Paxlovid shows organ-specific and age-specific impacts on risk of developing post-acute sequelae of COVID-19
et al., Communications Medicine, doi:10.1038/s43856-026-01535-4, Mar 2026
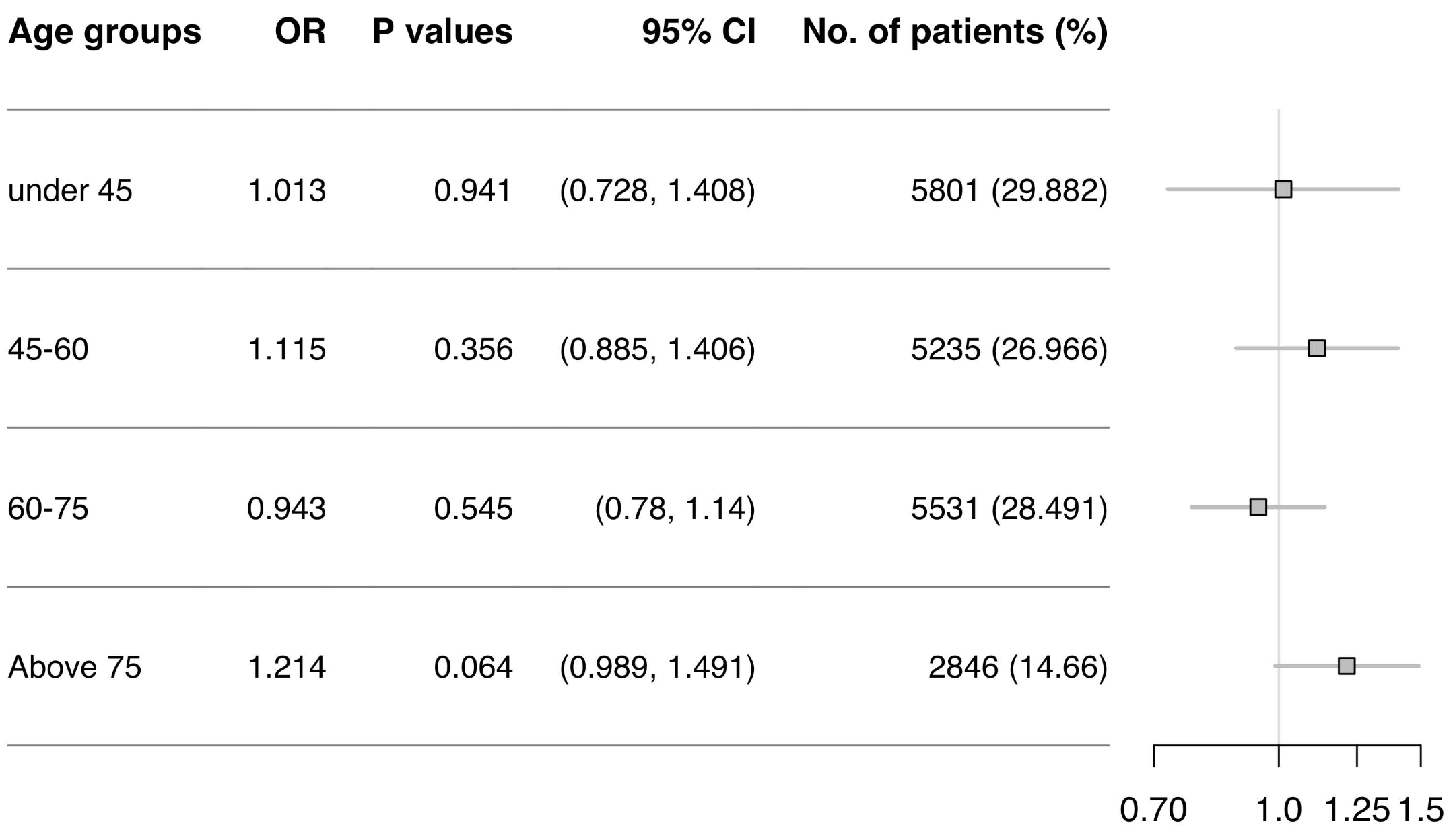
Retrospective 19,413 COVID-19 outpatient and hospitalized patients showing no overall reduction in long COVID risk with paxlovid. Stratification by organ system found a 37% reduction in gastrointestinal PASC (OR 0.63, p=0.002) - biologically plausible given oral drug absorption through gut tissue with established viral tropism - and a 97.4% increase in eye/ear PASC (OR 1.97, p=0.035). A sensitivity analysis of non-hospitalized patients aged 60-75 found a modest 16% reduction (OR 0.84, p=0.044), but this required subsetting to reach significance while the authors did not discuss a near-significant harm signal in the 75+ group (OR 1.21, p=0.064) - the primary target population for paxlovid.
