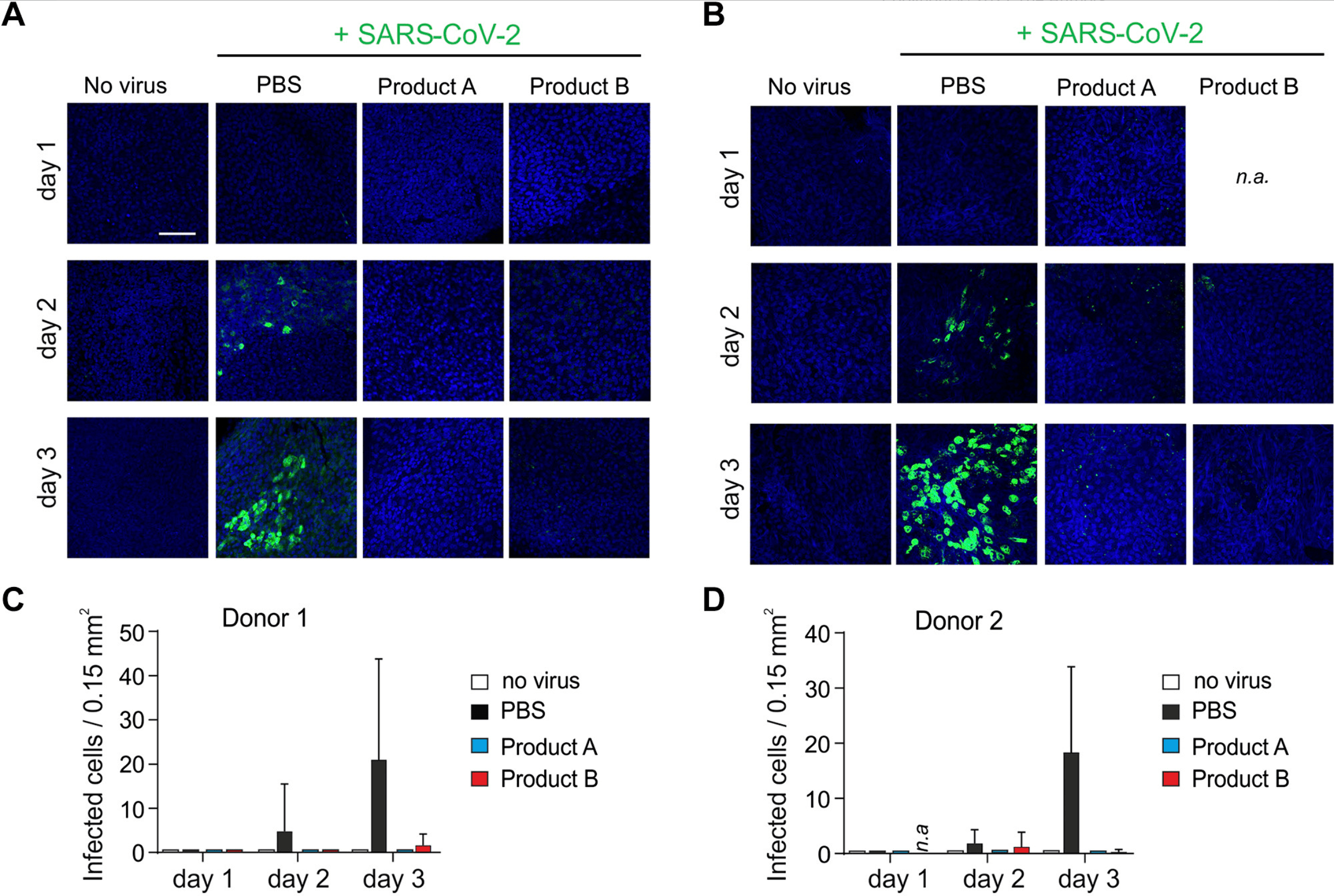
Carrageenan-containing over-the-counter nasal and oral sprays inhibit SARS-CoV-2 infection of airway epithelial cultures
et al., American Journal of Physiology: Lung Cellular and Molecular Physiology, doi:10.1152/ajplung.00552.2020, Apr 2021
In vitro study showing that carrageenan-containing nose spray and mouth spray inhibit SARS-CoV-2 in human airway epithelial cultures.
