Liposomal Lactoferrin Exerts Antiviral Activity against HCoV-229E and SARS-CoV-2 Pseudoviruses In Vitro
et al., Viruses, doi:10.3390/v15040972, Apr 2023
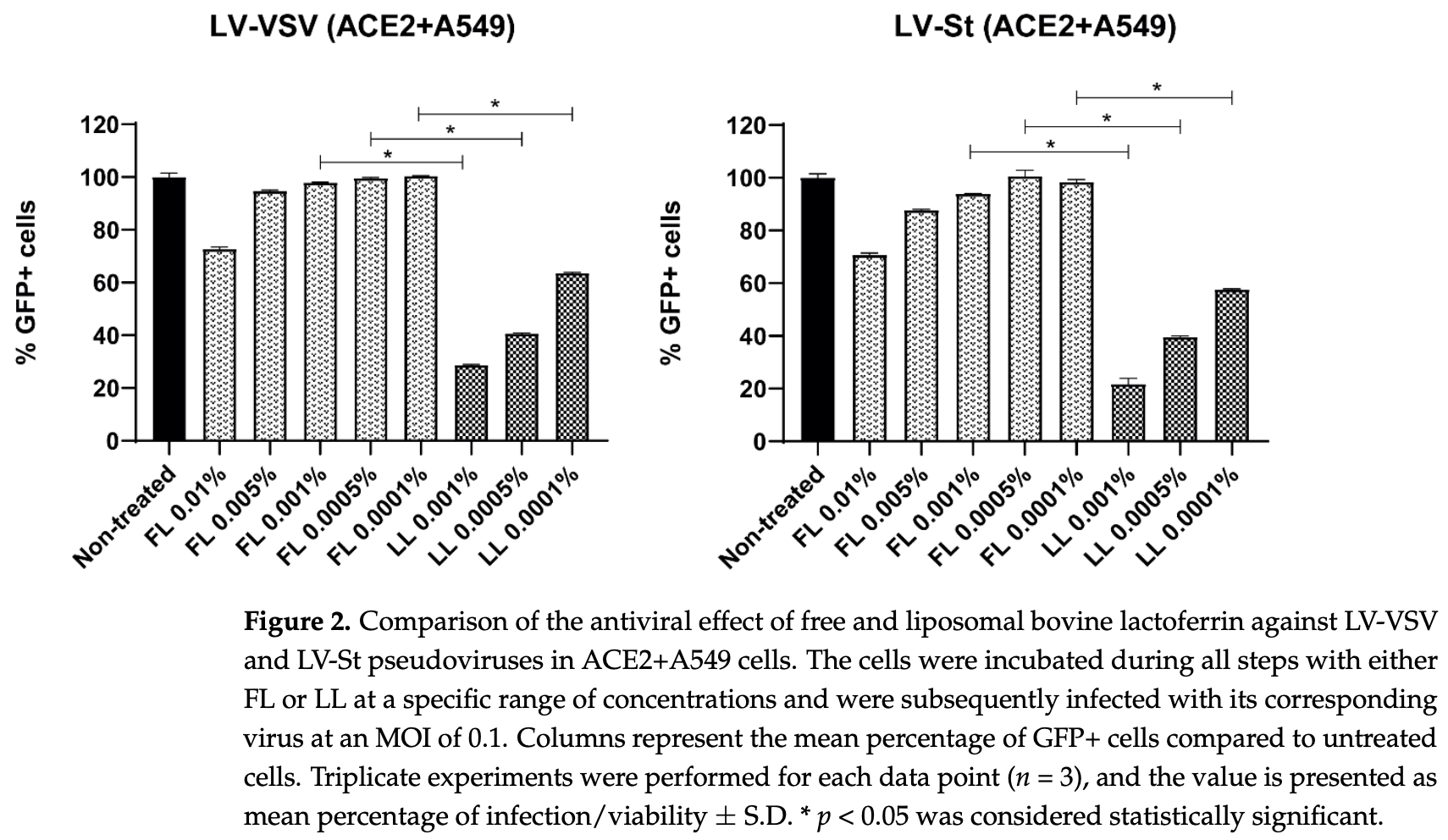
In vitro study showing more potent antiviral activity for SARS-CoV-2 and HCoV229E with liposomal lactoferrin compared with free lactoferrin. Authors note that liposomal encapsulation has been shown to increase permeability, bioavailability, and time release.
18 preclinical studies support the efficacy of lactoferrin for COVID-19:
1.
da Silva et al., Immunomodulatory effect of bovine lactoferrin during SARS-CoV-2 infection, Frontiers in Immunology, doi:10.3389/fimmu.2024.1456634.
2.
Cutone et al., Lactoferrin binding to Sars-CoV-2 Spike glycoprotein protects host from infection, inflammation and iron dysregulation., Research Square, doi:10.21203/rs.3.rs-1605740/v1.
3.
