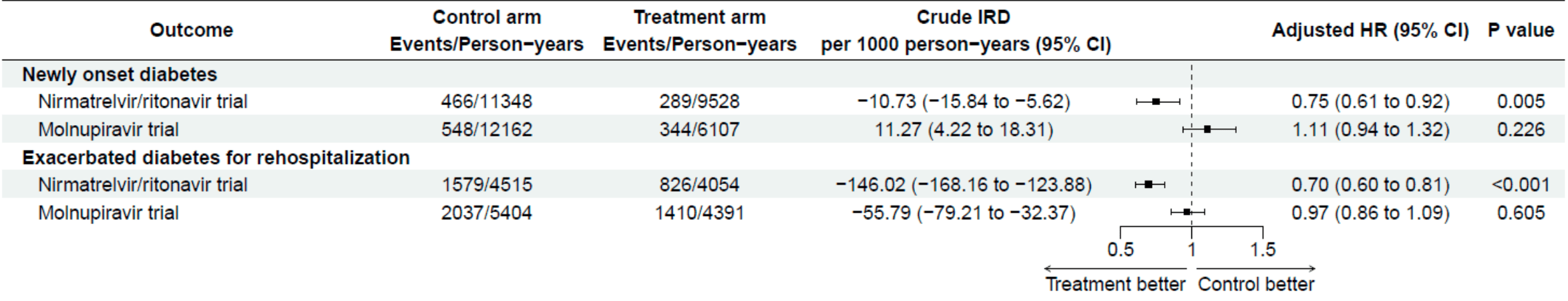
Effectiveness of nirmatrelvir/ritonavir and molnupiravir on post-COVID diabetes risk among an older adult cohort: a target trial emulation study
et al., BMC Medicine, doi:10.1186/s12916-026-04791-2, Mar 2026
Retrospective analysis finding lower risk of post-COVID diabetes with nirmatrelvir/ritonavir but not molnupiravir. The effect may be entirely due to confounding.
Authors excluded only 8% of non-diabetic patients for nirmatrelvir/ritonavir contraindications, compared to the ~20% prevalence expected based on external data1, a gap that may be attributed to limited data and an unusually narrow drug interaction list covering roughly half the FDA-contraindicated medications. Notably the prevalence of renal disease (in the molnupiravir arm where there is no contraindication) is remarkably low, suggesting under-ascertainment from the EHR data. The undetected contraindicated patients (e.g., severe CKD, liver cirrhosis) carry substantially elevated diabetes risk (estimated 1.5-3x baseline) and, critically, would accumulate asymmetrically in the nirmatrelvir/ritonavir control arm through the clone-censor-weight mechanism: clinicians who knew the real-world contraindication would not prescribe nirmatrelvir/ritonavir, causing these patients to be censored from the treatment arm while remaining in the control arm, inflating its event rate. Quantitative modeling shows that if 10% of the trial population had undetected contraindications with 2x the diabetes risk, the observed hazard ratio of 0.75 corrects to approximately 1.0, i.e., the entire effect disappears. This bias is structurally absent from the molnupiravir trial (which has no renal or hepatic contraindication exclusions), providing a parsimonious explanation for why nirmatrelvir/ritonavir appears protective while molnupiravir shows no benefit. Authors do not acknowledge this issue, do not test broader contraindication criteria in sensitivity analyses, and did not recognize that their chosen negative control outcome (injury/trauma) is insensitive to this confounding.
Table S1 shows an exclusion for drug contraindications to nirmatrelvir/ritonavir in the molnupiravir arm, however this conflicts with Figure 1.
Potential risks of molnupiravir include the creation of dangerous variants, and mutagenicity, carcinogenicity, teratogenicity, and embryotoxicity2-16. Multiple analyses have identified variants potentially created by molnupiravir17-21. Studies show significantly increased risk of acute kidney injury22, cardiovascular toxocity23, and neurological symptoms22. Treatment may increase viral rebound24,25.
Study covers molnupiravir and paxlovid.
1.
Lim et al., Prevalence of Medical Contraindications to Nirmatrelvir/Ritonavir in a Cohort of Hospitalized and Nonhospitalized Patients With COVID-19, Open Forum Infectious Diseases, doi:10.1093/ofid/ofac389.
2.
Swanstrom et al., Lethal mutagenesis as an antiviral strategy, Science, doi:10.1126/science.abn0048.
3.
Hadj Hassine et al., Lethal Mutagenesis of RNA Viruses and Approved Drugs with Antiviral Mutagenic Activity, Viruses, doi:10.3390/v14040841.
4.
Shum, C., An investigational study into the drug-associated mutational signature in SARS-CoV-2 viruses, The University of Hong Kong, PhD Thesis, hub.hku.hk/handle/10722/344396.
5.
Waters et al., Human genetic risk of treatment with antiviral nucleoside analog drugs that induce lethal mutagenesis: the special case of molnupiravir, Environmental and Molecular Mutagenesis, doi:10.1002/em.22471.
6.
Huntsman, M., An assessment of the reproductive toxicity of the anti-COVID-19 drug molnupiravir using stem cell-based embryo models, Master's Thesis, scholarspace.manoa.hawaii.edu/items/cd11342c-b4dc-44c0-8b44-ce6e3369c40b.
7.
Huntsman (B) et al., Detection of developmental toxicity of the anti-COVID-19 drug molnupiravir using gastruloid-based in vitro assays, Toxicological Sciences, doi:10.1093/toxsci/kfaf093.
8.
Zibat et al., N4-hydroxycytidine, the active compound of Molnupiravir, promotes SARS-CoV-2 mutagenesis and escape from a neutralizing nanobody, iScience, doi:10.1016/j.isci.2023.107786.
9.
Shiraki et al., Convenient screening of the reproductive toxicity of favipiravir and antiviral drugs in Caenorhabditis elegans, Heliyon, doi:10.1016/j.heliyon.2024.e35331.
10.
