Evaluation of the Effects of Remdesivir and Hydroxychloroquine on Viral Clearance in COVID-19
et al., Annals of Internal Medicine, doi:10.7326/M21-0653, NOR-Solidarity, NCT04321616, Jul 2021
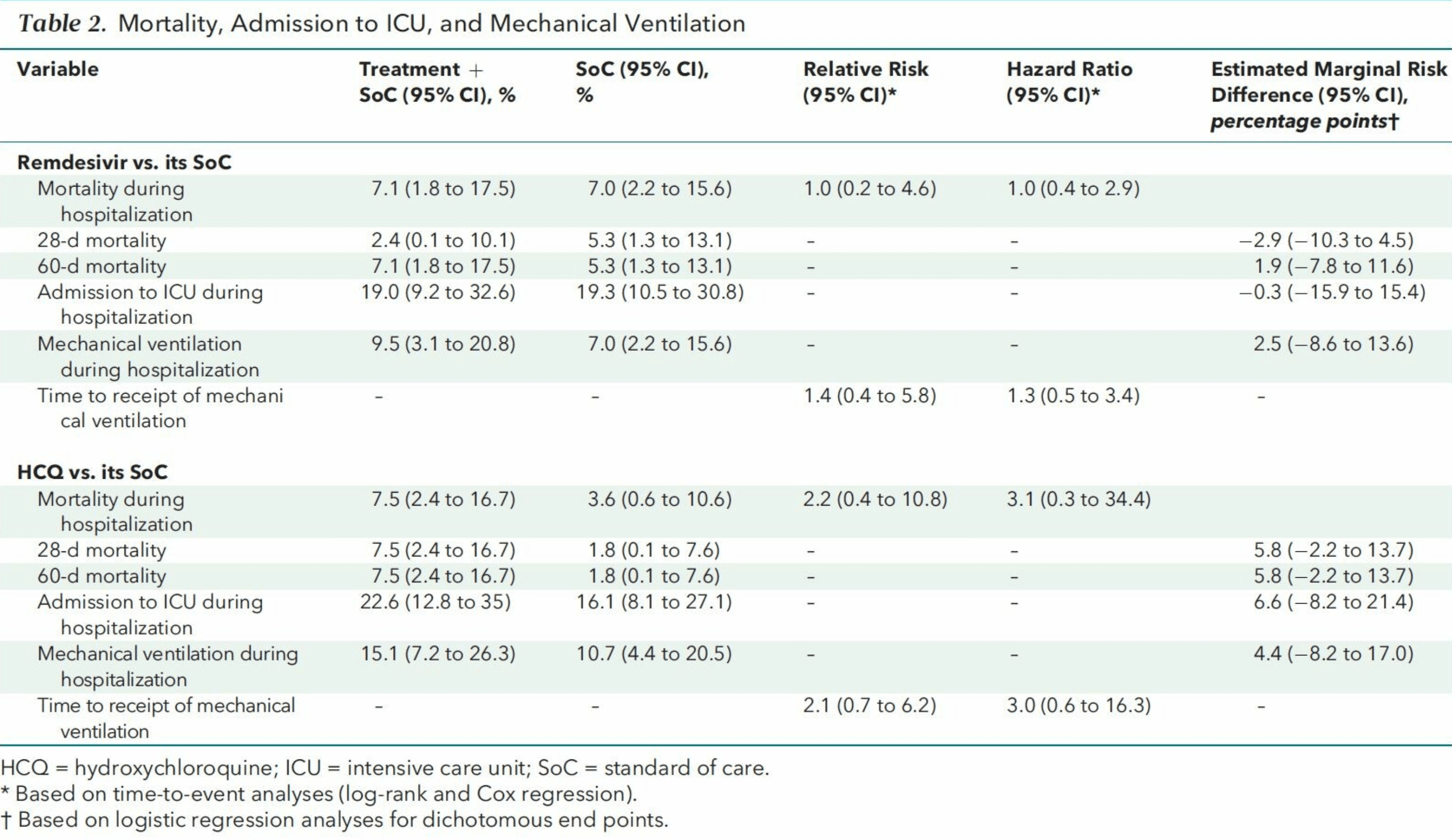
Small RCT in Norway showing no significant differences with remdesivir treatment. Add-on trial to WHO Solidarity. Longer term recovery results are from Patrick-Brown et al.
