Analyzing the Difference in the Length of Stay (LOS) in Moderate to Severe COVID-19 Patients Receiving Hydroxychloroquine or Favipiravir
et al., Pharmaceuticals, doi:10.3390/ph15121456, Nov 2022
Retrospective 200 hospitalized COVID-19 patients in Saudi Arabia, showing no significant difference in outcomes between HCQ and favipiravir.
Potential risks of favipiravir include kidney injury1-3, liver injury2-5, cardiovascular events5,6, pulmonary toxicity6,7, and mutagenicity, carcinogenicity, teratogenicity, embryotoxicity, and the creation of dangerous variants8-14.
Study covers favipiravir and HCQ.
|
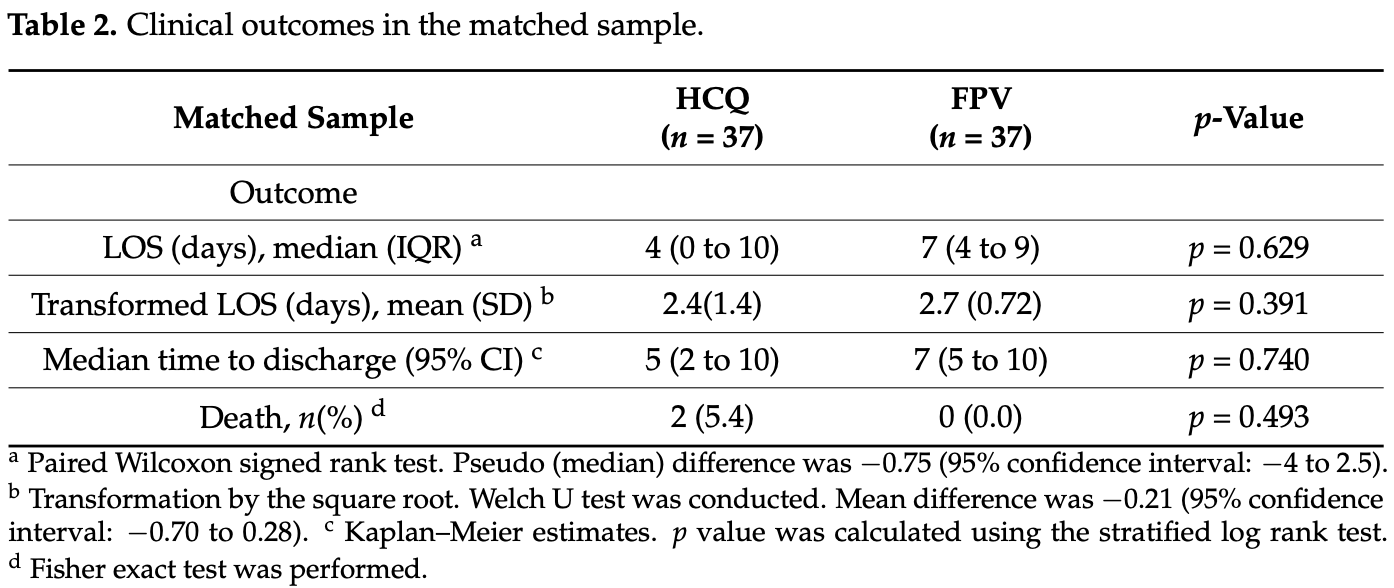
risk of death, 80.0% lower, RR 0.20, p = 0.49, treatment 0 of 37 (0.0%), control 2 of 37 (5.4%), NNT 18, relative risk is not 0 because of continuity correction due to zero events (with reciprocal of the contrasting arm), propensity score matching.
|
|
hospitalization time, 75.0% higher, relative time 1.75, p = 0.63, treatment 37, control 37, propensity score matching.
|
|
time to discharge, 40.0% higher, relative time 1.40, p = 0.74, treatment 37, control 37, propensity score matching.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
1.
Abdulaziz et al., Clinical Features and Prognosis of Acute Kidney Injury in Hospital-Admitted Patients with COVID-19 in Egypt: A Single-Center Experience, Mansoura Medical Journal, doi:10.58775/2735-3990.1433.
2.
Ülger et al., Experimental evaluation of favipiravir (T-705)-induced liver and kidney toxicity in rats, Food and Chemical Toxicology, doi:10.1016/j.fct.2025.115472.
3.
El-Fetouh et al., Experimental Studies on Some Drugs Used in Covid-19 Treatment (Favipiravir and Dexamethasone) in Albino Rats, Journal of Advanced Veterinary Research, 13:10, www.advetresearch.com/index.php/AVR/article/view/1635.
4.
