Iota-carrageenan and xylitol inhibit SARS-CoV-2 in Vero cell culture
et al., PLoS ONE, doi:10.1371/journal.pone.0259943, Aug 2020 (preprint)
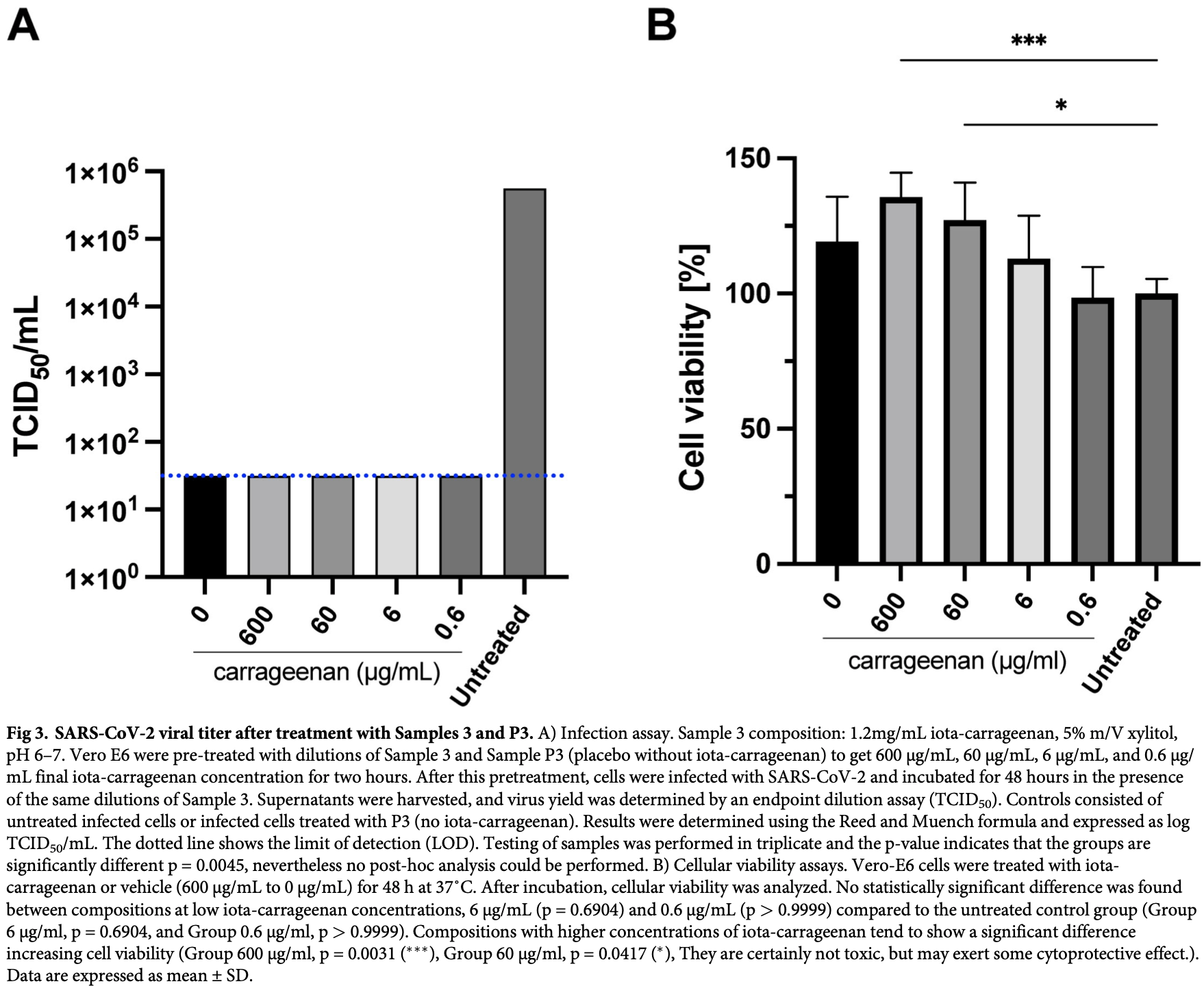
In vitro study showing that iota-carrageenan in concentrations as low as 6 µg/mL (easily achievable with existing nasal sprays) inhibits SARS-CoV-2 in Vero cell cultures.
20 preclinical studies support the efficacy of iota-carrageenan for COVID-19:
1.
Herida et al., Chemical Insights into the Antiviral Mechanisms of Marine Sulfated Polysaccharides: An In-Silico Screening and Molecular Docking Study, Biointerface Research in Applied Chemistry, doi:10.33263/BRIAC155.071.
2.
Krylova et al., Carrageenans and the Carrageenan-Echinochrome Complex as Anti-SARS-CoV-2 Agents, International Journal of Molecular Sciences, doi:10.3390/ijms26136175.
3.
Rohilla et al., Algae Polysaccharides (Carrageenan and Alginate)—A Treasure-Trove of Antiviral Compounds: An In Silico Approach to Identify Potential Candidates for Inhibition of S1-RBD Spike Protein of SARS-CoV-2, Stresses, doi:10.3390/stresses3030039.
