Azvudine for the treatment of cancer patients with COVID-19: a multicenter, real-world, retrospective, cohort study
et al., BMC Cancer, doi:10.1186/s12885-026-15800-1, Mar 2026
Azvudine for COVID-19
48th treatment shown to reduce risk in
January 2023, now with p = 0.0000000021 from 42 studies.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
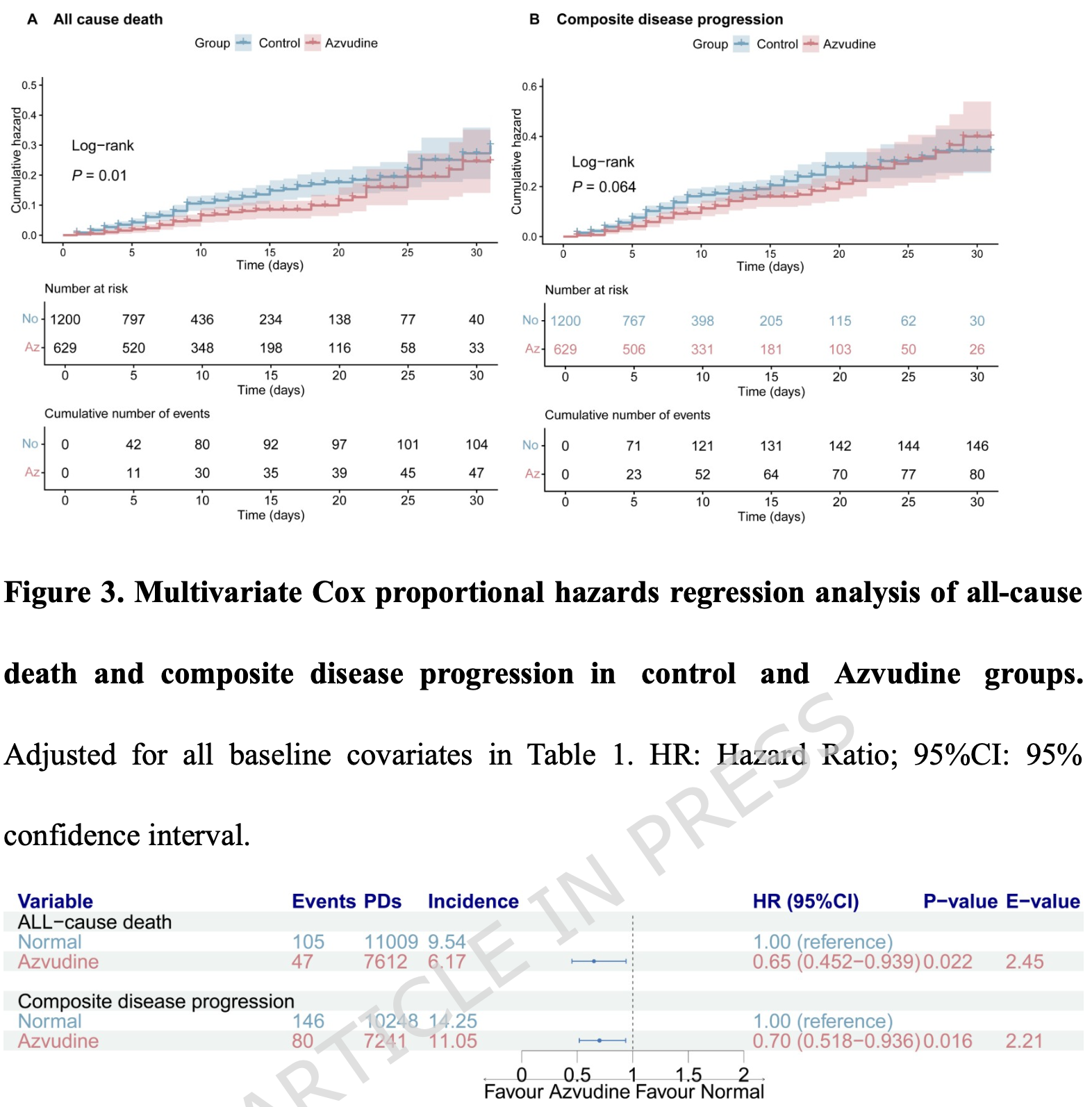
Retrospective 1,829 hospitalized cancer patients with COVID-19 in China showing lower mortality and disease progression with azvudine treatment.
