Efficacy and Safety of Favipiravir, an Oral RNA-Dependent RNA Polymerase Inhibitor, in Mild-to-Moderate COVID-19: A Randomized, Comparative, Open-Label, Multicenter, Phase 3 Clinical Trial
et al., International Journal of Infectious Diseases, doi:10.1016/j.ijid.2020.11.142, CTRI/2020/05/025114, Nov 2020
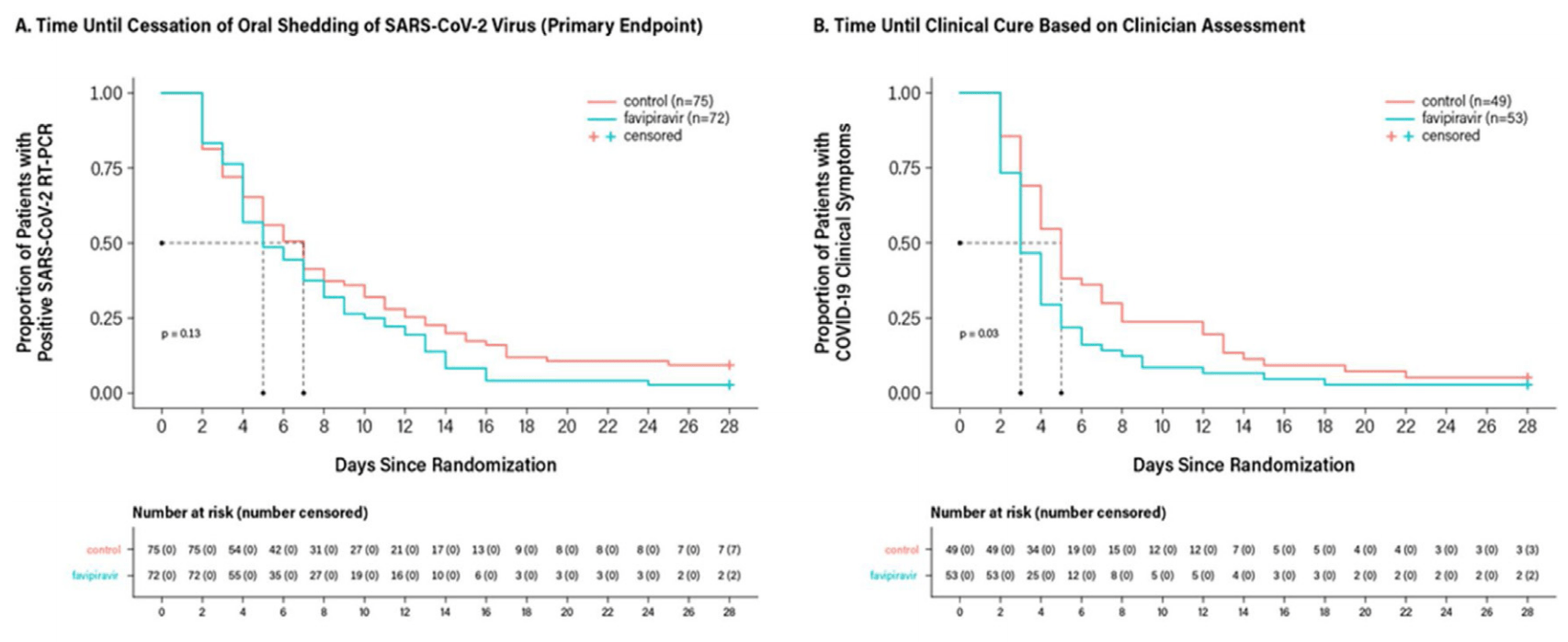
RCT with 75 favipiravir patients and 75 control patients showing improved recovery with treatment.
Potential risks of favipiravir include kidney injury1-3, liver injury2-5, cardiovascular events5,6, pulmonary toxicity6,7, and mutagenicity, carcinogenicity, teratogenicity, embryotoxicity, and the creation of dangerous variants8-14.
|
risk of death, 66.4% lower, RR 0.34, p = 1.00, treatment 0 of 73 (0.0%), control 1 of 75 (1.3%), NNT 75, relative risk is not 0 because of continuity correction due to zero events (with reciprocal of the contrasting arm).
|
|
time to discharge, 28.9% lower, HR 0.71, p = 0.07, treatment 75, control 72, inverted to make HR<1 favor treatment.
|
|
time to clinical cure, 42.8% lower, HR 0.57, p = 0.02, treatment 75, control 72, inverted to make HR<1 favor treatment.
|
|
time to viral clearance, 26.8% lower, HR 0.73, p = 0.10, treatment 75, control 72, inverted to make HR<1 favor treatment.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
1.
