Evaluating the effectiveness and safety of Azvudine for hospitalised patients with COVID-19 and hypertension: a multicenter retrospective cohort study
et al., Scientific Reports, doi:10.1038/s41598-025-34514-1, NCT06349655, Feb 2026
Azvudine for COVID-19
48th treatment shown to reduce risk in
January 2023, now with p = 0.0000000021 from 42 studies.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
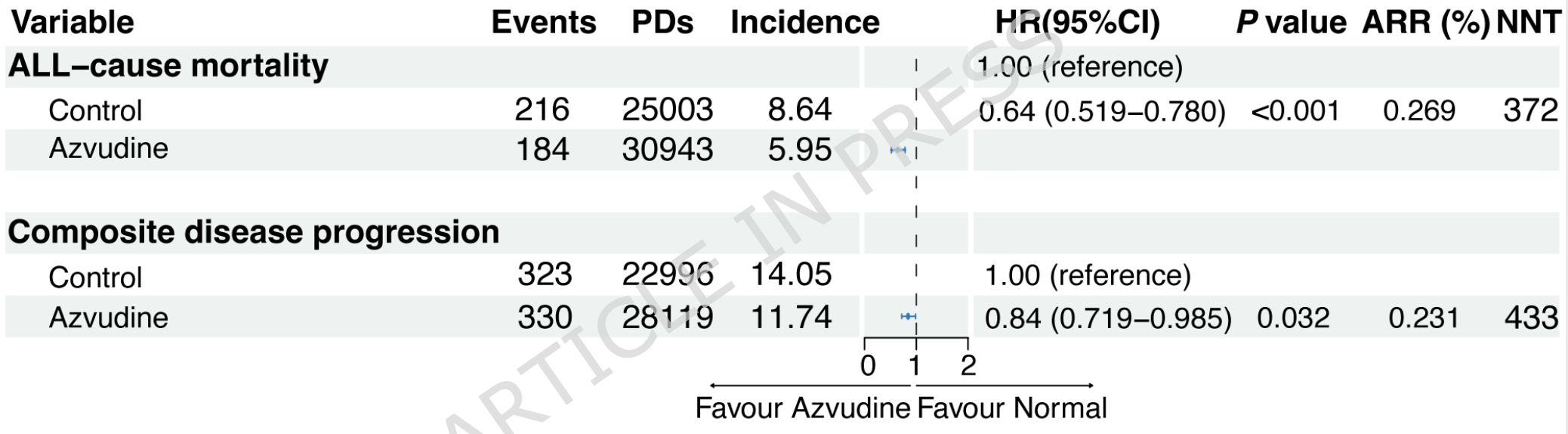
PSM retrospective 4,868 hospitalized COVID-19 patients with hypertension showing reduced mortality with azvudine.
For composite disease progression, the azvudine group had more raw events (330) than the control group (320) out of identical starting populations (2434 per group). Yet, the study reports a significantly protective Hazard Ratio (0.84). The data suggests a large differential in person-days driven by informative censoring (e.g., early discharge of control patients), biasing the Cox model. The person-days calculated for the composite progression endpoint drastically differ between groups (approx 28,100 for azvudine vs 22,700 for control) despite starting with identical sample sizes and following a 30-day max horizon. This suggests control patients were systematically censored earlier, creating a survival bias in the time-to-event analysis. Authors also acknowledge that the composite disease progression finding was not robust across sensitivity analyses.
The mortality result should have higher reliability, with consistent results across all five sensitivity analyses - probabilistic PSM, mean imputation, excluding first-day discharges, doubly robust estimation, and IPTW. However, differential censoring likely affects the results here as well. The raw counts in the PSM groups shows a more modest ~15% benefit.
In Table 3, the reported percentages do not match the counts. For example, serum phosphorus in the control group reports 74 cases out of 2223 (which is 3.3%) but shows 23%. Table 3 reports 70 Grade >= 3 events for hypertriglyceridemia in the control group and 100 in the azvudine group, however these are much larger than the number of events for all grades. Table 3 lists available data for hypertriglyceridemia as control = 2465 and azvudine = 2454. These numbers exceed the post-matching cohort size of 2434 patients per group.
The text states the mortality rate in the control group was 8.79% (n = 214), but later claims there were 216 cases.
