Lactoferrin Inhibition of the Complex Formation between ACE2 Receptor and SARS CoV-2 Recognition Binding Domain
et al., International Journal of Molecular Sciences, doi:10.3390/ijms23105436, May 2022
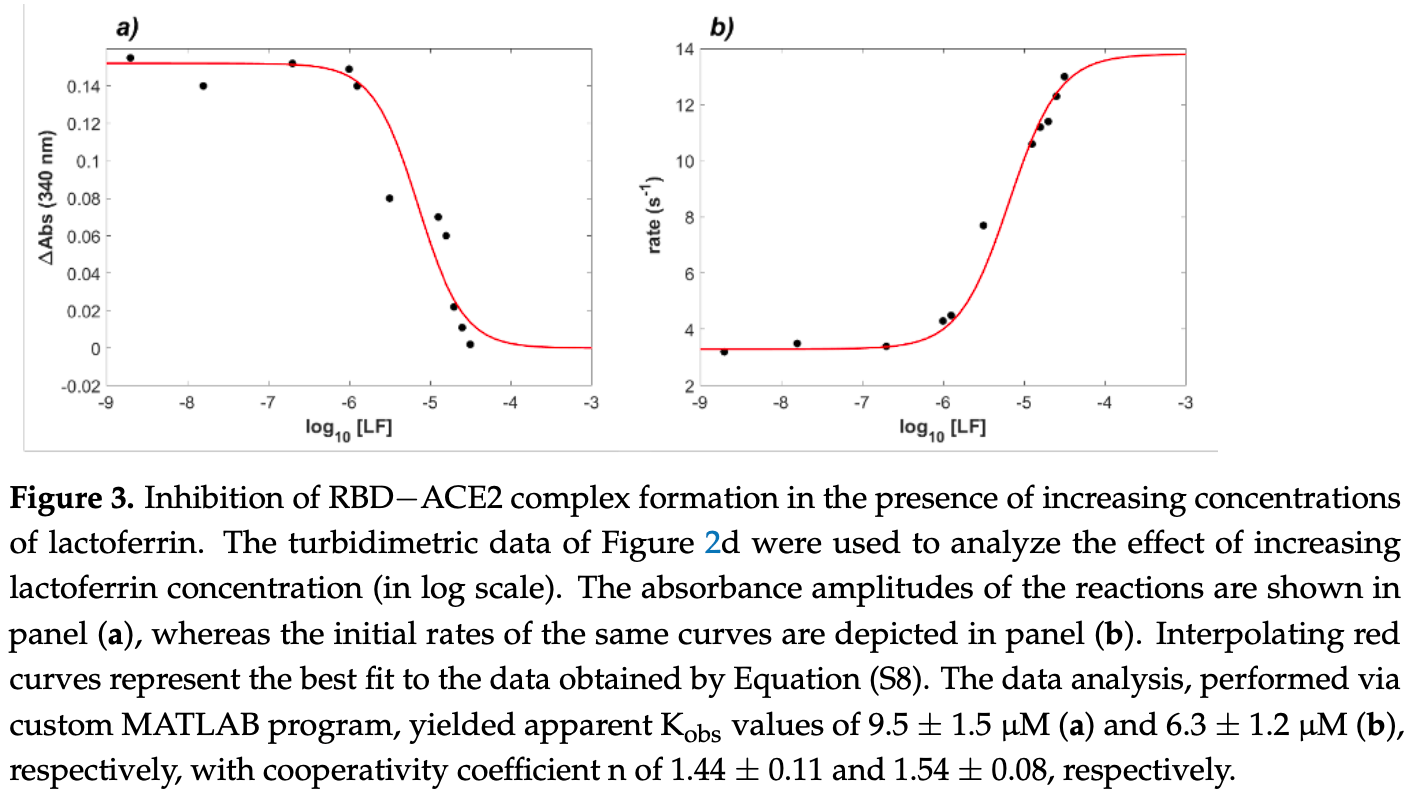
Biolayer interferometry and turbidimetry study showing lactoferrin inhibits ACE2 - SARS-CoV-2 RBD binding.
