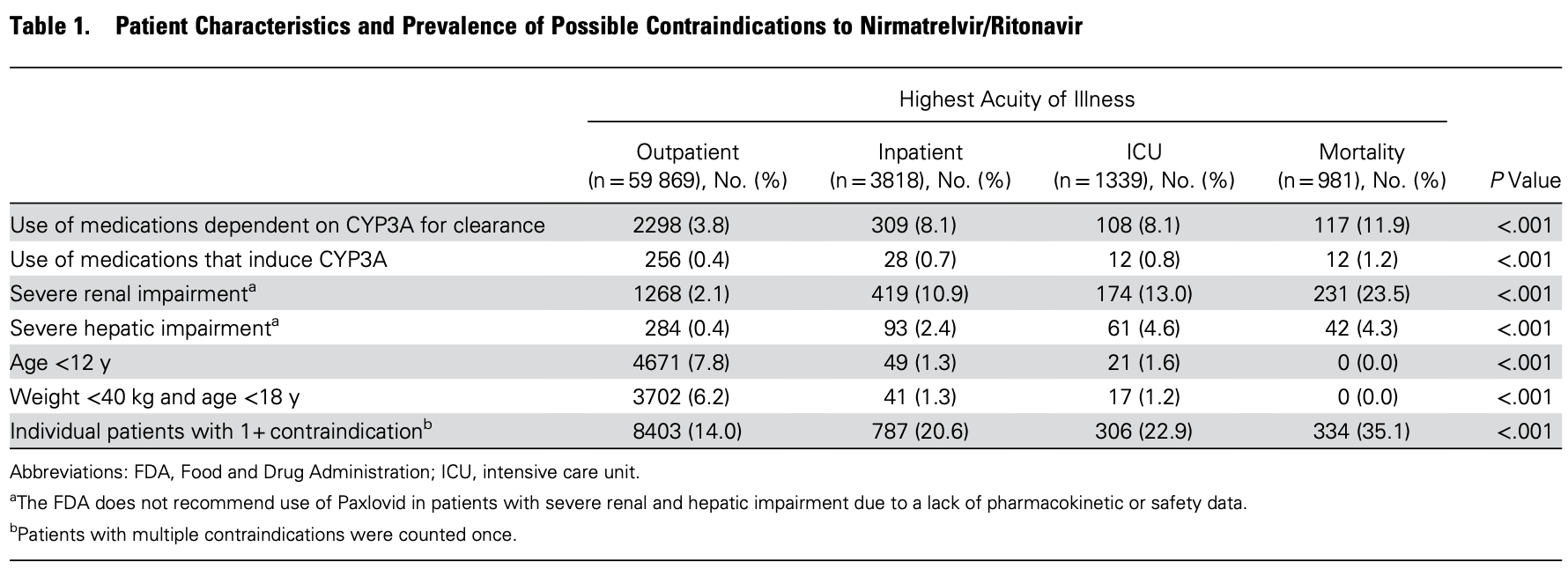
Prevalence of Medical Contraindications to Nirmatrelvir/Ritonavir in a Cohort of Hospitalized and Nonhospitalized Patients With COVID-19
et al., Open Forum Infectious Diseases, doi:10.1093/ofid/ofac389, Aug 2022
Analysis of 66,007 hospitalized and non-hospitalized patients showing a 14% prevalence of medical contraindications with nirmatrelvir/ritonavir for outpatients, 21% for inpatients, and 35% for patients that died.
Resistance. Variants may be resistant to paxlovid1-9. Use may promote the emergence of variants that weaken host immunity and potentially contribute to long COVID10. Confounding by contraindication. Hoertel et al. find that over 50% of patients that died had a contraindication for the use of Paxlovid11. Retrospective studies that do not exclude contraindicated patients may significantly overestimate efficacy. Black box warning. The FDA notes that severe, life-threatening, and/or fatal adverse reactions due to drug interactions have been reported in patients treated with paxlovid12. Kidney and liver injury. Studies show significantly increased risk of acute kidney injury13 and liver injury14,15. Viral rebound. Studies show significantly increased risk of replication-competent viral rebound16-18.