Miotto et al., Molecular Mechanisms Behind Anti SARS-CoV-2 Action of Lactoferrin, Frontiers in Molecular Biosciences, doi:10.3389/fmolb.2021.607443.
4.
Babulic et al., Lactoferrin Binds through Its N-Terminus to the Receptor-Binding Domain of the SARS-CoV-2 Spike Protein, Pharmaceuticals, doi:10.3390/ph17081021.
5.
Yathindranath et al., Lipid Nanoparticle-Based Inhibitors for SARS-CoV-2 Host Cell Infection, International Journal of Nanomedicine, doi:10.2147/IJN.S448005.
6.
Alves et al., Inhibition of SARS-CoV-2 Infection in Vero Cells by Bovine Lactoferrin under Different Iron-Saturation States, Pharmaceuticals, doi:10.3390/ph16101352.
7.
Kobayashi-Sakamoto et al., Bovine lactoferrin suppresses the cathepsin-dependent pathway of SARS-CoV-2 entry in vitro, International Dairy Journal, doi:10.1016/j.idairyj.2023.105805.
8.
Andreu et al., Liposomal Lactoferrin Exerts Antiviral Activity against HCoV-229E and SARS-CoV-2 Pseudoviruses In Vitro, Viruses, doi:10.3390/v15040972.
9.
Yazawa et al., Evaluation of SARS-CoV-2 isolation in cell culture from nasal/nasopharyngeal swabs or saliva specimens of patients with COVID-19, Research Square, doi:10.21203/rs.3.rs-2676422/v1.
10.
Piacentini et al., Lactoferrin Inhibition of the Complex Formation between ACE2 Receptor and SARS CoV-2 Recognition Binding Domain, International Journal of Molecular Sciences, doi:10.3390/ijms23105436.
11.
Ostrov et al., Highly Specific Sigma Receptor Ligands Exhibit Anti-Viral Properties in SARS-CoV-2 Infected Cells, Pathogens, doi:10.3390/pathogens10111514.
12.
Mirabelli et al., Morphological cell profiling of SARS-CoV-2 infection identifies drug repurposing candidates for COVID-19, Proceedings of the National Academy of Sciences, doi:10.1073/pnas.2105815118.
13.
Salaris et al., Protective Effects of Lactoferrin against SARS-CoV-2 Infection In Vitro, Nutrients, doi:10.3390/nu13020328.
Andreu et al., 15 Apr 2023, Spain, peer-reviewed, 4 authors.
Contact: sandreu@cbm.csic.es (corresponding author), ines.ripa@cbm.csic.es, raquel.bello-morales@uam.es, ja.lopez@uam.es.
In vitro studies are an important part of preclinical research, however results may be very different in vivo.
Liposomal Lactoferrin Exerts Antiviral Activity against HCoV-229E and SARS-CoV-2 Pseudoviruses In Vitro
Viruses, doi:10.3390/v15040972
A limited number of effective therapies are currently available to treat human coronavirus SARS-CoV-2 and other human coronaviruses, which are responsible for nearly a third of global cases of the common cold. The possibility of new emerging coronaviruses demands powerful new antiviral strategies. Lactoferrin is a well-known protein that possesses anti-inflammatory and immunomodulatory activities, and it has previously shown antiviral activity against several viruses, including SARS-CoV-2. To increase this antiviral activity, here we present bovine liposomal lactoferrin. Liposomal encapsulation of the compound was proven to increase permeability, bioavailability, and time release. In the present work, we compare the antiviral activity of free and liposomal bovine lactoferrin against HCoV229E and SARS-CoV-2 in vitro and in human primary bronchial epithelial cells, and we demonstrated that the liposomal form exerts a more potent antiviral activity than its free form at non-cytotoxic doses.
Informed Consent Statement: Written informed consent was obtained from the patient(s) to publish this paper.
Conflicts of Interest: The authors declare no conflict of interest.
