Optimal Dose and Safety of Intravenous Favipiravir in Hospitalized Patients With COVID-19: A Dose-Escalating, Randomized Controlled Phase Ib Study
et al., Clinical Pharmacology & Therapeutics, doi:10.1002/cpt.70261, NCT04746183, Mar 2026
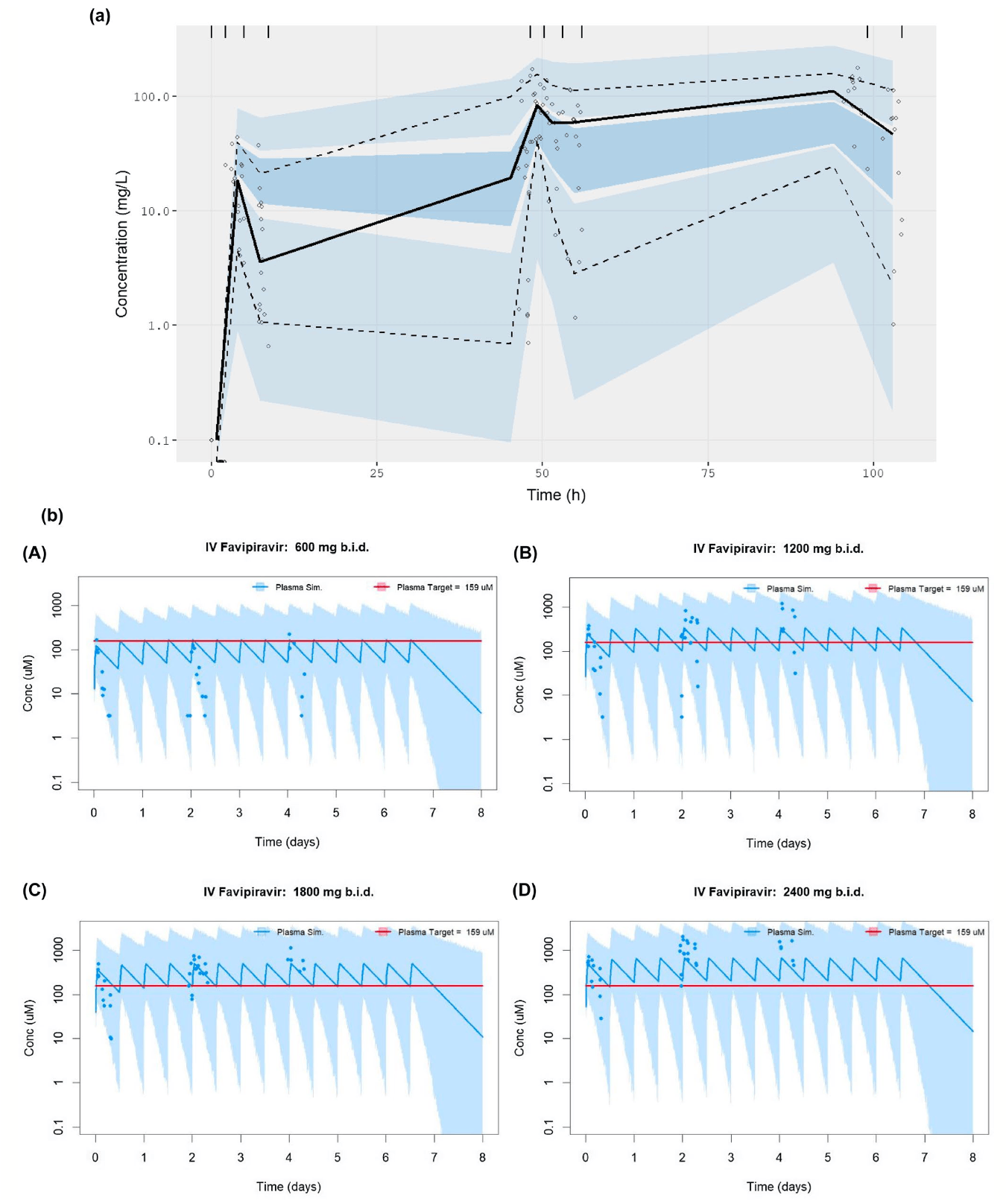
Phase Ib dose-escalating RCT of 24 hospitalized COVID-19 patients showing safety and tolerability of intravenous (IV) favipiravir at doses up to 2,400 mg twice daily.
Potential risks of favipiravir include kidney injury1-3, liver injury2-5, cardiovascular events5,6, pulmonary toxicity6,7, and mutagenicity, carcinogenicity, teratogenicity, embryotoxicity, and the creation of dangerous variants8-14.
1.
Abdulaziz et al., Clinical Features and Prognosis of Acute Kidney Injury in Hospital-Admitted Patients with COVID-19 in Egypt: A Single-Center Experience, Mansoura Medical Journal, doi:10.58775/2735-3990.1433.
2.
Ülger et al., Experimental evaluation of favipiravir (T-705)-induced liver and kidney toxicity in rats, Food and Chemical Toxicology, doi:10.1016/j.fct.2025.115472.
3.
El-Fetouh et al., Experimental Studies on Some Drugs Used in Covid-19 Treatment (Favipiravir and Dexamethasone) in Albino Rats, Journal of Advanced Veterinary Research, 13:10, www.advetresearch.com/index.php/AVR/article/view/1635.
4.
Almutairi et al., Liver Injury in Favipiravir-Treated COVID-19 Patients: Retrospective Single-Center Cohort Study, Tropical Medicine and Infectious Disease, doi:10.3390/tropicalmed8020129.
5.
Siby et al., Temporal Trends in Serious Adverse Events Associated with Oral Antivirals During the COVID-19 Pandemic: Insights from the FAERS Database (2020–2023), Open Forum Infectious Diseases, doi:10.1093/ofid/ofaf695.1825.
6.
