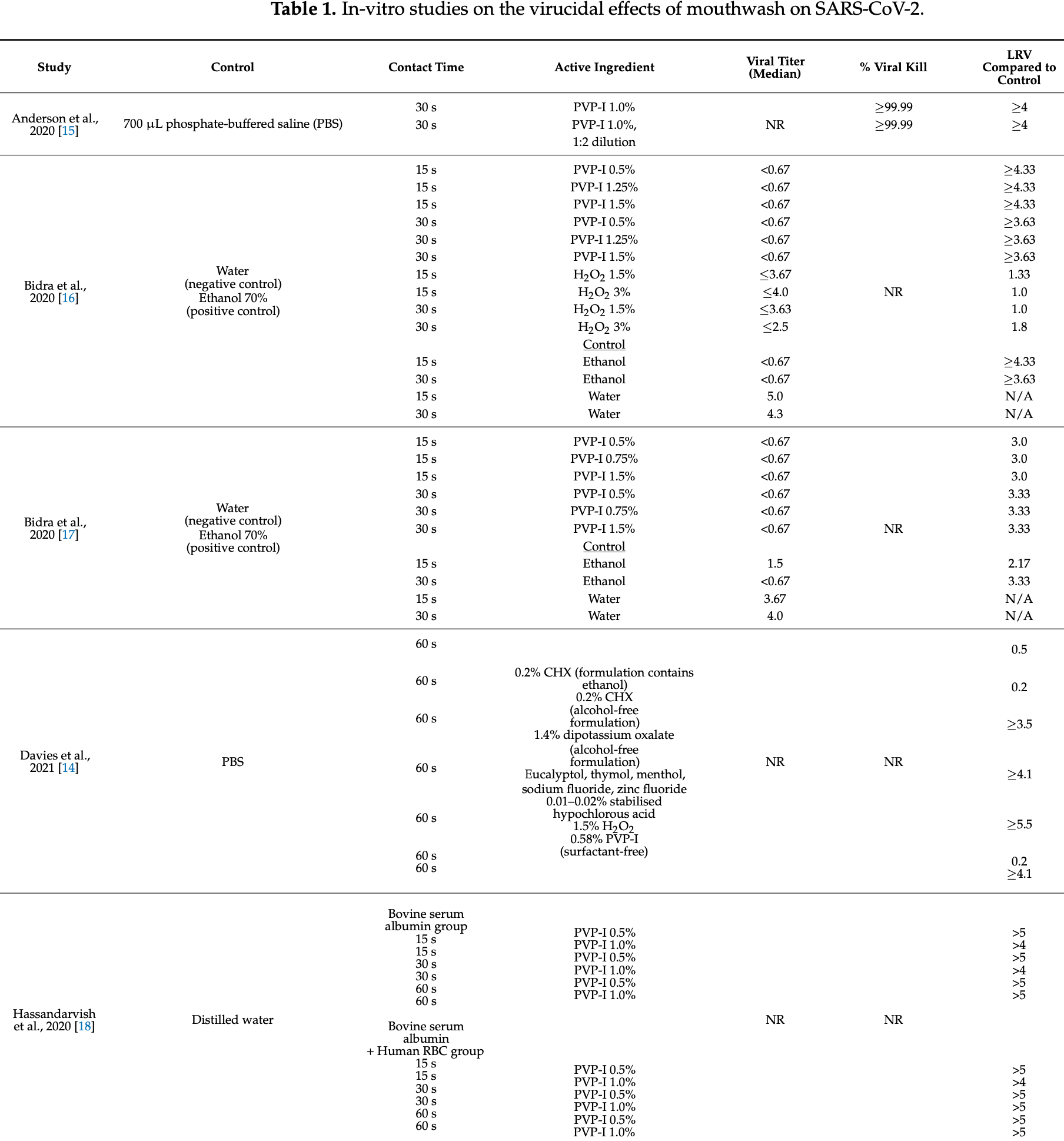
The In Vitro Virucidal Effects of Mouthwashes on SARS-CoV-2
et al., International Journal of Translational Medicine, doi:10.3390/ijtm2030030, Jul 2022
PVP-I for COVID-19
14th treatment shown to reduce risk in
February 2021, now with p = 0.0000000037 from 21 studies.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
Review of in vitro studies of mouthwashes, showing antiviral activity for SARS-CoV-2 with many compounds including PVP-I, cetylpyridinium chloride, chlorohexidine gluconate, dequalinium chloride, benzalkonium chloride, anionic phthalocyanine derivative, hypochlorous acid, and hydrogen peroxide.
Review covers povidone-iodine, cetylpyridinium chloride, chlorhexidine, hydrogen peroxide, and hypochlorous acid.
1.
Angjelova et al., Effects of Antiseptic Formulations on Oral Microbiota and Related Systemic Diseases: A Scoping Review, Antibiotics, doi:10.3390/antibiotics14080815.
2.
Thangamani et al., Rinsing away the threat: Antiviral mouthwashes and their efficacy, World Academy of Sciences Journal, doi:10.3892/wasj.2025.366.
3.
Gunawan et al., The Role of Poly Vinyl Pyrrolidone Iodine (PVP-I) in Preventing Cross-Infection during Dental Procedures: A Systematic Review in the COVID-19 Context, Journal of International Society of Preventive and Community Dentistry, doi:10.4103/jispcd.jispcd_78_24.
4.
Okeke et al., Antiseptics: An expeditious third force in the prevention and management of coronavirus diseases, Current Research in Microbial Sciences, doi:10.1016/j.crmicr.2024.100293.