References
Andreu, Ripa, Praena, López-Guerrero, Bello-Morales, The Valproic Acid Derivative Valpromide Inhibits Pseudorabies Virus Infection in Swine Epithelial and Mouse Neuroblastoma Cell Lines, Viruses, doi:10.3390/v13122522
Berlutti, Pantanella, Natalizi, Frioni, Paesano et al., Antiviral Properties of Lactoferrin-A Natural Immunity Molecule, Molecules, doi:10.3390/molecules16086992
Bolat, Eker, Kaplan, Duman, Arslan et al., Lactoferrin for COVID-19 Prevention, Treatment, and Recovery, Front. Nutr, doi:10.3389/fnut.2022.992733
Campione, Lanna, Cosio, Rosa, Conte et al., Lactoferrin as Antiviral Treatment in COVID-19 Management: Preliminary Evidence, Int J. Environ. Res. Public Health, doi:10.3390/ijerph182010985
Cutone, Rosa, Bonaccorsi Di Patti, Iacovelli, Conte et al., Lactoferrin Binding to SARS-CoV-2 Spike Glycoprotein Blocks Pseudoviral Entry and Relieves Iron Protein Dysregulation in Several In Vitro Models, Pharmaceutics, doi:10.3390/pharmaceutics14102111
Einerhand, Van Loo-Bouwman, Weiss, Wang, Ba et al., Can Lactoferrin, a Natural Mammalian Milk Protein, Assist in the Battle against COVID-19?, Nutrients, doi:10.3390/nu14245274
Enjuanes, Zuñiga, Castaño-Rodriguez, Gutierrez-Alvarez, Canton et al., Molecular Basis of Coronavirus Virulence and Vaccine Development, Adv. Virus Res, doi:10.1016/BS.AIVIR.2016.08.003
Farid, El Shemy, Nafie, Hegazy, Abdelhiee, Anti-Inflammatory, Anti-Oxidant and Hepatoprotective Effects of Lactoferrin in Rats, Drug Chem. Toxicol, doi:10.1080/01480545.2019.1585868
Gorbalenya, Baker, Baric, De Groot, Drosten et al., The Species Severe Acute Respiratory Syndrome-Related Coronavirus: Classifying 2019-NCoV and Naming It SARS-CoV-2, Nat. Microbiol, doi:10.1038/S41564-020-0695-Z
Grau-Expósito, Perea, Suppi, Massana, Vergara et al., Evaluation of SARS-CoV-2 Entry, Inflammation and New Therapeutics in Human Lung Tissue Cells, PLoS Pathog, doi:10.1371/journal.ppat.1010171
He, Qin, Guan, Liu, Hong et al., Bovine Lactoferrin Inhibits SARS-CoV-2 and SARS-CoV-1 by Targeting the RdRp Complex and Alleviates Viral Infection in the Hamster Model, J. Med. Virol, doi:10.1002/jmv.28281
Hoffmann, Hofmann-Winkler, Smith, Krüger, Arora et al., Camostat Mesylate Inhibits SARS-CoV-2 Activation by TMPRSS2-Related Proteases and Its Metabolite GBPA Exerts Antiviral Activi, ity. EBioMedicine, doi:10.1016/j.ebiom.2021.103255
Horndler, Delgado, Abia, Balabanov, Martínez-Fleta et al., Flow Cytometry Multiplexed Method for the Detection of Neutralizing Human Antibodies to the Native SARS-CoV-2.2 Spike Prot.tein, EMBO Mol. Med, doi:10.15252/emmm.202013549
Hu, Meng, Zhang, Xiang, Wang, The in Vitro Antiviral Activity of Lactoferrin against Common Human Coronaviruses and SARS-CoV-2 Is Mediated by Targeting the Heparan Sulfate Co-Receptor, Emerg Microbes Infect, doi:10.1080/22221751.2021.1888660
Indrayanto, Putra, Suhud, Validation of In-Vitro Bioassay Methods: Application in Herbal Drug Research, Profiles Drug Subst. Excip. Relat. Methodol, doi:10.1016/BS.PODRM.2020.07.005
Ishikado, Imanaka, Takeuchi, Harada, Makino, Liposomalization of Lactoferrin Enhanced It's Anti-Inflammatory Effects via Oral Administration, Biol. Pharm. Bull, doi:10.1248/bpb.28.1717
Kopaeva, Alchinova, Cherepov, Demorzhi, Nesterenko et al., New Properties of a Well-Known Antioxidant: Pleiotropic Effects of Human Lactoferrin in Mice Exposed to Gamma Irradiation in a Sublethal Dose, Antioxidants, doi:10.3390/antiox11091833
Lang, Yang, Deng, Liu, Yang et al., Inhibition of SARS Pseudovirus Cell Entry by Lactoferrin Binding to Heparan Sulfate Proteoglycans, PLoS ONE, doi:10.1371/journal.pone.0023710
Legrand, Elass, Carpentier, Mazurier, Interactions of Lactoferrin with Cells Involved in Immune Function, Biochem. Cell Biol, doi:10.1139/o06-045
Liu, Chen, Zhang, A Review of Liposomes as a Drug Delivery System: Current Status of Approved Products, Regulatory Environments, and Future Perspectives, Molecules, doi:10.3390/molecules27041372
Mann, Ndung'u, The Potential of Lactoferrin, Ovotransferrin and Lysozyme as Antiviral and Immune-Modulating Agents in COVID-19, Future Virol, doi:10.2217/fvl-2020-0170
