Lactoferrin as Antiviral Treatment in COVID-19 Management: Preliminary Evidence
et al., International Journal of Environmental Research and Public Health, doi:10.3390/ijerph182010985, Oct 2021
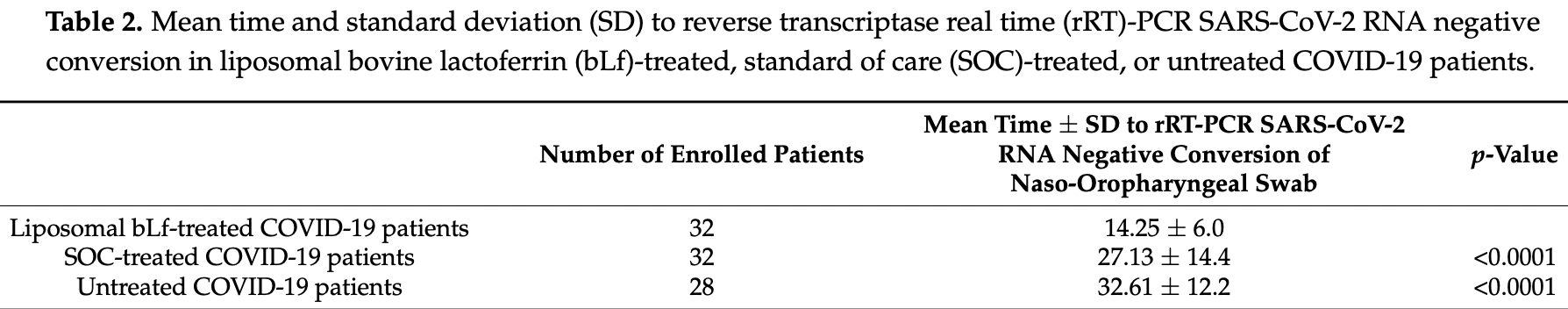
Small prospective study in Italy with 32 lactoferrin patients, 32 SOC, and 28 patients with no treatment, showing significantly faster viral clearance and improved recovery with treatment in unadjusted results. Oral and intranasal lactoferrin.
Targeted administration to the respiratory tract provides treatment directly
to the typical source of initial SARS-CoV-2 infection and replication, and
allows for rapid onset of action, higher local drug concentration, and reduced systemic side effects.
|
time to viral-, 47.5% lower, relative time 0.53, p < 0.001, treatment 32, control 32, vs. SOC.
|
|
time to viral-, 56.3% lower, relative time 0.44, p < 0.001, treatment 32, control 28, vs. untreated.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Campione et al., 19 Oct 2021, prospective, Italy, peer-reviewed, 32 authors.
Lactoferrin as Antiviral Treatment in COVID-19 Management: Preliminary Evidence
International Journal of Environmental Research and Public Health, doi:10.3390/ijerph182010985
Lactoferrin (Lf), a multifunctional cationic glycoprotein synthesized by exocrine glands and neutrophils, possesses an in vitro antiviral activity against SARS-CoV-2. Thus, we conducted an in vivo preliminary study to investigate the antiviral effect of oral and intranasal liposomal bovine Lf (bLf) in asymptomatic and mild-to-moderate COVID-19 patients. From April 2020 to June 2020, a total of 92 mild-to-moderate (67/92) and asymptomatic (25/92) COVID-19 patients were recruited and divided into three groups. Thirty-two patients (14 hospitalized and 18 in home-based isolation) received only oral and intranasal liposomal bLf; 32 hospitalized patients were treated only with standard of care (SOC) treatment; and 28, in home-based isolation, did not take any medication. Furthermore, 32 COVID-19 negative, untreated, healthy subjects were added for ancillary analysis. Liposomal bLf-treated COVID-19 patients obtained an earlier and significant (p < 0.0001) SARS-CoV-2 RNA negative conversion compared to the SOC-treated and untreated respectively). Liposomal bLf-treated COVID-19 patients showed fast clinical symptoms recovery compared to the SOC-treated COVID-19 patients. In bLf-treated patients, a
Funding: This study was supported by the contribution of "Fondazione Terzo Pilastro Internazionale", president Prof. Emmanuele Francesco Maria Emanuele.
Institutional Review Board Statement: The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the Tor Vergata University Hospital Ethics Committee (Code 42/20). It was registered at www.clinicaltrials.gov (Identifier: NCT04475120) (accessed on 2 August 2021). Informed Consent Statement: Informed consent was obtained from all subjects involved in the study.
Conflicts of Interest: The authors declare no conflict of interest.
