Comprehensive evaluation of six interventions for hospitalized patients with COVID-19: A propensity score matching study
et al., Saudi Pharmaceutical Journal, doi:10.1016/j.jsps.2023.02.004, Feb 2023
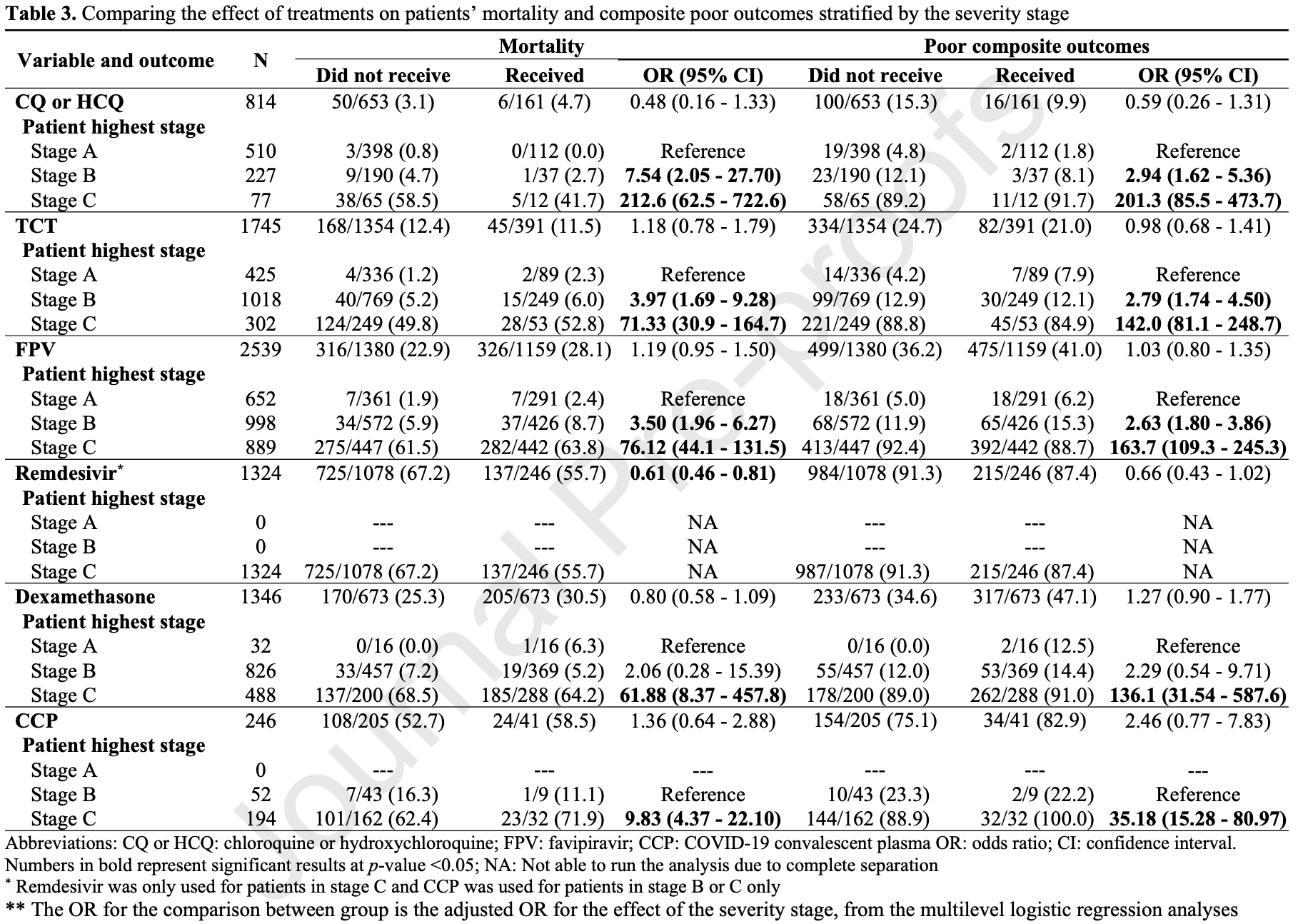
PSM retrospective 29 hospitals in Saudi Arabia, showing lower mortality with remdesivir treatment.
Gérard, Zhou, Wu, Kamo, Choi, Kim show increased risk of acute kidney injury, Leo, Briciu, Muntean, Petrov, Arch show increased risk of liver injury, Negru, Cheng, Mohammed, Kwok, Zhu show increased risk of cardiac disorders, and Kwok, Merches, Akinci, Tang, Bagheri show increased risk of mitochondrial toxicity with remdesivir.
Remdesivir efficacy disappears with longer
followup. Mixed-effects meta-regression of efficacy as a function of
followup duration across all remdesivir studies shows decreasing efficacy with
longer followup21. This may reflect
antiviral efficacy being offset by serious adverse effects of treatment.
|
risk of death, 17.3% lower, RR 0.83, p = 0.003, treatment 137 of 246 (55.7%), control 725 of 1,078 (67.3%), NNT 8.6, adjusted per study, odds ratio converted to relative risk, propensity score matching, multivariable.
|
|
risk of progression, 4.3% lower, RR 0.96, p = 0.12, treatment 215 of 246 (87.4%), control 984 of 1,078 (91.3%), NNT 26, adjusted per study, odds ratio converted to relative risk, AKI, ARDS, multi-organ failure, or mortality, propensity score matching, multivariable.
|
|
ICU time, 42.6% higher, relative time 1.43, p = 0.003, treatment 245, control 995, propensity score matching.
|
|
hospitalization time, 7.4% lower, relative time 0.93, p = 0.25, treatment 246, control 1,078, propensity score matching.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
1.
Gérard et al., Remdesivir and Acute Renal Failure: A Potential Safety Signal From Disproportionality Analysis of the WHO Safety Database, Clinical Pharmacology & Therapeutics, doi:10.1002/cpt.2145.
2.
Zhou et al., Acute Kidney Injury and Drugs Prescribed for COVID-19 in Diabetes Patients: A Real-World Disproportionality Analysis, Frontiers in Pharmacology, doi:10.3389/fphar.2022.833679.
3.
