Phase 2/3 Trial of Molnupiravir for Treatment of Covid-19 in Nonhospitalized Adults
et al., NEJM Evidence, doi:10.1056/EVIDoa2100043, MOVe-OUT Phase 2, NCT04575597, Jan 2022
Phase 2 RCT of 302 nonhospitalized adults showing possible benefit with molnupiravir (200, 400, or 800 mg twice daily for 5 days) for treatment of mild-to-moderate COVID-19.
Potential risks of molnupiravir include the creation of dangerous variants, and mutagenicity, carcinogenicity, teratogenicity, and embryotoxicity1-15. Multiple analyses have identified variants potentially created by molnupiravir16-20. Studies show significantly increased risk of acute kidney injury21, cardiovascular toxocity22, and neurological symptoms21. Treatment may increase viral rebound23,24.
|
risk of death, 66.7% lower, RR 0.33, p = 1.00, treatment 0 of 74 (0.0%), control 1 of 74 (1.4%), NNT 74, relative risk is not 0 because of continuity correction due to zero events (with reciprocal of the contrasting arm), 800mg, day 29.
|
|
risk of hospitalization, 25.0% lower, RR 0.75, p = 1.00, treatment 3 of 74 (4.1%), control 4 of 74 (5.4%), NNT 74, 800mg, day 29.
|
|
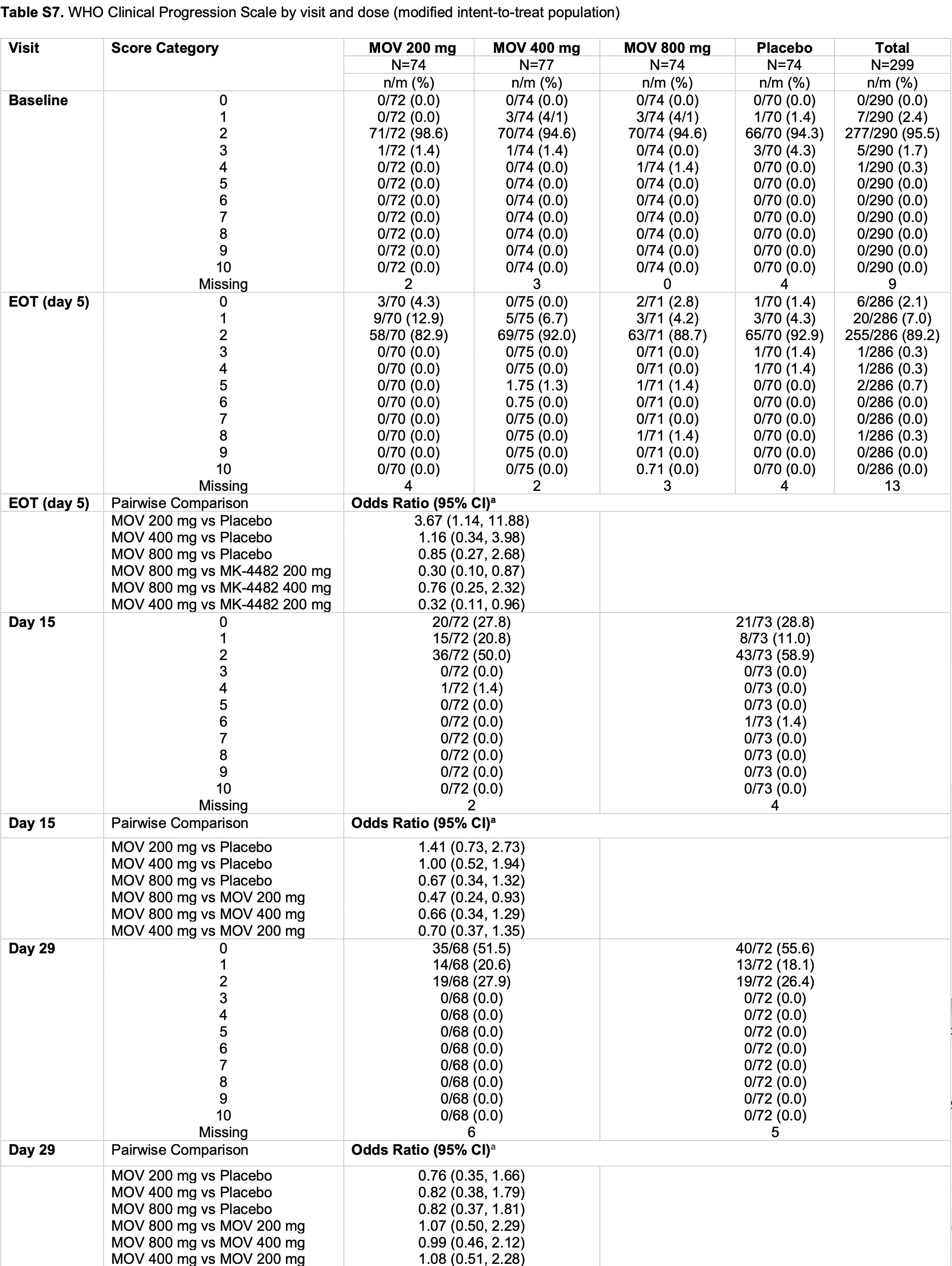
risk of no recovery, 22.0% higher, OR 1.22, p = 0.63, treatment 74, control 74, inverted to make OR<1 favor treatment, 800mg, day 29, Table S7, RR approximated with OR.