References
Amani, Khanijahani, Amani, Hydroxychloroquine plus standard of care compared with standard of care alone in COVID-19: A meta-analysis of randomized controlled trials, Sci. Rep, doi:10.1038/s41598-021-91089-3
Ashida, Sasaki, Suzuki, Lönnerdal, Cellular internalization of lactoferrin in intestinal epithelial cells, Biometals, doi:10.1023/B:BIOM.0000027710.13543.3f
Aziz, Fatima, Assaly, Elevated interleukin-6 and severe COVID-19: A meta-analysis, J. Med. Virol, doi:10.1002/jmv.25948
Berlutti, Pantanella, Natalizi, Frioni, Paesano et al., Antiviral properties of lactoferrin-A natural immunity molecule, Molecules, doi:10.3390/molecules16086992
Bolondi, Russo, Gamberini, Circelli, Meca et al., Iron metabolism and lymphocyte characterisation during Covid-19 infection in ICU patients: An observational cohort study, World J. Emerg. Surg, doi:10.1186/s13017-020-00323-2
Butowt, Bilinska, SARS-CoV-2: Olfaction, Brain Infection, and the Urgent Need for Clinical Samples Allowing Earlier Virus Detection, ACS Chem. Neurosci, doi:10.1021/acschemneuro.0c00172
Campione, Cosio, Rosa, Lanna, Di Girolamo et al., Lactoferrin as Protective Natural Barrier of Respiratory and Intestinal Mucosa against Coronavirus Infection and Inflammation, Int. J. Mol. Sci, doi:10.3390/ijms21144903
Campione, Lanna, Cosio, Rosa, Conte et al., Lactoferrin Against SARS-CoV-2: In vitro and in silico Evidences, Front. Pharmacol, doi:10.3389/fphar.2021.666600
Chan, Yuan, Kok, To, Chu et al., A familial cluster of pneumonia associated with the 2019 novel coronavirus indicating person-to-person transmission: A study of a family cluster, Lancet, doi:10.1016/S0140-6736(20)30154-9
Chen, Zhang, Ju, He, Advances in the research of mechanism and related immunotherapy on the cytokine storm induced by coronavirus disease
Chen, Zhou, Dong, Qu, Gong et al., Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: A descriptive study, Lancet, doi:10.1016/S0140-6736(20)30211-7
Chorin, Wadhwani, Magnani, Dai, Shulman et al., QT interval prolongation and torsade de pointes in patients with COVID-19 treated with hydroxychloroquine/azithromycin, Heart Rhythm, doi:10.1016/j.hrthm.2020.05.014
Cortegiani, Ippolito, Greco, Granone, Protti et al., Rationale and evidence on the use of tocilizumab in COVID-19: A systematic review, Pulmonology, doi:10.1016/j.pulmoe.2020.07.003
Cui, Li, Shi, Origin and evolution of pathogenic coronaviruses, Nat. Rev. Microbiol, doi:10.1038/s41579-018-0118-9
Cutone, Rosa, Lepanto, Scotti, Berlutti et al., Lactoferrin Efficiently Counteracts the Inflammation-Induced Changes of the Iron Homeostasis System in Macrophages, Front. Immunol, doi:10.3389/fimmu.2017.00705
Dalamaga, Karampela, Mantzoros, Commentary: Could iron chelators prove to be useful as an adjunct to COVID-19 Treatment Regimens?, Metab. Clin. Exp, doi:10.1016/j.metabol.2020.154260
Deng, Liu, Li, Zhang, Zhao et al., Blood biochemical characteristics of patients with coronavirus disease 2019 (COVID-19): A systemic review and meta-analysis, Clin. Chem. Lab. Med, doi:10.1515/cclm-2020-0338
Efsa Panel On, Products, Nutrition and Allergies (NDA). Scientific Opinion on bovine lactoferrin, EFSA J, doi:10.2903/j.efsa.2012.2701
Fda, Cow's Milk-Derived Lactoferrin
Goldberg, Goldberg, Cerejo, Tayal, Cerebrovascular Disease in COVID-19, AJNR Am. J. Neuroradiol, doi:10.3174/ajnr.A6588
Guan, Ni, Hu, Liang, Ou et al., Clinical characteristics of coronavirus disease 2019 in China, N. Engl. J. Med, doi:10.1056/NEJMoa2002032
Hu, Meng, Zhang, Xiang, Wang, The In Vitro antiviral activity of lactoferrin against common human coronaviruses and SARS-CoV-2 is mediated by targeting the heparan sulfate co-receptor, Emerg. Microbes Infect, doi:10.1080/22221751.2021.1888660
Huang, Ling, Cheng, Wen, Dai et al., Characteristics of the coronavirus disease 2019 and related therapeutic options, Mol. Ther. Methods Clin. Dev, doi:10.1016/j.omtm.2020.06.013