Ozhan et al., Evaluation of the cardiopulmonary effects of repurposed COVID-19 therapeutics in healthy rats, Scientific Reports, doi:10.1038/s41598-025-31048-4.
7.
Ülger (B) et al., Evaluation of the effects of favipiravir (T-705) on the lung tissue of healty rats: An experimental study, Food and Chemical Toxicology, doi:10.1016/j.fct.2025.115235.
8.
Zhirnov et al., Favipiravir: the hidden threat of mutagenic action, Journal of microbiology, epidemiology and immunobiology, doi:10.36233/0372-9311-114.
9.
Waters et al., Human genetic risk of treatment with antiviral nucleoside analog drugs that induce lethal mutagenesis: the special case of molnupiravir, Environmental and Molecular Mutagenesis, doi:10.1002/em.22471.
10.
Hadj Hassine et al., Lethal Mutagenesis of RNA Viruses and Approved Drugs with Antiviral Mutagenic Activity, Viruses, doi:10.3390/v14040841.
11.
Shum, C., An investigational study into the drug-associated mutational signature in SARS-CoV-2 viruses, The University of Hong Kong, PhD Thesis, hub.hku.hk/handle/10722/344396.
12.
Shiraki et al., Convenient screening of the reproductive toxicity of favipiravir and antiviral drugs in Caenorhabditis elegans, Heliyon, doi:10.1016/j.heliyon.2024.e35331.
Rowland et al., 18 Mar 2026, Randomized Controlled Trial, United Kingdom, peer-reviewed, median age 74.0, 33 authors, study period 10 September, 2022 - 1 November, 2023, trial NCT04746183 (history).
Contact: tom.fletcher@lstmed.ac.uk.
Optimal Dose and Safety of Intravenous Favipiravir in Hospitalized Patients With COVID ‐19: A Dose‐Escalating, Randomized Controlled Phase Ib Study
Clinical Pharmacology & Therapeutics, doi:10.1002/cpt.70261
AGILE (NCT04746183 ) is a Phase Ib/IIa platform, evaluating candidates to treat COVID-19. Candidate Specific Trial 6 evaluated the safety and optimal dose of a novel intravenous formulation of favipiravir in a dose-escalating, openlabel, randomized, controlled, Bayesian adaptive Phase Ib trial. Hospitalized adults with PCR-confirmed SARS-CoV-2 infection, within 14 days of symptomatic COVID-19 were randomized 2:1 in groups of 6 (n = 4 favipiravir, n = 2 standard of care) to ascending doses of intravenous favipiravir twice daily (b.i.d.) for 7 days or standard of care. Clinical data, safety evaluations, virology and pharmacokinetic samples were collected. The primary outcome was safety. Secondary outcomes included clinical, pharmacokinetic and virological endpoints. Twenty-four participants enrolled between September 10, 2022 and November 1, 2023 [10/24 female; median age 74 years (range 52-93)]. Favipiravir was well tolerated despite a high background rate of unrelated adverse events. No dose limiting toxicities were observed, with a model-predicted dose limiting toxicity risk of 16.8% and probability of unacceptable toxicity of 2.7% at the highest dose level. No serious adverse events were deemed related to favipiravir but an expected association with asymptomatic, transient hyperuricemia was observed. Favipiravir exposures increased disproportionally to dose with significant
AUTHOR CONTRIBUTIONS T.R., S.H.K., T.E.F. and R.F. wrote the manuscript; S.H.K., G.G., R.F., T.E.F., T.J., T.R., P.M., D.G.L., A.O., M.J. designed the research; T.E.F., R.L., R.F., L.W., T.R., K.D., M.I., Y.E., H.R., M.T., J.G., J.C., O.O. performed the research; S.H.K., G.G., G.S., W.W., H.P., A.O., T.E.F., R.F., T.R., L.D., E.C., L.J.E., V.S., W.G., C.H., C.K., K.B., J.A.H. analyzed the data.
References
Abdelnabi, The combined treatment of molnupiravir and favipiravir results in a potentiation of antiviral efficacy in a SARS-CoV-2 hamster infection model, EBioMedicine
Arshad, Prioritization of anti-SARS-Cov-2 drug repurposing opportunities based on plasma and target site concentrations derived from their established human pharmacokinetics, Clin. Pharmacol. Ther
Bixler, Efficacy of favipiravir (T-705) in nonhuman primates infected with Ebola virus or Marburg virus, Antiviral Res
Bixler, Intracellular conversion and in vivo dose response of favipiravir (T-705) in rodents infected with Ebola virus, Antiviral Res
Challenger, Development and validation of an LC-MS/ MS method for quantification of favipiravir in human plasma, J. Pharm. Biomed. Anal
Choi, Antiviral activity and safety of remdesivir against SARS-CoV-2 infection in human pluripotent stem cell-derived cardiomyocytes, Antiviral Res
Choy, lopinavir, emetine, and homoharringtonine inhibit SARS-CoV-2 replication in vitro, Antiviral Res
Driouich, Favipiravir antiviral efficacy against SARS-CoV-2 in a hamster model, Nat. Commun
Du, Chen, Favipiravir: pharmacokinetics and concerns about clinical trials for 2019-nCoV infection, Clin. Pharmacol. Ther
Eloy, Dose rationale for favipiravir use in patients infected with SARS-CoV-2, Clin. Pharmacol. Ther
Fidler, Nonlinear mixed-effects model development and simulation using nlmixr and related R open-source packages, CPT Pharmacometrics Syst. Pharmacol
Furuta, Gowen, Takahashi, Shiraki, Smee et al., Favipiravir (T-705), a novel viral RNA polymerase inhibitor, Antiviral Res
Furuta, Komeno, Nakamura, Favipiravir (T-705), a broad spectrum inhibitor of viral RNA polymerase, Proc. Jpn. Acad. Ser. B Phys. Biol. Sci