Martorell, Llopis, Gonzalez, Ramón, Serrano et al., A Nutritional Supplement Containing Lactoferrin Stimulates the Immune System, Extends Lifespan, and Reduces Amyloid β Peptide Toxicity in Caenorhabditis Elegans, Food Sci. Nutr, doi:10.1002/fsn3.388
Menéndez, Approaches to the Potential Therapy of COVID-19: A General Overview from the Medicinal Chemistry Perspective, Molecules, doi:10.3390/molecules27030658
Mesel-Lemoine, Millet, Vidalain, Law, Vabret et al., A Human Coronavirus Responsible for the Common Cold Massively Kills Dendritic Cells but Not Monocytes, J. Virol, doi:10.1128/JVI.00269-12
Nakabayashi, Miyano, Sato, Yamane, Taketa, Growth of Human Hepatoma Cells Lines with Differentiated Functions in Chemically Defined Medium, Cancer Res
Reed, Muench, A Simple Method of Estimating Fifty per Cent Endpoints, Am. J. Epidemiol, doi:10.1093/oxfordjournals.aje.a118408
Salaris, Scarpa, Elli, Bertolini, Guglielmetti et al., Protective Effects of Lactoferrin against SARS-CoV-2 Infection In Vitro, Nutrients, doi:10.3390/nu13020328
Serrano, Almudéver, Serrano, Milara, Torrens et al., Phosphatidylcholine Liposomes as Carriers to Improve Topical Ascorbic Acid Treatment of Skin Disorders, Clin. Cosmet. Investig. Dermatol, doi:10.2147/CCID.S90781
Serrano, Kochergina, Albors, Diaz, Oroval et al., Liposomal Lactoferrin as Potential Preventative and Cure for COVID-19, Int. J. Res. Health Sci, doi:10.5530/ijrhs.8.1.3
Serrano, Mullor, Sanchez, Kochergina, Albors et al., Liposomal Lactoferrin Effect in Preventing SARS-CoV-2 Binding in HACAT Keratinocytes, Int. J. Res. Health Sci, doi:10.5530/ijrhs.8.2.1
Siqueiros-Cendón, Arévalo-Gallegos, Iglessias-Figueroa, García-Montoya, Salazar-Martínez et al., Immunomodulatory Effects of Lactoferrin, Acta Pharmacol. Sin, doi:10.1038/aps.2013.200
Sokolov, Isakova-Sivak, Grudinina, Mezhenskaya, Litasova et al., Ferristatin II Efficiently Inhibits SARS-CoV-2 Replication in Vero Cells, Viruses, doi:10.3390/v14020317
Sokolov, Isakova-Sivak, Mezhenskaya, Kostevich, Gorbunov et al., Molecular Mimicry of the Receptor-Binding Domain of the SARS-CoV-2
Trie, Guillen, Vaughan, Telfer, Brewer et al., Liposomes as Possible Carriers for Lactoferrin in the Local Treatment of Inflammatory Diseases, Exp. Biol. Med, doi:10.1177/153537020122600608
Trif, Roseanu, Brock, Brewer, Designing Lipid Nanostructures for Local Delivery of Biologically Active Macromolecules, J. Liposome Res, doi:10.1080/08982100701530027
Vert, The Non-Specific Antiviral Activity of Polysulfates to Fight SARS-CoV-2, Its Mutants and Viruses with Cationic Spikes, J. Biomater. Sci. Polym. Ed, doi:10.1080/09205063.2021.1925391
Wang, Timilsena, Blanch, Adhikari, Lactoferrin: Structure, Function, Denaturation and Digestion, Crit. Rev. Food Sci. Nutr, doi:10.1080/10408398.2017.1381583
Wang, Wang, Wang, Luo, Wan et al., Lactoferrin for the Treatment of COVID-19 (Review), Exp. Ther. Med, doi:10.3892/etm.2020.9402
DOI record:
{
"DOI": "10.3390/v15040972",
"ISSN": [
"1999-4915"
],
"URL": "http://dx.doi.org/10.3390/v15040972",
"abstract": "<jats:p>A limited number of effective therapies are currently available to treat human coronavirus SARS-CoV-2 and other human coronaviruses, which are responsible for nearly a third of global cases of the common cold. The possibility of new emerging coronaviruses demands powerful new antiviral strategies. Lactoferrin is a well-known protein that possesses anti-inflammatory and immunomodulatory activities, and it has previously shown antiviral activity against several viruses, including SARS-CoV-2. To increase this antiviral activity, here we present bovine liposomal lactoferrin. Liposomal encapsulation of the compound was proven to increase permeability, bioavailability, and time release. In the present work, we compare the antiviral activity of free and liposomal bovine lactoferrin against HCoV229E and SARS-CoV-2 in vitro and in human primary bronchial epithelial cells, and we demonstrated that the liposomal form exerts a more potent antiviral activity than its free form at non-cytotoxic doses.</jats:p>",
"alternative-id": [
"v15040972"
],
"author": [
{
"ORCID": "http://orcid.org/0000-0003-0600-4152",
"affiliation": [
{