Jiang, Zhang, Zhao, Li, Lei et al., Transmission Potential of Asymptomatic and Paucisymptomatic Severe Acute Respiratory Syndrome Coronavirus 2 Infections: A 3-Family Cluster Study in China, J. Infect. Dis, doi:10.1093/infdis/jiaa206
Kappert, Jahić, Tauber, Assessment of serum ferritin as a biomarker in COVID-19: Bystander or participant? Insights by comparison with other infectious and non-infectious diseases, Biomarkers, doi:10.1080/1354750X.2020.1797880
Kim, An, Kim, Hwang, Comparative efficacy and safety of pharmacological interventions for the treatment of COVID-19: A systematic review and network meta-analysis, PLoS Med, doi:10.1371/journal.pmed.1003501
Kim, Read, Fauci, Therapy for Early COVID-19: A Critical Need, JAMA, doi:10.1001/jama.2020.22813
Kruzel, Zimecki, Actor, Lactoferrin in a Context of Inflammation-Induced Pathology, Front. Immunol, doi:10.3389/fimmu.2017.01438
Lan, Ge, Yu, Shan, Zhou et al., Structure of the SARS-CoV-2 spike receptor-binding domain bound to the ACE2 receptor, Nature, doi:10.1038/s41586-020-2180-5
Lang, Yang, Deng, Liu, Yang et al., Inhibition of SARS pseudovirus cell entry by lactoferrin binding to heparan sulfate proteoglycans, PLoS ONE, doi:10.1371/journal.pone.0023710
Lepanto, Rosa, Cutone, Conte, Paesano et al., Efficacy of Lactoferrin Oral Administration in the Treatment of Anemia and Anemia of Inflammation in Pregnant and Non-pregnant Women: An Interventional Study, Front. Immunol, doi:10.3389/fimmu.2018.02123
Lepanto, Rosa, Paesano, Valenti, Cutone, Lactoferrin in Aseptic and Septic Inflammation, Molecules, doi:10.3390/molecules24071323
Li, Geng, Peng, Meng, Lu, Molecular immune pathogenesis and diagnosis of COVID-19, J. Pharm. Anal, doi:10.1016/j.jpha.2020.03.001
Li, Huang, Wang, Wang, Liang et al., COVID-19 patients' clinical characteristics, discharge rate, and fatality rate of meta-analysis, J. Med. Virol, doi:10.1002/jmv.25757
Li, Structure, Function, and Evolution of Coronavirus Spike Proteins, Annu. Rev. Virol, doi:10.1146/annurev-virology-110615-042301
Li, Su, Zhi, Huang, Zhuang et al., Viral shedding dynamics in asymptomatic and mildly symptomatic patients infected with SARS-CoV-2, Clin. Microbiol. Infect, doi:10.1016/j.cmi.2020.07.008
Liao, Jiang, Lönnerdal, Biochemical and molecular impacts of lactoferrin on small intestinal growth and development during early life, Biochem. Cell Biol, doi:10.1139/o11-075
Liu, Chopra, Li, Wolfert, Tompkins et al., SARS-CoV-2 spike protein binds heparan sulfate in a length and sequence dependent manner, BiorXiv, doi:10.1101/2020.05.10.087288
Liu, Li, COVID-19: Attacks the 1-beta chain of hemoglobin and captures the porphyrin to inhibit human heme metabolism, ChemRxiv, doi:10.26434/chemrxiv.11938173.v8
Liu, Wang, Potential inhibitors against 2019-nCoV coronavirus M protease from clinically approved medicines, J. Genet. Genom, doi:10.1016/j.jgg.2020.02.001
Liu, Ye, Liu, Liu, Singh, Stability during In Vitro digestion of lactoferrin-loaded liposomes prepared from milk fat globule membrane-derived phospholipids, J. Dairy Sci, doi:10.3168/jds.2012-6072
Loganathan, Kuppusamy, Wankhar, Gurugubelli, Mahadevappa et al., Angiotensinconverting enzyme 2 (ACE2): COVID 19 gate way to multiple organ failure syndromes, Respir. Physiol. Neurobiol, doi:10.1016/j.resp.2020.103548
Lu, Zhao, Li, Niu, Yang et al., Genomic characterisation and epidemiology of 2019 novel coronavirus: Implications for virus origins and receptor binding, Lancet, doi:10.1016/S0140-6736(20)30251-8
Maeda, Obata, Do, Kuno, The Association of Interleukin-6 value, Interleukin inhibitors and Outcomes of Patients with COVID-19 in New York City, J. Med. Virol, doi:10.1002/jmv.26365
Mancinelli, Rosa, Cutone, Lepanto, Franchitto et al., Viral Hepatitis and Iron Dysregulation: Molecular Pathways and the Role of Lactoferrin, Molecules, doi:10.3390/molecules25081997