Gérard, Zhou, Wu, Kamo, Choi, Kim show increased risk of acute kidney injury, Leo, Briciu, Muntean, Petrov, Arch show increased risk of liver injury, Negru, Cheng, Mohammed, Kwok, Zhu show increased risk of cardiac disorders, and Kwok, Merches, Akinci, Tang, Bagheri show increased risk of mitochondrial toxicity with remdesivir.
Study covers remdesivir and HCQ.
|
risk of death, no change, RR 1.00, p = 1.00, treatment 3 of 42 (7.1%), control 4 of 57 (7.0%), adjusted per study.
|
|
risk of death, 35.7% higher, RR 1.36, p = 0.70, treatment 3 of 42 (7.1%), control 3 of 57 (5.3%), day 60.
|
|
risk of death, 54.8% lower, RR 0.45, p = 0.63, treatment 1 of 42 (2.4%), control 3 of 57 (5.3%), NNT 35, day 28.
|
|
risk of no recovery, 47.4% higher, RR 1.47, p = 0.01, treatment mean 16.8 (±11.4) n=42, control mean 11.4 (±10.4) n=76, relative CAT total score at 3 months.
|
|
risk of no recovery, 42.9% higher, RR 1.43, p = 0.009, treatment mean 3.0 (±1.7) n=42, control mean 2.1 (±1.8) n=76, relative CAT dyspnea score at 3 months.
|
|
risk of no recovery, 23.8% higher, RR 1.24, p = 0.10, treatment mean 2.6 (±1.5) n=42, control mean 2.1 (±1.6) n=76, relative CAT fatigue score at 3 months.
|
|
risk of no recovery, 50.0% higher, RR 1.50, p = 0.04, treatment mean 1.8 (±1.6) n=42, control mean 1.2 (±1.5) n=76, relative CAT cough score at 3 months.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
1.
Patrick-Brown et al., The effects of remdesivir on long-term symptoms in patients hospitalised for COVID-19: a pre-specified exploratory analysis, Communications Medicine, doi:10.1038/s43856-024-00650-4.
2.
Gérard et al., Remdesivir and Acute Renal Failure: A Potential Safety Signal From Disproportionality Analysis of the WHO Safety Database, Clinical Pharmacology & Therapeutics, doi:10.1002/cpt.2145.
3.
Zhou et al., Acute Kidney Injury and Drugs Prescribed for COVID-19 in Diabetes Patients: A Real-World Disproportionality Analysis, Frontiers in Pharmacology, doi:10.3389/fphar.2022.833679.
4.
Wu et al., Acute Kidney Injury Associated With Remdesivir: A Comprehensive Pharmacovigilance Analysis of COVID-19 Reports in FAERS, Frontiers in Pharmacology, doi:10.3389/fphar.2022.692828.
5.
Kamo et al., Association of Antiviral Drugs for the Treatment of COVID-19 With Acute Renal Failure, In Vivo, doi:10.21873/invivo.13637.
6.
Choi et al., Comparative effectiveness of combination therapy with nirmatrelvir–ritonavir and remdesivir versus monotherapy with remdesivir or nirmatrelvir–ritonavir in patients hospitalised with COVID-19: a target trial emulation study, The Lancet Infectious Diseases, doi:10.1016/S1473-3099(24)00353-0.
7.
Kim et al., Investigating the Safety Profile of Fast‐Track COVID‐19 Drugs Using the FDA Adverse Event Reporting System Database: A Comparative Observational Study, Pharmacoepidemiology and Drug Safety, doi:10.1002/pds.70043.
8.
Leo et al., Hepatocellular liver injury in hospitalized patients affected by COVID-19: Presence of different risk factors at different time points, Digestive and Liver Disease, doi:10.1016/j.dld.2021.12.014.
9.
Briciu et al., Evolving Clinical Manifestations and Outcomes in COVID-19 Patients: A Comparative Analysis of SARS-CoV-2 Variant Waves in a Romanian Hospital Setting, Pathogens, doi:10.3390/pathogens12121453.
10.