Gruber et al., Molnupiravir increases SARS‐CoV‐2 genome diversity and complexity: A case‐control cohort study, Journal of Medical Virology, doi:10.1002/jmv.29642.
11.
Marikawa et al., An active metabolite of the anti-COVID-19 drug molnupiravir impairs mouse preimplantation embryos at clinically relevant concentrations, Reproductive Toxicology, doi:10.1016/j.reprotox.2023.108475.
12.
Rahman, M., Elucidation of the DNA repair mechanisms involved in the repair of DNA damage caused by the Arabinosides and Anti-COVID-19 drugs, tokyo-metro-u.repo.nii.ac.jp/records/2000972.
13.
Zhou et al., β-D-N4-hydroxycytidine Inhibits SARS-CoV-2 Through Lethal Mutagenesis But Is Also Mutagenic To Mammalian Cells, The Journal of Infectious Diseases, doi:10.1093/infdis/jiab247.
14.
Chamod et al., Molnupiravir Metabolite--N4-hydroxycytidine Causes Cytotoxicity and DNA Damage in Mammalian Cells in vitro: N4-hydroxycytidine Induced Cytotoxicity DNA Damage, Asian Medical Journal and Alternative Medicine, 23:3, asianmedjam.com/index.php/amjam/article/view/1448.
15.
Standing et al., Randomized controlled trial of molnupiravir SARS-CoV-2 viral and antibody response in at-risk adult outpatients, Nature Communications, doi:10.1038/s41467-024-45641-0.
16.
Mori et al., Reactive oxygen species-mediated cytotoxic and DNA-damaging mechanism of N4-hydroxycytidine, a metabolite of the COVID-19 therapeutic drug molnupiravir, Free Radical Research, doi:10.1080/10715762.2025.2469738.
17.
Focosi et al., The fitness of molnupiravir-signed SARS-CoV-2 variants: imputation analysis based on prescription counts and GISAID analyses by country, Intervirology, doi:10.1159/000540282.
18.
Sanderson et al., A molnupiravir-associated mutational signature in global SARS-CoV-2 genomes, Nature, doi:10.1038/s41586-023-06649-6.
19.
Fountain-Jones et al., Effect of molnupiravir on SARS-CoV-2 evolution in immunocompromised patients: a retrospective observational study, The Lancet Microbe, doi:10.1016/S2666-5247(23)00393-2.
20.
Kosakovsky Pond et al., Anti-COVID drug accelerates viral evolution, Nature, doi:10.1038/d41586-023-03248-3.
22.
Siby et al., Temporal Trends in Serious Adverse Events Associated with Oral Antivirals During the COVID-19 Pandemic: Insights from the FAERS Database (2020–2023), Open Forum Infectious Diseases, doi:10.1093/ofid/ofaf695.1825.
23.
Ozhan et al., Evaluation of the cardiopulmonary effects of repurposed COVID-19 therapeutics in healthy rats, Scientific Reports, doi:10.1038/s41598-025-31048-4.
Guo et al., 17 Mar 2026, retrospective, China, peer-reviewed, mean age 73.0, 17 authors, study period 11 March, 2022 - 10 October, 2023.
Contact: marc@cuhk.edu.hk, yeoh_ek@cuhk.edu.hk.
Abstract: BMC Medicine
https://doi.org/10.1186/s12916-026-04791-2
Article in Press
Effectiveness of nirmatrelvir/ritonavir and
molnupiravir on post-COVID diabetes risk among an
older adult cohort: a target trial emulation study
Zihao Guo, Yuchen Wei, Aimin Yang, Carlos Wong, Xi Xiong, Kailu Wang, Guozhang Lin,
Huwen Wang, Chi Hung, Conglu Li, Carrie Yam, Tsz Chow, Shi Zhao, Chris Mok, David
Hui, Eng Yeoh & Ka Chong
Received: 19 Nov 2025
Accepted: 09 Mar 2026
Cite this article as: Guo, Z., Wei,
Y., Yang, A. et al. Effectiveness
of nirmatrelvir/ritonavir and
molnupiravir on post-COVID diabetes
risk among an older adult cohort: a
target trial emulation study. BMC
Med (2026).
https://doi.org/10.1186/s1291
6-026-04791-2
A
E
R
P
S
S
We are providing an unedited version of this manuscript to give early access to its
findings. Before final publication, the manuscript will undergo further editing. Please
note there may be errors present which affect the content, and all legal disclaimers
apply.
IN
If this paper is publishing under a Transparent Peer Review model then Peer
Review reports will publish with the final article.
I
T
R
E
L
C
©The Author(s) 2026. Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits
use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original
author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third
party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the
material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory
regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this
licence, visit http://creativecommons.org/licenses/by/4.0/
ACCEPTED
ARTICLE IN
MANUSCRIPT
PRESS
Effectiveness of nirmatrelvir/ritonavir and molnupiravir on post-COVID diabetes risk
among an older adult cohort: A target trial emulation study
Zihao Guo, PhD1#; Yuchen Wei, PhD1#; Aimin Yang, PhD2; Carlos King Ho Wong, PhD3,4,5;
Xi Xiong, PhD3,6; Kailu Wang, PhD1; Guozhang Lin, MSc1; Huwen Wang, PhD7; Chi Tim
Hung, MBBS1; Conglu Li, MSc1; Carrie Ho Kwan Yam, PhD1; Tsz Yu Chow, BSc1; Shi
Zhao, PhD1,8; Chris Ka Pun Mok, PhD9; David SC Hui, MBBS10; Eng Kiong Yeoh, MBBS1*;
Ka Chun Chong, PhD1*
1
School of Public Health and Primary Care, The Chinese University of Hong Kong, Hong
Kong Special Administrative Region, China
2
Department of Medicine & Therapeutics, Faculty of Medicine, The Chinese University of
Hong Kong, Hong Kong
3
Laboratory of Data Discovery for Health, Hong Kong Science Park, Hong Kong Special
Administrative Region, China
4
Department of Family Medicine and Primary Care, School of Clinical Medicine, Li Ka
Shing Faculty of Medicine, The University of Hong Kong, Hong Kong Special
Administrative Region, China
5
Department of Infectious Disease Epidemiology, London School of Hygiene and Tropical
Medicine, London, United Kingdom
6
Research Department of Practice and Policy, School of Pharmacy, University College
London, London, United Kingdom
7
Duke-NUS Medical School, Singapore
8
School of Public Health, Tianjin Medical University, China
9
Li Ka Shing Institute of Health Sciences, Chinese University of..