4 preclinical studies support the efficacy of azvudine for COVID-19:
Standard of Care (SOC) for COVID-19 in the study country,
China, is poor with low average efficacy for approved treatments5.
This study is excluded in the after exclusion results of meta-analysis:
potential significant differential censoring.
|
risk of death, 36.0% lower, HR 0.64, p < 0.001, treatment 2,434, control 2,434, propensity score matching.
|
|
risk of progression, 16.0% lower, HR 0.84, p = 0.03, treatment 2,434, control 2,434, propensity score matching.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
1.
Xiong et al., Real-world data of Azvudine-induced hepatotoxicity among hospitalized COVID-19 patients in China: a retrospective case-control study, Frontiers in Pharmacology, doi:10.3389/fphar.2025.1558054.
2.
Wang et al., Development and validation of a nomogram to assess the occurrence of liver dysfunction in patients with COVID-19 pneumonia in the ICU, BMC Infectious Diseases, doi:10.1186/s12879-025-10684-1.
3.
Li et al., Azvudine alleviates SARS-CoV-2-induced inflammation by targeting myeloperoxidase in NETosis, Chinese Chemical Letters, doi:10.1016/j.cclet.2024.110238.
Chen et al., 26 Feb 2026, retrospective, China, peer-reviewed, 15 authors, study period December 2022 - January 2023, trial NCT06349655 (history).
Contact: 20390@sias.edu.cn.
Evaluating the effectiveness and safety of Azvudine for hospitalised patients with COVID-19 and hypertension: a multicenter retrospective cohort study
Scientific Reports, doi:10.1038/s41598-025-34514-1
Hypertension is widely acknowledged as a major risk factor for disease severity and death in patients with coronavirus disease 2019 (COVID-19). Azvudine is recommended for COVID-19 patients in China. However, its clinical efficacy and safety for individuals with hypertension remain unclear. This nine-center retrospective cohort study included 32864 hospitalized COVID-19 patients in Henan Province, China, from December 2022 to January 2023. Among these patients, those with hypertension were identified and divided into the Azvudine and control groups (standard treatment without antiviral medication) after propensity score matching (PSM) at a 1:1 ratio. The primary outcomes measured were all-cause mortality and composite disease progression. Subgroup analyses and sensitivity tests were conducted to verify the robustness of the results. Safety was assessed based on adverse events (AEs). After PSM to balance baseline characteristics, the analysis included 2,434 Azvudine recipients and 2,434 controls, forming a final matched cohort. Azvudine was associated with a lower risk of all-cause mortality (HR: 0.64, 95% CI: 0.519-0.780; P < 0.001) and composite disease progression (HR: 0.84, 95% CI: 0.719-0.985; P = 0.032) in hypertensive patients with COVID-19. In five sensitivity analyses, Azvudine showed a highly robust effect in reducing allcause mortality, while the evidence for a reduction in the progression of composite disease progression was less consistent. No significant difference in severe AEs (≥ Grade 3) was observed between groups. These real-world findings suggest Azvudine may be a promising antiviral option for hypertensive COVID-19 patients, but further prospective trials are necessary to confirm these results.
References
Bernal, Molnupiravir for Oral Treatment of Covid-19 in Nonhospitalized Patients, The New England journal of medicine, doi:10.1056/NEJMoa2116044
Cascella, Rajnik, Aleem, Dulebohn, Napoli, None
Hammond, Oral Nirmatrelvir for High-Risk, Nonhospitalized Adults with Covid-19, The New England journal of medicine, doi:10.1056/NEJMoa2118542
Hayden, The Infectious Diseases Society of America Guidelines on the Diagnosis of COVID-19: Antigen Testing (January 2023), Clin Infect Dis, doi:10.1093/cid/ciad032
Who, WHO COVID-19 dashboard. Coronavirus biology and replication: implications for SARS-CoV-2, Nat Rev Microbiol, doi:10.1038/s41579-020-00468-6
Yu, Chang, The first Chinese oral anti-COVID-19 drug Azvudine launched, Innovation, doi:10.1016/j.xinn.2022.100321
DOI record:
{
"DOI": "10.1038/s41598-025-34514-1",
"ISSN": [
"2045-2322"
],
"URL": "http://dx.doi.org/10.1038/s41598-025-34514-1",
"alternative-id": [
"34514"
],
"assertion": [
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "Received",
"name": "received",
"order": 1,
"value": "10 July 2025"
},
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "Accepted",
"name": "accepted",
"order": 2,
"value": "29 December 2025"
},
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "First Online",
"name": "first_online",
"order": 3,
"value": "26 February 2026"
},
{
"group": {
"label": "Declarations",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 1
},
{
"group": {
"label": "Competing interests",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 2,
"value": "The authors declare no competing interests."