Abdulaziz et al., Clinical Features and Prognosis of Acute Kidney Injury in Hospital-Admitted Patients with COVID-19 in Egypt: A Single-Center Experience, Mansoura Medical Journal, doi:10.58775/2735-3990.1433.
2.
Ülger et al., Experimental evaluation of favipiravir (T-705)-induced liver and kidney toxicity in rats, Food and Chemical Toxicology, doi:10.1016/j.fct.2025.115472.
3.
El-Fetouh et al., Experimental Studies on Some Drugs Used in Covid-19 Treatment (Favipiravir and Dexamethasone) in Albino Rats, Journal of Advanced Veterinary Research, 13:10, www.advetresearch.com/index.php/AVR/article/view/1635.
4.
Almutairi et al., Liver Injury in Favipiravir-Treated COVID-19 Patients: Retrospective Single-Center Cohort Study, Tropical Medicine and Infectious Disease, doi:10.3390/tropicalmed8020129.
5.
Siby et al., Temporal Trends in Serious Adverse Events Associated with Oral Antivirals During the COVID-19 Pandemic: Insights from the FAERS Database (2020–2023), Open Forum Infectious Diseases, doi:10.1093/ofid/ofaf695.1825.
6.
Ozhan et al., Evaluation of the cardiopulmonary effects of repurposed COVID-19 therapeutics in healthy rats, Scientific Reports, doi:10.1038/s41598-025-31048-4.
7.
Ülger (B) et al., Evaluation of the effects of favipiravir (T-705) on the lung tissue of healty rats: An experimental study, Food and Chemical Toxicology, doi:10.1016/j.fct.2025.115235.
8.
Zhirnov et al., Favipiravir: the hidden threat of mutagenic action, Journal of microbiology, epidemiology and immunobiology, doi:10.36233/0372-9311-114.
9.
Waters et al., Human genetic risk of treatment with antiviral nucleoside analog drugs that induce lethal mutagenesis: the special case of molnupiravir, Environmental and Molecular Mutagenesis, doi:10.1002/em.22471.
10.
Hadj Hassine et al., Lethal Mutagenesis of RNA Viruses and Approved Drugs with Antiviral Mutagenic Activity, Viruses, doi:10.3390/v14040841.
11.
Shum, C., An investigational study into the drug-associated mutational signature in SARS-CoV-2 viruses, The University of Hong Kong, PhD Thesis, hub.hku.hk/handle/10722/344396.
12.
Shiraki et al., Convenient screening of the reproductive toxicity of favipiravir and antiviral drugs in Caenorhabditis elegans, Heliyon, doi:10.1016/j.heliyon.2024.e35331.
Udwadia et al., 16 Nov 2020, Randomized Controlled Trial, India, peer-reviewed, 11 authors, study period 14 May, 2020 - 3 July, 2020, trial CTRI/2020/05/025114.
Efficacy and safety of favipiravir, an oral RNA-dependent RNA polymerase inhibitor, in mild-to-moderate COVID-19: A randomized, comparative, open-label, multicenter, phase 3 clinical trial
International Journal of Infectious Diseases, doi:10.1016/j.ijid.2020.11.142
To assess the efficacy and safety of favipiravir in adults with mild-to-moderate coronavirus disease 2019 . Methods: In this randomized, open-label, parallel-arm, multicenter, phase 3 trial, adults (18-75 years) with RT-PCR confirmed COVID-19 and mild-to-moderate symptoms (including asymptomatic) were randomized 1:1 to oral favipiravir (day 1: 1800 mg BID and days 2À14: 800 mg BID) plus standard supportive care versus supportive care alone. The primary endpoint was time to the cessation of viral shedding; time to clinical cure was also measured. Results: From May 14 to July 3, 2020, 150 patients were randomized to favipiravir (n = 75) or control (n = 75). Median time to the cessation of viral shedding was 5 days (95% CI: 4 days, 7 days) versus 7 days (95% CI: 5 days, 8 days), P = 0.129, and median time to clinical cure was 3 days (95% CI: 3 days, 4 days) versus 5 days (95% CI: 4 days, 6 days), P = 0.030, for favipiravir and control, respectively. Adverse events were observed in 36% of favipiravir and 8% of control patients. One control patient died due to worsening disease.