Disease severity was measured using only three crude categories (outpatient/hospitalized/ICU), with 85-99% of each group classified as outpatient, meaning the severity covariate had no discriminating power for the vast majority of the cohort and cannot address confounding by indication within outpatients. The control group was not restricted to paxlovid-eligible patients, meaning contraindicated (higher-risk) patients contaminate the comparator, expected to overestimate benefit. Paxlovid recipients differ substantially by race, vaccination, and hospitalization (2% vs 9%), suggesting significant healthy-user and access bias that entropy balancing on observed covariates is unlikely to fully resolve. No adjustment for multiple comparisons was performed across dozens of tests - the eye/ear and non-hospitalized 60-75 findings do not survive Bonferroni correction. The abstract contradicts the main text, reporting OR 0.832 for non-hospitalized age range 65-75, while the results report OR 0.837 for a different age range 60-75. The supplementary methodology describes a different matched case-control design spanning 2020-2023 with 2018 pre-pandemic controls and minimum one-year follow-up, contradicting the main text's January-June 2022 cohort design with median 4.5-month follow-up.
The biases in this study all point towards making paxlovid look better than it actually is - confounding by contraindication, healthy-user bias, access bias, crude severity measurement and confounding by indication, and confounding by adjuvant treatment will all overestimate benefit. After correction, the signal for harm in the 75+ group is likely to become significant (currently p=0.06). This is biologically plausible - the 75+ group is where paxlovid's drug interaction profile creates the greatest risk of iatrogenic harm.
Ritonavir is one of the most potent CYP3A4 inhibitors known. It was originally developed as an HIV protease inhibitor but is used in paxlovid purely as a pharmacokinetic booster to block the liver enzyme that would otherwise rapidly metabolize nirmatrelvir. However, CYP3A4 also metabolizes a huge number of other drugs, and blocking it for 5 days dramatically raises blood levels of other CYP3A4-metabolized drugs the patient is taking. Even after the 5-day course ends, the downstream effects of the pharmacokinetic disruption may trigger a cascade of complications. There is also a more direct mechanism - elderly patients are more likely to have age-related liver, kidney, or heart problems, which may require an adjustment in the dose for patients receiving ritonavir. Reduced hepatic and renal clearance means both nirmatrelvir and ritonavir persist at higher concentrations for longer, potentially turning the standard 5-day dose into an overdose in some elderly patients with subclinical organ impairment.
Resistance. Variants may be resistant to paxlovid1-9. Use may promote the emergence of variants that weaken host immunity and potentially contribute to long COVID10. Confounding by contraindication. Hoertel et al. find that over 50% of patients that died had a contraindication for the use of Paxlovid11. Retrospective studies that do not exclude contraindicated patients may significantly overestimate efficacy. Black box warning. The FDA notes that severe, life-threatening, and/or fatal adverse reactions due to drug interactions have been reported in patients treated with paxlovid12. Kidney and liver injury. Studies show significantly increased risk of acute kidney injury13 and liver injury14,15. Viral rebound. Studies show significantly increased risk of replication-competent viral rebound16-18.
Standard of Care (SOC) for COVID-19 in the study country,
the USA, is very poor with very low average efficacy for approved treatments19.
Only expensive, high-profit treatments were approved for early treatment. Low-cost treatments were excluded, reducing the probability of early treatment due to access and cost barriers, and eliminating complementary and synergistic benefits seen with many low-cost treatments.
|
risk of long COVID, 6.7% higher, HR 1.07, p = 0.29, all patients.
|
|
risk of long COVID, 1.3% higher, HR 1.01, p = 0.94, <45.
|
|
risk of long COVID, 11.5% higher, HR 1.11, p = 0.36, 45-60.
|
|
risk of long COVID, 5.7% lower, HR 0.94, p = 0.55, 60-75.
|
|
risk of long COVID, 21.4% higher, HR 1.21, p = 0.06, >75.
|
|
risk of long COVID, 1.7% higher, HR 1.02, p = 0.85, all patients.
|
|
risk of long COVID, 5.5% higher, HR 1.05, p = 0.73, outpatients, <45.
|
|
risk of long COVID, 8.2% higher, HR 1.08, p = 0.51, outpatients, 45-60.
|
|
risk of long COVID, 16.3% lower, HR 0.84, p = 0.04, outpatients, 60-75.
|
|
risk of long COVID, 16.9% higher, HR 1.17, p = 0.13, outpatients, >75.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
1.
Zhou et al., Nirmatrelvir-resistant SARS-CoV-2 variants with high fitness in an infectious cell culture system, Science Advances, doi:10.1126/sciadv.add7197.
2.
Moghadasi et al., Rapid resistance profiling of SARS-CoV-2 protease inhibitors, npj Antimicrobials and Resistance, doi:10.1038/s44259-023-00009-0.
3.
Jochmans et al., The Substitutions L50F, E166A, and L167F in SARS-CoV-2 3CLpro Are Selected by a Protease Inhibitor In Vitro and Confer Resistance To Nirmatrelvir, mBio, doi:10.1128/mbio.02815-22.