"name": "Department of Molecular Biology, Universidad Autónoma de Madrid, C/Darwin, 2 Cantoblanco, 28049 Madrid, Spain"
},
{
"name": "Centro de Biología Molecular Severo Ochoa, Spanish National Research Council—Universidad Autónoma de Madrid (CSIC-UAM), C/Nicolás Cabrera, 1 Cantoblanco, 28049 Madrid, Spain"
}
],
"authenticated-orcid": false,
"family": "Andreu",
"given": "Sabina",
"sequence": "first"
},
{
"ORCID": "http://orcid.org/0000-0001-7007-5401",
"affiliation": [
{
"name": "Department of Molecular Biology, Universidad Autónoma de Madrid, C/Darwin, 2 Cantoblanco, 28049 Madrid, Spain"
},
{
"name": "Centro de Biología Molecular Severo Ochoa, Spanish National Research Council—Universidad Autónoma de Madrid (CSIC-UAM), C/Nicolás Cabrera, 1 Cantoblanco, 28049 Madrid, Spain"
}
],
"authenticated-orcid": false,
"family": "Ripa",
"given": "Inés",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-4415-6483",
"affiliation": [
{
"name": "Department of Molecular Biology, Universidad Autónoma de Madrid, C/Darwin, 2 Cantoblanco, 28049 Madrid, Spain"
},
{
"name": "Centro de Biología Molecular Severo Ochoa, Spanish National Research Council—Universidad Autónoma de Madrid (CSIC-UAM), C/Nicolás Cabrera, 1 Cantoblanco, 28049 Madrid, Spain"
}
],
"authenticated-orcid": false,
"family": "Bello-Morales",
"given": "Raquel",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0003-2817-5956",
"affiliation": [
{
"name": "Department of Molecular Biology, Universidad Autónoma de Madrid, C/Darwin, 2 Cantoblanco, 28049 Madrid, Spain"
},
{
"name": "Centro de Biología Molecular Severo Ochoa, Spanish National Research Council—Universidad Autónoma de Madrid (CSIC-UAM), C/Nicolás Cabrera, 1 Cantoblanco, 28049 Madrid, Spain"
}
],
"authenticated-orcid": false,
"family": "López-Guerrero",
"given": "José Antonio",
"sequence": "additional"
}
],
"container-title": "Viruses",
"container-title-short": "Viruses",
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2023,
4,
17
]
],
"date-time": "2023-04-17T08:45:32Z",
"timestamp": 1681721132000
},
"deposited": {
"date-parts": [
[
2023,
4,
17
]
],
"date-time": "2023-04-17T09:08:14Z",
"timestamp": 1681722494000
},
"indexed": {
"date-parts": [
[
2023,
4,
18
]
],
"date-time": "2023-04-18T05:11:10Z",
"timestamp": 1681794670902
},
"is-referenced-by-count": 0,
"issue": "4",
"issued": {
"date-parts": [
[
2023,
4,
15
]
]
},
"journal-issue": {
"issue": "4",
"published-online": {
"date-parts": [
[
2023,
4
]
]
}
},
"language": "en",
"license": [
{
"URL": "https://creativecommons.org/licenses/by/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2023,
4,
15
]
],
"date-time": "2023-04-15T00:00:00Z",
"timestamp": 1681516800000
}
}
],
"link": [
{
"URL": "https://www.mdpi.com/1999-4915/15/4/972/pdf",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "1968",
"original-title": [],
"page": "972",
"prefix": "10.3390",
"published": {
"date-parts": [
[
2023,
4,
15
]
]
},
"published-online": {
"date-parts": [
[
2023,
4,
15
]
]
},
"publisher": "MDPI AG",
"reference": [
{
"DOI": "10.1038/s41564-020-0695-z",
"article-title": "The Species Severe Acute Respiratory Syndrome-Related Coronavirus: Classifying 2019-NCoV and Naming It SARS-CoV-2",
"author": "Gorbalenya",
"doi-asserted-by": "crossref",
"first-page": "536",
"journal-title": "Nat. Microbiol.",
"key": "ref_1",
"volume": "5",
"year": "2020"
},
{
"DOI": "10.1016/bs.aivir.2016.08.003",
"article-title": "Molecular Basis of Coronavirus Virulence and Vaccine Development",
"author": "Enjuanes",
"doi-asserted-by": "crossref",
"first-page": "245",
"journal-title": "Adv. Virus Res.",
"key": "ref_2",
"volume": "96",
"year": "2016"
},
{
"DOI": "10.1128/JVI.00269-12",
"article-title": "A Human Coronavirus Responsible for the Common Cold Massively Kills Dendritic Cells but Not Monocytes",
"author": "Millet",
"doi-asserted-by": "crossref",
"first-page": "7577",
"journal-title": "J. Virol.",
"key": "ref_3",
"volume": "86",
"year": "2012"
},
{
"DOI": "10.3390/molecules27030658",
"doi-asserted-by": "crossref",
"key": "ref_4",
"unstructured": "Menéndez, J.C. (2022). Approaches to the Potential Therapy of COVID-19: A General Overview from the Medicinal Chemistry Perspective. Molecules, 27."