Schütz et al., 28 Apr 2021, peer-reviewed, 12 authors.
Carrageenan-containing over-the-counter nasal and oral sprays inhibit SARS-CoV-2 infection of airway epithelial cultures
American Journal of Physiology-Lung Cellular and Molecular Physiology, doi:10.1152/ajplung.00552.2020
Pharmaceutical interventions are urgently needed to prevent severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection and transmission. As SARS-CoV-2 infects and spreads via the nasopharyngeal airways, we analyzed the antiviral effect of selected nasal and oral sprays on virus infection in vitro. Two nose sprays showed virucidal activity but were cytotoxic precluding further analysis in cell culture. One nasal and one mouth spray suppressed SARS-CoV-2 infection of TMPRSS2-expressing Vero E6 cells and primary differentiated human airway epithelial cultures. The antiviral activity in both sprays could be attributed to polyanionic iand κ-carrageenans. Thus, application of carrageenan-containing nasal and mouth sprays may reduce the risk of acquiring SARS-CoV-2 infection and may limit viral spread, warranting further clinical evaluation.
References
Anderson, Sivalingam, Kang, Ananthanarayanan, Arumugam et al., Povidone-iodine demonstrates rapid in vitro virucidal activity against SARS-CoV-2, the virus causing COVID-19 disease, Infect Dis Ther, doi:10.1007/s40121-020-00316-3
Armstrong, Froelich, Inactivation of viruses by benzalkonium chloride, Appl Microbiol, doi:10.1128/AM.12.2.132-137.1964
Bansal, Jonsson, Taylor, Figueroa, Dugour et al., Iota-carrageenan and Xylitol inhibit SARS-CoV-2 in cell culture, bioRxiv, doi:10.1101/2020.08.19.225854
Buck, Thompson, Roberts, M€ Uller, Lowy et al., Carrageenan is a potent inhibitor of papillomavirus infection, PLoS Pathog, doi:10.1371/journal.ppat.0020069
Choi, Lee, Lim, Kim, Lee et al., Assessment of respiratory and systemic toxicity of Benzalkonium chloride following a 14-day inhalation study in rats, Part Fibre Toxicol, doi:10.1186/s12989-020-0339-8
Conzelmann, Gilg, Groß, Sch€ Utz, Preising et al., An enzyme-based immunodetection assay to quantify SARS-CoV-2 infection, Antiviral Res, doi:10.1016/j.antiviral.2020.104882
Dawes, Salivary flow patterns and the health of hard and soft oral tissues, J Am Dent Assoc, doi:10.14219/jada.archive.2008.0351
Deckx, Sutter, Guo, Mir, Van Driel, Nasal decongestants in monotherapy for the common cold, Cochrane Database Syst Rev, doi:10.1002/14651858.CD009612.pub2
Detalle, Stohr, Palomo, Piedra, Gilbert et al., Generation and characterization of ALX-0171, a potent novel therapeutic nanobody for the treatment of respiratory syncytial virus infection, Antimicrob Agents Chemother, doi:10.1128/AAC.01802-15
Djupesland, Skretting, Nasal deposition and clearance in man: comparison of a bidirectional powder device and a traditional liquid spray pump, J Aerosol Med Pulm Drug Deliv, doi:10.1089/jamp.2011.0924
Eccles, Meier, Jawad, Weinm€ Ullner R, Grassauer et al., Efficacy and safety of an antiviral Iota-Carrageenan nasal spray: a randomized, double-blind, placebo-controlled exploratory study in volunteers with early symptoms of the common cold, Respir Res, doi:10.1186/1465-9921-11-108
