Efficacy of favipiravir in adults with mild COVID-19: a randomized, double-blind, multicenter, placebo-controlled trial clinical trial
et al., Clinical Microbiology and Infection, doi:10.1016/j.cmi.2021.12.026, NCT04464408, Jan 2022
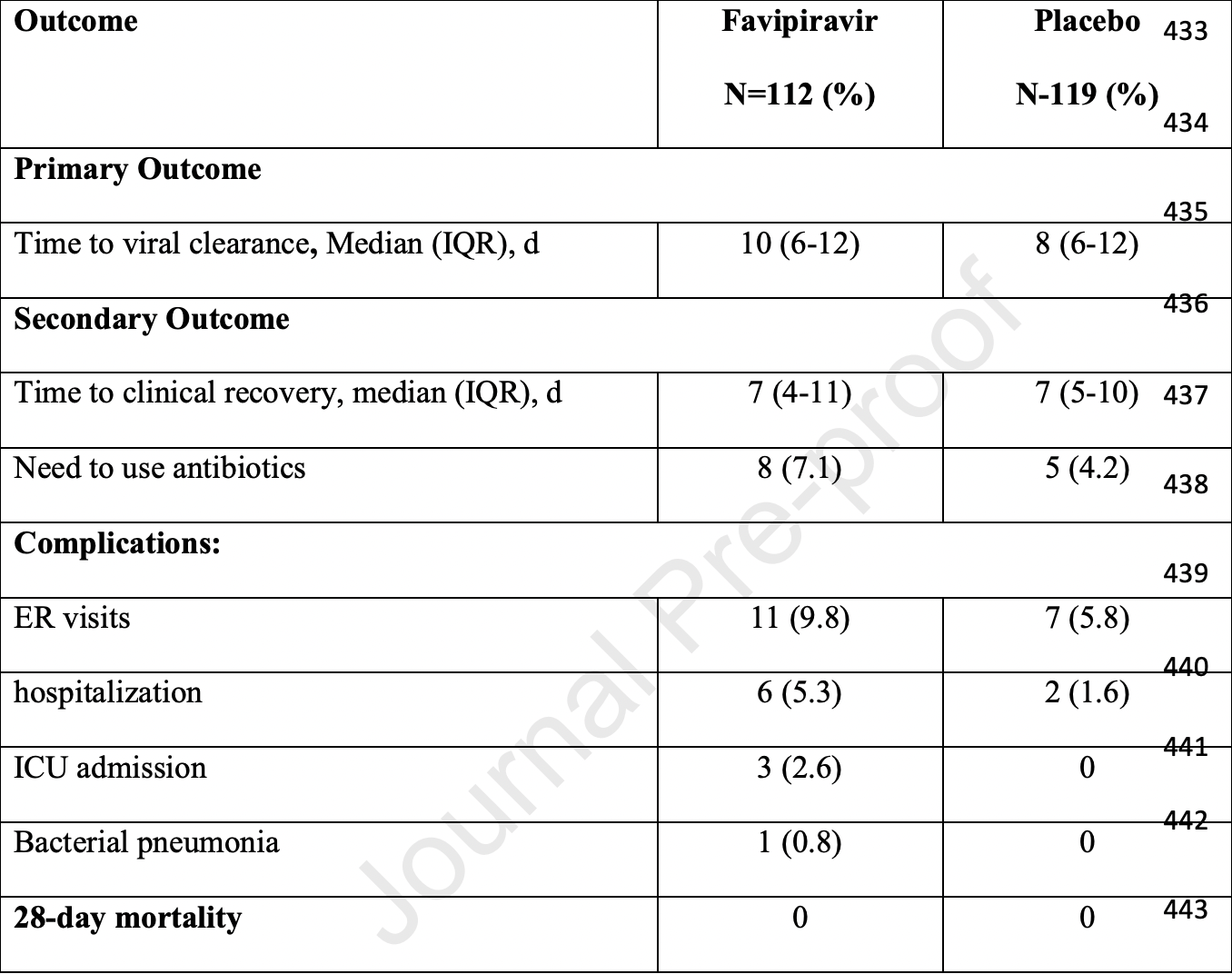
RCT with 112 favipiravir and 119 control patients showing no significant differences in outcomes. Viral clearance and clinical recovery for patients treated within 48 hours was better than those treated later. NCT04464408 (history).