Conclusion: The lack of statistical significance on the primary endpoint was confounded by limitations of the RT-PCR assay. Significant improvement in time to clinical cure suggests favipiravir may be beneficial in mild-to-moderate COVID-19.
Conflict of interest Monika Tandon, Pawan Singh, Hanmant Barkate, Saiprasad Patil, Shabbir Rangwala, Amol Pendse, and Jatin Kadam are fulltime employees of Glenmark Pharmaceuticals Limited, India. Wen Wu is a full-time employee of Glenmark Pharmaceuticals Ltd, United Kingdom. Cynthia Caracta is a full-time employee of Glenmark Pharmaceuticals Inc., USA. Zarir F Udwadia received an investigator grant from Glenmark Pharmaceuticals Limited, India, as a site principal investigator for this study.
Ethical approval The protocol and informed consent form were approved by institutional/independent ethics committees and the Drugs Controller General of India (April 26, 2020) . The trial was undertaken in accordance with current guidelines of the Central Drugs Standard Control Organization, which is the National Regulatory Authority in India, the International Conference on Harmonization Good Clinical Practice, and the Declaration of Helsinki. All patients provided written informed consent.
Contributions Author contributions: All authors contributed equally to this report. Zarir F Udwadia, Monika Tandon, Pawan Singh, Hanmant Barkate, Shabbir Rangwala, Amol Pendse, and Wen Wu had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. Study concept and design: Monika Tandon, Pawan Singh, Hanmant Barkate, Saiprasad Patil, and Wen Wu. Acquisition, analysis, or interpretation of data: Jatin Kadam, Amol..
References
Agrawal, Raju, Udwadia, Favipiravir: a new and emerging antiviral option in COVID-19, Med J Armed Forces India, doi:[368_TD$DIFF]10.1016/j.mjafi.2020.08.004
Buonaguro, Tagliamonte, Tornesello, Buonaguro, SARS-CoV-2 RNA polymerase as target for antiviral therapy, J Transl Med
Cai, Yang, Liu, Experimental treatment with favipiravir for COVID-19: an open-label control study, Engineering
Carmo, Pereira-Vaz, Mota, Clearance and persistence of SARS-CoV-2 RNA in patients with COVID-19, J Med Virol
Chen, Huang, Cheng, Favipiravir versus Arbidol for COVID-19: a randomized clinical trial, MedRxiv, doi:10.1101/2020.03.17.20037432
Delang, Abdelnabi, Neyts, Favipiravir as a potential countermeasure against neglected and emerging RNA viruses, Antiviral Res
Doi, Hibino, Hase, A prospective, randomized, open-label trial of early versus late favipiravir in hospitalized patients with COVID-19, Antimicrob Agents Chemother, doi:10.1128/AAC.01897-20
Dong, Hu, Gao, Discovering drugs to treat coronavirus disease 2019 (COVID-19), Drug Discov Ther
Fu, Han, Zhu, Risk factors for viral RNA shedding in COVID-19 patients, Eur Respir J, doi:10.1183/13993003.01190-2020
Furuta, Gowen, Takahashi, Shiraki, Smee et al., Favipiravir (T-705), a novel viral RNA polymerase inhibitor, Antiviral Res
Furuta, Komeno, Nakamura, Favipiravir (T-705), a broad spectrum inhibitor of viral RNA polymerase, Proc Jpn Acad Ser B Phys Biol Sci
Gogtay, Ravi, Thatte, Regulatory requirements for clinical trials in India: what academicians need to know, Indian J Anaesth
Goyal, Cardozo-Ojeda Fabian, Schiffer, Potency and timing of antiviral therapy as determinants of duration of SARS CoV-2 shedding and intensity of inflammatory response, Sci Adv, doi:10.1126/sciadv.abc7112
Heneghan, Onakpoya, Jones, Neuraminidase inhibitors for influenza: a systematic review and meta-analysis of regulatory and mortality data, Health Technol Assess
Ivashchenko, Dmitriev, Vostokova, AVIFAVIR for treatment of patients with moderate COVID-19: interim results of a phase II/III multicenter randomized clinical trial, Clin Infect Dis, doi:[382_TD$DIFF]10.1093/cid/ciaa1176
