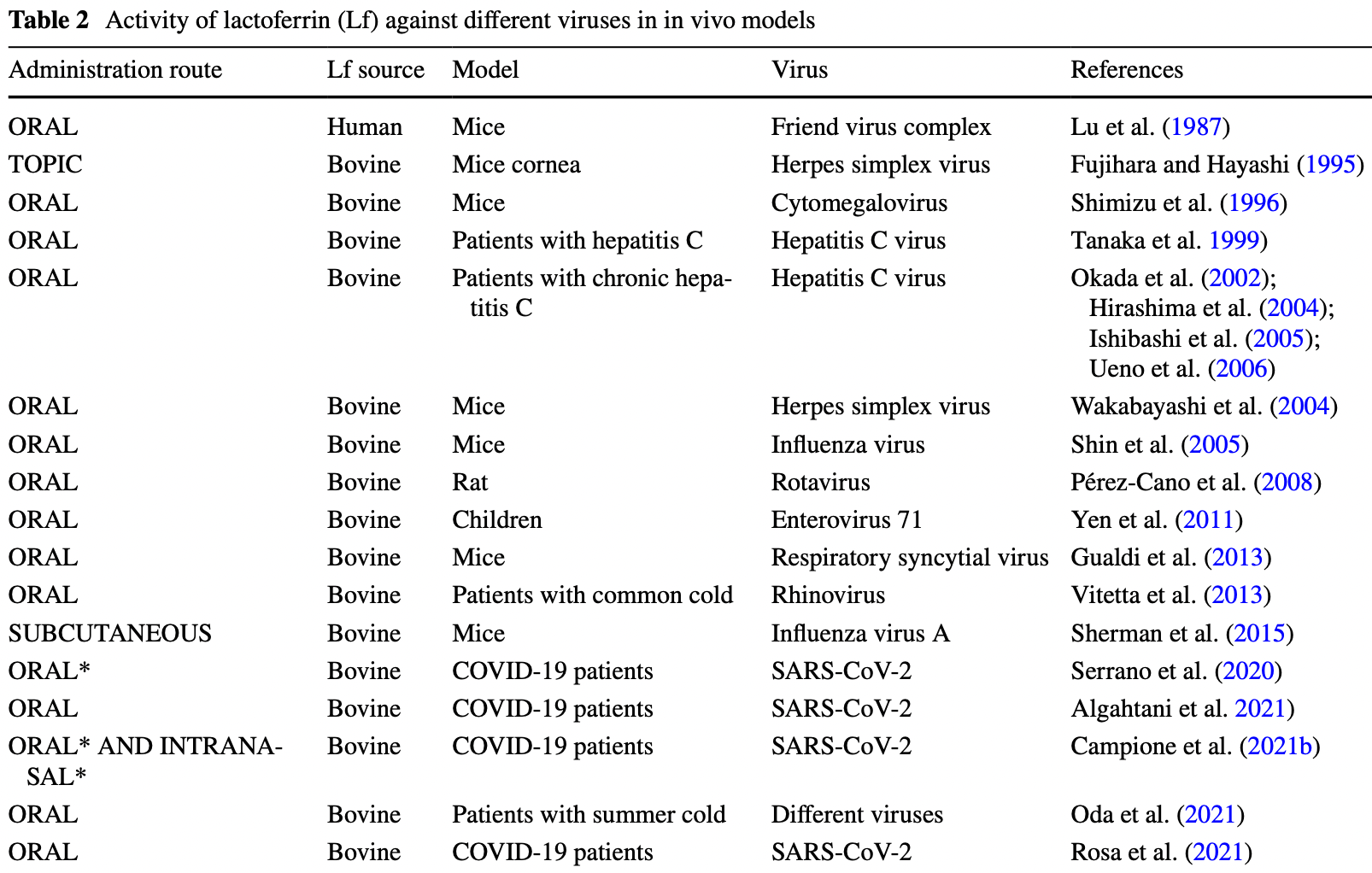
An overview on in vitro and in vivo antiviral activity of lactoferrin: its efficacy against SARS-CoV-2 infection
et al., BioMetals, doi:10.1007/s10534-022-00427-z, Aug 2022
Review of the antiviral activity of lactoferrin and efficacy for SARS-CoV-2.