Potential risks of favipiravir include kidney injury1-3, liver injury2-5, cardiovascular events5,6, pulmonary toxicity6,7, and mutagenicity, carcinogenicity, teratogenicity, embryotoxicity, and the creation of dangerous variants8-14.
|
risk of ICU admission, 618.8% higher, RR 7.19, p = 0.11, treatment 3 of 112 (2.7%), control 0 of 119 (0.0%), continuity correction due to zero event (with reciprocal of the contrasting arm).
|
|
risk of hospitalization, 218.8% higher, RR 3.19, p = 0.16, treatment 6 of 112 (5.4%), control 2 of 119 (1.7%).
|
|
time to clinical improvement, 11.9% higher, HR 1.12, p = 0.51, treatment 112, control 119, adjusted per study, inverted to make HR<1 favor treatment.
|
|
time to viral clearance, 14.9% higher, HR 1.15, p = 0.51, treatment 112, control 119, adjusted per study, inverted to make HR<1 favor treatment, primary outcome.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
1.
Abdulaziz et al., Clinical Features and Prognosis of Acute Kidney Injury in Hospital-Admitted Patients with COVID-19 in Egypt: A Single-Center Experience, Mansoura Medical Journal, doi:10.58775/2735-3990.1433.
2.
Ülger et al., Experimental evaluation of favipiravir (T-705)-induced liver and kidney toxicity in rats, Food and Chemical Toxicology, doi:10.1016/j.fct.2025.115472.
3.
El-Fetouh et al., Experimental Studies on Some Drugs Used in Covid-19 Treatment (Favipiravir and Dexamethasone) in Albino Rats, Journal of Advanced Veterinary Research, 13:10, www.advetresearch.com/index.php/AVR/article/view/1635.
4.
Almutairi et al., Liver Injury in Favipiravir-Treated COVID-19 Patients: Retrospective Single-Center Cohort Study, Tropical Medicine and Infectious Disease, doi:10.3390/tropicalmed8020129.
5.
Siby et al., Temporal Trends in Serious Adverse Events Associated with Oral Antivirals During the COVID-19 Pandemic: Insights from the FAERS Database (2020–2023), Open Forum Infectious Diseases, doi:10.1093/ofid/ofaf695.1825.
6.
Ozhan et al., Evaluation of the cardiopulmonary effects of repurposed COVID-19 therapeutics in healthy rats, Scientific Reports, doi:10.1038/s41598-025-31048-4.
7.
Ülger (B) et al., Evaluation of the effects of favipiravir (T-705) on the lung tissue of healty rats: An experimental study, Food and Chemical Toxicology, doi:10.1016/j.fct.2025.115235.
8.
Zhirnov et al., Favipiravir: the hidden threat of mutagenic action, Journal of microbiology, epidemiology and immunobiology, doi:10.36233/0372-9311-114.
9.
Waters et al., Human genetic risk of treatment with antiviral nucleoside analog drugs that induce lethal mutagenesis: the special case of molnupiravir, Environmental and Molecular Mutagenesis, doi:10.1002/em.22471.
10.
Hadj Hassine et al., Lethal Mutagenesis of RNA Viruses and Approved Drugs with Antiviral Mutagenic Activity, Viruses, doi:10.3390/v14040841.
11.
Shum, C., An investigational study into the drug-associated mutational signature in SARS-CoV-2 viruses, The University of Hong Kong, PhD Thesis, hub.hku.hk/handle/10722/344396.
12.
Shiraki et al., Convenient screening of the reproductive toxicity of favipiravir and antiviral drugs in Caenorhabditis elegans, Heliyon, doi:10.1016/j.heliyon.2024.e35331.