Pilkington, Pepperrell, Hill, A review of the safety of favipiravir-a potential treatment in the COVID-19 pandemic?, J Virus Erad
Qi, Yang, Jiang, Factors associated with the duration of viral shedding in adults with COVID-19 outside of Wuhan, China: a retrospective cohort study, Int J Infect Dis
Sanders, Monogue, Jodlowski, Cutrell, Pharmacologic treatments for coronavirus disease 2019 (COVID-19): a review, JAMA
Schiffer, Johnston, Corey, Song, Zhang et al., COVID-19 treatment: close to a cure? A rapid review of pharmacotherapies for the novel coronavirus, Int J Antimicrob Agents
Spinner, Gottlieb, Criner, Effect of remdesivir vs standard care on clinical status at 11 days in patients with moderate COVID-19: a randomized clinical trial, JAMA
Udwadia, Singh, Barkate, None, International Journal of Infectious Diseases
Udwadia, Singh, Barkate, None, International Journal of Infectious Diseases
Widders, Broom, Broom, SARS-CoV-2: the viral shedding vs infectivity dilemma, Infect Dis Health
Wu, Mcgoogan, Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72314 cases from the Chinese Center for Disease Control and Prevention, JAMA
Xu, Chen, Yuan, Factors associated with prolonged viral RNA shedding in patients with coronavirus disease 2019 (COVID-19), Clin Infect Dis
Zhou, Yang, Wang, A pneumonia outbreak associated with a new coronavirus of probable bat origin, Nature
DOI record:
{
"DOI": "10.1016/j.ijid.2020.11.142",
"ISSN": [
"1201-9712"
],
"URL": "http://dx.doi.org/10.1016/j.ijid.2020.11.142",
"alternative-id": [
"S120197122032453X"
],
"assertion": [
{
"label": "This article is maintained by",
"name": "publisher",
"value": "Elsevier"
},
{
"label": "Article Title",
"name": "articletitle",
"value": "Efficacy and safety of favipiravir, an oral RNA-dependent RNA polymerase inhibitor, in mild-to-moderate COVID-19: A randomized, comparative, open-label, multicenter, phase 3 clinical trial"
},
{
"label": "Journal Title",
"name": "journaltitle",
"value": "International Journal of Infectious Diseases"
},
{
"label": "CrossRef DOI link to publisher maintained version",
"name": "articlelink",
"value": "https://doi.org/10.1016/j.ijid.2020.11.142"
},
{
"label": "Content Type",
"name": "content_type",
"value": "article"
},
{
"label": "Copyright",
"name": "copyright",
"value": "© 2020 The Author(s). Published by Elsevier Ltd on behalf of International Society for Infectious Diseases."
}
],
"author": [
{
"affiliation": [],
"family": "Udwadia",
"given": "Zarir F.",
"sequence": "first"
},
{
"affiliation": [],
"family": "Singh",
"given": "Pawan",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Barkate",
"given": "Hanmant",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-4937-3188",
"affiliation": [],
"authenticated-orcid": false,
"family": "Patil",
"given": "Saiprasad",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-7563-2593",
"affiliation": [],
"authenticated-orcid": false,
"family": "Rangwala",
"given": "Shabbir",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-6333-7098",
"affiliation": [],
"authenticated-orcid": false,
"family": "Pendse",
"given": "Amol",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-1391-988X",
"affiliation": [],
"authenticated-orcid": false,
"family": "Kadam",
"given": "Jatin",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Wu",
"given": "Wen",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-8841-873X",
"affiliation": [],
"authenticated-orcid": false,
"family": "Caracta",
"given": "Cynthia F.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Tandon",
"given": "Monika",
"sequence": "additional"
}
],
"container-title": "International Journal of Infectious Diseases",
"container-title-short": "International Journal of Infectious Diseases",
"content-domain": {
