Cardiovascular Risks of COVID-19 Therapeutics: Integrated Analysis of FAERS, Electronic Health Records, and Transcriptomics
et al., Pharmaceuticals, doi:10.3390/ph19040574, Apr 2026
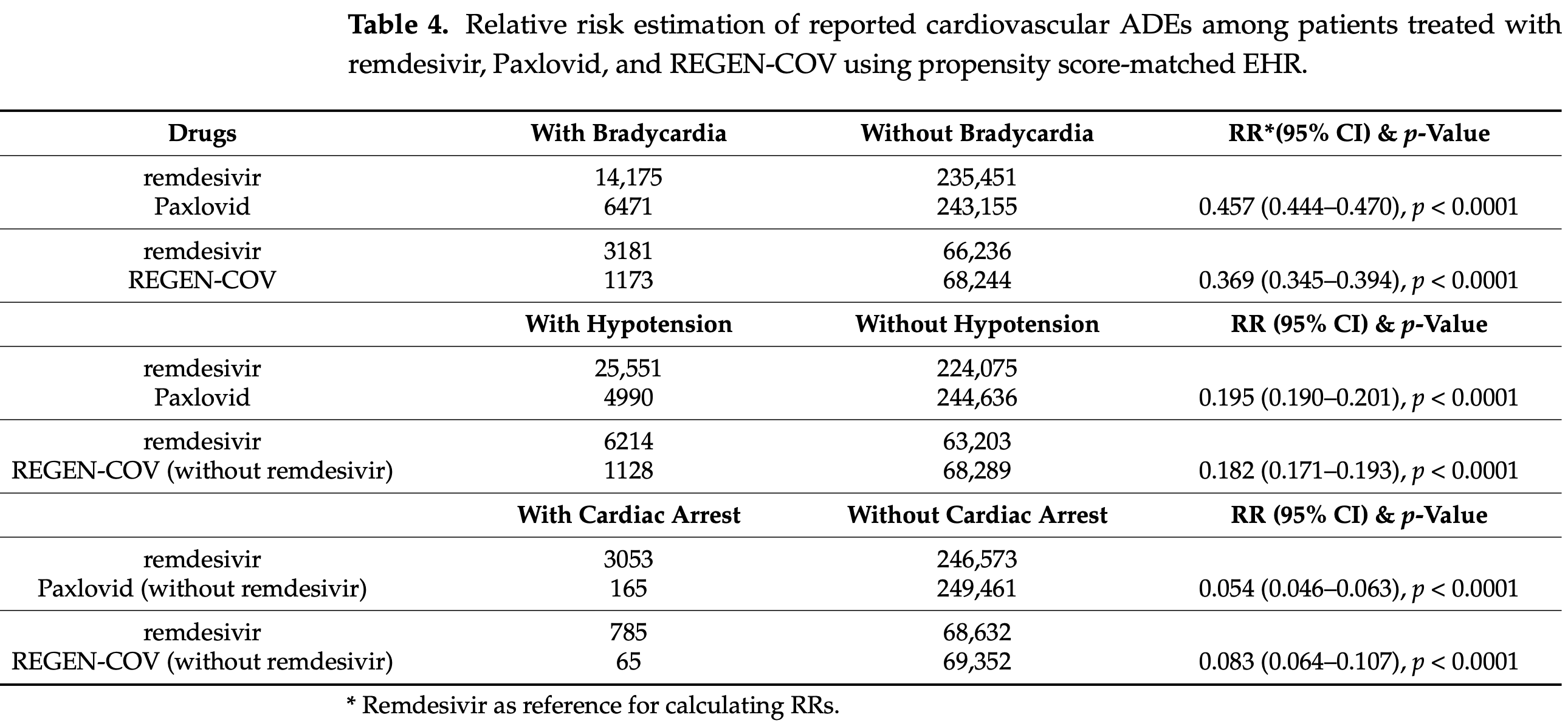
Pharmacovigilance and EHR analysis of 8,143 FAERS remdesivir reports and over 255,000 TriNetX EHR records, showing significantly higher risk of cardiovascular adverse events (bradycardia, hypotension, and cardiac arrest) with remdesivir compared to paxlovid and REGEN-COV in COVID-19 patients.
After propensity score matching, remdesivir-treated patients had approximately 2x higher risk of bradycardia, approximately 5x higher risk of hypotension, and approximately 19x higher risk of cardiac arrest compared to paxlovid-treated patients.
Transcriptomic analysis of remdesivir-treated human embryonic stem cell-derived cardiomyocytes identified enrichment of cGMP-PKG signaling, dilated cardiomyopathy, and calcium signaling pathways among upregulated genes, which the authors suggest may provide biological context for observed cardiovascular signals.