4.
Lopez et al., SARS-CoV-2 Resistance to Small Molecule Inhibitors, Current Clinical Microbiology Reports, doi:10.1007/s40588-024-00229-6.
5.
Zvornicanin et al., Molecular Mechanisms of Drug Resistance and Compensation in SARS-CoV-2 Main Protease: The Interplay Between E166 and L50, bioRxiv, doi:10.1101/2025.01.24.634813.
6.
Vukovikj et al., Impact of SARS-CoV-2 variant mutations on susceptibility to monoclonal antibodies and antiviral drugs: a non-systematic review, April 2022 to October 2024, Eurosurveillance, doi:10.2807/1560-7917.ES.2025.30.10.2400252.
7.
Deschenes et al., Functional and structural characterization of treatment-emergent nirmatrelvir resistance mutations at low frequencies in the main protease (Mpro) reveals a unique evolutionary route for SARS-CoV-2 to gain resistance, The Journal of Infectious Diseases, doi:10.1093/infdis/jiaf294.
8.
Zhou (B) et al., SARS-CoV-2 Mpro inhibitor ensitrelvir: asymmetrical cross-resistance with nirmatrelvir and emerging resistance hotspots, Emerging Microbes & Infections, doi:10.1080/22221751.2025.2552716.
9.
Chen et al., SARS-CoV-2 3CLpro mutations T21I and E166A confer differential resistance to simnotrelvir, bofutrelvir, and ensitrelvir, Journal of Virology, doi:10.1128/jvi.02223-25.
10.
Thomas et al., Nirmatrelvir-Resistant Mutations in SARS-CoV-2 Mpro Enhance Host Immune Evasion via Cleavage of NF-κB Essential Modulator, bioRxiv, doi:10.1101/2024.10.18.619137.
11.
Hoertel et al., Prevalence of Contraindications to Nirmatrelvir-Ritonavir Among Hospitalized Patients With COVID-19 at Risk for Progression to Severe Disease, JAMA Network Open, doi:10.1001/jamanetworkopen.2022.42140.
12.
FDA, Fact sheet for healthcare providers: emergency use authorization for paxlovid, www.fda.gov/media/155050/download.
13.
Kamo et al., Association of Antiviral Drugs for the Treatment of COVID-19 With Acute Renal Failure, In Vivo, doi:10.21873/invivo.13637.
14.
Wang et al., Development and validation of a nomogram to assess the occurrence of liver dysfunction in patients with COVID-19 pneumonia in the ICU, BMC Infectious Diseases, doi:10.1186/s12879-025-10684-1.
15.
Siby et al., Temporal Trends in Serious Adverse Events Associated with Oral Antivirals During the COVID-19 Pandemic: Insights from the FAERS Database (2020–2023), Open Forum Infectious Diseases, doi:10.1093/ofid/ofaf695.1825.
16.
Edelstein et al., SARS-CoV-2 virologic rebound with nirmatrelvir-ritonavir therapy, medRxiv, doi:10.1101/2023.06.23.23288598.
17.
Shah et al., SARS-CoV-2 infectious shedding and rebound among adults with and without oral antiviral use: two case-ascertained prospective household studies, The Lancet Microbe, doi:10.1016/j.lanmic.2025.101227.
Azhir et al., 24 Mar 2026, retrospective, USA, peer-reviewed, 5 authors, study period 1 January, 2022 - 7 June, 2022.
Contact: hestiri@mgh.harvard.edu.
Abstract: ARTICLE IN PRESS
Communications Medicine
https://doi.org/10.1038/s43856-026-01535-4
Article in Press
Paxlovid shows organ-specific and age-specific
impacts on risk of developing post-acute sequelae
of COVID-19
Alaleh Azhir, Jingya Cheng, Jiazi Tian, Shawn N. Murphy & Hossein Estiri
S
Received: 1 May 2025
PR
We are providing an unedited version of this manuscript to give early access to its
findings. Before final publication, the manuscript will undergo further editing. Please
note there may be errors present which affect the content, and all legal disclaimers
apply.
LE
IN
If this paper is publishing under a Transparent Peer Review model then Peer
Review reports will publish with the final article.