},
{
"group": {
"label": "Ethics approval and consent to participate",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 3,
"value": "This study was reviewed and approved by the Institutional Review Board of The First Affiliated Hospital of Zhengzhou University (2023-KY-0865-001). This study is retrospective, and no patient privacy information is exposed. Informed consent from the subject was not obtained."
}
],
"author": [
{
"affiliation": [],
"family": "Chen",
"given": "Yu",
"sequence": "first"
},
{
"affiliation": [],
"family": "Li",
"given": "Huan",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ma",
"given": "Yichen",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Wang",
"given": "Ling",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Qian",
"given": "Guowu",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Li",
"given": "Silin",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Luo",
"given": "Hong",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Zhang",
"given": "Shixi",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Li",
"given": "Guangming",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Zhang",
"given": "Donghua",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Li",
"given": "Guotao",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Zheng",
"given": "Yun",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Bai",
"given": "Qin",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Wang",
"given": "Haiyu",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ren",
"given": "Zhigang",
"sequence": "additional"
}
],
"container-title": "Scientific Reports",
"container-title-short": "Sci Rep",
"content-domain": {
"crossmark-restriction": false,
"domain": [
"link.springer.com"
]
},
"created": {
"date-parts": [
[
2026,
2,
26
]
],
"date-time": "2026-02-26T11:25:16Z",
"timestamp": 1772105116000
},
"deposited": {
"date-parts": [
[
2026,
2,
26
]
],
"date-time": "2026-02-26T11:25:18Z",
"timestamp": 1772105118000
},
"funder": [
{
"award": [
"232102311084"
],
"award-info": [
{
"award-number": [
"232102311084"
]
}
],
"name": "the Medical Science and Technique Foundation of Henan Province, China"
},
{
"award": [
"242102310046"
],
"award-info": [
{
"award-number": [
"242102310046"
]
}
],
"name": "the Medical Science and Technique Foundation of Henan Province, China"
},
{
"award": [
"24B320034"
],
"award-info": [
{
"award-number": [
"24B320034"
]
}
],
"name": "Key Scientific Research Projects of Colleges and Universities in Henan Province"
},
{
"award": [
"2025ZD01906305"
],
"award-info": [
{
"award-number": [
"2025ZD01906305"
]
}
],
"name": "Prevention and Control of Emerging and Major Infectious Diseases-National Science and Technology Major Project"
},
{
"award": [
"ZYYC202301ZD"
],
"award-info": [
{
"award-number": [
"ZYYC202301ZD"
]
}
],
"name": "Henan Zhongyuan Medical Science and Technology Innovation and Development Foundation"
},
{
"award": [
"232300421124"
],
"award-info": [
{
"award-number": [
"232300421124"
]
}
],
"name": "the Natural Science Foundation Key Project of Henan Province"
}
],
"indexed": {
"date-parts": [
[
2026,
2,
26
]
],
"date-time": "2026-02-26T12:09:45Z",
"timestamp": 1772107785734,
"version": "3.50.1"
},
"is-referenced-by-count": 0,
"issued": {
"date-parts": [
[
2026,
2,
26
]
]
},
"language": "en",
"license": [
{
"URL": "https://creativecommons.org/licenses/by-nc-nd/4.0",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
2,
26
]
],
"date-time": "2026-02-26T00:00:00Z",
"timestamp": 1772064000000
}
},
{
"URL": "https://creativecommons.org/licenses/by-nc-nd/4.0",
"content-version": "am",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
2,
26
]
],
"date-time": "2026-02-26T00:00:00Z",
"timestamp": 1772064000000
}
}
],
"link": [
{
"URL": "https://www.nature.com/articles/s41598-025-34514-1",
"content-type": "text/html",
"content-version": "vor",
"intended-application": "text-mining"
}
],
"member": "297",
"original-title": [],
"prefix": "10.1038",
"published": {
"date-parts": [
[
2026,
2,
26
]
]
},
"published-online": {
"date-parts": [
[
2026,
2,
26
]
]
},
"publisher": "Springer Science and Business Media LLC",
"reference": [
{
"key": "34514_CR1",
"unstructured": "WHO. WHO COVID-19 dashboard. https://data.who.int/dashboards/covid19/cases?n=c (2024)."