1.
Zhou et al., Nirmatrelvir-resistant SARS-CoV-2 variants with high fitness in an infectious cell culture system, Science Advances, doi:10.1126/sciadv.add7197.
2.
Moghadasi et al., Rapid resistance profiling of SARS-CoV-2 protease inhibitors, npj Antimicrobials and Resistance, doi:10.1038/s44259-023-00009-0.
3.
Jochmans et al., The Substitutions L50F, E166A, and L167F in SARS-CoV-2 3CLpro Are Selected by a Protease Inhibitor In Vitro and Confer Resistance To Nirmatrelvir, mBio, doi:10.1128/mbio.02815-22.
4.
Lopez et al., SARS-CoV-2 Resistance to Small Molecule Inhibitors, Current Clinical Microbiology Reports, doi:10.1007/s40588-024-00229-6.
5.
Zvornicanin et al., Molecular Mechanisms of Drug Resistance and Compensation in SARS-CoV-2 Main Protease: The Interplay Between E166 and L50, bioRxiv, doi:10.1101/2025.01.24.634813.
6.
Vukovikj et al., Impact of SARS-CoV-2 variant mutations on susceptibility to monoclonal antibodies and antiviral drugs: a non-systematic review, April 2022 to October 2024, Eurosurveillance, doi:10.2807/1560-7917.ES.2025.30.10.2400252.
7.
Deschenes et al., Functional and structural characterization of treatment-emergent nirmatrelvir resistance mutations at low frequencies in the main protease (Mpro) reveals a unique evolutionary route for SARS-CoV-2 to gain resistance, The Journal of Infectious Diseases, doi:10.1093/infdis/jiaf294.
8.
Zhou (B) et al., SARS-CoV-2 Mpro inhibitor ensitrelvir: asymmetrical cross-resistance with nirmatrelvir and emerging resistance hotspots, Emerging Microbes & Infections, doi:10.1080/22221751.2025.2552716.
9.
Chen et al., SARS-CoV-2 3CLpro mutations T21I and E166A confer differential resistance to simnotrelvir, bofutrelvir, and ensitrelvir, Journal of Virology, doi:10.1128/jvi.02223-25.
10.
Thomas et al., Nirmatrelvir-Resistant Mutations in SARS-CoV-2 Mpro Enhance Host Immune Evasion via Cleavage of NF-κB Essential Modulator, bioRxiv, doi:10.1101/2024.10.18.619137.
11.
Hoertel et al., Prevalence of Contraindications to Nirmatrelvir-Ritonavir Among Hospitalized Patients With COVID-19 at Risk for Progression to Severe Disease, JAMA Network Open, doi:10.1001/jamanetworkopen.2022.42140.
12.
FDA, Fact sheet for healthcare providers: emergency use authorization for paxlovid, www.fda.gov/media/155050/download.
13.
Kamo et al., Association of Antiviral Drugs for the Treatment of COVID-19 With Acute Renal Failure, In Vivo, doi:10.21873/invivo.13637.
14.
Wang et al., Development and validation of a nomogram to assess the occurrence of liver dysfunction in patients with COVID-19 pneumonia in the ICU, BMC Infectious Diseases, doi:10.1186/s12879-025-10684-1.
15.
Siby et al., Temporal Trends in Serious Adverse Events Associated with Oral Antivirals During the COVID-19 Pandemic: Insights from the FAERS Database (2020–2023), Open Forum Infectious Diseases, doi:10.1093/ofid/ofaf695.1825.
16.
Edelstein et al., SARS-CoV-2 virologic rebound with nirmatrelvir-ritonavir therapy, medRxiv, doi:10.1101/2023.06.23.23288598.
Lim et al., 1 Aug 2022, retrospective, USA, peer-reviewed, 5 authors, study period 4 March, 2020 - 22 March, 2022.