},
{
"DOI": "10.1080/01480545.2019.1585868",
"article-title": "Anti-Inflammatory, Anti-Oxidant and Hepatoprotective Effects of Lactoferrin in Rats",
"author": "Farid",
"doi-asserted-by": "crossref",
"first-page": "286",
"journal-title": "Drug Chem. Toxicol.",
"key": "ref_5",
"volume": "44",
"year": "2019"
},
{
"DOI": "10.3390/antiox11091833",
"doi-asserted-by": "crossref",
"key": "ref_6",
"unstructured": "Kopaeva, M.Y., Alchinova, I.B., Cherepov, A.B., Demorzhi, M.S., Nesterenko, M.V., Zarayskaya, I.Y., and Karganov, M.Y. (2022). New Properties of a Well-Known Antioxidant: Pleiotropic Effects of Human Lactoferrin in Mice Exposed to Gamma Irradiation in a Sublethal Dose. Antioxidants, 11."
},
{
"DOI": "10.1139/o06-045",
"article-title": "Interactions of Lactoferrin with Cells Involved in Immune Function",
"author": "Legrand",
"doi-asserted-by": "crossref",
"first-page": "282",
"journal-title": "Biochem. Cell Biol.",
"key": "ref_7",
"volume": "84",
"year": "2006"
},
{
"DOI": "10.3390/molecules16086992",
"doi-asserted-by": "crossref",
"key": "ref_8",
"unstructured": "Berlutti, F., Pantanella, F., Natalizi, T., Frioni, A., Paesano, R., Polimeni, A., and Valenti, P. (2011). Antiviral Properties of Lactoferrin—A Natural Immunity Molecule. Molecules, 16."
},
{
"article-title": "Liposomal Lactoferrin as Potential Preventative and Cure for COVID-19",
"author": "Serrano",
"first-page": "08",
"journal-title": "Int. J. Res. Health Sci.",
"key": "ref_9",
"volume": "8",
"year": "2020"
},
{
"DOI": "10.3390/nu14245274",
"doi-asserted-by": "crossref",
"key": "ref_10",
"unstructured": "Einerhand, A.W.C., van Loo-Bouwman, C.A., Weiss, G.A., Wang, C., Ba, G., Fan, Q., He, B., and Smit, G. (2022). Can Lactoferrin, a Natural Mammalian Milk Protein, Assist in the Battle against COVID-19?. Nutrients, 14."
},
{
"DOI": "10.1080/10408398.2017.1381583",
"article-title": "Lactoferrin: Structure, Function, Denaturation and Digestion",
"author": "Wang",
"doi-asserted-by": "crossref",
"first-page": "580",
"journal-title": "Crit. Rev. Food Sci. Nutr.",
"key": "ref_11",
"volume": "59",
"year": "2019"
},
{
"DOI": "10.3389/fnut.2022.992733",
"doi-asserted-by": "crossref",
"key": "ref_12",
"unstructured": "Bolat, E., Eker, F., Kaplan, M., Duman, H., Arslan, A., Saritaş, S., Şahutoğlu, A.S., and Karav, S. (2022). Lactoferrin for COVID-19 Prevention, Treatment, and Recovery. Front. Nutr., 9."
},
{
"DOI": "10.1371/journal.pone.0023710",
"doi-asserted-by": "crossref",
"key": "ref_13",
"unstructured": "Lang, J., Yang, N., Deng, J., Liu, K., Yang, P., Zhang, G., and Jiang, C. (2011). Inhibition of SARS Pseudovirus Cell Entry by Lactoferrin Binding to Heparan Sulfate Proteoglycans. PLoS ONE, 6."
},
{
"DOI": "10.1002/jmv.28281",
"doi-asserted-by": "crossref",
"key": "ref_14",
"unstructured": "He, S.T., Qin, H., Guan, L., Liu, K., Hong, B., Zhang, X., Lou, F., Li, M., Lin, W., and Chen, Y. (2023). Bovine Lactoferrin Inhibits SARS-CoV-2 and SARS-CoV-1 by Targeting the RdRp Complex and Alleviates Viral Infection in the Hamster Model. J. Med. Virol., 95."
},
{
"article-title": "Liposomal Lactoferrin Effect in Preventing SARS-CoV-2 Binding in HACAT Keratinocytes",
"author": "Serrano",
"first-page": "16",
"journal-title": "Int. J. Res. Health Sci.",
"key": "ref_15",
"volume": "8",
"year": "2020"
},
{
"DOI": "10.3390/ijerph182010985",
"doi-asserted-by": "crossref",
"key": "ref_16",
"unstructured": "Campione, E., Lanna, C., Cosio, T., Rosa, L., Conte, M.P., Iacovelli, F., Romeo, A., Falconi, M., Del Vecchio, C., and Franchin, E. (2021). Lactoferrin as Antiviral Treatment in COVID-19 Management: Preliminary Evidence. Int J. Environ. Res. Public Health, 18."