4.
Thet, H., The potential of carrageenan for the drug discovery of COVID-19 via molecular docking with angiotensin-converting enzyme 2 (ACE2) and the main protease (Mpro) of SARS-CoV-2, Journal of Bioinformatics and Genomics, doi:10.18454/jbg.2022.18.2.001.
5.
Alsaidi et al., Griffithsin and Carrageenan Combination Results in Antiviral Synergy against SARS-CoV-1 and 2 in a Pseudoviral Model, Marine Drugs, doi:10.3390/md19080418.
6.
Sattari et al., Repositioning Therapeutics for COVID-19: Virtual Screening of the Potent Synthetic and Natural Compounds as SARS-CoV-2 3CLpro Inhibitors, Research Square, doi:10.21203/rs.3.rs-37994/v1.
7.
Setz et al., Synergistic Virus Neutralizing Activities of European Black Elderberry Fruit Extract and Iota-Carrageenan Against SARS-CoV-2, Influenza A Virus and Respiratory Syncytial Virus, Nutrients, doi:10.3390/nu18081205.
8.
Hoffmann et al., Controlling the Sulfation Density of Glycosaminoglycan Glycopolymer Mimetics Enables High Antiviral Activity against SARS-CoV-2 and Reduces Anticoagulant Activity, Biomacromolecules, doi:10.1021/acs.biomac.5c00576.
9.
Yathindranath et al., Lipid Nanoparticle-Based Inhibitors for SARS-CoV-2 Host Cell Infection, International Journal of Nanomedicine, doi:10.2147/IJN.S448005.
10.
Setz (B) et al., Iota-Carrageenan Inhibits Replication of the SARS-CoV-2 Variants of Concern Omicron BA.1, BA.2 and BA.5, Nutraceuticals, doi:10.3390/nutraceuticals3030025.
11.
Meister et al., Virucidal activity of nasal sprays against severe acute respiratory syndrome coronavirus-2, Journal of Hospital Infection, doi:10.1016/j.jhin.2021.10.019.
12.
Bovard et al., Iota-carrageenan extracted from red algae is a potent inhibitor of SARS-CoV-2 infection in reconstituted human airway epithelia, Biochemistry and Biophysics Reports, doi:10.1016/j.bbrep.2021.101187.
13.
Fröba et al., Iota-Carrageenan Inhibits Replication of SARS-CoV-2 and the Respective Variants of Concern Alpha, Beta, Gamma and Delta, International Journal of Molecular Sciences, doi:10.3390/ijms222413202.
14.
Varese et al., Iota-carrageenan prevents the replication of SARS-CoV-2 on an in vitro respiratory epithelium model, bioRxiv, doi:10.1101/2021.04.27.441512.
15.
Morokutti-Kurz et al., Iota-carrageenan neutralizes SARS-CoV-2 and inhibits viral replication in vitro, PLoS ONE, doi:10.1371/journal.pone.0237480.
16.
Song et al., Inhibitory activities of marine sulfated polysaccharides against SARS-CoV-2, Food & Function, doi:10.1039/D0FO02017F.
Bansal et al., 21 Aug 2020, peer-reviewed, 7 authors.
In vitro studies are an important part of preclinical research, however results may be very different in vivo.
Iota-carrageenan and xylitol inhibit SARS-CoV-2 in Vero cell culture
PLOS ONE, doi:10.1371/journal.pone.0259943
Last year observed a global pandemic caused by SARS-CoV-2 (severe acute respiratory syndrome-coronavirus 2) infection affecting millions of individuals worldwide. There is an urgent unmet need to provide an easily producible and affordable medicine to prevent transmission and provide early treatment for this disease. Since the nasal cavity and the rhinopharynx are the sites of initial replication of SARS-CoV-2, a nasal spray may be an effective option to target SARS-CoV-2 infection. In this study, we tested the antiviral action of three candidate nasal spray formulations against SARS-CoV-2 in vitro. We determined that iotacarrageenan in concentrations as low as 6 μg/mL inhibits SARS-CoV-2 in vitro. The concentrations of iota-carrageenan with activity against SARS-CoV-2 in vitro may be easily achieved through the application of nasal sprays as commonly used in several countries. Recently a double-blind, placebo-controlled study showed that iota-carrageenan in isotonic sodium chloride reduces ca. five times the risk of infection by SARS-CoV-2 in health care personnel. Further, xylitol at a concentration of 50 mg/mL (ca. 329 mM) was found to exert some antiviral action, though this preliminary finding needs further confirmation.