Eccles, Winther, Johnston, Robinson, Trampisch et al., Efficacy and safety of iota-carrageenan nasal spray versus placebo in early treatment of the common cold in adults: the ICICC trial, Respir Res, doi:10.1186/s12931-015-0281-8
Fazekas, Eickhoff, Pruckner, Vollnhofer, Fischmeister et al., Lessons learned from a double-blind randomised placebo-controlled study with a iota-carrageenan nasal spray as medical device in children with acute symptoms of common cold, BMC Complement Altern Med, doi:10.1186/1472-6882-12-147
Girond, Crance, Van Cuyck-Gandre, Renaudet, Deloince, Antiviral activity of carrageenan on hepatitis A virus replication in cell culture, Res Virol, doi:10.1016/0923-2516(91)90011-Q
Graf, Bernkop-Schn€ Urch, Egyed, Koller, Prieschl-Grassauer et al., Development of a nasal spray containing xylometazoline hydrochloride and iota-carrageenan for the symptomatic relief of nasal congestion caused by rhinitis and sinusitis, Int J Gen Med, doi:10.2147/IJGM.S167123
Grassauer, Weinmuellner, Meier, Pretsch, Prieschl-Grassauer et al., Iota-Carrageenan is a potent inhibitor of rhinovirus infection, Virol J, doi:10.1186/1743-422X-5-107
Gupta, Madhavan, Sehgal, Nair, Mahajan et al., Extrapulmonary manifestations of COVID-19, Nat Med, doi:10.1038/s41591-020-0968-3
Hou, Okuda, Edwards, Martinez, Asakura et al., SARS-CoV-2 reverse genetics reveals a variable infection gradient in the respiratory tract, Cell, doi:10.1016/j.cell.2020.05.042
Huang, Lin, Wu, Wu, Assessment of the inhibition of Dengue virus infection by carrageenan via real-time monitoring of cellular oxygen consumption rates within a microfluidic device, Biomicrofluidics, doi:10.1063/1.4870772
Hui, Povidone-iodine and carrageenan are candidates for SARS-CoV-2 infection control, Hong Kong Med J, doi:10.12809/hkmj208889
Hunter, Saliva and oral health, 4th edition, Br Dent J, doi:10.1038/sj.bdj.2013.421
Illum, Nasal drug delivery-possibilities, problems and solutions, J Control Release, doi:10.1016/S0168-3659(02)00363-2
Jang, Shin, Lee, Kwon, Shin et al., Antiviral activity of lambda-carrageenan against influenza viruses in mice and severe acute respiratory syndrome coronavirus 2, Sci Rep, doi:10.1038/s41598-020-80896-9
Jiao, Zhang, Influence of intranasal drugs on human nasal mucociliary clearance and ciliary beat frequency, Allergy Asthma Immunol Res, doi:10.4168/aair.2019.11.3.306
Johnson, Pulmonary toxicity of benzalkonium chloride, J Aerosol Med Pulm Drug Deliv, doi:10.1089/jamp.2017.1390
Koenighofer, Lion, Bodenteich, Prieschl-Grassauer, Grassauer et al., Carrageenan nasal spray in virus confirmed common cold: individual patient data analysis of two randomized controlled trials, Multidiscip Respir Med, doi:10.1186/2049-6958-9-57
Kublik, Vidgren, Nasal delivery systems and their effect on deposition and absorption, Adv Drug Deliv Rev, doi:10.1016/S0169-409X(97)00067-7
Leibbrandt, Meier, K€ Onig-Schuster, Weinm€ Ullner R, Kalthoff et al., Iota-carrageenan is a potent inhibitor of influenza A virus infection, PLoS One, doi:10.1371/journal.pone.0014320
Little, Read, Amlôt, Chadborn, Rice et al., Reducing risks from coronavirus transmission in the home-the role of viral load, BMJ