5.
Gandhi et al., Washing Illness Away: A Systematic Review of the Impact of Nasal Irrigation and Spray on COVID‐19, The Laryngoscope, doi:10.1002/lary.31761.
6.
Brito-Reia et al., Antiviral Mechanism and Clinical Benefits of Mouthwash Active Against SARS-CoV-2, Current Oral Health Reports, doi:10.1007/s40496-024-00368-1.
7.
Donzelli, A., Neglected Effective Early Therapies against COVID-19: Focus on Functional Foods and Related Active Substances. A Review, MDPI AG, doi:10.20944/preprints202312.1178.v1.
8.
Chavda et al., Nasal sprays for treating COVID-19: a scientific note, Pharmacological Reports, doi:10.1007/s43440-023-00463-7.
9.
Ting et al., The In Vitro Virucidal Effects of Mouthwashes on SARS-CoV-2, International Journal of Translational Medicine, doi:10.3390/ijtm2030030.
10.
Lim et al., Repurposing povidone-iodine to reduce the risk of SARS-CoV-2 infection and transmission: a narrative review, Annals of Medicine, doi:10.1080/07853890.2022.2076902.
Ting et al., 31 Jul 2022, peer-reviewed, 2 authors.
The In Vitro Virucidal Effects of Mouthwashes on SARS-CoV-2
International Journal of Translational Medicine, doi:10.3390/ijtm2030030
Oral antiseptic mouthwashes have been widely used for their antibacterial activity. As a result of the SARS-CoV-2 pandemic, the antiviral properties of these oral antiseptics have been aggressively studied. To demonstrate the direct antiviral activity of mouthwashes against SARS-CoV-2, this review will focus on the in vitro virucidal effects of these mouthwashes. Knowledge of the type, concentration, and exposure time of available mouthwashes can provide insights into effective protocols for their clinical use. With an understanding of the characteristics of each oral antiseptic mouthwash, proper mouthwash selection against SARS-CoV-2 may become a useful adjunct to personal protective equipment.
Author Contributions: M.T. participated in the design of the review, literature search and article selection, drafting and revising of manuscript and table, and final approval of submitted version. J.B.S. participated in the design of the review, article selection, drafting and revising of manuscript and table, and final approval of submitted version. All authors have read and agreed to the published version of the manuscript.
Conflicts of Interest: The authors declare no conflict of interest in the writing of this manuscript. J.B.S. is a U.S. Government Special Government Employee with the US Food and Drug Administration, Silver Spring, MD, USA.
References
Abdel-Moneim, Abdelwhab, Memish, Insights into SARS-CoV-2 evolution, potential antivirals, and vaccines, Virology, doi:10.1016/j.virol.2021.02.007
Ader, Paul, Reinhardt, Safran, Pino et al., Effect of mouth rinsing with two polyvinylpyrrolidone-iodine mixtures on iodine absorption and thyroid function, J. Clin. Endocrinol. Metab, doi:10.1210/jcem-66-3-632
Anderson, Sivalingam, Kang, Ananthanarayanan, Arumugam et al., Povidone-Iodine Demonstrates Rapid In Vitro Virucidal Activity Against SARS-CoV-2, The Virus Causing COVID-19 Disease, Infect. Dis. Ther, doi:10.1007/s40121-020-00316-3
Bai, Yao, Wei, Tian, Jin et al., Presumed Asymptomatic Carrier Transmission of COVID-19, JAMA, doi:10.1001/jama.2020.2565
Banerjee, Nasir, Budylowski, Yip, Aftanas et al., Isolation, Sequence, Infectivity, and Replication Kinetics of Severe Acute Respiratory Syndrome Coronavirus 2, Emerg. Infect. Dis, doi:10.3201/eid2609.201495
Bidra, Pelletier, Westover, Frank, Brown et al., Comparison of In Vitro Inactivation of SARS CoV-2 with Hydrogen Peroxide and Povidone-Iodine Oral Antiseptic Rinses, J. Prosthodont, doi:10.1111/jopr.13220
Bidra, Pelletier, Westover, Frank, Brown et al., Rapid In-Vitro Inactivation of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) Using Povidone-Iodine Oral Antiseptic Rinse, J. Prosthodont, doi:10.1111/jopr.13209
Bordea, Candrea, Salagean, Pop, Lucaciu et al., Impact of COVID-19 Pandemic on Healthcare Professionals and Oral Care Operational Services: A Systemic Review, Risk Manag. Healthc. Policy, doi:10.2147/RMHP.S284557
Bordea, Xhajanka, Candrea, Bran, Onisor et al., Coronavirus (SARS-CoV-2) Pandemic: Future Challenges for Dental Practitioners, Microorganisms, doi:10.3390/microorganisms8111704
Caruso, Del Prete, Lazzarino, Hydrogen peroxide and viral infections: A literature review with research hypothesis definition in relation to the current covid-19 pandemic, Med. Hypotheses, doi:10.1016/j.mehy.2020.109910