Wu et al., Acute Kidney Injury Associated With Remdesivir: A Comprehensive Pharmacovigilance Analysis of COVID-19 Reports in FAERS, Frontiers in Pharmacology, doi:10.3389/fphar.2022.692828.
4.
Kamo et al., Association of Antiviral Drugs for the Treatment of COVID-19 With Acute Renal Failure, In Vivo, doi:10.21873/invivo.13637.
5.
Choi et al., Comparative effectiveness of combination therapy with nirmatrelvir–ritonavir and remdesivir versus monotherapy with remdesivir or nirmatrelvir–ritonavir in patients hospitalised with COVID-19: a target trial emulation study, The Lancet Infectious Diseases, doi:10.1016/S1473-3099(24)00353-0.
6.
Kim et al., Investigating the Safety Profile of Fast‐Track COVID‐19 Drugs Using the FDA Adverse Event Reporting System Database: A Comparative Observational Study, Pharmacoepidemiology and Drug Safety, doi:10.1002/pds.70043.
7.
Leo et al., Hepatocellular liver injury in hospitalized patients affected by COVID-19: Presence of different risk factors at different time points, Digestive and Liver Disease, doi:10.1016/j.dld.2021.12.014.
8.
Briciu et al., Evolving Clinical Manifestations and Outcomes in COVID-19 Patients: A Comparative Analysis of SARS-CoV-2 Variant Waves in a Romanian Hospital Setting, Pathogens, doi:10.3390/pathogens12121453.
9.
Muntean et al., Effects of COVID-19 on the Liver and Mortality in Patients with SARS-CoV-2 Pneumonia Caused by Delta and Non-Delta Variants: An Analysis in a Single Centre, Pharmaceuticals, doi:10.3390/ph17010003.
10.
Petrov et al., The Effect of Potentially Hepatotoxic Medicinal Products on Alanine Transaminase Levels in COVID-19 Patients: A Case–Control Study, Safety and Risk of Pharmacotherapy, doi:10.30895/2312-7821-2025-458.
11.
Arch et al., Evaluation of the effectiveness of remdesivir in treating severe COVID-19 using data from the ISARIC WHO Clinical Characterisation Protocol UK: a prospective, national cohort study, medRxiv, doi:10.1101/2021.06.18.21259072.
12.
Negru et al., Comparative Pharmacovigilance Analysis of Approved and Repurposed Antivirals for COVID-19: Insights from EudraVigilance Data, Biomedicines, doi:10.3390/biomedicines13061387.
13.
Cheng et al., Cardiovascular Safety of COVID-19 Treatments: A Disproportionality Analysis of Adverse Event Reports from the WHO VigiBase, Infectious Diseases and Therapy, doi:10.1007/s40121-025-01225-z.
14.
Mohammed et al., Bradycardia associated with remdesivir treatment in coronavirus disease 2019 patients: A propensity score-matched analysis, Medicine, doi:10.1097/MD.0000000000044501.
15.
Kwok et al., Remdesivir induces persistent mitochondrial and structural damage in human induced pluripotent stem cell-derived cardiomyocytes, Cardiovascular Research, doi:10.1093/cvr/cvab311.
16.
Zhu et al., Cardiovascular Risks of COVID-19 Therapeutics: Integrated Analysis of FAERS, Electronic Health Records, and Transcriptomics, Pharmaceuticals, doi:10.3390/ph19040574.
17.
Merches et al., The potential of remdesivir to affect function, metabolism and proliferation of cardiac and kidney cells in vitro, Archives of Toxicology, doi:10.1007/s00204-022-03306-1.
18.
Akinci et al., Elucidation of remdesivir cytotoxicity pathways through genome-wide CRISPR-Cas9 screening and transcriptomics, bioRxiv, doi:10.1101/2020.08.27.270819.
19.
Tang et al., Modeling drug-induced mitochondrial toxicity with human primary cardiomyocytes, Science China Life Sciences, doi:10.1007/s11427-023-2369-3.
Alshamrani et al., 15 Feb 2023, retrospective, Saudi Arabia, peer-reviewed, 3 authors, study period March 2020 - January 2021.
Comprehensive evaluation of six interventions for hospitalized patients with COVID-19: A propensity score matching study
Saudi Pharmaceutical Journal, doi:10.1016/j.jsps.2023.02.004
This is a PDF file of an article that has undergone enhancements after acceptance, such as the addition of a cover page and metadata, and formatting for readability, but it is not yet the definitive version of record. This version will undergo additional copyediting, typesetting and review before it is published in its final form, but we are providing this version to give early visibility of the article. Please note that, during the production process, errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Authors' contributions: OAA designed the study, analyzed the data and revised and edited the manuscript. AAA and AMA contributed to the study design and writing the first draft of the manuscript. All authors read and approved the final manuscript.