Ludwig, Enzenhofer, Schneider, Rauch, Bodenteich et al., Efficacy of a carrageenan nasal spray in patients with common cold: a randomized controlled trial, Respir Res, doi:10.1186/1465-9921-14-124
Matza-Porges, Eisen, Ibrahim, Haberman, Fridlender et al., A new antiviral screening method that simultaneously detects viral replication, cell viability, and cell toxicity, J Virol Methods, doi:10.1016/j.jviromet.2014.08.007
Meister, Br€ Uggemann, Todt, Conzelmann, M€ Uller et al., Virucidal efficacy of different oral rinses against severe acute respiratory syndrome coronavirus 2, J Infect Dis, doi:10.1093/infdis/jiaa471
Meselson, Droplets and aerosols in the transmission of SARS-CoV-2, N Engl J Med, doi:10.1056/NEJMc2009324
Moakes, Davies, Stamataki, Grover, Formulation of a composite nasal spray enabling enhanced surface coverage and prophylaxis of SARS-CoV-2, bioRxiv, doi:10.1101/2020.11.18.388645
Morokutti-Kurz, Graf, Grassauer, Prieschl-Grassauer, SARS-CoV-2 in-vitro neutralization assay reveals inhibition of virus entry by iota-carrageenan, bioRxiv, doi:10.1101/2020.07.28.224733
Morokutti-Kurz, Graf, Prieschl-Grassauer, Amylmetacresol/ 2,4-dichlorobenzyl alcohol, hexylresorcinol, or carrageenan lozenges as active treatments for sore throat, Int J Gen Med, doi:10.2147/IJGM.S120665
M€ Osges, Shah-Hosseini, Hucke, Joisten, Dexpanthenol: an overview of its contribution to symptom relief in acute rhinitis treated with decongestant nasal sprays, Adv Ther, doi:10.1007/s12325-017-0581-0
M€ Uller, Groß, Conzelmann, Kr€ Uger, Koepke et al., SARS-CoV-2 infects and replicates in cells of the human endocrine and exocrine pancreas, Nat Metab, doi:10.1038/s42255-021-00347-1
Passàli, Salerni, Passàli, Passàli, Bellussi, Nasal decongestants in the treatment of chronic nasal obstruction: efficacy and safety of use, Expert Opin Drug Saf, doi:10.1517/14740338.5.6.783
Pedersen, Belstrøm, The role of natural salivary defences in maintaining a healthy oral microbiota, J Dent, doi:10.1016/j.jdent.2018.08.010
Pennington, Ratcliffe, Wilson, Hardy, The influence of solution viscosity on nasal spray deposition and clearance, Int J Pharm, doi:10.1016/0378-5173(88)90277-3
Pereira, Critchley, The COVID 19 novel coronavirus pandemic 2020: seaweeds to the rescue? Why does substantial, supporting research about the antiviral properties of seaweed polysaccharides seem to go unrecognized by the pharmaceutical community in these desperate times?, J Appl Phycol, doi:10.1007/s10811-020-02143-y
Quadir, Zia, Needham, Toxicological implications of nasal formulations, Drug Deliv, doi:10.1080/107175499266823
Rheinbaben, K€ Ohnlein, Naujox, Werner, Zur Wirksamkeit der Kombination von Xylometazolinhydrochlorid und Dexpanthenol gegen Bakterien, Hefen und Viren, Krankenhaus-Hygiene þ Infekt, doi:10.1016/j.khinf.2017.03.002
Sahin-Yilmaz, Naclerio, Anatomy and physiology of the upper airway, Proc Am Thorac Soc, doi:10.1513/pats.201007-050RN
Saint, Brignole, Bringuier, Bauchet, Feldmann et al., Effects of benzalkonium chloride on growth and survival of Chang conjunctival cells, Invest Ophthalmol Vis Sci
