Compassionate Use of Remdesivir for Patients with Severe Covid-19
et al., NEJM, doi:10.1056/NEJMoa2007016, Apr 2020
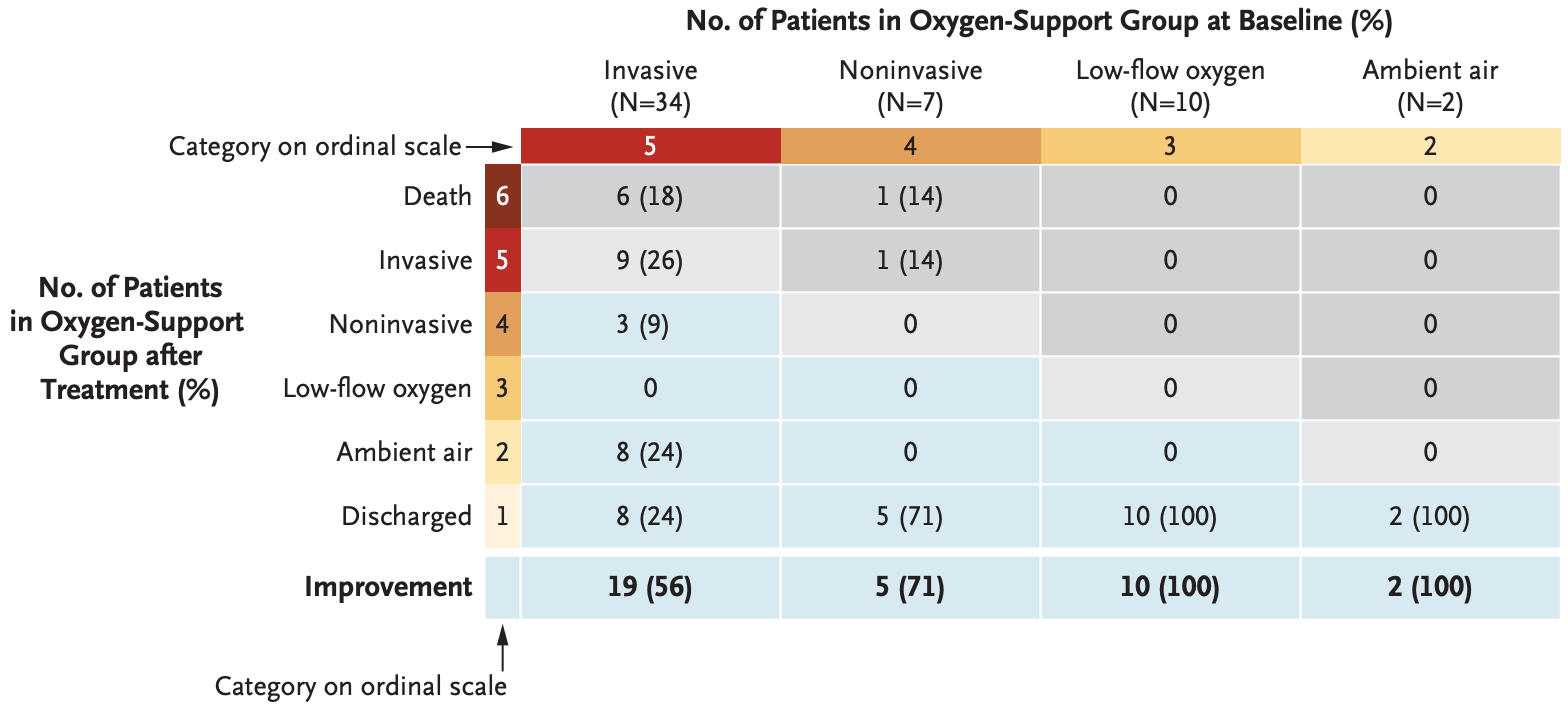
Report on compassionate use of remdesivir with 61 severe COVID-19 patients, showing clinical improvement in 36 of 53 patients.
Gérard, Zhou, Wu, Kamo, Choi, Kim show increased risk of acute kidney injury, Leo, Briciu, Muntean, Petrov, Arch show increased risk of liver injury, Negru, Cheng, Mohammed, Kwok, Zhu show increased risk of cardiac disorders, and Kwok, Merches, Akinci, Tang, Bagheri show increased risk of mitochondrial toxicity with remdesivir.
1.
Gérard et al., Remdesivir and Acute Renal Failure: A Potential Safety Signal From Disproportionality Analysis of the WHO Safety Database, Clinical Pharmacology & Therapeutics, doi:10.1002/cpt.2145.
2.