Supporting information S1 Table . Residual virus titer (TCID50/mL) after treatment with iota-carrageenan solutions in Diluent P1 (sodium chloride 9 mg/mL adjusted to pH 6-7). (PDF)
S2 Table. Statistical analysis of residual virus titers determined after each treatment with different concentrations of iota-carrageenan in
S8 Table. Statistical analysis of residual virus titers determined after each treatment with different concentrations of iota-carrageenan in Diluent P3 (xylitol 50 mg/mL adjusted to pH 6-7). (PDF)
S9 Table. Cell viability found by MTT assay after treatment with diluents and solutions of iota carrageenan without the addition of virus expressed as optical density and statistical analysis compared to untreated cells. (PDF) Author Contributions
References
Ahmadi, Moghadamtousi, Abubakar, Zandi, Antiviral Potential of Algae Polysaccharides Isolated from Marine Sources: A Review, Biomed Res Int, doi:10.1155/2015/825203
Bitter, Suter-Zimmermann, Surber, Nasal Drug Delivery in Humans, Curr Probl Dermatol, doi:10.1159/000321044
Buck, Thompson, Roberts, Mu ¨ller, Lowy et al., Carrageenan Is a Potent Inhibitor of Papillomavirus Infection, Plos Pathog, doi:10.1371/journal.ppat.0020069
Callahan, Lee, Lee, Zulauf, Kirby et al., Nasal-Swab Testing Misses Patients with Low SARS-CoV-2 Viral Loads, Medrxiv, doi:10.1101/2020.06.12.20128736
Dong, Du, Gardner, An interactive web-based dashboard to track COVID-19 in real time, Lancet Infect Dis, doi:10.1016/S1473-3099(20)30120-1
Durairaj, Launspach, Watt, Businga, Kline et al., Safety assessment of inhaled xylitol in mice and healthy volunteers, Respir Res, doi:10.1186/1465-9921-5-13
Eccles, Iota-Carrageenan as an Antiviral Treatment for the Common Cold, Open Virology J, doi:10.2174/1874357902014010009
Eccles, Meier, Jawad, Weinmu ¨llner R, Grassauer et al., Efficacy and safety of an antiviral Iota-Carrageenan nasal spray: a randomized, double-blind, placebo-controlled exploratory study in volunteers with early symptoms of the common cold, Respir Res, doi:10.1186/1465-9921-11-108
Fazekas, Eickhoff, Pruckner, Vollnhofer, Fischmeister et al., Lessons learned from a double-blind randomised placebo-controlled study with a iota-carrageenan nasal spray as medical device in children with acute symptoms of common cold, Bmc Complem Altern M, doi:10.1186/1472-6882-12-147
Figueroa, Lombardo, Dogliotti, Flynn, Giugliano et al., Efficacy of a Nasal Spray Containing Iota-Carrageenan in the Postexposure Prophylaxis of COVID-19 in Hospital Personnel Dedicated to Patients Care with COVID-19 Disease, Int J Gen Medicine, doi:10.2147/IJGM.S328486
Garcia, Schroeter, Segal, Stanek, Foureman et al., Dosimetry of nasal uptake of water-soluble and reactive gases: A first study of interhuman variability, Inhal Toxicol, doi:10.1080/08958370802320186
Girond, Crance, Cuyck-Gandre, Renaudet, Deloince, Antiviral activity of carrageenan on hepatitis A virus replication in cell culture, Res Virology, doi:10.1016/0923-2516(91)90011-q
Gizurarson, Anatomical and Histological Factors Affecting Intranasal Drug and Vaccine Delivery, Curr Drug Deliv, doi:10.2174/156720112803529828
Graf, Bernkop-Schnu ¨rch, Egyed, Koller, Prieschl-Grassauer et al., Development of a nasal spray containing xylometazoline hydrochloride and iota-carrageenan for the symptomatic relief of nasal congestion caused by rhinitis and sinusitis, Int J Gen Medicine, doi:10.2147/IJGM.S167123
Grassauer, Prieschl-Grassauer, Bodenteich, Koller, Morokutti-Kurz, Stuffy nose deblocking composition having antiviral activity-Google Patents
Grassauer, Weinmuellner, Meier, Pretsch, Prieschl-Grassauer et al., Iota-Carrageenan is a potent inhibitor of rhinovirus infection, Virol J, doi:10.1186/1743-422X-5-107
Hebar, Koller, Seifert, Chabicovsky, Bodenteich et al., Non-Clinical Safety Evaluation of Intranasal Iota-Carrageenan, Plos One, doi:10.1371/journal.pone.0122911
Helassa, Garnett, Farrant, Khan, Pickup et al., A novel fluorescent sensor protein for detecting changes in airway surface liquid glucose concentration, Biochem J, doi:10.1042/BJ20141041
Kelvin, Halperin, COVID-19 in children: the link in the transmission chain, Lancet Infect Dis, doi:10.1016/S1473-3099(20)30236-X