Griffiths, AGILE: a seamless phase I/IIa platform for the rapid evaluation of candidates for COVID-19 treatment: an update to the structured summary of a study protocol for a randomised platform trial letter, Trials
Gülhan, Pharmacokinetic characterization of favipiravir in patients with COVID-19, Br. J. Clin. Pharmacol
Hattori, GRL-0920, an indole Chloropyridinyl Ester, completely blocks SARS-CoV-2 infection, MBio
Hayden, Favipiravir treatment of uncomplicated influenza in adults: results of two phase 3, randomized, doubleblind, placebo-controlled trials, J. Infect. Dis
Hayden, Lenk, Epstein, Kang, Oral favipiravir exposure and pharmacodynamic effects in outpatient adults with acute influenza, J. Infect. Dis
Huchting, Vanderlinden, Van Berwaer, Meier, Naesens, Cell line-dependent activation and antiviral activity of T-1105, the non-fluorinated analogue of T-705 (favipiravir), Antiviral Res
Irie, Pharmacokinetics of favipiravir in critically ill patients with COVID-19, Clin. Transl. Sci
Irie, Population pharmacokinetics of favipiravir in patients with COVID-19, CPT Pharmacometrics Syst. Pharmacol
Jaki, Barnett, Titman, Mozgunov, A seamless phase I/II platform design with a time-to-event efficacy endpoint for potential COVID-19 therapies, Stat. Methods Med. Res
Jeon, Identification of antiviral drug candidates against SARS-CoV-2 from FDA-approved drugs, Antimicrob. Agents Chemother
Kaptein, Favipiravir at high doses has potent antiviral activity in SARS-CoV-2-infected hamsters, whereas hydroxychloroquine lacks activity, Proc. Natl. Acad. Sci. U.S.A
Khoo, Optimal dose and safety of molnupiravir in patients with early SARS-CoV-2: a phase I, open-label, doseescalating, randomized controlled study, J. Antimicrob. Chemother
Korula, Favipiravir for treating COVID-19, Cochrane Database Syst. Rev
Madelain, Favipiravir pharmacokinetics in nonhuman primates and insights for future efficacy studies of hemorrhagic fever viruses, Antimicrob. Agents Chemother
Mozgunov, Jaki, Paoletti, Randomized dose-escalation designs for drug combination cancer trials with immunotherapy, J. Biopharm. Stat
Nguyen, Favipiravir pharmacokinetics in Ebola-infected patients of the JIKI trial reveals concentrations lower than targeted, PLoS Negl. Trop. Dis
Pertinez, Rajoli, Khoo, Owen, Pharmacokinetic modelling to estimate intracellular favipiravir ribofuranosyl-5′-triphosphate exposure to support posology for SARS-CoV-2, J. Antimicrob. Chemother
Pizzorno, In vitro evaluation of antiviral activity of single and combined repurposable drugs against SARS-CoV-2, Antiviral Res
Sakurai, A phase I, randomized, double-blind, placebocontrolled, single ascending-dose study to evaluate the pharmacokinetics, safety and tolerability of injectable favipiravir in healthy subjects <https
Shannon, Rapid incorporation of Favipiravir by the fast and permissive viral RNA polymerase complex results in SARS-CoV-2 lethal mutagenesis, Nat. Commun
Sissoko, Experimental treatment with favipiravir for Ebola virus disease (the JIKI trial): a historically controlled, singlearm proof-of-concept trial in Guinea, PLoS Med
Walker, An open label, adaptive, phase 1 trial of highdose oral nitazoxanide in healthy volunteers: an antiviral candidate for SARS-CoV-2, Clin. Pharmacol. Ther
Wang, Phase 2a, open-label, dose-escalating, multicenter pharmacokinetic study of favipiravir (T-705) in combination with oseltamivir in patients with severe influenza, EBioMedicine
Wang, Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro, Cell Res
DOI record:
{
"DOI": "10.1002/cpt.70261",
"ISSN": [
"0009-9236",
"1532-6535"
],
"URL": "http://dx.doi.org/10.1002/cpt.70261",
"abstract": "<jats:p>\n AGILE (NCT04746183) is a Phase Ib/IIa platform, evaluating candidates to treat COVID‐19. Candidate Specific Trial 6 evaluated the safety and optimal dose of a novel intravenous formulation of favipiravir in a dose‐escalating, open‐label, randomized, controlled, Bayesian adaptive Phase Ib trial. Hospitalized adults with PCR‐confirmed SARS‐CoV‐2 infection, within 14 days of symptomatic COVID‐19 were randomized 2:1 in groups of 6 (\n <jats:italic>n</jats:italic>\n = 4 favipiravir,\n <jats:italic>n</jats:italic>\n = 2 standard of care) to ascending doses of intravenous favipiravir twice daily (b.i.d.) for 7 days or standard of care. Clinical data, safety evaluations, virology and pharmacokinetic samples were collected. The primary outcome was safety. Secondary outcomes included clinical, pharmacokinetic and virological endpoints. Twenty‐four participants enrolled between September 10, 2022 and November 1, 2023 [10/24 female; median age 74 years (range 52–93)]. Favipiravir was well tolerated despite a high background rate of unrelated adverse events. No dose limiting toxicities were observed, with a model‐predicted dose limiting toxicity risk of 16.8% and probability of unacceptable toxicity of 2.7% at the highest dose level. No serious adverse events were deemed related to favipiravir but an expected association with asymptomatic, transient hyperuricemia was observed. Favipiravir exposures increased disproportionally to dose with significant accumulation in plasma, but with marked variability between participants within each cohort. This novel formulation of favipiravir was safe at sustained high doses that reached pre‐specified pharmacokinetic targets in a study group with frailty and complex health profiles. We consider doses up to 2,400 mg b.i.d. to be safe for further evaluation.\n </jats:p>",