While the authors used a PSM-matched cohort for efficacy, they used 'Available data' (unmatched cohorts: Control n=5609, Azvudine n=593) for the safety analysis in Table 2. In the unmatched data, the azvudine group had significantly higher baseline severity (21.8% severe vs 8.7% in control). Therefore the reported 'distinct toxicity profile' (e.g., higher hypokalemia, lymphocyte changes) is confounded by the fact that the azvudine group was substantially sicker at baseline.
The control group has 105 events over 11,009 person-days (1,200 patients), while the azvudine group has 47 events over 7,612 person-days (629 patients), or roughly 9.2 person-days per patient in the control group versus 12.1 person-days per patient in the azvudine group. If censoring at discharge were non-informative, we would expect roughly similar average follow-up times between groups. Instead, control patients have substantially shorter follow-up on average. This suggests control patients were being discharged (and thus censored) earlier, and since these are the patients who survived to discharge, healthier control patients are being removed from the risk set sooner. That preferentially leaves sicker control patients under observation, inflating the apparent mortality rate in the control group relative to the azvudine group. The raw HR of ~0.86 from the death counts in the PSM groups may be more accurate.
On page 12, the text states '47 deaths occurred in the azvudine group compared with 104 in the control group.' However, the forest plot in Figure 3 explicitly lists '105' events for the control group under all-cause death.
The observation endpoint is defined as 'discharge date, or day 30'. Patients who recover and are discharged early are censored from the mortality analysis at that time. Because healthier patients have systematically shorter follow-up times, this introduces informative censoring that biases the Kaplan-Meier estimates and person-time incidence calculations.
Table 2 contains several calculation errors. For example, azvudine hypokalemia is reported as 193/456 (42.32%) but listed as 43%; control ALT increased is 256/1779 (14.39%) but listed as 14.7%; azvudine lymphocyte count decreased is 220/593 (37.09%) but listed as 36.9%.
The authors state 'All authors declare that they have no competing interests.' However, the senior author (Z.R.) was the lead investigator on previous trials establishing avudine's efficacy.
Table 1 reports continuous variables using mean and standard deviation for laboratory parameters where the standard deviation drastically exceeds the mean. Because these values cannot be negative, this indicates extreme right-skewness, making mean/SD statistically misleading.
Standard of Care (SOC) for COVID-19 in the study country,
China, is poor with low average efficacy for approved treatments3.
This study is excluded in the after exclusion results of meta-analysis:
potential significant differential censoring.
|
risk of death, 35.0% lower, HR 0.65, p = 0.02, treatment 629, control 1,200, propensity score matching, Cox proportional hazards.
|
|
risk of progression, 30.0% lower, HR 0.70, p = 0.02, treatment 629, control 1,200, propensity score matching, Cox proportional hazards.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
1.
Xiong et al., Real-world data of Azvudine-induced hepatotoxicity among hospitalized COVID-19 patients in China: a retrospective case-control study, Frontiers in Pharmacology, doi:10.3389/fphar.2025.1558054.
Li et al., 4 Mar 2026, retrospective, China, peer-reviewed, 14 authors, study period 5 December, 2022 - 31 January, 2023.
Contact: fccrenzg@zzu.edu.cn, johnyuem@zzu.edu.cn.