Marietta, Coluccio, Luppi, COVID-19, coagulopathy and venous thromboembolism: More questions than answers, Intern. Emerg. Med, doi:10.1007/s11739-020-02432-x
Merad, Martin, Pathological inflammation in patients with COVID-19: A key role for monocytes and macrophages, Nat. Rev. Immunol, doi:10.1038/s41577-020-0331-4
Mercuro, Yen, Shim, Maher, Mccoy et al., Risk of QT interval prolongation associated with use of hydroxychloroquine with or without concomitant azithromycin among hospitalized patients testing positive for coronavirus disease 2019 (COVID-19), JAMA Cardiol, doi:10.1001/jamacardio.2020.1834
Ng, Cheung, Wong, Wang, Ip et al., Antiviral activities of whey proteins, Appl. Microbiol. Biotechnol, doi:10.1007/s00253-015-6818-4
Nguyen, Dolladille, Drici, Fenioux, Alexandre et al., Cardiovascular toxicities associated with hydroxychloroquine and azithromycin: An analysis of the World Health Organization pharmacovigilance database, Circulation, doi:10.1161/CIRCULATIONAHA.120.048238
Prévost, Gasser, Beaudoin-Bussières, Richard, Duerr et al., Cross-sectional evaluation of humoral responses against SARS-CoV-2 Spike, Cell. Rep. Med, doi:10.1016/j.xcrm.2020.100126
Rosa, Cutone, Lepanto, Paesano, Valenti, Lactoferrin: A Natural Glycoprotein Involved in Iron and Inflammatory Homeostasis, Int. J. Mol. Sci, doi:10.3390/ijms18091985
Rosa, Lepanto, Cutone, Siciliano, Paesano et al., Influence of oral administration mode on the efficacy of commercial bovine Lactoferrin against iron and inflammatory homeostasis disorders, Biometals, doi:10.1007/s10534-020-00236-2
Rosa, Tripepi, Naldi, Aimati, Santangeli et al., Ambulatory COVID-19 Patients Treated with Lactoferrin as a Supplementary Antiviral Agent: A Preliminary Study, J. Clin. Med, doi:10.3390/jcm10184276
Salaris, Scarpa, Elli, Bertolini, Guglielmetti et al., Protective Effects of Lactoferrin against SARS-CoV-2 Infection In Vitro, Nutrients, doi:10.3390/nu13020328
Satış, Özger, Aysert Yıldız, Hızel, Gulbahar et al., Prognostic value of interleukin-18 and its association with other inflammatory markers and disease severity in COVID-19, Cytokine, doi:10.1016/j.cyto.2020.155302
Shao, Za, None, doi:10.3760/cma.j.cn501120-20200224-00088
Spring, Md, Usa, Food and Drug Administration
Suzuki, Wong, Ashida, Schryvers, Lönnerdal, The N1 domain of human lactoferrin is required for internalization by caco-2 cells and targeting to the nucleus, Biochemistry, doi:10.1021/bi8012164
Tahir Ul Qamar, Alqahtani, Alamri, Chen, Structural basis of SARS-CoV-2 3CLpro and antiCOVID-19 drug discovery from medicinal plants, J. Pharm. Anal, doi:10.1016/j.jpha.2020.03.009
Tang, Li, Wang, Sun, Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia, J. Thromb. Haemost, doi:10.1111/jth.14768
Thachil, What do monitoring platelet counts in COVID-19 teach us?, J. Thromb. Haemost, doi:10.1111/jth.14879
Tian, Li, Huang, Xia, Lu et al., Potent binding of 2019 novel coronavirus spike protein by a SARS coronavirus-specific human monoclonal antibody, Emerg. Microbes Infect, doi:10.1080/22221751.2020.1729069
Tsai, Lai, Lin, Luo, Lin et al., Clinical manifestation and disease progression in COVID-19 infection, J. Chin. Med. Assoc, doi:10.1097/JCMA.0000000000000463
Valenti, Antonini, Lactoferrin: An important host defence against microbial and viral attack, Cell. Mol. Life Sci, doi:10.1007/s00018-005-5372-0
Valenti, Frioni, Rossi, Ranucci, De Fino et al., Aerosolized bovine lactoferrin reduces neutrophils and pro-inflammatory cytokines in mouse models of Pseudomonas aeruginosa lung infections, Biochem. Cell Biol, doi:10.1139/bcb-2016-0050
Van Der Strate, Beljaars, Molema, Harmsen, Meijer, Antiviral activities of lactoferrin, Antiviral Res, doi:10.1016/S0166-3542(01)00195-4
Vincent, Bergeron, Benjannet, Erickson, Rollin et al., Chloroquine is a potent inhibitor of SARS coronavirus infection and spread, Virol. J, doi:10.1186/1743-422X-2-69
Wakabayashi, Oda, Yamauchi, Abe, Lactoferrin for prevention of common viral infections, J. Infect. Chemother, doi:10.1016/j.jiac.2014.08.003