DOI record:
{
"DOI": "10.1186/s12916-026-04791-2",
"ISSN": [
"1741-7015"
],
"URL": "http://dx.doi.org/10.1186/s12916-026-04791-2",
"abstract": "<jats:title>Abstract</jats:title>\n <jats:sec>\n <jats:title>Background</jats:title>\n <jats:p>Accumulating evidence indicates that SARS-CoV-2 infection is associated with a broad spectrum of post-acute COVID sequelae, including diabetes. While nirmatrelvir/ritonavir and molnupiravir have demonstrated efficacy in reducing acute COVID-19 severity, their protective effects against post-COVID diabetes remain uncertain. In this study, we aimed to evaluate the effectiveness of these antiviral agents in reducing post-COVID diabetes risks, including new-onset diabetes in non-diabetic individuals and exacerbated diabetes in those with pre-existing diabetes.</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Methods</jats:title>\n <jats:p>We emulate target randomized controlled trials of COVID-19 antivirals in hospitalized patients who tested positive for SARS-CoV-2 between March 11, 2022, and October 10, 2023, in Hong Kong. Two analytic patient cohorts for assessing incident diabetes and exacerbation of diabetes for rehospitalization, including those with or without diabetes confirmed before the index date, were identified. Cloning, censoring, and weighting were used to emulate the target trials of nirmatrelvir/ritonavir and molnupiravir, involving treatment arm and control arm within each trial. Cause-specific Cox proportional hazard model and an extended form of Cox model for modeling recurrent hospitalizations were used to estimate the hazard ratio (HR) between arms in each trial, adjusting for baseline covariates.</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Results</jats:title>\n <jats:p>Among 88,643 hospitalized patients first time infected by SARS-CoV-2 identified, 35,997 and 18,865 eligible patients were included in the two analytic cohorts for the analysis on newly onset diabetes and exacerbated diabetes for rehospitalization, respectively. The median follow-up period ranged from 344 to 365 days across treatment and control arms of target trials. Compared with the no treatment arm, non-diabetic patients who received nirmatrelvir/ritonavir showed a significantly lower risk of post-COVID incident diabetes (HR: 0.75, 95% CI: 0.61 to 0.92). A reduced risk of diabetes rehospitalizations (HR: 0.70, 95% CI: 0.60 to 0.81) was observed among the diabetic patients. No significant associations were found for the use of molnupiravir and post-COVID diabetes outcomes.</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Conclusions</jats:title>\n <jats:p>Our study demonstrates the effectiveness of nirmatrelvir/ritonavir in reducing the risks of post-acute COVID sequelae of diabetes in the hospitalized population, regardless of their diabetic status, whereas molnupiravir showed no significant benefit. Our findings offer valuable clinical insights for managing diabetes during the post-acute phase of SARS-CoV-2 infection.</jats:p>\n </jats:sec>",
"alternative-id": [
"4791"
],
"assertion": [
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "Received",
"name": "received",
"order": 1,
"value": "19 November 2025"
},
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "Accepted",
"name": "accepted",
"order": 2,
"value": "9 March 2026"
},
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "First Online",
"name": "first_online",
"order": 3,
"value": "17 March 2026"
},
{
"group": {
"label": "Declarations",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 1
},
{
"group": {
"label": "Ethics approval and consent to participate",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 2,
"value": "Ethics approval was obtained from the Joint CUHK-NTEC Clinical Research Ethics Committee (No. 2023.006)."
},
{
"group": {
"label": "Consent for publication",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 3,
"value": "Not applicable."
},
{
"group": {
"label": "Competing interests",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 4,
"value": "The authors declare no competing interests."
}
],
"author": [
{
"affiliation": [],
"family": "Guo",
"given": "Zihao",
"sequence": "first"
},
{
"affiliation": [],
"family": "Wei",
"given": "Yuchen",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Yang",
"given": "Aimin",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Wong",
"given": "Carlos King Ho",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Xiong",
"given": "Xi",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Wang",
"given": "Kailu",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Lin",
"given": "Guozhang",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Wang",
"given": "Huwen",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Hung",
"given": "Chi Tim",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Li",
"given": "Conglu",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Yam",
"given": "Carrie Ho Kwan",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Chow",
"given": "Tsz Yu",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Zhao",
"given": "Shi",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Mok",
"given": "Chris Ka Pun",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Hui",
"given": "David S. C.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Yeoh",
"given": "Eng Kiong",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Chong",
"given": "Ka Chun",
"sequence": "additional"
}
],
"container-title": "BMC Medicine",
"container-title-short": "BMC Med",
"content-domain": {
"crossmark-restriction": false,
"domain": [
"link.springer.com"
]
},
"created": {
"date-parts": [
[
2026,
3,
17
]
],
"date-time": "2026-03-17T11:06:37Z",