18 preclinical studies support the efficacy of lactoferrin for COVID-19:
1.
da Silva et al., Immunomodulatory effect of bovine lactoferrin during SARS-CoV-2 infection, Frontiers in Immunology, doi:10.3389/fimmu.2024.1456634.
2.
Cutone et al., Lactoferrin binding to Sars-CoV-2 Spike glycoprotein protects host from infection, inflammation and iron dysregulation., Research Square, doi:10.21203/rs.3.rs-1605740/v1.
3.
Miotto et al., Molecular Mechanisms Behind Anti SARS-CoV-2 Action of Lactoferrin, Frontiers in Molecular Biosciences, doi:10.3389/fmolb.2021.607443.
4.
Babulic et al., Lactoferrin Binds through Its N-Terminus to the Receptor-Binding Domain of the SARS-CoV-2 Spike Protein, Pharmaceuticals, doi:10.3390/ph17081021.
5.
Yathindranath et al., Lipid Nanoparticle-Based Inhibitors for SARS-CoV-2 Host Cell Infection, International Journal of Nanomedicine, doi:10.2147/IJN.S448005.
6.
Alves et al., Inhibition of SARS-CoV-2 Infection in Vero Cells by Bovine Lactoferrin under Different Iron-Saturation States, Pharmaceuticals, doi:10.3390/ph16101352.
7.
Kobayashi-Sakamoto et al., Bovine lactoferrin suppresses the cathepsin-dependent pathway of SARS-CoV-2 entry in vitro, International Dairy Journal, doi:10.1016/j.idairyj.2023.105805.
8.
Andreu et al., Liposomal Lactoferrin Exerts Antiviral Activity against HCoV-229E and SARS-CoV-2 Pseudoviruses In Vitro, Viruses, doi:10.3390/v15040972.
9.
Yazawa et al., Evaluation of SARS-CoV-2 isolation in cell culture from nasal/nasopharyngeal swabs or saliva specimens of patients with COVID-19, Research Square, doi:10.21203/rs.3.rs-2676422/v1.
10.
Piacentini et al., Lactoferrin Inhibition of the Complex Formation between ACE2 Receptor and SARS CoV-2 Recognition Binding Domain, International Journal of Molecular Sciences, doi:10.3390/ijms23105436.
11.
Ostrov et al., Highly Specific Sigma Receptor Ligands Exhibit Anti-Viral Properties in SARS-CoV-2 Infected Cells, Pathogens, doi:10.3390/pathogens10111514.
12.
Mirabelli et al., Morphological cell profiling of SARS-CoV-2 infection identifies drug repurposing candidates for COVID-19, Proceedings of the National Academy of Sciences, doi:10.1073/pnas.2105815118.
13.
Salaris et al., Protective Effects of Lactoferrin against SARS-CoV-2 Infection In Vitro, Nutrients, doi:10.3390/nu13020328.
Piacentini et al., 13 May 2022, Italy, peer-reviewed, 10 authors.
Contact: giacomo.parisi@iit.it (corresponding author), roberta.piacentini@uniroma1.it, centi.laura95@gmail.com, alberto.boffi@uniroma1.it, miottomattia1@gmail.com, edoardo.milanetti@uniroma1.it, lorenzo.dirienzo@iit.it, martina.pitea@iit.it, giancarlo.ruocco@iit.it, paolopiazza@edifinstruments.com.
In vitro studies are an important part of preclinical research, however results may be very different in vivo.
Lactoferrin Inhibition of the Complex Formation between ACE2 Receptor and SARS CoV-2 Recognition Binding Domain
International Journal of Molecular Sciences, doi:10.3390/ijms23105436
The present investigation focuses on the analysis of the interactions among human lactoferrin (LF), SARS-CoV-2 receptor-binding domain (RBD) and human angiotensin-converting enzyme 2 (ACE2) receptor in order to assess possible mutual interactions that could provide a molecular basis of the reported preventative effect of lactoferrin against CoV-2 infection. In particular, kinetic and thermodynamic parameters for the pairwise interactions among the three proteins were measured via two independent techniques, biolayer interferometry and latex nanoparticle-enhanced turbidimetry. The results obtained clearly indicate that LF is able to bind the ACE2 receptor ectodomain with significantly high affinity, whereas no binding to the RBD was observed up to the maximum "physiological" lactoferrin concentration range. Lactoferrin, above 1 µM concentration, thus appears to directly interfere with RBD-ACE2 binding, bringing about a measurable, up to 300-fold increase of the K D value relative to RBD-ACE2 complex formation.