Risk may be increased in part due to confounding - the propensity score matching did not match patients for COVID-19 severity.
Gérard, Zhou, Wu, Kamo, Choi, Kim show increased risk of acute kidney injury, Leo, Briciu, Muntean, Petrov, Arch show increased risk of liver injury, Negru, Cheng, Mohammed, Kwok, Zhu show increased risk of cardiac disorders, and Kwok, Merches, Akinci, Tang, Bagheri show increased risk of mitochondrial toxicity with remdesivir.
1.
Gérard et al., Remdesivir and Acute Renal Failure: A Potential Safety Signal From Disproportionality Analysis of the WHO Safety Database, Clinical Pharmacology & Therapeutics, doi:10.1002/cpt.2145.
2.
Zhou et al., Acute Kidney Injury and Drugs Prescribed for COVID-19 in Diabetes Patients: A Real-World Disproportionality Analysis, Frontiers in Pharmacology, doi:10.3389/fphar.2022.833679.
3.
Wu et al., Acute Kidney Injury Associated With Remdesivir: A Comprehensive Pharmacovigilance Analysis of COVID-19 Reports in FAERS, Frontiers in Pharmacology, doi:10.3389/fphar.2022.692828.
4.
Kamo et al., Association of Antiviral Drugs for the Treatment of COVID-19 With Acute Renal Failure, In Vivo, doi:10.21873/invivo.13637.
5.
Choi et al., Comparative effectiveness of combination therapy with nirmatrelvir–ritonavir and remdesivir versus monotherapy with remdesivir or nirmatrelvir–ritonavir in patients hospitalised with COVID-19: a target trial emulation study, The Lancet Infectious Diseases, doi:10.1016/S1473-3099(24)00353-0.
6.
Kim et al., Investigating the Safety Profile of Fast‐Track COVID‐19 Drugs Using the FDA Adverse Event Reporting System Database: A Comparative Observational Study, Pharmacoepidemiology and Drug Safety, doi:10.1002/pds.70043.
7.
Leo et al., Hepatocellular liver injury in hospitalized patients affected by COVID-19: Presence of different risk factors at different time points, Digestive and Liver Disease, doi:10.1016/j.dld.2021.12.014.
8.
Briciu et al., Evolving Clinical Manifestations and Outcomes in COVID-19 Patients: A Comparative Analysis of SARS-CoV-2 Variant Waves in a Romanian Hospital Setting, Pathogens, doi:10.3390/pathogens12121453.
9.
Muntean et al., Effects of COVID-19 on the Liver and Mortality in Patients with SARS-CoV-2 Pneumonia Caused by Delta and Non-Delta Variants: An Analysis in a Single Centre, Pharmaceuticals, doi:10.3390/ph17010003.
10.
Petrov et al., The Effect of Potentially Hepatotoxic Medicinal Products on Alanine Transaminase Levels in COVID-19 Patients: A Case–Control Study, Safety and Risk of Pharmacotherapy, doi:10.30895/2312-7821-2025-458.
11.
Arch et al., Evaluation of the effectiveness of remdesivir in treating severe COVID-19 using data from the ISARIC WHO Clinical Characterisation Protocol UK: a prospective, national cohort study, medRxiv, doi:10.1101/2021.06.18.21259072.
12.
Negru et al., Comparative Pharmacovigilance Analysis of Approved and Repurposed Antivirals for COVID-19: Insights from EudraVigilance Data, Biomedicines, doi:10.3390/biomedicines13061387.
13.
Cheng et al., Cardiovascular Safety of COVID-19 Treatments: A Disproportionality Analysis of Adverse Event Reports from the WHO VigiBase, Infectious Diseases and Therapy, doi:10.1007/s40121-025-01225-z.
14.
Mohammed et al., Bradycardia associated with remdesivir treatment in coronavirus disease 2019 patients: A propensity score-matched analysis, Medicine, doi:10.1097/MD.0000000000044501.
15.
Kwok et al., Remdesivir induces persistent mitochondrial and structural damage in human induced pluripotent stem cell-derived cardiomyocytes, Cardiovascular Research, doi:10.1093/cvr/cvab311.
16.
Zhu et al., Cardiovascular Risks of COVID-19 Therapeutics: Integrated Analysis of FAERS, Electronic Health Records, and Transcriptomics, Pharmaceuticals, doi:10.3390/ph19040574.
17.
Merches et al., The potential of remdesivir to affect function, metabolism and proliferation of cardiac and kidney cells in vitro, Archives of Toxicology, doi:10.1007/s00204-022-03306-1.
18.
Akinci et al., Elucidation of remdesivir cytotoxicity pathways through genome-wide CRISPR-Cas9 screening and transcriptomics, bioRxiv, doi:10.1101/2020.08.27.270819.