Muntean et al., Effects of COVID-19 on the Liver and Mortality in Patients with SARS-CoV-2 Pneumonia Caused by Delta and Non-Delta Variants: An Analysis in a Single Centre, Pharmaceuticals, doi:10.3390/ph17010003.
11.
Petrov et al., The Effect of Potentially Hepatotoxic Medicinal Products on Alanine Transaminase Levels in COVID-19 Patients: A Case–Control Study, Safety and Risk of Pharmacotherapy, doi:10.30895/2312-7821-2025-458.
12.
Arch et al., Evaluation of the effectiveness of remdesivir in treating severe COVID-19 using data from the ISARIC WHO Clinical Characterisation Protocol UK: a prospective, national cohort study, medRxiv, doi:10.1101/2021.06.18.21259072.
13.
Negru et al., Comparative Pharmacovigilance Analysis of Approved and Repurposed Antivirals for COVID-19: Insights from EudraVigilance Data, Biomedicines, doi:10.3390/biomedicines13061387.
14.
Cheng et al., Cardiovascular Safety of COVID-19 Treatments: A Disproportionality Analysis of Adverse Event Reports from the WHO VigiBase, Infectious Diseases and Therapy, doi:10.1007/s40121-025-01225-z.
15.
Mohammed et al., Bradycardia associated with remdesivir treatment in coronavirus disease 2019 patients: A propensity score-matched analysis, Medicine, doi:10.1097/MD.0000000000044501.
16.
Kwok et al., Remdesivir induces persistent mitochondrial and structural damage in human induced pluripotent stem cell-derived cardiomyocytes, Cardiovascular Research, doi:10.1093/cvr/cvab311.
17.
Zhu et al., Cardiovascular Risks of COVID-19 Therapeutics: Integrated Analysis of FAERS, Electronic Health Records, and Transcriptomics, Pharmaceuticals, doi:10.3390/ph19040574.
18.
Merches et al., The potential of remdesivir to affect function, metabolism and proliferation of cardiac and kidney cells in vitro, Archives of Toxicology, doi:10.1007/s00204-022-03306-1.
19.
Akinci et al., Elucidation of remdesivir cytotoxicity pathways through genome-wide CRISPR-Cas9 screening and transcriptomics, bioRxiv, doi:10.1101/2020.08.27.270819.
Barrat-Due et al., 13 Jul 2021, Double Blind Randomized Controlled Trial, Norway, peer-reviewed, 43 authors, average treatment delay 8.0 days, trial NCT04321616 (history) (NOR-Solidarity).
Evaluation of the Effects of Remdesivir and Hydroxychloroquine on Viral Clearance in COVID-19
Annals of Internal Medicine, doi:10.7326/m21-0653
Background: New treatment modalities are urgently needed for patients with COVID-19. The World Health Organization (WHO) Solidarity trial showed no effect of remdesivir or hydroxychloroquine (HCQ) on mortality, but the antiviral effects of these drugs are not known. Objective: To evaluate the effects of remdesivir and HCQ on all-cause, in-hospital mortality; the degree of respiratory failure and inflammation; and viral clearance in the oropharynx. Design: NOR-Solidarity is an independent, add-on, randomized controlled trial to the WHO Solidarity trial that included biobanking and 3 months of clinical follow-up (ClinicalTrials. gov: NCT04321616) Setting: 23 hospitals in Norway. Patients: Eligible patients were adults hospitalized with confirmed SARS-CoV-2 infection. Intervention: Between 28 March and 4 October 2020, a total of 185 patients were randomly assigned and 181 were included in the full analysis set. Patients received remdesivir (n = 42), HCQ (n = 52), or standard of care (SoC) (n = 87). Measurements: In addition to the primary end point of WHO Solidarity, study-specific outcomes were viral clearance in oropharyngeal specimens, the degree of respiratory failure, and inflammatory variables. Results: No significant differences were seen between treatment groups in mortality during hospitalization. There was a marked decrease in SARS-CoV-2 load in the oropharynx during the first week overall, with similar decreases and 10-day viral loads among the remdesivir, HCQ, and SoC groups. Remdesivir and HCQ did not affect the degree of respiratory failure or inflammatory variables in plasma or serum. The lack of antiviral effect was not associated with symptom duration, level of viral load, degree of inflammation, or presence of antibodies against SARS-CoV-2 at hospital admittance.