"timestamp": 1773745597000
},
"deposited": {
"date-parts": [
[
2026,
3,
17
]
],
"date-time": "2026-03-17T11:06:48Z",
"timestamp": 1773745608000
},
"funder": [
{
"award": [
"C6036-21GF"
],
"award-info": [
{
"award-number": [
"C6036-21GF"
]
}
],
"name": "RGC Collaborative Research Fund"
},
{
"award": [
"T11-705/21-N"
],
"award-info": [
{
"award-number": [
"T11-705/21-N"
]
}
],
"name": "RGC theme-based research schemes"
},
{
"DOI": "10.13039/501100005847",
"award": [
"COVID190105, COVID19F03, INF-CUHK-1, COVID1903003"
],
"award-info": [
{
"award-number": [
"COVID190105, COVID19F03, INF-CUHK-1, COVID1903003"
]
}
],
"doi-asserted-by": "publisher",
"id": [
{
"asserted-by": "publisher",
"id": "10.13039/501100005847",
"id-type": "DOI"
}
],
"name": "Health and Medical Research Fund"
},
{
"award": [
"2025.059"
],
"award-info": [
{
"award-number": [
"2025.059"
]
}
],
"name": "CUHK Direct Grant"
},
{
"DOI": "10.13039/501100001809",
"award": [
"72574190"
],
"award-info": [
{
"award-number": [
"72574190"
]
}
],
"doi-asserted-by": "publisher",
"id": [
{
"asserted-by": "publisher",
"id": "10.13039/501100001809",
"id-type": "DOI"
}
],
"name": "National Natural Science Foundation of China"
}
],
"indexed": {
"date-parts": [
[
2026,
3,
17
]
],
"date-time": "2026-03-17T12:01:20Z",
"timestamp": 1773748880844,
"version": "3.50.1"
},
"is-referenced-by-count": 0,
"issued": {
"date-parts": [
[
2026,
3,
17
]
]
},
"language": "en",
"license": [
{
"URL": "https://creativecommons.org/licenses/by/4.0",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
3,
17
]
],
"date-time": "2026-03-17T00:00:00Z",
"timestamp": 1773705600000
}
},
{
"URL": "https://creativecommons.org/licenses/by/4.0",
"content-version": "am",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
3,
17
]
],
"date-time": "2026-03-17T00:00:00Z",
"timestamp": 1773705600000
}
}
],
"link": [
{
"URL": "https://link.springer.com/article/10.1186/s12916-026-04791-2",
"content-type": "text/html",
"content-version": "vor",
"intended-application": "text-mining"
}
],
"member": "297",
"original-title": [],
"prefix": "10.1186",
"published": {
"date-parts": [
[
2026,
3,
17
]
]
},
"published-online": {
"date-parts": [
[
2026,
3,
17
]
]
},
"publisher": "Springer Science and Business Media LLC",
"reference": [
{
"DOI": "10.1038/s41591-022-02001-z",
"author": "E Xu",
"doi-asserted-by": "crossref",
"first-page": "2406",
"issue": "11",
"journal-title": "Nat Med",
"key": "4791_CR1",
"unstructured": "Xu E, Xie Y, Al-Aly Z. Long-term neurologic outcomes of COVID-19. Nat Med. 2022;28(11):2406–15.",
"volume": "28",
"year": "2022"
},
{
"DOI": "10.1038/s41591-022-01689-3",
"author": "Y Xie",
"doi-asserted-by": "crossref",
"first-page": "583",
"issue": "3",
"journal-title": "Nat Med",
"key": "4791_CR2",
"unstructured": "Xie Y, Xu E, Bowe B, Al-Aly Z. Long-term cardiovascular outcomes of COVID-19. Nat Med. 2022;28(3):583–90.",
"volume": "28",
"year": "2022"
},
{
"DOI": "10.1016/S2213-8587(22)00355-2",
"author": "Y Xie",
"doi-asserted-by": "crossref",
"first-page": "120",
"issue": "2",
"journal-title": "Lancet Diabetes Endocrinol",
"key": "4791_CR3",
"unstructured": "Xie Y, Al-Aly Z. Risks and burdens of incident diabetes in long COVID: a cohort study. Lancet Diabetes Endocrinol. 2023;11(2):120–8.",
"volume": "11",
"year": "2023"
},
{
"DOI": "10.1038/s41598-022-24185-7",
"author": "P Ssentongo",
"doi-asserted-by": "crossref",
"issue": "1",
"journal-title": "Sci Rep",
"key": "4791_CR4",
"unstructured": "Ssentongo P, Zhang Y, Witmer L, Chinchilli VM, Ba DM. Association of COVID-19 with diabetes: a systematic review and meta-analysis. Sci Rep. 2022;12(1):20191.",
"volume": "12",
"year": "2022"
},
{
"author": "JL Harding",
"issue": "110202",
"journal-title": "Diabetes Res Clin Pract",
"key": "4791_CR5",
"unstructured": "Harding JL, Oviedo SA, Ali MK, Ofotokun I, Gander JC, Patel SA, et al. The bidirectional association between diabetes and long-COVID-19 - a systematic review. Diabetes Res Clin Pract. 2023;195(110202):110202.",
"volume": "195",
"year": "2023"
},
{
"DOI": "10.1001/jamanetworkopen.2022.33014",
"author": "EK Kendall",
"doi-asserted-by": "crossref",
"issue": "9",
"journal-title": "JAMA Netw Open",
"key": "4791_CR6",
"unstructured": "Kendall EK, Olaker VR, Kaelber DC, Xu R, Davis PB. Association of SARS-CoV-2 infection with new-onset type 1 diabetes among pediatric patients from 2020 to 2021. JAMA Netw Open. 2022;5(9):e2233014.",
"volume": "5",
"year": "2022"
},
{
"DOI": "10.1056/NEJMoa2118542",
"author": "J Hammond",
"doi-asserted-by": "crossref",
"first-page": "1397",
"issue": "15",
"journal-title": "N Engl J Med",
"key": "4791_CR7",
"unstructured": "Hammond J, Leister-Tebbe H, Gardner A, Abreu P, Bao W, Wisemandle W, et al. Oral nirmatrelvir for high-risk, nonhospitalized adults with COVID-19. N Engl J Med. 2022;386(15):1397–408.",
"volume": "386",
"year": "2022"
},
{
"DOI": "10.1016/S1473-3099(22)00507-2",
"author": "CKH Wong",
"doi-asserted-by": "crossref",
"first-page": "1681",
"issue": "12",
"journal-title": "Lancet Infect Dis",
"key": "4791_CR8",
"unstructured": "Wong CKH, Au ICH, Lau KTK, Lau EHY, Cowling BJ, Leung GM. Real-world effectiveness of early molnupiravir or nirmatrelvir-ritonavir in hospitalised patients with COVID-19 without supplemental oxygen requirement on admission during Hong Kong’s omicron BA.2 wave: a retrospective cohort study. Lancet Infect Dis. 2022;22(12):1681–93.",
"volume": "22",
"year": "2022"
},
{
"DOI": "10.1056/NEJMoa2116044",
"doi-asserted-by": "crossref",
"key": "4791_CR9",
"unstructured": "Jayk Bernal A, Gomes da Silva MM, Musungaie DB, et al. Molnupiravir for oral treatment of COVID-19 in nonhospitalized patients. N Engl J Med. 2022;386(6):509–20."