Koenighofer, Lion, Bodenteich, Prieschl-Grassauer, Grassauer et al., Carrageenan nasal spray in virus confirmed common cold: individual patient data analysis of two randomized controlled trials, Multidiscip Resp Med, doi:10.1186/2049-6958-9-57
Leibbrandt, Meier, Ko ¨nig-Schuster, Weinmu ¨llner R, Kalthoff et al., Iota-Carrageenan Is a Potent Inhibitor of Influenza A Virus Infection, Plos One, doi:10.1371/journal.pone.0014320
Lin, Tang, Wei, Dai, Sun, Xylitol nasal irrigation in the treatment of chronic rhinosinusitis, Am J Otolaryng, doi:10.1016/j.amjoto.2017.03.006
Liu, Liao, Wan, Xiang, Zhang, Correlation Between Relative Nasopharyngeal Virus RNA Load and Lymphocyte Count Disease Severity in Patients with COVID-19, Viral Immunol, doi:10.1089/vim.2020.0062
Liu, Yan, Xiang, Liu, Viral dynamics in mild and severe cases of COVID-19, Lancet Infect Dis, doi:10.1016/S1473-3099(20)30232-2
Ludwig, Enzenhofer, Schneider, Rauch, Bodenteich et al., Efficacy of a Carrageenan nasal spray in patients with common cold: a randomized controlled trial, Respir Res, doi:10.1186/1465-9921-14-124
Mathieu, Ritchie, Ortiz-Ospina, Roser, Hasell et al., A global database of COVID-19 vaccinations, Nat Hum Behav, doi:10.1038/s41562-021-01122-8
Mendoza, Manguiat, Wood, Drebot, Two Detailed Plaque Assay Protocols for the Quantification of Infectious SARS-CoV-2, Curr Protoc Microbiol, doi:10.1002/cpmc.105
Morokutti-Kurz, Ko ¨nig-Schuster, Koller, Graf, Graf et al., The Intranasal Application of Zanamivir and Carrageenan Is Synergistically Active against Influenza A Virus in the Murine Model, Plos One, doi:10.1371/journal.pone.0128794
Morokutti-Kurz, ¨ba, Graf, Große, Grassauer et al., Iota-carrageenan neutralizes SARS-CoV-2 and inhibits viral replication in vitro, Plos One, doi:10.1371/journal.pone.0237480
Pires, Fortuna, Alves, Falcão, Intranasal Drug Delivery: How, Why and What for?, J Pharm Pharm Sci, doi:10.18433/j3nc79
Reed, Muench, A simple method of estimating fifty per cent endpoints, Am J Epidemiol, doi:10.1093/oxfordjournals.aje.a118408
Sakallioğlu, Gu ¨venc, Cingi, Xylitol and its usage in ENT practice, J Laryngology X0026 Otology, doi:10.1017/S0022215114001340
Salli, Lehtinen, Tiihonen, Ouwehand, Xylitol's Health Benefits beyond Dental Health: A Comprehensive Review, Nutrients, doi:10.3390/nu11081813
Schu ¨tz, Conzelmann, Fois, Groß, Weil et al., Carrageenan-containing over-thecounter nasal and oral sprays inhibit SARS-CoV-2 infection of airway epithelial cultures, Am J Physiollung C, doi:10.1152/ajplung.00552.2020
Shao, Guo, Xu W Ping, Li, Zhao et al., Specific Inhibitory Effect of κ-Carrageenan Polysaccharide on Swine Pandemic 2009 H1N1 Influenza Virus, Plos One, doi:10.1371/journal.pone.0126577
Talarico, Damonte, Interference in dengue virus adsorption and uncoating by carrageenans, Virology, doi:10.1016/j.virol.2007.01.043
Wagenmann, Naclerio, Anatomic and physiologic considerations in sinusitis, J Allergy Clin Immun, doi:10.1016/0091-6749(92)90161-t
Weissman, Fernandez, Hwang, Xylitol nasal irrigation in the management of chronic rhinosinusitis: A pilot study, Laryngoscope, doi:10.1002/lary.22176
Wu, Mcgoogan, Characteristics of and Important Lessons From the Coronavirus Disease 2019 (COVID-19) Outbreak in China, Jama, doi:10.1001/jama.2020.2648
Xu, Wi, Kim, Kim, Ameliorating Effect of Dietary Xylitol on Human Respiratory Syncytial Virus (hRSV) Infection, Biological Pharm Bulletin, doi:10.1248/bpb.b15-00773
Zou, Ruan, Huang, Liang, Huang et al., SARS-CoV-2 Viral Load in Upper Respiratory Specimens of Infected Patients, New Engl J Med, doi:10.1056/NEJMc2001737
DOI record:
{
"DOI": "10.1371/journal.pone.0259943",
"ISSN": [
"1932-6203"
],
"URL": "http://dx.doi.org/10.1371/journal.pone.0259943",