Limitation: The trial had no placebo group.
Conclusion: Neither remdesivir nor HCQ affected viral clearance in hospitalized patients with COVID-19.
EDTA plasma samples were taken at randomization, between days 3 and 5, between days 7 and 9, and weekly thereafter. CRP, ferritin, procalcitonin, LDH, lymphocytes, and neutrophils were analyzed by the routine laboratory at the hospitals included in the study. Treatment effects are given as estimated differences in daily decrease rates (slopes) of the variables between remdesivir and its SoC during the first week, and in differences in point estimates at day 10. Results are presented as estimated treatment differences with 95% CIs. CRP = C-reactive protein; LDH = lactate dehydrogenase; SoC = standard of care. * To convert to SI units (mkat/L), multiply by 0.0167. EDTA plasma samples were taken at randomization, between days 3 and 5, between days 7 and 9, and weekly thereafter. CRP, ferritin, procalcitonin, LDH, lymphocytes, and neutrophils were analyzed by the routine laboratory at the hospitals included in the study. Treatment effects are given as estimated differences in daily decrease rates (slopes) of the variables between HCQ and its SoC during the first week, and in differences in point estimates at day 10. Results are presented as estimated treatment differences with 95% CIs. CRP = C-reactive protein; HCQ = hydroxychloroquine; LDH = lactate dehydrogenase; SoC = standard of care. * To convert to SI units (mkat/L), multiply by 0.0167. Subgroup analyses evaluating the effect on viral clearance of remdesivir compared with SoC in patients with short (<7 d) and long (≥7 d)..
References
Amanat, Stadlbauer, Strohmeier, A serological assay to detect SARS-CoV-2 seroconversion in humans, Nat Med, doi:10.1038/s41591-020-0913-5
Beigel, Tomashek, Dodd, ACTT-1 Study Group Members. Remdesivir for the treatment of Covid-19 -final report, N Engl J Med, doi:10.1056/NEJMoa2007764
Bergen, Dr ; Grålum, Johannessen, Mielnik, Olsen et al., Critical revision of the article for important intellectual content: A
Bimonte, Crispo, Amore, Potential antiviral drugs for SARS-cov-2 treatment: preclinical findings and ongoing clinical research, Vivo, doi:10.21873/invivo.11949
Dolin, Hirsch, Remdesivir-an important first step
Dr, Berg, None
Dr, Blomberg, None, Jonas Lies veg
Dr, Haugli, None, Postboks
Dr, Johannessen, None
Dr, Mathiessen, None
Dr, None
Dr, None, Finbråten: Lovisenberg Diaconal Hospital, Lovisenberggata
Dr, Tholin: Molde Hospital, Parkvegen 84 6412
Dreier, Störmer, Kleesiek, Use of bacteriophage MS2 as an internal control in viral reverse transcription-PCR assays, J Clin Microbiol, doi:10.2807/1560-7917.ES.2020.25.3.2000045
Drs, Barratt-Due, Olsen, Nezvalova-Henriksen, Lund-Johansen et al., Current Author Addresses
Elverum, Dr, Skei, Trøndelag Hospital Trust/NTNU, Postboks
Førde, Hospital, Førde, Norway, None
Gordon, Winer, Hoogendijk, Immunosuppressant and immunomodulatory treatment for dermatomyositis and polymyositis, Cochrane Database Syst Rev, doi:10.1002/14651858.CD003643.pub4
Haugesund, Dr, Thoresen, None, Arnhold Dybjords vei
Holter, Pischke, De Boer, Systemic complement activation is associated with respiratory failure in COVID-19 hospitalized patients, Proc Natl Acad Sci U S A, doi:10.1073/pnas.2010540117
Horby, Mafham, Linsell, Effect of hydroxychloroquine in hospitalized patients with Covid-19, N Engl J Med, doi:10.1056/NEJMoa2022926
Hospital, None
Hospital, None
Hospital, Viken, Trust, None
Hønefoss, Dr, Hoff, None
Investigators, Effect of remdesivir vs standard care on clinical status at 11 days in patients with moderate COVID-19: a randomized clinical trial, JAMA, doi:10.1001/jama.2020.16349