References
Almohammed, Pharmd, None
Almutairi, Assiri, Almohammed, Predictors of Poor Outcome among Critically Ill COVID-19 Patients: A Nationally Representative Sample of the Saudi Arabian Population, Journal of Clinical Medicine, doi:10.3390/jcm11102818
Arabi, Asiri, Assiri, Balkhy, Al Bshabshe et al., Interferon Beta-1b and Lopinavir-Ritonavir for Middle East Respiratory Syndrome, N Engl J Med, doi:10.1056/NEJMoa2015294
Beigel, Tomashek, Dodd, Mehta, Zingman et al., Remdesivir for the Treatment of Covid-19 -Final Report, N Engl J Med, doi:10.1056/NEJMoa2007764
Bosaeed, Alharbi, Mahmoud, Alrehily, Bahlaq et al., Efficacy of favipiravir in adults with mild COVID-19: a randomized, double-blind, multicentre, placebo-controlled clinical trial, Clin Microbiol Infect, doi:10.1016/j.cmi.2021.12.026
Cao, Ming, Chen, Zhu, Shi, The Effectiveness of Convalescent Plasma for the Treatment of Novel Corona Virus Disease 2019: A Systematic Review and Meta-Analysis, Front Med, doi:10.3389/fmed.2021.641429
Casadevall, Pirofski, The convalescent sera option for containing COVID-19, J Clin Invest, doi:10.1172/JCI138003
Chen, Chan, Jiang, Kao, Lu et al., In vitro susceptibility of 10 clinical isolates of SARS coronavirus to selected antiviral compounds, J Clin Virol, doi:10.1016/j.jcv.2004.03.003
Consortium, Pan, Peto, Henao-Restrepo, Preziosi et al., Repurposed Antiviral Drugs for Covid-19 -Interim WHO Solidarity Trial Results, N Engl J Med, doi:10.1056/NEJMoa2023184
Di Castelnuovo, Costanzo, Cassone, Cauda, De Gaetano et al., Hydroxychloroquine and mortality in COVID-19 patients: a systematic review and a meta-analysis of observational studies and randomized controlled trials, Pathog Glob Health, doi:10.1080/20477724.2021.1936818
Doi, Hibino, Hase, Yamamoto, Kasamatsu et al., A Prospective, Randomized, Open-Label Trial of Early versus Late Favipiravir Therapy in Hospitalized Patients with COVID-19, Antimicrob Agents Chemother, doi:10.1128/AAC.01897-20
Elsawah, Elsokary, Abdallah, Elshafie, Efficacy and safety of remdesivir in hospitalized Covid-19 patients: Systematic review and meta-analysis including network meta-analysis, Rev Med Virol, doi:10.1002/rmv.2187
Fda, Coronavirus (COVID-19) Update: FDA Issues Emergency Use Authorization for Potential COVID-19 Treatment
Fda, FDA Authorizes Baricitinib Alone as Treatment for COVID-19 -Drug Information Update
Fda, FDA Authorizes Tocilizumab for Treatment of COVID-19
Gautret, Lagier, Parola, Hoang, Meddeb et al., Hydroxychloroquine and azithromycin as a treatment of COVID-19: results of an open-label non-randomized clinical trial, Int J Antimicrob Agents, doi:10.1016/j.ijantimicag.2020.105949
Geleris, Sun, Platt, Zucker, Baldwin et al., Observational Study of Hydroxychloroquine in Hospitalized Patients with Covid-19, N Engl J Med, doi:10.1056/NEJMoa2012410
Gharbharan, Jordans, Geurtsvankessel, Hollander, Karim et al., Effects of potent neutralizing antibodies from convalescent plasma in patients hospitalized for severe SARS-CoV-2 infection, Nat Commun, doi:10.1038/s41467-021-23469-2
Goldman, Lye, Hui, Marks, Bruno et al., Remdesivir for 5 or 10 Days in Patients with Severe Covid-19, N Engl J Med, doi:10.1056/NEJMoa2015301
Grein, Ohmagari, Shin, Diaz, Asperges et al., Compassionate Use of Remdesivir for Patients with Severe Covid-19, N Engl J Med, doi:10.1056/NEJMoa2007016
Group, Horby, Lim, Emberson, Mafham et al., Dexamethasone in Hospitalized Patients with Covid-19, N Engl J Med, doi:10.1056/NEJMoa2021436
Hajjar, Costa, Rizk, Biselli, Gomes et al., Intensive care management of patients with COVID-19: a practical approach, Ann Intensive Care, doi:10.1186/s13613-021-00820-w
Heimfarth, Serafini, Martins-Filho, Quintans, Quintans-Junior, Drug repurposing and cytokine management in response to COVID-19: A review, Int Immunopharmacol, doi:10.1016/j.intimp.2020.106947
Hoertel, Sanchez-Rico, Vernet, Beeker, Neuraz et al., Dexamethasone use and mortality in hospitalized patients with coronavirus disease 2019: A multicentre retrospective observational study, Br J Clin Pharmacol, doi:10.1111/bcp.14784
Hung, Lung, Tso, Liu, Chung et al., Triple combination of interferon beta-1b, lopinavir-ritonavir, and ribavirin in the treatment of patients admitted to hospital with COVID-19: an open-label, randomised, phase 2 trial, Lancet, doi:10.1016/S0140-6736(20)31042-4
Ivashchenko, Dmitriev, Vostokova, Azarova, Blinow et al., AVIFAVIR for Treatment of Patients With Moderate Coronavirus Disease 2019 (COVID-19): Interim Results of a Phase II/III Multicenter Randomized Clinical Trial, Clin Infect Dis, doi:10.1093/cid/ciaa1176
Janiaud, Axfors, Schmitt, Gloy, Ebrahimi et al., Association of Convalescent Plasma Treatment With Clinical Outcomes in Patients With COVID-19: A Systematic Review and Meta-analysis, JAMA, doi:10.1001/jama.2021.2747
Klassen, Senefeld, Johnson, Carter, Wiggins et al., The Effect of Convalescent Plasma Therapy on Mortality Among Patients With COVID-19: Systematic Review and Meta-analysis, Mayo Clin Proc, doi:10.1016/j.mayocp.2021.02.008
Kloypan, Saesong, Sangsuemoon, Chantharit, Mongkhon, CONVALESCENT plasma for COVID-19: A meta-analysis of clinical trials and real-world evidence, Eur J Clin Invest, doi:10.1111/eci.13663
Kocayigit, Ozmen, Suner, Tomak, Demir et al., Observational study of the effects of Favipiravir vs Lopinavir/Ritonavir on clinical outcomes in critically Ill patients with COVID-19, J Clin Pharm Ther, doi:10.1111/jcpt.13305