Zhou et al., Acute Kidney Injury and Drugs Prescribed for COVID-19 in Diabetes Patients: A Real-World Disproportionality Analysis, Frontiers in Pharmacology, doi:10.3389/fphar.2022.833679.
3.
Wu et al., Acute Kidney Injury Associated With Remdesivir: A Comprehensive Pharmacovigilance Analysis of COVID-19 Reports in FAERS, Frontiers in Pharmacology, doi:10.3389/fphar.2022.692828.
4.
Kamo et al., Association of Antiviral Drugs for the Treatment of COVID-19 With Acute Renal Failure, In Vivo, doi:10.21873/invivo.13637.
5.
Choi et al., Comparative effectiveness of combination therapy with nirmatrelvir–ritonavir and remdesivir versus monotherapy with remdesivir or nirmatrelvir–ritonavir in patients hospitalised with COVID-19: a target trial emulation study, The Lancet Infectious Diseases, doi:10.1016/S1473-3099(24)00353-0.
6.
Kim et al., Investigating the Safety Profile of Fast‐Track COVID‐19 Drugs Using the FDA Adverse Event Reporting System Database: A Comparative Observational Study, Pharmacoepidemiology and Drug Safety, doi:10.1002/pds.70043.
7.
Leo et al., Hepatocellular liver injury in hospitalized patients affected by COVID-19: Presence of different risk factors at different time points, Digestive and Liver Disease, doi:10.1016/j.dld.2021.12.014.
8.
Briciu et al., Evolving Clinical Manifestations and Outcomes in COVID-19 Patients: A Comparative Analysis of SARS-CoV-2 Variant Waves in a Romanian Hospital Setting, Pathogens, doi:10.3390/pathogens12121453.
9.
Muntean et al., Effects of COVID-19 on the Liver and Mortality in Patients with SARS-CoV-2 Pneumonia Caused by Delta and Non-Delta Variants: An Analysis in a Single Centre, Pharmaceuticals, doi:10.3390/ph17010003.
10.
Petrov et al., The Effect of Potentially Hepatotoxic Medicinal Products on Alanine Transaminase Levels in COVID-19 Patients: A Case–Control Study, Safety and Risk of Pharmacotherapy, doi:10.30895/2312-7821-2025-458.
11.
Arch et al., Evaluation of the effectiveness of remdesivir in treating severe COVID-19 using data from the ISARIC WHO Clinical Characterisation Protocol UK: a prospective, national cohort study, medRxiv, doi:10.1101/2021.06.18.21259072.
12.
Negru et al., Comparative Pharmacovigilance Analysis of Approved and Repurposed Antivirals for COVID-19: Insights from EudraVigilance Data, Biomedicines, doi:10.3390/biomedicines13061387.
13.
Cheng et al., Cardiovascular Safety of COVID-19 Treatments: A Disproportionality Analysis of Adverse Event Reports from the WHO VigiBase, Infectious Diseases and Therapy, doi:10.1007/s40121-025-01225-z.
14.
Mohammed et al., Bradycardia associated with remdesivir treatment in coronavirus disease 2019 patients: A propensity score-matched analysis, Medicine, doi:10.1097/MD.0000000000044501.
15.
Kwok et al., Remdesivir induces persistent mitochondrial and structural damage in human induced pluripotent stem cell-derived cardiomyocytes, Cardiovascular Research, doi:10.1093/cvr/cvab311.
16.
Zhu et al., Cardiovascular Risks of COVID-19 Therapeutics: Integrated Analysis of FAERS, Electronic Health Records, and Transcriptomics, Pharmaceuticals, doi:10.3390/ph19040574.
17.
Merches et al., The potential of remdesivir to affect function, metabolism and proliferation of cardiac and kidney cells in vitro, Archives of Toxicology, doi:10.1007/s00204-022-03306-1.
18.
Akinci et al., Elucidation of remdesivir cytotoxicity pathways through genome-wide CRISPR-Cas9 screening and transcriptomics, bioRxiv, doi:10.1101/2020.08.27.270819.
Grein et al., 10 Apr 2020, peer-reviewed, 59 authors.
Compassionate Use of Remdesivir for Patients with Severe Covid-19
New England Journal of Medicine, doi:10.1056/nejmoa2007016
BACKGROUND Remdesivir, a nucleotide analogue prodrug that inhibits viral RNA polymerases, has shown in vitro activity against SARS-CoV-2.