Kristiansand, Dr, Eiken, None, Innlandet Hospital Trust Anders Sandvigs Gate
Laake, Buanes, Småstuen, Characteristics, management and survival of ICU patients with coronavirus disease-19 in Norway, March-June 2020. A prospective observational study, Acta Anaesthesiol Scand, doi:10.1111/aas.13785
Levanger, Dr, Hannula, None
Lyngbakken, Berdal, Eskesen, A pragmatic randomized controlled trial reports lack of efficacy of hydroxychloroquine on coronavirus disease 2019 viral kinetics, Nat Commun, doi:10.1038/s41467-020-19056-6
Maciorowski, Idrissi, Gupta, A review of the preclinical and clinical efficacy of remdesivir, hydroxychloroquine, and lopinavir-ritonavir treatments against COVID-19, SLAS Discov, doi:10.1177/247255522095838516
Nydalen, None
Oslo, Dr, Mielnik, Helse Førde, Svanehaugvegen 2
Oslo, Drs, Tveita, None, Postboks
Pan, Peto, Henao-Restrepo, WHO Solidarity Trial Consortium. Repurposed antiviral drugs for Covid-19-interim WHO Solidarity trial results, N Engl J Med, doi:10.1056/NEJMoa2023184
Rochwerg, Siemieniuk, Agoritsas, A living WHO guideline on drugs for covid-19, BMJ, doi:10.1136/bmj.m3379
Skien, Dr, Kittang, Haraldsplass Deaconess Hospital
Spinner, Gottlieb, Criner, None
Torgarden, None
Tønsberg, Dr, Dahl, None
Wang, Cao, Zhang, Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV)
Wang, Zhang, Du, Remdesivir in adults with severe COVID-19: a randomised, double-blind, placebo-controlled, multicentre trial, Lancet, doi:10.1016/S0140-6736(20)31022-9
DOI record:
{
"DOI": "10.7326/m21-0653",
"ISSN": [
"0003-4819",
"1539-3704"
],
"URL": "http://dx.doi.org/10.7326/M21-0653",
"alternative-id": [
"10.7326/M21-0653"
],
"author": [
{
"ORCID": "http://orcid.org/0000-0001-5408-7162",
"affiliation": [
{
"name": "Oslo University Hospital, Oslo, Norway (A.B., I.C.O., T.K., T.T., C.F.)"
}
],
"authenticated-orcid": false,
"family": "Barratt-Due",
"given": "Andreas",
"sequence": "first"
},
{
"ORCID": "http://orcid.org/0000-0001-6889-5873",
"affiliation": [
{
"name": "Oslo University Hospital, Oslo, Norway (A.B., I.C.O., T.K., T.T., C.F.)"
}
],
"authenticated-orcid": false,
"family": "Olsen",
"given": "Inge Christoffer",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Oslo University Hospital and Hospital Pharmacies, South-Eastern Norway Enterprise, Oslo, Norway (K.N.)"
}
],
"family": "Nezvalova-Henriksen",
"given": "Katerina",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0003-0868-9861",
"affiliation": [
{
"name": "Oslo University Hospital, Oslo, Norway (A.B., I.C.O., T.K., T.T., C.F.)"
}
],
"authenticated-orcid": false,
"family": "Kåsine",
"given": "Trine",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-2445-1258",
"affiliation": [
{
"name": "Oslo University Hospital and ImmunoLingo Convergence Centre, University of Oslo, Oslo, Norway (F.L.)"
}
],
"authenticated-orcid": false,
"family": "Lund-Johansen",
"given": "Fridtjof",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Institute of Clinical Medicine and Research Institute of Internal Medicine, Oslo University Hospital, and Lovisenberg Diaconal Hospital, Oslo, Norway (H.H.)"
}
],
"family": "Hoel",
"given": "Hedda",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-7916-0717",
"affiliation": [
{
"name": "Institute of Clinical Medicine, Oslo University Hospital, Oslo, Norway (A.R.H., S.D., J.T.A., K.T., A.S., F.M., A.M.D., M.T.)"
}
],
"authenticated-orcid": false,
"family": "Holten",
"given": "Aleksander Rygh",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-0668-9262",
"affiliation": [
{
"name": "Bærum Hospital, Vestre Viken Hospital Trust, Drammen, Norway (A.T.)"