Bosaeed et al., 11 Jan 2022, Double Blind Randomized Controlled Trial, Saudi Arabia, peer-reviewed, 31 authors, study period 23 July, 2020 - 4 August, 2021, average treatment delay 3.0 days, trial NCT04464408 (history).
Efficacy of favipiravir in adults with mild COVID-19: a randomized, double-blind, multicentre, placebo-controlled clinical trial
Clinical Microbiology and Infection, doi:10.1016/j.cmi.2021.12.026
This is a PDF file of an article that has undergone enhancements after acceptance, such as the addition of a cover page and metadata, and formatting for readability, but it is not yet the definitive version of record. This version will undergo additional copyediting, typesetting and review before it is published in its final form, but we are providing this version to give early visibility of the article. Please note that, during the production process, errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
clinical/microbiological benefits of re-purposing antiviral therapy, particularly favipiravir, for SARS-CoV-2 infections. In conclusion, this randomized double-blinded placebo-controlled clinical trial found no clinical/virological benefit in treating mild COVID-19 patients with favipiravir. The trial result may influence decisions to remove favipiravir from national protocols for COVID-19 treatment.
Transparency declaration All authors declare no conflict of interests.
Author Contributions: Bosaeed M, Alharbi A, Mahmoud E, and Abalkhail M jointly formed the study concept and design,. All authors critically revised the report and approved the final version to be submitted for publication. The corresponding author confirms that he had full access to all the data in the study and had final responsibility for the decision to submit for publication Funding: This work was supported by King Abdullah International Medical Research Center, Saudi Arabia. Protocol No.RC20/220/R. The funder had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication. All the authors have full access to all the data in the study and had final responsibility for the decision to submit for publication. The study Statistical Analysis Plan is available with this publication as part of the supplementary material. Individual participant data are..
References
Alamer, Alrashed, Alfaifi, Alosaimi, Alhassar et al., Effectiveness and safety of favipiravir compared to supportive care in moderately to critically ill COVID-19 patients: a retrospective study with propensity score matching sensitivity analysis, Current Medical Research and Opinion
Baranovich, Wong, Armstrong, Marjuki, Webby et al., T-705 (Favipiravir) Induces Lethal Mutagenesis in Influenza A H1N1 Viruses In Vitro, Journal of Virology
Bosaeed, Hussein, Alharbi, Alsaedy, Alothman, A Trial of Favipiravir and Hydroxychloroquine combination in Adults Hospitalized with moderate and severe Covid-19: A structured summary of a study protocol for a randomised controlled trial, Trials
Buonaguro, Tagliamonte, Tornesello, Buonaguro, SARS-CoV-2 RNA polymerase as target for antiviral therapy, Journal of Translational Medicine
Cai, Yang, Liu, Chen, Shu et al., Experimental Treatment with Favipiravir for COVID-19: An Open-Label Control Study
Chen, Zhang, Huang, Yin, Cheng et al., Favipiravir Versus Arbidol for Clinical Recovery Rate in Moderate and Severe Adult COVID-19 Patients: A Prospective, Multicenter, Open-Label, Randomized Controlled Clinical Trial, Frontiers in Pharmacology
Delang, Abdelnabi, Neyts, Favipiravir as a potential countermeasure against neglected and emerging RNA viruses, Antiviral Research
Doi, Hibino, Hase, Yamamoto, Kasamatsu et al., Randomized, Open-Label Trial of Early versus Late Favipiravir Therapy in Hospitalized Patients with COVID-19, Antimicrobial Agents and Chemotherapy
Dong, Shamsuddin, Campbell, Theodoratou, Current COVID-19 treatments: Rapid review of the literature, Journal of Global Health
Forrest, Rayner, Park, Mills, Early Treatment of COVID-19 Disease: A Missed Opportunity, Infectious Diseases and Therapy
Furuta, Komeno, Nakamura T, Favipiravir (T-705), a broad spectrum inhibitor of viral RNA polymerase, Proceedings of the Japan Academy, Series B