References
Baker, Baker, Molecular structure, binding properties and dynamics of Lactoferrin, Cell. Mol. Life Sci, doi:10.1007/s00018-005-5368-9
Berlutti, Pantanella, Natalizi, Frioni, Paesano et al., Antiviral Properties of Lactoferrin-A Natural Immunity Molecule, Molecules, doi:10.3390/molecules16086992
Bishop, Gagneux, Evolution of carbohydrate antigens-microbial forces shaping host glycomes?, Glycobiology, doi:10.1093/glycob/cwm005
Bò, Miotto, Di Rienzo, Milanetti, Ruocco, Exploring the Association Between Sialic Acid and SARS-CoV-2 Spike Protein Through a Molecular Dynamics-Based Approach, Front. Med Technol, doi:10.3389/fmedt.2020.614652
Campione, Cosio, Rosa, Lanna, Di Girolamo et al., Lactoferrin as Protective Natural Barrier of Respiratory and Intestinal Mucosa against Coronavirus Infection and Inflammation, Int. J. Mol. Sci, doi:10.3390/ijms21144903
Campione, Lanna, Cosio, Rosa, Conte et al., Lactoferrin Against SARS-CoV-2: In Vitro and In Silico Evidences, Front. Pharmacol, doi:10.3389/fphar.2021.666600
Chang, Ng, Sun, Lactoferrin as potential preventative and adjunct treatment for COVID-19, Int. J. Antimicrob. Agents, doi:10.1016/j.ijantimicag.2020.106118
Coletta, Amendola, Numerical Modelling of the Optical Properties of Plasmonic and Latex Nanoparticles to Improve the Detection Limit of Immuno-Turbidimetric Assays, Nanomaterials, doi:10.3390/nano11051147
Concepcion, Witte, Wartchow, Choo, Yao et al., Label-Free Detection of Biomolecular Interactions Using BioLayer Interferometry for Kinetic Characterization, Comb. Chem. High Throughput Screen, doi:10.2174/138620709789104915
Cölfen, Völkel, Eda, Kobold, Kaufmann et al., Mechanism of Nanoparticle-Enhanced Turbidimetric Assays Applying Nanoparticles of Different Size and Immunoreactivity, Langmuir
Dzimianski, Lorig-Roach, O'rourke, Alexander, Kimmey et al., Rapid and sensitive detection of SARS-CoV-2 antibodies by biolayer interferometry, Sci. Rep, doi:10.1038/s41598-020-78895-x
Giansanti, Panella, Leboffe, Antonini, Lactoferrin from Milk: Nutraceutical and Pharmacological Properties, Pharmaceuticals, doi:10.3390/ph9040061
Gurnani, Lunn, Perrier, Synthesis of mannosylated and PEGylated nanoparticles via RAFT emulsion polymerisation, and investigation of particle-lectin aggregation using turbidimetric and DLS techniques, Polymer, doi:10.1016/j.polymer.2016.08.093
Harmsen, Swart, De Béthune, Pauwels, De Clercq et al., Antiviral Effects of Plasma and Milk Proteins: Lactoferrin Shows Potent Activity against Both Human Immunodeficiency Virus and Human Cytomegalovirus Replication In Vitro, J. Infect. Dis, doi:10.1093/infdis/172.2.380
Hulswit, Lang, Bakkers, Li, Li et al., Human coronaviruses OC43 and HKU1 bind to 9-O -acetylated sialic acids via a conserved receptor-binding site in spike protein domain A, Proc. Natl. Acad. Sci, doi:10.1073/pnas.1809667116
Kamat, Rafique, Designing binding kinetic assay on the bio-layer interferometry (BLI) biosensor to characterize antibodyantigen interactions, Anal. Biochem, doi:10.1016/j.ab.2017.08.002
Kell, Heyden, Pretorius, The Biology of Lactoferrin, an Iron-Binding Protein That Can Help Defend Against Viruses and Bacteria, Front. Immunol, doi:10.3389/fimmu.2020.01221