Abstract: BMC Cancer
https://doi.org/10.1186/s12885-026-15800-1
Article in Press
Azvudine for the treatment of cancer patients
with COVID-19: a multicenter, real-world,
retrospective, cohort study
Lei Li, Wenjie Liu, Ling Wang, Mengzhao Yang, Juan Wang, Guotao Li, Guowu Qian,
Shixi Zhang, Donghai Liu, Hong Luo, Silin Li, Donghua Zhang, Zujiang Yu & Zhigang
Ren
Received: 19 August 2025
Accepted: 24 February 2026
Cite this article as: Li L., Liu W.,
Wang L. et al. Azvudine for the
treatment of cancer patients with
COVID-19: a multicenter, real-world,
retrospective, cohort study. BMC
Cancer (2026). https://doi.org/10.1186/
s12885-026-15800-1
A
S
S
We are providing an unedited version of this manuscript to give early access to its
findings. Before final publication, the manuscript will undergo further editing. Please
note there may be errors present which affect the content, and all legal disclaimers
apply.
IN
E
R
P
If this paper is publishing under a Transparent Peer Review model then Peer
Review reports will publish with the final article.
I
T
R
E
L
C
© The Author(s) 2026. Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International
License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit
to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do
not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this
article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the
article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain
permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
ARTICLE IN PRESS
Title Page
Azvudine for the Treatment of Cancer Patients with COVID-19: A
multicenter, real-world, retrospective, cohort study
Authors
Lei Li 1†, Wenjie Liu 2†, Ling Wang 3, Mengzhao Yang 1, Juan Wang 4,
Guotao Li 5, Guowu Qian 6, Shixi Zhang 7, Donghai Liu 3, Hong Luo 8, Silin
S
S
E
R
P
Li 9, Donghua Zhang 10, Zujiang Yu 1*, Zhigang Ren 1*
Affiliations
1
IN
E
L
C
I
T
R
A
Department of Infectious Diseases, State Key Laboratory of Antiviral Drugs, Pingyuan
Laboratory, the First Affiliated Hospital of Zhengzhou University, Zhengzhou 450052,
China;
2
Pain Department of the First Affiliated Hospital of Zhengzhou University, Zhengzhou
450052, China;
3
Department of Clinical Laboratory, Henan Provincial Chest Hospital Affiliated of
Zhengzhou University, Zhengzhou 450008, China;
4
Department of Liver Disease, the Affiliated Infectious Disease Hospital of Zhengzhou
University, Zhengzhou 450052, China;
5
Department of Infectious Diseases, Luoyang Central Hospital Affiliated of Zhengzhou
University, Luoyang 471000, China;
1
ARTICLE IN PRESS
6
Department of Gastrointestinal Surgery, Nanyang Central Hospital, Nanyang 473009,
China;
7
Department of Infectious Diseases, Shangqiu Municipal Hospital, Shangqiu 476000,
China;
8
Guangshan County People’s Hospital, Guangshan County, Xinyang 465450, China;
9
Department of Respiratory and..
DOI record:
{
"DOI": "10.1186/s12885-026-15800-1",
"ISSN": [
"1471-2407"
],
"URL": "http://dx.doi.org/10.1186/s12885-026-15800-1",
"alternative-id": [
"15800"
],
"assertion": [
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "Received",
"name": "received",
"order": 1,
"value": "19 August 2025"
},
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "Accepted",
"name": "accepted",
"order": 2,
"value": "24 February 2026"
},
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "First Online",
"name": "first_online",
"order": 3,
"value": "4 March 2026"
},
{
"group": {
"label": "Declarations",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 1
},
{
"group": {
"label": "Ethics approval and consent to participate",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 2,
"value": "The study followed the Helsinki Declaration and Rules of Good Clinical Practice. This study was approved by the Institutional Review Board of the First Affiliated Hospital of Zhengzhou University (Approval No. 2023-KY-0865-001). The need for informed consent to participate was waived by the Institutional Review Board of the First Affiliated Hospital of Zhengzhou University due to the retrospective nature of the study and the use of anonymized clinical data."
},
{
"group": {
"label": "Consent for publication",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 3,
"value": "Not applicable. This retrospective cohort study employed anonymized data and did not require individual informed consent for publication."
},
{
"group": {
"label": "Competing interests",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 4,
"value": "The authors declare no competing interests."