Zhu et al., 2 Apr 2026, prospective, peer-reviewed, 9 authors.
Contact: fcheng1@usf.edu (corresponding author), xinranzhu@usf.edu, skuppa@usf.edu, gibreti@gmail.com, rpmj528@gmail.com, buil@usf.edu, kunbu@usf.edu, jzhang7@bu.edu, jwei7@bu.edu.
Abstract:
Article
Cardiovascular Risks of COVID-19 Therapeutics: Integrated Analysis of FAERS, Electronic Health Records, and Transcriptomics
Xinran Zhu 1 , Suguna Aishwarya Kuppa 1 , Gibret Umeukeje 1 , Robert Morris 1 , Lan Bui 2 , Kun Bu 2 , Jie Zhang 3 , Jin Wei 3 and Feng Cheng 1, *
- 1 Department of Pharmaceutical Sciences, College of Pharmacy, University of South Florida, Tampa, FL 33612, USA; xinranzhu@usf.edu (X.Z.); skuppa@usf.edu (S.A.K.); gibreti@gmail.com (G.U.); rpmj528@gmail.com (R.M.)
- 2 Department of Mathematics & Statistics, College of Art and Science, University of South Florida, Tampa, FL 33620, USA; buil@usf.edu (L.B.); kunbu@usf.edu (K.B.)
- 3 Section of Nephrology, Department of Medicine, Boston University Chobanian & Avedisian School of Medicine, Boston, MA 02118, USA; jzhang7@bu.edu (J.Z.); jwei7@bu.edu (J.W.)
* Correspondence: fcheng1@usf.edu
Abstract
Background/Objectives : The purpose of this study was to investigate the association between cardiovascular adverse drug events (ADEs) and the use of COVID-19 medicines. Methods : The analyses were conducted by leveraging pharmacovigilance data from the Food and Drug Authority (FDA) Adverse Event Reporting System (FAERS) and TriNetX electronic health records (EHRs). Transcriptomic data from human embryonic stem cellderived cardiomyocytes (hESC-CMs) exposed to remdesivir were analyzed to provide supportive biological context for the observed cardiovascular safety signals. Results : Comparative analysis of three approved COVID-19 therapies revealed that COVID-19 patients treated with remdesivir had a higher risk of cardiovascular events than those treated with Paxlovid or REGEN-COV. FAERS analysis further indicated that bradycardia, hypotension, and cardiac arrest were the most frequently reported cardiovascular events associated with remdesivir, which was validated by propensity score-matched EHR data. These findings suggest an association between remdesivir exposure and increased cardiovascular ADEs relative to other COVID-19 therapies. Sex-stratified analysis using FAERS and EHR did not show strong sex-dependent patterns for remdesivir-associated cardiovascular ADEs. Age-stratified analyses of EHR data showed age-associated variation across the three cardiovascular ADEs. Bradycardia displayed a non-uniform pattern with higher prevalence in the youngest and oldest age groups, hypotension showed an overall age-associated increase, and cardiac arrest showed only a weak age-associated effect. Pathway enrichment analysis on transcriptomic data revealed that the 'cGMP-PKG signaling pathway', 'dilated cardiomyopathy', and 'calcium signaling pathway' were enriched among genes up-regulated by remdesivir exposure. Conclusions : In summary, our integrated analysis of pharmacovigilance, EHR, and transcriptomic data provides convergent evidence for associations between remdesivir and cardiovascular ADEs and offers biological context into these associations.
Keywords: FAERS; pharmacovigilance; TriNetX; EHR; COVID-19 drugs; remdesivir; Paxlovid; REGEN-COV; bradycardia; ADE
Academic Editor: Eleni Gavriilaki
Received: 19 February 2026
Revised: 27 March 2026
Accepted: 29 March 2026
Published: 2 April 2026
Copyright:
©2026 by the authors.
Licensee MDPI, Basel, Switzerland.
This article is an open access article
distributed under the terms and
conditions of the Creative Commons
Attribution (CC BY) license.