}
],
"authenticated-orcid": false,
"family": "Tveita",
"given": "Anders",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-9696-2081",
"affiliation": [
{
"name": "Diakonhjemmet Hospital, Oslo, Norway (A.M.)"
}
],
"authenticated-orcid": false,
"family": "Mathiessen",
"given": "Alexander",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Sørlandet Hospital SSK, Kristiansand, Norway (M.H.)"
}
],
"family": "Haugli",
"given": "Mette",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Innlandet Hospital Trust, Lillehammer, Norway (R.E.)"
}
],
"family": "Eiken",
"given": "Ragnhild",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-1319-6511",
"affiliation": [
{
"name": "University Hospital of North Norway, Tromsø, Norway (A.B.K.)"
}
],
"authenticated-orcid": false,
"family": "Kildal",
"given": "Anders Benjamin",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-6937-3580",
"affiliation": [
{
"name": "Stavanger University Hospital, Stavanger, Norway (Å.B.)"
}
],
"authenticated-orcid": false,
"family": "Berg",
"given": "Åse",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-5966-7166",
"affiliation": [
{
"name": "Institute of Clinical Medicine, Oslo University Hospital, Oslo, and Vestfold Hospital Trust, Tønsberg, Norway (A.J.)"
}
],
"authenticated-orcid": false,
"family": "Johannessen",
"given": "Asgeir",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-1669-1032",
"affiliation": [
{
"name": "Drammen Hospital, Vestre Viken Hospital Trust, Drammen, and University of Bergen, Bergen, Norway (L.H.)"
}
],
"authenticated-orcid": false,
"family": "Heggelund",
"given": "Lars",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-7818-9411",
"affiliation": [
{
"name": "Research Institute of Internal Medicine, Oslo University Hospital, Oslo, Norway (T.B.D., K.H.S.)"
}
],
"authenticated-orcid": false,
"family": "Dahl",
"given": "Tuva Børresdatter",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Research Institute of Internal Medicine, Oslo University Hospital, Oslo, Norway (T.B.D., K.H.S.)"
}
],
"family": "Skåra",
"given": "Karoline Hansen",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-3894-5371",
"affiliation": [
{
"name": "Førde Central Hospital, Førde, Norway (P.M.)"
}
],
"authenticated-orcid": false,
"family": "Mielnik",
"given": "Pawel",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Haugesund Hospital, Haugesund, Norway (L.A.K.)"
}
],
"family": "Le",
"given": "Lan Ai Kieu",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-4149-0451",
"affiliation": [
{
"name": "Ringerike Hospital, Vestre Viken Hospital Trust, Ringerike, Norway (L.T.)"
}
],
"authenticated-orcid": false,
"family": "Thoresen",
"given": "Lars",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-5224-0338",
"affiliation": [
{
"name": "Kongsberg Hospital, Vestre Viken Hospital Trust, Drammen, Norway (G.E.)"
}
],
"authenticated-orcid": false,
"family": "Ernst",
"given": "Gernot",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-7861-2125",
"affiliation": [
{
"name": "Ålesund Hospital, Møre and Romsdal Hospital Trust, Ålesund, Norway (D.A.L.)"
}
],
"authenticated-orcid": false,
"family": "Hoff",
"given": "Dag Arne Lihaug",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-0707-8236",
"affiliation": [
{
"name": "Telemark Hospital Trust, Skien, Norway (H.S.)"
}
],
"authenticated-orcid": false,
"family": "Skudal",
"given": "Hilde",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Haraldsplass Deaconess Hospital, Bergen, Norway (B.R.K.)"
}
],
"family": "Kittang",
"given": "Bård Reiakvam",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Sorlandet Hospital, Arendal, Norway (R.B.O.)"
}
],
"family": "Olsen",
"given": "Roy Bjørkholt",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-6768-541X",
"affiliation": [
{
"name": "Molde Hospital, Møre and Romsdal Hospital Trust, Molde, Norway (B.T.)"
}
],
"authenticated-orcid": false,
"family": "Tholin",
"given": "Birgitte",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-7103-8426",
"affiliation": [
{
"name": "Innlandet Hospital Trust, Elverum, Norway (C.M.Y.)"