"abstract": "<jats:p>Last year observed a global pandemic caused by SARS-CoV-2 (severe acute respiratory syndrome-coronavirus 2) infection affecting millions of individuals worldwide. There is an urgent unmet need to provide an easily producible and affordable medicine to prevent transmission and provide early treatment for this disease. Since the nasal cavity and the rhinopharynx are the sites of initial replication of SARS-CoV-2, a nasal spray may be an effective option to target SARS-CoV-2 infection. In this study, we tested the antiviral action of three candidate nasal spray formulations against SARS-CoV-2<jats:italic>in vitro</jats:italic>. We determined that iota-carrageenan in concentrations as low as 6 μg/mL inhibits SARS-CoV-2<jats:italic>in vitro</jats:italic>. The concentrations of iota-carrageenan with activity against SARS-CoV-2<jats:italic>in vitro</jats:italic>may be easily achieved through the application of nasal sprays as commonly used in several countries. Recently a double-blind, placebo-controlled study showed that iota-carrageenan in isotonic sodium chloride reduces ca. five times the risk of infection by SARS-CoV-2 in health care personnel. Further, xylitol at a concentration of 50 mg/mL (ca. 329 mM) was found to exert some antiviral action, though this preliminary finding needs further confirmation.</jats:p>",
"author": [
{
"affiliation": [],
"family": "Bansal",
"given": "Shruti",
"sequence": "first"
},
{
"affiliation": [],
"family": "Jonsson",
"given": "Colleen B.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Taylor",
"given": "Shannon L.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Figueroa",
"given": "Juan Manuel",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-0187-0548",
"affiliation": [],
"authenticated-orcid": true,
"family": "Dugour",
"given": "Andrea Vanesa",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-3368-0538",
"affiliation": [],
"authenticated-orcid": true,
"family": "Palacios",
"given": "Carlos",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-9735-0135",
"affiliation": [],
"authenticated-orcid": true,
"family": "Vega",
"given": "Julio César",
"sequence": "additional"
}
],
"container-title": "PLOS ONE",
"container-title-short": "PLoS ONE",
"content-domain": {
"crossmark-restriction": false,
"domain": [
"www.plosone.org"
]
},
"created": {
"date-parts": [
[
2021,
11,
19
]
],
"date-time": "2021-11-19T18:33:01Z",
"timestamp": 1637346781000
},
"deposited": {
"date-parts": [
[
2023,
1,
15
]
],
"date-time": "2023-01-15T21:36:32Z",
"timestamp": 1673818592000
},
"editor": [
{
"affiliation": [],
"family": "Polyak",
"given": "Stephen J.",
"sequence": "first"
}
],
"funder": [
{
"name": "Amcyte Pharma Inc."
},
{
"name": "Laboratorio Pablo Cassara"
},
{
"name": "Fundación Pablo Cassará"
},
{
"name": "Fundación Pablo Cassará"
},
{
"name": "Fundación Pablo Cassará"
}
],
"indexed": {
"date-parts": [
[
2024,
4,
23
]
],
"date-time": "2024-04-23T10:20:56Z",
"timestamp": 1713867656160
},
"is-referenced-by-count": 17,
"issue": "11",
"issued": {
"date-parts": [
[
2021,
11,
19
]
]
},
"journal-issue": {
"issue": "11",
"published-online": {
"date-parts": [
[
2021,
11,
19
]
]
}
},
"language": "en",
"license": [
{
"URL": "http://creativecommons.org/licenses/by/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
11,
19
]
],
"date-time": "2021-11-19T00:00:00Z",
"timestamp": 1637280000000
}
}
],
"link": [
{
"URL": "https://dx.plos.org/10.1371/journal.pone.0259943",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "340",
"original-title": [],
"page": "e0259943",
"prefix": "10.1371",
"published": {
"date-parts": [
[
2021,
11,
19
]
]
},
"published-online": {
"date-parts": [
[
2021,
11,
19
]
]
},
"publisher": "Public Library of Science (PLoS)",
"reference": [
{
"DOI": "10.1016/S1473-3099(20)30120-1",
"article-title": "An interactive web-based dashboard to track COVID-19 in real time",
"author": "E Dong",
"doi-asserted-by": "crossref",
"first-page": "533",
"journal-title": "Lancet Infect Dis. 2020",
"key": "pone.0259943.ref001",
"volume": "20"
},
{
"DOI": "10.1001/jama.2020.2648",
"article-title": "Characteristics of and Important Lessons From the Coronavirus Disease 2019 (COVID-19) Outbreak in China",
"author": "Z Wu",
"doi-asserted-by": "crossref",
"first-page": "1239",
"journal-title": "Jama",
"key": "pone.0259943.ref002",
"volume": "323",
"year": "2020"
},
{
"DOI": "10.1016/S1473-3099(20)30236-X",
"article-title": "COVID-19 in children: the link in the transmission chain",
"author": "AA Kelvin",
"doi-asserted-by": "crossref",
"first-page": "633",
"journal-title": "Lancet Infect Dis",
"key": "pone.0259943.ref003",
"volume": "20",
"year": "2020"