DOI record:
{
"DOI": "10.3390/ph19040574",
"ISSN": [
"1424-8247"
],
"URL": "http://dx.doi.org/10.3390/ph19040574",
"abstract": "<jats:p>Background/Objectives: The purpose of this study was to investigate the association between cardiovascular adverse drug events (ADEs) and the use of COVID-19 medicines. Methods: The analyses were conducted by leveraging pharmacovigilance data from the Food and Drug Authority (FDA) Adverse Event Reporting System (FAERS) and TriNetX electronic health records (EHRs). Transcriptomic data from human embryonic stem cell-derived cardiomyocytes (hESC-CMs) exposed to remdesivir were analyzed to provide supportive biological context for the observed cardiovascular safety signals. Results: Comparative analysis of three approved COVID-19 therapies revealed that COVID-19 patients treated with remdesivir had a higher risk of cardiovascular events than those treated with Paxlovid or REGEN-COV. FAERS analysis further indicated that bradycardia, hypotension, and cardiac arrest were the most frequently reported cardiovascular events associated with remdesivir, which was validated by propensity score-matched EHR data. These findings suggest an association between remdesivir exposure and increased cardiovascular ADEs relative to other COVID-19 therapies. Sex-stratified analysis using FAERS and EHR did not show strong sex-dependent patterns for remdesivir-associated cardiovascular ADEs. Age-stratified analyses of EHR data showed age-associated variation across the three cardiovascular ADEs. Bradycardia displayed a non-uniform pattern with higher prevalence in the youngest and oldest age groups, hypotension showed an overall age-associated increase, and cardiac arrest showed only a weak age-associated effect. Pathway enrichment analysis on transcriptomic data revealed that the “cGMP-PKG signaling pathway”, “dilated cardiomyopathy”, and “calcium signaling pathway” were enriched among genes up-regulated by remdesivir exposure. Conclusions: In summary, our integrated analysis of pharmacovigilance, EHR, and transcriptomic data provides convergent evidence for associations between remdesivir and cardiovascular ADEs and offers biological context into these associations.</jats:p>",
"alternative-id": [
"ph19040574"
],
"author": [
{
"affiliation": [
{
"name": "Department of Pharmaceutical Sciences, College of Pharmacy, University of South Florida, Tampa, FL 33612, USA"
}
],
"family": "Zhu",
"given": "Xinran",
"sequence": "first"
},
{
"ORCID": "https://orcid.org/0009-0007-3438-9340",
"affiliation": [
{
"name": "Department of Pharmaceutical Sciences, College of Pharmacy, University of South Florida, Tampa, FL 33612, USA"
}
],
"authenticated-orcid": false,
"family": "Kuppa",
"given": "Suguna Aishwarya",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0002-6708-884X",
"affiliation": [
{
"name": "Department of Pharmaceutical Sciences, College of Pharmacy, University of South Florida, Tampa, FL 33612, USA"
}
],
"authenticated-orcid": false,
"family": "Umeukeje",
"given": "Gibret",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Pharmaceutical Sciences, College of Pharmacy, University of South Florida, Tampa, FL 33612, USA"
}
],
"family": "Morris",
"given": "Robert",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Mathematics & Statistics, College of Art and Science, University of South Florida, Tampa, FL 33620, USA"
}
],
"family": "Bui",
"given": "Lan",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0009-0006-9526-3023",
"affiliation": [
{
"name": "Department of Mathematics & Statistics, College of Art and Science, University of South Florida, Tampa, FL 33620, USA"
}
],
"authenticated-orcid": false,
"family": "Bu",
"given": "Kun",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Section of Nephrology, Department of Medicine, Boston University Chobanian & Avedisian School of Medicine, Boston, MA 02118, USA"
}
],
"family": "Zhang",
"given": "Jie",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Section of Nephrology, Department of Medicine, Boston University Chobanian & Avedisian School of Medicine, Boston, MA 02118, USA"
}
],
"family": "Wei",
"given": "Jin",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0003-3619-8090",
"affiliation": [
{
"name": "Department of Pharmaceutical Sciences, College of Pharmacy, University of South Florida, Tampa, FL 33612, USA"
}
],
"authenticated-orcid": false,
"family": "Cheng",
"given": "Feng",
"sequence": "additional"
}
],
"container-title": "Pharmaceuticals",
"container-title-short": "Pharmaceuticals",
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2026,
4,
2
]
],
"date-time": "2026-04-02T14:09:54Z",
"timestamp": 1775138994000
},
"deposited": {
"date-parts": [
[
2026,
4,
2
]
],
"date-time": "2026-04-02T14:15:32Z",
"timestamp": 1775139332000
},
"indexed": {
"date-parts": [
[
2026,
4,
2
]
],
"date-time": "2026-04-02T14:59:45Z",
"timestamp": 1775141985565,
"version": "3.50.1"
},
"is-referenced-by-count": 0,
"issue": "4",
"issued": {
"date-parts": [
[
2026,
4,
2
]
]
},
"journal-issue": {
"issue": "4",
"published-online": {
"date-parts": [
[
2026,
4
]
]
}
},
"language": "en",
"license": [
{
"URL": "https://creativecommons.org/licenses/by/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
4,
2
]
],
"date-time": "2026-04-02T00:00:00Z",
"timestamp": 1775088000000
}
}
],
"link": [
{
"URL": "https://www.mdpi.com/1424-8247/19/4/574/pdf",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "1968",
"original-title": [],
"page": "574",
"prefix": "10.3390",
"published": {
"date-parts": [
[
2026,
4,
2
]
]
},
"published-online": {
"date-parts": [
[
2026,
4,
2
]
]
},
"publisher": "MDPI AG",
"reference": [
{
"DOI": "10.1186/s12879-021-06536-3",
"doi-asserted-by": "crossref",
"key": "ref_1",
"unstructured": "Dessie, Z.G., and Zewotir, T. (2021). Mortality-related risk factors of COVID-19: A systematic review and meta-analysis of 42 studies and 423,117 patients. BMC Infect. Dis., 21."