A
R
TI
C
Cite this article as: Azhir, A., Cheng, J.,
Tian, J. et al. Paxlovid shows organspecific and age-specific impacts on
risk of developing post-acute
sequelae of COVID-19. Commun Med
(2026). https://doi.org/10.1038/
s43856-026-01535-4
ES
Accepted: 9 March 2026
© The Author(s) 2026. Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International
License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit
to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not
have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this
article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the
article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain
permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
ARTICLE IN PRESS
Paxlovid shows organ-specific and age-specific
impacts on risk of developing post-acute sequelae of
COVID-19
Alaleh Azhir, MD, MSc1,2*; Jingya Cheng, M.B.1*; Jiazi Tian, M.Sc.1; Shawn N. Murphy, MD, PhD3
Hossein Estiri, PhD1†
1 Department of Medicine, Massachusetts General Hospital, Boston, MA, USA
S
2 Department of Medicine, Brigham and Women's Hospital, Boston, MA, USA
A
R
TI
C
LE
IN
PR
ES
3 Department of Neurology, Massachusetts General Hospital, Boston, MA, USA
* contributed equally
† Corresponding author: Hossein Estiri
Email: hestiri@mgh.harvard.edu
399 Revolution Drive, Suite 790, Somerville, MA, 02145, USA
ARTICLE IN PRESS
Abstract
Background: The impact of antiviral therapies, including Paxlovid, on post-acute sequelae of
COVID-19 (PASC) remains inconclusive.
S
Methods: We analyzed data from 19,413 patients (age > 18) from a validated PASC research
cohort in New England who experienced at least one COVID-19 infection episode between
January 1, 2022, and June 7, 2022, totaling 22,094 episodes. Multivariable logistic regression
with inverse probability weights was used to infer the causal effects of Paxlovid treatment during
acute infection and the risk of PASC overall (primary outcome), stratified by age group and organ
system.
IN
PR
ES
Results: Across all age groups, Paxlovid shows no statistically significant effect in lowering overall
PASC risk. Stratification by organ system reveals a..
DOI record:
{
"DOI": "10.1038/s43856-026-01535-4",
"ISSN": [
"2730-664X"
],
"URL": "http://dx.doi.org/10.1038/s43856-026-01535-4",
"alternative-id": [
"1535"
],
"assertion": [
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "Received",
"name": "received",
"order": 1,
"value": "1 May 2025"
},
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "Accepted",
"name": "accepted",
"order": 2,
"value": "9 March 2026"
},
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "First Online",
"name": "first_online",
"order": 3,
"value": "24 March 2026"
},
{
"group": {
"label": "Competing interests",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 1,
"value": "The authors declare no competing interests."
}
],
"author": [
{
"affiliation": [],
"family": "Azhir",
"given": "Alaleh",
"sequence": "first"
},
{
"ORCID": "https://orcid.org/0009-0006-1236-1614",
"affiliation": [],
"authenticated-orcid": false,
"family": "Cheng",
"given": "Jingya",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0003-0599-3438",
"affiliation": [],
"authenticated-orcid": false,
"family": "Tian",
"given": "Jiazi",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0002-1905-8806",
"affiliation": [],
"authenticated-orcid": false,
"family": "Murphy",
"given": "Shawn N.",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0002-0204-8978",
"affiliation": [],
"authenticated-orcid": false,
"family": "Estiri",
"given": "Hossein",
"sequence": "additional"
}
],
"container-title": "Communications Medicine",
"container-title-short": "Commun Med",
"content-domain": {
"crossmark-restriction": false,
"domain": [
"link.springer.com"
]
},
"created": {
"date-parts": [
[
2026,
3,
24
]
],
"date-time": "2026-03-24T20:35:39Z",
"timestamp": 1774384539000
},
"deposited": {
"date-parts": [
[
2026,
3,
24
]
],
"date-time": "2026-03-24T20:35:49Z",
"timestamp": 1774384549000
},
"funder": [
{
"DOI": "10.13039/100000060",
"award": [
"R01AI165535"
],
"award-info": [
{
"award-number": [
"R01AI165535"
]
}
],
"doi-asserted-by": "publisher",
"id": [
{
"asserted-by": "publisher",
"id": "10.13039/100000060",
"id-type": "DOI"
}
],
"name": "U.S. Department of Health & Human Services | NIH | National Institute of Allergy and Infectious Diseases"
}
],
"indexed": {
"date-parts": [
[
2026,
3,
24
]
],
"date-time": "2026-03-24T22:26:31Z",
"timestamp": 1774391191549,
"version": "3.50.1"
},
"is-referenced-by-count": 0,
"issued": {
"date-parts": [
[
2026,
3,
24
]
]
},
"language": "en",
"license": [
{
"URL": "https://creativecommons.org/licenses/by-nc-nd/4.0",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
3,
24
]
],
"date-time": "2026-03-24T00:00:00Z",
"timestamp": 1774310400000
}
},
{
"URL": "https://creativecommons.org/licenses/by-nc-nd/4.0",
"content-version": "am",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
3,
24
]
],
"date-time": "2026-03-24T00:00:00Z",
"timestamp": 1774310400000
}
}
],
"link": [
{
"URL": "https://www.nature.com/articles/s43856-026-01535-4",
"content-type": "text/html",
"content-version": "vor",
"intended-application": "text-mining"
}
],
"member": "297",
"original-title": [],
"prefix": "10.1038",
"published": {
"date-parts": [
[
2026,
3,
24
]
]
},
"published-online": {
"date-parts": [
[
2026,
3,
24
]
]
},
"publisher": "Springer Science and Business Media LLC",
"reference": [
{
"DOI": "10.15620/cdc:132417",
"doi-asserted-by": "publisher",
"key": "1535_CR1",
"unstructured": "Adjaye-Gbewonyo, D., Vahratian, A., Cria, G. P. & Bertolli, J. Long COVID in Adults: United States, 2022. (National Center for Health Statistics (U.S.), 2023). https://doi.org/10.15620/cdc:132417."