Ceri, Olson, Stremick, Read, Morck et al., The Calgary Biofilm Device: New technology for rapid determination of antibiotic susceptibilities of bacterial biofilms, J. Clin. Microbiol, doi:10.1128/JCM.37.6.1771-1776.1999
Chin, Chu, Perera, Hui, Yen et al., Stability of SARS-CoV-2 in different environmental conditions, Lancet Microbe, doi:10.1016/S2666-5247(20)30003-3
Cieplik, Jakubovics, Buchalla, Maisch, Hellwig et al., Resistance Toward Chlorhexidine in Oral Bacteria-Is There Cause for Concern? Front, Microbiol, doi:10.3389/fmicb.2019.00587
Davies, Buczkowski, Welch, Green, Mawer et al., Effective in vitro inactivation of SARS-CoV-2 by commercially available mouthwashes, J. Gen. Virol, doi:10.1099/jgv.0.001578
De Clercq, Stewart, None
De Somer, Studies on the mechanism of the priming effect of interferon on interferon production by cell cultures exposed to poly(rI)-poly(rC), Infect. Immun, doi:10.1128/iai.8.3.309-316.1973
Domingo, Farrales, Loya, Pura, Uy, The effect of 1% povidone iodine as a pre-procedural mouthrinse in 20 patients with varying degrees of oral hygiene, J. Philipp. Dent. Assoc
Drake, Villhauer, An in vitro comparative study determining bactericidal activity of stabilized chlorine dioxide and other oral rinses, J. Clin. Dent
Eggers, Koburger-Janssen, Eickmann, Zorn, In Vitro Bactericidal and Virucidal Efficacy of Povidone-Iodine Gargle/Mouthwash Against Respiratory and Oral Tract Pathogens, Infect. Dis. Ther, doi:10.1007/s40121-018-0200-7
Elliot, Herbert, Jack, Stable Compositions of Uncomplexed Iodine and Methods of Use, U.S. Patent No
Elworthy, Greenman, Doherty, Newcombe, Addy, The substantivity of a number of oral hygiene products determined by the duration of effects on salivary bacteria, J. Periodontol, doi:10.1902/jop.1996.67.6.572
Emilson, Fornell, Effect of toothbrushing with chlorhexidine gel on salivary microflora, oral hygiene, and caries, Scand. J. Dent. Res, doi:10.1111/j.1600-0722.1976.tb00495.x
Enciso, Keaton, Saleh, Ahmadieh, Clark et al., Assessing the utility of serum C-telopeptide crosslink of type 1 collagen as a predictor of bisphosphonate-related osteonecrosis of the jaw: A systematic review and meta-analysis, J. Am. Dent. Assoc, doi:10.1016/j.adaj.2016.02.011
Fernandez, Guedes, Langa, Rosing, Cavagni et al., Virucidal efficacy of chlorhexidine: A systematic review, Odontology, doi:10.1007/s10266-021-00660-x
Fogorv, Szle, None
Foley, Jr, The relationship between autoimmune thyroid disease and iodine intake: A review, Endokrynol. Pol
Frank, Brown, Capriotti, Westover, Pelletier et al., In Vitro Efficacy of a Povidone-Iodine Nasal Antiseptic for Rapid Inactivation of SARS-CoV-2, JAMA Otolaryngol. Head Neck Surg, doi:10.1001/jamaoto.2020.3053
Furudate, Nishimaki, Muto, 125I uptake competing with iodine absorption by the thyroid gland following povidoneiodine skin application, Exp. Anim, doi:10.1538/expanim.46.197
Geneva, Cuzzo, Fazili, Javaid, Normal Body Temperature: A Systematic Review, Open Forum Infect. Dis, doi:10.1093/ofid/ofz032
Gillim-Ross, Taylor, Scholl, Ridenour, Masters et al., Discovery of novel human and animal cells infected by the severe acute respiratory syndrome coronavirus by replication-specific multiplex reverse transcription-PCR, J. Clin. Microbiol, doi:10.1128/JCM.42.7.3196-3206.2004
Gray, Katelaris, Lipson, Recurrent anaphylaxis caused by topical povidone-iodine (Betadine), J. Paediatr Child. Health, doi:10.1111/jpc.12232
Grootveld, Silwood, Gill, Lynch, Evidence for the microbicidal activity of a chlorine dioxide-containing oral rinse formulation in vivo, J. Clin. Dent
Harrel, Molinari, Aerosols and splatter in dentistry: A brief review of the literature and infection control implications, J. Am. Dent. Assoc, doi:10.14219/jada.archive.2004.0207
Hassandarvish, Tiong, Mohamed, Arumugam, Ananthanarayanan et al., In vitro virucidal activity of povidone iodine gargle and mouthwash against SARS-CoV-2: Implications for dental practice, Br. Dent. J, doi:10.1038/s41415-020-2402-0
Hoffmann, Kleine-Weber, Schroeder, Kruger, Herrler et al., SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor, Cell, doi:10.1016/j.cell.2020.02.052
Huang, Huang, Use of chlorhexidine to eradicate oropharyngeal SARS-CoV-2 in COVID-19 patients, J. Med. Virol, doi:10.1002/jmv.26954