METHODS We provided remdesivir on a compassionate-use basis to patients hospitalized with Covid-19, the illness caused by infection with SARS-CoV-2. Patients were those with confirmed SARS-CoV-2 infection who had an oxygen saturation of 94% or less while they were breathing ambient air or who were receiving oxygen support. Patients received a 10-day course of remdesivir, consisting of 200 mg administered intravenously on day 1, followed by 100 mg daily for the remaining 9 days of treatment. This report is based on data from patients who received remdesivir during the period from January 25, 2020, through March 7, 2020, and have clinical data for at least 1 subsequent day.
RESULTS Of the 61 patients who received at least one dose of remdesivir, data from 8 could not be analyzed (including 7 patients with no post-treatment data and 1 with a dosing error). Of the 53 patients whose data were analyzed, 22 were in the United States, 22 in Europe or Canada, and 9 in Japan. At baseline, 30 patients (57%) were receiving mechanical ventilation and 4 (8%) were receiving extracorporeal membrane oxygenation. During a median follow-up of 18 days, 36 patients (68%) had an improvement in oxygen-support class, including 17 of 30 patients (57%) receiving mechanical ventilation who were extubated. A total of 25 patients (47%) were discharged, and 7 patients (13%) died; mortality was 18% (6 of 34) among patients receiving invasive ventilation and 5% (1 of 19) among those not receiving invasive ventilation.
CONCLUSIONS In this cohort of patients hospitalized for severe Covid-19 who were treated with compassionate-use remdesivir, clinical improvement was observed in 36 of 53 patients (68%). Measurement of efficacy will require ongoing randomized, placebocontrolled trials of remdesivir therapy. (Funded by Gilead Sciences.
The authors' affiliations are as follows: Cedars-Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S. Copyright © 2020 Massachusetts Medical Society. All rights reserved. n engl j med 382;24 nejm.org June 11, 2020
References
Arentz, Yim, Klaff, Characteristics and outcomes of 21 critically ill patients with COVID-19 in Washington State, JAMA
Baden, Rubin, Covid-19 -the search for effective therapy, N Engl J Med
Bhatraju, Ghassemieh, Nichols, Covid-19 in critically ill patients in the Seattle region -case series, N Engl J Med
Cao, Wang, Wen, A trial of lopinavir-ritonavir in adults hospitalized with severe Covid-19, N Engl J Med
Cucinotta, Vanelli, WHO declares COVID-19 a pandemic, Acta Biomed
De Wit, Feldmann, Cronin, Prophylactic and therapeutic remdesivir (GS-5734) treatment in the rhesus macaque model of MERS-CoV infection, Proc Natl Acad Sci U S A
Fauci, Lane, Redfield, Covid-19 -navigating the uncharted, N Engl J Med
Holshue, Debolt, Lindquist, First case of 2019 novel coronavirus in the United States, N Engl J Med
Kujawski, Wong, Collins, First 12 patients with coronavirus disease 2019 (COVID-19) in the United States. medRxiv, doi:10.1101/2020.03.09.20032896v1
Lescure, Bouadma, Nguyen, Clinical and virological data of the first cases of COVID-19 in Europe: a case series, Lancet Infect Dis
Mahase, Kmietowicz, Covid-19: doctors are told not to perform CPR on patients in cardiac arrest, BMJ
Mulangu, Dodd, Rt, A randomized, controlled trial of Ebola virus disease therapeutics, N Engl J Med
Onder, Rezza, Brusaferro, Case-fatality rate and characteristics of patients dying in relation to COVID-19 in Italy, JAMA
Poston, Patel, Davis, Management of critically ill adults with COVID-19, JAMA
Rodriguez-Morales, Cardona-Ospina, Ocampo, Clinical, laboratory and imaging features of COVID-19: a systematic review and meta-analysis, Travel Med Infect Dis
Sheahan, Sims, Graham, Broad-spectrum antiviral GS-5734 inhibits both epidemic and zoonotic coronaviruses, Sci Transl Med
Sheahan, Sims, Leist, Comparative therapeutic efficacy of remdesivir and combination lopinavir, ritonavir, and interferon beta against MERS-CoV, Nat Commun
Shen, Wang, Zhao, Treatment of 5 critically ill patients with COVID-19 with convalescent plasma, JAMA
Spinelli, Pellino, COVID-19 pandemic: perspectives on an unfolding crisis, Br J Surg
Touret, De Lamballerie, Of chloroquine and COVID-19, Antiviral Res
Wang, Cao, Zhang, Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro, Cell Res
Wang, Hu, Hu, Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China, JAMA
Weiss, Murdoch, Clinical course and mortality risk of severe COVID-19, Lancet
Wu, Chen, Cai, Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China, JAMA Intern Med
Wu, Mcgoogan, Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72 314 cases from the Chinese Center for Disease Control and Prevention, JAMA
Zhang, Shi, Wang, Liver injury in COVID-19: management and challenges, Lancet Gastroenterol Hepatol
Zhou, Yu, Du, Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study, Lancet
DOI record:
{
"DOI": "10.1056/nejmoa2007016",
"ISSN": [
"0028-4793",
"1533-4406"
],
"URL": "http://dx.doi.org/10.1056/NEJMoa2007016",
"alternative-id": [
"10.1056/NEJMoa2007016"
],
"author": [
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Grein",
"given": "Jonathan",
