The Relationship Between Plasma Favipiravir Concentrations and Clinical Outcomes in COVID-19
et al., ERJ Open Research, doi:10.1183/23120541.01560-2025, Mar 2026
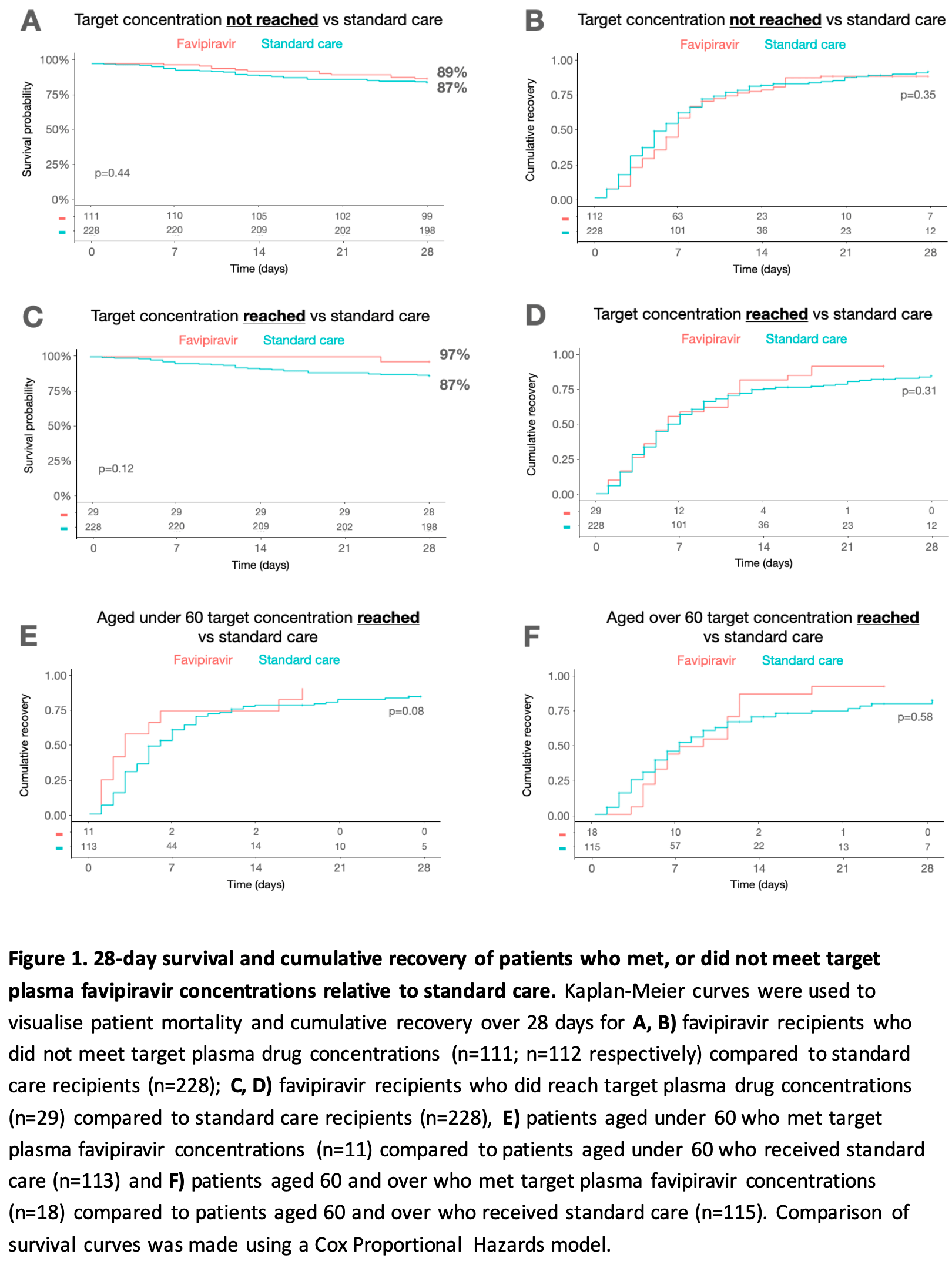
Post hoc pharmacokinetic analysis of 140 hospitalized COVID-19 patients from the PIONEER RCT, showing no statistically significant improvement in clinical outcomes among favipiravir-treated patients who achieved target plasma concentrations compared to those who did not, or compared to standard care recipients. Only 21% of patients reached the target plasma concentration. Time to clinical improvement was 7.7 vs 9.1 days for target achievers vs non-achievers (p=0.26), and 28-day survival was 97% vs 87% for target achievers vs standard care recipients (p=0.12), neither reaching statistical significance.