},
{
"DOI": "10.7326/M22-3565",
"doi-asserted-by": "crossref",
"key": "4791_CR10",
"unstructured": "Bajema KL, Berry K, Streja E, Rajeevan N, Li Y, Mutalik P, et al. Effectiveness of COVID-19 treatment with nirmatrelvir-ritonavir or molnupiravir among U.S. veterans: target trial emulation studies with one-month and six-month outcomes. Ann Intern Med. 2023;176(6):807–16."
},
{
"DOI": "10.1001/jamainternmed.2023.5099",
"author": "KW Fung",
"doi-asserted-by": "crossref",
"first-page": "1404",
"issue": "12",
"journal-title": "JAMA Intern Med",
"key": "4791_CR11",
"unstructured": "Fung KW, Baye F, Baik SH, McDonald CJ. Nirmatrelvir and molnupiravir and post-COVID-19 condition in older patients. JAMA Intern Med. 2023;183(12):1404–6.",
"volume": "183",
"year": "2023"
},
{
"DOI": "10.1016/S1473-3099(24)00217-2",
"author": "H Wang",
"doi-asserted-by": "crossref",
"first-page": "1130",
"issue": "10",
"journal-title": "Lancet Infect Dis",
"key": "4791_CR12",
"unstructured": "Wang H, Wei Y, Hung CT, Lin G, Jiang X, Li C, et al. Association of nirmatrelvir-ritonavir with post-acute sequelae and mortality in patients admitted to hospital with COVID-19: a retrospective cohort study. Lancet Infect Dis. 2024;24(10):1130–40.",
"volume": "24",
"year": "2024"
},
{
"DOI": "10.1016/S2665-9913(24)00224-8",
"author": "G Lin",
"doi-asserted-by": "crossref",
"first-page": "e108",
"issue": "2",
"journal-title": "Lancet Rheumatol",
"key": "4791_CR13",
"unstructured": "Lin G, Wei Y, Wang H, Boyer C, Jia KM, Hung CT, et al. Association of nirmatrelvir-ritonavir with post-acute sequelae and mortality among patients who are immunocompromised with COVID-19 in Hong Kong: a retrospective cohort study. Lancet Rheumatol. 2025;7(2):e108–17.",
"volume": "7",
"year": "2025"
},
{
"DOI": "10.1080/22221751.2025.2469648",
"author": "Y Wei",
"doi-asserted-by": "crossref",
"first-page": "2469648",
"issue": "1",
"journal-title": "Emerg Microbes Infect",
"key": "4791_CR14",
"unstructured": "Wei Y, Boyer C, Jia KM, Lin G, Wang H, Li C, et al. Effectiveness of nirmatrelvir/ritonavir and molnupiravir on post-COVID-19 outcomes among outpatients: a target trial emulation investigation. Emerg Microbes Infect. 2025;14(1):2469648.",
"volume": "14",
"year": "2025"
},
{
"DOI": "10.1001/jamainternmed.2024.2007",
"author": "LN Geng",
"doi-asserted-by": "crossref",
"first-page": "1024",
"issue": "9",
"journal-title": "JAMA Intern Med",
"key": "4791_CR15",
"unstructured": "Geng LN, Bonilla H, Hedlin H, Jacobson KB, Tian L, Jagannathan P, et al. Nirmatrelvir-ritonavir and symptoms in adults with postacute sequelae of SARS-CoV-2 infection: the STOP-PASC randomized clinical trial. JAMA Intern Med. 2024;184(9):1024–34.",
"volume": "184",
"year": "2024"
},
{
"DOI": "10.1016/S1473-3099(25)00073-8",
"author": "M Sawano",
"doi-asserted-by": "crossref",
"first-page": "936",
"issue": "8",
"journal-title": "Lancet Infect Dis",
"key": "4791_CR16",
"unstructured": "Sawano M, Bhattacharjee B, Caraballo C, Khera R, Li S-X, Herrin J, et al. Nirmatrelvir-ritonavir versus placebo-ritonavir in individuals with long COVID in the USA (PAX LC): a double-blind, randomised, placebo-controlled, phase 2, decentralised trial. Lancet Infect Dis. 2025;25(8):936–46.",
"volume": "25",
"year": "2025"
},
{
"DOI": "10.7326/M23-1394",
"doi-asserted-by": "crossref",
"key": "4791_CR17",
"unstructured": "Ioannou GN, Berry K, Rajeevan N, Li Y, Mutalik P, Yan L, et al. Effectiveness of nirmatrelvir-ritonavir against the development of post-COVID-19 conditions among U.S. veterans: a target trial emulation. Ann Intern Med. 2023;176(11):1486–97."
},
{
"DOI": "10.1001/jamainternmed.2023.0743",
"author": "Y Xie",
"doi-asserted-by": "crossref",
"first-page": "554",
"issue": "6",
"journal-title": "JAMA Intern Med",
"key": "4791_CR18",
"unstructured": "Xie Y, Choi T, Al-Aly Z. Association of treatment with nirmatrelvir and the risk of post-COVID-19 condition. JAMA Intern Med. 2023;183(6):554–64.",
"volume": "183",
"year": "2023"
},
{
"DOI": "10.1016/j.isci.2025.111907",
"doi-asserted-by": "crossref",
"key": "4791_CR19",
"unstructured": "Su G, Li S, Zhang D, Zhou Y, Wang L, Yuan Y, et al. Real-world effectiveness of azvudine versus nirmatrelvir-ritonavir in hospitalized patients with COVID-19 and pre-existing diabetes. iScience. 2025;28(2):111907."