}
],
"authenticated-orcid": false,
"family": "Ystrøm",
"given": "Carl Magnus",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Levanger Hospital, Nord-Trøndelag Hospital Trust, Levanger, Norway (N.V.S.)"
}
],
"family": "Skei",
"given": "Nina Vibeche",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Oslo University Hospital, Oslo, Norway (A.B., I.C.O., T.K., T.T., C.F.)"
}
],
"family": "Tran",
"given": "Trung",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-5047-4982",
"affiliation": [
{
"name": "Institute of Clinical Medicine, Oslo University Hospital, Oslo, Norway (A.R.H., S.D., J.T.A., K.T., A.S., F.M., A.M.D., M.T.)"
}
],
"authenticated-orcid": false,
"family": "Dudman",
"given": "Susanne",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0003-1710-1628",
"affiliation": [
{
"name": "Institute of Clinical Medicine, Oslo University Hospital, Oslo, Norway (A.R.H., S.D., J.T.A., K.T., A.S., F.M., A.M.D., M.T.)"
}
],
"authenticated-orcid": false,
"family": "Andersen",
"given": "Jan Terje",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0003-4073-7843",
"affiliation": [
{
"name": "Trondheim University Hospital, Trondheim, Norway (R.H.)"
}
],
"authenticated-orcid": false,
"family": "Hannula",
"given": "Raisa",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Institute of Clinical Medicine, Oslo University Hospital, Oslo, and Akershus University Hospital, Lørenskog, Norway (O.D.)"
}
],
"family": "Dalgard",
"given": "Olav",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Unger-Vetlesen Institute, Lovisenberg Diaconal Hospital, Oslo, Norway (A.F.)"
}
],
"family": "Finbråten",
"given": "Ane-Kristine",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Institute of Clinical Medicine, Oslo University Hospital, Oslo, Norway (A.R.H., S.D., J.T.A., K.T., A.S., F.M., A.M.D., M.T.)"
}
],
"family": "Tonby",
"given": "Kristian",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-5647-4297",
"affiliation": [
{
"name": "Haukeland University Hospital and University of Bergen, Bergen, Norway (B.B.)"
}
],
"authenticated-orcid": false,
"family": "Blomberg",
"given": "Bjorn",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0003-1864-9740",
"affiliation": [
{
"name": "Østfold Hospital Kalnes, Grålum, Norway (S.A.)"
}
],
"authenticated-orcid": false,
"family": "Aballi",
"given": "Saad",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Oslo University Hospital, Oslo, Norway (A.B., I.C.O., T.K., T.T., C.F.)"
}
],
"family": "Fladeby",
"given": "Cathrine",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-5322-728X",
"affiliation": [
{
"name": "Institute of Clinical Medicine, Oslo University Hospital, Oslo, Norway (A.R.H., S.D., J.T.A., K.T., A.S., F.M., A.M.D., M.T.)"
}
],
"authenticated-orcid": false,
"family": "Steffensen",
"given": "Anne",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-8934-2784",
"affiliation": [
{
"name": "Institute of Clinical Medicine, Oslo University Hospital, Oslo, Norway (A.R.H., S.D., J.T.A., K.T., A.S., F.M., A.M.D., M.T.)"
}
],
"authenticated-orcid": false,
"family": "Müller",
"given": "Fredrik",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0003-4009-4032",
"affiliation": [
{
"name": "Institute of Clinical Medicine, Oslo University Hospital, Oslo, Norway (A.R.H., S.D., J.T.A., K.T., A.S., F.M., A.M.D., M.T.)"
}
],
"authenticated-orcid": false,
"family": "Dyrhol-Riise",
"given": "Anne Ma",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-7775-3404",
"affiliation": [
{
"name": "Institute of Clinical Medicine, Oslo University Hospital, Oslo, Norway (A.R.H., S.D., J.T.A., K.T., A.S., F.M., A.M.D., M.T.)"
}
],
"authenticated-orcid": false,
"family": "Trøseid",
"given": "Marius",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0003-0919-836X",
"affiliation": [
{
"name": "Institute of Clinical Medicine and Research Institute of Internal Medicine, Oslo University Hospital, Oslo, Norway (P.A.)."