Krause, Fleming, Longini, Peto, Briand et al., SARS-CoV-2 Variants and Vaccines, N Engl J Med, doi:10.1056/NEJMsr2105280
Li, Zhang, Hu, Tong, Zheng et al., Effect of Convalescent Plasma Therapy on Time to Clinical Improvement in Patients With Severe and Life-threatening COVID-19: A Randomized Clinical Trial, JAMA, doi:10.1001/jama.2020.10044
Liu, Cao, Xu, Wang, Zhang et al., Hydroxychloroquine, a less toxic derivative of chloroquine, is effective in inhibiting SARS-CoV-2 infection in vitro, Cell Discov, doi:10.1038/s41421-020-0156-0
Liu, Chen, Liu, Nie, Lu, Comorbid Chronic Diseases are Strongly Correlated with Disease Severity among COVID-19 Patients: A Systematic Review and Meta-Analysis, Aging Dis, doi:10.14336/AD.2020.0502
Ma, Xu, Liu, Sun, Li et al., Efficacy and safety of systematic corticosteroids among severe COVID-19 patients: a systematic review and meta-analysis of randomized controlled trials, Signal Transduct Target Ther, doi:10.1038/s41392-021-00521-7
Malhani, Enani, Sharif-Askari, Alghareeb, Bin-Brikan et al., Combination of (interferon beta-1b, lopinavir/ritonavir and ribavirin) versus favipiravir in hospitalized patients with non-critical COVID-19: A cohort study, PLoS One, doi:10.1371/journal.pone.0252984
Martins-Filho, Ferreira, Heimfarth, Araujo, Quintans-Junior, Efficacy and safety of hydroxychloroquine as pre-and post-exposure prophylaxis and treatment of COVID-19: A systematic review and meta-analysis of blinded, placebo-controlled, randomized clinical trials, Lancet Reg Health Am, doi:10.1016/j.lana.2021.100062
Million, Gautret, Colson, Roussel, Dubourg et al., Clinical efficacy of chloroquine derivatives in COVID-19 infection: comparative meta-analysis between the big data and the real world, New Microbes New Infect, doi:10.1016/j.nmni.2020.100709
Moh, Saudi MoH Protocol for Patients Suspected of/Confirmed with COVID-19
Pan, Bai, Propensity score methods for causal inference: an overview, Behaviormetrika, doi:10.1007/s41237-018-0058-8
Simonovich, Burgos Pratx, Scibona, Beruto, Vallone et al., A Randomized Trial of Convalescent Plasma in Covid-19 Severe Pneumonia, N Engl J Med, doi:10.1056/NEJMoa2031304
Singh, Parida, Lingaraju, Kesavan, Kumar et al., Drug repurposing approach to fight COVID-19, Pharmacol Rep, doi:10.1007/s43440-020-00155-6
Snow, Saleem, Ambler, Nastouli, Mccoy et al., Convalescent plasma for COVID-19: a meta-analysis, trial sequential analysis, and metaregression, Br J Anaesth, doi:10.1016/j.bja.2021.07.033
Spinner, Gottlieb, Criner, Lopez, Cattelan et al., Effect of Remdesivir vs Standard Care on Clinical Status at 11 Days in Patients With Moderate COVID-19: A Randomized Clinical Trial, JAMA, doi:10.1001/jama.2020.16349
Tay, Poh, Renia, Macary, Ng, The trinity of COVID-19: immunity, inflammation and intervention, Nat Rev Immunol, doi:10.1038/s41577-020-0311-8
Tomazini, Maia, Cavalcanti, Berwanger, Rosa et al., Effect of Dexamethasone on Days Alive and Ventilator-Free in Patients With Moderate or Severe Acute Respiratory Distress Syndrome and COVID-19: The CoDEX Randomized Clinical Trial, JAMA, doi:10.1001/jama.2020.17021
Tortajada, Colomer, Andreu-Ballester, Esparcia, Oltra et al., Corticosteroids for COVID-19 patients requiring oxygen support? Yes, but not for everyone: Effect of corticosteroids on mortality and intensive care unit admission in patients with COVID-19 according to patients' oxygen requirements, J Med Virol, doi:10.1002/jmv.26635
Troxel, Petkova, Goldfeld, Liu, Tarpey et al., Association of Convalescent Plasma Treatment With Clinical Status in Patients Hospitalized With COVID-19: A Meta-analysis, JAMA Netw Open, doi:10.1001/jamanetworkopen.2021.47331
Udwadia, Singh, Barkate, Patil, Rangwala et al., Efficacy and safety of favipiravir, an oral RNA-dependent RNA polymerase inhibitor, in mild-to-moderate COVID-19: A randomized, comparative, open-label, multicenter, phase 3 clinical trial, Int J Infect Dis, doi:10.1016/j.ijid.2020.11.142
Vecchie, Batticciotto, Tangianu, Bonaventura, Pennella et al., Highdose dexamethasone treatment for COVID-19 severe acute respiratory distress syndrome: a retrospective study, Intern Emerg Med, doi:10.1007/s11739-021-02800-1
Wang, Cao, Zhang, Yang, Liu et al., Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro, Cell Res, doi:10.1038/s41422-020-0282-0
Wang, Zhang, Du, Du, Zhao et al., Remdesivir in adults with severe COVID-19: a randomised, double-blind, placebo-controlled, multicentre trial, Lancet, doi:10.1016/S0140-6736(20)31022-9
DOI record:
{
"DOI": "10.1016/j.jsps.2023.02.004",
"ISSN": [
"1319-0164"
],
"URL": "http://dx.doi.org/10.1016/j.jsps.2023.02.004",
"alternative-id": [
"S1319016423000348"
],
"assertion": [
{
"label": "This article is maintained by",
"name": "publisher",
"value": "Elsevier"
},
{
"label": "Article Title",
"name": "articletitle",
"value": "Comprehensive evaluation of six interventions for hospitalized patients with COVID-19: A propensity score matching study"
},
{
"label": "Journal Title",
"name": "journaltitle",
"value": "Saudi Pharmaceutical Journal"
},
{
"label": "CrossRef DOI link to publisher maintained version",
"name": "articlelink",
"value": "https://doi.org/10.1016/j.jsps.2023.02.004"
},
{
"label": "Content Type",
"name": "content_type",
"value": "article"
},
{
"label": "Copyright",
"name": "copyright",
"value": "© 2023 The Author(s). Published by Elsevier B.V. on behalf of King Saud University."