"sequence": "first"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Ohmagari",
"given": "Norio",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Shin",
"given": "Daniel",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Diaz",
"given": "George",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Asperges",
"given": "Erika",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Castagna",
"given": "Antonella",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Feldt",
"given": "Torsten",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Green",
"given": "Gary",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Green",
"given": "Margaret L.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Lescure",
"given": "François-Xavier",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-5606-8712",
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"authenticated-orcid": false,
"family": "Nicastri",
"given": "Emanuele",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-6066-2853",
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"authenticated-orcid": false,
"family": "Oda",
"given": "Rentaro",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Yo",
"given": "Kikuo",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Quiros-Roldan",
"given": "Eugenia",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Studemeister",
"given": "Alex",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Redinski",
"given": "John",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Ahmed",
"given": "Seema",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Bernett",
"given": "Jorge",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Chelliah",
"given": "Daniel",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Chen",
"given": "Danny",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Chihara",
"given": "Shingo",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Cohen",
"given": "Stuart H.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Cunningham",
"given": "Jennifer",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "D’Arminio Monforte",
"given": "Antonella",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Ismail",
"given": "Saad",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Kato",
"given": "Hideaki",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Lapadula",
"given": "Giuseppe",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "L’Her",
"given": "Erwan",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Maeno",
"given": "Toshitaka",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Majumder",
"given": "Sumit",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Massari",
"given": "Marco",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Mora-Rillo",
"given": "Marta",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Mutoh",
"given": "Yoshikazu",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Nguyen",
"given": "Duc",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Verweij",
"given": "Ewa",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Zoufaly",
"given": "Alexander",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Osinusi",
"given": "Anu O.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "DeZure",
"given": "Adam",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Zhao",
"given": "Yang",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Zhong",
"given": "Lijie",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Chokkalingam",
"given": "Anand",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Elboudwarej",
"given": "Emon",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Telep",
"given": "Laura",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Timbs",
"given": "Leighann",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Henne",
"given": "Ilana",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Sellers",
"given": "Scott",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Cao",
"given": "Huyen",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Tan",
"given": "Susanna K.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Winterbourne",
"given": "Lucinda",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Desai",
"given": "Polly",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Mera",
"given": "Robertino",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Gaggar",
"given": "Anuj",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Myers",
"given": "Robert P.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Brainard",
"given": "Diana M.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Childs",
"given": "Richard",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Cedars–Sinai Medical Center, Los Angeles (J.G.), El Camino Hospital, Mountain View (D.S., D. Chelliah), Sutter Santa Rosa Regional Hospital, Santa Rosa (G.G.), Regional Medical Center (A.S., J.R.) and Good Samaritan Hospital (S.M.), San Jose, John Muir Health, Walnut Creek (J.B.), UC Davis Health, Sacramento (S.H.C.), NorthBay Medical Center, Fairfield (S.I.), and Gilead Sciences, Foster City (A.O.O., A.D., Y.Z., L.Z., A. Chokkalingam, E.E., L. Telep, L. Timbs, I.H., S.S., H.C., S.K.T., L.W., P.D.,..."