Kumaraswamy, Tobias, Label-Free Kinetic Analysis of an Antibody-Antigen Interaction Using Biolayer Interferometry, doi:10.1007/978-1-4939-2425-7_10
Lang, Yang, Deng, Liu, Yang et al., Inhibition of SARS Pseudovirus Cell Entry by Lactoferrin Binding to Heparan Sulfate Proteoglycans, PLoS ONE, doi:10.1371/journal.pone.0023710
Langford-Smith, Day, Bishop, Clark, Complementing the Sugar Code: Role of GAGs and Sialic Acid in Complement Regulation, Front. Immunol, doi:10.3389/fimmu.2015.00025
Mahmood, Poma, Okazaki, Optimizing G ō-MARTINI Coarse-Grained Model for F-BAR Protein on Lipid Membrane, Front. Mol. Biosci, doi:10.3389/fmolb.2021.619381
Malatesta, The study of bimolecular reactions under non-pseudo-first order conditions, Biophys. Chem, doi:10.1016/j.bpc.2005.04.006
Martínez, Cooper, Poma, Guzman, Free Energies of the Disassembly of Viral Capsids from a Multiscale Molecular Simulation Approach, J. Chem. Inf. Model, doi:10.1021/acs.jcim.9b00883
Milanetti, Miotto, Di Rienzo, Monti, Gosti et al., Zernike polynomial expansion: Finding the protein-protein binding regions, Comput. Struct. Biotechnol. J, doi:10.1016/j.csbj.2020.11.051
Milanetti, Miotto, Di Rienzo, Nagaraj, Monti et al., In-Silico Evidence for a Two Receptor Based Strategy of SARS-CoV-2, Front. Mol. Biosci, doi:10.3389/fmolb.2021.690655
Miotto, Di Rienzo, Bò, Boffi, Ruocco et al., Molecular Mechanisms Behind Anti SARS-CoV-2 Action of Lactoferrin, Front. Mol. Biosci, doi:10.3389/fmolb.2021.607443
Miotto, Di Rienzo, Gosti, Milanetti, Ruocco, Does blood type affect the COVID-19 infection pattern?, PLoS ONE, doi:10.1371/journal.pone.0251535
Müller-Esparza, Osorio-Valeriano, Steube, Thanbichler, Randau, Bio-Layer Interferometry Analysis of the Target Binding Activity of CRISPR-Cas Effector Complexes, Front. Mol. Biosci, doi:10.3389/fmolb.2020.00098
Nedyalkova, Vasighi, Sappati, Kumar, Madurga et al., Inhibition Ability of Natural Compounds on Receptor-Binding Domain of SARS-CoV2: An in Silico Approach, Pharmaceuticals, doi:10.3390/ph14121328
Odolczyk, Marzec, Winiewska-Szajewska, Pozna Ński, Zielenkiewicz, Native Structure-Based Peptides as Potential Protein-Protein Interaction Inhibitors of SARS-CoV-2 Spike Protein and Human ACE2 Receptor, Molecules, doi:10.3390/molecules26082157
Petersen, Strategies Using Bio-Layer Interferometry Biosensor Technology for Vaccine Research and Development, Biosensors, doi:10.3390/bios7040049
Puddu, Borghi, Gessani, Valenti, Belardelli et al., Antiviral effect of bovine lactoferrin saturated with metal ions on early steps of human immunodeficiency virus type 1 infection, Int. J. Biochem. Cell Biol, doi:10.1016/S1357-2725(98)00066-1
Rascón-Cruz, Espinoza-Sánchez, Siqueiros-Cendón, Nakamura-Bencomo, Arévalo-Gallegos et al., Lactoferrin: A Glycoprotein Involved in Immunomodulation, Anticancer, and Antimicrobial Processes, Molecules, doi:10.3390/molecules26010205
Redwan, Uversky, El-Fakharany, Al-Mehdar, Potential lactoferrin activity against pathogenic viruses, Comptes Rendus Biol, doi:10.1016/j.crvi.2014.08.003
Richards, Areas, volumes, packing and protein structure, Annu. Rev. Biophys. Bioeng, doi:10.1146/annurev.bb.06.060177.001055
Rosa, Cutone, Lepanto, Paesano, Valenti, Lactoferrin: A Natural Glycoprotein Involved in Iron and Inflammatory Homeostasis, Int. J. Mol. Sci, doi:10.3390/ijms18091985