},
{
"DOI": "10.1056/NEJMoa2118542",
"doi-asserted-by": "crossref",
"key": "1535_CR2",
"unstructured": "Hammond, J. et al. Oral nirmatrelvir for high-risk, nonhospitalized adults with Covid-19. N. Engl. J. Med. 386, 1397–1408 (2022)."
},
{
"DOI": "10.1016/S1473-3099(23)00011-7",
"doi-asserted-by": "crossref",
"key": "1535_CR3",
"unstructured": "Aggarwal, N. R. et al. Real-world use of nirmatrelvir-ritonavir in outpatients with COVID-19 during the era of omicron variants including BA.4 and BA.5 in Colorado, USA: a retrospective cohort study. Lancet Infect. Dis. 23, 696–705 (2023)."
},
{
"DOI": "10.1001/jamainternmed.2023.0743",
"author": "Y Xie",
"doi-asserted-by": "crossref",
"first-page": "554",
"journal-title": "JAMA Intern. Med.",
"key": "1535_CR4",
"unstructured": "Xie, Y., Choi, T. & Al-Aly, Z. Association of treatment with nirmatrelvir and the risk of post-COVID-19 condition. JAMA Intern. Med. 183, 554–564 (2023).",
"volume": "183",
"year": "2023"
},
{
"DOI": "10.1016/j.jinf.2024.106190",
"doi-asserted-by": "crossref",
"key": "1535_CR5",
"unstructured": "Jiang, J. et al. Early use of oral antiviral drugs and the risk of post COVID-19 syndrome: a systematic review and network meta-analysis. J. Infect. 89, 106190 (2024)."
},
{
"DOI": "10.1002/jmv.29333",
"doi-asserted-by": "crossref",
"key": "1535_CR6",
"unstructured": "Durstenfeld, M. S. et al. Association of nirmatrelvir for acute SARS-CoV-2 infection with subsequent Long COVID symptoms in an observational cohort study. J. Med. Virol. 96, e29333 (2024)."
},
{
"DOI": "10.7326/M23-1394",
"author": "GN Ioannou",
"doi-asserted-by": "crossref",
"first-page": "1486",
"journal-title": "Ann. Intern. Med.",
"key": "1535_CR7",
"unstructured": "Ioannou, G. N. et al. Effectiveness of nirmatrelvir-ritonavir against the development of post-COVID-19 conditions among U.S. veterans: a target trial emulation. Ann. Intern. Med. 176, 1486–1497 (2023).",
"volume": "176",
"year": "2023"
},
{
"DOI": "10.7326/M22-3565",
"author": "KL Bajema",
"doi-asserted-by": "crossref",
"first-page": "807",
"journal-title": "Ann. Intern. Med.",
"key": "1535_CR8",
"unstructured": "Bajema, K. L. et al. Effectiveness of COVID-19 treatment with nirmatrelvir-ritonavir or molnupiravir among U.S. veterans: target trial emulation studies with one-month and six-month outcomes. Ann. Intern. Med. 176, 807–816 (2023).",
"volume": "176",
"year": "2023"
},
{
"DOI": "10.1001/jamainternmed.2023.5099",
"author": "KW Fung",
"doi-asserted-by": "crossref",
"first-page": "1404",
"journal-title": "JAMA Intern. Med.",
"key": "1535_CR9",
"unstructured": "Fung, K. W., Baye, F., Baik, S. H. & McDonald, C. J. Nirmatrelvir and molnupiravir and post-COVID-19 condition in older patients. JAMA Intern. Med. 183, 1404–1406 (2023).",
"volume": "183",
"year": "2023"
},
{
"DOI": "10.1016/S1473-3099(24)00217-2",
"author": "H Wang",
"doi-asserted-by": "crossref",
"first-page": "1130",
"journal-title": "Lancet Infect. Dis.",
"key": "1535_CR10",
"unstructured": "Wang, H. et al. Association of nirmatrelvir-ritonavir with post-acute sequelae and mortality in patients admitted to hospital with COVID-19: a retrospective cohort study. Lancet Infect. Dis. 24, 1130–1140 (2024).",
"volume": "24",
"year": "2024"
},
{
"DOI": "10.1001/jamainternmed.2024.2007",
"author": "LN Geng",
"doi-asserted-by": "crossref",
"first-page": "1024",
"journal-title": "JAMA Intern. Med.",
"key": "1535_CR11",
"unstructured": "Geng, L. N. et al. Nirmatrelvir-ritonavir and symptoms in adults with postacute sequelae of SARS-CoV-2 infection: the STOP-PASC randomized clinical trial: the STOP-PASC randomized clinical trial. JAMA Intern. Med. 184, 1024–1034 (2024).",
"volume": "184",
"year": "2024"
},
{
"DOI": "10.1002/jmv.28750",
"author": "MH Chuang",
"doi-asserted-by": "crossref",
"journal-title": "J. Med. Virol.",
"key": "1535_CR12",
"unstructured": "Chuang, M. H. et al. Efficacy of nirmatrelvir and ritonavir for post-acute COVID-19 sequelae beyond 3 months of SARS-CoV-2 infection. J. Med. Virol. 95, e28750 (2023).",
"volume": "95",
"year": "2023"
},
{
"DOI": "10.21203/rs.3.rs-4536807/v1",
"doi-asserted-by": "publisher",
"key": "1535_CR13",
"unstructured": "Wang, F. et al. Real-World Effectiveness of Nirmatrelvir in Protecting Long COVID for Outpatient Adult Patients-A Large-Scale Observational Cohort Study from the RECOVER Initiative. Res sq https://doi.org/10.21203/rs.3.rs-4536807/v1 (2024)."