Wang, Cao, Zhang, Yang, Liu et al., Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) In, Vitro. Cell Res, doi:10.1038/s41422-020-0282-0
Wang, Zhao, Liu, Wang, Zhang et al., Pattern of liver injury in adult patients with COVID-19: A retrospective analysis of 105 patients, Mil. Med. Res, doi:10.1186/s40779-020-00256-6
Xu, Dong, An, Lv, Yin et al., Clinical and computed tomographic imaging features of novel coronavirus pneumonia caused by SARS-CoV-2, J. Infect, doi:10.1016/j.jinf.2020.02.017
Xu, Shi, Wang, Zhang, Huang et al., Pathological findings of COVID-19 associated with acute respiratory distress syndrome, Lancet Respir. Med, doi:10.1016/S2213-2600(20)30076-X
Xu, Zhong, Deng, Peng, Dan et al., High expression of ACE2 receptor of 2019-nCoV on the epithelial cells of oral mucosa, Int. J. Oral Sci, doi:10.1038/s41368-020-0074-x
Zhang, Chen, Swaroop, Xu, Wang et al., Heparan sulfate assists SARS-CoV-2 in cell entry and can be targeted by approved drugs In Vitro, Cell. Discov, doi:10.1038/s41421-020-00222-5
Zhang, Han, Ye, Liu, Tian et al., Influence of Phospholipids Structure on the Physicochemical Properties and In Vitro Digestibility of Lactoferrin-Loaded Liposomes, Food Biophys
Zhou, Yu, Du, Fan, Liu et al., Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study, Lancet, doi:10.1016/S0140-6736(20)30566-3
Zou, Chen, Zou, Han, Hao et al., Single-cell RNA-seq data analysis on the receptor ACE2 expression reveals the potential risk of different human organs vulnerable to 2019-nCoV infection, Front. Med, doi:10.1007/s11684-020-0754-0
Zwirzitz, Reiter, Skrabana, Ohradanova-Repic, Majdic et al., Lactoferrin is a natural inhibitor of plasminogen activation, J. Biol. Chem, doi:10.1074/jbc.RA118.003145
DOI record:
{
"DOI": "10.3390/ijerph182010985",
"ISSN": [
"1660-4601"
],
"URL": "http://dx.doi.org/10.3390/ijerph182010985",
"abstract": "<jats:p>Lactoferrin (Lf), a multifunctional cationic glycoprotein synthesized by exocrine glands and neutrophils, possesses an in vitro antiviral activity against SARS-CoV-2. Thus, we conducted an in vivo preliminary study to investigate the antiviral effect of oral and intranasal liposomal bovine Lf (bLf) in asymptomatic and mild-to-moderate COVID-19 patients. From April 2020 to June 2020, a total of 92 mild-to-moderate (67/92) and asymptomatic (25/92) COVID-19 patients were recruited and divided into three groups. Thirty-two patients (14 hospitalized and 18 in home-based isolation) received only oral and intranasal liposomal bLf; 32 hospitalized patients were treated only with standard of care (SOC) treatment; and 28, in home-based isolation, did not take any medication. Furthermore, 32 COVID-19 negative, untreated, healthy subjects were added for ancillary analysis. Liposomal bLf-treated COVID-19 patients obtained an earlier and significant (p < 0.0001) SARS-CoV-2 RNA negative conversion compared to the SOC-treated and untreated COVID-19 patients (14.25 vs. 27.13 vs. 32.61 days, respectively). Liposomal bLf-treated COVID-19 patients showed fast clinical symptoms recovery compared to the SOC-treated COVID-19 patients. In bLf-treated patients, a significant decrease in serum ferritin, IL-6, and D-dimers levels was observed. No adverse events were reported. These observations led us to speculate a potential role of bLf in the management of mild-to-moderate and asymptomatic COVID-19 patients.</jats:p>",
"alternative-id": [
"ijerph182010985"
],
"author": [
{
"affiliation": [],
"family": "Campione",
"given": "Elena",
"sequence": "first"
},
{
"affiliation": [],
"family": "Lanna",
"given": "Caterina",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0003-4025-7882",
"affiliation": [],
"authenticated-orcid": false,
"family": "Cosio",
"given": "Terenzio",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-1252-6080",
"affiliation": [],
"authenticated-orcid": false,