},
{
"DOI": "10.1248/bpb.28.1717",
"article-title": "Liposomalization of Lactoferrin Enhanced It’s Anti-Inflammatory Effects via Oral Administration",
"author": "Ishikado",
"doi-asserted-by": "crossref",
"first-page": "1717",
"journal-title": "Biol. Pharm. Bull.",
"key": "ref_17",
"volume": "28",
"year": "2005"
},
{
"article-title": "Liposomes as Possible Carriers for Lactoferrin in the Local Treatment of Inflammatory Diseases",
"author": "Trie",
"first-page": "559",
"journal-title": "Exp. Biol. Med.",
"key": "ref_18",
"volume": "226",
"year": "2016"
},
{
"DOI": "10.1080/08982100701530027",
"article-title": "Designing Lipid Nanostructures for Local Delivery of Biologically Active Macromolecules",
"author": "Trif",
"doi-asserted-by": "crossref",
"first-page": "237",
"journal-title": "J. Liposome Res.",
"key": "ref_19",
"volume": "17",
"year": "2008"
},
{
"DOI": "10.3390/molecules27041372",
"doi-asserted-by": "crossref",
"key": "ref_20",
"unstructured": "Liu, P., Chen, G., and Zhang, J. (2022). A Review of Liposomes as a Drug Delivery System: Current Status of Approved Products, Regulatory Environments, and Future Perspectives. Molecules, 27."
},
{
"article-title": "Growth of Human Hepatoma Cells Lines with Differentiated Functions in Chemically Defined Medium",
"author": "Nakabayashi",
"first-page": "3858",
"journal-title": "Cancer Res.",
"key": "ref_21",
"volume": "42",
"year": "1982"
},
{
"DOI": "10.15252/emmm.202013549",
"article-title": "Flow Cytometry Multiplexed Method for the Detection of Neutralizing Human Antibodies to the Native SARS-CoV-2.2 Spike Prot.tein",
"author": "Horndler",
"doi-asserted-by": "crossref",
"first-page": "e13549",
"journal-title": "EMBO Mol. Med.",
"key": "ref_22",
"volume": "13",
"year": "2021"
},
{
"DOI": "10.1371/journal.ppat.1010171",
"doi-asserted-by": "crossref",
"key": "ref_23",
"unstructured": "Grau-Expósito, J., Perea, D., Suppi, M., Massana, N., Vergara, A., Soler, M.J., Trinite, B., Blanco, J., García-Pérez, J., and Alcamí, J. (2022). Evaluation of SARS-CoV-2 Entry, Inflammation and New Therapeutics in Human Lung Tissue Cells. PLoS Pathog., 18."
},
{
"DOI": "10.1093/oxfordjournals.aje.a118408",
"article-title": "A Simple Method of Estimating Fifty per Cent Endpoints",
"author": "Reed",
"doi-asserted-by": "crossref",
"first-page": "493",
"journal-title": "Am. J. Epidemiol.",
"key": "ref_24",
"volume": "27",
"year": "1938"
},
{
"DOI": "10.3390/v13122522",
"doi-asserted-by": "crossref",
"key": "ref_25",
"unstructured": "Andreu, S., Ripa, I., Praena, B., López-guerrero, J.A., and Bello-morales, R. (2021). The Valproic Acid Derivative Valpromide Inhibits Pseudorabies Virus Infection in Swine Epithelial and Mouse Neuroblastoma Cell Lines. Viruses, 13."
},
{
"article-title": "Phosphatidylcholine Liposomes as Carriers to Improve Topical Ascorbic Acid Treatment of Skin Disorders",
"author": "Serrano",
"first-page": "591",
"journal-title": "Clin. Cosmet. Investig. Dermatol.",
"key": "ref_26",
"volume": "8",
"year": "2015"
},
{
"DOI": "10.1002/fsn3.388",
"article-title": "A Nutritional Supplement Containing Lactoferrin Stimulates the Immune System, Extends Lifespan, and Reduces Amyloid β Peptide Toxicity in Caenorhabditis Elegans",
"author": "Martorell",
"doi-asserted-by": "crossref",
"first-page": "255",
"journal-title": "Food Sci. Nutr.",
"key": "ref_27",
"volume": "5",
"year": "2017"
},
{
"DOI": "10.1016/j.ebiom.2021.103255",
"article-title": "Camostat Mesylate Inhibits SARS-CoV-2 Activation by TMPRSS2-Related Proteases and Its Metabolite GBPA Exerts Antiviral Activi.ity",
"author": "Hoffmann",
"doi-asserted-by": "crossref",
"first-page": "103255",
"journal-title": "EBioMedicine",
"key": "ref_28",
"volume": "65",
"year": "2021"
},
{
"DOI": "10.1080/22221751.2021.1888660",
"article-title": "The in Vitro Antiviral Activity of Lactoferrin against Common Human Coronaviruses and SARS-CoV-2 Is Mediated by Targeting the Heparan Sulfate Co-Receptor",
"author": "Hu",
"doi-asserted-by": "crossref",
"first-page": "317",
"journal-title": "Emerg Microbes Infect.",
"key": "ref_29",
"volume": "10",
"year": "2021"
},
{
"DOI": "10.1007/s10534-022-00458-6",
"doi-asserted-by": "crossref",
"key": "ref_30",
"unstructured": "Sokolov, A.V., Isakova-Sivak, I.N., Mezhenskaya, D.A., Kostevich, V.A., Gorbunov, N.P., Elizarova, A.Y., Matyushenko, V.A., Berson, Y.M., Grudinina, N.A., and Kolmakov, N.N. (2022). Molecular Mimicry of the Receptor-Binding Domain of the SARS-CoV-2 Spike Protein: From the Interaction of Spike-Specific Antibodies with Transferrin and Lactoferrin to the Antiviral Effects of Human Recombinant Lactoferrin. Biometals, 1–26."