Inchingolo, Inchingolo, Bordea, Malcangi, Xhajanka et al., SARS-CoV-2 Disease through Viral Genomic and Receptor Implications: An Overview of Diagnostic and Immunology Breakthroughs, Microorganisms, doi:10.3390/microorganisms9040793
Iorgulescu, Saliva between normal and pathological. Important factors in determining systemic and oral health, J. Med. Life
Jain, Bhat, Asawa, Tak, Singh et al., Effect of Training School Teachers on Oral Hygiene Status of 8-10 Years Old Government School Children of Udaipur City, India, J. Clin. Diagn. Res. JCDR, doi:10.7860/JCDR/2016/18576.8330
Kabir, Ahmed, Chowdhury, Iqbal, Paulmurugan et al., Management of COVID-19: Current status and future prospects, Microbes. Infect, doi:10.1016/j.micinf.2021.104832
Kampf, Acquired resistance to chlorhexidine-is it time to establish an 'antiseptic stewardship' initiative?, J. Hosp. Infect, doi:10.1016/j.jhin.2016.08.018
Kampf, Antibiotic Resistance Can Be Enhanced in Gram-Positive Species by Some Biocidal Agents Used for Disinfection, Antibiotics, doi:10.3390/antibiotics8010013
Kampf, Biocidal Agents Used for Disinfection Can Enhance Antibiotic Resistance in Gram-Negative Species, Antibiotics, doi:10.3390/antibiotics7040110
Kampf, Todt, Pfaender, Steinmann, Persistence of coronaviruses on inanimate surfaces and their inactivation with biocidal agents, J. Hosp. Infect, doi:10.1016/j.jhin.2020.01.022
Kariwa, Fujii, Takashima, Inactivation of SARS coronavirus by means of povidone-iodine, physical conditions and chemical reagents, Dermatology, doi:10.1159/000089211
Kaye, SARS-associated coronavirus replication in cell lines, Emerg. Infect. Dis, doi:10.3201/eid1201.050496
Koch-Heier, Hoffmann, Schindler, Lussi, Planz, Inactivation of SARS-CoV-2 through Treatment with the Mouth Rinsing Solutions ViruProX((R)) and BacterX((R)) Pro, Microorganisms, doi:10.3390/microorganisms9030521
Komine, Yamaguchi, Okamoto, Yamamoto, Virucidal activity of oral care products against SARS-CoV-2 in vitro, J. Oral. Maxillofac. Surg. Med. Pathol, doi:10.1016/j.ajoms.2021.02.002
Kovesi, The use of Betadine antiseptic in the treatment of oral surgical, parodontological and oral mucosal diseases
Kraus, Perry, Nickerson, Salivary catalase and peroxidase values in normal subjects and in persons with periodontal disease, Oral Surg. Oral Med. Oral Pathol, doi:10.1016/0030-4220(58)90228-7
Kärber, Beitrag zur kollektiven Behandlung pharmakologischer Reihenversuche. Naunyn-Schmiedebergs Arch, Für Exp. Pathol. Pharmakol, doi:10.1007/BF01863914
Lee, Suprono, Stephens, Withers, Li, Efficacy of stabilized chlorine dioxide-based unflavored mouthwash in reducing oral malodor: An 8-week randomized controlled study, Am. J. Dent
Loe, Schiott, The effect of mouthrinses and topical application of chlorhexidine on the development of dental plaque and gingivitis in man, J. Periodontal Res, doi:10.1111/j.1600-0765.1970.tb00696.x
Madan, Sequeira, Shenoy, Shetty, The effect of three mouthwashes on radiation-induced oral mucositis in patients with head and neck malignancies: A randomized control trial, J. Cancer Res, doi:10.4103/0973-1482.39597
Matsuyama, Nao, Shirato, Kawase, Saito et al., Enhanced isolation of SARS-CoV-2 by TMPRSS2-expressing cells, doi:10.1073/pnas.2002589117
Maynard, Jenkins, Moran, Addy, Newcombe et al., A 6-month home usage trial of a 1% chlorhexidine toothpaste (II). Effects on the oral microflora, J. Clin. Periodontol, doi:10.1111/j.1600-051X.1993.tb00345.x
Meister, Bruggemann, Todt, Conzelmann, Muller et al., Virucidal Efficacy of Different Oral Rinses Against Severe Acute Respiratory Syndrome Coronavirus 2, J. Infect. Dis, doi:10.1093/infdis/jiaa471
Micik, Miller, Mazzarella, Ryge, Studies on dental aerobiology. I. Bacterial aerosols generated during dental procedures, J. Dent. Res, doi:10.1177/00220345690480012401
Moskowitz, Mendenhall, Comparative Analysis of Antiviral Efficacy of Four Different Mouthwashes against Severe Acute Respiratory Syndrome Coronavirus 2: An In Vitro Study, Int. J. Exp. Dent. Sci, doi:10.5005/jp-journals-10029-1209
Mossel, Huang, Narayanan, Makino, Tesh et al., Exogenous ACE2 expression allows refractory cell lines to support severe acute respiratory syndrome coronavirus replication, J. Virol, doi:10.1128/JVI.79.6.3846-3850.2005
Munoz-Basagoiti, Perez-Zsolt, Leon, Blanc, Raich-Regue et al., Mouthwashes with CPC Reduce the Infectivity of SARS-CoV-2 Variants In Vitro, J. Dent. Res, doi:10.1177/00220345211029269
O'donnell, Thomas, Stanton, Maillard, Murphy et al., Potential Role of Oral Rinses Targeting the Viral Lipid Envelope in SARS-CoV-2 Infection, Function, doi:10.1093/function/zqaa002
