilities of reducing viral loads in COVID-19 treatment, Eur J Pharmacol
Wo ¨lfel, Corman, Guggemos, Virological assessment of hospitalized patients with COVID-2019, Nature
Zhou, Yu, Du, Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study, Lancet
DOI record:
{
"DOI": "10.1007/s40121-021-00517-4",
"ISSN": [
"2193-8229",
"2193-6382"
],
"URL": "http://dx.doi.org/10.1007/s40121-021-00517-4",
"alternative-id": [
"517"
],
"assertion": [
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "Received",
"name": "received",
"order": 1,
"value": "21 May 2021"
},
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "Accepted",
"name": "accepted",
"order": 2,
"value": "27 July 2021"
},
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "First Online",
"name": "first_online",
"order": 3,
"value": "27 August 2021"
}
],
"author": [
{
"affiliation": [],
"family": "Shinkai",
"given": "Masaharu",
"sequence": "first"
},
{
"affiliation": [],
"family": "Tsushima",
"given": "Kenji",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Tanaka",
"given": "Shingo",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Hagiwara",
"given": "Eri",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Tarumoto",
"given": "Norihito",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Kawada",
"given": "Ichiro",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Hirai",
"given": "Yuji",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Fujiwara",
"given": "Sho",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Komase",
"given": "Yuko",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Saraya",
"given": "Takeshi",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Koh",
"given": "Hidefumi",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Kagiyama",
"given": "Naho",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Shimada",
"given": "Megumi",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Kanou",
"given": "Daiki",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Antoku",
"given": "Shinichi",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Uchida",
"given": "Yujiro",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Tokue",
"given": "Yutaka",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Takamori",
"given": "Mikio",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Gon",
"given": "Yasuhiro",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ie",
"given": "Kenya",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Yamazaki",
"given": "Yoshitaka",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Harada",
"given": "Kazumasa",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Miyao",
"given": "Naoki",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Naka",
"given": "Takashi",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Iwata",
"given": "Mitsunaga",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Nakagawa",
"given": "Atsushi",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Hiyama",
"given": "Kazutoshi",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ogawa",
"given": "Yoshihiko",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Shinoda",
"given": "Masahiro",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ota",
"given": "Shinichiro",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Hirouchi",
"given": "Takatomo",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Terada",
"given": "Jiro",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Kawano",
"given": "Shuichi",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ogura",
"given": "Takashi",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Sakurai",
"given": "Tsutomu",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Matsumoto",
"given": "Yoshihiko",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Kunishima",
"given": "Hiroyuki",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Kobayashi",
"given": "Osamu",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Iwata",
"given": "Satoshi",
"sequence": "additional"
}
],
"container-title": "Infectious Diseases and Therapy",
"container-title-short": "Infect Dis Ther",
"content-domain": {
"crossmark-restriction": false,
"domain": [
"link.springer.com"
]
},
"created": {
"date-parts": [
[
2021,
8,
27
]
],
"date-time": "2021-08-27T18:02:36Z",
"timestamp": 1630087356000
},
"deposited": {
"date-parts": [
[
2021,
11,
7
]
],
"date-time": "2021-11-07T14:15:07Z",
"timestamp": 1636294507000
},
"funder": [
{
"DOI": "10.13039/501100019949",
"doi-asserted-by": "crossref",
"name": "FUJIFILM Toyama Chemical CO"
}
],
"indexed": {
"date-parts": [
[
2024,
3,
28
]
],
"date-time": "2024-03-28T10:36:05Z",
"timestamp": 1711622165879
},
"is-referenced-by-count": 49,
"issue": "4",
"issued": {
"date-parts": [
[
2021,
8,
27
]
]
},
"journal-issue": {
"issue": "4",
"published-print": {
"date-parts": [
[
2021,
12
]
]
}
},
"language": "en",
"license": [
{
"URL": "https://creativecommons.org/licenses/by-nc/4.0",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
8,
27
]
],
"date-time": "2021-08-27T00:00:00Z",
"timestamp": 1630022400000
}
},
{
"URL": "https://creativecommons.org/licenses/by-nc/4.0",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
8,
27
]
],
"date-time": "2021-08-27T00:00:00Z",
"timestamp": 1630022400000
}
}
],
"link": [
{
"URL": "https://link.springer.com/content/pdf/10.1007/s40121-021-00517-4.pdf",
"content-type": "application/pdf",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://link.springer.com/article/10.1007/s40121-021-00517-4/fulltext.html",
"content-type": "text/html",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://link.springer.com/content/pdf/10.1007/s40121-021-00517-4.pdf",
"content-type": "application/pdf",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "297",
"original-title": [],
"page": "2489-2509",
"prefix": "10.1007",
"published": {
"date-parts": [
[
2021,
8,
27
]
]
},
"published-online": {
"date-parts": [
[
2021,
8,
27
]
]
},
"published-print": {
"date-parts": [
[
2021,
12
]
]
},
"publisher": "Springer Science and Business Media LLC",
"reference": [
{
"key": "517_CR1",
"unstructured": "Coronavirus disease (COVID-19) Weekly Epidemiological Update and Weekly Operational Upd. Geneva: World Health Organization. 2021. https://www.who.int/publications/m/item/weekly-operational-update-on-covid-19---10-may-2021. Accessed 10 May 2021."