Almutairi et al., Liver Injury in Favipiravir-Treated COVID-19 Patients: Retrospective Single-Center Cohort Study, Tropical Medicine and Infectious Disease, doi:10.3390/tropicalmed8020129.
5.
Siby et al., Temporal Trends in Serious Adverse Events Associated with Oral Antivirals During the COVID-19 Pandemic: Insights from the FAERS Database (2020–2023), Open Forum Infectious Diseases, doi:10.1093/ofid/ofaf695.1825.
6.
Ozhan et al., Evaluation of the cardiopulmonary effects of repurposed COVID-19 therapeutics in healthy rats, Scientific Reports, doi:10.1038/s41598-025-31048-4.
7.
Ülger (B) et al., Evaluation of the effects of favipiravir (T-705) on the lung tissue of healty rats: An experimental study, Food and Chemical Toxicology, doi:10.1016/j.fct.2025.115235.
8.
Zhirnov et al., Favipiravir: the hidden threat of mutagenic action, Journal of microbiology, epidemiology and immunobiology, doi:10.36233/0372-9311-114.
9.
Waters et al., Human genetic risk of treatment with antiviral nucleoside analog drugs that induce lethal mutagenesis: the special case of molnupiravir, Environmental and Molecular Mutagenesis, doi:10.1002/em.22471.
10.
Hadj Hassine et al., Lethal Mutagenesis of RNA Viruses and Approved Drugs with Antiviral Mutagenic Activity, Viruses, doi:10.3390/v14040841.
11.
Shum, C., An investigational study into the drug-associated mutational signature in SARS-CoV-2 viruses, The University of Hong Kong, PhD Thesis, hub.hku.hk/handle/10722/344396.
12.
Shiraki et al., Convenient screening of the reproductive toxicity of favipiravir and antiviral drugs in Caenorhabditis elegans, Heliyon, doi:10.1016/j.heliyon.2024.e35331.
Alosaimi et al., 24 Nov 2022, retrospective, Saudi Arabia, peer-reviewed, 13 authors, study period April 2020 - March 2021, this trial compares with another treatment - results may be better when compared to placebo.
Contact: w.alturaiki@mu.edu.sa (corresponding author).
Analyzing the Difference in the Length of Stay (LOS) in Moderate to Severe COVID-19 Patients Receiving Hydroxychloroquine or Favipiravir
Pharmaceuticals, doi:10.3390/ph15121456
Background: The coronavirus 2019 (COVID-19) disease, caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) virus led to a global pandemic. HCQ and FPV were used early in the pandemic as a treatment modality for COVID-19. Various studies evaluated the HCQ and FPV effectiveness, based on the mortality endpoint and showed conflicting results. We hypothesize that analyzing the difference in the LOS as a significant endpoint would be of a major interest, especially for healthcare providers, to prevent a lengthy hospitalization and disease progression. Methods: This is a retrospective observational study, conducted via a medical chart review of COVD-19 patients who were admitted between April 2020 and March 2021 with a moderate to severe illness. The LOS endpoint was tested using the paired Wilcoxon signed-rank (WSR) model. Prior to using the WSR model, the balance between the HCQ and FPV groups, the propensity score matching, the LOS distribution, and the normality assumptions were tested. Two sensitivity statistical analyses were conducted to confirm the results (stratified log-rank test and U Welch test after transforming the LOS by the squared root values). Results: A total of 200 patients were included for the analysis: 83 patients in the HCQ group and 117 patients in the FPV group. Thirty-seven patients were matched in each group. The LOS data was positively skewed and violated the normality (Shapiro-Wilk p < 0.001) and had an unequal variance (Levene's test, p = 0.019). The WSR test showed no statistical significance in the LOS endpoint, with a median of −0.75 days (95% confidence interval: −4.0 to 2.5, p = 0.629), in favor of the HCQ group (four days), in comparison to seven days of the FPV group. The WSR findings were further confirmed with the stratified log rank test (p = 740) and the U Welch test (p = 391). Conclusions: The study concluded that the HCQ and FPV treatments have a comparable effectiveness in terms of the LOS in the moderate to severe COVID-19 patients. This study highlights the importance of analyzing the LOS as a relevant endpoint, in order to prevent the costs of a lengthy hospitalization and disease progression. The current study also emphasizes the
Funding: Princess Nourah bint Abdulrahman University Researchers Supporting Project number (PNURSP2022R299), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia.
Institutional Review Board Statement: The protocol was approved by the Institutional Review Board at King Fahad Medical City (IRB 22-175). Hence, the written informed consent was waived due to the retrospective nature of the study and it was not required, since only unidentifiable data were extracted from the medical records. Informed Consent Statement: Informed consent was waived due to the retrospective nature of the study. Data Availability Statement: Data is contained within the article and Supplementary Material.