}
],
"author": [
{
"affiliation": [],
"family": "Alshamrani",
"given": "Ali A.",
"sequence": "first"
},
{
"affiliation": [],
"family": "Assiri",
"given": "Ahmed M.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Almohammed",
"given": "Omar A.",
"sequence": "additional"
}
],
"container-title": "Saudi Pharmaceutical Journal",
"container-title-short": "Saudi Pharmaceutical Journal",
"content-domain": {
"crossmark-restriction": true,
"domain": [
"elsevier.com",
"sciencedirect.com"
]
},
"created": {
"date-parts": [
[
2023,
2,
15
]
],
"date-time": "2023-02-15T17:07:11Z",
"timestamp": 1676480831000
},
"deposited": {
"date-parts": [
[
2023,
2,
15
]
],
"date-time": "2023-02-15T17:07:34Z",
"timestamp": 1676480854000
},
"indexed": {
"date-parts": [
[
2023,
2,
16
]
],
"date-time": "2023-02-16T05:52:35Z",
"timestamp": 1676526755170
},
"is-referenced-by-count": 0,
"issued": {
"date-parts": [
[
2023,
2
]
]
},
"language": "en",
"license": [
{
"URL": "https://www.elsevier.com/tdm/userlicense/1.0/",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2023,
2,
1
]
],
"date-time": "2023-02-01T00:00:00Z",
"timestamp": 1675209600000
}
},
{
"URL": "http://creativecommons.org/licenses/by-nc-nd/4.0/",
"content-version": "vor",
"delay-in-days": 11,
"start": {
"date-parts": [
[
2023,
2,
12
]
],
"date-time": "2023-02-12T00:00:00Z",
"timestamp": 1676160000000
}
}
],
"link": [
{
"URL": "https://api.elsevier.com/content/article/PII:S1319016423000348?httpAccept=text/xml",
"content-type": "text/xml",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://api.elsevier.com/content/article/PII:S1319016423000348?httpAccept=text/plain",
"content-type": "text/plain",
"content-version": "vor",
"intended-application": "text-mining"
}
],
"member": "78",
"original-title": [],
"prefix": "10.1016",
"published": {
"date-parts": [
[
2023,
2
]
]
},
"published-print": {
"date-parts": [
[
2023,
2
]
]
},
"publisher": "Elsevier BV",
"reference": [
{
"DOI": "10.3390/jcm11102818",
"article-title": "Predictors of Poor Outcome among Critically Ill COVID-19 Patients: A Nationally Representative Sample of the Saudi Arabian Population",
"author": "Almutairi",
"doi-asserted-by": "crossref",
"first-page": "2818",
"issue": "10",
"journal-title": "Journal of Clinical Medicine",
"key": "10.1016/j.jsps.2023.02.004_b0005",
"volume": "11",
"year": "2022"
},
{
"DOI": "10.1056/NEJMoa2015294",
"article-title": "Interferon Beta-1b and Lopinavir-Ritonavir for Middle East Respiratory Syndrome",
"author": "Arabi",
"doi-asserted-by": "crossref",
"first-page": "1645",
"issue": "17",
"journal-title": "N Engl J Med",
"key": "10.1016/j.jsps.2023.02.004_b0010",
"volume": "383",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa2007764",
"article-title": "Remdesivir for the Treatment of Covid-19 - Final Report",
"author": "Beigel",
"doi-asserted-by": "crossref",
"first-page": "1813",
"issue": "19",
"journal-title": "N Engl J Med",
"key": "10.1016/j.jsps.2023.02.004_b0015",
"volume": "383",
"year": "2020"
},
{
"DOI": "10.1016/j.cmi.2021.12.026",
"article-title": "Efficacy of favipiravir in adults with mild COVID-19: a randomized, double-blind, multicentre, placebo-controlled clinical trial",
"author": "Bosaeed",
"doi-asserted-by": "crossref",
"first-page": "602",
"issue": "4",
"journal-title": "Clin Microbiol Infect",
"key": "10.1016/j.jsps.2023.02.004_b0020",
"volume": "28",
"year": "2022"
},
{
"article-title": "The Effectiveness of Convalescent Plasma for the Treatment of Novel Corona Virus Disease 2019: A Systematic Review and Meta-Analysis",
"author": "Cao",
"journal-title": "Front Med (Lausanne)",
"key": "10.1016/j.jsps.2023.02.004_b0025",
"volume": "8",
"year": "2021"
},
{
"DOI": "10.1172/JCI138003",
"article-title": "The convalescent sera option for containing COVID-19",
"author": "Casadevall",
"doi-asserted-by": "crossref",
"first-page": "1545",
"issue": "4",
"journal-title": "J Clin Invest",
"key": "10.1016/j.jsps.2023.02.004_b0030",
"volume": "130",
"year": "2020"
},
{
"DOI": "10.1016/j.jcv.2004.03.003",
"article-title": "In vitro susceptibility of 10 clinical isolates of SARS coronavirus to selected antiviral compounds",
"author": "Chen",
"doi-asserted-by": "crossref",
"first-page": "69",
"issue": "1",
"journal-title": "J Clin Virol",
"key": "10.1016/j.jsps.2023.02.004_b0035",
"volume": "31",
"year": "2004"
},
{
"DOI": "10.1056/NEJMoa2023184",
"article-title": "Repurposed Antiviral Drugs for Covid-19 - Interim WHO Solidarity Trial Results",
"author": "Consortium",
"doi-asserted-by": "crossref",
"first-page": "497",
"issue": "6",
"journal-title": "N Engl J Med",
"key": "10.1016/j.jsps.2023.02.004_b0040",
"volume": "384",
"year": "2021"
},
{
"DOI": "10.1080/20477724.2021.1936818",
"article-title": "Hydroxychloroquine and mortality in COVID-19 patients: a systematic review and a meta-analysis of observational studies and randomized controlled trials",
"author": "Di Castelnuovo",
"doi-asserted-by": "crossref",
"first-page": "456",
"issue": "7–8",
"journal-title": "Pathog Glob Health",
"key": "10.1016/j.jsps.2023.02.004_b0045",
"volume": "115",
"year": "2021"
},
{
"DOI": "10.1128/AAC.01897-20",
"article-title": "A Prospective, Randomized, Open-Label Trial of Early versus Late Favipiravir Therapy in Hospitalized Patients with COVID-19",
"author": "Doi",
"doi-asserted-by": "crossref",
"issue": "12",
"journal-title": "Antimicrob Agents Chemother",
"key": "10.1016/j.jsps.2023.02.004_b0050",
"volume": "64",
"year": "2020"
},
{
"DOI": "10.1002/rmv.2187",
"article-title": "Efficacy and safety of remdesivir in hospitalized Covid-19 patients: Systematic review and meta-analysis including network meta-analysis",
"author": "Elsawah",
"doi-asserted-by": "crossref",
"first-page": "e2187",
"issue": "4",
"journal-title": "Rev Med Virol",
"key": "10.1016/j.jsps.2023.02.004_b0055",
"volume": "31",
"year": "2021"
},
{
"key": "10.1016/j.jsps.2023.02.004_b0060",
"unstructured": "FDA, 2020. Coronavirus (COVID-19) Update: FDA Issues Emergency Use Authorization for Potential COVID-19 Treatment. https://www.fda.gov/news-events/press-announcements/coronavirus-covid-19-update-fda-issues-emergency-use-authorization-potential-covid-19-treatment"
},
{
"key": "10.1016/j.jsps.2023.02.004_b0065",
"unstructured": "FDA, 2021a. FDA Authorizes Baricitinib Alone as Treatment for COVID-19 - Drug Information Update. https://content.govdelivery.com/accounts/USFDA/bulletins/2ea74e3"
},
{
"key": "10.1016/j.jsps.2023.02.004_b0070",
"unstructured": "FDA, 2021b. FDA Authorizes Tocilizumab for Treatment of COVID-19 https://www.fda.gov/media/150319/download."