},
{
"DOI": "10.1016/j.jacc.2020.08.059",
"article-title": "Coronavirus and Cardiovascular Disease, Myocardial Injury, and Arrhythmia: JACC Focus Seminar",
"author": "Giustino",
"doi-asserted-by": "crossref",
"first-page": "2011",
"journal-title": "J. Am. Coll. Cardiol.",
"key": "ref_2",
"volume": "76",
"year": "2020"
},
{
"DOI": "10.1128/CMR.00162-20",
"article-title": "Remdesivir against COVID-19 and Other Viral Diseases",
"author": "Malin",
"doi-asserted-by": "crossref",
"first-page": "e00162-20",
"journal-title": "Clin. Microbiol. Rev.",
"key": "ref_3",
"volume": "34",
"year": "2020"
},
{
"DOI": "10.1007/s40265-020-01378-w",
"article-title": "Remdesivir: First Approval",
"author": "Lamb",
"doi-asserted-by": "crossref",
"first-page": "1355",
"journal-title": "Drugs",
"key": "ref_4",
"volume": "80",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa2116846",
"article-title": "Early Remdesivir to Prevent Progression to Severe COVID-19 in Outpatients",
"author": "Gottlieb",
"doi-asserted-by": "crossref",
"first-page": "305",
"journal-title": "N. Engl. J. Med.",
"key": "ref_5",
"volume": "386",
"year": "2022"
},
{
"DOI": "10.1002/jmv.28037",
"article-title": "Combination therapy with remdesivir and immunomodulators improves respiratory status in COVID-19: A retrospective study",
"author": "Kojima",
"doi-asserted-by": "crossref",
"first-page": "5702",
"journal-title": "J. Med. Virol.",
"key": "ref_6",
"volume": "94",
"year": "2022"
},
{
"DOI": "10.1093/bmb/ldac037",
"article-title": "Drug repurposing for COVID-19: Current evidence from randomized controlled adaptive platform trials and living systematic reviews",
"author": "Augustin",
"doi-asserted-by": "crossref",
"first-page": "31",
"journal-title": "Br. Med. Bull.",
"key": "ref_7",
"volume": "147",
"year": "2023"
},
{
"DOI": "10.7861/clinmed.2022-0080",
"article-title": "A practical update on the management of patients with COVID-19",
"author": "Mehta",
"doi-asserted-by": "crossref",
"first-page": "468",
"journal-title": "Clin. Med.",
"key": "ref_8",
"volume": "22",
"year": "2022"
},
{
"DOI": "10.1021/acs.jmedchem.6b01594",
"article-title": "Discovery and Synthesis of a Phosphoramidate Prodrug of a Pyrrolo [2,1-f][triazin-4-amino] Adenine C-Nucleoside (GS-5734) for the Treatment of Ebola and Emerging Viruses",
"author": "Siegel",
"doi-asserted-by": "crossref",
"first-page": "1648",
"journal-title": "J. Med. Chem.",
"key": "ref_9",
"volume": "60",
"year": "2017"
},
{
"DOI": "10.1074/jbc.AC120.015720",
"article-title": "Template-dependent inhibition of coronavirus RNA-dependent RNA polymerase by remdesivir reveals a second mechanism of action",
"author": "Tchesnokov",
"doi-asserted-by": "crossref",
"first-page": "16156",
"journal-title": "J. Biol. Chem.",
"key": "ref_10",
"volume": "295",
"year": "2020"
},
{
"DOI": "10.1038/d41573-022-00047-9",
"article-title": "Top companies and drugs by sales in 2021",
"author": "Urquhart",
"doi-asserted-by": "crossref",
"first-page": "251",
"journal-title": "Nat. Rev. Drug Discov.",
"key": "ref_11",
"volume": "21",
"year": "2022"
},
{
"DOI": "10.1002/cpt.3337",
"article-title": "Pharmacokinetics, Mass Balance, Safety, and Tolerability of Obeldesivir in Healthy Participants",
"author": "Anoshchenko",
"doi-asserted-by": "crossref",
"first-page": "1231",
"journal-title": "Clin. Pharmacol. Ther.",
"key": "ref_12",
"volume": "116",
"year": "2024"
},
{