Conflicts of Interest: The authors declare no conflict of interest.
References
Abd-Elsalam, Esmail, Khalaf, Abdo, Medhat et al., Hydroxychloroquine in the treatment of COVID-19: A multicenter randomized controlled study, Am. J. Trop. Med. Hyg, doi:10.4269/ajtmh.20-0873
Ader, Peiffer-Smadja, Poissy, Bouscambert-Duchamp, Belhadi et al., An open-label randomized controlled trial of the effect of lopinavir/ritonavir, lopinavir/ritonavir plus IFN-β-1a and hydroxychloroquine in hospitalized patients with COVID-19, Clin. Microbiol. Infect, doi:10.1016/j.cmi.2021.05.020
Akbari, Razmi, Sedaghat, Dana, Amiri et al., Comparative effectiveness of pharmacological interventions on mortality and the average length of hospital stay of patients with COVID-19: A systematic review and meta-analysis of randomized controlled trials, Expert Rev. Anti-Infect. Ther, doi:10.1080/14787210.2022.1997587
Alamer, Alrashed, Alfaifi, Alosaimi, Alhassar et al., Effectiveness and safety of favipiravir compared to supportive care in moderately to critically ill COVID-19 patients: A retrospective study with propensity score matching sensitivity analysis, Curr. Med. Res. Opin
Alotaibi, Ali, Bakhshwin, Alatawi, Alotaibi et al., Effectiveness and Safety of Favipiravir Compared to Hydroxychloroquine for Management of COVID-19: A Retrospective Study, Int. J. Gen. Med, doi:10.2147/IJGM.S329881
Austin, A Tutorial and Case Study in Propensity Score Analysis: An Application to Estimating the Effect of In-Hospital Smoking Cessation Counseling on Mortality, Multivar. Behav. Res, doi:10.1080/00273171.2011.540480
Borba, Val, Sampaio, Alexandre, Melo et al., Chloroquine diphosphate in two different dosages as adjunctive therapy of hospitalized patients with severe respiratory syndrome in the context of coronavirus (SARS-CoV-2) infection: Preliminary safety results of a randomized, double-blinded, phase IIb clinical trial (CloroCOVID-19 Study), MedRxiv, doi:10.1101/2020.04.07.20056424
Buuren, Mice: Multivariate Imputation by Chained Equations
Chazard, Ficheur, Beuscart, Preda, How to Compare the Length of Stay of Two Samples of Inpatients? A Simulation Study to Compare Type I and Type II Errors of 12 Statistical Tests, Value Health, doi:10.1016/j.jval.2017.02.009
Costanzo, De Giglio, Roviello, SARS-CoV-2: Recent Reports on Antiviral Therapies Based on Lopinavir/Ritonavir, Darunavir/Umifenovir, Hydroxychloroquine, Remdesivir, Favipiravir and other Drugs for the Treatment of the New Coronavirus, Curr. Med. Chem, doi:10.2174/0929867327666200416131117
Eroglu, Kartal, Kongur, Comparison of the intensive care unit admission rate and mortalities of COVID-19 patients who received Hydroxychloroquine and Favipiravir in the ward, J. Anesth. Crit. Care Open Access, doi:10.15406/jaccoa.2022.14.00504
Extance, COVID-19: What is the evidence for the antiviral Paxlovid?, BMJ, doi:10.1136/bmj.o1037
Gao, Tian, Yang, Breakthrough: Chloroquine phosphate has shown apparent efficacy in treatment of COVID-19 associated pneumonia in clinical studies, Biosci. Trends, doi:10.5582/bst.2020.01047
Gautret, Lagier, Parola, Hoang, Meddeb et al., Hydroxychloroquine and azithromycin as a treatment of COVID-19: Results of an open-label non-randomized clinical trial, Int. J. Antimicrob. Agents, doi:10.1016/j.ijantimicag.2020.105949
Geleris, Sun, Platt, Zucker, Baldwin et al., Observational study of hydroxychloroquine in hospitalized patients with COVID-19, N. Engl. J. Med
Greife, Cobalt: Covariate Balance Tables and Plots
Group, Dexamethasone in hospitalized patients with COVID-19-Preliminary report, N. Engl. J. Med
Guo, Lu, Tan, Kuang, Luo et al., Risk factors on admission associated with hospital length of stay in patients with COVID-19: A retrospective cohort study, Sci. Rep, doi:10.1038/s41598-021-86853-4
Hoekstra, Kiers, Johnson, Are Assumptions of Well-Known Statistical Techniques Checked, and Why (Not)?, Front. Psychol, doi:10.3389/fpsyg.2012.00137
Kim, Sparks, Liew, Putman, Berenbaum et al., A Rush to Judgment? Rapid Reporting and Dissemination of Results and Its Consequences Regarding the Use of Hydroxychloroquine for COVID-19, Ann. Intern. Med, doi:10.7326/M20-1223
Kohl, Package MKpower: Wilcoxon Rank Sum and Signed Rank Tests