},
{
"DOI": "10.1128/AAC.00356-07",
"author": "BB Gowen",
"doi-asserted-by": "publisher",
"first-page": "3168",
"journal-title": "Antimicrob Agents Chemother",
"key": "517_CR2",
"unstructured": "Gowen BB, Wong MH, Jung KH, et al. In vitro and in vivo activities of T-705 against arenavirus and bunyavirus infections. Antimicrob Agents Chemother. 2007;51:3168–76.",
"volume": "51",
"year": "2007"
},
{
"DOI": "10.1371/journal.pntd.0001342",
"author": "M Mendenhall",
"doi-asserted-by": "publisher",
"first-page": "e1342",
"journal-title": "PLoS Negl Trop Dis",
"key": "517_CR3",
"unstructured": "Mendenhall M, Russell A, Smee DF, et al. Effective oral favipiravir (T-705) therapy initiated after the onset of clinical disease in a model of arenavirus hemorrhagic fever. PLoS Negl Trop Dis. 2011;5:e1342.",
"volume": "5",
"year": "2011"
},
{
"DOI": "10.1038/s41422-020-0282-0",
"author": "M Wang",
"doi-asserted-by": "publisher",
"first-page": "269",
"journal-title": "Cell Res",
"key": "517_CR4",
"unstructured": "Wang M, Cao R, Zhang L, et al. Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro. Cell Res. 2020;30:269–71.",
"volume": "30",
"year": "2020"
},
{
"DOI": "10.1038/s41467-020-18463-z",
"author": "A Shanon",
"doi-asserted-by": "publisher",
"first-page": "4682",
"journal-title": "Nat Commun",
"key": "517_CR5",
"unstructured": "Shanon A, Selisko B, Le NT, et al. Rapid incorporation of favipiravir by the fast and permissive viral RNA polymerase complex results in SARS-CoV-2 lethal mutagenesis. Nat Commun. 2020;11:4682.",
"volume": "11",
"year": "2020"
},
{
"DOI": "10.1074/jbc.RA120.013679",
"author": "CJ Gordon",
"doi-asserted-by": "publisher",
"first-page": "6785",
"journal-title": "J Biol Chem",
"key": "517_CR6",
"unstructured": "Gordon CJ, Tchesnokov EP, Woolner E, et al. Remdesivir is a direct-acting antiviral that inhibits RNA-dependent RNA polymerase from severe acute respiratory syndrome coronavirus 2 with high potency. J Biol Chem. 2020;295:6785–97.",
"volume": "295",
"year": "2020"
},
{
"author": "Q Cai",
"first-page": "1192",
"journal-title": "Eng (Beijing)",
"key": "517_CR7",
"unstructured": "Cai Q, Yang M, Liu D, et al. Experimental treatment with favipiravir for COVID-19: An open-label control study. Eng (Beijing). 2020;6:1192–8.",
"volume": "6",
"year": "2020"
},
{
"key": "517_CR8",
"unstructured": "Favipiravir Observational Study Group, Fujita Health University. Interim report of the Favipiravir Observational Study in Japan. 2020. https://www.kansensho.or.jp/uploads/files/topics/2019ncov/covid19_favip_210419_eng.pdf."