Contact: sarah.lim@state.mn.us, permissions@oup.com.
Abstract: Open Forum Infectious Diseases
BRIEF REPORT
Prevalence of Medical
Contraindications to Nirmatrelvir/
Ritonavir in a Cohort of Hospitalized
and Nonhospitalized Patients With
COVID-19
Sarah Lim,1 Christopher J. Tignanelli,2, Nicolas Hoertel,3, David R. Boulware,4,
and Michael G. Usher4
1
This analysis describes the prevalence of contraindications
to nirmatrelvir/ritonavir among 66 007 patients with
coronavirus disease 2019 in a large health care system. A
possible contradiction was present in 9830 patients (14.8%),
with the prevalence of contraindications increasing with
higher acuity of illness.
Keywords. nirmatrelvir/ritonavir; COVID-19; contraindications; Palovid.
The authorization of 2 novel oral antiviral therapies for severe
acute respiratory syndrome coronavirus 2 (SARS-CoV-2) by
the US Food and Drug Administration (FDA) in December
2021, nirmaltrelvir/ritonavir and molnupiravir [1, 2], has re
sulted in broader availability of effective coronavirus disease
2019 (COVID-19) early treatments in high-income countries
and has spurred new initiatives for improved access, such as
the Test to Treat program [3]. Other therapeutics such as
monoclonal antibodies and intravenous remdesivir are avail
able but require skilled health care personnel resources to ad
minister. Of these 2 oral antivirals, nirmaltrelvir/ritonavir
reduced the risk of hospitalization by 89%, as compared with
30% with molnupiravir [4, 5], and is the preferred therapy
for mild to moderate COVID-19 in nonhospitalized high-risk
adults [6].
Received 19 May 2022; editorial decision 28 July 2022; accepted 01 August 2022; published
online 3 August 2022
Correspondence: S. Lim, MBBCh, MPH, Minnesota Department of Health, 625 North Robert
Street, St. Paul, MN 55155 (sarah.lim@state.mn.us).
Open Forum Infectious Diseases®
© The Author(s) 2022. Published by Oxford University Press on behalf of Infectious Diseases
Society of America. This is an Open Access article distributed under the terms of the
Creative Commons Attribution-NonCommercial-NoDerivs licence (https://creativecommons.
org/licenses/by-nc-nd/4.0/), which permits non-commercial reproduction and distribution of
the work, in any medium, provided the original work is not altered or transformed in any
way, and that the work is properly cited. For commercial re-use, please contact journals.
permissions@oup.com
https://doi.org/10.1093/ofid/ofac389
METHODS
We conducted a retrospective review of all patients evaluated
for a diagnosis of COVID-19 in an integrated health system
consisting of 11 hospitals and 55 clinics, between March 4,
2020, and March 22, 2022. This health system provides care
for ∼20% of the population of Minnesota. Approval was ob
tained from the institutional review board at the University
BRIEF REPORT • OFID • 1
Minnesota Department of Health, St. Paul, Minnesota, USA, 2Department of Surgery,
University of Minnesota, Minneapolis, Minnesota, USA, 3Université Paris Cité, AP-HP, Hôpital
Corentin-Celton, DMU Psychiatrie et Addictologie, INSERM U1266, Institut de Psychiatrie et
Neuroscience de Paris, Paris, France, and 4Department of Medicine, University of Minnesota,
Minneapolis, Minnesota, USA
However, nirmaltrelvir/ritonavir is not suitable for all
patients, including many who are at high risk for severe illness.
Ritonavir strongly inhibits cytochrome P450 (CYP) 3A4 me
tabolism, which results in..