Hassanipour, Arab-Zozani, Amani, Heidarzad, Fathalipour et al., The efficacy and safety of Favipiravir in treatment of COVID-19: a systematic review and meta-analysis of clinical trials, Scientific Reports
Hemoglobin, median (IQR)
Ivashchenko, Dmitriev, Vostokova, Azarova, Blinow et al., AVIFAVIR for Treatment of Patients With Moderate Coronavirus Disease 2019 (COVID-19): Interim Results of a Phase II/III Multicenter Randomized Clinical Trial, Clinical Infectious Diseases: the Infectious Diseases Society of America
J O U R N A L P R E, -p r o o f
Kumar, Early versus late oseltamivir treatment in severely ill patients with 2009 pandemic influenza A (H1N1): speed is life, Journal of Antimicrobial Chemotherapy
Manabe, Kambayashi, Akatsu, Kudo, Favipiravir for the treatment of patients with COVID-19: a systematic review and meta-analysis, BMC infectious diseases
Oboho, Reed, Leon, Rothrock, Aragon et al., 1341The benefit of early influenza antiviral treatment of pregnant women hospitalized with laboratory-confirmed influenza, Open Forum Infectious Diseases
Pan, Peto, Karim, Alejandria, Henao-Restrepo et al., Repurposed antiviral drugs for COVID-19 -interim WHO SOLIDARITY trial results
Reis, Silva Ea Dos, Silva, Thabane, Singh et al., Effect of Early Treatment With Hydroxychloroquine or Lopinavir and Ritonavir on Risk of Hospitalization Among Patients With COVID-19, JAMA Network Open
Shinkai, Tsushima, Tanaka, Hagiwara, Tarumoto et al., Efficacy and Safety of Favipiravir in Moderate COVID-19 Pneumonia Patients without Oxygen Therapy: A Randomized, Phase III Clinical Trial, Infectious Diseases and Therapy
Shiraki, Daikoku, Favipiravir, an anti-influenza drug against life-threatening RNA virus infections, Pharmacology & Therapeutics
Udwadia, Singh, Barkate, Patil, Rangwala et al., Efficacy and safety of favipiravir, an oral RNA-dependent RNA polymerase inhibitor, in mild-to-moderate COVID-19: A randomized, comparative, open-label, multicenter, phase 3 clinical trial, International Journal of Infectious Diseases
Who Director, General's opening remarks at the media briefing on COVID-19 -11
Who, None
Who, None
Wu, Mcgoogan, Characteristics of and Important Lessons From the Coronavirus Disease 2019 (COVID-19) Outbreak in China, JAMA
Yamakawa, Yamamoto, Ishimaru, Hashimoto, Terayama et al., Japanese rapid/living recommendations on drug management for COVID-19, Acute Medicine & Surgery
Yao, Ye, Zhang, Cui, Huang et al., In Vitro Antiviral Activity and Projection of Optimized Dosing Design of Hydroxychloroquine for the Treatment of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2), Clinical Infectious Diseases
Özlüşen, Kozan, Akcan, Kalender, Yaprak et al., Effectiveness of favipiravir in COVID-19: a live systematic review, European Journal of Clinical Microbiology & Infectious Diseases
DOI record:
{
"DOI": "10.1016/j.cmi.2021.12.026",
"ISSN": [
"1198-743X"
],
"URL": "http://dx.doi.org/10.1016/j.cmi.2021.12.026",
"alternative-id": [
"S1198743X21007345"
],
"author": [
{
"affiliation": [],
"family": "Bosaeed",
"given": "Mohammad",
"sequence": "first"
},
{
"affiliation": [],
"family": "Alharbi",
"given": "Ahmad",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Mahmoud",
"given": "Ebrahim",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Alrehily",
"given": "Sanaa",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Bahlaq",
"given": "Mohannad",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Gaifer",
"given": "Zied",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Alturkistani",
"given": "Hanan",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Alhagan",
"given": "Khaled",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Alshahrani",
"given": "Saad",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Tolbah",
"given": "Ali",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Musattat",
"given": "Abrar",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Alanazi",
"given": "Maha",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Jaha",
"given": "Raniah",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Sultana",
"given": "Khizra",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Alqahtani",
"given": "Hajar",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Al Aamer",