Rosa, Tripepi, Naldi, Aimati, Santangeli et al., Ambulatory COVID-19 Patients Treated with Lactoferrin as a Supplementary Antiviral Agent: A Preliminary Study, J. Clin. Med, doi:10.3390/jcm10184276
Saponaro, Rutigliano, Sestito, Bandini, Storti et al., ACE2 in the Era of SARS-CoV-2: Controversies and Novel Perspectives, Front. Mol. Biosci, doi:10.3389/fmolb.2020.588618
Superti, Siciliano, Rega, Giansanti, Valenti et al., Involvement of bovine lactoferrin metal saturation, sialic acid and protein fragments in the inhibition of rotavirus infection, Biochim. Biophys. Acta, doi:10.1016/S0304-4165(01)00178-7
Tortorici, Walls, Lang, Wang, Li et al., Structural basis for human coronavirus attachment to sialic acid receptors, Nat. Struct. Mol. Biol, doi:10.1038/s41594-019-0233-y
Varki, Biological roles of glycans, Glycobiology, doi:10.1093/glycob/cww086
Wang, Wang, Wang, Luo, Wan et al., Lactoferrin for the treatment of COVID-19 (Review), Exp. Ther. Med, doi:10.3892/etm.2020.9402
Yan, Zhang, Li, Xia, Guo et al., Structural basis for the recognition of SARS-CoV-2 by full-length human ACE2, Science, doi:10.1126/science.abb2762
Yi, Kaneko, Yu, Murakami, Hepatitis C virus envelope proteins bind lactoferrin, J. Virol
Zhou, Yang, Wang, Hu, Zhang et al., A pneumonia outbreak associated with a new coronavirus of probable bat origin, Nature, doi:10.1038/s41586-020-2012-7
Zhu, Zhang, Wang, Li, Yang et al., A Novel Coronavirus from Patients with Pneumonia in China, N. Engl. J. Med, doi:10.1056/NEJMoa2001017
DOI record:
{
"DOI": "10.3390/ijms23105436",
"ISSN": [
"1422-0067"
],
"URL": "http://dx.doi.org/10.3390/ijms23105436",
"abstract": "<jats:p>The present investigation focuses on the analysis of the interactions among human lactoferrin (LF), SARS-CoV-2 receptor-binding domain (RBD) and human angiotensin-converting enzyme 2 (ACE2) receptor in order to assess possible mutual interactions that could provide a molecular basis of the reported preventative effect of lactoferrin against CoV-2 infection. In particular, kinetic and thermodynamic parameters for the pairwise interactions among the three proteins were measured via two independent techniques, biolayer interferometry and latex nanoparticle-enhanced turbidimetry. The results obtained clearly indicate that LF is able to bind the ACE2 receptor ectodomain with significantly high affinity, whereas no binding to the RBD was observed up to the maximum “physiological” lactoferrin concentration range. Lactoferrin, above 1 µM concentration, thus appears to directly interfere with RBD–ACE2 binding, bringing about a measurable, up to 300-fold increase of the KD value relative to RBD–ACE2 complex formation.</jats:p>",
"alternative-id": [
"ijms23105436"
],
"author": [
{
"ORCID": "http://orcid.org/0000-0003-1381-9560",
"affiliation": [],
"authenticated-orcid": false,
"family": "Piacentini",
"given": "Roberta",
"sequence": "first"
},
{
"ORCID": "http://orcid.org/0000-0002-8253-940X",
"affiliation": [],
"authenticated-orcid": false,
"family": "Centi",
"given": "Laura",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-0043-8921",
"affiliation": [],
"authenticated-orcid": false,
"family": "Miotto",
"given": "Mattia",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Milanetti",