Potential risks of favipiravir include kidney injury1-3, liver injury2-5, cardiovascular events5,6, pulmonary toxicity6,7, and mutagenicity, carcinogenicity, teratogenicity, embryotoxicity, and the creation of dangerous variants8-14.
1.
Abdulaziz et al., Clinical Features and Prognosis of Acute Kidney Injury in Hospital-Admitted Patients with COVID-19 in Egypt: A Single-Center Experience, Mansoura Medical Journal, doi:10.58775/2735-3990.1433.
2.
Ülger et al., Experimental evaluation of favipiravir (T-705)-induced liver and kidney toxicity in rats, Food and Chemical Toxicology, doi:10.1016/j.fct.2025.115472.
3.
El-Fetouh et al., Experimental Studies on Some Drugs Used in Covid-19 Treatment (Favipiravir and Dexamethasone) in Albino Rats, Journal of Advanced Veterinary Research, 13:10, www.advetresearch.com/index.php/AVR/article/view/1635.
4.
Almutairi et al., Liver Injury in Favipiravir-Treated COVID-19 Patients: Retrospective Single-Center Cohort Study, Tropical Medicine and Infectious Disease, doi:10.3390/tropicalmed8020129.
5.
Siby et al., Temporal Trends in Serious Adverse Events Associated with Oral Antivirals During the COVID-19 Pandemic: Insights from the FAERS Database (2020–2023), Open Forum Infectious Diseases, doi:10.1093/ofid/ofaf695.1825.
6.
Ozhan et al., Evaluation of the cardiopulmonary effects of repurposed COVID-19 therapeutics in healthy rats, Scientific Reports, doi:10.1038/s41598-025-31048-4.
7.
Ülger (B) et al., Evaluation of the effects of favipiravir (T-705) on the lung tissue of healty rats: An experimental study, Food and Chemical Toxicology, doi:10.1016/j.fct.2025.115235.
8.
Zhirnov et al., Favipiravir: the hidden threat of mutagenic action, Journal of microbiology, epidemiology and immunobiology, doi:10.36233/0372-9311-114.
9.
Waters et al., Human genetic risk of treatment with antiviral nucleoside analog drugs that induce lethal mutagenesis: the special case of molnupiravir, Environmental and Molecular Mutagenesis, doi:10.1002/em.22471.
10.
Hadj Hassine et al., Lethal Mutagenesis of RNA Viruses and Approved Drugs with Antiviral Mutagenic Activity, Viruses, doi:10.3390/v14040841.
11.
Shum, C., An investigational study into the drug-associated mutational signature in SARS-CoV-2 viruses, The University of Hong Kong, PhD Thesis, hub.hku.hk/handle/10722/344396.
12.
Shiraki et al., Convenient screening of the reproductive toxicity of favipiravir and antiviral drugs in Caenorhabditis elegans, Heliyon, doi:10.1016/j.heliyon.2024.e35331.
Wawman et al., 26 Mar 2026, United Kingdom, peer-reviewed, mean age 59.5, 19 authors, study period May 2020 - May 2021.
Contact: p.bhavsar@imperial.ac.uk.
Abstract: ERJ
research
THE BEST IN OPEN ACCESS BASIC,
TRANSLATIONAL & CLINICAL
RESPIRATORY RESEARCH
Early View
Original Research Article
The Relationship Between Plasma Favipiravir Concentrations and Clinical Outcomes in COVID19
Rebecca E. Wawman, Pallav L. Shah, Marta Boffito, James Tonkin, Francesca Conway, Anand Tana, Breno R. Santos, Beatriz Grinsztejn, Brenda Crabtree Ramírez, Henry Pertinez, Andrew Owen, Paul Curley, Usman Arshad, Helen Cox, Mark R. Johnson, Anton Pozniak, Michael Pelly, Christopher M. Orton, Pankaj K. Bhavsar
Please cite this article as: Wawman RE, Shah PL, Boffito M, et al . The Relationship Between Plasma Favipiravir Concentrations and Clinical Outcomes in COVID-19. ERJ Open Res 2026; in press (https://doi.org/10.1183/23120541.01560-2025).