},
{
"DOI": "10.1016/j.ijantimicag.2020.105949",
"article-title": "Hydroxychloroquine and azithromycin as a treatment of COVID-19: results of an open-label non-randomized clinical trial",
"author": "Gautret",
"doi-asserted-by": "crossref",
"issue": "1",
"journal-title": "Int J Antimicrob Agents",
"key": "10.1016/j.jsps.2023.02.004_b0075",
"volume": "56",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa2012410",
"article-title": "Observational Study of Hydroxychloroquine in Hospitalized Patients with Covid-19",
"author": "Geleris",
"doi-asserted-by": "crossref",
"first-page": "2411",
"issue": "25",
"journal-title": "N Engl J Med",
"key": "10.1016/j.jsps.2023.02.004_b0080",
"volume": "382",
"year": "2020"
},
{
"DOI": "10.1038/s41467-021-23469-2",
"article-title": "Effects of potent neutralizing antibodies from convalescent plasma in patients hospitalized for severe SARS-CoV-2 infection",
"author": "Gharbharan",
"doi-asserted-by": "crossref",
"first-page": "3189",
"issue": "1",
"journal-title": "Nat Commun",
"key": "10.1016/j.jsps.2023.02.004_b0085",
"volume": "12",
"year": "2021"
},
{
"DOI": "10.1056/NEJMoa2015301",
"article-title": "Remdesivir for 5 or 10 Days in Patients with Severe Covid-19",
"author": "Goldman",
"doi-asserted-by": "crossref",
"first-page": "1827",
"issue": "19",
"journal-title": "N Engl J Med",
"key": "10.1016/j.jsps.2023.02.004_b0090",
"volume": "383",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa2007016",
"article-title": "Compassionate Use of Remdesivir for Patients with Severe Covid-19",
"author": "Grein",
"doi-asserted-by": "crossref",
"first-page": "2327",
"issue": "24",
"journal-title": "N Engl J Med",
"key": "10.1016/j.jsps.2023.02.004_b0095",
"volume": "382",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa2021436",
"article-title": "Dexamethasone in Hospitalized Patients with Covid-19",
"author": "Group",
"doi-asserted-by": "crossref",
"first-page": "693",
"issue": "8",
"journal-title": "N Engl J Med",
"key": "10.1016/j.jsps.2023.02.004_b0100",
"volume": "384",
"year": "2021"
},
{
"DOI": "10.1186/s13613-021-00820-w",
"article-title": "Intensive care management of patients with COVID-19: a practical approach",
"author": "Hajjar",
"doi-asserted-by": "crossref",
"first-page": "36",
"issue": "1",
"journal-title": "Ann Intensive Care",
"key": "10.1016/j.jsps.2023.02.004_b0105",
"volume": "11",
"year": "2021"
},
{
"DOI": "10.1016/j.intimp.2020.106947",
"article-title": "Drug repurposing and cytokine management in response to COVID-19: A review",
"author": "Heimfarth",
"doi-asserted-by": "crossref",
"journal-title": "Int Immunopharmacol",
"key": "10.1016/j.jsps.2023.02.004_b0110",
"volume": "88",
"year": "2020"
},
{
"DOI": "10.1111/bcp.14784",
"article-title": "Dexamethasone use and mortality in hospitalized patients with coronavirus disease 2019: A multicentre retrospective observational study",
"author": "Hoertel",
"doi-asserted-by": "crossref",
"first-page": "3766",
"issue": "10",
"journal-title": "Br J Clin Pharmacol",
"key": "10.1016/j.jsps.2023.02.004_b0115",
"volume": "87",
"year": "2021"
},
{
"DOI": "10.1016/S0140-6736(20)31042-4",
"article-title": "Triple combination of interferon beta-1b, lopinavir-ritonavir, and ribavirin in the treatment of patients admitted to hospital with COVID-19: an open-label, randomised, phase 2 trial",
"author": "Hung",
"doi-asserted-by": "crossref",
"first-page": "1695",
"issue": "10238",
"journal-title": "Lancet",
"key": "10.1016/j.jsps.2023.02.004_b0120",
"volume": "395",
"year": "2020"
},
{
"key": "10.1016/j.jsps.2023.02.004_b0125",
"unstructured": "SAS Institute Inc. (2016). The PSMATCH procedure. In SAS/STAT® 14.2 User’s Guide. SAS Institute Inc., Cary, NC."