Satpati, Algal sulfated polysaccharides: potent immunomodulators against COVID-19 in pandemic 2020, Biosci Biotechnol Res Asia, doi:10.13005/bbra/2863
Shrestha, Canosa, Nowacki, Procop, Vogel et al., Distribution of transmission potential during nonsevere COVID-19 illness, Clin Infect Dis, doi:10.1093/cid/ciaa886
Song, Peng, Wang, Liu, Dong et al., Inhibitory activities of marine sulfated polysaccharides against SARS-CoV-2, Food Funct, doi:10.1039/D0FO02017F
Sungnak, Huang, B Ecavin, Berg, Queen et al., SARS-CoV-2 entry factors are highly expressed in nasal epithelial cells together with innate immune genes, Nat Med, doi:10.1038/s41591-020-0868-6
Valerieva, Popov, Staevska, Kralimarkova, Petkova et al., Effect of micronized cellulose powder on the efficacy of topical oxymetazoline in allergic rhinitis, Allergy Asthma Proc
Weil, Groß, R€ Ocker, Bravo-Rodriguez, Heid et al., Supramolecular mechanism of viral envelope disruption by molecular tweezers, J Am Chem Soc, doi:10.1021/jacs.0c06400
Winkelmann, Thompson, Neuland, Jaramillo, Fois et al., Inflammation-induced upregulation of P2X 4 expression augments mucin secretion in airway epithelia, Am J Physiol Lung Cell Mol Physiol, doi:10.1152/ajplung.00157.2018
W€ Olfel, Corman, Guggemos, Seilmaier, Zange et al., Virological assessment of hospitalized patients with COVID-2019, Nature, doi:10.1038/s41586-020-2196-x
Xia, Liu, Wang, Xu, Lan et al., Inhibition of SARS-CoV-2 (previously 2019-nCoV) infection by a highly potent pan-coronavirus fusion inhibitor targeting its spike protein that harbors a high capacity to mediate membrane fusion, Cell Res, doi:10.1038/s41422-020-0305-x
Xia, Yan, Xu, Agrawal, Algaissi et al., A pan-coronavirus fusion inhibitor targeting the HR1 domain of human coronavirus spike, Sci Adv, doi:10.1126/sciadv.aav4580
Zhu, Zhang, Li, Yang, Song et al., A novel coronavirus from patients with pneumonia in China, 2019, N Engl J Med, doi:10.1056/NEJMoa2001017
DOI record:
{
"DOI": "10.1152/ajplung.00552.2020",
"ISSN": [
"1040-0605",
"1522-1504"
],
"URL": "http://dx.doi.org/10.1152/ajplung.00552.2020",
"abstract": "<jats:p> Pharmaceutical interventions are urgently needed to prevent severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection and transmission. As SARS-CoV-2 infects and spreads via the nasopharyngeal airways, we analyzed the antiviral effect of selected nasal and oral sprays on virus infection in vitro. Two nose sprays showed virucidal activity but were cytotoxic precluding further analysis in cell culture. One nasal and one mouth spray suppressed SARS-CoV-2 infection of TMPRSS2-expressing Vero E6 cells and primary differentiated human airway epithelial cultures. The antiviral activity in both sprays could be attributed to polyanionic ι- and κ-carrageenans. Thus, application of carrageenan-containing nasal and mouth sprays may reduce the risk of acquiring SARS-CoV-2 infection and may limit viral spread, warranting further clinical evaluation. </jats:p>",
"alternative-id": [
"10.1152/ajplung.00552.2020"
],
"assertion": [
{
"group": {
"label": "Publication History",
"name": "publication_history"
},
"label": "Received",
"name": "received",
"order": 0,
"value": "2020-11-09"
},
{
"group": {
"label": "Publication History",