This manuscript has recently been accepted for publication in the ERJ Open Research . It is published here in its accepted form prior to copyediting and typesetting by our production team. After these production processes are complete and the authors have approved the resulting proofs, the article will move to the latest issue of the ERJOR online.
Copyright ©The authors 2026. This version is distributed under the terms of the Creative Commons Attribution Licence 4.0.
open
The Relationship Between Plasma Favipiravir Concentrations and Clinical Outcomes in COVID19
Rebecca E Wawman 1 , Pallav L Shah 1, 2, 3 , Marta Boffito 1, 3 , James Tonkin 1, 2, 3 , Francesca Conway 1, 2, 3 , Anand Tana 1, 2, 3 , Breno R Santos 4 , Beatriz Grinsztejn 5 , Brenda Crabtree Ramírez 6 , Henry Pertinez 8 , Andrew Owen 8 , Paul Curley 8 , Usman Arshad 8 , Helen Cox 8 , Mark R Johnson 3 , Anton Pozniak 3, 7, , Michael Pelly 3 , Christopher M Orton 1, 2, 3 , Pankaj K Bhavsar 1,2*
1 National Heart and Lung Institute, Imperial College London, London, United Kingdom
2 Department of Respiratory Medicine, Royal Brompton Hospital, London, United Kingdom
3 Chelsea and Westminster NHS Foundation Trust, London, United Kingdom
4 Departamento de Infectología, Hospital Nossa Senhora da Conceição-Grupo Hospitalar Conceição, Porto Alegre, Brazil
5 Instituto Nacional de Infectologia Evandro Chagas, Rio de Janeiro, Brazil
6 Departamento de Infectología. Instituto Nacional de Ciencias Médicas y Nutrición, Salvador Zubirán, Mexico City, Mexico
7 Department of Clinical Research, London School of Hygiene and Tropical Medicine, London, UK
8 Department of Pharmacology and Therapeutics, Institute of Systems, Molecular and Integrative Biology, University of Liverpool, Liverpool, United Kingdom
*Correspondence:
Dr Pankaj K Bhavsar, Airway Disease, National Heart & Lung Institute, Imperial College London, Dovehouse St, London SW3 6LY, United Kingdom. Email: p.bhavsar@imperial.ac.uk
- 2 Figures, 3 Tables
Word Count: 2,692
ABSTRACT
Question addressed by the study: Favipiravir has shown efficacy against SARS-CoV-2 in patients under 60, but data linking plasma concentrations to clinical outcomes are limited. This study investigated whether favipiravir plasma concentrations influence clinical efficacy and outcomes in hospitalised COVID-19 patients and asked: how can antiviral dosing strategies be optimised to improve pandemic preparedness and treatment efficacy?
Materials/patients and methods: Adult participants were drawn from the PIONEER trial, in which patients received oral favipiravir (1800 mg twice daily on day 1, then 800 mg twice daily for nine days) plus standard care. This analysis..