Mahévas, Tran, Roumier, Chabrol, Paule et al., No evidence of clinical efficacy of hydroxychloroquine in patients hospitalised for COVID-19 infection and requiring oxygen: Results of a study using routinely collected data to emulate a target trial, MedRxiv, doi:10.1101/2020.04.10.20060699
Membrillo, Ramírez-Olivencia, Estébanez, De Dios, Herrero et al., Early hydroxychloroquine is associated with an increase of survival in COVID-19 patients: An observational study, Preprints, doi:10.20944/preprints202005.0057.v1
Patino, Ferreira, Meeting the assumptions of statistical tests: An important and often forgotten step to reporting valid results, J. Bras. Pneumol, doi:10.1590/s1806-37562018000000303
Pilkington, Pepperrell, Hill, A review of the safety of favipiravir-A potential treatment in the COVID-19 pandemic?, J. Virus Erad, doi:10.1016/S2055-6640(20)30016-9
Pishgar, MatchThem: Matching and Weighting Multiply Imputed Datasets
Qualls, Pallin, Schuur, Parametric Versus Nonparametric Statistical Tests: The Length of Stay Example, Acad. Emerg. Med, doi:10.1111/j.1553-2712.2010.00874.x
Scheff, Fundamental Statistical Principles for the Neurobiologist, Chapter 8-Nonparametric Statistics, doi:10.1016/B978-0-12-804753-8.00008-7
Therneau, Survival: Survival Analysis
Torjesen, COVID-19: Hydroxychloroquine does not benefit hospitalised patients, UK trial finds, BMJ, doi:10.1136/bmj.m2263
Turan, Menteş, Özel, Şerefhano Glu, Aydo Gan et al., The effect of favipiravir versus hydroxychloroquine on clinical and laboratory findings in COVID-19 in healthcare workers, Braz. J. Infect. Dis, doi:10.1016/j.bjid.2022.102328
Udwadia, Singh, Barkate, Patil, Rangwala et al., Efficacy and safety of favipiravir, an oral RNA-dependent RNA polymerase inhibitor, in mild-to-moderate COVID-19: A randomized, comparative, open-label, multicenter, phase 3 clinical trial, Int. J. Infect. Dis, doi:10.1016/j.ijid.2020.11.142
Wei, Diagnosis and Treatment Protocol for Novel Coronavirus Pneumonia (Trial Version 7), Chin. Med. J
Who, Coronavirus Disease (COVID-19) Situation Report; World Health Organization
Wickham, Create Elegant Data Visualisations Using the Grammar of Graphics
Wu, Mcgoogan, Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: Summary of a report of 72,314 cases from the Chinese Center for Disease Control and Prevention, JAMA, doi:10.1001/jama.2020.2648
Yan, Muller, Why Remdesivir Failed: Preclinical Assumptions Overestimate the Clinical Efficacy of Remdesivir for COVID-19 and Ebola, Antimicrob. Agents Chemother, doi:10.1128/AAC.01117-21
DOI record:
{
"DOI": "10.3390/ph15121456",
"ISSN": [
"1424-8247"
],
"URL": "http://dx.doi.org/10.3390/ph15121456",
"abstract": "<jats:p>Background: The coronavirus 2019 (COVID-19) disease, caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) virus led to a global pandemic. HCQ and FPV were used early in the pandemic as a treatment modality for COVID-19. Various studies evaluated the HCQ and FPV effectiveness, based on the mortality endpoint and showed conflicting results. We hypothesize that analyzing the difference in the LOS as a significant endpoint would be of a major interest, especially for healthcare providers, to prevent a lengthy hospitalization and disease progression. Methods: This is a retrospective observational study, conducted via a medical chart review of COVD-19 patients who were admitted between April 2020 and March 2021 with a moderate to severe illness. The LOS endpoint was tested using the paired Wilcoxon signed-rank (WSR) model. Prior to using the WSR model, the balance between the HCQ and FPV groups, the propensity score matching, the LOS distribution, and the normality assumptions were tested. Two sensitivity statistical analyses were conducted to confirm the results (stratified log-rank test and U Welch test after transforming the LOS by the squared root values). Results: A total of 200 patients were included for the analysis: 83 patients in the HCQ group and 117 patients in the FPV group. Thirty-seven patients were matched in each group. The LOS data was positively skewed and violated the normality (Shapiro–Wilk p < 0.001) and had an unequal variance (Levene’s test, p = 0.019). The WSR test showed no statistical significance in the LOS endpoint, with a median of −0.75 days (95% confidence interval: −4.0 to 2.5, p = 0.629), in favor of the HCQ group (four days), in comparison to seven days of the FPV group. The WSR findings were further confirmed with the stratified log rank test (p = 740) and the U Welch test (p = 391). Conclusions: The study concluded that the HCQ and FPV treatments have a comparable effectiveness in terms of the LOS in the moderate to severe COVID-19 patients. This study highlights the importance of analyzing the LOS as a relevant endpoint, in order to prevent the costs of a lengthy hospitalization and disease progression. The current study also emphasizes the importance of applying the appropriate statistical testing when dealing with two-sample paired data and analyzing non-parametric data such as the LOS.</jats:p>",
"alternative-id": [
"ph15121456"
],
"author": [
{
"ORCID": "http://orcid.org/0000-0003-4719-2655",
"affiliation": [],
"authenticated-orcid": false,
"family": "Alosaimi",
"given": "Bandar",
"sequence": "first"
},
{
"ORCID": "http://orcid.org/0000-0001-5154-7477",
"affiliation": [],
"authenticated-orcid": false,
"family": "Alshanbari",
"given": "Huda M.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Alturaiqy",
"given": "Muath",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-6761-9519",
"affiliation": [],
"authenticated-orcid": false,
"family": "AlRawi",
"given": "Halah Z.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Alamri",
"given": "Saad",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Albujaidy",
"given": "Asma",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Bin Sabaan",
"given": "Aljawharah",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Alrashed",
"given": "Ahmed A.",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-2091-1376",
"affiliation": [],
"authenticated-orcid": false,
"family": "Alamer",
"given": "Ahmad",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-1505-3581",
"affiliation": [],
"authenticated-orcid": false,
"family": "Alghofaili",
"given": "Fayez",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Al-Duraymih",
"given": "Khaled",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Alshalani",
"given": "Abdulaziz J.",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-6947-7582",
"affiliation": [],
"authenticated-orcid": false,
"family": "Alturaiki",
"given": "Wael",
"sequence": "additional"
}
],
"container-title": "Pharmaceuticals",
"container-title-short": "Pharmaceuticals",
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2022,
11,
24
]
],
"date-time": "2022-11-24T08:24:09Z",
"timestamp": 1669278249000
},
"deposited": {
"date-parts": [
[
2022,
11,
24
]
],
"date-time": "2022-11-24T09:10:46Z",
"timestamp": 1669281046000
},
"funder": [
{
"award": [
"PNURSP2022R299"
],
"name": "Princess Nourah bint Abdulrahman University"
}
],
"indexed": {
"date-parts": [
[
2022,
11,
25
]
],
"date-time": "2022-11-25T06:13:11Z",
"timestamp": 1669356791483
},
"is-referenced-by-count": 0,
"issue": "12",
"issued": {
"date-parts": [
[
2022,
11,
24
]
]
},
"journal-issue": {
"issue": "12",
"published-online": {
"date-parts": [
[
2022,
12
]
]
}
},
"language": "en",
"license": [
{
"URL": "https://creativecommons.org/licenses/by/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2022,
11,
24
]
],
"date-time": "2022-11-24T00:00:00Z",
"timestamp": 1669248000000
}
}
],
"link": [
{
"URL": "https://www.mdpi.com/1424-8247/15/12/1456/pdf",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "1968",
"original-title": [],
"page": "1456",
"prefix": "10.3390",
"published": {
"date-parts": [
[
2022,
11,
24
]
]
},
"published-online": {
"date-parts": [
[
2022,
11,
24
]
]
},
"publisher": "MDPI AG",
"reference": [
{
"key": "ref_1",
"unstructured": "WHO (2022). Coronavirus Disease (COVID-19) Situation Report, World Health Organization."
},
{
"DOI": "10.1001/jama.2020.2648",
"article-title": "Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: Summary of a report of 72,314 cases from the Chinese Center for Disease Control and Prevention",
"doi-asserted-by": "crossref",
"first-page": "1239",
"journal-title": "JAMA",
"key": "ref_2",
"volume": "323",
"year": "2020"
},
{
"key": "ref_3",
"unstructured": "COVID-19 Treatment Guidelines Panel (2022, April 04). Coronavirus Disease 2019 (COVID-19) Treatment Guidelines. National Institutes of Health, Available online: https://www.covid19treatmentguidelines.nih.gov/."