"crossmark-restriction": true,
"domain": [
"clinicalkey.fr",
"clinicalkey.jp",
"clinicalkey.es",
"clinicalkey.com.au",
"clinicalkey.com",
"ijidonline.com",
"elsevier.com",
"sciencedirect.com"
]
},
"created": {
"date-parts": [
[
2020,
11,
16
]
],
"date-time": "2020-11-16T16:40:12Z",
"timestamp": 1605544812000
},
"deposited": {
"date-parts": [
[
2023,
6,
30
]
],
"date-time": "2023-06-30T16:45:55Z",
"timestamp": 1688143555000
},
"funder": [
{
"name": "lenmark Pharmaceuticals Limited, India"
}
],
"indexed": {
"date-parts": [
[
2024,
4,
8
]
],
"date-time": "2024-04-08T02:17:57Z",
"timestamp": 1712542677450
},
"is-referenced-by-count": 185,
"issued": {
"date-parts": [
[
2021,
2
]
]
},
"language": "en",
"license": [
{
"URL": "https://www.elsevier.com/tdm/userlicense/1.0/",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
2,
1
]
],
"date-time": "2021-02-01T00:00:00Z",
"timestamp": 1612137600000
}
},
{
"URL": "http://creativecommons.org/licenses/by-nc-nd/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2020,
11,
9
]
],
"date-time": "2020-11-09T00:00:00Z",
"timestamp": 1604880000000
}
}
],
"link": [
{
"URL": "https://api.elsevier.com/content/article/PII:S120197122032453X?httpAccept=text/xml",
"content-type": "text/xml",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://api.elsevier.com/content/article/PII:S120197122032453X?httpAccept=text/plain",
"content-type": "text/plain",
"content-version": "vor",
"intended-application": "text-mining"
}
],
"member": "78",
"original-title": [],
"page": "62-71",
"prefix": "10.1016",
"published": {
"date-parts": [
[
2021,
2
]
]
},
"published-print": {
"date-parts": [
[
2021,
2
]
]
},
"publisher": "Elsevier BV",
"reference": [
{
"DOI": "10.1016/j.mjafi.2020.08.004",
"article-title": "Favipiravir: a new and emerging antiviral option in COVID-19",
"author": "Agrawal",
"doi-asserted-by": "crossref",
"journal-title": "Med J Armed Forces India",
"key": "10.1016/j.ijid.2020.11.142_bib0005",
"year": "2020"
},
{
"key": "10.1016/j.ijid.2020.11.142_bib0010",
"unstructured": "Anti-influenza drug Avigan® Tablet Meets Primary Endpoint in Phase III Clinical Trial in Japan for COVID-19 patients. Press Release. September 23, 2020. https://www.fujifilm.com/jp/en/news/hq/5451."
},
{
"key": "10.1016/j.ijid.2020.11.142_bib0015",
"unstructured": "Duration of Isolation and Precautions for Adults with COVID-19. July 22, 2020. https://www.cdc.gov/coronavirus/2019-ncov/hcp/duration-isolation.html [Accessed 30 July 2020]."
},
{
"DOI": "10.1186/s12967-020-02355-3",
"article-title": "SARS-CoV-2 RNA polymerase as target for antiviral therapy",
"author": "Buonaguro",
"doi-asserted-by": "crossref",
"first-page": "185",
"issue": "1",
"journal-title": "J Transl Med",
"key": "10.1016/j.ijid.2020.11.142_bib0020",
"volume": "18",
"year": "2020"
},
{
"article-title": "Experimental treatment with favipiravir for COVID-19: an open-label control study",
"author": "Cai",
"first-page": "1192",
"journal-title": "Engineering (Beijing)",
"key": "10.1016/j.ijid.2020.11.142_bib0025",
"volume": "10",
"year": "2020"
},
{
"DOI": "10.1002/jmv.26103",
"article-title": "Clearance and persistence of SARS-CoV-2 RNA in patients with COVID-19",
"author": "Carmo",
"doi-asserted-by": "crossref",
"first-page": "2227",
"journal-title": "J Med Virol",
"key": "10.1016/j.ijid.2020.11.142_bib0030",
"volume": "10",
"year": "2020"
},
{
"article-title": "Favipiravir versus Arbidol for COVID-19: a randomized clinical trial",
"author": "Chen",
"issue": "March",
"journal-title": "MedRxiv",
"key": "10.1016/j.ijid.2020.11.142_bib0035",
"year": "2020"
},
{
"DOI": "10.1016/j.antiviral.2018.03.003",
"article-title": "Favipiravir as a potential countermeasure against neglected and emerging RNA viruses",
"author": "Delang",
"doi-asserted-by": "crossref",
"first-page": "85",