}
],
"family": "Flanigan",
"given": "Timothy",
"sequence": "additional"
}
],
"container-title": "New England Journal of Medicine",
"container-title-short": "N Engl J Med",
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2020,
4,
10
]
],
"date-time": "2020-04-10T19:09:56Z",
"timestamp": 1586545796000
},
"deposited": {
"date-parts": [
[
2020,
6,
10
]
],
"date-time": "2020-06-10T20:58:06Z",
"timestamp": 1591822686000
},
"funder": [
{
"DOI": "10.13039/100005564",
"award": [
"N/A"
],
"doi-asserted-by": "publisher",
"name": "Gilead Sciences"
}
],
"indexed": {
"date-parts": [
[
2024,
5,
14
]
],
"date-time": "2024-05-14T09:46:00Z",
"timestamp": 1715679960887
},
"is-referenced-by-count": 1842,
"issue": "24",
"issued": {
"date-parts": [
[
2020,
6,
11
]
]
},
"journal-issue": {
"issue": "24",
"published-print": {
"date-parts": [
[
2020,
6,
11
]
]
}
},
"language": "en",
"license": [
{
"URL": "http://www.nejmgroup.org/legal/terms-of-use.htm",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2020,
6,
11
]
],
"date-time": "2020-06-11T00:00:00Z",
"timestamp": 1591833600000
}
}
],
"link": [
{
"URL": "http://www.nejm.org/doi/pdf/10.1056/NEJMoa2007016",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "150",
"original-title": [],
"page": "2327-2336",
"prefix": "10.1056",
"published": {
"date-parts": [
[
2020,
6,
11
]
]
},
"published-print": {
"date-parts": [
[
2020,
6,
11
]
]
},
"publisher": "Massachusetts Medical Society",
"reference": [
{
"author": "Cucinotta D",
"first-page": "157",
"journal-title": "Acta Biomed",
"key": "r1",
"volume": "91",
"year": "2020"
},
{
"DOI": "10.1002/bjs.11627",
"doi-asserted-by": "publisher",
"key": "r2"
},
{
"DOI": "10.1056/NEJMe2002387",
"doi-asserted-by": "publisher",
"key": "r3"
},
{
"DOI": "10.1136/bmj.m1282",
"doi-asserted-by": "publisher",
"key": "r4"
},
{
"DOI": "10.1016/j.tmaid.2020.101623",
"doi-asserted-by": "publisher",
"key": "r5"
},
{
"DOI": "10.1016/S0140-6736(20)30633-4",
"doi-asserted-by": "publisher",
"key": "r6"
},
{
"DOI": "10.1001/jamainternmed.2020.0994",
"doi-asserted-by": "publisher",
"key": "r7"
},
{
"DOI": "10.1001/jama.2020.4683",
"doi-asserted-by": "publisher",
"key": "r8"
},
{
"DOI": "10.1001/jama.2020.4914",
"doi-asserted-by": "publisher",
"key": "r9"
},
{
"DOI": "10.1056/NEJMoa2001282",
"doi-asserted-by": "publisher",
"key": "r10"
},
{
"DOI": "10.1001/jama.2020.4783",
"doi-asserted-by": "publisher",
"key": "r11"
},
{
"DOI": "10.1016/j.antiviral.2020.104762",
"doi-asserted-by": "publisher",
"key": "r12"
},
{
"DOI": "10.1056/NEJMe2005477",
"doi-asserted-by": "publisher",
"key": "r13"
},
{
"DOI": "10.1073/pnas.1922083117",
"doi-asserted-by": "publisher",
"key": "r14"
},
{
"DOI": "10.1126/scitranslmed.aal3653",
"doi-asserted-by": "publisher",
"key": "r15"
},
{
"DOI": "10.1038/s41467-019-13940-6",
"doi-asserted-by": "publisher",
"key": "r16"
},
{
"DOI": "10.1038/s41422-020-0282-0",
"doi-asserted-by": "publisher",
"key": "r17"
},
{
"DOI": "10.1056/NEJMoa1910993",
"doi-asserted-by": "publisher",
"key": "r18"
},
{
"DOI": "10.1056/NEJMoa2001191",
"doi-asserted-by": "publisher",
"key": "r21"
},
{
"DOI": "10.1016/S1473-3099(20)30200-0",
"doi-asserted-by": "publisher",
"key": "r22"
},
{
"DOI": "10.1001/jama.2020.4326",
"doi-asserted-by": "publisher",
"key": "r23"
},
{
"DOI": "10.1001/jama.2020.1585",
"doi-asserted-by": "publisher",
"key": "r24"
},
{
"DOI": "10.1001/jama.2020.2648",
"doi-asserted-by": "publisher",
"key": "r25"
},
{
"DOI": "10.1016/S0140-6736(20)30566-3",
"doi-asserted-by": "publisher",
"key": "r26"
},
{
"DOI": "10.1056/NEJMoa2004500",
"doi-asserted-by": "publisher",
"key": "r27"
},
{
"DOI": "10.1016/S2468-1253(20)30057-1",
"doi-asserted-by": "publisher",
"key": "r29"
}
],
"reference-count": 26,
"references-count": 26,
"relation": {
"has-review": [
{
"asserted-by": "object",
"id": "10.3410/f.737712055.793588342",
"id-type": "doi"
}
]
},
"resource": {
"primary": {
"URL": "http://www.nejm.org/doi/10.1056/NEJMoa2007016"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "Compassionate Use of Remdesivir for Patients with Severe Covid-19",
"type": "journal-article",
"volume": "382"
}