},
{
"DOI": "10.2147/IJGM.S329881",
"article-title": "Effectiveness and Safety of Favipiravir Compared to Hydroxychloroquine for Management of COVID-19: A Retrospective Study",
"doi-asserted-by": "crossref",
"first-page": "5597",
"journal-title": "Int. J. Gen. Med.",
"key": "ref_4",
"volume": "14",
"year": "2021"
},
{
"DOI": "10.2174/0929867327666200416131117",
"article-title": "SARS-CoV-2: Recent Reports on Antiviral Therapies Based on Lopinavir/Ritonavir, Darunavir/Umifenovir, Hydroxychloroquine, Remdesivir, Favipiravir and other Drugs for the Treatment of the New Coronavirus",
"doi-asserted-by": "crossref",
"first-page": "4536",
"journal-title": "Curr. Med. Chem.",
"key": "ref_5",
"volume": "27",
"year": "2020"
},
{
"article-title": "Dexamethasone in hospitalized patients with COVID-19—Preliminary report",
"first-page": "693",
"journal-title": "N. Engl. J. Med.",
"key": "ref_6",
"volume": "384",
"year": "2020"
},
{
"DOI": "10.1097/CM9.0000000000000819",
"article-title": "Diagnosis and Treatment Protocol for Novel Coronavirus Pneumonia (Trial Version 7)",
"doi-asserted-by": "crossref",
"first-page": "1087",
"journal-title": "Chin. Med. J.",
"key": "ref_7",
"volume": "133",
"year": "2020"
},
{
"article-title": "Efficacy and safety of favipiravir, an oral RNA-dependent RNA polymerase inhibitor, in mild-to-moderate COVID-19: A randomized, comparative, open-label, multicenter, phase 3 clinical trial",
"first-page": "62",
"journal-title": "Int. J. Infect. Dis.",
"key": "ref_8",
"volume": "103",
"year": "2020"
},
{
"key": "ref_9",
"unstructured": "Saudi Arabia Ministry of Health (MOH) (2020, September 02). Saudi MoH Protocol for Patients Suspected of/Confirmed with COVID-19, Available online: https://www.moh.gov.sa/Ministry/MediaCenter/Publications/Documents/MOH-therapeutic-protocol-for-COVID-19.pdf."
},
{
"key": "ref_10",
"unstructured": "Saudi Arabia Ministry of Health (MOH) (2020, September 02). Coronavirus Disease COVID-19 Guidelines, v1.3, Available online: https://www.moh.gov.sa/Ministry/MediaCenter/Publications/Documents/Coronavirus-Disease-2019-Guidelines-v1.2.pdf."
},
{
"key": "ref_11",
"unstructured": "Kohl, M. (2022, July 19). Package MKpower: Wilcoxon Rank Sum and Signed Rank Tests. Available online: https://cran.r-project.org/web/packages/MKpower/vignettes/MKpower.html#wilcoxon-rank-sum-and-signed-rank-tests."
},
{
"DOI": "10.1016/B978-0-12-804753-8.00008-7",
"doi-asserted-by": "crossref",
"key": "ref_12",
"unstructured": "Scheff, S. (2016). Fundamental Statistical Principles for the Neurobiologist, Chapter 8—Nonparametric Statistics, Academic Press."
},
{
"DOI": "10.1080/00273171.2011.540480",
"article-title": "A Tutorial and Case Study in Propensity Score Analysis: An Application to Estimating the Effect of In-Hospital Smoking Cessation Counseling on Mortality",
"doi-asserted-by": "crossref",
"first-page": "119",
"journal-title": "Multivar. Behav. Res.",
"key": "ref_13",
"volume": "46",
"year": "2011"
},
{
"key": "ref_14",
"unstructured": "Wickham, H. (2022, July 19). ggplot2: Create Elegant Data Visualisations Using the Grammar of Graphics. Available online: https://cran.r-project.org/web/packages/ggplot2/index.html."
},
{
"key": "ref_15",
"unstructured": "Pishgar, F. (2022, July 19). MatchThem: Matching and Weighting Multiply Imputed Datasets. Available online: https://cran.r-project.org/web/packages/MatchThem/index.html."
},
{
"key": "ref_16",
"unstructured": "Greife, N. (2022, July 19). Cobalt: Covariate Balance Tables and Plots. Available online: https://cran.r-project.org/web/packages/cobalt/index.html."
},
{
"key": "ref_17",
"unstructured": "Buuren, S.V. (2022, July 19). Mice: Multivariate Imputation by Chained Equations. Available online: https://cran.r-project.org/web/packages/mice/index.html."