|
|
risk of no recovery, 49.3% higher, OR 1.49, p = 0.25, treatment 74, control 74, inverted to make OR<1 favor treatment, 800mg, day 15, Table S7, RR approximated with OR.
|
|
risk of no recovery, 17.6% higher, OR 1.18, p = 0.79, treatment 74, control 74, inverted to make OR<1 favor treatment, 800mg, day 5, Table S7, RR approximated with OR.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
1.
Swanstrom et al., Lethal mutagenesis as an antiviral strategy, Science, doi:10.1126/science.abn0048.
2.
Hadj Hassine et al., Lethal Mutagenesis of RNA Viruses and Approved Drugs with Antiviral Mutagenic Activity, Viruses, doi:10.3390/v14040841.
3.
Shum, C., An investigational study into the drug-associated mutational signature in SARS-CoV-2 viruses, The University of Hong Kong, PhD Thesis, hub.hku.hk/handle/10722/344396.
4.
Waters et al., Human genetic risk of treatment with antiviral nucleoside analog drugs that induce lethal mutagenesis: the special case of molnupiravir, Environmental and Molecular Mutagenesis, doi:10.1002/em.22471.
5.
Huntsman, M., An assessment of the reproductive toxicity of the anti-COVID-19 drug molnupiravir using stem cell-based embryo models, Master's Thesis, scholarspace.manoa.hawaii.edu/items/cd11342c-b4dc-44c0-8b44-ce6e3369c40b.
6.
Huntsman (B) et al., Detection of developmental toxicity of the anti-COVID-19 drug molnupiravir using gastruloid-based in vitro assays, Toxicological Sciences, doi:10.1093/toxsci/kfaf093.
7.
Zibat et al., N4-hydroxycytidine, the active compound of Molnupiravir, promotes SARS-CoV-2 mutagenesis and escape from a neutralizing nanobody, iScience, doi:10.1016/j.isci.2023.107786.
8.
Shiraki et al., Convenient screening of the reproductive toxicity of favipiravir and antiviral drugs in Caenorhabditis elegans, Heliyon, doi:10.1016/j.heliyon.2024.e35331.
9.
Gruber et al., Molnupiravir increases SARS‐CoV‐2 genome diversity and complexity: A case‐control cohort study, Journal of Medical Virology, doi:10.1002/jmv.29642.
10.
Marikawa et al., An active metabolite of the anti-COVID-19 drug molnupiravir impairs mouse preimplantation embryos at clinically relevant concentrations, Reproductive Toxicology, doi:10.1016/j.reprotox.2023.108475.
11.
Rahman, M., Elucidation of the DNA repair mechanisms involved in the repair of DNA damage caused by the Arabinosides and Anti-COVID-19 drugs, tokyo-metro-u.repo.nii.ac.jp/records/2000972.
12.
Zhou et al., β-D-N4-hydroxycytidine Inhibits SARS-CoV-2 Through Lethal Mutagenesis But Is Also Mutagenic To Mammalian Cells, The Journal of Infectious Diseases, doi:10.1093/infdis/jiab247.