},
{
"DOI": "10.1128/AAC.01051-06",
"author": "RW Sidwell",
"doi-asserted-by": "publisher",
"first-page": "845",
"journal-title": "Antimicrob Agents Chemother",
"key": "517_CR9",
"unstructured": "Sidwell RW, Barnard DL, Day CW, et al. Efficacy of orally administered T-705 on lethal avian influenza A (H5N1) virus infections in mice. Antimicrob Agents Chemother. 2007;51:845–51.",
"volume": "51",
"year": "2007"
},
{
"DOI": "10.1371/journal.pntd.0009103",
"author": "K Suemori",
"doi-asserted-by": "publisher",
"first-page": "e0009103",
"journal-title": "PLoS Negl Trop Dis",
"key": "517_CR10",
"unstructured": "Suemori K, Saijo M, Yamanaka A, et al. A multicenter non-randomized, uncontrolled single arm trial for evaluation of the efficacy and the safety of the treatment with favipiravir for patients with severe fever with thrombocytopenia syndrome. PLoS Negl Trop Dis. 2021;22:e0009103.",
"volume": "22",
"year": "2021"
},
{
"DOI": "10.1111/j.0006-341X.1999.00853.x",
"author": "L Cui",
"doi-asserted-by": "publisher",
"first-page": "853",
"journal-title": "Biometrics",
"key": "517_CR11",
"unstructured": "Cui L, Hung HM, Wang SJ. Modification of sample size in group sequential clinical trials. Biometrics. 1999;55:853–7.",
"volume": "55",
"year": "1999"
},
{
"DOI": "10.2169/internalmedicine.5528-20",
"author": "M Morikawa",
"doi-asserted-by": "publisher",
"first-page": "31",
"journal-title": "Intern Med",
"key": "517_CR12",
"unstructured": "Morikawa M, Shinoda M, Ota S, et al. Clinical features of 154 COVID-19 patients and parameters for effective detection of pneumonia at the time of initial diagnosis in Japan. Intern Med. 2021;60:31–7.",
"volume": "60",
"year": "2021"
},
{
"DOI": "10.1016/j.ijid.2020.11.142",
"author": "ZF Udwadia",
"doi-asserted-by": "publisher",
"first-page": "62",
"journal-title": "Int J Infect Dis",
"key": "517_CR13",
"unstructured": "Udwadia ZF, Singh P, Barkate H, et al. Efficacy and safety of favipiravir, an oral RNA-dependent RNA polymerase inhibitor, in mild-to-moderate COVID-19: a randomized, comparative, open-label, multicenter, phase 3 clinical trial. Int J Infect Dis. 2021;103:62–71.",
"volume": "103",
"year": "2021"
},
{
"author": "AA Ivashchenko",
"first-page": "ciaa1176",
"journal-title": "Clin Infect Dis",
"key": "517_CR14",
"unstructured": "Ivashchenko AA, Dmitriev KA, Vostokova NV, et al. AVIFAVIR for treatment of patients with moderate COVID-19: interim results of a phase II/III multicenter randomized clinical trial. Clin Infect Dis. 2020;2020:ciaa1176.",
"volume": "2020",
"year": "2020"
},
{
"DOI": "10.1128/AAC.01897-20",
"author": "Y Doi",
"doi-asserted-by": "publisher",
"first-page": "e01897-20",
"journal-title": "Antimicrob Agents Chemother",
"key": "517_CR15",
"unstructured": "Doi Y, Hibino M, Hase R, et al. A prospective, randomized, open-label trial of early versus late favipiravir therapy in hospitalized patients with COVID-19. Antimicrob Agents Chemother. 2020;64:e01897-20.",
"volume": "64",
"year": "2020"
},
{
"DOI": "10.1073/pnas.2014441117",
"author": "SJF Kaptein",
"doi-asserted-by": "publisher",
"first-page": "26955",
"journal-title": "Proc Natl Acad Sci USA",
"key": "517_CR16",
"unstructured": "Kaptein SJF, Jacobs S, Langendries L, et al. Favipiravir at high doses has potent antiviral activity in SARS-CoV-2-infected hamsters, whereas hydroxychloroquine lacks activity. Proc Natl Acad Sci USA. 2020;117:26955–65.",