},
{
"key": "ref_18",
"unstructured": "Therneau, T. (2022, July 19). Survival: Survival Analysis. Available online: https://cran.r-project.org/web/packages/survival/index.html."
},
{
"DOI": "10.15406/jaccoa.2022.14.00504",
"article-title": "Comparison of the intensive care unit admission rate and mortalities of COVID-19 patients who received Hydroxychloroquine and Favipiravir in the ward",
"doi-asserted-by": "crossref",
"first-page": "40",
"journal-title": "J. Anesth. Crit. Care Open Access",
"key": "ref_19",
"volume": "14",
"year": "2022"
},
{
"DOI": "10.1016/j.bjid.2022.102328",
"article-title": "The effect of favipiravir versus hydroxychloroquine on clinical and laboratory findings in COVID-19 in healthcare workers",
"doi-asserted-by": "crossref",
"first-page": "102328",
"journal-title": "Braz. J. Infect. Dis.",
"key": "ref_20",
"volume": "26",
"year": "2022"
},
{
"DOI": "10.4269/ajtmh.20-0873",
"article-title": "Hydroxychloroquine in the treatment of COVID-19: A multicenter randomized controlled study",
"doi-asserted-by": "crossref",
"first-page": "1635",
"journal-title": "Am. J. Trop. Med. Hyg.",
"key": "ref_21",
"volume": "103",
"year": "2020"
},
{
"DOI": "10.1016/j.cmi.2021.05.020",
"article-title": "An open-label randomized controlled trial of the effect of lopinavir/ritonavir, lopinavir/ritonavir plus IFN-β-1a and hydroxychloroquine in hospitalized patients with COVID-19",
"doi-asserted-by": "crossref",
"first-page": "1826",
"journal-title": "Clin. Microbiol. Infect.",
"key": "ref_22",
"volume": "27",
"year": "2021"
},
{
"article-title": "Comparative effectiveness of pharmacological interventions on mortality and the average length of hospital stay of patients with COVID-19: A systematic review and meta-analysis of randomized controlled trials",
"first-page": "585",
"journal-title": "Expert Rev. Anti-Infect. Ther.",
"key": "ref_23",
"volume": "20",
"year": "2021"
},
{
"DOI": "10.1080/03007995.2021.1920900",
"article-title": "Effectiveness and safety of favipiravir compared to supportive care in moderately to critically ill COVID-19 patients: A retrospective study with propensity score matching sensitivity analysis",
"doi-asserted-by": "crossref",
"first-page": "1085",
"journal-title": "Curr. Med. Res. Opin.",
"key": "ref_24",
"volume": "37",
"year": "2021"
},
{
"DOI": "10.20944/preprints202005.0057.v2",
"doi-asserted-by": "crossref",
"key": "ref_25",
"unstructured": "Membrillo, F.J., Ramírez-Olivencia, G., Estébanez, M., de Dios, B., Herrero, M.D., Mata, T., Borobia, A.M., Gutiérrez, C., Simón, M., and Ochoa, A. (2020). Early hydroxychloroquine is associated with an increase of survival in COVID-19 patients: An observational study. Preprints, 2020050057."
},
{
"DOI": "10.1101/2020.04.10.20060699",
"doi-asserted-by": "crossref",
"key": "ref_26",
"unstructured": "Mahévas, M., Tran, V.T., Roumier, M., Chabrol, A., Paule, R., Guillaud, C., Gallien, S., Lepeule, R., Szwebel, T.A., and Lescure, X. (2020). No evidence of clinical efficacy of hydroxychloroquine in patients hospitalised for COVID-19 infection and requiring oxygen: Results of a study using routinely collected data to emulate a target trial. MedRxiv."
},
{
"DOI": "10.1056/NEJMoa2012410",
"article-title": "Observational study of hydroxychloroquine in hospitalized patients with COVID-19",
"doi-asserted-by": "crossref",
"first-page": "2411",
"journal-title": "N. Engl. J. Med.",
"key": "ref_27",
"volume": "382",
"year": "2020"
},
{
"key": "ref_28",
"unstructured": "Borba, M.G.S., Val, F.F.A., Sampaio, V.S., Alexandre, M.A.A., Melo, G.C., Brito, M., Mourão, M.P.G., Brito-Sousa, J.D., Baía-da-Silva, D., and Guerra, M.V.F. (2020). Chloroquine diphosphate in two different dosages as adjunctive therapy of hospitalized patients with severe respiratory syndrome in the context of coronavirus (SARS-CoV-2) infection: Preliminary safety results of a randomized, double-blinded, phase IIb clinical trial (CloroCOVID-19 Study). MedRxiv."