},
{
"author": "J-Y Wu",
"issue": "6",
"journal-title": "J Med Virol",
"key": "4791_CR20",
"unstructured": "Wu J-Y, Liu M-Y, Liu T-H, Chuang M-H, Hsu W-H, Huang P-Y, et al. Clinical efficacy of nirmatrelvir and ritonavir combination for treating diabetic patients with COVID-19. J Med Virol. 2023;95(6):e28866.",
"volume": "95",
"year": "2023"
},
{
"author": "DTW Lui",
"issue": "5",
"journal-title": "JAMA Netw Open",
"key": "4791_CR21",
"unstructured": "Lui DTW, Chung MSH, Lau EHY, Lau KTK, Au ICH, Lee CH, et al. Analysis of all-cause hospitalization and death among nonhospitalized patients with type 2 diabetes and SARS-CoV-2 infection treated with molnupiravir or nirmatrelvir-ritonavir during the omicron wave in Hong Kong. JAMA Netw Open. 2023;6(5):e2314393.",
"volume": "6",
"year": "2023"
},
{
"DOI": "10.1111/dom.15830",
"author": "EYF Wan",
"doi-asserted-by": "crossref",
"first-page": "4653",
"issue": "10",
"journal-title": "Diabetes Obes Metab",
"key": "4791_CR22",
"unstructured": "Wan EYF, Wong ZCT, Yan VKC, Chui CSL, Lai FTT, Li X, et al. Comparing the effectiveness of molnupiravir and nirmatrelvir-ritonavir in non-hospitalized and hospitalized COVID-19 patients with type 2 diabetes: a target trial emulation study. Diabetes Obes Metab. 2024;26(10):4653–64.",
"volume": "26",
"year": "2024"
},
{
"DOI": "10.1001/jamanetworkopen.2023.8872",
"author": "PB Davis",
"doi-asserted-by": "crossref",
"issue": "4",
"journal-title": "JAMA Netw Open",
"key": "4791_CR23",
"unstructured": "Davis PB, Xu R. COVID-19 and incident diabetes-recovery is not so sweet after all. JAMA Netw Open. 2023;6(4):e238872.",
"volume": "6",
"year": "2023"
},
{
"DOI": "10.1016/j.jclinepi.2016.04.014",
"author": "MA Hernán",
"doi-asserted-by": "crossref",
"first-page": "70",
"journal-title": "J Clin Epidemiol",
"key": "4791_CR24",
"unstructured": "Hernán MA, Sauer BC, Hernández-Díaz S, Platt R, Shrier I. Specifying a target trial prevents immortal time bias and other self-inflicted injuries in observational analyses. J Clin Epidemiol. 2016;79:70–5.",
"volume": "79",
"year": "2016"
},
{
"DOI": "10.1093/ije/dyaa057",
"doi-asserted-by": "crossref",
"key": "4791_CR25",
"unstructured": "Maringe C, Benitez Majano S, Exarchakou A, Smith M, Rachet B, Belot A, et al. Reflection on modern methods: trial emulation in the presence of immortal-time bias. Assessing the benefit of major surgery for elderly lung cancer patients using observational data. Int J Epidemiol. 2020;49(5):1719–29."
},
{
"DOI": "10.1001/jama.2025.13350",
"author": "AG Cashin",
"doi-asserted-by": "crossref",
"first-page": "1084",
"issue": "12",
"journal-title": "JAMA",
"key": "4791_CR26",
"unstructured": "Cashin AG, Hansford HJ, Hernán MA, Swanson SA, Lee H, Jones MD, et al. Transparent reporting of observational studies emulating a target trial-the TARGET statement. JAMA. 2025;334(12):1084–93.",
"volume": "334",
"year": "2025"
},
{
"DOI": "10.7326/M22-3057",
"author": "EYF Wan",
"doi-asserted-by": "crossref",
"first-page": "505",
"issue": "4",
"journal-title": "Ann Intern Med",
"key": "4791_CR27",
"unstructured": "Wan EYF, Yan VKC, Mok AHY, Wang B, Xu W, Cheng FWT, et al. Effectiveness of molnupiravir and nirmatrelvir–ritonavir in hospitalized patients with COVID-19. Ann Intern Med. 2023;176(4):505–14.",
"volume": "176",
"year": "2023"
},
{
"key": "4791_CR28",
"unstructured": "Norwegian Institute of Public Health. WHOCC. ATCDDD - ATC/DDD index. Fhi.no. [Accessed 13 Aug 2025]. Available from: https://atcddd.fhi.no/atc_ddd_index/?code=a10"
},
{
"key": "4791_CR29",
"unstructured": "Hospital Authority. Hospital authority drug formulary management. [Accessed 13 Aug 2025]. Available from: https://www.ha.org.hk/hadf/en-us/"
},
{
"DOI": "10.2337/dc20-2850",
"author": "B Zheng",
"doi-asserted-by": "crossref",
"first-page": "1556",
"issue": "7",
"journal-title": "Diabetes Care",
"key": "4791_CR30",
"unstructured": "Zheng B, Su B, Price G, Tzoulaki I, Ahmadi-Abhari S, Middleton L. Glycemic control, diabetic complications, and risk of dementia in patients with diabetes: results from a large U.K. cohort study. Diabetes Care. 2021;44(7):1556–63.",
"volume": "44",
"year": "2021"
},
{
"DOI": "10.1001/jamasurg.2025.3081",
"author": "X Liu",
"doi-asserted-by": "crossref",
"first-page": "1125",
"issue": "10",
"journal-title": "JAMA Surg",
"key": "4791_CR31",
"unstructured": "Liu X, Lui DTW, Xiong X, Li L, Luk Y, Wong CH, et al. Parathyroidectomy and risk of incident diabetes in patients with primary hyperparathyroidism. JAMA Surg. 2025;160(10):1125–32.",
"volume": "160",
"year": "2025"
},
{
"DOI": "10.1001/jamainternmed.2021.5714",