"family": "Rosa",
"given": "Luigi",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0003-0753-6003",
"affiliation": [],
"authenticated-orcid": false,
"family": "Conte",
"given": "Maria Pia",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-7511-6575",
"affiliation": [],
"authenticated-orcid": false,
"family": "Iacovelli",
"given": "Federico",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-6162-6814",
"affiliation": [],
"authenticated-orcid": false,
"family": "Romeo",
"given": "Alice",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-3990-4758",
"affiliation": [],
"authenticated-orcid": false,
"family": "Falconi",
"given": "Mattia",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Del Vecchio",
"given": "Claudia",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Franchin",
"given": "Elisa",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Lia",
"given": "Maria Stella",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Minieri",
"given": "Marilena",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Chiaramonte",
"given": "Carlo",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-9943-9130",
"affiliation": [],
"authenticated-orcid": false,
"family": "Ciotti",
"given": "Marco",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-8702-1116",
"affiliation": [],
"authenticated-orcid": false,
"family": "Nuccetelli",
"given": "Marzia",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-7442-2252",
"affiliation": [],
"authenticated-orcid": false,
"family": "Terrinoni",
"given": "Alessandro",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Iannuzzi",
"given": "Ilaria",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Coppeta",
"given": "Luca",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Magrini",
"given": "Andrea",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Bernardini",
"given": "Sergio",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Sabatini",
"given": "Stefano",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Rosapepe",
"given": "Felice",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Bartoletti",
"given": "Pier Luigi",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Moricca",
"given": "Nicola",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Di Lorenzo",
"given": "Andrea",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Andreoni",
"given": "Massimo",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0003-1452-0333",
"affiliation": [],
"authenticated-orcid": false,
"family": "Sarmati",
"given": "Loredana",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Miani",
"given": "Alessandro",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Piscitelli",
"given": "Prisco",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Squillaci",
"given": "Ettore",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Valenti",
"given": "Piera",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-8697-6896",
"affiliation": [],
"authenticated-orcid": false,
"family": "Bianchi",
"given": "Luca",
"sequence": "additional"
}
],
"container-title": [
"International Journal of Environmental Research and Public Health"
],
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2021,
10,
20
]
],
"date-time": "2021-10-20T07:01:13Z",
"timestamp": 1634713273000
},
"deposited": {
"date-parts": [
[
2021,
10,
20
]
],
"date-time": "2021-10-20T13:31:59Z",
"timestamp": 1634736719000
},
"indexed": {
"date-parts": [
[
2021,
12,
10
]
],
"date-time": "2021-12-10T22:43:51Z",
"timestamp": 1639176231504
},
"is-referenced-by-count": 0,
"issn-type": [
{
"type": "electronic",
"value": "1660-4601"
}
],
"issue": "20",
"issued": {
"date-parts": [
[
2021,
10,
19
]
]
},
"journal-issue": {
"issue": "20",
"published-online": {
"date-parts": [
[
2021,
10
]
]
}
},
"language": "en",
"license": [
{
"URL": "https://creativecommons.org/licenses/by/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
10,
19
]
],