"alternative-id": [
"10.1002/cpt.70261"
],
"article-number": "cpt.70261",
"assertion": [
{
"group": {
"label": "Publication History",
"name": "publication_history"
},
"label": "Received",
"name": "received",
"order": 0,
"value": "2025-10-08"
},
{
"group": {
"label": "Publication History",
"name": "publication_history"
},
"label": "Accepted",
"name": "accepted",
"order": 2,
"value": "2026-02-23"
},
{
"group": {
"label": "Publication History",
"name": "publication_history"
},
"label": "Published",
"name": "published",
"order": 3,
"value": "2026-03-18"
}
],
"author": [
{
"ORCID": "https://orcid.org/0000-0001-5059-065X",
"affiliation": [
{
"name": "Liverpool School of Tropical Medicine Liverpool UK"
},
{
"name": "Liverpool University Hospitals Foundation Trust Liverpool UK"
}
],
"authenticated-orcid": false,
"family": "Rowland",
"given": "Tim",
"sequence": "first"
},
{
"ORCID": "https://orcid.org/0000-0003-0227-4200",
"affiliation": [
{
"name": "Liverpool University Hospitals Foundation Trust Liverpool UK"
},
{
"name": "NIHR Liverpool Clinical Research Facility Liverpool UK"
}
],
"authenticated-orcid": false,
"family": "FitzGerald",
"given": "Richard",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0002-8978-6067",
"affiliation": [
{
"name": "Department of Pharmacology and Therapeutics, Institute of Integrative, Systems and Molecular Biology, Centre for Experimental Therapeutics (TherEx) University of Liverpool Liverpool UK"
}
],
"authenticated-orcid": false,
"family": "Challenger",
"given": "Elizabeth",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0001-5557-9396",
"affiliation": [
{
"name": "Department of Pharmacology and Therapeutics, Institute of Integrative, Systems and Molecular Biology, Centre for Experimental Therapeutics (TherEx) University of Liverpool Liverpool UK"
}
],
"authenticated-orcid": false,
"family": "Dickinson",
"given": "Laura",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0009-0007-0078-3294",
"affiliation": [
{
"name": "Department of Pharmacology and Therapeutics, Institute of Integrative, Systems and Molecular Biology, Centre for Experimental Therapeutics (TherEx) University of Liverpool Liverpool UK"
}
],
"authenticated-orcid": false,
"family": "Else",
"given": "Laura J.",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0002-3827-4387",
"affiliation": [
{
"name": "Liverpool University Hospitals Foundation Trust Liverpool UK"
},
{
"name": "NIHR Liverpool Clinical Research Facility Liverpool UK"
}
],
"authenticated-orcid": false,
"family": "Walker",
"given": "Lauren",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "NIHR Liverpool Clinical Research Facility Liverpool UK"
}
],
"family": "Hale",
"given": "Colin",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0002-0429-0186",
"affiliation": [
{
"name": "Department of Pharmacology and Therapeutics, Institute of Integrative, Systems and Molecular Biology, Centre for Experimental Therapeutics (TherEx) University of Liverpool Liverpool UK"
}
],
"authenticated-orcid": false,
"family": "Shaw",
"given": "Victoria",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "NIHR Liverpool Clinical Research Facility Liverpool UK"
}
],
"family": "Kelly",
"given": "Callum",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "NIHR Liverpool Clinical Research Facility Liverpool UK"
}
],
"family": "Lyon",
"given": "Rebecca",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "NIHR Liverpool Clinical Research Facility Liverpool UK"
}
],
"family": "Gibney",
"given": "Jennifer",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "PHARMExcel Welwyn Garden City UK"
}
],
"family": "Dhamani",
"given": "Karim",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "PHARMExcel Welwyn Garden City UK"
}
],
"family": "Irwin",
"given": "Margaret",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "PHARMExcel Welwyn Garden City UK"
}
],
"family": "Enever",
"given": "Yvanne",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0002-1064-3187",
"affiliation": [
{
"name": "Department of Pharmacology and Therapeutics, Institute of Integrative, Systems and Molecular Biology, Centre for Experimental Therapeutics (TherEx) University of Liverpool Liverpool UK"
}
],
"authenticated-orcid": false,
"family": "Tetlow",
"given": "Michelle",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0003-0683-8085",
"affiliation": [
{
"name": "Department of Pharmacology and Therapeutics, Institute of Integrative, Systems and Molecular Biology, Centre for Experimental Therapeutics (TherEx) University of Liverpool Liverpool UK"
}
],
"authenticated-orcid": false,
"family": "Wood",
"given": "William",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0001-7443-4520",
"affiliation": [
{
"name": "Department of Pharmacology and Therapeutics, Institute of Integrative, Systems and Molecular Biology, Centre for Experimental Therapeutics (TherEx) University of Liverpool Liverpool UK"
}
],
"authenticated-orcid": false,
"family": "Reynolds",