}
],
"author": [
{
"affiliation": [],
"family": "Li",
"given": "Lei",
"sequence": "first"
},
{
"affiliation": [],
"family": "Liu",
"given": "Wenjie",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Wang",
"given": "Ling",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Yang",
"given": "Mengzhao",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Wang",
"given": "Juan",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Li",
"given": "Guotao",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Qian",
"given": "Guowu",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Zhang",
"given": "Shixi",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Liu",
"given": "Donghai",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Luo",
"given": "Hong",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Li",
"given": "Silin",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Zhang",
"given": "Donghua",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Yu",
"given": "Zujiang",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ren",
"given": "Zhigang",
"sequence": "additional"
}
],
"container-title": "BMC Cancer",
"container-title-short": "BMC Cancer",
"content-domain": {
"crossmark-restriction": false,
"domain": [
"link.springer.com"
]
},
"created": {
"date-parts": [
[
2026,
3,
4
]
],
"date-time": "2026-03-04T01:03:47Z",
"timestamp": 1772586227000
},
"deposited": {
"date-parts": [
[
2026,
3,
4
]
],
"date-time": "2026-03-04T01:03:52Z",
"timestamp": 1772586232000
},
"funder": [
{
"DOI": "10.13039/501100012166",
"award": [
"2023YFC3043514"
],
"award-info": [
{
"award-number": [
"2023YFC3043514"
]
}
],
"doi-asserted-by": "publisher",
"id": [
{
"asserted-by": "publisher",
"id": "10.13039/501100012166",
"id-type": "DOI"
}
],
"name": "National Key Research and Development Program of China"
},
{
"DOI": "10.13039/501100012166",
"award": [
"2022YFC2303100"
],
"award-info": [
{
"award-number": [
"2022YFC2303100"
]
}
],
"doi-asserted-by": "publisher",
"id": [
{
"asserted-by": "publisher",
"id": "10.13039/501100012166",
"id-type": "DOI"
}
],
"name": "National Key Research and Development Program of China"
},
{
"award": [
"ZYCXTD2023002"
],
"award-info": [
{
"award-number": [
"ZYCXTD2023002"
]
}
],
"name": "Scientific Research and Innovation Team of The First Affiliated Hospital of Zhengzhou University"
},
{
"award": [
"QNCXTD2023002"
],
"award-info": [
{
"award-number": [
"QNCXTD2023002"
]
}
],
"name": "Scientific Research and Innovation Team of The First Affiliated Hospital of Zhengzhou University"
},
{
"name": "Central Plains Talent Program-Central Plains Youth Top Talents"
},
{
"award": [
"HNSWJW-2022013"
],
"award-info": [
{
"award-number": [
"HNSWJW-2022013"
]
}
],
"name": "Young and Middle-aged Academic Leaders of Henan Provincial Health Commission"
},
{
"award": [
"24HASTIT063"
],
"award-info": [
{
"award-number": [
"24HASTIT063"
]
}
],
"name": "Scientific and Technological Innovation Talents of Universities in Henan Province"
}
],
"indexed": {
"date-parts": [
[
2026,
3,
4
]
],
"date-time": "2026-03-04T01:54:28Z",
"timestamp": 1772589268797,
"version": "3.50.1"
},
"is-referenced-by-count": 0,
"issued": {
"date-parts": [
[
2026,
3,
4
]
]
},
"language": "en",
"license": [
{
"URL": "https://creativecommons.org/licenses/by-nc-nd/4.0",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
3,
4
]
],
"date-time": "2026-03-04T00:00:00Z",
"timestamp": 1772582400000
}
},
{
"URL": "https://creativecommons.org/licenses/by-nc-nd/4.0",
"content-version": "am",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
3,
4
]
],
"date-time": "2026-03-04T00:00:00Z",
"timestamp": 1772582400000
}
}
],
"link": [
{
"URL": "https://link.springer.com/article/10.1186/s12885-026-15800-1",
"content-type": "text/html",
"content-version": "vor",
"intended-application": "text-mining"
}
],
"member": "297",
"original-title": [],
"prefix": "10.1186",
"published": {
"date-parts": [
[
2026,
3,
4
]
]
},
"published-online": {
"date-parts": [
[
2026,
3,
4
]
]
},
"publisher": "Springer Science and Business Media LLC",
"reference": [
{
"key": "15800_CR1",
"unstructured": "WHO COVID-19 dashboard. https://data.who.int/dashboards/covid19/cases?n=c."