"given": "Kholoud",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Jaser",
"given": "Saud",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Alsaedy",
"given": "Abdulrahman",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ahmad",
"given": "Ayoub",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Abalkhail",
"given": "Mohammed",
"sequence": "additional"
},
{
"affiliation": [],
"family": "AlJohani",
"given": "Sameera",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Al Jeraisy",
"given": "Majed",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Almaziad",
"given": "Sultan",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Albaalharith",
"given": "Nahlah",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Alabdulkareem",
"given": "Khaled",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Alshowair",
"given": "Abdulmajeed",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Alharbi",
"given": "Naif Khalaf",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Alrabiah",
"given": "Fahad",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Alshamrani",
"given": "Majid",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Aldibasi",
"given": "Omar",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Alaskar",
"given": "Ahmed",
"sequence": "additional"
}
],
"container-title": [
"Clinical Microbiology and Infection"
],
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2022,
1,
11
]
],
"date-time": "2022-01-11T02:11:07Z",
"timestamp": 1641867067000
},
"deposited": {
"date-parts": [
[
2022,
1,
11
]
],
"date-time": "2022-01-11T02:11:26Z",
"timestamp": 1641867086000
},
"funder": [
{
"DOI": "10.13039/501100013302",
"doi-asserted-by": "publisher",
"name": "King Abdullah International Medical Research Center"
}
],
"indexed": {
"date-parts": [
[
2022,
1,
11
]
],
"date-time": "2022-01-11T05:43:47Z",
"timestamp": 1641879827010
},
"is-referenced-by-count": 0,
"issn-type": [
{
"type": "print",
"value": "1198-743X"
}
],
"issued": {
"date-parts": [
[
2022,
1
]
]
},
"language": "en",
"license": [
{
"URL": "https://www.elsevier.com/tdm/userlicense/1.0/",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2022,
1,
1
]
],
"date-time": "2022-01-01T00:00:00Z",
"timestamp": 1640995200000
}
},
{
"URL": "http://creativecommons.org/licenses/by-nc-nd/4.0/",
"content-version": "vor",
"delay-in-days": 6,
"start": {
"date-parts": [
[
2022,
1,
7
]
],
"date-time": "2022-01-07T00:00:00Z",
"timestamp": 1641513600000
}
}
],
"link": [
{
"URL": "https://api.elsevier.com/content/article/PII:S1198743X21007345?httpAccept=text/xml",
"content-type": "text/xml",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://api.elsevier.com/content/article/PII:S1198743X21007345?httpAccept=text/plain",
"content-type": "text/plain",
"content-version": "vor",
"intended-application": "text-mining"
}
],
"member": "78",
"original-title": [],
"prefix": "10.1016",
"published": {
"date-parts": [
[
2022,
1
]
]
},
"published-print": {
"date-parts": [
[
2022,
1
]
]
},
"publisher": "Elsevier BV",
"reference": [
{
"article-title": "COVID-19 Map - Johns Hopkins Coronavirus Resource Center [Internet]",
"journal-title": "Johns Hopkins Coronavirus Resource Center",
"key": "10.1016/j.cmi.2021.12.026_bib1",
"year": "2020"
},
{
"article-title": "WHO Director-General's opening remarks at the media briefing on COVID-19 - 11 March 2020 [Internet]",
"journal-title": "Who.int",
"key": "10.1016/j.cmi.2021.12.026_bib2",
"year": "2020"
},
{
"article-title": "WHO's COVID-19 response [Internet]",
"author": "Timeline",
"journal-title": "Who.int",
"key": "10.1016/j.cmi.2021.12.026_bib3",
"year": "2021"
},
{
"DOI": "10.1093/cid/ciaa237",
"article-title": "In Vitro Antiviral Activity and Projection of Optimized Dosing Design of Hydroxychloroquine for the Treatment of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2)",
"author": "Yao",
"doi-asserted-by": "crossref",
"first-page": "732",
"issue": "15",
"journal-title": "Clinical Infectious Diseases",
"key": "10.1016/j.cmi.2021.12.026_bib4",
"volume": "71",
"year": "2020"
},
{
"key": "10.1016/j.cmi.2021.12.026_bib5",
"unstructured": "Pan H, Peto R, Karim Q, Alejandria M, Henao-Restrepo A, García C et al. Repurposed antiviral drugs for COVID-19 –interim WHO SOLIDARITY trial results. 2020."