Pelletier, Tessema, Frank, Westover, Brown et al., Efficacy of Povidone-Iodine Nasal and Oral Antiseptic Preparations Against Severe Acute Respiratory Syndrome-Coronavirus 2 (SARS-CoV-2), Ear. Nose. Throat. J, doi:10.1177/0145561320957237
Popkin, Zilka, Dimaano, Fujioka, Rackley et al., Cetylpyridinium Chloride (CPC) Exhibits Potent, Rapid Activity Against Influenza Viruses in vitro and in vivo, Pathog. Immun, doi:10.20411/pai.v2i2.200
Radford, Beighton, Nugent, Jackson, Effect of use of 0.05% cetylpyridinium chloride mouthwash on normal oral flora, J. Dent, doi:10.1016/S0300-5712(95)00116-6
Ramalingam, Cai, Wong, Twomey, Chen et al., Antiviral innate immune response in non-myeloid cells is augmented by chloride ions via an increase in intracellular hypochlorous acid levels, Sci. Rep, doi:10.1038/s41598-018-31936-y
Reimer, Wichelhaus, Schafer, Rudolph, Kramer et al., Antimicrobial effectiveness of povidone-iodine and consequences for new application areas, Dermatology, doi:10.1159/000057738
Rodríguez-Casanovas, La Rosa, Bello-Lemus, Rasperini, Acosta-Hoyos, Virucidal Activity of Different Mouthwashes Using a Novel Biochemical Assay, Healthcare, doi:10.3390/healthcare10010063
Rupel, Ottaviani, Gobbo, Contardo, Tirelli et al., A systematic review of therapeutical approaches in bisphosphonates-related osteonecrosis of the jaw (BRONJ), Oral Oncol
Russell, Biocide use and antibiotic resistance: The relevance of laboratory findings to clinical and environmental situations, Lancet Infect. Dis, doi:10.1016/S1473-3099(03)00833-8
Santos, Da Fonseca Orcina, Brito Reia, Ribeiro, Grotto et al., Virucidal Activity of the Antiseptic Mouthwash and Dental Gel Containing Anionic Phthalocyanine Derivative: In vitro Study, Clin. Cosmet. Investig. Dent, doi:10.2147/CCIDE.S315419
Santos, Teodoro, Sibelino, Novaes, Farias et al., Antibiofilm action of PHTALOX®-containing oral care formulations, J. Dent. Res
Shen, Niu, Wang, Huang, Wang et al., High-Throughput Screening and Identification of Potent Broad-Spectrum Inhibitors.s of Coronaviruses, J. Virol, doi:10.1128/JVI.00023-19
Shet, Westover, Hong, Igo, Cataldo et al., In vitro inactivation of SARS-CoV-2 using a povidone-iodine oral rinse, BMC Oral Health, doi:10.1186/s12903-022-02082-9
Shewale, Gelhaus, Ratcliff, Hernandez-Kapila, In vitro antiviral activity of stabilized chlorine dioxide containing oral care products, Oral Dis, doi:10.1111/odi.14044
Spearman, The method of 'Right and Wrong cases' ("Constant Stimuli") without Gauss's Formulae, Br. J. Psychol, doi:10.1111/j.2044-8295.1908.tb00176.x
Steinhauer, Meister, Todt, Krawczyk, Passvogel et al., Comparison of the in-vitro efficacy of different mouthwash solutions targeting SARS-CoV-2 based on the European Standard EN 14476, J. Hosp. Infect, doi:10.1016/j.jhin.2021.01.031
Sungnak, Huang, Becavin, Berg, Queen et al., SARS-CoV-2 entry factors are highly expressed in nasal epithelial cells together with innate immune genes, Nat. Med, doi:10.1038/s41591-020-0868-6
Tateyama-Makino, Abe-Yutori, Iwamoto, Tsutsumi, Tsuji et al., The inhibitory effects of toothpaste and mouthwash ingredients on the interaction between the SARS-CoV-2 spike protein and ACE2, and the protease activity of TMPRSS2 in vitro, PLoS ONE, doi:10.1371/journal.pone.0257705
Ting, Suzuki, SARS-CoV-2: Overview and Its Impact on Oral Health, Biomedicines, doi:10.3390/biomedicines9111690
Tiong, Hassandarvish, Bakar, Mohamed, Wan Sulaiman et al., The effectiveness of various gargle formulations and salt water against SARS-CoV-2, Sci. Rep, doi:10.1038/s41598-021-99866-w
To, Tsang, Leung, Tam, Wu et al., Temporal profiles of viral load in posterior oropharyngeal saliva samples and serum antibody responses during infection by SARS-CoV-2: An observational cohort study, Lancet Infect. Dis, doi:10.1016/S1473-3099(20)30196-1
Tseng, Tseng, Perrone, Worthy, Popov et al., Apical entry and release of severe acute respiratory syndrome-associated coronavirus in polarized Calu-3 lung epithelial cells, J. Virol, doi:10.1128/JVI.79.15.9470-9479.2005
Vogt, Hauser, Mueller, Bosse, Hopp, Efficacy of Conventional and Liposomal Povidone-Iodine in Infected Mesh Skin Grafts: An Exploratory Study, Infect. Dis. Ther, doi:10.1007/s40121-017-0172-z
Walsh, Jordan, Clyne, Rohde, Drummond et al., SARS-CoV-2 detection, viral load and infectivity over the course of an infection, J. Infect, doi:10.1016/j.jinf.2020.06.067