"journal-title": "Antiviral Res",
"key": "10.1016/j.ijid.2020.11.142_bib0040",
"volume": "153",
"year": "2018"
},
{
"DOI": "10.1128/AAC.01897-20",
"article-title": "A prospective, randomized, open-label trial of early versus late favipiravir in hospitalized patients with COVID-19",
"author": "Doi",
"doi-asserted-by": "crossref",
"first-page": "e01897",
"issue": "12",
"journal-title": "Antimicrob Agents Chemother",
"key": "10.1016/j.ijid.2020.11.142_bib0045",
"volume": "64",
"year": "2020"
},
{
"DOI": "10.5582/ddt.2020.01012",
"article-title": "Discovering drugs to treat coronavirus disease 2019 (COVID-19)",
"author": "Dong",
"doi-asserted-by": "crossref",
"first-page": "58",
"issue": "1",
"journal-title": "Drug Discov Ther",
"key": "10.1016/j.ijid.2020.11.142_bib0050",
"volume": "14",
"year": "2020"
},
{
"DOI": "10.1183/13993003.01190-2020",
"article-title": "Risk factors for viral RNA shedding in COVID-19 patients",
"author": "Fu",
"doi-asserted-by": "crossref",
"first-page": "2001190",
"issue": "1",
"journal-title": "Eur Respir J",
"key": "10.1016/j.ijid.2020.11.142_bib0055",
"volume": "56",
"year": "2020"
},
{
"DOI": "10.1016/j.antiviral.2013.09.015",
"article-title": "Favipiravir (T-705), a novel viral RNA polymerase inhibitor",
"author": "Furuta",
"doi-asserted-by": "crossref",
"first-page": "446",
"issue": "2",
"journal-title": "Antiviral Res",
"key": "10.1016/j.ijid.2020.11.142_bib0060",
"volume": "100",
"year": "2013"
},
{
"DOI": "10.2183/pjab.93.027",
"article-title": "Favipiravir (T-705), a broad spectrum inhibitor of viral RNA polymerase",
"author": "Furuta",
"doi-asserted-by": "crossref",
"first-page": "449",
"issue": "7",
"journal-title": "Proc Jpn Acad Ser B Phys Biol Sci",
"key": "10.1016/j.ijid.2020.11.142_bib0065",
"volume": "93",
"year": "2017"
},
{
"DOI": "10.4103/ija.IJA_143_17",
"article-title": "Regulatory requirements for clinical trials in India: what academicians need to know",
"author": "Gogtay",
"doi-asserted-by": "crossref",
"first-page": "192",
"issue": "3",
"journal-title": "Indian J Anaesth",
"key": "10.1016/j.ijid.2020.11.142_bib0070",
"volume": "61",
"year": "2017"
},
{
"DOI": "10.1126/sciadv.abc7112",
"article-title": "Potency and timing of antiviral therapy as determinants of duration of SARS CoV-2 shedding and intensity of inflammatory response",
"author": "Goyal",
"doi-asserted-by": "crossref",
"first-page": "eabc7112",
"issue": "47",
"journal-title": "Sci Adv",
"key": "10.1016/j.ijid.2020.11.142_bib0075",
"volume": "6",
"year": "2020"
},
{
"DOI": "10.3310/hta20420",
"article-title": "Neuraminidase inhibitors for influenza: a systematic review and meta-analysis of regulatory and mortality data",
"author": "Heneghan",
"doi-asserted-by": "crossref",
"first-page": "1",
"issue": "42",
"journal-title": "Health Technol Assess",
"key": "10.1016/j.ijid.2020.11.142_bib0080",
"volume": "20",
"year": "2016"
},
{
"article-title": "AVIFAVIR for treatment of patients with moderate COVID-19: interim results of a phase II/III multicenter randomized clinical trial",
"author": "Ivashchenko",
"journal-title": "Clin Infect Dis",
"key": "10.1016/j.ijid.2020.11.142_bib0085",
"year": "2020"
},
{
"DOI": "10.1016/S2055-6640(20)30016-9",
"article-title": "A review of the safety of favipiravir-a potential treatment in the COVID-19 pandemic?",
"author": "Pilkington",
"doi-asserted-by": "crossref",
"first-page": "45",
"issue": "2",
"journal-title": "J Virus Erad",
"key": "10.1016/j.ijid.2020.11.142_bib0090",
"volume": "6",
"year": "2020"
},
{
"DOI": "10.1016/j.ijid.2020.05.045",
"article-title": "Factors associated with the duration of viral shedding in adults with COVID-19 outside of Wuhan, China: a retrospective cohort study",
"author": "Qi",
"doi-asserted-by": "crossref",
"first-page": "531",