},
{
"DOI": "10.1038/s41562-021-01122-8",
"article-title": "A global database of COVID-19 vaccinations",
"author": "E Mathieu",
"doi-asserted-by": "crossref",
"first-page": "947",
"journal-title": "Nat Hum Behav",
"key": "pone.0259943.ref004",
"volume": "5",
"year": "2021"
},
{
"DOI": "10.1089/vim.2020.0062",
"article-title": "Correlation Between Relative Nasopharyngeal Virus RNA Load and Lymphocyte Count Disease Severity in Patients with COVID-19",
"author": "Y Liu",
"doi-asserted-by": "crossref",
"first-page": "330",
"journal-title": "Viral Immunol",
"key": "pone.0259943.ref005",
"volume": "34",
"year": "2021"
},
{
"DOI": "10.1016/S1473-3099(20)30232-2",
"article-title": "Viral dynamics in mild and severe cases of COVID-19",
"author": "Y Liu",
"doi-asserted-by": "crossref",
"first-page": "656",
"journal-title": "Lancet Infect Dis",
"key": "pone.0259943.ref006",
"volume": "20",
"year": "2020"
},
{
"DOI": "10.1056/NEJMc2001737",
"article-title": "SARS-CoV-2 Viral Load in Upper Respiratory Specimens of Infected Patients",
"author": "L Zou",
"doi-asserted-by": "crossref",
"first-page": "1177",
"journal-title": "New Engl J Med",
"key": "pone.0259943.ref007",
"volume": "382",
"year": "2020"
},
{
"article-title": "Nasal-Swab Testing Misses Patients with Low SARS-CoV-2 Viral Loads",
"author": "C Callahan",
"journal-title": "Medrxiv",
"key": "pone.0259943.ref008",
"year": "2020"
},
{
"DOI": "10.1186/1465-9921-11-108",
"article-title": "Efficacy and safety of an antiviral Iota-Carrageenan nasal spray: a randomized, double-blind, placebo-controlled exploratory study in volunteers with early symptoms of the common cold",
"author": "R Eccles",
"doi-asserted-by": "crossref",
"first-page": "108",
"journal-title": "Respir Res",
"key": "pone.0259943.ref009",
"volume": "11",
"year": "2010"
},
{
"DOI": "10.1186/2049-6958-9-57",
"article-title": "Carrageenan nasal spray in virus confirmed common cold: individual patient data analysis of two randomized controlled trials",
"author": "M Koenighofer",
"doi-asserted-by": "crossref",
"first-page": "57",
"journal-title": "Multidiscip Resp Med",
"key": "pone.0259943.ref010",
"volume": "9",
"year": "2014"
},
{
"DOI": "10.1186/1465-9921-14-124",
"article-title": "Efficacy of a Carrageenan nasal spray in patients with common cold: a randomized controlled trial",
"author": "M Ludwig",
"doi-asserted-by": "crossref",
"first-page": "124",
"journal-title": "Respir Res",
"key": "pone.0259943.ref011",
"volume": "14",
"year": "2013"
},
{
"DOI": "10.1155/2015/825203",
"article-title": "Antiviral Potential of Algae Polysaccharides Isolated from Marine Sources",
"author": "A Ahmadi",
"doi-asserted-by": "crossref",
"first-page": "1",
"journal-title": "A Review. Biomed Res Int",
"key": "pone.0259943.ref012",
"volume": "2015",
"year": "2015"
},
{
"DOI": "10.1371/journal.ppat.0020069",
"article-title": "Carrageenan Is a Potent Inhibitor of Papillomavirus Infection",
"author": "CB Buck",
"doi-asserted-by": "crossref",
"first-page": "e69",
"journal-title": "Plos Pathog",
"key": "pone.0259943.ref013",
"volume": "2",
"year": "2006"
},
{
"DOI": "10.1016/0923-2516(91)90011-Q",
"article-title": "Antiviral activity of carrageenan on hepatitis A virus replication in cell culture",
"author": "S Girond",
"doi-asserted-by": "crossref",
"first-page": "261",
"journal-title": "Res Virology",
"key": "pone.0259943.ref014",
"volume": "142",
"year": "1991"
},
{
"DOI": "10.1186/1743-422X-5-107",
"article-title": "Iota-Carrageenan is a potent inhibitor of rhinovirus infection",
"author": "A Grassauer",
"doi-asserted-by": "crossref",
"first-page": "107",
"journal-title": "Virol J",
"key": "pone.0259943.ref015",
"volume": "5",
"year": "2008"
},
{
"DOI": "10.1371/journal.pone.0126577",
"article-title": "Specific Inhibitory Effect of κ-Carrageenan Polysaccharide on Swine Pandemic 2009 H1N1 Influenza Virus",
"author": "Q Shao",
"doi-asserted-by": "crossref",
"first-page": "e0126577",
"journal-title": "Plos One",
"key": "pone.0259943.ref016",
"volume": "10",
"year": "2015"
},
{
"DOI": "10.1016/j.virol.2007.01.043",
"article-title": "Interference in dengue virus adsorption and uncoating by carrageenans",
"author": "LB Talarico",
"doi-asserted-by": "crossref",