},
{
"DOI": "10.1093/cid/ciaa1176",
"article-title": "AVIFAVIR for Treatment of Patients With Moderate Coronavirus Disease 2019 (COVID-19): Interim Results of a Phase II/III Multicenter Randomized Clinical Trial",
"author": "Ivashchenko",
"doi-asserted-by": "crossref",
"first-page": "531",
"issue": "3",
"journal-title": "Clin Infect Dis",
"key": "10.1016/j.jsps.2023.02.004_b0130",
"volume": "73",
"year": "2021"
},
{
"DOI": "10.1001/jama.2021.2747",
"article-title": "Association of Convalescent Plasma Treatment With Clinical Outcomes in Patients With COVID-19: A Systematic Review and Meta-analysis",
"author": "Janiaud",
"doi-asserted-by": "crossref",
"first-page": "1185",
"issue": "12",
"journal-title": "JAMA",
"key": "10.1016/j.jsps.2023.02.004_b0135",
"volume": "325",
"year": "2021"
},
{
"DOI": "10.1016/j.mayocp.2021.02.008",
"article-title": "The Effect of Convalescent Plasma Therapy on Mortality Among Patients With COVID-19: Systematic Review and Meta-analysis",
"author": "Klassen",
"doi-asserted-by": "crossref",
"first-page": "1262",
"issue": "5",
"journal-title": "Mayo Clin Proc",
"key": "10.1016/j.jsps.2023.02.004_b0140",
"volume": "96",
"year": "2021"
},
{
"DOI": "10.1111/eci.13663",
"article-title": "CONVALESCENT plasma for COVID-19: A meta-analysis of clinical trials and real-world evidence",
"author": "Kloypan",
"doi-asserted-by": "crossref",
"first-page": "e13663",
"issue": "11",
"journal-title": "Eur J Clin Invest",
"key": "10.1016/j.jsps.2023.02.004_b0145",
"volume": "51",
"year": "2021"
},
{
"DOI": "10.1111/jcpt.13305",
"article-title": "Observational study of the effects of Favipiravir vs Lopinavir/Ritonavir on clinical outcomes in critically Ill patients with COVID-19",
"author": "Kocayigit",
"doi-asserted-by": "crossref",
"first-page": "454",
"issue": "2",
"journal-title": "J Clin Pharm Ther",
"key": "10.1016/j.jsps.2023.02.004_b0150",
"volume": "46",
"year": "2021"
},
{
"DOI": "10.1056/NEJMsr2105280",
"article-title": "SARS-CoV-2 Variants and Vaccines",
"author": "Krause",
"doi-asserted-by": "crossref",
"first-page": "179",
"issue": "2",
"journal-title": "N Engl J Med",
"key": "10.1016/j.jsps.2023.02.004_b0155",
"volume": "385",
"year": "2021"
},
{
"DOI": "10.1001/jama.2020.10044",
"article-title": "Effect of Convalescent Plasma Therapy on Time to Clinical Improvement in Patients With Severe and Life-threatening COVID-19: A Randomized Clinical Trial",
"author": "Li",
"doi-asserted-by": "crossref",
"first-page": "460",
"issue": "5",
"journal-title": "JAMA",
"key": "10.1016/j.jsps.2023.02.004_b0160",
"volume": "324",
"year": "2020"
},
{
"DOI": "10.1038/s41421-020-0156-0",
"article-title": "Hydroxychloroquine, a less toxic derivative of chloroquine, is effective in inhibiting SARS-CoV-2 infection in vitro",
"author": "Liu",
"doi-asserted-by": "crossref",
"first-page": "16",
"journal-title": "Cell Discov",
"key": "10.1016/j.jsps.2023.02.004_b0165",
"volume": "6",
"year": "2020"
},
{
"DOI": "10.14336/AD.2020.0502",
"article-title": "Comorbid Chronic Diseases are Strongly Correlated with Disease Severity among COVID-19 Patients: A Systematic Review and Meta-Analysis",
"author": "Liu",
"doi-asserted-by": "crossref",
"first-page": "668",
"issue": "3",
"journal-title": "Aging Dis",
"key": "10.1016/j.jsps.2023.02.004_b0170",
"volume": "11",
"year": "2020"
},
{
"DOI": "10.1038/s41392-021-00521-7",
"article-title": "Efficacy and safety of systematic corticosteroids among severe COVID-19 patients: a systematic review and meta-analysis of randomized controlled trials",
"author": "Ma",
"doi-asserted-by": "crossref",
"first-page": "83",
"issue": "1",
"journal-title": "Signal Transduct Target Ther",
"key": "10.1016/j.jsps.2023.02.004_b0175",
"volume": "6",
"year": "2021"
},
{
"DOI": "10.1371/journal.pone.0252984",
"article-title": "Combination of (interferon beta-1b, lopinavir/ritonavir and ribavirin) versus favipiravir in hospitalized patients with non-critical COVID-19: A cohort study",
"author": "Malhani",
"doi-asserted-by": "crossref",
"first-page": "e0252984",
"issue": "6",
"journal-title": "PLoS One",
"key": "10.1016/j.jsps.2023.02.004_b0180",
"volume": "16",
"year": "2021"
},
{
"article-title": "Efficacy and safety of hydroxychloroquine as pre-and post-exposure prophylaxis and treatment of COVID-19: A systematic review and meta-analysis of blinded, placebo-controlled, randomized clinical trials",
"author": "Martins-Filho",
"journal-title": "Lancet Reg Health Am",
"key": "10.1016/j.jsps.2023.02.004_b0185",
"volume": "2",
"year": "2021"
},
{
"DOI": "10.1016/j.nmni.2020.100709",
"article-title": "Clinical efficacy of chloroquine derivatives in COVID-19 infection: comparative meta-analysis between the big data and the real world",
"author": "Million",
"doi-asserted-by": "crossref",
"journal-title": "New Microbes New Infect",
"key": "10.1016/j.jsps.2023.02.004_b0190",
"volume": "38",
"year": "2020"
},
{
"key": "10.1016/j.jsps.2023.02.004_b0195",
"unstructured": "Saudi MoH, 2020. Saudi MoH Protocol for Patients Suspected of/Confirmed with COVID-19 (Version 2.0)."