"name": "publication_history"
},
"label": "Accepted",
"name": "accepted",
"order": 2,
"value": "2021-02-04"
},
{
"group": {
"label": "Publication History",
"name": "publication_history"
},
"label": "Published",
"name": "published",
"order": 3,
"value": "2021-04-28"
}
],
"author": [
{
"ORCID": "http://orcid.org/0000-0003-3782-0240",
"affiliation": [
{
"name": "Institute of Molecular Virology, Ulm University Medical Center, Ulm, Germany"
}
],
"authenticated-orcid": false,
"family": "Schütz",
"given": "Desiree",
"sequence": "first"
},
{
"affiliation": [
{
"name": "Institute of Molecular Virology, Ulm University Medical Center, Ulm, Germany"
}
],
"family": "Conzelmann",
"given": "Carina",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Institute of General Physiology, Ulm University, Ulm, Germany"
}
],
"family": "Fois",
"given": "Giorgio",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Institute of Molecular Virology, Ulm University Medical Center, Ulm, Germany"
}
],
"family": "Groß",
"given": "Rüdiger",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0003-0925-2426",
"affiliation": [
{
"name": "Institute of Molecular Virology, Ulm University Medical Center, Ulm, Germany"
}
],
"authenticated-orcid": false,
"family": "Weil",
"given": "Tatjana",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-8182-9309",
"affiliation": [
{
"name": "Institute of Molecular Virology, Ulm University Medical Center, Ulm, Germany"
}
],
"authenticated-orcid": false,
"family": "Wettstein",
"given": "Lukas",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Institute for Microbiology and Hygiene, Ulm University Medical Center, Ulm, Germany"
}
],
"family": "Stenger",
"given": "Steffen",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Chemistry and iNano Interdisciplinary Nanoscience Centre, Aarhus University, Aarhus, Denmark"
}
],
"family": "Zelikin",
"given": "Alexander",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Otorhinolaryngology, Head and Neck Surgery, Ulm University, Ulm, Germany"
}
],
"family": "Hoffmann",
"given": "Thomas K.",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-4763-1104",
"affiliation": [
{
"name": "Institute of General Physiology, Ulm University, Ulm, Germany"
}
],
"authenticated-orcid": false,
"family": "Frick",
"given": "Manfred",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Institute of Molecular Virology, Ulm University Medical Center, Ulm, Germany"
}
],
"family": "Müller",
"given": "Janis A.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Institute of Molecular Virology, Ulm University Medical Center, Ulm, Germany"
}
],
"family": "Münch",
"given": "Jan",
"sequence": "additional"
}
],
"container-title": "American Journal of Physiology-Lung Cellular and Molecular Physiology",
"container-title-short": "American Journal of Physiology-Lung Cellular and Molecular Physiology",
"content-domain": {
"crossmark-restriction": true,
"domain": [
"journals.physiology.org"
]
},
"created": {
"date-parts": [
[
2021,
2,
9
]
],
"date-time": "2021-02-09T22:23:50Z",
"timestamp": 1612909430000
},
"deposited": {
"date-parts": [
[
2021,
6,
23
]
],
"date-time": "2021-06-23T09:20:45Z",
"timestamp": 1624440045000
},