DOI record:
{
"DOI": "10.1093/ofid/ofac389",
"ISSN": [
"2328-8957"
],
"URL": "http://dx.doi.org/10.1093/ofid/ofac389",
"abstract": "<jats:title>Abstract</jats:title>\n <jats:p>This analysis describes the prevalence of contraindications to nirmatrelvir/ritonavir among 66 007 patients with coronavirus disease 2019 in a large health care system. A possible contradiction was present in 9830 patients (14.8%), with the prevalence of contraindications increasing with higher acuity of illness.</jats:p>",
"article-number": "ofac389",
"author": [
{
"affiliation": [
{
"name": "Minnesota Department of Health , St. Paul, Minnesota , USA"
}
],
"family": "Lim",
"given": "Sarah",
"sequence": "first"
},
{
"ORCID": "https://orcid.org/0000-0002-8079-5565",
"affiliation": [
{
"name": "Department of Surgery, University of Minnesota , Minneapolis, Minnesota , USA"
}
],
"authenticated-orcid": false,
"family": "Tignanelli",
"given": "Christopher J",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0002-7890-1349",
"affiliation": [
{
"name": "Université Paris Cité, AP-HP, Hôpital Corentin-Celton, DMU Psychiatrie et Addictologie, INSERM U1266, Institut de Psychiatrie et Neuroscience de Paris , Paris , France"
}
],
"authenticated-orcid": false,
"family": "Hoertel",
"given": "Nicolas",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0002-4715-0060",
"affiliation": [
{
"name": "Department of Medicine, University of Minnesota , Minneapolis, Minnesota , USA"
}
],
"authenticated-orcid": false,
"family": "Boulware",
"given": "David R",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Medicine, University of Minnesota , Minneapolis, Minnesota , USA"
}
],
"family": "Usher",
"given": "Michael G",
"sequence": "additional"
}
],
"container-title": "Open Forum Infectious Diseases",
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2022,
8,
3
]
],
"date-time": "2022-08-03T19:06:47Z",
"timestamp": 1659553607000
},
"deposited": {
"date-parts": [
[
2022,
8,
22
]
],
"date-time": "2022-08-22T17:18:58Z",
"timestamp": 1661188738000
},
"indexed": {
"date-parts": [
[
2026,
1,
8
]
],
"date-time": "2026-01-08T05:15:31Z",
"timestamp": 1767849331274,
"version": "3.49.0"
},
"is-referenced-by-count": 23,
"issue": "8",
"issued": {
"date-parts": [
[
2022,
8,
1
]
]
},
"journal-issue": {
"issue": "8",
"published-print": {
"date-parts": [
[
2022,
8,
2
]
]
}
},
"language": "en",
"license": [
{
"URL": "https://creativecommons.org/licenses/by-nc-nd/4.0/",
"content-version": "vor",
"delay-in-days": 2,
"start": {
"date-parts": [
[
2022,
8,
3
]
],
"date-time": "2022-08-03T00:00:00Z",
"timestamp": 1659484800000
}
}
],
"link": [
{
"URL": "https://academic.oup.com/ofid/advance-article-pdf/doi/10.1093/ofid/ofac389/45228963/ofac389.pdf",
"content-type": "application/pdf",
"content-version": "am",
"intended-application": "syndication"
},
{
"URL": "https://academic.oup.com/ofid/article-pdf/9/8/ofac389/45495742/ofac389.pdf",
"content-type": "application/pdf",
"content-version": "vor",
"intended-application": "syndication"
},
{
"URL": "https://academic.oup.com/ofid/article-pdf/9/8/ofac389/45495742/ofac389.pdf",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "286",
"original-title": [],
"prefix": "10.1093",
"published": {
"date-parts": [
[
2022,
8,
1
]
]
},
"published-online": {
"date-parts": [
[
2022,
8,
3
]
]
},
"published-other": {
"date-parts": [
[
2022,
8,
1
]
]
},
"published-print": {
"date-parts": [
[
2022,
8,
2
]
]
},
"publisher": "Oxford University Press (OUP)",
"reference": [
{
"article-title": "Paxlovid Emergency Use Authorization 105 approval letter",
"author": "US Food and Drug Administration",