"first-page": "473",
"journal-title": "Virology",
"key": "pone.0259943.ref017",
"volume": "363",
"year": "2007"
},
{
"DOI": "10.1371/journal.pone.0014320",
"article-title": "Iota-Carrageenan Is a Potent Inhibitor of Influenza A Virus Infection",
"author": "A Leibbrandt",
"doi-asserted-by": "crossref",
"first-page": "e14320",
"journal-title": "Plos One",
"key": "pone.0259943.ref018",
"volume": "5",
"year": "2010"
},
{
"DOI": "10.1186/1472-6882-12-147",
"article-title": "Lessons learned from a double-blind randomised placebo-controlled study with a iota-carrageenan nasal spray as medical device in children with acute symptoms of common cold",
"author": "T Fazekas",
"doi-asserted-by": "crossref",
"first-page": "147",
"journal-title": "Bmc Complem Altern M",
"key": "pone.0259943.ref019",
"volume": "12",
"year": "2012"
},
{
"DOI": "10.2147/IJGM.S167123",
"article-title": "Development of a nasal spray containing xylometazoline hydrochloride and iota-carrageenan for the symptomatic relief of nasal congestion caused by rhinitis and sinusitis",
"author": "C Graf",
"doi-asserted-by": "crossref",
"first-page": "275",
"journal-title": "Int J Gen Medicine",
"key": "pone.0259943.ref020",
"volume": "11",
"year": "2018"
},
{
"DOI": "10.1371/journal.pone.0128794",
"article-title": "The Intranasal Application of Zanamivir and Carrageenan Is Synergistically Active against Influenza A Virus in the Murine Model",
"author": "M Morokutti-Kurz",
"doi-asserted-by": "crossref",
"first-page": "e0128794",
"journal-title": "Plos One",
"key": "pone.0259943.ref021",
"volume": "10",
"year": "2015"
},
{
"DOI": "10.3390/nu11081813",
"article-title": "Xylitol’s Health Benefits beyond Dental Health: A Comprehensive Review",
"author": "K Salli",
"doi-asserted-by": "crossref",
"first-page": "1813",
"journal-title": "Nutrients",
"key": "pone.0259943.ref022",
"volume": "11",
"year": "2019"
},
{
"DOI": "10.1017/S0022215114001340",
"article-title": "Xylitol and its usage in ENT practice",
"author": "Ö Sakallioğlu",
"doi-asserted-by": "crossref",
"first-page": "580",
"journal-title": "J Laryngology X0026 Otology",
"key": "pone.0259943.ref023",
"volume": "128",
"year": "2014"
},
{
"DOI": "10.1016/j.amjoto.2017.03.006",
"article-title": "Xylitol nasal irrigation in the treatment of chronic rhinosinusitis",
"author": "L Lin",
"doi-asserted-by": "crossref",
"first-page": "383",
"journal-title": "Am J Otolaryng",
"key": "pone.0259943.ref024",
"volume": "38",
"year": "2017"
},
{
"DOI": "10.1248/bpb.b15-00773",
"article-title": "Ameliorating Effect of Dietary Xylitol on Human Respiratory Syncytial Virus (hRSV) Infection",
"author": "ML Xu",
"doi-asserted-by": "crossref",
"first-page": "540",
"journal-title": "Biological Pharm Bulletin",
"key": "pone.0259943.ref025",
"volume": "39",
"year": "2016"
},
{
"DOI": "10.1371/journal.pone.0122911",
"article-title": "Non-Clinical Safety Evaluation of Intranasal Iota-Carrageenan",
"author": "A Hebar",
"doi-asserted-by": "crossref",
"first-page": "e0122911",
"journal-title": "Plos One",
"key": "pone.0259943.ref026",
"volume": "10",
"year": "2015"
},
{
"DOI": "10.1002/lary.22176",
"article-title": "Xylitol nasal irrigation in the management of chronic rhinosinusitis: A pilot study",
"author": "JD Weissman",
"doi-asserted-by": "crossref",
"first-page": "2468",
"journal-title": "Laryngoscope",
"key": "pone.0259943.ref027",
"volume": "121",
"year": "2011"
},
{
"DOI": "10.1186/1465-9921-5-13",
"article-title": "Safety assessment of inhaled xylitol in mice and healthy volunteers",
"author": "L Durairaj",
"doi-asserted-by": "crossref",
"first-page": "13",
"journal-title": "Respir Res",
"key": "pone.0259943.ref028",
"volume": "5",
"year": "2004"
},
{
"DOI": "10.2174/1874357902014010009",
"article-title": "Iota-Carrageenan as an Antiviral Treatment for the Common Cold",
"author": "R Eccles",
"doi-asserted-by": "crossref",
"first-page": "9",
"journal-title": "Open Virology J",
"key": "pone.0259943.ref029",
"volume": "14",
"year": "2020"
},
{
"article-title": "Stuffy nose deblocking composition having antiviral activity—Google Patents",
"author": "A Grassauer",
"first-page": "1",
"journal-title": "US10660914B2",
"key": "pone.0259943.ref030",
"year": "2020"