13.
Chamod et al., Molnupiravir Metabolite--N4-hydroxycytidine Causes Cytotoxicity and DNA Damage in Mammalian Cells in vitro: N4-hydroxycytidine Induced Cytotoxicity DNA Damage, Asian Medical Journal and Alternative Medicine, 23:3, asianmedjam.com/index.php/amjam/article/view/1448.
14.
Standing et al., Randomized controlled trial of molnupiravir SARS-CoV-2 viral and antibody response in at-risk adult outpatients, Nature Communications, doi:10.1038/s41467-024-45641-0.
15.
Mori et al., Reactive oxygen species-mediated cytotoxic and DNA-damaging mechanism of N4-hydroxycytidine, a metabolite of the COVID-19 therapeutic drug molnupiravir, Free Radical Research, doi:10.1080/10715762.2025.2469738.
16.
Focosi et al., The fitness of molnupiravir-signed SARS-CoV-2 variants: imputation analysis based on prescription counts and GISAID analyses by country, Intervirology, doi:10.1159/000540282.
17.
Sanderson et al., A molnupiravir-associated mutational signature in global SARS-CoV-2 genomes, Nature, doi:10.1038/s41586-023-06649-6.
18.
Fountain-Jones et al., Effect of molnupiravir on SARS-CoV-2 evolution in immunocompromised patients: a retrospective observational study, The Lancet Microbe, doi:10.1016/S2666-5247(23)00393-2.
19.
Kosakovsky Pond et al., Anti-COVID drug accelerates viral evolution, Nature, doi:10.1038/d41586-023-03248-3.
21.
Siby et al., Temporal Trends in Serious Adverse Events Associated with Oral Antivirals During the COVID-19 Pandemic: Insights from the FAERS Database (2020–2023), Open Forum Infectious Diseases, doi:10.1093/ofid/ofaf695.1825.
22.
Ozhan et al., Evaluation of the cardiopulmonary effects of repurposed COVID-19 therapeutics in healthy rats, Scientific Reports, doi:10.1038/s41598-025-31048-4.
Caraco et al., 25 Jan 2022, Double Blind Randomized Controlled Trial, placebo-controlled, multiple countries, peer-reviewed, mean age 49.2, 19 authors, trial NCT04575597 (history) (MOVe-OUT Phase 2).
Contact: matthew.johnson1@merck.com.
Phase 2/3 Trial of Molnupiravir for Treatment of Covid-19 in Nonhospitalized Adults
NEJM Evidence, doi:10.1056/evidoa2100043
BACKGROUND Safe and effective oral treatments are needed to improve clinical outcomes for nonhospitalized patients with Covid-19. Molnupiravir is an orally administered, small-molecule ribonucleoside prodrug shown to inhibit replication of severe acute respiratory syndrome coronavirus 2 in vitro and in animal models. METHODS MOVe-OUT is an ongoing, phase 2/3, randomized, placebo-controlled, double-blind study evaluating the safety, efficacy, and pharmacokinetics of molnupiravir in nonhospitalized adults. In the phase 2 component, participants had mild or moderate, laboratory-confirmed Covid-19 with sign/symptom onset up to (and including) 7 days before randomization. Participants were randomly assigned 1:1:1:1 to receive 200, 400, or 800 mg of molnupiravir or placebo twice daily for 5 days, stratified by time since sign/ symptom onset and by being at increased risk for severe illness from Covid-19. The primary efficacy end point was the proportion of participants who were hospitalized and/or died through day 29.
RESULTS The phase 2 component randomly assigned 302 participants to treatment; baseline characteristics were comparable across treatment groups. Molnupiravir had no apparent dose-related effect on adverse events, and no clinically meaningful abnormalities in laboratory test results were observed in relation to dose or treatment. Eleven participants were hospitalized or died through day 29. Of 225 participants in the combined molnupiravir group, 7 (3.1%) were hospitalized or died, compared with 4 of 74 participants (5.4%) in the placebo group. Subgroup analyses suggested lower incidences of hospitalization and/or death in the molnupiravir versus placebo groups in participants older than 60 years of age, those with increased risk for severe illness, those with symptom onset up to (and including) 5 days before randomization, and those with both symptom onset up to (and including) 5 days before randomization and increased risk for severe illness.