"journal-title": "Int J Infect Dis",
"key": "10.1016/j.ijid.2020.11.142_bib0095",
"volume": "96",
"year": "2020"
},
{
"article-title": "Pharmacologic treatments for coronavirus disease 2019 (COVID-19): a review",
"author": "Sanders",
"first-page": "1824",
"issue": "18",
"journal-title": "JAMA",
"key": "10.1016/j.ijid.2020.11.142_bib0100",
"volume": "323",
"year": "2020"
},
{
"DOI": "10.1093/ofid/ofaa232",
"article-title": "An early test-and-treat strategy for severe acute respiratory syndrome coronavirus 2",
"author": "Schiffer",
"doi-asserted-by": "crossref",
"issue": "7",
"journal-title": "Open Forum Infect Dis",
"key": "10.1016/j.ijid.2020.11.142_bib0105",
"volume": "7",
"year": "2020"
},
{
"DOI": "10.1016/j.ijantimicag.2020.106080",
"article-title": "COVID-19 treatment: close to a cure? A rapid review of pharmacotherapies for the novel coronavirus",
"author": "Song",
"doi-asserted-by": "crossref",
"first-page": "106080",
"issue": "2",
"journal-title": "Int J Antimicrob Agents",
"key": "10.1016/j.ijid.2020.11.142_bib0110",
"volume": "56",
"year": "2020"
},
{
"DOI": "10.1001/jama.2020.16349",
"article-title": "Effect of remdesivir vs standard care on clinical status at 11 days in patients with moderate COVID-19: a randomized clinical trial",
"author": "Spinner",
"doi-asserted-by": "crossref",
"first-page": "1048",
"issue": "11",
"journal-title": "JAMA",
"key": "10.1016/j.ijid.2020.11.142_bib0115",
"volume": "324",
"year": "2020"
},
{
"author": "The Center for Systems Science and Engineering at Johns Hopkins",
"key": "10.1016/j.ijid.2020.11.142_bib0120",
"series-title": "COVID-19 Global Case Dashboard",
"year": "2020"
},
{
"DOI": "10.1016/j.idh.2020.05.002",
"article-title": "SARS-CoV-2: the viral shedding vs infectivity dilemma",
"author": "Widders",
"doi-asserted-by": "crossref",
"first-page": "210",
"issue": "3",
"journal-title": "Infect Dis Health",
"key": "10.1016/j.ijid.2020.11.142_bib0125",
"volume": "25",
"year": "2020"
},
{
"author": "World Health Organization",
"key": "10.1016/j.ijid.2020.11.142_bib0130",
"series-title": "Timeline of WHO’s Response to COVID-19",
"year": "2020"
},
{
"DOI": "10.1001/jama.2020.2648",
"article-title": "Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72314 cases from the Chinese Center for Disease Control and Prevention",
"author": "Wu",
"doi-asserted-by": "crossref",
"first-page": "1239",
"issue": "13",
"journal-title": "JAMA",
"key": "10.1016/j.ijid.2020.11.142_bib0135",
"volume": "323",
"year": "2020"
},
{
"DOI": "10.1093/cid/ciaa351",
"article-title": "Factors associated with prolonged viral RNA shedding in patients with coronavirus disease 2019 (COVID-19)",
"author": "Xu",
"doi-asserted-by": "crossref",
"first-page": "799",
"issue": "15",
"journal-title": "Clin Infect Dis",
"key": "10.1016/j.ijid.2020.11.142_bib0140",
"volume": "71",
"year": "2020"
},
{
"DOI": "10.1038/s41586-020-2012-7",
"article-title": "A pneumonia outbreak associated with a new coronavirus of probable bat origin",
"author": "Zhou",
"doi-asserted-by": "crossref",
"first-page": "270",
"issue": "7798",
"journal-title": "Nature",
"key": "10.1016/j.ijid.2020.11.142_bib0145",
"volume": "579",
"year": "2020"
}
],
"reference-count": 29,
"references-count": 29,
"relation": {},
"resource": {
"primary": {
"URL": "https://linkinghub.elsevier.com/retrieve/pii/S120197122032453X"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"Infectious Diseases",
"Microbiology (medical)",
"General Medicine"
],
"subtitle": [],
"title": "Efficacy and safety of favipiravir, an oral RNA-dependent RNA polymerase inhibitor, in mild-to-moderate COVID-19: A randomized, comparative, open-label, multicenter, phase 3 clinical trial",
"type": "journal-article",
"update-policy": "http://dx.doi.org/10.1016/elsevier_cm_policy",
"volume": "103"
}