},
{
"DOI": "10.1016/j.cytogfr.2023.11.004",
"author": "D Wekking",
"doi-asserted-by": "crossref",
"first-page": "110",
"journal-title": "Cytokine Growth Factor Rev",
"key": "15800_CR2",
"unstructured": "Wekking D, Senevirathne TH, Pearce JL, Aiello M, Scartozzi M, Lambertini M, De Silva P, Solinas C. The impact of COVID-19 on cancer patients. Cytokine Growth Factor Rev. 2024;75:110–8.",
"volume": "75",
"year": "2024"
},
{
"DOI": "10.1002/jmv.26232",
"author": "B Hu",
"doi-asserted-by": "crossref",
"first-page": "250",
"issue": "1",
"journal-title": "J Med Virol",
"key": "15800_CR3",
"unstructured": "Hu B, Huang S, Yin L. The cytokine storm and COVID-19. J Med Virol. 2021;93(1):250–6.",
"volume": "93",
"year": "2021"
},
{
"DOI": "10.1056/NEJMra2026131",
"author": "DC Fajgenbaum",
"doi-asserted-by": "crossref",
"first-page": "2255",
"issue": "23",
"journal-title": "N Engl J Med",
"key": "15800_CR4",
"unstructured": "Fajgenbaum DC, June CH. Cytokine Storm. N Engl J Med. 2020;383(23):2255–73.",
"volume": "383",
"year": "2020"
},
{
"DOI": "10.1016/j.cytogfr.2020.05.002",
"author": "SH Nile",
"doi-asserted-by": "crossref",
"first-page": "66",
"journal-title": "Cytokine Growth Factor Rev",
"key": "15800_CR5",
"unstructured": "Nile SH, Nile A, Qiu J, Li L, Jia X, Kai G. COVID-19: pathogenesis, cytokine storm and therapeutic potential of interferons. Cytokine Growth Factor Rev. 2020;53:66–70.",
"volume": "53",
"year": "2020"
},
{
"DOI": "10.1007/s00262-023-03524-1",
"author": "N Sobhani",
"doi-asserted-by": "crossref",
"first-page": "3427",
"issue": "11",
"journal-title": "Cancer Immunol Immunother",
"key": "15800_CR6",
"unstructured": "Sobhani N, Mondani G, Roviello G, Catalano M, Sirico M, D’Angelo A, Scaggiante B, Generali D. Cancer management during the COVID-19 world pandemic. Cancer Immunol Immunother. 2023;72(11):3427–44.",
"volume": "72",
"year": "2023"
},
{
"DOI": "10.1002/advs.202001435",
"author": "Z Ren",
"doi-asserted-by": "crossref",
"first-page": "e2001435",
"issue": "19",
"journal-title": "Adv Sci (Weinh)",
"key": "15800_CR7",
"unstructured": "Ren Z, Luo H, Yu Z, Song J, Liang L, Wang L, Wang H, Cui G, Liu Y, Wang J, et al. A Randomized, Open-Label, Controlled Clinical Trial of Azvudine Tablets in the Treatment of Mild and Common COVID-19, a Pilot Study. Adv Sci (Weinh). 2020;7(19):e2001435.",
"volume": "7",
"year": "2020"
},
{
"DOI": "10.1038/s41392-021-00835-6",
"author": "JL Zhang",
"doi-asserted-by": "crossref",
"first-page": "414",
"issue": "1",
"journal-title": "Signal Transduct Target Ther",
"key": "15800_CR8",
"unstructured": "Zhang JL, Li YH, Wang LL, Liu HQ, Lu SY, Liu Y, Li K, Liu B, Li SY, Shao FM, et al. Azvudine is a thymus-homing anti-SARS-CoV-2 drug effective in treating COVID-19 patients. Signal Transduct Target Ther. 2021;6(1):414.",
"volume": "6",
"year": "2021"
},
{
"DOI": "10.7150/jca.91530",
"author": "F Li",
"doi-asserted-by": "crossref",
"first-page": "2442",
"issue": "8",
"journal-title": "J Cancer",
"key": "15800_CR9",