},
{
"DOI": "10.1056/NEJMoa2021436",
"article-title": "Dexamethasone in Hospitalized Patients with Covid-19",
"doi-asserted-by": "crossref",
"first-page": "693",
"issue": "8",
"journal-title": "New England Journal of Medicine",
"key": "10.1016/j.cmi.2021.12.026_bib6",
"volume": "384",
"year": "2021"
},
{
"DOI": "10.1016/j.antiviral.2018.03.003",
"article-title": "Favipiravir as a potential countermeasure against neglected and emerging RNA viruses",
"author": "Delang",
"doi-asserted-by": "crossref",
"first-page": "85",
"journal-title": "Antiviral Research",
"key": "10.1016/j.cmi.2021.12.026_bib7",
"volume": "153",
"year": "2018"
},
{
"DOI": "10.2183/pjab.93.027",
"article-title": "Favipiravir (T-705), a broad spectrum inhibitor of viral RNA polymerase",
"author": "FURUTA",
"doi-asserted-by": "crossref",
"first-page": "449",
"issue": "7",
"journal-title": "Proceedings of the Japan Academy, Series B.",
"key": "10.1016/j.cmi.2021.12.026_bib8",
"volume": "93",
"year": "2017"
},
{
"DOI": "10.1186/s12967-020-02355-3",
"article-title": "SARS-CoV-2 RNA polymerase as target for antiviral therapy",
"author": "Buonaguro",
"doi-asserted-by": "crossref",
"issue": "1",
"journal-title": "Journal of Translational Medicine",
"key": "10.1016/j.cmi.2021.12.026_bib9",
"volume": "18",
"year": "2020"
},
{
"DOI": "10.1080/03007995.2021.1920900",
"article-title": "Effectiveness and safety of favipiravir compared to supportive care in moderately to critically ill COVID-19 patients: a retrospective study with propensity score matching sensitivity analysis",
"author": "Alamer",
"doi-asserted-by": "crossref",
"first-page": "1085",
"issue": "7",
"journal-title": "Current Medical Research and Opinion",
"key": "10.1016/j.cmi.2021.12.026_bib10",
"volume": "37",
"year": "2021"
},
{
"DOI": "10.1016/j.ijid.2020.11.142",
"article-title": "Efficacy and safety of favipiravir, an oral RNA-dependent RNA polymerase inhibitor, in mild-to-moderate COVID-19: A randomized, comparative, open-label, multicenter, phase 3 clinical trial",
"author": "Udwadia",
"doi-asserted-by": "crossref",
"first-page": "62",
"journal-title": "International Journal of Infectious Diseases",
"key": "10.1016/j.cmi.2021.12.026_bib11",
"volume": "103",
"year": "2021"
},
{
"DOI": "10.1038/s41598-021-90551-6",
"article-title": "The efficacy and safety of Favipiravir in treatment of COVID-19: a systematic review and meta-analysis of clinical trials",
"author": "Hassanipour",
"doi-asserted-by": "crossref",
"issue": "1",
"journal-title": "Scientific Reports",
"key": "10.1016/j.cmi.2021.12.026_bib12",
"volume": "11",
"year": "2021"
},
{
"article-title": "Japanese rapid/living recommendations on drug management for COVID‐19",
"author": "Yamakawa",
"issue": "1",
"journal-title": "Acute Medicine & Surgery",
"key": "10.1016/j.cmi.2021.12.026_bib13",
"volume": "8",
"year": "2021"
},
{
"key": "10.1016/j.cmi.2021.12.026_bib14",
"unstructured": "[Internet]. Ministry Of Health Saudi Arabia. [cited 7 November 2021]. Available from: https://www.moh.gov.sa/en/Pages/Default.aspx"
},
{