},
{
"DOI": "10.1101/2025.04.02.25324861",
"doi-asserted-by": "publisher",
"key": "1535_CR14",
"unstructured": "Shen Y. et al. Assessment of Nirmatrelvir-Ritonavir Effects on Acute and Post-Acute COVID-19 Illness in US Adolescents: Target Trial Emulation. MedRXiv https://doi.org/10.1101/2025.04.02.25324861 (2025)."
},
{
"author": "FS Sharif-Askari",
"journal-title": "Sci. Rep.",
"key": "1535_CR15",
"unstructured": "Sharif-Askari, F. S. et al. Nirmatrelvir plus ritonavir reduces COVID-19 hospitalization and prevents long COVID in adult outpatients. Sci. Rep. 14, 25901 (2024).",
"volume": "14",
"year": "2024"
},
{
"DOI": "10.1186/s12889-024-19264-5",
"author": "R Sk Abd Razak",
"doi-asserted-by": "crossref",
"journal-title": "BMC Public Health",
"key": "1535_CR16",
"unstructured": "Sk Abd Razak, R. et al. Post-COVID syndrome prevalence: a systematic review and meta-analysis. BMC Public Health 24, 1785 (2024).",
"volume": "24",
"year": "2024"
},
{
"DOI": "10.1016/j.medj.2024.10.009",
"doi-asserted-by": "publisher",
"key": "1535_CR17",
"unstructured": "Azhir, A. et al. Precision phenotyping for curating research cohorts of patients with unexplained post-acute sequelae of COVID-19. Med https://doi.org/10.1016/j.medj.2024.10.009 (2024)."
},
{
"key": "1535_CR18",
"unstructured": "WHO. Coronavirus disease (COVID-19): Post COVID-19 condition. Accessed 2023. https://www.who.int/news-room/questions-and-answers/item/coronavirus-disease-(covid-19)-post-covid-19-condition."
},
{
"DOI": "10.1056/NEJMsb2408466",
"author": "EW Ely",
"doi-asserted-by": "crossref",
"first-page": "1746",
"journal-title": "N. Engl. J. Med.",
"key": "1535_CR19",
"unstructured": "Ely, E. W., Brown, L. M. & Fineberg, H. V. National academies of sciences, engineering, and medicine committee on examining the working definition for long covid. Long Covid defined. N. Engl. J. Med. 391, 1746–1753 (2024).",
"volume": "391",
"year": "2024"
},
{
"DOI": "10.1001/jamanetworkopen.2022.38354",
"author": "ZH Strasser",
"doi-asserted-by": "crossref",
"first-page": "e2238354",
"journal-title": "JAMA Netw. Open",
"key": "1535_CR20",
"unstructured": "Strasser, Z. H., Greifer, N., Hadavand, A., Murphy, S. N. & Estiri, H. Estimates of SARS-CoV-2 Omicron BA.2 subvariant severity in New England. JAMA Netw. Open 5, e2238354 (2022).",
"volume": "5",
"year": "2022"
},
{
"DOI": "10.1016/j.cmi.2020.11.028",
"author": "D Yahav",
"doi-asserted-by": "crossref",
"first-page": "315",
"journal-title": "Clin. Microbiol. Infect.",
"key": "1535_CR21",
"unstructured": "Yahav, D. et al. Definitions for coronavirus disease 2019 reinfection, relapse and PCR re-positivity. Clin. Microbiol. Infect. 27, 315–318 (2021).",
"volume": "27",
"year": "2021"
},
{
"DOI": "10.1016/S1473-3099(21)00703-9",
"author": "JB Soriano",
"doi-asserted-by": "crossref",
"first-page": "e102",
"journal-title": "Lancet Infect. Dis.",
"key": "1535_CR22",
"unstructured": "Soriano, J. B., Murthy, S., Marshall, J. C., Relan, P. & Diaz, J. V. WHO clinical case definition working group on post-COVID-19 condition. A clinical case definition of post-COVID-19 condition by a Delphi consensus. Lancet Infect. Dis. 22, e102–e107 (2022).",
"volume": "22",
"year": "2022"
},
{
"DOI": "10.21105/joss.00648",
"author": "A Gasparini",
"doi-asserted-by": "crossref",
"first-page": "648",
"journal-title": "J. Open Source Softw.",
"key": "1535_CR23",
"unstructured": "Gasparini, A. comorbidity: An R package for computing comorbidity scores. J. Open Source Softw. 3, 648 (2018).",
"volume": "3",