Walsh, Safety issues relating to the use of hydrogen peroxide in dentistry, Aust Dent. J, doi:10.1111/j.1834-7819.2000.tb00261.x
Wolfel, Corman, Guggemos, Seilmaier, Zange et al., Virological assessment of hospitalized patients with COVID-2019, Nature, doi:10.1038/s41586-020-2196-x
Yasumura, Kawakita, The research for the SV40 by means of tissue culture technique, Nippon. Rinsho
Zanelli, Ragazzi, De Marco, Chemical gastritis and colitis related to hydrogen peroxide mouthwash, Br. J. Clin. Pharmacol, doi:10.1111/bcp.13100
Zhao, Lin, Ran, Musa, Yang et al., Preliminary estimation of the basic reproduction number of novel coronavirus (2019-nCoV) in China, from 2019 to 2020: A data-driven analysis in the early phase of the outbreak, Int. J. Infect. Dis, doi:10.1016/j.ijid.2020.01.050
Zou, Ruan, Huang, Liang, Huang et al., SARS-CoV-2 Viral Load in Upper Respiratory Specimens of Infected Patients, N. Engl. J. Med, doi:10.1056/NEJMc2001737
DOI record:
{
"DOI": "10.3390/ijtm2030030",
"ISSN": [
"2673-8937"
],
"URL": "http://dx.doi.org/10.3390/ijtm2030030",
"abstract": "<jats:p>Oral antiseptic mouthwashes have been widely used for their antibacterial activity. As a result of the SARS-CoV-2 pandemic, the antiviral properties of these oral antiseptics have been aggressively studied. To demonstrate the direct antiviral activity of mouthwashes against SARS-CoV-2, this review will focus on the in vitro virucidal effects of these mouthwashes. Knowledge of the type, concentration, and exposure time of available mouthwashes can provide insights into effective protocols for their clinical use. With an understanding of the characteristics of each oral antiseptic mouthwash, proper mouthwash selection against SARS-CoV-2 may become a useful adjunct to personal protective equipment.</jats:p>",
"alternative-id": [
"ijtm2030030"
],
"author": [
{
"ORCID": "http://orcid.org/0000-0001-8503-2279",
"affiliation": [],
"authenticated-orcid": false,
"family": "Ting",
"given": "Miriam",
"sequence": "first"
},
{
"ORCID": "http://orcid.org/0000-0003-3348-8893",
"affiliation": [],
"authenticated-orcid": false,
"family": "Suzuki",
"given": "Jon B.",
"sequence": "additional"
}
],
"container-title": "International Journal of Translational Medicine",
"container-title-short": "IJTM",
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2022,
8,
1
]
],
"date-time": "2022-08-01T06:06:42Z",
"timestamp": 1659334002000
},
"deposited": {
"date-parts": [
[
2022,
8,
1
]
],
"date-time": "2022-08-01T06:20:05Z",
"timestamp": 1659334805000
},
"indexed": {
"date-parts": [
[
2022,
8,
1
]
],
"date-time": "2022-08-01T06:42:48Z",
"timestamp": 1659336168277
},
"is-referenced-by-count": 0,
"issue": "3",
"issued": {
"date-parts": [
[
2022,
7,
31
]
]
},
"journal-issue": {
"issue": "3",
"published-online": {
"date-parts": [
[
2022,
9
]
]
}
},
"language": "en",
"license": [
{
"URL": "https://creativecommons.org/licenses/by/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2022,
7,
31
]
],
"date-time": "2022-07-31T00:00:00Z",
"timestamp": 1659225600000
}
}
],
"link": [
{
"URL": "https://www.mdpi.com/2673-8937/2/3/30/pdf",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "1968",
"original-title": [],
"page": "387-397",
"prefix": "10.3390",
"published": {
"date-parts": [
[
2022,
7,
31
]
]
},
"published-online": {
"date-parts": [
[
2022,
7,
31
]
]
},
"publisher": "MDPI AG",
"reference": [
{
"DOI": "10.3390/biomedicines9111690",
"doi-asserted-by": "publisher",
"key": "ref1"
},
{
"DOI": "10.1016/j.ijid.2020.01.050",
"doi-asserted-by": "publisher",
"key": "ref2"
},
{
"DOI": "10.1056/NEJMc2001737",
"doi-asserted-by": "publisher",
"key": "ref3"
},
{
"DOI": "10.1038/s41591-020-0868-6",
"doi-asserted-by": "publisher",
"key": "ref4"
},
{
"DOI": "10.14219/jada.archive.2004.0207",
"doi-asserted-by": "publisher",
"key": "ref5"
},
{
"DOI": "10.1177/00220345690480012401",
"doi-asserted-by": "publisher",
"key": "ref6"
},
{
"DOI": "10.1016/j.adaj.2016.02.011",
"doi-asserted-by": "publisher",
"key": "ref7"
},
{
"DOI": "10.3390/microorganisms8111704",
"doi-asserted-by": "publisher",
"key": "ref8"
},
{
"DOI": "10.1038/s41586-020-2196-x",
"doi-asserted-by": "publisher",
"key": "ref9"
},
{
"DOI": "10.1016/S1473-3099(20)30196-1",
"doi-asserted-by": "publisher",
"key": "ref10"
},
{
"DOI": "10.1001/jama.2020.2565",
"doi-asserted-by": "publisher",
"key": "ref11"
},
{
"DOI": "10.2147/RMHP.S284557",