"DOI": "10.1001/jama.2020.2648",
"article-title": "Characteristics of and Important Lessons From the Coronavirus Disease 2019 (COVID-19) Outbreak in China",
"author": "Wu",
"doi-asserted-by": "crossref",
"first-page": "1239",
"issue": "13",
"journal-title": "JAMA",
"key": "10.1016/j.cmi.2021.12.026_bib15",
"volume": "323",
"year": "2020"
},
{
"DOI": "10.1007/s40121-020-00349-8",
"article-title": "Early Treatment of COVID-19 Disease: A Missed Opportunity",
"author": "Forrest",
"doi-asserted-by": "crossref",
"first-page": "715",
"issue": "4",
"journal-title": "Infectious Diseases and Therapy",
"key": "10.1016/j.cmi.2021.12.026_bib16",
"volume": "9",
"year": "2020"
},
{
"DOI": "10.1001/jamanetworkopen.2021.6468",
"article-title": "Effect of Early Treatment With Hydroxychloroquine or Lopinavir and Ritonavir on Risk of Hospitalization Among Patients With COVID-19",
"author": "Reis",
"doi-asserted-by": "crossref",
"issue": "4",
"journal-title": "JAMA Network Open",
"key": "10.1016/j.cmi.2021.12.026_bib17",
"volume": "4",
"year": "2021"
},
{
"article-title": "Current COVID-19 treatments: Rapid review of the literature",
"author": "Dong",
"first-page": "11",
"journal-title": "Journal of Global Health",
"key": "10.1016/j.cmi.2021.12.026_bib18",
"year": "2021"
},
{
"DOI": "10.1016/j.pharmthera.2020.107512",
"article-title": "Favipiravir, an anti-influenza drug against life-threatening RNA virus infections",
"author": "Shiraki",
"doi-asserted-by": "crossref",
"first-page": "107512",
"journal-title": "Pharmacology & Therapeutics [Internet]",
"key": "10.1016/j.cmi.2021.12.026_bib19",
"volume": "209",
"year": "2020"
},
{
"DOI": "10.1128/JVI.02346-12",
"article-title": "T-705 (Favipiravir) Induces Lethal Mutagenesis in Influenza A H1N1 Viruses In Vitro",
"author": "Baranovich",
"doi-asserted-by": "crossref",
"first-page": "3741",
"issue": "7",
"journal-title": "Journal of Virology",
"key": "10.1016/j.cmi.2021.12.026_bib20",
"volume": "87",
"year": "2013"
},
{
"DOI": "10.1007/s40121-021-00517-4",
"article-title": "Efficacy and Safety of Favipiravir in Moderate COVID-19 Pneumonia Patients without Oxygen Therapy: A Randomized, Phase III Clinical Trial",
"author": "Shinkai",
"doi-asserted-by": "crossref",
"journal-title": "Infectious Diseases and Therapy",
"key": "10.1016/j.cmi.2021.12.026_bib21",
"year": "2021"
},
{
"DOI": "10.1186/s13063-020-04825-x",
"article-title": "A Trial of Favipiravir and Hydroxychloroquine combination in Adults Hospitalized with moderate and severe Covid-19: A structured summary of a study protocol for a randomised controlled trial",
"author": "Bosaeed",
"doi-asserted-by": "crossref",
"issue": "1",
"journal-title": "Trials",
"key": "10.1016/j.cmi.2021.12.026_bib22",
"volume": "21",
"year": "2020"
},
{
"DOI": "10.1007/s10096-021-04307-1",
"article-title": "Effectiveness of favipiravir in COVID-19: a live systematic review",
"author": "Özlüşen",
"doi-asserted-by": "crossref",
"journal-title": "European Journal of Clinical Microbiology & Infectious Diseases",
"key": "10.1016/j.cmi.2021.12.026_bib23",
"year": "2021"