"DOI": "10.1016/S1473-3099(25)00238-5",
"article-title": "Efficacy and safety of obeldesivir in low-risk, non-hospitalised patients with COVID-19 (OAKTREE): A phase 3, randomised, double-blind, placebo-controlled study",
"author": "Ogbuagu",
"doi-asserted-by": "crossref",
"first-page": "1282",
"journal-title": "Lancet Infect. Dis.",
"key": "ref_13",
"volume": "25",
"year": "2025"
},
{
"DOI": "10.1128/AAC.01155-21",
"article-title": "Rethinking Remdesivir: Synthesis, Antiviral Activity, and Pharmacokinetics of Oral Lipid Prodrugs",
"author": "Schooley",
"doi-asserted-by": "crossref",
"first-page": "e0115521",
"journal-title": "Antimicrob. Agents Chemother.",
"key": "ref_14",
"volume": "65",
"year": "2021"
},
{
"DOI": "10.1056/NEJMoa2118542",
"article-title": "Oral Nirmatrelvir for High-Risk, Nonhospitalized Adults with COVID-19",
"author": "Hammond",
"doi-asserted-by": "crossref",
"first-page": "1397",
"journal-title": "N. Engl. J. Med.",
"key": "ref_15",
"volume": "386",
"year": "2022"
},
{
"DOI": "10.1126/science.abl4784",
"article-title": "An oral SARS-CoV-2 M(pro) inhibitor clinical candidate for the treatment of COVID-19",
"author": "Owen",
"doi-asserted-by": "crossref",
"first-page": "1586",
"journal-title": "Science",
"key": "ref_16",
"volume": "374",
"year": "2021"
},
{
"DOI": "10.1007/s40121-024-01023-z",
"article-title": "Proportion of Patients in the United States Who Fill Their Nirmatrelvir/Ritonavir Prescriptions",
"author": "Rudolph",
"doi-asserted-by": "crossref",
"first-page": "2035",
"journal-title": "Infect. Dis. Ther.",
"key": "ref_17",
"volume": "13",
"year": "2024"
},
{
"DOI": "10.1056/NEJMoa2108163",
"article-title": "REGEN-COV Antibody Combination and Outcomes in Outpatients with Covid-19",
"author": "Weinreich",
"doi-asserted-by": "crossref",
"first-page": "e81",
"journal-title": "N. Engl. J. Med.",
"key": "ref_18",
"volume": "385",
"year": "2021"
},
{
"DOI": "10.1007/s12012-021-09703-9",
"article-title": "Potential Cardiotoxic Effects of Remdesivir on Cardiovascular System: A Literature Review",
"author": "Nabati",
"doi-asserted-by": "crossref",
"first-page": "268",
"journal-title": "Cardiovasc. Toxicol.",
"key": "ref_19",
"volume": "22",
"year": "2022"
},
{
"DOI": "10.1056/NEJMoa2007016",
"article-title": "Compassionate Use of Remdesivir for Patients with Severe COVID-19",
"author": "Grein",
"doi-asserted-by": "crossref",
"first-page": "2327",
"journal-title": "N. Engl. J. Med.",
"key": "ref_20",
"volume": "382",
"year": "2020"
},
{
"article-title": "Cardiac Adverse Events With Remdesivir in COVID-19 Infection",
"author": "Gupta",
"first-page": "e11132",
"journal-title": "Cureus",
"key": "ref_21",
"volume": "12",
"year": "2020"
},
{
"DOI": "10.1161/CIRCEP.121.009811",
"article-title": "Remdesivir-Induced Bradycardia in COVID-19: A Single Center Prospective Study",
"author": "Attena",
"doi-asserted-by": "crossref",
"first-page": "e009811",
"journal-title": "Circ. Arrhythm. Electrophysiol.",
"key": "ref_22",
"volume": "14",
"year": "2021"
},
{
"DOI": "10.3390/ph14070611",
"doi-asserted-by": "crossref",
"key": "ref_23",
"unstructured": "Rafaniello, C., Ferrajolo, C., Sullo, M.G., Gaio, M., Zinzi, A., Scavone, C., Gargano, F., Coscioni, E., Rossi, F., and Capuano, A. (2021). Cardiac Events Potentially Associated to Remdesivir: An Analysis from the European Spontaneous Adverse Event Reporting System. Pharmaceuticals, 14."