},
{
"article-title": "COVID-19: Hydroxychloroquine does not benefit hospitalised patients, UK trial finds",
"first-page": "m2263",
"journal-title": "BMJ",
"key": "ref_29",
"volume": "369",
"year": "2020"
},
{
"article-title": "COVID-19: What is the evidence for the antiviral Paxlovid?",
"first-page": "o10377",
"journal-title": "BMJ",
"key": "ref_30",
"volume": "37",
"year": "2022"
},
{
"DOI": "10.5582/bst.2020.01047",
"article-title": "Breakthrough: Chloroquine phosphate has shown apparent efficacy in treatment of COVID-19 associated pneumonia in clinical studies",
"doi-asserted-by": "crossref",
"first-page": "72",
"journal-title": "Biosci. Trends",
"key": "ref_31",
"volume": "14",
"year": "2020"
},
{
"DOI": "10.1016/j.ijantimicag.2020.105949",
"article-title": "Hydroxychloroquine and azithromycin as a treatment of COVID-19: Results of an open-label non-randomized clinical trial",
"doi-asserted-by": "crossref",
"first-page": "105949",
"journal-title": "Int. J. Antimicrob. Agents",
"key": "ref_32",
"volume": "56",
"year": "2020"
},
{
"DOI": "10.1016/S2055-6640(20)30016-9",
"article-title": "A review of the safety of favipiravir—A potential treatment in the COVID-19 pandemic?",
"doi-asserted-by": "crossref",
"first-page": "45",
"journal-title": "J. Virus Erad.",
"key": "ref_33",
"volume": "6",
"year": "2020"
},
{
"DOI": "10.1128/AAC.01117-21",
"article-title": "Why Remdesivir Failed: Preclinical Assumptions Overestimate the Clinical Efficacy of Remdesivir for COVID-19 and Ebola",
"doi-asserted-by": "crossref",
"first-page": "e0111721",
"journal-title": "Antimicrob. Agents Chemother.",
"key": "ref_34",
"volume": "65",
"year": "2021"
},
{
"DOI": "10.1038/s41598-021-86853-4",
"article-title": "Risk factors on admission associated with hospital length of stay in patients with COVID-19: A retrospective cohort study",
"doi-asserted-by": "crossref",
"first-page": "7310",
"journal-title": "Sci. Rep.",
"key": "ref_35",
"volume": "11",
"year": "2021"
},
{
"DOI": "10.1590/s1806-37562018000000303",
"article-title": "Meeting the assumptions of statistical tests: An important and often forgotten step to reporting valid results",
"doi-asserted-by": "crossref",
"first-page": "353",
"journal-title": "J. Bras. Pneumol.",
"key": "ref_36",
"volume": "44",
"year": "2018"
},
{
"article-title": "Are Assumptions of Well-Known Statistical Techniques Checked, and Why (Not)?",
"first-page": "137",
"journal-title": "Front. Psychol.",
"key": "ref_37",
"volume": "3",
"year": "2012"
},
{
"DOI": "10.7326/M20-1223",
"article-title": "A Rush to Judgment? Rapid Reporting and Dissemination of Results and Its Consequences Regarding the Use of Hydroxychloroquine for COVID-19",
"doi-asserted-by": "crossref",
"first-page": "819",
"journal-title": "Ann. Intern. Med.",
"key": "ref_38",
"volume": "172",
"year": "2020"
},
{
"DOI": "10.1111/j.1553-2712.2010.00874.x",
"article-title": "Parametric Versus Nonparametric Statistical Tests: The Length of Stay Example",
"doi-asserted-by": "crossref",
"first-page": "1113",
"journal-title": "Acad. Emerg. Med.",
"key": "ref_39",
"volume": "17",
"year": "2010"
},
{
"DOI": "10.1016/j.jval.2017.02.009",
"article-title": "How to Compare the Length of Stay of Two Samples of Inpatients? A Simulation Study to Compare Type I and Type II Errors of 12 Statistical Tests",
"doi-asserted-by": "crossref",
"first-page": "992",
"journal-title": "Value Health",
"key": "ref_40",
"volume": "20",
"year": "2017"
}
],
"reference-count": 40,
"references-count": 40,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.mdpi.com/1424-8247/15/12/1456"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"Drug Discovery",
"Pharmaceutical Science",
"Molecular Medicine"
],
"subtitle": [],
"title": "Analyzing the Difference in the Length of Stay (LOS) in Moderate to Severe COVID-19 Patients Receiving Hydroxychloroquine or Favipiravir",
"type": "journal-article",
"volume": "15"
}
alosaimi