"author": "IA Mansi",
"doi-asserted-by": "crossref",
"first-page": "1562",
"issue": "12",
"journal-title": "JAMA Intern Med",
"key": "4791_CR32",
"unstructured": "Mansi IA, Chansard M, Lingvay I, Zhang S, Halm EA, Alvarez CA. Association of statin therapy initiation with diabetes progression: a retrospective matched-cohort study: a retrospective matched-cohort study. JAMA Intern Med. 2021;181(12):1562–74.",
"volume": "181",
"year": "2021"
},
{
"author": "ICH Lam",
"issue": "102000",
"journal-title": "EClinicalMedicine",
"key": "4791_CR33",
"unstructured": "Lam ICH, Wong CKH, Zhang R, Chui CSL, Lai FTT, Li X, et al. Long-term post-acute sequelae of COVID-19 infection: a retrospective, multi-database cohort study in Hong Kong and the UK. EClinicalMedicine. 2023;60(102000):102000.",
"volume": "60",
"year": "2023"
},
{
"author": "Z Guo",
"issue": "1",
"journal-title": "Nat Commun",
"key": "4791_CR34",
"unstructured": "Guo Z, Wei Y, Lin G, Jia KM, Boyer C, Wang H, et al. Effectiveness of nirmatrelvir/ritonavir and molnupiravir in reducing the risk of short-term and long-term cardiovascular complications of COVID-19: a target trial emulation study. Nat Commun. 2025;17(1):1031.",
"volume": "17",
"year": "2025"
},
{
"DOI": "10.1093/ckj/sfae164",
"doi-asserted-by": "crossref",
"key": "4791_CR35",
"unstructured": "Zhang Y, Ba DM, Risher K, Liao D, Parent LJ, Ghahramani N, et al. Effects of ACE inhibitor/ARB therapy and long COVID on kidney disease: a retrospective cohort study using real-world data. Clin Kidney J. 2024;17(7):sfae164."
},
{
"key": "4791_CR36",
"unstructured": "Fact sheet for healthcare providers: emergency use authorization for Paxlovid™. Fda.gov. [Accessed 20 Jun 2025]. Available from: https://www.fda.gov/media/155050/download"
},
{
"key": "4791_CR37",
"unstructured": "Fact sheet for healthcare providers: emergency use authorization for Lagevrio™ (molnupiravir) capsules. Fda.gov. [Accessed 20 Jun 2025]. Available from: https://www.fda.gov/media/155054/download"
},
{
"DOI": "10.1080/03610910902859574",
"author": "PC Austin",
"doi-asserted-by": "crossref",
"first-page": "1228",
"issue": "6",
"journal-title": "Commun Stat",
"key": "4791_CR38",
"unstructured": "Austin PC. Using the standardized difference to compare the prevalence of a binary variable between two groups in observational research. Commun Stat. 2009;38(6):1228–34.",
"volume": "38",
"year": "2009"
},
{
"DOI": "10.1001/jama.2024.4970",
"author": "PC Austin",
"doi-asserted-by": "crossref",
"first-page": "2125",
"issue": "24",
"journal-title": "JAMA",
"key": "4791_CR39",
"unstructured": "Austin PC, Ibrahim M, Putter H. Accounting for competing risks in clinical research. JAMA. 2024;331(24):2125–6.",
"volume": "331",
"year": "2024"
},
{
"DOI": "10.1161/CIRCULATIONAHA.115.017719",
"author": "PC Austin",
"doi-asserted-by": "crossref",
"first-page": "601",
"issue": "6",
"journal-title": "Circulation",
"key": "4791_CR40",
"unstructured": "Austin PC, Lee DS, Fine JP. Introduction to the analysis of survival data in the presence of competing risks. Circulation. 2016;133(6):601–9.",
"volume": "133",
"year": "2016"
},
{
"DOI": "10.1093/ije/dyu222",
"author": "LDAF Amorim",
"doi-asserted-by": "crossref",
"first-page": "324",
"issue": "1",
"journal-title": "Int J Epidemiol",
"key": "4791_CR41",
"unstructured": "Amorim LDAF, Cai J. Modelling recurrent events: a tutorial for analysis in epidemiology. Int J Epidemiol. 2015;44(1):324–33.",
"volume": "44",
"year": "2015"
},
{
"DOI": "10.1093/aje/kwn164",
"author": "SR Cole",
"doi-asserted-by": "crossref",
"first-page": "656",
"issue": "6",
"journal-title": "Am J Epidemiol",
"key": "4791_CR42",
"unstructured": "Cole SR, Hernán MA. Constructing inverse probability weights for marginal structural models. Am J Epidemiol. 2008;168(6):656–64.",
"volume": "168",
"year": "2008"
},
{
"DOI": "10.1097/EDE.0b013e3181d61eeb",
"author": "M Lipsitch",
"doi-asserted-by": "crossref",
"first-page": "383",
"issue": "3",
"journal-title": "Epidemiology",
"key": "4791_CR43",
"unstructured": "Lipsitch M, Tchetgen Tchetgen E, Cohen T. Negative controls: a tool for detecting confounding and bias in observational studies: a tool for detecting confounding and bias in observational studies. Epidemiology. 2010;21(3):383–8.",
"volume": "21",
"year": "2010"
},
{
"DOI": "10.1016/j.diabet.2020.10.002",
"author": "T Sathish",
"doi-asserted-by": "crossref",
"issue": "2",
"journal-title": "Diabetes Metab",
"key": "4791_CR44",
"unstructured": "Sathish T, Tapp RJ, Cooper ME, Zimmet P. Potential metabolic and inflammatory pathways between COVID-19 and new-onset diabetes. Diabetes Metab. 2021;47(2):101204.",