},
{
"DOI": "10.1016/j.cmi.2021.02.013",
"article-title": "Serious bradycardia and remdesivir for coronavirus 2019 (COVID-19): A new safety concerns",
"author": "Touafchia",
"doi-asserted-by": "crossref",
"first-page": "791.e5",
"journal-title": "Clin. Microbiol. Infect.",
"key": "ref_24",
"volume": "27",
"year": "2021"
},
{
"DOI": "10.1002/jmv.28441",
"article-title": "Efficacy and safety of nirmatrelvir/ritonavir (Paxlovid) for COVID-19: A rapid review and meta-analysis",
"author": "Amani",
"doi-asserted-by": "crossref",
"first-page": "e28441",
"journal-title": "J. Med. Virol.",
"key": "ref_25",
"volume": "95",
"year": "2023"
},
{
"DOI": "10.3346/jkms.2023.38.e347",
"article-title": "Effectiveness and Adverse Events of Nirmatrelvir/Ritonavir Versus Molnupiravir for COVID-19 in Outpatient Setting: Multicenter Prospective Observational Study",
"author": "Park",
"doi-asserted-by": "crossref",
"first-page": "e347",
"journal-title": "J. Korean Med. Sci.",
"key": "ref_26",
"volume": "38",
"year": "2023"
},
{
"DOI": "10.2174/0113816128280987240214103432",
"article-title": "Safety Profile of Paxlovid in the Treatment of COVID-19",
"author": "Lv",
"doi-asserted-by": "crossref",
"first-page": "666",
"journal-title": "Curr. Pharm. Des.",
"key": "ref_27",
"volume": "30",
"year": "2024"
},
{
"DOI": "10.1002/ohn.278",
"article-title": "Postmarketing Reporting of Paxlovid-Related Dysgeusia: A Real-World Pharmacovigilance Study",
"author": "Cvancara",
"doi-asserted-by": "crossref",
"first-page": "55",
"journal-title": "Otolaryngol. Head Neck Surg.",
"key": "ref_28",
"volume": "169",
"year": "2023"
},
{
"DOI": "10.1080/14740338.2025.2471509",
"article-title": "Effects of nirmatrelvir/ritonavir (Paxlovid) on the nervous system: Analysis on adverse events released by FDA",
"author": "Gao",
"doi-asserted-by": "crossref",
"first-page": "669",
"journal-title": "Expert Opin. Drug Saf.",
"key": "ref_29",
"volume": "25",
"year": "2026"
},
{
"DOI": "10.1053/j.ajkd.2025.02.603",
"article-title": "Safety and Effectiveness of Nirmatrelvir-Ritonavir in Patients With Advanced Kidney Dysfunction and COVID-19",
"author": "Anand",
"doi-asserted-by": "crossref",
"first-page": "202",
"journal-title": "Am. J. Kidney Dis.",
"key": "ref_30",
"volume": "86",
"year": "2025"
},
{
"DOI": "10.15420/ecr.2024.04",
"article-title": "Cardiovascular Drug Interactions with Nirmatrelvir/Ritonavir for COVID-19: Considerations for Daily Practice",
"author": "Ferri",
"doi-asserted-by": "crossref",
"first-page": "e15",
"journal-title": "Eur. Cardiol.",
"key": "ref_31",
"volume": "19",
"year": "2024"
},
{
"article-title": "Paxlovid-Induced Symptomatic Bradycardia and Syncope",
"author": "Ganipisetti",
"first-page": "e33831",
"journal-title": "Cureus",
"key": "ref_32",
"volume": "15",
"year": "2023"
},
{
"DOI": "10.3390/ph15121472",
"doi-asserted-by": "crossref",
"key": "ref_33",
"unstructured": "Zou, J., and Jing, F. (2022). Cardiovascular Adverse Events Associated with Monoclonal Antibody Products in Patients with COVID-19. Pharmaceuticals, 15."
},
{
"article-title": "Acute Hypersensitivity Reaction After Casirivimab/Imdevimab Infusion in a COVID-19-Positive Young Male: Myopericarditis or Kounis Syndrome?",
"author": "Rivera",
"first-page": "e31125",
"journal-title": "Cureus",
"key": "ref_34",
"volume": "14",
"year": "2022"
},
{
"DOI": "10.1177/0018578718795271",
"article-title": "The Newly Available FAERS Public Dashboard: Implications for Health Care Professionals",
"author": "Kumar",
"doi-asserted-by": "crossref",
"first-page": "75",
"journal-title": "Hosp. Pharm.",
"key": "ref_35",
"volume": "54",
"year": "2019"
},
{
"DOI": "10.1002/advs.202203388",
"article-title": "Investigating and Resolving Cardiotoxicity Induced by COVID-19 Treatments using Human Pluripotent Stem Cell-Derived Cardiomyocytes and Engineered Heart Tissues",
"author": "Xu",
"doi-asserted-by": "crossref",
"first-page": "e2203388",
"journal-title": "Adv. Sci.",
"key": "ref_36",
"volume": "9",
"year": "2022"
},
{
"DOI": "10.3390/antibiotics10121477",
"doi-asserted-by": "crossref",
"key": "ref_37",
"unstructured": "Poliseno, M., Gallo, C., Cibelli, D.C., Minafra, G.A., Bottalico, I.F., Bruno, S.R., D’Errico, M.L., Montemurro, L., Rizzo, M., and Barbera, L. (2021). Efficacy and Safety of Remdesivir over Two Waves of the SARS-CoV-2 Pandemic. Antibiotics, 10."