DOI record:
{
"DOI": "10.1183/23120541.01560-2025",
"ISSN": [
"2312-0541"
],
"URL": "http://dx.doi.org/10.1183/23120541.01560-2025",
"abstract": "<jats:sec>\n <jats:title>Question addressed by the study</jats:title>\n <jats:p>Favipiravir has shown efficacy against SARS-CoV-2 in patients under 60, but data linking plasma concentrations to clinical outcomes are limited. This study investigated whether favipiravir plasma concentrations influence clinical efficacy and outcomes in hospitalised COVID-19 patients and asked: how can antiviral dosing strategies be optimised to improve pandemic preparedness and treatment efficacy?</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Materials/patients and methods</jats:title>\n <jats:p>Adult participants were drawn from the PIONEER trial, in which patients received oral favipiravir (1800 mg twice daily on day 1, then 800 mg twice daily for nine days) plus standard care. This analysis included patients with confirmed COVID-19 and ≥75% study adherence. Samples were collected between days 5–10 post-treatment initiation. The primary outcome was time to clinical improvement. Secondary outcomes included achievement of clinical improvement and mortality risk.</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Results</jats:title>\n <jats:p>\n Of 140 patients (50% male; mean age 59.5 [\n <jats:sc>sd</jats:sc>\n 14.8]), 29 (21%) reached target plasma concentrations. Mean time to improvement was 7.7 [5.9] vs 9.1 [7.2] days for target achievers vs non-achievers (p=0.26). Target was more often achieved in females (34%) than males (7%) (p=0.0002). Plasma concentration inversely correlated with BMI (r=–0.4; p<0.0001), with lower BMI in achievers (26.0 [5.1] vs 30.5 [6.9]; p=0.003). ALP and ALT levels were also lower in achievers (p=0.004 and p=0.02, respectively).\n </jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Answer to the question</jats:title>\n <jats:p>Most patients did not reach target favipiravir levels. Concentrations were influenced by sex, BMI, and liver function, confirming the need for pharmacokinetically guided dosing and therapeutic monitoring to optimise antiviral efficacy in future pandemic responses.</jats:p>\n </jats:sec>",
"accepted": {
"date-parts": [
[
2026,
1,
26
]
]
},
"alternative-id": [
"10.1183/23120541.01560-2025"
],
"author": [
{
"affiliation": [],
"family": "Wawman",
"given": "Rebecca E.",
"sequence": "first"
},
{
"ORCID": "https://orcid.org/0000-0002-9052-4638",
"affiliation": [],
"authenticated-orcid": false,
"family": "Shah",
"given": "Pallav L.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Boffito",
"given": "Marta",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Tonkin",
"given": "James",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Conway",
"given": "Francesca",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Tana",
"given": "Anand",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Santos",
"given": "Breno R.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Grinsztejn",
"given": "Beatriz",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ramírez",
"given": "Brenda Crabtree",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Pertinez",
"given": "Henry",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Owen",
"given": "Andrew",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Curley",
"given": "Paul",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Arshad",
"given": "Usman",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Cox",
"given": "Helen",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Johnson",
"given": "Mark R.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Pozniak",
"given": "Anton",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Pelly",
"given": "Michael",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Orton",
"given": "Christopher M.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Bhavsar",
"given": "Pankaj K.",
"sequence": "additional"
}
],
"container-title": "ERJ Open Research",
"container-title-short": "ERJ Open Res",
"content-domain": {
"crossmark-restriction": true,
"domain": [
"publications.ersnet.org"
]
},
"created": {
"date-parts": [
[
2026,
3,
26
]
],
"date-time": "2026-03-26T18:15:25Z",
"timestamp": 1774548925000
},
"deposited": {
"date-parts": [
[
2026,
3,
26
]
],
"date-time": "2026-03-26T18:15:26Z",
"timestamp": 1774548926000
},
"indexed": {
"date-parts": [
[
2026,
3,
26
]
],
"date-time": "2026-03-26T19:02:10Z",
"timestamp": 1774551730989,
"version": "3.50.1"
},
"is-referenced-by-count": 0,
"issued": {
"date-parts": [
[
2026,
3,
26
]
]
},
"language": "en",
"license": [
{
"URL": "http://creativecommons.org/licenses/by/4.0/",
"content-version": "am",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
3,
26
]
],
"date-time": "2026-03-26T00:00:00Z",
"timestamp": 1774483200000
}
}
],
"link": [
{
"URL": "https://syndication.highwire.org/content/doi/10.1183/23120541.01560-2025",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "81",
"original-title": [],
"page": "01560-2025",
"prefix": "10.1183",
"published": {
"date-parts": [
[
2026,
3,
26
]
]
},
"published-online": {
"date-parts": [
[
2026,
3,
26
]
]
},
"publisher": "European Respiratory Society (ERS)",
"reference-count": 0,
"references-count": 0,
"relation": {},
"resource": {
"primary": {
"URL": "https://publications.ersnet.org/lookup/doi/10.1183/23120541.01560-2025"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "The Relationship Between Plasma Favipiravir Concentrations and Clinical Outcomes in COVID-19",
"type": "journal-article",
"update-policy": "https://doi.org/10.1183/ers-crossmark-policy"
}