"given": "Edoardo",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Di Rienzo",
"given": "Lorenzo",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-3982-4141",
"affiliation": [],
"authenticated-orcid": false,
"family": "Pitea",
"given": "Martina",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Piazza",
"given": "Paolo",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-2762-9533",
"affiliation": [],
"authenticated-orcid": false,
"family": "Ruocco",
"given": "Giancarlo",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Boffi",
"given": "Alberto",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-3064-0928",
"affiliation": [],
"authenticated-orcid": false,
"family": "Parisi",
"given": "Giacomo",
"sequence": "additional"
}
],
"container-title": "International Journal of Molecular Sciences",
"container-title-short": "IJMS",
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2022,
5,
13
]
],
"date-time": "2022-05-13T12:37:02Z",
"timestamp": 1652445422000
},
"deposited": {
"date-parts": [
[
2022,
5,
13
]
],
"date-time": "2022-05-13T13:26:39Z",
"timestamp": 1652448399000
},
"indexed": {
"date-parts": [
[
2022,
5,
14
]
],
"date-time": "2022-05-14T12:15:39Z",
"timestamp": 1652530539449
},
"is-referenced-by-count": 0,
"issue": "10",
"issued": {
"date-parts": [
[
2022,
5,
13
]
]
},
"journal-issue": {
"issue": "10",
"published-online": {
"date-parts": [
[
2022,
5
]
]
}
},
"language": "en",
"license": [
{
"URL": "https://creativecommons.org/licenses/by/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2022,
5,
13
]
],
"date-time": "2022-05-13T00:00:00Z",
"timestamp": 1652400000000
}
}
],
"link": [
{
"URL": "https://www.mdpi.com/1422-0067/23/10/5436/pdf",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "1968",
"original-title": [],
"page": "5436",
"prefix": "10.3390",
"published": {
"date-parts": [
[
2022,
5,
13
]
]
},
"published-online": {
"date-parts": [
[
2022,
5,
13
]
]
},
"publisher": "MDPI AG",
"reference": [
{
"DOI": "10.1016/j.crvi.2014.08.003",
"doi-asserted-by": "publisher",
"key": "ref1"
},
{
"DOI": "10.1007/s00018-005-5368-9",
"doi-asserted-by": "publisher",
"key": "ref2"
},
{
"DOI": "10.3390/ph9040061",
"doi-asserted-by": "publisher",
"key": "ref3"
},
{
"DOI": "10.3390/ijms18091985",
"doi-asserted-by": "publisher",
"key": "ref4"
},
{
"DOI": "10.1093/glycob/cww086",
"doi-asserted-by": "publisher",
"key": "ref5"
},
{
"DOI": "10.1093/glycob/cwm005",
"doi-asserted-by": "publisher",
"key": "ref6"
},
{
"DOI": "10.1371/journal.pone.0251535",
"doi-asserted-by": "publisher",
"key": "ref7"
},
{
"DOI": "10.1128/jvi.71.8.5997-6002.1997",
"doi-asserted-by": "publisher",
"key": "ref8"
},
{
"DOI": "10.1093/infdis/172.2.380",
"doi-asserted-by": "publisher",
"key": "ref9"
},
{
"DOI": "10.1016/S0304-4165(01)00178-7",
"doi-asserted-by": "publisher",
"key": "ref10"
},
{
"DOI": "10.1016/S1357-2725(98)00066-1",
"doi-asserted-by": "publisher",
"key": "ref11"
},
{
"DOI": "10.3390/molecules16086992",
"doi-asserted-by": "publisher",
"key": "ref12"
},
{
"DOI": "10.3390/ijms21144903",
"doi-asserted-by": "publisher",
"key": "ref13"
},
{
"DOI": "10.3892/etm.2020.9402",
"doi-asserted-by": "publisher",
"key": "ref14"
},
{
"DOI": "10.3390/jcm10184276",
"doi-asserted-by": "publisher",
"key": "ref15"
},
{