},
{
"DOI": "10.3390/v14020317",
"doi-asserted-by": "crossref",
"key": "ref_31",
"unstructured": "Sokolov, A., Isakova-Sivak, I., Grudinina, N., Mezhenskaya, D., Litasova, E., Kostevich, V., Stepanova, E., Rak, A., Sychev, I., and Kirik, O. (2022). Ferristatin II Efficiently Inhibits SARS-CoV-2 Replication in Vero Cells. Viruses, 14."
},
{
"DOI": "10.21203/rs.3.rs-1605740/v1",
"doi-asserted-by": "crossref",
"key": "ref_32",
"unstructured": "Cutone, A., Rosa, L., Bonaccorsi di Patti, M.C., Iacovelli, F., Conte, M.P., Ianiro, G., Romeo, A., Campione, E., Bianchi, L., and Valenti, P. (2022). Lactoferrin Binding to SARS-CoV-2 Spike Glycoprotein Blocks Pseudoviral Entry and Relieves Iron Protein Dysregulation in Several In Vitro Models. Pharmaceutics, 14."
},
{
"DOI": "10.2217/fvl-2020-0170",
"article-title": "The Potential of Lactoferrin, Ovotransferrin and Lysozyme as Antiviral and Immune-Modulating Agents in COVID-19",
"author": "Mann",
"doi-asserted-by": "crossref",
"first-page": "609",
"journal-title": "Future Virol.",
"key": "ref_33",
"volume": "15",
"year": "2020"
},
{
"DOI": "10.1080/09205063.2021.1925391",
"article-title": "The Non-Specific Antiviral Activity of Polysulfates to Fight SARS-CoV-2, Its Mutants and Viruses with Cationic Spikes",
"author": "Vert",
"doi-asserted-by": "crossref",
"first-page": "1466",
"journal-title": "J. Biomater. Sci. Polym. Ed.",
"key": "ref_34",
"volume": "32",
"year": "2021"
},
{
"key": "ref_35",
"unstructured": "US Food and Drug Administration (2023, March 05). GRN 000456 Cow’s Milk-Derived Lactoferrin, Available online: https://www.fda.gov/media/153787/download."
},
{
"DOI": "10.1038/aps.2013.200",
"article-title": "Immunomodulatory Effects of Lactoferrin",
"doi-asserted-by": "crossref",
"first-page": "557",
"journal-title": "Acta Pharmacol. Sin.",
"key": "ref_36",
"volume": "35",
"year": "2014"
},
{
"DOI": "10.3390/nu13020328",
"doi-asserted-by": "crossref",
"key": "ref_37",
"unstructured": "Salaris, C., Scarpa, M., Elli, M., Bertolini, A., Guglielmetti, S., Pregliasco, F., Blandizzi, C., Brun, P., and Castagliuolo, I. (2021). Protective Effects of Lactoferrin against SARS-CoV-2 Infection In Vitro. Nutrients, 13."
},
{
"DOI": "10.3892/etm.2020.9402",
"article-title": "Lactoferrin for the Treatment of COVID-19 (Review)",
"author": "Wang",
"doi-asserted-by": "crossref",
"first-page": "272",
"journal-title": "Exp. Ther. Med.",
"key": "ref_38",
"volume": "20",
"year": "2020"
},
{
"DOI": "10.1016/bs.podrm.2020.07.005",
"article-title": "Validation of In-Vitro Bioassay Methods: Application in Herbal Drug Research",
"author": "Indrayanto",
"doi-asserted-by": "crossref",
"first-page": "273",
"journal-title": "Profiles Drug Subst. Excip. Relat. Methodol.",
"key": "ref_39",
"volume": "46",
"year": "2021"
}
],
"reference-count": 39,
"references-count": 39,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.mdpi.com/1999-4915/15/4/972"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"Virology",
"Infectious Diseases"
],
"subtitle": [],
"title": "Liposomal Lactoferrin Exerts Antiviral Activity against HCoV-229E and SARS-CoV-2 Pseudoviruses In Vitro",
"type": "journal-article",
"volume": "15"
}