"unstructured": "Li F, Zheng K, Qi X, Cui K, Yang J, Hao Z. A retrospective analysis of Azvudine in patients with COVID-19 and pre-existing cancer. J Cancer. 2024;15(8):2442–7.",
"volume": "15",
"year": "2024"
},
{
"DOI": "10.1002/jmv.28947",
"author": "H Yang",
"doi-asserted-by": "crossref",
"first-page": "e28947",
"issue": "7",
"journal-title": "J Med Virol",
"key": "15800_CR10",
"unstructured": "Yang H, Wang Z, Jiang C, Zhang Y, Zhang Y, Xu M, Zhang Y, Wang Y, Liu X, An Z, et al. Oral azvudine for mild-to-moderate COVID-19 in high risk, nonhospitalized adults: Results of a real-world study. J Med Virol. 2023;95(7):e28947.",
"volume": "95",
"year": "2023"
},
{
"DOI": "10.1002/advs.202404450",
"author": "R Sun",
"doi-asserted-by": "crossref",
"first-page": "e2404450",
"issue": "13",
"journal-title": "Adv Sci (Weinh)",
"key": "15800_CR11",
"unstructured": "Sun R, Wang H, Sun J, Yang M, Zhang S, Hu X, Yu B, Song Z, Han N, Luo H, et al. Effectiveness and Safety of Oral Azvudine for Elderly Hospitalized Patients With COVID-19: A Multicenter, Retrospective, Real-World Study. Adv Sci (Weinh). 2025;12(13):e2404450.",
"volume": "12",
"year": "2025"
},
{
"key": "15800_CR12",
"unstructured": "China,NHCotPsRo. COVID-19 diagnosis and treatment plan (trial version 9). 2022. https://www.gov.cn/zhengce/zhengceku/2022-03/15/5679257/files/49854a49c7004f4ea9e622f3f2c568d8.pdf."
},
{
"key": "15800_CR13",
"unstructured": "China,NHCotPsRo. COVID-19 diagnosis and treatment plan (trial version 10). 2023. https://www.gov.cn/zhengce/zhengceku/2023-01/06/5735343/files/5844ce04246b431dbd322d8ba10afb48.pdf."
},
{
"key": "15800_CR14",
"unstructured": "CommonTerminology Criteria for Adverse Events (CTCAE). Version 5.0. https://dctd.cancer.gov/research/ctep-trials/for-sites/adverse-events/ctcae-v5-5x7.pdf."
},
{
"DOI": "10.3389/fphar.2023.1228548",
"author": "KW Zhu",
"doi-asserted-by": "crossref",
"first-page": "1228548",
"journal-title": "Front Pharmacol",
"key": "15800_CR15",
"unstructured": "Zhu KW. Efficacy and safety evaluation of Azvudine in the prospective treatment of COVID-19 based on four phase III clinical trials. Front Pharmacol. 2023;14:1228548.",
"volume": "14",
"year": "2023"
},
{
"DOI": "10.1016/j.apsb.2023.07.007",
"author": "K Zong",
"doi-asserted-by": "crossref",
"first-page": "4655",
"issue": "11",
"journal-title": "Acta Pharm Sin B",
"key": "15800_CR16",
"unstructured": "Zong K, Zhou H, Li W, Jiang E, Liu Y, Li S. Azvudine reduces the in-hospital mortality of COVID-19 patients: a retrospective cohort study. Acta Pharm Sin B. 2023;13(11):4655–60.",
"volume": "13",
"year": "2023"
},
{
"DOI": "10.1016/j.ijantimicag.2020.105924",
"author": "CC Lai",
"doi-asserted-by": "crossref",
"first-page": "105924",
"issue": "3",
"journal-title": "Int J Antimicrob Agents",
"key": "15800_CR17",
"unstructured": "Lai CC, Shih TP, Ko WC, Tang HJ, Hsueh PR. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and coronavirus disease-2019 (COVID-19): The epidemic and the challenges. Int J Antimicrob Agents. 2020;55(3):105924.",
"volume": "55",