Author Affiliations
References
Agostini, Pruijssers, Chappell, Small-molecule antiviral b-d-N 4 -hydroxycytidine inhibits a proofreading-intact coronavirus with a high genetic barrier to resistance, J Virol
Arribas, Bhagani, Lobo, Randomized Trial of Molnupiravir or Placebo in Patients Hospitalized with Covid-19, NEJM Evid, doi:10.1056/EVIDoa2100044
Bernal, Da Silva, Musungaie, Molnupiravir for oral treatment of Covid-19 in nonhospitalized patients, N Engl J Med, doi:10.1056/NEJMoa2116044
Bioreliance, Big Blue® Transgenic Rodent Mutation Assay
Chawla, Cao, Stone, Model-based dose selection for the phase 3 evaluation of molnupiravir in the treatment of COVID-19 in adults
Chen, Zhang, Case, Resistance of SARS-CoV-2 variants to neutralization by monoclonal and serum-derived polyclonal antibodies, Nat Med
Collier, Marco, Ferreira, CITIID-NIHR Bio-Resource COVID-19 Collaboration; COVID-19 Genomics UK (COG-UK) Consortium. Sensitivity of SARS-CoV-2 B.1.1.7 to mRNA vaccine-elicited antibodies, Nature
Cox, Wolf, Plemper, Therapeutically administered ribonucleoside analogue MK-4482/EIDD-2801 blocks SARS-CoV-2 transmission in ferrets, Nat Microbiol
Davies, Abbott, Barnard, CMMID COVID-19 Working Group; COVID-19 Genomics UK (COG-UK) Consortium. Estimated transmissibility and impact of SARS-CoV-2 lineage B.1.1.7 in England, Science
Fiore, Fry, Shay, Gubareva, Bresee et al., Antiviral agents for the treatment and chemoprophylaxis of influenza-recommendations of the Advisory Committee on Immunization Practices
Gollapudi, Lynch, Heflich, The in vivo Pig-a assay: a report of the International Workshop on Genotoxicity Testing (IWGT) Workgroup, Mutat Res Genet Toxicol Environ Mutagen
Gottlieb, Nirula, Chen, Effect of bamlanivimab as monotherapy or in combination with etesevimab on viral load in patients with mild to moderate COVID-19: a randomized clinical trial, JAMA
Grobler, Strizki, Murgolo, Molnupiravir maintains antiviral activity against SARS-CoV-2 variants in vitro and in early clinical studies
Heflich, Dertinger, Dobrovolsky, The in vivo erythrocyte Pig-a gene mutation assay. Part 1: Detailed review paper and retrospective performance assessment
Miettinen, Nurminen, Comparative analysis of two rates, Stat Med
River, Vivo Pig-a Assay
Sheahan, Sims, Zhou, An orally bioavailable broadspectrum antiviral inhibits SARS-CoV-2 in human airway epithelial cell cultures and multiple coronaviruses in mice, Sci Transl Med
Stokes, Zambrano, Anderson, Coronavirus Disease 2019 Case Surveillance -United States, January 22, MMWR Morb Mortal Wkly Rep
Stuyver, Whitaker, Mcbrayer, Ribonucleoside analogue that blocks replication of bovine viral diarrhea and hepatitis C viruses in culture, Antimicrob Agents Chemother
Troth, Butterton, Deanda, Letter to the editor in response to Zhou et al, J Infect Dis
Urakova, Kuznetsova, Crossman, Beta-d-N(4)-hydroxycytidine is a potent anti-alphavirus compound that induces a high level of mutations in the viral genome, J Virol
Wahl, Gralinski, Johnson, SARS-CoV-2 infection is effectively treated and prevented by EIDD-2801, Nature
Weinreich, Sivapalasingam, Norton, Trial Investigators. REGN-COV2, a neutralizing antibody cocktail, in outpatients with Covid-19, N Engl J Med
Yoon, Toots, Lee, Orally efficacious broad-spectrum ribonucleoside analog inhibitor of influenza and respiratory syncytial viruses, Antimicrob Agents Chemother
Zhou, Hill, Sarkar, b-d-N4-hydroxycytidine inhibits SARS-CoV-2 through lethal mutagenesis but is also mutagenic to mammalian cells, J Infect Dis
DOI record:
{
"DOI": "10.1056/evidoa2100043",
"ISSN": [
"2766-5526"
],
"URL": "http://dx.doi.org/10.1056/EVIDoa2100043",
"alternative-id": [
"10.1056/EVIDoa2100043"
],
"author": [
{
"affiliation": [
{
"name": "Clinical Pharmacology Unit, Hadassah-Hebrew University Medical Center, Jerusalem, Israel"
}
],
"family": "Caraco",
"given": "Yoseph",
"sequence": "first"
},
{
"affiliation": [
{
"name": "Crofoot Research Center, Inc., Houston, TX"
}
],