"given": "Helen",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0001-8063-6399",
"affiliation": [
{
"name": "Department of Pharmacology and Therapeutics, Institute of Integrative, Systems and Molecular Biology, Centre for Experimental Therapeutics (TherEx) University of Liverpool Liverpool UK"
}
],
"authenticated-orcid": false,
"family": "Chiong",
"given": "Justin",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0003-2921-0522",
"affiliation": [
{
"name": "Bangor University Bangor UK"
}
],
"authenticated-orcid": false,
"family": "Osanlou",
"given": "Orod",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0009-0004-4522-9201",
"affiliation": [
{
"name": "Centre of Excellence for Long‐acting Therapeutics University of Liverpool Liverpool UK"
}
],
"authenticated-orcid": false,
"family": "Pertinez",
"given": "Henry",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0002-5758-0179",
"affiliation": [
{
"name": "Department of Pharmacology and Therapeutics, Institute of Integrative, Systems and Molecular Biology, Centre for Experimental Therapeutics (TherEx) University of Liverpool Liverpool UK"
}
],
"authenticated-orcid": false,
"family": "Bullock",
"given": "Katie",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0002-1865-3195",
"affiliation": [
{
"name": "Department of Pharmacology and Therapeutics, Institute of Integrative, Systems and Molecular Biology, Centre for Experimental Therapeutics (TherEx) University of Liverpool Liverpool UK"
}
],
"authenticated-orcid": false,
"family": "Greenhalf",
"given": "William",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0002-9819-7651",
"affiliation": [
{
"name": "Centre of Excellence for Long‐acting Therapeutics University of Liverpool Liverpool UK"
}
],
"authenticated-orcid": false,
"family": "Owen",
"given": "Andrew",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0001-7680-2200",
"affiliation": [
{
"name": "Liverpool School of Tropical Medicine Liverpool UK"
}
],
"authenticated-orcid": false,
"family": "Lalloo",
"given": "David G.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Liverpool School of Tropical Medicine Liverpool UK"
},
{
"name": "Keble College Oxford UK"
}
],
"family": "Jacobs",
"given": "Michael",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0002-6582-0275",
"affiliation": [
{
"name": "Institute of Infection, Veterinary and Ecological Sciences, Faculty of Health and Life Science University of Liverpool Liverpool UK"
}
],
"authenticated-orcid": false,
"family": "Hiscox",
"given": "Julian A.",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0002-1096-188X",
"affiliation": [
{
"name": "MRC Biostatistics Unit University of Cambridge Cambridge UK"
},
{
"name": "Faculty of Informatics and Data Science University of Regensburg Regensburg Delaware USA"
}
],
"authenticated-orcid": false,
"family": "Jaki",
"given": "Thomas",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0001-6810-0284",
"affiliation": [
{
"name": "MRC Biostatistics Unit University of Cambridge Cambridge UK"
}
],
"authenticated-orcid": false,
"family": "Mozgunov",
"given": "Pavel",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0009-0004-0582-839X",
"affiliation": [
{
"name": "Southampton Clinical Trials Unit University of Southampton & University Hospital Southampton NHS Foundation Trust Southampton UK"
}
],
"authenticated-orcid": false,
"family": "Saunders",
"given": "Geoffrey",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0002-9579-8021",
"affiliation": [
{
"name": "Southampton Clinical Trials Unit University of Southampton & University Hospital Southampton NHS Foundation Trust Southampton UK"
}
],
"authenticated-orcid": false,
"family": "Griffiths",
"given": "Gareth",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0002-2769-0967",
"affiliation": [
{
"name": "Liverpool University Hospitals Foundation Trust Liverpool UK"
},
{
"name": "Department of Pharmacology and Therapeutics, Institute of Integrative, Systems and Molecular Biology, Centre for Experimental Therapeutics (TherEx) University of Liverpool Liverpool UK"
}
],
"authenticated-orcid": false,
"family": "Khoo",
"given": "Saye H.",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0002-3712-415X",
"affiliation": [
{
"name": "Liverpool School of Tropical Medicine Liverpool UK"
},
{
"name": "Liverpool University Hospitals Foundation Trust Liverpool UK"
}
],
"authenticated-orcid": false,
"family": "Fletcher",
"given": "Thomas E.",
"sequence": "additional"
},
{
"affiliation": [],
"name": "the AGILE CST‐6 Study Group",
"sequence": "additional"
}
],
"container-title": "Clinical Pharmacology & Therapeutics",
"container-title-short": "Clin Pharma and Therapeutics",
"content-domain": {
"crossmark-restriction": true,
"domain": [
"ascpt.onlinelibrary.wiley.com"
]
},
"created": {
"date-parts": [
[
2026,
3,
18
]
],
"date-time": "2026-03-18T15:29:41Z",
"timestamp": 1773847781000
},
"deposited": {
"date-parts": [
[
2026,
3,
18
]
],
"date-time": "2026-03-18T15:29:47Z",
"timestamp": 1773847787000
},
"funder": [
{
"DOI": "10.13039/501100000265",
"award": [
"MR/V028391/1"
],
"award-info": [
{
"award-number": [
"MR/V028391/1"
]
}
],
"doi-asserted-by": "publisher",
"id": [
{
"asserted-by": "publisher",