},
{
"DOI": "10.1002/cpmc.105",
"article-title": "Two Detailed Plaque Assay Protocols for the Quantification of Infectious SARS-CoV-2",
"author": "EJ Mendoza",
"doi-asserted-by": "crossref",
"journal-title": "Curr Protoc Microbiol",
"key": "pone.0259943.ref031",
"volume": "57",
"year": "2020"
},
{
"DOI": "10.1093/oxfordjournals.aje.a118408",
"article-title": "A simple method of estimating fifty per cent endpoints",
"author": "LJ Reed",
"doi-asserted-by": "crossref",
"first-page": "493",
"journal-title": "Am J Epidemiol",
"key": "pone.0259943.ref032",
"volume": "27",
"year": "1938"
},
{
"DOI": "10.1371/journal.pone.0237480",
"article-title": "Iota-carrageenan neutralizes SARS-CoV-2 and inhibits viral replication in vitro",
"author": "M Morokutti-Kurz",
"doi-asserted-by": "crossref",
"first-page": "e0237480",
"journal-title": "Plos One",
"key": "pone.0259943.ref033",
"volume": "16",
"year": "2021"
},
{
"DOI": "10.1152/ajplung.00552.2020",
"article-title": "Carrageenan-containing over-the-counter nasal and oral sprays inhibit SARS-CoV-2 infection of airway epithelial cultures",
"author": "D Schütz",
"doi-asserted-by": "crossref",
"first-page": "L750",
"journal-title": "Am J Physiol-lung C",
"key": "pone.0259943.ref034",
"volume": "320",
"year": "2021"
},
{
"DOI": "10.1159/000321044",
"article-title": "Nasal Drug Delivery in Humans",
"author": "C Bitter",
"doi-asserted-by": "crossref",
"first-page": "20",
"journal-title": "Curr Probl Dermatol",
"key": "pone.0259943.ref035",
"volume": "40",
"year": "2011"
},
{
"DOI": "10.1080/08958370802320186",
"article-title": "Dosimetry of nasal uptake of water-soluble and reactive gases: A first study of interhuman variability",
"author": "GJM Garcia",
"doi-asserted-by": "crossref",
"first-page": "607",
"journal-title": "Inhal Toxicol",
"key": "pone.0259943.ref036",
"volume": "21",
"year": "2009"
},
{
"DOI": "10.2174/156720112803529828",
"article-title": "Anatomical and Histological Factors Affecting Intranasal Drug and Vaccine Delivery",
"author": "S Gizurarson",
"doi-asserted-by": "crossref",
"first-page": "566",
"journal-title": "Curr Drug Deliv",
"key": "pone.0259943.ref037",
"volume": "9",
"year": "2012"
},
{
"DOI": "10.18433/J3NC79",
"article-title": "Intranasal Drug Delivery: How, Why and What for?",
"author": "A Pires",
"doi-asserted-by": "crossref",
"first-page": "288",
"journal-title": "J Pharm Pharm Sci",
"key": "pone.0259943.ref038",
"volume": "12",
"year": "2009"
},
{
"DOI": "10.1042/BJ20141041",
"article-title": "A novel fluorescent sensor protein for detecting changes in airway surface liquid glucose concentration",
"author": "N Helassa",
"doi-asserted-by": "crossref",
"first-page": "213",
"journal-title": "Biochem J",
"key": "pone.0259943.ref039",
"volume": "464",
"year": "2014"
},
{
"DOI": "10.1016/0091-6749(92)90161-T",
"article-title": "Anatomic and physiologic considerations in sinusitis",
"author": "M Wagenmann",
"doi-asserted-by": "crossref",
"first-page": "419",
"journal-title": "J Allergy Clin Immun",
"key": "pone.0259943.ref040",
"volume": "90",
"year": "1992"
},
{
"DOI": "10.1016/j.rmed.2020.105987",
"article-title": "Practical strategies for a safe and effective delivery of aerosolized medications to patients with COVID-19",
"author": "A Ari",
"doi-asserted-by": "crossref",
"first-page": "105987",
"journal-title": "Resp Med",
"key": "pone.0259943.ref041",
"volume": "167",
"year": "2020"
},
{
"DOI": "10.2147/IJGM.S328486",
"article-title": "Efficacy of a Nasal Spray Containing Iota-Carrageenan in the Postexposure Prophylaxis of COVID-19 in Hospital Personnel Dedicated to Patients Care with COVID-19 Disease",
"author": "JM Figueroa",
"doi-asserted-by": "crossref",
"first-page": "6277",
"journal-title": "Int J Gen Medicine",
"key": "pone.0259943.ref042",
"volume": "14",
"year": "2021"
}
],
"reference-count": 42,
"references-count": 42,
"relation": {},
"resource": {
"primary": {
"URL": "https://dx.plos.org/10.1371/journal.pone.0259943"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "Iota-carrageenan and xylitol inhibit SARS-CoV-2 in Vero cell culture",
"type": "journal-article",
"update-policy": "http://dx.doi.org/10.1371/journal.pone.corrections_policy",
"volume": "16"
}