"family": "Crofoot",
"given": "Gordon E.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Internal Medicine, Infectious Disease Service, Fundación Valle del Lili, Cali, Colombia"
}
],
"family": "Moncada",
"given": "Pablo Andres",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Saint Petersburg State Pediatric Medical University, Saint Petersburg, Russia"
},
{
"name": "Strategic Medical System LLC, Saint Petersburg, Russia"
}
],
"family": "Galustyan",
"given": "Anna Nikolaevna",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Jongaie Research, Gauteng, South Africa"
}
],
"family": "Musungaie",
"given": "Dany Badibanga",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Departments of Infectious Diseases and Medical Virology, Newcastle upon Tyne Hospitals, Newcastle upon Tyne, UK"
}
],
"family": "Payne",
"given": "Brendan",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Medical Research Institute LLC, Saint Petersburg, Russia"
}
],
"family": "Kovalchuk",
"given": "Evgeniy",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Advanced Research for Health Improvement LLC, Immokalee, FL"
}
],
"family": "Gonzalez",
"given": "Antonio",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Merck Research Laboratories, Merck & Co., Inc., Kenilworth, NJ"
}
],
"family": "Brown",
"given": "Michelle L.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Merck Research Laboratories, Merck & Co., Inc., Kenilworth, NJ"
}
],
"family": "Williams-Diaz",
"given": "Angela",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Merck Research Laboratories, Merck & Co., Inc., Kenilworth, NJ"
}
],
"family": "Gao",
"given": "Wei",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Merck Research Laboratories, Merck & Co., Inc., Kenilworth, NJ"
}
],
"family": "Strizki",
"given": "Julie M.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Merck Research Laboratories, Merck & Co., Inc., Kenilworth, NJ"
}
],
"family": "Grobler",
"given": "Jay",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Merck Research Laboratories, Merck & Co., Inc., Kenilworth, NJ"
}
],
"family": "Du",
"given": "Jiejun",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Merck Research Laboratories, Merck & Co., Inc., Kenilworth, NJ"
}
],
"family": "Assaid",
"given": "Christopher A.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Merck Research Laboratories, Merck & Co., Inc., Kenilworth, NJ"
}
],
"family": "Paschke",
"given": "Amanda",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Merck Research Laboratories, Merck & Co., Inc., Kenilworth, NJ"
}
],
"family": "Butterton",
"given": "Joan R.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Merck Research Laboratories, Merck & Co., Inc., Kenilworth, NJ"
}
],
"family": "Johnson",
"given": "Matthew G.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Merck Research Laboratories, Merck & Co., Inc., Kenilworth, NJ"
}
],
"family": "De Anda",
"given": "Carisa",
"sequence": "additional"
}
],
"container-title": "NEJM Evidence",
"container-title-short": "NEJM Evidence",
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2021,
12,
21
]
],
"date-time": "2021-12-21T23:46:05Z",
"timestamp": 1640130365000
},
"deposited": {
"date-parts": [
[
2022,
4,
28
]
],
"date-time": "2022-04-28T18:11:51Z",
"timestamp": 1651169511000
},
"indexed": {
"date-parts": [
[
2023,
4,
20
]
],
"date-time": "2023-04-20T10:07:49Z",
"timestamp": 1681985269591
},
"is-referenced-by-count": 30,
"issue": "2",
"issued": {
"date-parts": [
[
2022,
1,
25
]
]
},
"journal-issue": {
"issue": "2",
"published-print": {
"date-parts": [
[
2022,
1,
25
]
]
}
},
"language": "en",
"member": "150",
"original-title": [],
"prefix": "10.1056",
"published": {
"date-parts": [
[
2022,
1,
25
]
]
},
"published-print": {
"date-parts": [
[
2022,
1,
25
]
]
},
"publisher": "Massachusetts Medical Society",
"reference": [
{
"key": "e_1_3_5_2_2",
"unstructured": "World Health Organization. WHO Coronavirus (COVID-19) Dashboard (https://covid19.who.int/)."