"DOI": "10.1016/j.ijantimicag.2020.106118",
"doi-asserted-by": "publisher",
"key": "ref16"
},
{
"DOI": "10.3390/ph14121328",
"doi-asserted-by": "publisher",
"key": "ref17"
},
{
"DOI": "10.3390/molecules26082157",
"doi-asserted-by": "publisher",
"key": "ref18"
},
{
"DOI": "10.1056/NEJMoa2001017",
"doi-asserted-by": "publisher",
"key": "ref19"
},
{
"DOI": "10.1038/s41586-020-2012-7",
"doi-asserted-by": "publisher",
"key": "ref20"
},
{
"DOI": "10.1126/science.abb2762",
"doi-asserted-by": "publisher",
"key": "ref21"
},
{
"DOI": "10.1371/journal.pone.0023710",
"doi-asserted-by": "publisher",
"key": "ref22"
},
{
"DOI": "10.3389/fimmu.2015.00025",
"doi-asserted-by": "publisher",
"key": "ref23"
},
{
"DOI": "10.1073/pnas.1809667116",
"doi-asserted-by": "publisher",
"key": "ref24"
},
{
"DOI": "10.1016/j.csbj.2020.11.051",
"doi-asserted-by": "publisher",
"key": "ref25"
},
{
"DOI": "10.3389/fmolb.2021.607443",
"doi-asserted-by": "publisher",
"key": "ref26"
},
{
"DOI": "10.1021/la025983n",
"doi-asserted-by": "publisher",
"key": "ref27"
},
{
"DOI": "10.3390/nano11051147",
"doi-asserted-by": "publisher",
"key": "ref28"
},
{
"DOI": "10.1016/j.polymer.2016.08.093",
"doi-asserted-by": "publisher",
"key": "ref29"
},
{
"DOI": "10.3389/fmolb.2020.588618",
"doi-asserted-by": "publisher",
"key": "ref30"
},
{
"DOI": "10.3390/molecules26010205",
"doi-asserted-by": "publisher",
"key": "ref31"
},
{
"DOI": "10.3389/fimmu.2020.01221",
"doi-asserted-by": "publisher",
"key": "ref32"
},
{
"DOI": "10.1038/s41594-019-0233-y",
"doi-asserted-by": "publisher",
"key": "ref33"
},
{
"DOI": "10.3389/fmolb.2021.690655",
"doi-asserted-by": "publisher",
"key": "ref34"
},
{
"DOI": "10.3389/fphar.2021.666600",
"doi-asserted-by": "publisher",
"key": "ref35"
},
{
"DOI": "10.1021/acs.jcim.9b00883",
"doi-asserted-by": "publisher",
"key": "ref36"
},
{
"DOI": "10.3389/fmolb.2021.619381",
"doi-asserted-by": "publisher",
"key": "ref37"
},
{
"DOI": "10.3389/fmedt.2020.614652",
"doi-asserted-by": "publisher",
"key": "ref38"
},
{
"DOI": "10.2174/138620709789104915",
"doi-asserted-by": "publisher",
"key": "ref39"
},
{
"DOI": "10.1016/j.ab.2017.08.002",
"doi-asserted-by": "publisher",
"key": "ref40"
},
{
"DOI": "10.3390/bios7040049",
"doi-asserted-by": "publisher",
"key": "ref41"
},
{
"DOI": "10.1038/s41598-020-78895-x",
"doi-asserted-by": "publisher",
"key": "ref42"
},
{
"DOI": "10.1016/j.bpc.2005.04.006",
"doi-asserted-by": "publisher",
"key": "ref43"
},
{
"DOI": "10.1007/978-1-4939-2425-7_10",
"doi-asserted-by": "publisher",
"key": "ref44"
},
{
"DOI": "10.3389/fmolb.2020.00098",
"doi-asserted-by": "publisher",
"key": "ref45"
},
{
"DOI": "10.1146/annurev.bb.06.060177.001055",
"doi-asserted-by": "publisher",
"key": "ref46"
}
],
"reference-count": 46,
"references-count": 46,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.mdpi.com/1422-0067/23/10/5436"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"Inorganic Chemistry",
"Organic Chemistry",
"Physical and Theoretical Chemistry",
"Computer Science Applications",
"Spectroscopy",
"Molecular Biology",
"General Medicine",
"Catalysis"
],
"subtitle": [],
"title": "Lactoferrin Inhibition of the Complex Formation between ACE2 Receptor and SARS CoV-2 Recognition Binding Domain",
"type": "journal-article",
"volume": "23"
}