},
{
"DOI": "10.1136/bmj.m1985",
"author": "AB Docherty",
"doi-asserted-by": "publisher",
"journal-title": "BMJ",
"key": "34514_CR2",
"unstructured": "Docherty, A. B. et al. Features of 20 133 UK patients in hospital with covid-19 using the ISARIC WHO clinical characterisation protocol: Prospective observational cohort study. BMJ 369, m1985. https://doi.org/10.1136/bmj.m1985 (2020).",
"volume": "369",
"year": "2020"
},
{
"DOI": "10.1016/s0140-6736(20)30566-3",
"author": "F Zhou",
"doi-asserted-by": "publisher",
"first-page": "1054",
"journal-title": "Lancet",
"key": "34514_CR3",
"unstructured": "Zhou, F. et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 395, 1054–1062. https://doi.org/10.1016/s0140-6736(20)30566-3 (2020).",
"volume": "395",
"year": "2020"
},
{
"DOI": "10.1038/s41421-020-00235-0",
"author": "LK Zhang",
"doi-asserted-by": "publisher",
"first-page": "96",
"journal-title": "Cell. Discov.",
"key": "34514_CR4",
"unstructured": "Zhang, L. K. et al. Calcium channel blocker amlodipine besylate therapy is associated with reduced case fatality rate of COVID-19 patients with hypertension. Cell. Discov. 6, 96. https://doi.org/10.1038/s41421-020-00235-0 (2020).",
"volume": "6",
"year": "2020"
},
{
"DOI": "10.1001/jama.2016.19043",
"author": "MH Forouzanfar",
"doi-asserted-by": "publisher",
"first-page": "165",
"journal-title": "JAMA",
"key": "34514_CR5",
"unstructured": "Forouzanfar, M. H. et al. Global burden of hypertension and systolic blood pressure of at least 110 to 115 mm Hg, 1990–2015. JAMA 317, 165–182. https://doi.org/10.1001/jama.2016.19043 (2017).",
"volume": "317",
"year": "2017"
},
{
"DOI": "10.1080/07853890.2021.1931957",
"author": "L Zhong",
"doi-asserted-by": "publisher",
"first-page": "770",
"journal-title": "Ann. Med.",
"key": "34514_CR6",
"unstructured": "Zhong, L. et al. Effects of hypertension on the outcomes of COVID-19: a multicentre retrospective cohort study. Ann. Med. 53, 770–776. https://doi.org/10.1080/07853890.2021.1931957 (2021).",
"volume": "53",
"year": "2021"
},
{
"DOI": "10.1038/s41440-020-0485-2",
"author": "S Huang",
"doi-asserted-by": "publisher",
"first-page": "824",
"journal-title": "Hypertens. Res.",
"key": "34514_CR7",
"unstructured": "Huang, S. et al. COVID-19 patients with hypertension have more severe disease: A multicenter retrospective observational study. Hypertens. Res. 43, 824–831. https://doi.org/10.1038/s41440-020-0485-2 (2020).",
"volume": "43",
"year": "2020"
},
{
"DOI": "10.1080/10641963.2022.2071914",
"author": "Z Qian",
"doi-asserted-by": "publisher",
"first-page": "451",
"journal-title": "Clin. Exp. Hypertens.",
"key": "34514_CR8",
"unstructured": "Qian, Z. et al. Association between hypertension and prognosis of patients with COVID-19: A systematic review and meta-analysis. Clin. Exp. Hypertens. 44, 451–458. https://doi.org/10.1080/10641963.2022.2071914 (2022).",
"volume": "44",
"year": "2022"
},
{
"DOI": "10.1038/s41579-020-00468-6",
"author": "P V’Kovski",
"doi-asserted-by": "publisher",
"first-page": "155",
"journal-title": "Nat. Rev. Microbiol.",
"key": "34514_CR9",
"unstructured": "V’Kovski, P., Kratzel, A., Steiner, S., Stalder, H. & Thiel, V. Coronavirus biology and replication: implications for SARS-CoV-2. Nat. Rev. Microbiol. 19, 155–170. https://doi.org/10.1038/s41579-020-00468-6 (2021).",
"volume": "19",
"year": "2021"
},
{
"DOI": "10.1093/cid/ciad032",
"author": "MK Hayden",
"doi-asserted-by": "publisher",
"first-page": "e350",
"journal-title": "Clin. Infect. Dis.",
"key": "34514_CR10",
"unstructured": "Hayden, M. K. et al. The infectious diseases society of America guidelines on the diagnosis of COVID-19: Antigen testing (January 2023). Clin. Infect. Dis. 78, e350–e384. https://doi.org/10.1093/cid/ciad032 (2024).",