},
{
"DOI": "10.15585/mmwr.mm6924e2",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_3_2"
},
{
"DOI": "10.1128/AAC.00766-18",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_4_2"
},
{
"DOI": "10.1038/s41564-020-00835-2",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_5_2"
},
{
"DOI": "10.1126/scitranslmed.abb5883",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_6_2"
},
{
"DOI": "10.1038/s41586-021-03312-w",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_7_2"
},
{
"DOI": "10.1128/JVI.01965-17",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_8_2"
},
{
"DOI": "10.1093/infdis/jiab247",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_9_2"
},
{
"DOI": "10.1016/j.mrgentox.2014.09.007",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_13_2"
},
{
"DOI": "10.1093/infdis/jiab362",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_14_2"
},
{
"DOI": "10.1056/NEJMoa2116044",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_17_2"
},
{
"key": "e_1_3_5_18_2",
"unstructured": "Chawla A Cao Y Stone J et al. Model-based dose selection for the phase 3 evaluation of molnupiravir in the treatment of COVID-19 in adults. Poster presented at the 2021 ECCMID Annual Meeting Vienna Austria. July 9-12 2021 (https://eacademy.escmid.org/escmid/2021/eccmid-2021/332297/)."
},
{
"DOI": "10.1002/sim.4780040211",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_19_2"
},
{
"article-title": "Antiviral agents for the treatment and chemoprophylaxis of influenza — recommendations of the Advisory Committee on Immunization Practices (ACIP)",
"first-page": "1",
"journal-title": "MMWR Recomm Rep",
"key": "e_1_3_5_20_2",
"unstructured": "Fiore AE, Fry A, Shay D, Gubareva L, Bresee JS, Uyeki TM. Antiviral agents for the treatment and chemoprophylaxis of influenza — recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep 2011;60:1-24.",
"volume": "60",
"year": "2011"
},
{
"key": "e_1_3_5_21_2",
"unstructured": "GlaxoSmithKline. Fact Sheet for Healthcare Providers Emergency Use Authorization (EUA) of Sotrovimab. Updated November 2021 (https://gskpro.com/content/dam/global/hcpportal/en_US/Prescribing_Information/Sotrovimab/pdf/SOTROVIMAB-EUA.PDF#nameddest=HCPFS2021)."
},
{
"DOI": "10.1001/jama.2021.0202",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_22_2"
},
{
"DOI": "10.1056/NEJMoa2035002",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_23_2"
},
{
"DOI": "10.1128/JVI.01348-19",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_24_2"
},
{
"DOI": "10.1128/AAC.47.1.244-254.2003",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_25_2"
},
{
"DOI": "10.1038/s41591-021-01294-w",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_26_2"
},
{
"DOI": "10.1038/s41586-021-03412-7",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_27_2"
},
{
"DOI": "10.1126/science.abg3055",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_28_2"
},
{
"key": "e_1_3_5_29_2",
"unstructured": "Grobler J Strizki J Murgolo N et al. Molnupiravir maintains antiviral activity against SARS-CoV-2 variants in vitro and in early clinical studies. Presented at ID Week September 19 to October 3 2021 (https://idweek.junolive.co/welcome2021)."
},
{
"DOI": "10.1056/EVIDoa2100044",
"doi-asserted-by": "publisher",
"key": "e_1_3_5_30_2"
}
],
"reference-count": 24,
"references-count": 24,
"relation": {},
"resource": {
"primary": {
"URL": "https://evidence.nejm.org/doi/10.1056/EVIDoa2100043"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subtitle": [],
"title": "Phase 2/3 Trial of Molnupiravir for Treatment of Covid-19 in Nonhospitalized Adults",
"type": "journal-article",
"volume": "1"
}