"id": "10.13039/501100000265",
"id-type": "DOI"
}
],
"name": "Medical Research Council"
},
{
"DOI": "10.13039/501100000265",
"award": [
"MR/W005611/1"
],
"award-info": [
{
"award-number": [
"MR/W005611/1"
]
}
],
"doi-asserted-by": "publisher",
"id": [
{
"asserted-by": "publisher",
"id": "10.13039/501100000265",
"id-type": "DOI"
}
],
"name": "Medical Research Council"
},
{
"DOI": "10.13039/501100000265",
"award": [
"MC_UU_00040/03"
],
"award-info": [
{
"award-number": [
"MC_UU_00040/03"
]
}
],
"doi-asserted-by": "publisher",
"id": [
{
"asserted-by": "publisher",
"id": "10.13039/501100000265",
"id-type": "DOI"
}
],
"name": "Medical Research Council"
},
{
"DOI": "10.13039/100010269",
"award": [
"221590/Z/20/Z"
],
"award-info": [
{
"award-number": [
"221590/Z/20/Z"
]
}
],
"doi-asserted-by": "publisher",
"id": [
{
"asserted-by": "publisher",
"id": "10.13039/100010269",
"id-type": "DOI"
}
],
"name": "Wellcome Trust"
},
{
"DOI": "10.13039/100000038",
"award": [
"75F40120C00085"
],
"award-info": [
{
"award-number": [
"75F40120C00085"
]
}
],
"doi-asserted-by": "publisher",
"id": [
{
"asserted-by": "publisher",
"id": "10.13039/100000038",
"id-type": "DOI"
}
],
"name": "U.S. Food and Drug Administration"
},
{
"DOI": "10.13039/501100000272",
"award": [
"NIHR300576"
],
"award-info": [
{
"award-number": [
"NIHR300576"
]
}
],
"doi-asserted-by": "publisher",
"id": [
{
"asserted-by": "publisher",
"id": "10.13039/501100000272",
"id-type": "DOI"
}
],
"name": "National Institute for Health and Care Research"
},
{
"DOI": "10.13039/501100022419",
"doi-asserted-by": "publisher",
"id": [
{
"asserted-by": "publisher",
"id": "10.13039/501100022419",
"id-type": "DOI"
}
],
"name": "National Institute for Health Research Southampton Biomedical Research Centre"
}
],
"indexed": {
"date-parts": [
[
2026,
3,
18
]
],
"date-time": "2026-03-18T23:14:52Z",
"timestamp": 1773875692380,
"version": "3.50.1"
},
"is-referenced-by-count": 0,
"issued": {
"date-parts": [
[
2026,
3,
18
]
]
},
"language": "en",
"license": [
{
"URL": "http://creativecommons.org/licenses/by/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
3,
18
]
],
"date-time": "2026-03-18T00:00:00Z",
"timestamp": 1773792000000
}
},
{
"URL": "http://doi.wiley.com/10.1002/tdm_license_1.1",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
3,
18
]
],
"date-time": "2026-03-18T00:00:00Z",
"timestamp": 1773792000000
}
}
],
"link": [
{
"URL": "https://ascpt.onlinelibrary.wiley.com/doi/pdf/10.1002/cpt.70261",
"content-type": "application/pdf",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://ascpt.onlinelibrary.wiley.com/doi/full-xml/10.1002/cpt.70261",
"content-type": "application/xml",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://ascpt.onlinelibrary.wiley.com/doi/pdf/10.1002/cpt.70261",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "311",
"original-title": [],
"prefix": "10.1002",
"published": {
"date-parts": [
[
2026,
3,
18
]
]
},
"published-online": {
"date-parts": [
[
2026,
3,
18
]
]
},
"publisher": "Wiley",
"reference": [
{
"DOI": "10.1186/s13063-021-05458-4",
"article-title": "AGILE: a seamless phase I/IIa platform for the rapid evaluation of candidates for COVID‐19 treatment: an update to the structured summary of a study protocol for a randomised platform trial letter",
"author": "Griffiths G.O.",
"doi-asserted-by": "crossref",
"first-page": "1",
"journal-title": "Trials",
"key": "e_1_2_10_1_1",
"volume": "22",
"year": "2021"
},
{
"DOI": "10.2183/pjab.93.027",
"article-title": "Favipiravir (T‐705), a broad spectrum inhibitor of viral RNA polymerase",
"author": "Furuta Y.",
"doi-asserted-by": "crossref",
"first-page": "449",
"journal-title": "Proc. Jpn. Acad. Ser. B Phys. Biol. Sci.",
"key": "e_1_2_10_2_1",
"volume": "93",
"year": "2017"
},
{
"DOI": "10.1016/j.ebiom.2020.103125",
"article-title": "Phase 2a, open‐label, dose‐escalating, multi‐center pharmacokinetic study of favipiravir (T‐705) in combination with oseltamivir in patients with severe influenza",
"author": "Wang Y.",
"doi-asserted-by": "crossref",
"journal-title": "EBioMedicine",
"key": "e_1_2_10_3_1",
"volume": "62",
"year": "2020"
},
{
"DOI": "10.1093/infdis/jiad409",
"article-title": "Oral favipiravir exposure and pharmacodynamic effects in outpatient adults with acute influenza",
"author": "Hayden F.G.",
"doi-asserted-by": "crossref",
"first-page": "e395",
"journal-title": "J. Infect. Dis.",
"key": "e_1_2_10_4_1",
"volume": "230",
"year": "2024"
},
{
"DOI": "10.1371/journal.pmed.1001967",
"article-title": "Experimental treatment with favipiravir for Ebola virus disease (the JIKI trial): a historically controlled, single‐arm proof‐of‐concept trial in Guinea",
"author": "Sissoko D.",
"doi-asserted-by": "crossref",
"journal-title": "PLoS Med.",
"key": "e_1_2_10_5_1",
"volume": "13",
"year": "2016"
},
{
"article-title": "Favipiravir for treating COVID‐19",
"author": "Korula P.",
"journal-title": "Cochrane Database Syst. Rev.",
"key": "e_1_2_10_6_1",
"volume": "2",
"year": "2024"
},
{
"key": "e_1_2_10_7_1",
"unstructured": "Evaluation and Licensing Division Pharmaceutical and Food Safety Bureau Ministry of Health Labour and Welfare.Report on the deliberation results<https://www.pmda.go.jp/files/000210319.pdf>."