},
{
"DOI": "10.1186/s12879-021-06164-x",
"article-title": "Favipiravir for the treatment of patients with COVID-19: a systematic review and meta-analysis",
"author": "Manabe",
"doi-asserted-by": "crossref",
"first-page": "489",
"issue": "1",
"journal-title": "BMC Infectious Diseases [Internet]",
"key": "10.1016/j.cmi.2021.12.026_bib24",
"volume": "21",
"year": "2021"
},
{
"DOI": "10.1093/ofid/ofu051.158",
"article-title": "1341The benefit of early influenza antiviral treatment of pregnant women hospitalized with laboratory-confirmed influenza",
"author": "Oboho",
"doi-asserted-by": "crossref",
"first-page": "S58",
"issue": "suppl_1",
"journal-title": "Open Forum Infectious Diseases",
"key": "10.1016/j.cmi.2021.12.026_bib25",
"volume": "1",
"year": "2014"
},
{
"DOI": "10.1093/jac/dkr090",
"article-title": "Early versus late oseltamivir treatment in severely ill patients with 2009 pandemic influenza A (H1N1): speed is life",
"author": "Kumar",
"doi-asserted-by": "crossref",
"first-page": "959",
"issue": "5",
"journal-title": "Journal of Antimicrobial Chemotherapy",
"key": "10.1016/j.cmi.2021.12.026_bib26",
"volume": "66",
"year": "2011"
},
{
"DOI": "10.1093/cid/ciaa1176",
"article-title": "AVIFAVIR for Treatment of Patients With Moderate Coronavirus Disease 2019 (COVID-19): Interim Results of a Phase II/III Multicenter Randomized Clinical Trial",
"author": "Ivashchenko",
"doi-asserted-by": "crossref",
"first-page": "531",
"issue": "3",
"journal-title": "Clinical Infectious Diseases: The Infectious Diseases Society of America [Internet]",
"key": "10.1016/j.cmi.2021.12.026_bib27",
"volume": "73",
"year": "2020"
},
{
"DOI": "10.1016/j.eng.2020.03.007",
"article-title": "Experimental Treatment with Favipiravir for COVID-19: An Open-Label Control Study",
"author": "Cai",
"doi-asserted-by": "crossref",
"journal-title": "Engineering [Internet]",
"key": "10.1016/j.cmi.2021.12.026_bib28",
"year": "2020"
},
{
"article-title": "Favipiravir Versus Arbidol for Clinical Recovery Rate in Moderate and Severe Adult COVID-19 Patients: A Prospective, Multicenter, Open-Label, Randomized Controlled Clinical Trial",
"author": "Chen",
"first-page": "12",
"journal-title": "Frontiers in Pharmacology",
"key": "10.1016/j.cmi.2021.12.026_bib29",
"year": "2021"
},
{
"article-title": "A Prospective, Randomized, Open-Label Trial of Early versus Late Favipiravir Therapy in Hospitalized Patients with COVID-19",
"author": "Doi",
"first-page": "64",
"issue": "12",
"journal-title": "Antimicrobial Agents and Chemotherapy",
"key": "10.1016/j.cmi.2021.12.026_bib30",
"year": "2020"
}
],
"reference-count": 30,
"references-count": 30,
"relation": {},
"score": 1,
"short-container-title": [
"Clinical Microbiology and Infection"
],
"short-title": [],
"source": "Crossref",
"subject": [
"Infectious Diseases",
"Microbiology (medical)",
"General Medicine"
],
"subtitle": [],
"title": [
"Efficacy of favipiravir in adults with mild COVID-19: a randomized, double-blind, multicenter, placebo-controlled trial clinical trial"
],
"type": "journal-article"
}