"volume": "78",
"year": "2024"
},
{
"DOI": "10.1056/NEJMoa2118542",
"author": "J Hammond",
"doi-asserted-by": "publisher",
"first-page": "1397",
"journal-title": "N. Engl. J. Med.",
"key": "34514_CR11",
"unstructured": "Hammond, J. et al. Oral nirmatrelvir for high-risk, nonhospitalized adults with COVID-19. N. Engl. J. Med. 386, 1397–1408. https://doi.org/10.1056/NEJMoa2118542 (2022).",
"volume": "386",
"year": "2022"
},
{
"DOI": "10.1056/NEJMoa2116044",
"author": "A Jayk Bernal",
"doi-asserted-by": "publisher",
"first-page": "509",
"journal-title": "N. Engl. J. Med.",
"key": "34514_CR12",
"unstructured": "Jayk Bernal, A. et al. Molnupiravir for oral treatment of COVID-19 in nonhospitalized patients. N. Engl. J. Med. 386, 509–520. https://doi.org/10.1056/NEJMoa2116044 (2022).",
"volume": "386",
"year": "2022"
},
{
"DOI": "10.1016/j.xinn.2022.100321",
"author": "B Yu",
"doi-asserted-by": "publisher",
"first-page": "100321",
"journal-title": "Innov. Camb. Mass.",
"key": "34514_CR13",
"unstructured": "Yu, B. & Chang, J. The first Chinese oral anti-COVID-19 drug Azvudine launched. Innov. Camb. Mass. 3, 100321. https://doi.org/10.1016/j.xinn.2022.100321 (2022).",
"volume": "3",
"year": "2022"
},
{
"key": "34514_CR14",
"unstructured": "Cascella, M., Rajnik, M., Aleem, A., Dulebohn, S. C. & Di Napoli, R. In StatPearls (StatPearls Publishing Copyright © 2025, StatPearls Publishing LLC., 2025)"
},
{
"DOI": "10.1016/s0140-6736(22)01586-0",
"author": "CKH Wong",
"doi-asserted-by": "publisher",
"first-page": "1213",
"journal-title": "Lancet",
"key": "34514_CR15",
"unstructured": "Wong, C. K. H. et al. Real-world effectiveness of molnupiravir and nirmatrelvir plus ritonavir against mortality, hospitalisation, and in-hospital outcomes among community-dwelling, ambulatory patients with confirmed SARS-CoV-2 infection during the omicron wave in Hong Kong: an observational study. Lancet 400, 1213–1222. https://doi.org/10.1016/s0140-6736(22)01586-0 (2022).",
"volume": "400",
"year": "2022"
},
{
"DOI": "10.1016/j.lanwpc.2022.100602",
"author": "AK Wai",
"doi-asserted-by": "publisher",
"journal-title": "Lancet Reg. Health West Pac.",
"key": "34514_CR16",
"unstructured": "Wai, A. K. et al. Association of Molnupiravir and Nirmatrelvir-Ritonavir with preventable mortality, hospital admissions and related avoidable healthcare system cost among high-risk patients with mild to moderate COVID-19. Lancet Reg. Health West Pac. 30, 100602. https://doi.org/10.1016/j.lanwpc.2022.100602 (2023).",
"volume": "30",
"year": "2023"
},
{
"DOI": "10.1007/s40121-023-00760-x",
"author": "C Weng",
"doi-asserted-by": "publisher",
"first-page": "649",
"journal-title": "Infect. Dis. Ther.",
"key": "34514_CR17",
"unstructured": "Weng, C. et al. Safety and efficacy of paxlovid against omicron variants of coronavirus disease 2019 in elderly patients. Infect. Dis. Ther. 12, 649–662. https://doi.org/10.1007/s40121-023-00760-x (2023).",
"volume": "12",
"year": "2023"
},
{
"DOI": "10.1038/s41392-021-00835-6",
"author": "JL Zhang",
"doi-asserted-by": "publisher",
"first-page": "414",
"journal-title": "Signal Transduct. Target Ther.",
"key": "34514_CR18",
"unstructured": "Zhang, J. L. et al. Azvudine is a thymus-homing anti-SARS-CoV-2 drug effective in treating COVID-19 patients. Signal Transduct. Target Ther. 6, 414. https://doi.org/10.1038/s41392-021-00835-6 (2021).",
"volume": "6",
"year": "2021"
},
{
"key": "34514_CR19",
"unstructured": "ADMINISTRATION, N. M. P. Domestically developed drug joins virus battle. http://english.nmpa.gov.cn/2022-08/15/c_797867.htm (2022)."