},
{
"DOI": "10.1016/j.antiviral.2013.09.015",
"article-title": "Favipiravir (T‐705), a novel viral RNA polymerase inhibitor",
"author": "Furuta Y.",
"doi-asserted-by": "crossref",
"first-page": "446",
"journal-title": "Antiviral Res.",
"key": "e_1_2_10_8_1",
"volume": "100",
"year": "2013"
},
{
"DOI": "10.1002/cpt.1844",
"article-title": "Favipiravir: pharmacokinetics and concerns about clinical trials for 2019‐nCoV infection",
"author": "Du Y.X.",
"doi-asserted-by": "crossref",
"first-page": "242",
"journal-title": "Clin. Pharmacol. Ther.",
"key": "e_1_2_10_9_1",
"volume": "108",
"year": "2020"
},
{
"DOI": "10.1002/cpt.1877",
"article-title": "Dose rationale for favipiravir use in patients infected with SARS‐CoV‐2",
"author": "Eloy P.",
"doi-asserted-by": "crossref",
"first-page": "188",
"journal-title": "Clin. Pharmacol. Ther.",
"key": "e_1_2_10_10_1",
"volume": "108",
"year": "2020"
},
{
"DOI": "10.1128/AAC.00819-20",
"article-title": "Identification of antiviral drug candidates against SARS‐CoV‐2 from FDA‐approved drugs",
"author": "Jeon S.",
"doi-asserted-by": "crossref",
"journal-title": "Antimicrob. Agents Chemother.",
"key": "e_1_2_10_11_1",
"volume": "64",
"year": "2020"
},
{
"DOI": "10.1038/s41467-020-18463-z",
"article-title": "Rapid incorporation of Favipiravir by the fast and permissive viral RNA polymerase complex results in SARS‐CoV‐2 lethal mutagenesis",
"author": "Shannon A.",
"doi-asserted-by": "crossref",
"first-page": "4682",
"journal-title": "Nat. Commun.",
"key": "e_1_2_10_12_1",
"volume": "11",
"year": "2020"
},
{
"DOI": "10.1016/j.antiviral.2020.104878",
"article-title": "In vitro evaluation of antiviral activity of single and combined repurposable drugs against SARS‐CoV‐2",
"author": "Pizzorno A.",
"doi-asserted-by": "crossref",
"journal-title": "Antiviral Res.",
"key": "e_1_2_10_13_1",
"volume": "181",
"year": "2020"
},
{
"DOI": "10.1016/j.antiviral.2020.104786",
"article-title": "Remdesivir, lopinavir, emetine, and homoharringtonine inhibit SARS‐CoV‐2 replication in vitro",
"author": "Choy K.T.",
"doi-asserted-by": "crossref",
"journal-title": "Antiviral Res.",
"key": "e_1_2_10_14_1",
"volume": "178",
"year": "2020"
},
{
"DOI": "10.1128/mBio.01833-20",
"article-title": "GRL‐0920, an indole Chloropyridinyl Ester, completely blocks SARS‐CoV‐2 infection",
"author": "Hattori S.I.",
"doi-asserted-by": "crossref",
"first-page": "1",
"journal-title": "MBio",
"key": "e_1_2_10_15_1",
"volume": "11",
"year": "2020"
},
{
"DOI": "10.1016/j.antiviral.2020.104955",
"article-title": "Antiviral activity and safety of remdesivir against SARS‐CoV‐2 infection in human pluripotent stem cell‐derived cardiomyocytes",
"author": "Choi S.W.",
"doi-asserted-by": "crossref",
"journal-title": "Antiviral Res.",
"key": "e_1_2_10_16_1",
"volume": "184",
"year": "2020"
},
{
"DOI": "10.1038/s41422-020-0282-0",
"article-title": "Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019‐nCoV) in vitro",
"author": "Wang M.",
"doi-asserted-by": "crossref",
"first-page": "269",
"journal-title": "Cell Res.",
"key": "e_1_2_10_17_1",
"volume": "30",
"year": "2020"
},
{
"DOI": "10.1073/pnas.2014441117",
"article-title": "Favipiravir at high doses has potent antiviral activity in SARS‐CoV‐2−infected hamsters, whereas hydroxychloroquine lacks activity",
"author": "Kaptein S.J.F.",
"doi-asserted-by": "crossref",
"first-page": "26955",
"journal-title": "Proc. Natl. Acad. Sci. U.S.A.",
"key": "e_1_2_10_18_1",
"volume": "117",
"year": "2020"
},
{
"DOI": "10.1016/j.ebiom.2021.103595",
"article-title": "The combined treatment of molnupiravir and favipiravir results in a potentiation of antiviral efficacy in a SARS‐CoV‐2 hamster infection model",
"author": "Abdelnabi R.",
"doi-asserted-by": "crossref",
"journal-title": "EBioMedicine",
"key": "e_1_2_10_19_1",
"volume": "72",
"year": "2021"
},
{
"DOI": "10.1038/s41467-021-21992-w",
"article-title": "Favipiravir antiviral efficacy against SARS‐CoV‐2 in a hamster model",
"author": "Driouich J.S.",
"doi-asserted-by": "crossref",
"journal-title": "Nat. Commun.",
"key": "e_1_2_10_20_1",
"volume": "12",
"year": "2021"
},
{
"DOI": "10.1093/infdis/jiac135",
"article-title": "Favipiravir treatment of uncomplicated influenza in adults: results of two phase 3, randomized, double‐blind, placebo‐controlled trials",
"author": "Hayden F.G.",
"doi-asserted-by": "crossref",
"first-page": "1790",
"journal-title": "J. Infect. Dis.",
"key": "e_1_2_10_21_1",
"volume": "226",
"year": "2022"
},
{
"DOI": "10.1111/cts.12827",
"article-title": "Pharmacokinetics of favipiravir in critically ill patients with COVID‐19",
"author": "Irie K.",
"doi-asserted-by": "crossref",
"first-page": "880",
"journal-title": "Clin. Transl. Sci.",
"key": "e_1_2_10_22_1",
"volume": "13",
"year": "2020"
},
{
"DOI": "10.1111/bcp.15227",
"article-title": "Pharmacokinetic characterization of favipiravir in patients with COVID‐19",
"author": "Gülhan R.",
"doi-asserted-by": "crossref",
"first-page": "3516",
"journal-title": "Br. J. Clin. Pharmacol.",
"key": "e_1_2_10_23_1",
"volume": "88",
"year": "2022"
},
{
"DOI": "10.1002/cpt.1909",
"article-title": "Prioritization of anti‐SARS‐Cov‐2 drug repurposing opportunities based on plasma and target site concentrations derived from their established human pharmacokinetics",
"author": "Arshad U.",
"doi-asserted-by": "crossref",
"first-page": "775",
"journal-title": "Clin. Pharmacol. Ther.",
"key": "e_1_2_10_24_1",
"volume": "108",
"year": "2020"
},
{
"DOI": "10.1093/jac/dkab135",
"article-title": "Pharmacokinetic modelling to estimate intracellular favipiravir ribofuranosyl‐5′‐triphosphate exposure to support posology for SARS‐CoV‐2",
"author": "Pertinez H.",
"doi-asserted-by": "crossref",
"first-page": "2121",
"journal-title": "J. Antimicrob. Chemother.",
"key": "e_1_2_10_25_1",
"volume": "76",
"year": "2021"
},
{
"DOI": "10.1128/AAC.01305-16",
"article-title": "Favipiravir pharmacokinetics in nonhuman primates and insights for future efficacy studies of hemorrhagic fever viruses",
"author": "Madelain V.",
"doi-asserted-by": "crossref",
"journal-title": "Antimicrob. Agents Chemother.",
"key": "e_1_2_10_26_1",
"volume": "61",
"year": "2017"
},
{
"DOI": "10.1016/j.antiviral.2017.12.021",
"article-title": "Efficacy of favipiravir (T‐705) in nonhuman primates infected with Ebola virus or Marburg virus",
"author": "Bixler S.L.",
"doi-asserted-by": "crossref",
"first-page": "97",
"journal-title": "Antiviral Res.",
"key": "e_1_2_10_27_1",
"volume": "151",
"year": "2018"
},
{
"key": "e_1_2_10_28_1",
"unstructured": "Sakurai T.A phase I randomized double‐blind placebo‐controlled single ascending‐dose study to evaluate the pharmacokinetics safety and tolerability of injectable favipiravir in healthy subjects<https://jrct.niph.go.jp/en‐latest‐detail/jRCT2071210042>."