},
{
"DOI": "10.1007/s41237-018-0058-8",
"article-title": "Propensity score methods for causal inference: an overview",
"author": "Pan",
"doi-asserted-by": "crossref",
"first-page": "317",
"issue": "2",
"journal-title": "Behaviormetrika",
"key": "10.1016/j.jsps.2023.02.004_b0200",
"volume": "45",
"year": "2018"
},
{
"DOI": "10.1056/NEJMoa2031304",
"article-title": "A Randomized Trial of Convalescent Plasma in Covid-19 Severe Pneumonia",
"author": "Simonovich, V. A., Burgos Pratx, L. D., Scibona, P., Beruto, M. V., Vallone, M. G., Vazquez, C., Savoy, N., Giunta, D. H., Perez, L. G., Sanchez, M. D. L., Gamarnik, A. V., Ojeda, D. S., Santoro, D. M., Camino, P. J., Antelo, S., Rainero, K., Vidiella, G. P., Miyazaki, E. A., Cornistein, W., Trabadelo, O. A., Ross, F. M., Spotti, M., Funtowicz, G., Scordo, W. E., Losso, M. H., Ferniot, I., Pardo, P. E., Rodriguez, E., Rucci, P., Pasquali, J., Fuentes, N. A., Esperatti, M., Speroni, G. A., Nannini, E. C., Matteaccio, A., Michelangelo, H. G., Follmann, D., Lane, H. C., Belloso, W. H., & PlasmAr Study, G",
"doi-asserted-by": "crossref",
"first-page": "619",
"issue": "7",
"journal-title": "N Engl J Med",
"key": "10.1016/j.jsps.2023.02.004_b0205",
"volume": "384",
"year": "2021"
},
{
"DOI": "10.1007/s43440-020-00155-6",
"article-title": "Drug repurposing approach to fight COVID-19",
"author": "Singh",
"doi-asserted-by": "crossref",
"first-page": "1479",
"issue": "6",
"journal-title": "Pharmacol Rep",
"key": "10.1016/j.jsps.2023.02.004_b0210",
"volume": "72",
"year": "2020"
},
{
"DOI": "10.1016/j.bja.2021.07.033",
"article-title": "Convalescent plasma for COVID-19: a meta-analysis, trial sequential analysis, and meta-regression",
"author": "Snow",
"doi-asserted-by": "crossref",
"first-page": "834",
"issue": "6",
"journal-title": "Br J Anaesth",
"key": "10.1016/j.jsps.2023.02.004_b0215",
"volume": "127",
"year": "2021"
},
{
"DOI": "10.1001/jama.2020.16349",
"article-title": "Effect of Remdesivir vs Standard Care on Clinical Status at 11 Days in Patients With Moderate COVID-19: A Randomized Clinical Trial",
"author": "Spinner",
"doi-asserted-by": "crossref",
"first-page": "1048",
"issue": "11",
"journal-title": "JAMA",
"key": "10.1016/j.jsps.2023.02.004_b0220",
"volume": "324",
"year": "2020"
},
{
"DOI": "10.1038/s41577-020-0311-8",
"article-title": "The trinity of COVID-19: immunity, inflammation and intervention",
"author": "Tay",
"doi-asserted-by": "crossref",
"first-page": "363",
"issue": "6",
"journal-title": "Nat Rev Immunol",
"key": "10.1016/j.jsps.2023.02.004_b0225",
"volume": "20",
"year": "2020"
},
{
"DOI": "10.1001/jama.2020.17021",
"article-title": "Effect of Dexamethasone on Days Alive and Ventilator-Free in Patients With Moderate or Severe Acute Respiratory Distress Syndrome and COVID-19: The CoDEX Randomized Clinical Trial",
"author": "Tomazini",
"doi-asserted-by": "crossref",
"first-page": "1307",
"issue": "13",
"journal-title": "JAMA",
"key": "10.1016/j.jsps.2023.02.004_b0230",
"volume": "324",
"year": "2020"
},
{
"DOI": "10.1002/jmv.26635",
"article-title": "Corticosteroids for COVID-19 patients requiring oxygen support? Yes, but not for everyone: Effect of corticosteroids on mortality and intensive care unit admission in patients with COVID-19 according to patients' oxygen requirements",
"author": "Tortajada",
"doi-asserted-by": "crossref",
"first-page": "1817",
"issue": "3",
"journal-title": "J Med Virol",
"key": "10.1016/j.jsps.2023.02.004_b0235",
"volume": "93",
"year": "2021"
},
{
"DOI": "10.1001/jamanetworkopen.2021.47331",
"article-title": "Association of Convalescent Plasma Treatment With Clinical Status in Patients Hospitalized With COVID-19: A Meta-analysis",
"author": "Troxel",
"doi-asserted-by": "crossref",
"first-page": "e2147331",
"issue": "1",
"journal-title": "JAMA Netw Open",
"key": "10.1016/j.jsps.2023.02.004_b0240",
"volume": "5",
"year": "2022"
},
{
"DOI": "10.1016/j.ijid.2020.11.142",
"article-title": "Efficacy and safety of favipiravir, an oral RNA-dependent RNA polymerase inhibitor, in mild-to-moderate COVID-19: A randomized, comparative, open-label, multicenter, phase 3 clinical trial",
"author": "Udwadia",
"doi-asserted-by": "crossref",
"first-page": "62",
"journal-title": "Int J Infect Dis",
"key": "10.1016/j.jsps.2023.02.004_b0245",
"volume": "103",
"year": "2021"
},
{
"DOI": "10.1007/s11739-021-02800-1",
"article-title": "High-dose dexamethasone treatment for COVID-19 severe acute respiratory distress syndrome: a retrospective study",
"author": "Vecchie",
"doi-asserted-by": "crossref",
"first-page": "1913",
"issue": "7",
"journal-title": "Intern Emerg Med",
"key": "10.1016/j.jsps.2023.02.004_b0250",
"volume": "16",
"year": "2021"
},
{
"DOI": "10.1038/s41422-020-0282-0",
"article-title": "Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro",
"author": "Wang",
"doi-asserted-by": "crossref",
"first-page": "269",
"issue": "3",
"journal-title": "Cell Res",
"key": "10.1016/j.jsps.2023.02.004_b0255",
"volume": "30",
"year": "2020"
},
{
"DOI": "10.1016/S0140-6736(20)31022-9",
"article-title": "Remdesivir in adults with severe COVID-19: a randomised, double-blind, placebo-controlled, multicentre trial",
"author": "Wang",
"doi-asserted-by": "crossref",
"first-page": "1569",
"issue": "10236",
"journal-title": "Lancet",
"key": "10.1016/j.jsps.2023.02.004_b0260",
"volume": "395",
"year": "2020"
}
],
"reference-count": 52,
"references-count": 52,
"relation": {},
"resource": {
"primary": {
"URL": "https://linkinghub.elsevier.com/retrieve/pii/S1319016423000348"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"Pharmaceutical Science",
"Pharmacology"
],
"subtitle": [],
"title": "Comprehensive evaluation of six interventions for hospitalized patients with COVID-19: A propensity score matching study",
"type": "journal-article",
"update-policy": "http://dx.doi.org/10.1016/elsevier_cm_policy"
}
alshamrani