"doi-asserted-by": "publisher",
"key": "ref12"
},
{
"DOI": "10.3390/microorganisms9040793",
"doi-asserted-by": "publisher",
"key": "ref13"
},
{
"DOI": "10.1099/jgv.0.001578",
"doi-asserted-by": "publisher",
"key": "ref14"
},
{
"DOI": "10.1007/s40121-020-00316-3",
"doi-asserted-by": "publisher",
"key": "ref15"
},
{
"DOI": "10.1111/jopr.13220",
"doi-asserted-by": "publisher",
"key": "ref16"
},
{
"DOI": "10.1111/jopr.13209",
"doi-asserted-by": "publisher",
"key": "ref17"
},
{
"DOI": "10.1038/s41415-020-2402-0",
"doi-asserted-by": "publisher",
"key": "ref18"
},
{
"DOI": "10.1159/000089211",
"doi-asserted-by": "publisher",
"key": "ref19"
},
{
"DOI": "10.3390/microorganisms9030521",
"doi-asserted-by": "publisher",
"key": "ref20"
},
{
"DOI": "10.1016/j.ajoms.2021.02.002",
"doi-asserted-by": "publisher",
"key": "ref21"
},
{
"DOI": "10.1093/infdis/jiaa471",
"doi-asserted-by": "publisher",
"key": "ref22"
},
{
"DOI": "10.5005/jp-journals-10029-1209",
"doi-asserted-by": "publisher",
"key": "ref23"
},
{
"DOI": "10.1177/0145561320957237",
"doi-asserted-by": "publisher",
"key": "ref24"
},
{
"DOI": "10.2147/CCIDE.S315419",
"doi-asserted-by": "publisher",
"key": "ref25"
},
{
"DOI": "10.1186/s12903-022-02082-9",
"doi-asserted-by": "publisher",
"key": "ref26"
},
{
"DOI": "10.1111/odi.14044",
"doi-asserted-by": "publisher",
"key": "ref27"
},
{
"DOI": "10.1016/j.jhin.2021.01.031",
"doi-asserted-by": "publisher",
"key": "ref28"
},
{
"DOI": "10.1038/s41598-021-99866-w",
"doi-asserted-by": "publisher",
"key": "ref29"
},
{
"DOI": "10.1007/s40121-018-0200-7",
"doi-asserted-by": "publisher",
"key": "ref30"
},
{
"DOI": "10.1111/j.2044-8295.1908.tb00176.x",
"doi-asserted-by": "publisher",
"key": "ref31"
},
{
"DOI": "10.1007/BF01863914",
"doi-asserted-by": "publisher",
"key": "ref32"
},
{
"DOI": "10.1016/j.oraloncology.2014.08.016",
"doi-asserted-by": "publisher",
"key": "ref33"
},
{
"article-title": "The research for the SV40 by means of tissue culture technique",
"author": "Yasumura",
"first-page": "1201",
"journal-title": "Nippon. Rinsho.",
"key": "ref34",
"volume": "21",
"year": "1963"
},
{
"DOI": "10.3201/eid2609.201495",
"doi-asserted-by": "publisher",
"key": "ref35"
},
{
"DOI": "10.1073/pnas.2002589117",
"doi-asserted-by": "publisher",
"key": "ref36"
},
{
"DOI": "10.1128/JVI.79.15.9470-9479.2005",
"doi-asserted-by": "publisher",
"key": "ref37"
},
{
"DOI": "10.1128/JVI.79.6.3846-3850.2005",
"doi-asserted-by": "publisher",
"key": "ref38"
},
{
"DOI": "10.3201/eid1201.050496",
"doi-asserted-by": "publisher",
"key": "ref39"
},
{
"DOI": "10.1128/JCM.42.7.3196-3206.2004",
"doi-asserted-by": "publisher",
"key": "ref40"
},
{
"DOI": "10.1128/iai.8.3.309-316.1973",
"doi-asserted-by": "publisher",
"key": "ref41"
},
{
"DOI": "10.1001/jamaoto.2020.3053",
"doi-asserted-by": "publisher",
"key": "ref42"
},
{
"DOI": "10.1159/000057738",
"doi-asserted-by": "publisher",
"key": "ref43"
},
{
"DOI": "10.4103/0973-1482.39597",
"doi-asserted-by": "publisher",
"key": "ref44"
},
{
"DOI": "10.1210/jcem-66-3-632",
"doi-asserted-by": "publisher",
"key": "ref45"
},
{
"article-title": "[The use of Betadine antiseptic in the treatment of oral surgical, parodontological and oral mucosal diseases]",
"author": "Kovesi",
"first-page": "243",
"journal-title": "Fogorv. Szle.",
"key": "ref46",
"volume": "92",
"year": "1999"
},
{
"article-title": "The effect of 1% povidone iodine as a pre-procedural mouthrinse in 20 patients with varying degrees of oral hygiene",
"author": "Domingo",
"first-page": "31",
"journal-title": "J. Philipp. Dent. Assoc.",
"key": "ref47",
"volume": "48",
"year": "1996"
},
{
"article-title": "The relationship between autoimmune thyroid disease and iodine intake: A review",
"author": "Foley",
"first-page": "53",
"journal-title": "Endokrynol. Pol.",
"key": "ref48",
"volume": "43",
"year": "1992"
},
{
"DOI": "10.1538/expanim.46.197",
"doi-asserted-by": "publisher",
"key": "ref49"
},
{
"DOI": "10.1111/jpc.12232",
"doi-asserted-by": "publisher",
"key": "ref50"
},
{
"DOI": "10.1016/j.jhin.2020.01.022",
"doi-asserted-by": "publisher",
"key": "ref51"
},
{
"article-title": "Interim Infection Prevention and Control Guidance for Dental Settings during the COVID-19 Response",
"author": "The Centers for Disease Control and Prevention (CDC)",
"key": "ref52"
},
{
"DOI": "10.1007/s40121-017-0172-z",
"doi-asserted-by": "publisher",
"key": "ref53"
},
{
"DOI": "10.1093/function/zqaa002",
"doi-asserted-by": "publisher",