"funder": [
{
"DOI": "10.13039/100008316",
"award": [
"316249678"
],
"doi-asserted-by": "publisher",
"name": "Baden-Württemberg Stiftung"
},
{
"DOI": "10.13039/501100001659",
"doi-asserted-by": "publisher",
"name": "Deutsche Forschungsgemeinschaft"
},
{
"DOI": "10.13039/501100001659",
"award": [
"458685747"
],
"doi-asserted-by": "publisher",
"name": "Deutsche Forschungsgemeinschaft"
},
{
"DOI": "10.13039/100010676",
"award": [
"101003555"
],
"doi-asserted-by": "publisher",
"name": "H2020 Societal Challenges"
},
{
"DOI": "10.13039/501100003542",
"doi-asserted-by": "publisher",
"name": "Ministerium für Wissenschaft, Forschung und Kunst Baden-Württemberg"
}
],
"indexed": {
"date-parts": [
[
2024,
4,
15
]
],
"date-time": "2024-04-15T23:36:26Z",
"timestamp": 1713224186714
},
"is-referenced-by-count": 42,
"issue": "5",
"issued": {
"date-parts": [
[
2021,
5,
1
]
]
},
"journal-issue": {
"issue": "5",
"published-print": {
"date-parts": [
[
2021,
5,
1
]
]
}
},
"language": "en",
"link": [
{
"URL": "https://journals.physiology.org/doi/pdf/10.1152/ajplung.00552.2020",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "24",
"original-title": [],
"page": "L750-L756",
"prefix": "10.1152",
"published": {
"date-parts": [
[
2021,
5,
1
]
]
},
"published-print": {
"date-parts": [
[
2021,
5,
1
]
]
},
"publisher": "American Physiological Society",
"reference": [
{
"DOI": "10.1056/NEJMoa2001017",
"doi-asserted-by": "publisher",
"key": "B1"
},
{
"DOI": "10.1038/s41591-020-0968-3",
"doi-asserted-by": "publisher",
"key": "B2"
},
{
"DOI": "10.1016/j.cell.2020.05.042",
"doi-asserted-by": "publisher",
"key": "B3"
},
{
"DOI": "10.1056/NEJMc2009324",
"doi-asserted-by": "publisher",
"key": "B4"
},
{
"DOI": "10.1038/s41586-020-2196-x",
"doi-asserted-by": "publisher",
"key": "B5"
},
{
"DOI": "10.1136/bmj.m1728",
"doi-asserted-by": "publisher",
"key": "B6"
},
{
"DOI": "10.1093/cid/ciaa886",
"doi-asserted-by": "publisher",
"key": "B7"
},
{
"DOI": "10.2147/IJGM.S167123",
"doi-asserted-by": "publisher",
"key": "B8"
},
{
"DOI": "10.1007/s12325-017-0581-0",
"doi-asserted-by": "publisher",
"key": "B9"
},
{
"DOI": "10.1002/14651858.CD009612.pub2",
"doi-asserted-by": "publisher",
"key": "B10"
},
{
"DOI": "10.1016/0378-5173(88)90277-3",
"doi-asserted-by": "publisher",
"key": "B11"
},
{
"DOI": "10.2500/aap.2015.36.3879",
"doi-asserted-by": "publisher",
"key": "B12"
},
{
"DOI": "10.1186/1465-9921-11-108",
"doi-asserted-by": "publisher",
"key": "B13"
},
{
"DOI": "10.1186/s12931-015-0281-8",
"doi-asserted-by": "publisher",
"key": "B14"
},
{
"DOI": "10.1186/1472-6882-12-147",
"doi-asserted-by": "publisher",
"key": "B15"
},
{
"DOI": "10.1186/2049-6958-9-57",
"doi-asserted-by": "publisher",
"key": "B16"
},
{
"DOI": "10.1186/1465-9921-14-124",
"doi-asserted-by": "publisher",
"key": "B17"
},
{
"DOI": "10.1152/ajplung.00157.2018",
"doi-asserted-by": "publisher",
"key": "B18"
},
{
"DOI": "10.1016/j.antiviral.2020.104882",
"doi-asserted-by": "publisher",
"key": "B19"
},
{
"DOI": "10.1016/j.jviromet.2014.08.007",
"doi-asserted-by": "publisher",
"key": "B20"
},
{
"DOI": "10.1371/journal.ppat.0020069",
"doi-asserted-by": "publisher",
"key": "B21"
},
{
"DOI": "10.1016/0923-2516(91)90011-Q",