"year": "2020"
},
{
"DOI": "10.1016/j.lanepe.2023.100741",
"author": "B Zheng",
"doi-asserted-by": "crossref",
"first-page": "100741",
"journal-title": "Lancet Reg Health Eur",
"key": "15800_CR18",
"unstructured": "Zheng B, Tazare J, Nab L, Green AC, Curtis HJ, Mahalingasivam V, Herrett EL, Costello RE, Eggo RM, Speed V, et al. Comparative effectiveness of nirmatrelvir/ritonavir versus sotrovimab and molnupiravir for preventing severe COVID-19 outcomes in non-hospitalised high-risk patients during Omicron waves: observational cohort study using the OpenSAFELY platform. Lancet Reg Health Eur. 2023;34:100741.",
"volume": "34",
"year": "2023"
},
{
"DOI": "10.1056/NEJMoa2116044",
"author": "A Jayk Bernal",
"doi-asserted-by": "crossref",
"first-page": "509",
"issue": "6",
"journal-title": "N Engl J Med",
"key": "15800_CR19",
"unstructured": "Jayk Bernal A, Gomes da Silva MM, Musungaie DB, Kovalchuk E, Gonzalez A, Delos Reyes V, Martín-Quirós A, Caraco Y, Williams-Diaz A, Brown ML, et al. Molnupiravir for Oral Treatment of Covid-19 in Nonhospitalized Patients. N Engl J Med. 2022;386(6):509–20.",
"volume": "386",
"year": "2022"
},
{
"DOI": "10.1016/j.bcp.2011.01.001",
"author": "Q Wang",
"doi-asserted-by": "crossref",
"first-page": "848",
"issue": "7",
"journal-title": "Biochem Pharmacol",
"key": "15800_CR20",
"unstructured": "Wang Q, Liu X, Wang Q, Zhang Y, Jiang J, Guo X, Fan Q, Zheng L, Yu X, Wang N, et al. FNC, a novel nucleoside analogue inhibits cell proliferation and tumor growth in a variety of human cancer cells. Biochem Pharmacol. 2011;81(7):848–55.",
"volume": "81",
"year": "2011"
},
{
"DOI": "10.3389/fmed.2023.1143485",
"author": "RM da Silva",
"doi-asserted-by": "crossref",
"first-page": "1143485",
"journal-title": "Front Med (Lausanne)",
"key": "15800_CR21",
"unstructured": "da Silva RM, Gebe Abreu Cabral P, de Souza SB, Arruda RF, Cabral SPF, de Assis A, Martins YPM, Tavares CAA, Viana Junior AB, Chang J, et al. Serial viral load analysis by DDPCR to evaluate FNC efficacy and safety in the treatment of mild cases of COVID-19. Front Med (Lausanne). 2023;10:1143485.",
"volume": "10",
"year": "2023"
},
{
"DOI": "10.1016/j.jinf.2024.106355",
"author": "Z Ren",
"doi-asserted-by": "crossref",
"first-page": "106355",
"issue": "6",
"journal-title": "J Infect",
"key": "15800_CR22",
"unstructured": "Ren Z, Yang M, Su G, Qian G, Yuan Y, Yu J, Li S, Wang C, Lu M, Luo H, et al. Real-world effectiveness and safety of azvudine in hospitalized patients with SARS-CoV-2 infection: A multicenter, retrospective cohort study. J Infect. 2024;89(6):106355.",
"volume": "89",
"year": "2024"
}
],
"reference-count": 22,
"references-count": 22,
"relation": {},
"resource": {
"primary": {
"URL": "https://link.springer.com/10.1186/s12885-026-15800-1"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "Azvudine for the treatment of cancer patients with COVID-19: a multicenter, real-world, retrospective, cohort study",
"type": "journal-article",
"update-policy": "https://doi.org/10.1007/springer_crossmark_policy"
}