"key": "ref54"
},
{
"article-title": "Stable Compositions of Uncomplexed Iodine and Methods of Use",
"author": "Elliot",
"journal-title": "U.S. Patent",
"key": "ref55",
"year": "2018"
},
{
"DOI": "10.20411/pai.v2i2.200",
"doi-asserted-by": "publisher",
"key": "ref56"
},
{
"DOI": "10.1128/JVI.00023-19",
"doi-asserted-by": "publisher",
"key": "ref57"
},
{
"DOI": "10.1177/00220345211029269",
"doi-asserted-by": "publisher",
"key": "ref58"
},
{
"DOI": "10.1902/jop.1996.67.6.572",
"doi-asserted-by": "publisher",
"key": "ref59"
},
{
"DOI": "10.3390/healthcare10010063",
"doi-asserted-by": "publisher",
"key": "ref60"
},
{
"DOI": "10.1002/jmv.26954",
"doi-asserted-by": "publisher",
"key": "ref61"
},
{
"DOI": "10.7860/JCDR/2016/18576.8330",
"doi-asserted-by": "publisher",
"key": "ref62"
},
{
"DOI": "10.1007/s10266-021-00660-x",
"doi-asserted-by": "publisher",
"key": "ref63"
},
{
"DOI": "10.1111/j.1834-7819.2000.tb00261.x",
"doi-asserted-by": "publisher",
"key": "ref64"
},
{
"DOI": "10.1016/j.mehy.2020.109910",
"doi-asserted-by": "publisher",
"key": "ref65"
},
{
"DOI": "10.1111/bcp.13100",
"doi-asserted-by": "publisher",
"key": "ref66"
},
{
"DOI": "10.1016/0030-4220(58)90228-7",
"doi-asserted-by": "publisher",
"key": "ref67"
},
{
"article-title": "Antibiofilm action of PHTALOX®-containing oral care formulations",
"author": "Santos",
"first-page": "3326",
"journal-title": "J. Dent. Res.",
"key": "ref68",
"volume": "99",
"year": "2020"
},
{
"article-title": "An in vitro comparative study determining bactericidal activity of stabilized chlorine dioxide and other oral rinses",
"author": "Drake",
"first-page": "1",
"journal-title": "J. Clin. Dent.",
"key": "ref69",
"volume": "22",
"year": "2011"
},
{
"article-title": "Evidence for the microbicidal activity of a chlorine dioxide-containing oral rinse formulation in vivo",
"author": "Grootveld",
"first-page": "67",
"journal-title": "J. Clin. Dent.",
"key": "ref70",
"volume": "12",
"year": "2001"
},
{
"article-title": "Efficacy of stabilized chlorine dioxide-based unflavored mouthwash in reducing oral malodor: An 8-week randomized controlled study",
"author": "Lee",
"first-page": "309",
"journal-title": "Am. J. Dent.",
"key": "ref71",
"volume": "31",
"year": "2018"
},
{
"DOI": "10.1038/s41598-018-31936-y",
"doi-asserted-by": "publisher",
"key": "ref72"
},
{
"DOI": "10.1371/journal.pone.0257705",
"doi-asserted-by": "publisher",
"key": "ref73"
},
{
"DOI": "10.1016/j.virol.2021.02.007",
"doi-asserted-by": "publisher",
"key": "ref74"
},
{
"DOI": "10.1016/j.micinf.2021.104832",
"doi-asserted-by": "publisher",
"key": "ref75"
},
{
"DOI": "10.1016/j.cell.2020.02.052",
"doi-asserted-by": "publisher",
"key": "ref76"
},
{
"DOI": "10.1111/j.1600-0765.1970.tb00696.x",
"doi-asserted-by": "publisher",
"key": "ref77"
},
{
"DOI": "10.1111/j.1600-0722.1976.tb00495.x",
"doi-asserted-by": "publisher",
"key": "ref78"
},
{
"DOI": "10.1111/j.1600-051X.1993.tb00345.x",
"doi-asserted-by": "publisher",
"key": "ref79"
},
{
"DOI": "10.1016/j.jhin.2016.08.018",
"doi-asserted-by": "publisher",
"key": "ref80"
},
{
"DOI": "10.3390/antibiotics7040110",
"doi-asserted-by": "publisher",
"key": "ref81"
},
{
"DOI": "10.3390/antibiotics8010013",
"doi-asserted-by": "publisher",
"key": "ref82"
},
{
"DOI": "10.1016/S1473-3099(03)00833-8",
"doi-asserted-by": "publisher",
"key": "ref83"
},
{
"DOI": "10.3389/fmicb.2019.00587",
"doi-asserted-by": "publisher",
"key": "ref84"
},
{
"DOI": "10.1016/S0300-5712(95)00116-6",
"doi-asserted-by": "publisher",
"key": "ref85"
},
{
"DOI": "10.1016/j.jinf.2020.06.067",
"doi-asserted-by": "publisher",
"key": "ref86"
},
{
"article-title": "Saliva between normal and pathological. Important factors in determining systemic and oral health",
"author": "Iorgulescu",
"first-page": "303",
"journal-title": "J. Med. Life",
"key": "ref87",
"volume": "2",
"year": "2009"
},
{
"DOI": "10.1128/JCM.37.6.1771-1776.1999",
"doi-asserted-by": "publisher",
"key": "ref88"
},
{
"DOI": "10.1093/ofid/ofz032",
"doi-asserted-by": "publisher",
"key": "ref89"
},
{
"DOI": "10.1016/S2666-5247(20)30003-3",
"doi-asserted-by": "publisher",
"key": "ref90"
}
],
"reference-count": 90,
"references-count": 90,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.mdpi.com/2673-8937/2/3/30"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subtitle": [],
"title": "The In Vitro Virucidal Effects of Mouthwashes on SARS-CoV-2",
"type": "journal-article",
"volume": "2"
}