"volume": "47",
"year": "2021"
},
{
"DOI": "10.1371/journal.pmed.1004052",
"author": "E Rezel-Potts",
"doi-asserted-by": "crossref",
"issue": "7",
"journal-title": "PLoS Med",
"key": "4791_CR45",
"unstructured": "Rezel-Potts E, Douiri A, Sun X, Chowienczyk PJ, Shah AM, Gulliford MC. Cardiometabolic outcomes up to 12 months after COVID-19 infection. A matched cohort study in the UK. PLoS Med. 2022;19(7):e1004052.",
"volume": "19",
"year": "2022"
},
{
"DOI": "10.1001/jamanetworkopen.2023.8866",
"author": "Z Naveed",
"doi-asserted-by": "crossref",
"issue": "4",
"journal-title": "JAMA Netw Open",
"key": "4791_CR46",
"unstructured": "Naveed Z, Velásquez García HA, Wong S, Wilton J, McKee G, Mahmood B, et al. Association of COVID-19 infection with incident diabetes. JAMA Netw Open. 2023;6(4):e238866.",
"volume": "6",
"year": "2023"
},
{
"DOI": "10.1016/j.cmet.2021.05.013",
"author": "CT Wu",
"doi-asserted-by": "crossref",
"first-page": "1565",
"issue": "8",
"journal-title": "Cell Metab",
"key": "4791_CR47",
"unstructured": "Wu CT, Lidsky PV, Xiao Y, Lee IT, Cheng R, Nakayama T, et al. SARS-CoV-2 infects human pancreatic β cells and elicits β cell impairment. Cell Metab. 2021;33(8):1565-1576.e5.",
"volume": "33",
"year": "2021"
},
{
"author": "A Extance",
"journal-title": "BMJ",
"key": "4791_CR48",
"unstructured": "Extance A. COVID-19: what is the evidence for the antiviral Paxlovid? BMJ. 2022;377:o1037.",
"volume": "377",
"year": "2022"
},
{
"author": "A Extance",
"journal-title": "BMJ",
"key": "4791_CR49",
"unstructured": "Extance A. COVID-19: what is the evidence for the antiviral molnupiravir? BMJ. 2022;377:o926.",
"volume": "377",
"year": "2022"
},
{
"DOI": "10.1016/S1473-3099(23)00493-0",
"author": "WHK Schilling",
"doi-asserted-by": "crossref",
"first-page": "36",
"issue": "1",
"journal-title": "Lancet Infect Dis",
"key": "4791_CR50",
"unstructured": "Schilling WHK, Jittamala P, Watson JA, Boyd S, Luvira V, Siripoon T, et al. Antiviral efficacy of molnupiravir versus ritonavir-boosted nirmatrelvir in patients with early symptomatic COVID-19 (PLATCOV): an open-label, phase 2, randomised, controlled, adaptive trial. Lancet Infect Dis. 2024;24(1):36–45.",
"volume": "24",
"year": "2024"
},
{
"author": "Z Guo",
"issue": "3",
"journal-title": "J Med Virol",
"key": "4791_CR51",
"unstructured": "Guo Z, Zhao S, Mok CKP, So RTY, Yam CHK, Chow TY, et al. Comparing the incubation period, serial interval, and infectiousness profile between SARS-CoV-2 omicron and delta variants. J Med Virol. 2023;95(3):e28648.",
"volume": "95",
"year": "2023"
},
{
"author": "EK Yeoh",
"journal-title": "One Health",
"key": "4791_CR52",
"unstructured": "Yeoh EK, Chong KC, Chiew CJ, Lee VJ, Ng CW, Hashimoto H, et al. Assessing the impact of non-pharmaceutical interventions on the transmissibility and severity of COVID-19 during the first five months in the Western Pacific Region. One Health. 2021;12:100213.",
"volume": "12",
"year": "2021"
},
{
"DOI": "10.7189/jogh.13.06017",
"author": "KC Chong",
"doi-asserted-by": "crossref",
"first-page": "06017",
"journal-title": "J Glob Health",
"key": "4791_CR53",
"unstructured": "Chong KC, Chan PK, Hung CT, Wong CK, Xiong X, Wei Y, et al. Changes in all-cause and cause-specific excess mortality before and after the omicron outbreak of COVID-19 in Hong Kong. J Glob Health. 2023;13:06017.",
"volume": "13",
"year": "2023"
},
{
"author": "K Li",
"issue": "101554",
"journal-title": "Lancet Reg Health West Pac",
"key": "4791_CR54",
"unstructured": "Li K, Wei Y, Hung CT, Wong CKH, Xiong X, Chan PKS, et al. Post-pandemic excess mortality of COVID-19 in Hong Kong: a retrospective study. Lancet Reg Health West Pac. 2025;58(101554):101554.",
"volume": "58",
"year": "2025"
},
{
"DOI": "10.1038/s41467-025-61737-7",
"author": "KC Chong",
"doi-asserted-by": "crossref",
"first-page": "6924",
"issue": "1",
"journal-title": "Nat Commun",
"key": "4791_CR55",
"unstructured": "Chong KC, Wei Y, Jia KM, Boyer C, Lin G, Wang H, et al. SARS-CoV-2 rebound and post-acute mortality and hospitalization among patients admitted with COVID-19: cohort study. Nat Commun. 2025;16(1):6924.",
"volume": "16",
"year": "2025"
}
],
"reference-count": 55,
"references-count": 55,
"relation": {},
"resource": {
"primary": {
"URL": "https://link.springer.com/10.1186/s12916-026-04791-2"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "Effectiveness of nirmatrelvir/ritonavir and molnupiravir on post-COVID diabetes risk among an older adult cohort: a target trial emulation study",
"type": "journal-article",
"update-policy": "https://doi.org/10.1007/springer_crossmark_policy"
}