"key": "2022082217181244700_ofac389-B1",
"year": "2021"
},
{
"article-title": "Molnupiravir Emergency Use Authorization 105 approval letter",
"author": "US Food and Drug Administration",
"key": "2022082217181244700_ofac389-B2"
},
{
"article-title": "HHS/ASPR fact sheet: COVID-19 Test to Treat",
"author": "Department of Health and Human Services",
"key": "2022082217181244700_ofac389-B3",
"year": "2022"
},
{
"article-title": "Fact sheet for health care providers: Emergency Use Authorization for Paxlovid",
"author": "US Food and Drug Administration",
"key": "2022082217181244700_ofac389-B4",
"year": "2021"
},
{
"article-title": "Fact sheet for health care providers: Emergency Use Authorization for Molnupiravir",
"author": "US Food and Drug Administration",
"key": "2022082217181244700_ofac389-B5",
"year": "2021"
},
{
"article-title": "Coronavirus disease 2019 (COVID-19) treatment guidelines",
"author": "National Institutes of Health, COVID-19 Treatment Guidelines Panel",
"key": "2022082217181244700_ofac389-B6"
},
{
"DOI": "10.2165/00003088-199835040-00002",
"article-title": "Ritonavir",
"author": "Hsu",
"doi-asserted-by": "crossref",
"first-page": "275",
"journal-title": "Clin Pharmacokinet",
"key": "2022082217181244700_ofac389-B7",
"volume": "35",
"year": "1998"
},
{
"article-title": "Underlying medical conditions associated with higher risk for severe COVID-19: information for healthcare professionals",
"author": "Centers for Disease Control and Prevention",
"key": "2022082217181244700_ofac389-B8"
},
{
"article-title": "Maximizing the benefit of COVID-19 therapeutics: considerations for state public health officials. ASTHO and Duke Margolis Center for Health Policy Brief",
"author": "Greene",
"key": "2022082217181244700_ofac389-B9"
},
{
"DOI": "10.1053/j.ajkd.2010.02.337",
"article-title": "Estimating GFR using the CKD epidemiology collaboration (CKD-EPI) creatinine equation: more accurate GFR estimates, lower CKD prevalence estimates, and better risk predictions",
"author": "Levey",
"doi-asserted-by": "crossref",
"first-page": "622",
"journal-title": "Am J Kidney Dis",
"key": "2022082217181244700_ofac389-B10",
"volume": "55",
"year": "2010"
},
{
"DOI": "10.1056/NEJMoa2116846",
"article-title": "Early remdesivir to prevent progression to severe COVID-19 in outpatients",
"author": "Gottlieb",
"doi-asserted-by": "crossref",
"first-page": "305",
"journal-title": "N Engl J Med",
"key": "2022082217181244700_ofac389-B11",
"volume": "386",
"year": "2022"
},
{
"article-title": "Fact sheet for health care providers: Emergency Use Authorization for Bebtelovimab",
"author": "US Food and Drug Administration",
"key": "2022082217181244700_ofac389-B12",
"year": "2021"
},
{
"DOI": "10.3390/vaccines9080914",
"article-title": "Effect of SARS-CoV-2 mutations on the efficacy of antibody therapy and response to vaccines",
"author": "Yaqinuddin",
"doi-asserted-by": "crossref",
"first-page": "914",
"journal-title": "Vaccines (Basel)",
"key": "2022082217181244700_ofac389-B13",
"volume": "9",
"year": "2021"
},
{
"article-title": "FDA approves first COVID-19 treatment for young children. FDA News Release",
"author": "US Food and Drug Administration",
"key": "2022082217181244700_ofac389-B14",
"year": "2022"
}
],
"reference-count": 14,
"references-count": 14,
"relation": {},
"resource": {
"primary": {
"URL": "https://academic.oup.com/ofid/article/doi/10.1093/ofid/ofac389/6654829"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "Prevalence of Medical Contraindications to Nirmatrelvir/Ritonavir in a Cohort of Hospitalized and Nonhospitalized Patients With COVID-19",
"type": "journal-article",
"volume": "9"
}