"date-time": "2021-10-19T00:00:00Z",
"timestamp": 1634601600000
}
}
],
"link": [
{
"URL": "https://www.mdpi.com/1660-4601/18/20/10985/pdf",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "1968",
"original-title": [],
"page": "10985",
"prefix": "10.3390",
"published": {
"date-parts": [
[
2021,
10,
19
]
]
},
"published-online": {
"date-parts": [
[
2021,
10,
19
]
]
},
"publisher": "MDPI AG",
"reference": [
{
"DOI": "10.1016/S0140-6736(20)30251-8",
"doi-asserted-by": "publisher",
"key": "ref1"
},
{
"DOI": "10.1080/22221751.2020.1729069",
"doi-asserted-by": "publisher",
"key": "ref2"
},
{
"DOI": "10.1038/s41586-020-2180-5",
"doi-asserted-by": "publisher",
"key": "ref3"
},
{
"DOI": "10.1038/s41579-018-0118-9",
"doi-asserted-by": "publisher",
"key": "ref4"
},
{
"DOI": "10.1146/annurev-virology-110615-042301",
"doi-asserted-by": "publisher",
"key": "ref5"
},
{
"DOI": "10.1371/journal.pone.0023710",
"doi-asserted-by": "publisher",
"key": "ref6"
},
{
"DOI": "10.1080/22221751.2021.1888660",
"doi-asserted-by": "publisher",
"key": "ref7"
},
{
"DOI": "10.1021/acschemneuro.0c00172",
"doi-asserted-by": "publisher",
"key": "ref8"
},
{
"DOI": "10.1038/s41368-020-0074-x",
"doi-asserted-by": "publisher",
"key": "ref9"
},
{
"DOI": "10.1007/s11684-020-0754-0",
"doi-asserted-by": "publisher",
"key": "ref10"
},
{
"DOI": "10.1016/j.resp.2020.103548",
"doi-asserted-by": "publisher",
"key": "ref11"
},
{
"DOI": "10.1016/S0140-6736(20)30211-7",
"doi-asserted-by": "publisher",
"key": "ref12"
},
{
"DOI": "10.1016/j.omtm.2020.06.013",
"doi-asserted-by": "publisher",
"key": "ref13"
},
{
"DOI": "10.1016/S0140-6736(20)30154-9",
"doi-asserted-by": "publisher",
"key": "ref14"
},
{
"DOI": "10.3760/cma.j.cn501120-20200224-00088",
"doi-asserted-by": "publisher",
"key": "ref15"
},
{
"DOI": "10.1056/NEJMoa2002032",
"doi-asserted-by": "publisher",
"key": "ref16"
},
{
"DOI": "10.1097/JCMA.0000000000000463",
"doi-asserted-by": "publisher",
"key": "ref17"
},
{
"DOI": "10.1002/jmv.25757",
"doi-asserted-by": "publisher",
"key": "ref18"
},
{
"DOI": "10.1038/s41577-020-0331-4",
"doi-asserted-by": "publisher",
"key": "ref19"
},
{
"DOI": "10.3174/ajnr.A6588",
"doi-asserted-by": "publisher",
"key": "ref20"
},
{
"DOI": "10.26434/chemrxiv.11938173.v8",
"doi-asserted-by": "publisher",
"key": "ref21"
},
{
"DOI": "10.1016/S0140-6736(20)30566-3",
"doi-asserted-by": "publisher",
"key": "ref22"
},
{
"DOI": "10.3390/molecules25081997",
"doi-asserted-by": "publisher",
"key": "ref23"
},
{
"key": "ref24"
},
{
"DOI": "10.1016/S0140-6736(20)32013-4",
"doi-asserted-by": "publisher",
"key": "ref25"
},
{
"DOI": "10.1016/j.jpha.2020.03.009",
"doi-asserted-by": "publisher",
"key": "ref26"
},
{
"DOI": "10.1016/j.jgg.2020.02.001",
"doi-asserted-by": "publisher",
"key": "ref27"
},
{
"DOI": "10.1038/s41422-020-0282-0",
"doi-asserted-by": "publisher",
"key": "ref28"
},
{
"DOI": "10.1186/1743-422X-2-69",
"doi-asserted-by": "publisher",
"key": "ref29"
},
{
"DOI": "10.3390/ijms18091985",
"doi-asserted-by": "publisher",
"key": "ref30"
},
{
"DOI": "10.1007/s00018-005-5372-0",
"doi-asserted-by": "publisher",
"key": "ref31"
},
{
"key": "ref32"
},
{
"DOI": "10.2903/j.efsa.2012.2701",
"doi-asserted-by": "publisher",
"key": "ref33"
},
{
"DOI": "10.3389/fphar.2021.666600",
"doi-asserted-by": "publisher",
"key": "ref34"
},
{
"DOI": "10.3390/nu13020328",
"doi-asserted-by": "publisher",
"key": "ref35"
},
{
"DOI": "10.3390/molecules16086992",
"doi-asserted-by": "publisher",
"key": "ref36"
},
{
"DOI": "10.1016/j.jiac.2014.08.003",
"doi-asserted-by": "publisher",
"key": "ref37"
},
{
"DOI": "10.1023/B:BIOM.0000027710.13543.3f",
"doi-asserted-by": "publisher",
"key": "ref38"
},
{
"DOI": "10.3390/molecules24071323",
"doi-asserted-by": "publisher",
"key": "ref39"
},
{
"DOI": "10.1021/bi8012164",
"doi-asserted-by": "publisher",
"key": "ref40"
},
{
"DOI": "10.1139/o11-075",
"doi-asserted-by": "publisher",
"key": "ref41"
},
{
"DOI": "10.3389/fimmu.2017.01438",