"year": "2018"
},
{
"DOI": "10.1093/pan/mpr025",
"author": "J Hainmueller",
"doi-asserted-by": "crossref",
"first-page": "25",
"journal-title": "Polit. Anal.",
"key": "1535_CR24",
"unstructured": "Hainmueller, J. Entropy balancing for causal effects: a multivariate reweighting method to produce balanced samples in observational studies. Polit. Anal. 20, 25–46 (2012).",
"volume": "20",
"year": "2012"
},
{
"author": "E Xu",
"journal-title": "Nat. Commun.",
"key": "1535_CR25",
"unstructured": "Xu, E., Xie, Y. & Al-Aly, Z. Long-term gastrointestinal outcomes of COVID-19. Nat. Commun. 14, 983 (2023).",
"volume": "14",
"year": "2023"
},
{
"DOI": "10.1016/S1473-3099(24)00171-3",
"author": "W Zuo",
"doi-asserted-by": "crossref",
"first-page": "845",
"journal-title": "Lancet Infect. Dis.",
"key": "1535_CR26",
"unstructured": "Zuo, W. et al. The persistence of SARS-CoV-2 in tissues and its association with long COVID symptoms: a cross-sectional cohort study in China. Lancet Infect. Dis. 24, 845–855 (2024).",
"volume": "24",
"year": "2024"
},
{
"DOI": "10.1038/s41590-017-0006-x",
"author": "J Nikolich-Žugich",
"doi-asserted-by": "crossref",
"first-page": "10",
"journal-title": "Nat. Immunol.",
"key": "1535_CR27",
"unstructured": "Nikolich-Žugich, J. The twilight of immunity: emerging concepts in aging of the immune system. Nat. Immunol. 19, 10–19 (2018).",
"volume": "19",
"year": "2018"
},
{
"DOI": "10.1038/s41392-023-01451-2",
"author": "Z Liu",
"doi-asserted-by": "crossref",
"first-page": "200",
"journal-title": "Sig. Transduct. Target. Ther.",
"key": "1535_CR28",
"unstructured": "Liu, Z. et al. Immunosenescence: molecular mechanisms and diseases. Sig. Transduct. Target. Ther. 8, 200 (2023).",
"volume": "8",
"year": "2023"
},
{
"DOI": "10.1016/j.arr.2021.101422",
"author": "A Santoro",
"doi-asserted-by": "crossref",
"journal-title": "Ageing Res. Rev.",
"key": "1535_CR29",
"unstructured": "Santoro, A., Bientinesi, E. & Monti, D. Immunosenescence and inflammaging in the aging process: age-related diseases or longevity? Ageing Res. Rev. 71, 101422 (2021).",
"volume": "71",
"year": "2021"
},
{
"DOI": "10.1016/j.cmi.2024.07.023",
"author": "G Sun",
"doi-asserted-by": "crossref",
"first-page": "1618",
"journal-title": "Clin. Microbiol. Infect.",
"key": "1535_CR30",
"unstructured": "Sun, G., Lin, K., Ai, J. & Zhang, W. The efficacy of antivirals, corticosteroids, and mAbs as acute COVID treatments in reducing the incidence of long COVID’-Author’s reply. Clin. Microbiol. Infect. 30, 1618–1619 (2024).",
"volume": "30",
"year": "2024"
},
{
"DOI": "10.1038/s41598-025-06052-3",
"author": "M Berry",
"doi-asserted-by": "crossref",
"journal-title": "Sci. Rep.",
"key": "1535_CR31",
"unstructured": "Berry, M. et al. Risk of long COVID in hospitalized individuals treated with remdesivir for acute COVID-19. Sci. Rep. 15, 27441 (2025).",
"volume": "15",
"year": "2025"
},
{
"DOI": "10.1093/jleuko/qiac001",
"author": "MS Islam",
"doi-asserted-by": "crossref",
"first-page": "236",
"journal-title": "J. Leukoc. Biol.",
"key": "1535_CR32",
"unstructured": "Islam, M. S., Wang, Z., Abdel-Mohsen, M., Chen, X. & Montaner, L. J. Tissue injury and leukocyte changes in post-acute sequelae of SARS-CoV-2: review of 2833 post-acute patient outcomes per immune dysregulation and microbial translocation in long COVID. J. Leukoc. Biol. 113, 236–254 (2023).",
"volume": "113",
"year": "2023"
},
{
"DOI": "10.1016/S0140-6736(24)01136-X",
"author": "T Greenhalgh",
"doi-asserted-by": "crossref",
"first-page": "707",