},
{
"DOI": "10.1093/jac/dkab318",
"article-title": "Optimal dose and safety of molnupiravir in patients with early SARS‐CoV‐2: a phase I, open‐label, dose‐escalating, randomized controlled study",
"author": "Khoo S.H.",
"doi-asserted-by": "crossref",
"first-page": "3286",
"journal-title": "J. Antimicrob. Chemother.",
"key": "e_1_2_10_29_1",
"volume": "76",
"year": "2021"
},
{
"DOI": "10.1002/cpt.2463",
"article-title": "An open label, adaptive, phase 1 trial of high‐dose oral nitazoxanide in healthy volunteers: an antiviral candidate for SARS‐CoV‐2",
"author": "Walker L.E.",
"doi-asserted-by": "crossref",
"first-page": "585",
"journal-title": "Clin. Pharmacol. Ther.",
"key": "e_1_2_10_30_1",
"volume": "111",
"year": "2022"
},
{
"DOI": "10.1080/10543406.2018.1535503",
"article-title": "Randomized dose‐escalation designs for drug combination cancer trials with immunotherapy",
"author": "Mozgunov P.",
"doi-asserted-by": "crossref",
"first-page": "359",
"journal-title": "J. Biopharm. Stat.",
"key": "e_1_2_10_31_1",
"volume": "29",
"year": "2019"
},
{
"DOI": "10.1177/09622802241288348",
"article-title": "A seamless phase I/II platform design with a time‐to‐event efficacy endpoint for potential COVID‐19 therapies",
"author": "Jaki T.",
"doi-asserted-by": "crossref",
"first-page": "2115",
"journal-title": "Stat. Methods Med. Res.",
"key": "e_1_2_10_32_1",
"volume": "33",
"year": "2024"
},
{
"DOI": "10.1016/j.jpba.2023.115436",
"article-title": "Development and validation of an LC‐MS/MS method for quantification of favipiravir in human plasma",
"author": "Challenger E.",
"doi-asserted-by": "crossref",
"journal-title": "J. Pharm. Biomed. Anal.",
"key": "e_1_2_10_33_1",
"volume": "233",
"year": "2023"
},
{
"DOI": "10.1002/psp4.12445",
"article-title": "Nonlinear mixed‐effects model development and simulation using nlmixr and related R open‐source packages",
"author": "Fidler M.",
"doi-asserted-by": "crossref",
"first-page": "621",
"journal-title": "CPT Pharmacometrics Syst. Pharmacol.",
"key": "e_1_2_10_34_1",
"volume": "8",
"year": "2019"
},
{
"DOI": "10.1002/psp4.12685",
"article-title": "Population pharmacokinetics of favipiravir in patients with COVID‐19",
"author": "Irie K.",
"doi-asserted-by": "crossref",
"first-page": "1161",
"journal-title": "CPT Pharmacometrics Syst. Pharmacol.",
"key": "e_1_2_10_35_1",
"volume": "10",
"year": "2021"
},
{
"DOI": "10.1371/journal.pntd.0005389",
"article-title": "Favipiravir pharmacokinetics in Ebola‐infected patients of the JIKI trial reveals concentrations lower than targeted",
"author": "Nguyen T.H.T.",
"doi-asserted-by": "crossref",
"journal-title": "PLoS Negl. Trop. Dis.",
"key": "e_1_2_10_36_1",
"volume": "11",
"year": "2017"
},
{
"DOI": "10.1016/j.antiviral.2019.04.002",
"article-title": "Cell line‐dependent activation and antiviral activity of T‐1105, the non‐fluorinated analogue of T‐705 (favipiravir)",
"author": "Huchting J.",
"doi-asserted-by": "crossref",
"first-page": "1",
"journal-title": "Antiviral Res.",
"key": "e_1_2_10_37_1",
"volume": "167",
"year": "2019"
},
{
"DOI": "10.1016/j.antiviral.2017.12.020",
"article-title": "Intracellular conversion and in vivo dose response of favipiravir (T‐705) in rodents infected with Ebola virus",
"author": "Bixler S.L.",
"doi-asserted-by": "crossref",
"first-page": "50",
"journal-title": "Antiviral Res.",
"key": "e_1_2_10_38_1",
"volume": "151",
"year": "2018"
}
],
"reference-count": 38,
"references-count": 38,
"relation": {},
"resource": {
"primary": {
"URL": "https://ascpt.onlinelibrary.wiley.com/doi/10.1002/cpt.70261"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "Optimal Dose and Safety of Intravenous Favipiravir in Hospitalized Patients With\n <scp>COVID</scp>\n ‐19: A Dose‐Escalating, Randomized Controlled Phase Ib Study",
"type": "journal-article",
"update-policy": "https://doi.org/10.1002/crossmark_policy"
}