},
{
"DOI": "10.1002/advs.202001435",
"author": "Z Ren",
"doi-asserted-by": "publisher",
"first-page": "e2001435",
"journal-title": "Adv. Sci.",
"key": "34514_CR20",
"unstructured": "Ren, Z. et al. A randomized, open-label, controlled clinical trial of azvudine tablets in the treatment of mild and common COVID-19, a pilot study. Adv. Sci. 7, e2001435. https://doi.org/10.1002/advs.202001435 (2020).",
"volume": "7",
"year": "2020"
},
{
"DOI": "10.1038/s41392-025-02126-w",
"author": "H Wang",
"doi-asserted-by": "publisher",
"first-page": "30",
"journal-title": "Signal Transduct. Target. Ther.",
"key": "34514_CR21",
"unstructured": "Wang, H. et al. Real-world effectiveness and safety of oral azvudine versus nirmatrelvir-ritonavir (Paxlovid) in hospitalized patients with COVID-19: A multicenter, retrospective, cohort study. Signal Transduct. Target. Ther. 10, 30. https://doi.org/10.1038/s41392-025-02126-w (2025).",
"volume": "10",
"year": "2025"
},
{
"DOI": "10.1002/advs.202404450",
"author": "R Sun",
"doi-asserted-by": "publisher",
"first-page": "e2404450",
"journal-title": "Adv. Sci.",
"key": "34514_CR22",
"unstructured": "Sun, R. et al. Effectiveness and safety of oral azvudine for elderly hospitalized patients with COVID-19: A multicenter, retrospective, real-world study. Adv. Sci. 12, e2404450. https://doi.org/10.1002/advs.202404450 (2025).",
"volume": "12",
"year": "2025"
},
{
"DOI": "10.1016/j.isci.2025.111907",
"author": "G Su",
"doi-asserted-by": "publisher",
"first-page": "111907",
"journal-title": "iScience",
"key": "34514_CR23",
"unstructured": "Su, G. et al. Real-world effectiveness of azvudine versus nirmatrelvir-ritonavir in hospitalized patients with COVID-19 and pre-existing diabetes. iScience 28, 111907. https://doi.org/10.1016/j.isci.2025.111907 (2025).",
"volume": "28",
"year": "2025"
},
{
"key": "34514_CR24",
"unstructured": "China, N. H. C. o. t. P. s. R. o. COVID-19 diagnosis and treatment plan (trial version 9). https://www.gov.cn/zhengce/zhengceku/2022-03/15/5679257/files/49854a49c7004f4ea9e622f3f2c568d8.pdf (2022)."
},
{
"key": "34514_CR25",
"unstructured": "China, N. H. C. o. t. P. s. R. o. COVID-19 Diagnosis and treatment plan (trial version 10). https://www.gov.cn/zhengce/zhengceku/2023-01/06/5735343/files/5844ce04246b431dbd322d8ba10afb48.pdf (2023)."
},
{
"key": "34514_CR26",
"unstructured": "Services, U. S. D. O. H. A. H. Common Terminology Criteria for Adverse Events (CTCAE) Version 5.0. https://ctep.cancer.gov/protocoldevelopment/electronic_applications/docs/ctcae_v5_quick_reference_5x7.pdf (2017)."