"journal-title": "Lancet",
"key": "1535_CR33",
"unstructured": "Greenhalgh, T., Sivan, M., Perlowski, A. & Nikolich, J. Ž Long COVID: a clinical update. Lancet 404, 707–724 (2024).",
"volume": "404",
"year": "2024"
},
{
"DOI": "10.7554/eLife.86002",
"author": "ZA Sherif",
"doi-asserted-by": "crossref",
"journal-title": "Elife",
"key": "1535_CR34",
"unstructured": "Sherif, Z. A., Gomez, C. R., Connors, T. J., Henrich, T. J. & Reeves, W. B. Pathogenic mechanisms of post-acute sequelae of SARS-CoV-2 infection (PASC). Elife 12, e86002 (2023).",
"volume": "12",
"year": "2023"
},
{
"author": "A Mantovani",
"first-page": "1891",
"journal-title": "Cell Death Differ.",
"key": "1535_CR35",
"unstructured": "Mantovani, A. et al. Covid-19 Commission of the Accademia Nazionale dei Lincei Cappuccinelli Pietro 13 Fitzgerald Garrett 14 Bacci Massimo Livi 15 Melino Gennaro 16 Parisi Giorgio 17 Rappuoli Rino 18 19 Rezza Giovanni 20 Vineis Paolo 21. Long Covid: where we stand and challenges ahead. Cell Death Differ. 29, 1891–1900 (2022).",
"volume": "29",
"year": "2022"
},
{
"DOI": "10.1371/journal.pone.0275274",
"author": "JA Frontera",
"doi-asserted-by": "crossref",
"first-page": "e0275274",
"journal-title": "Plos ONE",
"key": "1535_CR36",
"unstructured": "Frontera, J. A. et al. Post-acute sequelae of COVID-19 symptom phenotypes and therapeutic strategies: a prospective, observational study. Plos ONE 17, e0275274 (2022).",
"volume": "17",
"year": "2022"
},
{
"DOI": "10.1016/S1473-3099(25)00073-8",
"doi-asserted-by": "crossref",
"key": "1535_CR37",
"unstructured": "Sawano, M. et al. Nirmatrelvir–ritonavir versus placebo–ritonavir in individuals with long COVID in the USA (PAX LC): a double-blind, randomised, placebo-controlled, phase 2, decentralised trial. Lancet Infect. Dis. 25, 936–946 (2025)."
},
{
"key": "1535_CR38",
"unstructured": "Greifer, N. Weighting for covariate balance in observational studies [R package WeightIt version 0.13.1]. Published online 2022. Accessed July 7, 2022. https://CRAN.R-project.org/package=WeightIt."
},
{
"key": "1535_CR39",
"unstructured": "Greifer, N. Covariate balance tables and plots: a guide to the cobalt package. 2024. Accessed May 31, 2024. https://cloud.r-project.org/web/packages/cobalt/vignettes/cobalt.html."
},
{
"key": "1535_CR40",
"unstructured": "Arel-Bundock, V., Greifer, N. & Bacher, E. Marginaleffects: Predictions, Comparisons, Slopes, Marginal Means, and Hypothesis Tests.; 2024. Accessed May 31, 2024. https://CRAN.R-project.org/package=marginaleffects."
},
{
"key": "1535_CR41",
"unstructured": "Survey: analysis of complex survey samples. Comprehensive R Archive Network (CRAN). Accessed 2022. https://cran.r-project.org/web/packages/survey/index.html."
},
{
"DOI": "10.5281/zenodo.18749575",
"author": "A Azhir",
"doi-asserted-by": "publisher",
"journal-title": "Version 1.0.1. Zenodo",
"key": "1535_CR42",
"unstructured": "Azhir, A., Cheng, J. & Estiri, H. Paxlovid shows organ-specific and age-specific impacts on risk of developing post-acute sequelae of COVID-19. Version 1.0.1. Zenodo https://doi.org/10.5281/zenodo.18749575 (2026).",
"year": "2026"
}
],
"reference-count": 42,
"references-count": 42,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.nature.com/articles/s43856-026-01535-4"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "Paxlovid shows organ-specific and age-specific impacts on risk of developing post-acute sequelae of COVID-19",
"type": "journal-article",
"update-policy": "https://doi.org/10.1007/springer_crossmark_policy"
}