"volume": "117",
"year": "2020"
},
{
"DOI": "10.1016/j.ejphar.2020.173634",
"author": "Y Wang",
"doi-asserted-by": "publisher",
"first-page": "173634",
"journal-title": "Eur J Pharmacol",
"key": "517_CR17",
"unstructured": "Wang Y, Chen L. Tissue distributions of antiviral drugs affect their capabilities of reducing viral loads in COVID-19 treatment. Eur J Pharmacol. 2020;889:173634.",
"volume": "889",
"year": "2020"
},
{
"DOI": "10.1016/j.jiac.2021.04.013",
"author": "S Fujii",
"doi-asserted-by": "publisher",
"first-page": "1051",
"journal-title": "J Infect Chemother.",
"key": "517_CR18",
"unstructured": "Fujii S, Ibe Y, Ishigo T, et al. Early favipiravir treatment was associated with early defervescence in non-severe COVID-19 patients. J Infect Chemother. 2021;27:1051–7.",
"volume": "27",
"year": "2021"
},
{
"DOI": "10.1016/S0140-6736(20)30566-3",
"author": "F Zhou",
"doi-asserted-by": "publisher",
"first-page": "1054",
"journal-title": "Lancet",
"key": "517_CR19",
"unstructured": "Zhou F, Yu T, Du R, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395:1054–62.",
"volume": "395",
"year": "2020"
},
{
"DOI": "10.1056/NEJMc2009787",
"author": "TJ Oxley",
"doi-asserted-by": "publisher",
"first-page": "e60",
"journal-title": "N Engl J Med",
"key": "517_CR20",
"unstructured": "Oxley TJ, Mocco J, Majidi S, et al. Large-vessel stroke as a presenting feature of Covid-19 in the young. N Engl J Med. 2020;382:e60.",
"volume": "382",
"year": "2020"
},
{
"DOI": "10.1038/s41586-020-2196-x",
"author": "R Wölfel",
"doi-asserted-by": "publisher",
"first-page": "465",
"journal-title": "Nature",
"key": "517_CR21",
"unstructured": "Wölfel R, Corman VM, Guggemos W, et al. Virological assessment of hospitalized patients with COVID-2019. Nature. 2020;581:465–9.",
"volume": "581",
"year": "2020"
},
{
"DOI": "10.15585/mmwr.mm7003e2",
"doi-asserted-by": "crossref",
"key": "517_CR22",
"unstructured": "Galloway SE, Paul P, MacCannell DR, et al. Emergence of SARS-CoV-2 B.1.1.7 Lineage—United States, December 29, 2020-January 12, 2021. MMWR Morb Mortal Wkly Rep. 2021;70:95–9."
},
{
"DOI": "10.1016/j.cell.2020.07.012",
"author": "Q Li",
"doi-asserted-by": "publisher",
"first-page": "1284",
"journal-title": "Cell",
"key": "517_CR23",
"unstructured": "Li Q, Wu J, Nie J, et al. The impact of mutations in SARS-CoV-2 spike on viral infectivity and antigenicity. Cell. 2020;182:1284–94.",
"volume": "182",
"year": "2020"
},
{
"DOI": "10.1038/s41586-021-03471-w",
"author": "S Cele",
"doi-asserted-by": "publisher",
"first-page": "142",
"journal-title": "Nature",
"key": "517_CR24",
"unstructured": "Cele S, Gazy I, Jackson L, et al. Escape of SARS-CoV-2 501Y.V2 from neutralization by convalescent plasma. Nature. 2021;593:142–6.",
"volume": "593",
"year": "2021"
}
],
"reference-count": 24,
"references-count": 24,
"relation": {},
"resource": {
"primary": {
"URL": "https://link.springer.com/10.1007/s40121-021-00517-4"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"Infectious Diseases",
"Microbiology (medical)"
],
"subtitle": [],
"title": "Efficacy and Safety of Favipiravir in Moderate COVID-19 Pneumonia Patients without Oxygen Therapy: A Randomized, Phase III Clinical Trial",
"type": "journal-article",
"update-policy": "http://dx.doi.org/10.1007/springer_crossmark_policy",
"volume": "10"
}