"doi-asserted-by": "publisher",
"key": "B22"
},
{
"DOI": "10.1186/1743-422X-5-107",
"doi-asserted-by": "publisher",
"key": "B23"
},
{
"DOI": "10.1063/1.4870772",
"doi-asserted-by": "publisher",
"key": "B24"
},
{
"DOI": "10.1038/s41598-020-80896-9",
"doi-asserted-by": "publisher",
"key": "B25"
},
{
"DOI": "10.1038/s41422-020-0305-x",
"doi-asserted-by": "publisher",
"key": "B26"
},
{
"DOI": "10.1038/s41591-020-0868-6",
"doi-asserted-by": "publisher",
"key": "B28"
},
{
"DOI": "10.1016/j.khinf.2017.03.002",
"doi-asserted-by": "publisher",
"key": "B29"
},
{
"DOI": "10.1128/AM.12.2.132-137.1964",
"doi-asserted-by": "publisher",
"key": "B30"
},
{
"DOI": "10.1007/s40121-020-00316-3",
"doi-asserted-by": "publisher",
"key": "B31"
},
{
"DOI": "10.1186/s12989-020-0339-8",
"doi-asserted-by": "publisher",
"key": "B32"
},
{
"author": "De Saint Jean M",
"first-page": "619",
"journal-title": "Invest Ophthalmol Vis Sci",
"key": "B33",
"volume": "40",
"year": "1999"
},
{
"DOI": "10.1089/jamp.2017.1390",
"doi-asserted-by": "publisher",
"key": "B34"
},
{
"DOI": "10.4168/aair.2019.11.3.306",
"doi-asserted-by": "publisher",
"key": "B35"
},
{
"DOI": "10.1517/14740338.5.6.783",
"doi-asserted-by": "publisher",
"key": "B36"
},
{
"DOI": "10.1080/107175499266823",
"doi-asserted-by": "publisher",
"key": "B37"
},
{
"DOI": "10.1371/journal.pone.0014320",
"doi-asserted-by": "publisher",
"key": "B38"
},
{
"DOI": "10.12809/hkmj208889",
"doi-asserted-by": "publisher",
"key": "B39"
},
{
"DOI": "10.1007/s10811-020-02143-y",
"doi-asserted-by": "publisher",
"key": "B40"
},
{
"DOI": "10.13005/bbra/2863",
"doi-asserted-by": "publisher",
"key": "B41"
},
{
"DOI": "10.2147/IJGM.S120665",
"doi-asserted-by": "publisher",
"key": "B45"
},
{
"DOI": "10.1039/D0FO02017F",
"doi-asserted-by": "publisher",
"key": "B46"
},
{
"DOI": "10.1093/infdis/jiaa471",
"doi-asserted-by": "publisher",
"key": "B47"
},
{
"DOI": "10.1089/jamp.2011.0924",
"doi-asserted-by": "publisher",
"key": "B48"
},
{
"DOI": "10.1016/S0168-3659(02)00363-2",
"doi-asserted-by": "publisher",
"key": "B49"
},
{
"DOI": "10.1016/S0169-409X(97)00067-7",
"doi-asserted-by": "publisher",
"key": "B50"
},
{
"DOI": "10.1513/pats.201007-050RN",
"doi-asserted-by": "publisher",
"key": "B51"
},
{
"DOI": "10.14219/jada.archive.2008.0351",
"doi-asserted-by": "publisher",
"key": "B52"
},
{
"DOI": "10.1038/sj.bdj.2013.421",
"doi-asserted-by": "publisher",
"key": "B53"
},
{
"DOI": "10.1016/j.jdent.2018.08.010",
"doi-asserted-by": "publisher",
"key": "B54"
},
{
"DOI": "10.1021/jacs.0c06400",
"doi-asserted-by": "publisher",
"key": "B55"
},
{
"DOI": "10.1126/sciadv.aav4580",
"doi-asserted-by": "publisher",
"key": "B56"
},
{
"DOI": "10.1128/AAC.01802-15",
"doi-asserted-by": "publisher",
"key": "B57"
}
],
"reference-count": 53,
"references-count": 53,
"relation": {},
"resource": {
"primary": {
"URL": "https://journals.physiology.org/doi/10.1152/ajplung.00552.2020"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "Carrageenan-containing over-the-counter nasal and oral sprays inhibit SARS-CoV-2 infection of airway epithelial cultures",
"type": "journal-article",
"update-policy": "http://dx.doi.org/10.1152/aps-crossmark-policy",
"volume": "320"
}