},
{
"DOI": "10.1016/j.jinf.2024.106355",
"author": "Z Ren",
"doi-asserted-by": "publisher",
"journal-title": "J. Infect.",
"key": "34514_CR27",
"unstructured": "Ren, Z. et al. Real-world effectiveness and safety of azvudine in hospitalized patients with SARS-CoV-2 infection: A multicenter, retrospective cohort study. J. Infect. 89, 106355. https://doi.org/10.1016/j.jinf.2024.106355 (2024).",
"volume": "89",
"year": "2024"
},
{
"DOI": "10.3389/fendo.2025.1467303",
"author": "Y Zhou",
"doi-asserted-by": "publisher",
"first-page": "1467303",
"journal-title": "Front. Endocrinol.",
"key": "34514_CR28",
"unstructured": "Zhou, Y. et al. A multicenter, real-world cohort study: effectiveness and safety of Azvudine in hospitalized COVID-19 patients with pre-existing diabetes. Front. Endocrinol. 16, 1467303. https://doi.org/10.3389/fendo.2025.1467303 (2025).",
"volume": "16",
"year": "2025"
},
{
"DOI": "10.1002/advs.202405679",
"author": "J Sun",
"doi-asserted-by": "publisher",
"first-page": "e2405679",
"journal-title": "Adv. Sci.",
"key": "34514_CR29",
"unstructured": "Sun, J. et al. The antiviral efficacy and safety of azvudine in hospitalized SARS-CoV-2 infected patients with liver diseases based on a multicenter, retrospective cohort study. Adv. Sci. 12, e2405679. https://doi.org/10.1002/advs.202405679 (2025).",
"volume": "12",
"year": "2025"
},
{
"DOI": "10.1186/s12879-025-10643-w",
"author": "B Rao",
"doi-asserted-by": "publisher",
"first-page": "275",
"journal-title": "BMC Infect. Dis.",
"key": "34514_CR30",
"unstructured": "Rao, B. et al. Real-world effectiveness and safety of oral azvudine versus paxlovid for COVID-19 in patients with kidney disease: A multicenter, retrospective, cohort study. BMC Infect. Dis. 25, 275. https://doi.org/10.1186/s12879-025-10643-w (2025).",
"volume": "25",
"year": "2025"
},
{
"DOI": "10.1002/advs.202306050",
"author": "L Wu",
"doi-asserted-by": "publisher",
"first-page": "e2306050",
"journal-title": "Adv. Sci.",
"key": "34514_CR31",
"unstructured": "Wu, L. et al. Azvudine for the treatment of COVID-19 in pre-existing cardiovascular diseases: A single-center, real-world experience. Adv. Sci. 11, e2306050. https://doi.org/10.1002/advs.202306050 (2024).",
"volume": "11",
"year": "2024"
},
{
"DOI": "10.1056/NEJMoa2001282",
"author": "B Cao",
"doi-asserted-by": "publisher",
"first-page": "1787",
"journal-title": "N. Engl. J. Med.",
"key": "34514_CR32",
"unstructured": "Cao, B. et al. A trial of lopinavir-ritonavir in adults hospitalized with severe COVID-19. N. Engl. J. Med. 382, 1787–1799. https://doi.org/10.1056/NEJMoa2001282 (2020).",
"volume": "382",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa2021436",
"author": "P Horby",
"doi-asserted-by": "publisher",
"first-page": "693",
"journal-title": "N. Engl. J. Med.",
"key": "34514_CR33",
"unstructured": "Horby, P. et al. Dexamethasone in hospitalized patients with COVID-19. N. Engl. J. Med. 384, 693–704. https://doi.org/10.1056/NEJMoa2021436 (2021).",
"volume": "384",
"year": "2021"
},
{
"DOI": "10.3389/fmed.2023.1143485",
"author": "RM da Silva",
"doi-asserted-by": "publisher",
"first-page": "1143485",
"journal-title": "Front. Med.",
"key": "34514_CR34",
"unstructured": "da Silva, R. M. et al. Serial viral load analysis by DDPCR to evaluate FNC efficacy and safety in the treatment of mild cases of COVID-19. Front. Med. 10, 1143485. https://doi.org/10.3389/fmed.2023.1143485 (2023).",
"volume": "10",
"year": "2023"
}
],
"reference-count": 34,
"references-count": 34,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.nature.com/articles/s41598-025-34514-1"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "Evaluating the effectiveness and safety of Azvudine for hospitalised patients with COVID-19 and hypertension: a multicenter retrospective cohort study",
"type": "journal-article",
"update-policy": "https://doi.org/10.1007/springer_crossmark_policy"
}