},
{
"DOI": "10.1016/j.antiviral.2020.104955",
"article-title": "Antiviral activity and safety of remdesivir against SARS-CoV-2 infection in human pluripotent stem cell-derived cardiomyocytes",
"author": "Choi",
"doi-asserted-by": "crossref",
"first-page": "104955",
"journal-title": "Antivir. Res.",
"key": "ref_38",
"volume": "184",
"year": "2020"
},
{
"DOI": "10.1016/j.amjcard.2020.04.054",
"article-title": "Cardiovascular Safety of Potential Drugs for the Treatment of Coronavirus Disease 2019",
"author": "Aggarwal",
"doi-asserted-by": "crossref",
"first-page": "147",
"journal-title": "Am. J. Cardiol.",
"key": "ref_39",
"volume": "128",
"year": "2020"
},
{
"DOI": "10.3389/jpps.2023.11208",
"doi-asserted-by": "crossref",
"key": "ref_40",
"unstructured": "Kingsley, R., Rohlman, C., Otto, A., Chaudhary, R., Phelan, D., and Kirchoff, R. (2023). Remdesivir-induced conduction abnormalities: A molecular model-based explanation. J. Pharm. Pharm. Sci., 26."
},
{
"DOI": "10.1016/j.jacc.2022.08.800",
"article-title": "Cardiovascular Drug Interactions With Nirmatrelvir/Ritonavir in Patients With COVID-19: JACC Review Topic of the Week",
"author": "Abraham",
"doi-asserted-by": "crossref",
"first-page": "1912",
"journal-title": "J. Am. Coll. Cardiol.",
"key": "ref_41",
"volume": "80",
"year": "2022"
},
{
"DOI": "10.1002/jps.22109",
"article-title": "Review of the basic and clinical pharmacology of sulfobutylether-beta-cyclodextrin (SBECD)",
"author": "Luke",
"doi-asserted-by": "crossref",
"first-page": "3291",
"journal-title": "J. Pharm. Sci.",
"key": "ref_42",
"volume": "99",
"year": "2010"
},
{
"DOI": "10.1136/heartjnl-2021-319971",
"article-title": "Non-invasive markers for sudden cardiac death risk stratification in dilated cardiomyopathy",
"author": "Pooranachandran",
"doi-asserted-by": "crossref",
"first-page": "998",
"journal-title": "Heart",
"key": "ref_43",
"volume": "108",
"year": "2022"
},
{
"DOI": "10.3389/fphar.2020.00072",
"doi-asserted-by": "crossref",
"key": "ref_44",
"unstructured": "Kistamás, K., Veress, R., Horváth, B., Bányász, T., Nánási, P.P., and Eisner, D.A. (2020). Calcium Handling Defects and Cardiac Arrhythmia Syndromes. Front. Pharmacol., 11."
},
{
"DOI": "10.1001/jama.2023.12945",
"article-title": "Monoclonal Antibody and Oral Antiviral Treatment of SARS-CoV-2 Infection in US Nursing Homes",
"author": "McGarry",
"doi-asserted-by": "crossref",
"first-page": "561",
"journal-title": "JAMA",
"key": "ref_45",
"volume": "330",
"year": "2023"
},
{
"DOI": "10.1016/j.chembiol.2024.03.008",
"article-title": "SARS-CoV-2 resistance to monoclonal antibodies and small-molecule drugs",
"author": "Iketani",
"doi-asserted-by": "crossref",
"first-page": "632",
"journal-title": "Cell Chem. Biol.",
"key": "ref_46",
"volume": "31",
"year": "2024"
},
{
"DOI": "10.1016/j.jval.2021.04.1270",
"article-title": "PIN82 Real-World Utilization of Remdesivir in 2020: A Retrospective Cohort Study",
"author": "Mozaffari",
"doi-asserted-by": "crossref",
"first-page": "S121",
"journal-title": "Value Health",
"key": "ref_47",
"volume": "24",
"year": "2021"
}
],
"reference-count": 47,
"references-count": 47,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.mdpi.com/1424-8247/19/4/574"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "Cardiovascular Risks of COVID-19 Therapeutics: Integrated Analysis of FAERS, Electronic Health Records, and Transcriptomics",
"type": "journal-article",
"update-policy": "https://doi.org/10.3390/mdpi_crossmark_policy",
"volume": "19"
}