"doi-asserted-by": "publisher",
"key": "ref42"
},
{
"DOI": "10.3390/ijms21144903",
"doi-asserted-by": "publisher",
"key": "ref43"
},
{
"DOI": "10.1074/jbc.RA118.003145",
"doi-asserted-by": "publisher",
"key": "ref44"
},
{
"DOI": "10.1007/s11739-020-02432-x",
"doi-asserted-by": "publisher",
"key": "ref45"
},
{
"DOI": "10.1016/j.jinf.2020.02.017",
"doi-asserted-by": "publisher",
"key": "ref46"
},
{
"DOI": "10.1161/CIRCULATIONAHA.120.048238",
"doi-asserted-by": "publisher",
"key": "ref47"
},
{
"DOI": "10.1001/jamacardio.2020.1834",
"doi-asserted-by": "publisher",
"key": "ref48"
},
{
"DOI": "10.1016/j.hrthm.2020.05.014",
"doi-asserted-by": "publisher",
"key": "ref49"
},
{
"DOI": "10.1007/s10534-020-00236-2",
"doi-asserted-by": "publisher",
"key": "ref50"
},
{
"DOI": "10.1001/jama.2020.22813",
"doi-asserted-by": "publisher",
"key": "ref51"
},
{
"DOI": "10.1016/S0166-3542(01)00195-4",
"doi-asserted-by": "publisher",
"key": "ref52"
},
{
"DOI": "10.1007/s00253-015-6818-4",
"doi-asserted-by": "publisher",
"key": "ref53"
},
{
"DOI": "10.1101/2020.05.10.087288",
"doi-asserted-by": "publisher",
"key": "ref54"
},
{
"DOI": "10.1038/s41421-020-00222-5",
"doi-asserted-by": "publisher",
"key": "ref55"
},
{
"DOI": "10.1093/infdis/jiaa206",
"doi-asserted-by": "publisher",
"key": "ref56"
},
{
"DOI": "10.1016/j.cmi.2020.07.008",
"doi-asserted-by": "publisher",
"key": "ref57"
},
{
"DOI": "10.3390/jcm10184276",
"doi-asserted-by": "publisher",
"key": "ref58"
},
{
"DOI": "10.1002/jmv.25948",
"doi-asserted-by": "publisher",
"key": "ref59"
},
{
"DOI": "10.1016/j.jpha.2020.03.001",
"doi-asserted-by": "publisher",
"key": "ref60"
},
{
"DOI": "10.1111/jth.14768",
"doi-asserted-by": "publisher",
"key": "ref61"
},
{
"DOI": "10.1016/S2213-2600(20)30076-X",
"doi-asserted-by": "publisher",
"key": "ref62"
},
{
"DOI": "10.1016/j.pulmoe.2020.07.003",
"doi-asserted-by": "publisher",
"key": "ref63"
},
{
"DOI": "10.1002/jmv.26365",
"doi-asserted-by": "publisher",
"key": "ref64"
},
{
"DOI": "10.3389/fimmu.2017.00705",
"doi-asserted-by": "publisher",
"key": "ref65"
},
{
"DOI": "10.1139/bcb-2016-0050",
"doi-asserted-by": "publisher",
"key": "ref66"
},
{
"DOI": "10.3389/fimmu.2018.02123",
"doi-asserted-by": "publisher",
"key": "ref67"
},
{
"DOI": "10.1111/jth.14879",
"doi-asserted-by": "publisher",
"key": "ref68"
},
{
"DOI": "10.1186/s13017-020-00323-2",
"doi-asserted-by": "publisher",
"key": "ref69"
},
{
"DOI": "10.1080/1354750X.2020.1797880",
"doi-asserted-by": "publisher",
"key": "ref70"
},
{
"DOI": "10.1016/j.cyto.2020.155302",
"doi-asserted-by": "publisher",
"key": "ref71"
},
{
"DOI": "10.1016/j.metabol.2020.154260",
"doi-asserted-by": "publisher",
"key": "ref72"
},
{
"DOI": "10.1515/cclm-2020-0338",
"doi-asserted-by": "publisher",
"key": "ref73"
},
{
"DOI": "10.1186/s40779-020-00256-6",
"doi-asserted-by": "publisher",
"key": "ref74"
},
{
"DOI": "10.1371/journal.pmed.1003501",
"doi-asserted-by": "publisher",
"key": "ref75"
},
{
"DOI": "10.1038/s41598-021-91089-3",
"doi-asserted-by": "publisher",
"key": "ref76"
},
{
"DOI": "10.3168/jds.2012-6072",
"doi-asserted-by": "publisher",
"key": "ref77"
},
{
"DOI": "10.1007/s11483-019-09581-3",
"article-title": "Influence of Phospholipids Structure on the Physicochemical Properties and In Vitro Digestibility of Lactoferrin-Loaded Liposomes",
"author": "Zhang",
"doi-asserted-by": "crossref",
"journal-title": "Food Biophys.",
"key": "ref78",
"year": "2019"
},
{
"DOI": "10.1016/j.xcrm.2020.100126",
"doi-asserted-by": "publisher",
"key": "ref79"
}
],
"reference-count": 79,
"references-count": 79,
"relation": {},
"score": 1,
"short-container-title": [
"IJERPH"
],
"short-title": [],
"source": "Crossref",
"subject": [
"Health, Toxicology and Mutagenesis",
"Public Health, Environmental and Occupational Health"
],
"subtitle": [],
"title": [
"Lactoferrin as Antiviral Treatment in COVID-19 Management: Preliminary Evidence"
],
"type": "journal-article",
"volume": "18"
}