}
],
"authenticated-orcid": false,
"family": "Aukrust",
"given": "Pål",
"sequence": "additional"
},
{
"affiliation": [],
"name": "NOR-Solidarity trial",
"sequence": "additional"
}
],
"container-title": "Annals of Internal Medicine",
"container-title-short": "Ann Intern Med",
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2021,
7,
12
]
],
"date-time": "2021-07-12T21:00:52Z",
"timestamp": 1626123652000
},
"deposited": {
"date-parts": [
[
2021,
9,
20
]
],
"date-time": "2021-09-20T21:06:00Z",
"timestamp": 1632171960000
},
"indexed": {
"date-parts": [
[
2024,
3,
28
]
],
"date-time": "2024-03-28T05:58:00Z",
"timestamp": 1711605480644
},
"is-referenced-by-count": 78,
"issue": "9",
"issued": {
"date-parts": [
[
2021,
9
]
]
},
"journal-issue": {
"issue": "9",
"published-print": {
"date-parts": [
[
2021,
9
]
]
}
},
"language": "en",
"member": "4285",
"original-title": [],
"page": "1261-1269",
"prefix": "10.7326",
"published": {
"date-parts": [
[
2021,
9
]
]
},
"published-print": {
"date-parts": [
[
2021,
9
]
]
},
"publisher": "American College of Physicians",
"reference": [
{
"DOI": "10.1056/NEJMoa2023184",
"doi-asserted-by": "publisher",
"key": "r1-M210653"
},
{
"DOI": "10.1056/NEJMoa2022926",
"doi-asserted-by": "publisher",
"key": "r2-M210653"
},
{
"DOI": "10.1056/NEJMoa2007764",
"doi-asserted-by": "publisher",
"key": "r3-M210653"
},
{
"DOI": "10.1002/14651858.CD003643.pub4",
"doi-asserted-by": "publisher",
"key": "r4-M210653"
},
{
"DOI": "10.1038/s41422-020-0282-0",
"doi-asserted-by": "publisher",
"key": "r5-M210653"
},
{
"DOI": "10.1136/bmj.m3379",
"doi-asserted-by": "publisher",
"key": "r6-M210653"
},
{
"DOI": "10.1111/aas.13785",
"doi-asserted-by": "publisher",
"key": "r10-M210653"
},
{
"DOI": "10.21873/invivo.11949",
"doi-asserted-by": "publisher",
"key": "r11-M210653"
},
{
"DOI": "10.1016/S0140-6736(20)31022-9",
"doi-asserted-by": "publisher",
"key": "r12-M210653"
},
{
"DOI": "10.1038/s41467-020-19056-6",
"doi-asserted-by": "publisher",
"key": "r13-M210653"
},
{
"DOI": "10.1056/NEJMe2018715",
"doi-asserted-by": "publisher",
"key": "r14-M210653"
},
{
"DOI": "10.1177/2472555220958385",
"doi-asserted-by": "publisher",
"key": "r15-M210653"
},
{
"DOI": "10.1371/journal.pmed.1003252",
"doi-asserted-by": "publisher",
"key": "r16-M210653"
},
{
"DOI": "10.1001/jama.2020.16349",
"doi-asserted-by": "publisher",
"key": "r17-M210653"
},
{
"DOI": "10.2807/1560-7917.ES.2020.25.3.2000045",
"doi-asserted-by": "publisher",
"key": "r18-M210653"
},
{
"DOI": "10.1128/JCM.43.9.4551-4557.2005",
"doi-asserted-by": "publisher",
"key": "r19-M210653"
},
{
"DOI": "10.1073/pnas.2010540117",
"doi-asserted-by": "publisher",
"key": "r20-M210653"
},
{
"DOI": "10.1038/s41591-020-0913-5",
"doi-asserted-by": "publisher",
"key": "r21-M210653"
}
],
"reference-count": 18,
"references-count": 18,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.acpjournals.org/doi/10.7326/M21-0653"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"General Medicine",
"Internal Medicine"
],
"subtitle": [
"A Randomized Trial"
],
"title": "Evaluation of the Effects of Remdesivir and Hydroxychloroquine on Viral Clearance in COVID-19",
"type": "journal-article",
"volume": "174"
}
barratdue