1.
Palacios-Rosas et al., COVID-19 and Lactoferrin: A Systematic Review and Meta-Analysis, COVID, doi:10.3390/covid5100176.
2.
Eker et al., The potential of lactoferrin as antiviral and immune-modulating agent in viral infectious diseases, Frontiers in Immunology, doi:10.3389/fimmu.2024.1402135.
3.
Manzoni et al., Lactoferrin Supplementation in Preventing and Protecting from SARS-CoV-2 Infection: Is There Any Role in General and Special Populations? An Updated Review of Literature, International Journal of Molecular Sciences, doi:10.3390/ijms251910248.
4.
Rosa et al., An overview on in vitro and in vivo antiviral activity of lactoferrin: its efficacy against SARS-CoV-2 infection, BioMetals, doi:10.1007/s10534-022-00427-z.
5.
Mattar et al., Natural resources to control COVID-19: could lactoferrin amend SARS-CoV-2 infectivity?, PeerJ, doi:10.7717/peerj.11303.
Rosa et al., 3 Aug 2022, peer-reviewed, 6 authors.
Contact: piera.valenti@uniroma1.it.
An overview on in vitro and in vivo antiviral activity of lactoferrin: its efficacy against SARS-CoV-2 infection
BioMetals, doi:10.1007/s10534-022-00427-z
SARS-CoV-2. In vivo, Lf oral administration in early stage of SARS-CoV-2 infection counteracts COVID-19 pathogenesis. In particular, the effect of Lf on SARS-CoV-2 entry, inflammatory homeostasis, iron dysregulation, iron-proteins synthesis, reactive oxygen formation, oxidative stress, gut-lung axis regulation as well as on RNA negativization, and coagulation/fibrinolysis balance will be critically reviewed. Moreover, the molecular mechanisms underneath, including the Lf binding to HSPGs and spike glycoprotein, will be disclosed and discussed. Taken together, present data not only support the application of the oral administration of Lf alone in asymptomatic COVID-19 patients or as adjuvant of standard of care practice in symptomatic ones but also constitute the basis for enriching the limited literature on Lf effectiveness for COVID-19 treatment.
Keywords Lactoferrin • SARS-CoV-2 • COVID-19 • Inflammatory and iron homeostasis • Gut-lung axis • Coagulation and fibrinolysis
Lactoferrin and iron Lactoferrin (Lf), identified in 1939 in bovine milk and isolated in 1960 from both human (Johansson 1960; Montreuil et al. 1960 ) and bovine milk (Groves 1960), is constitutively synthesized by exocrine glands and secreted in human fluids. After induction,
Author contributions P.V.: conceptualization; L.R., A.C., and P.V.: writing: original
Declarations
Conflict of interest The authors declare no conflict of interest. Publisher's Note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations. Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
References
Abobaker, Can iron chelation as an adjunct treatment of COVID-19 improve the clinical outcome?, Eur J Clin Pharmacol, doi:10.1007/s00228-020-02942-9
Actor, Hwang, Kruzel, Lactoferrin as a natural immune modulator, Curr Pharm Des, doi:10.2174/138161209788453202
Al-Samkari, Leaf, Dzik, Carlson, Fogerty et al., COVID-19 and coagulation: bleeding and thrombotic manifestations of SARS-CoV-2 infection, Blood, doi:10.1182/blood.2020006520
Algahtani, Elabbasy, Samak, Adeboye, Yusuf et al., The Prospect of lactoferrin use as adjunctive agent in management of SARS-CoV-2 patients: a randomized pilot study, Medicina (kaunas), doi:10.3390/medicina57080842
Ali, Hasan, Kow, Merchant, Lactoferrin reduces the risk of respiratory tract infections: a meta-analysis of randomized controlled trials, Clin Nutr ESPEN, doi:10.1016/j.clnesp.2021.08.019
Ammendolia, Agamennone, Pietrantoni, Lannutti, Siciliano et al., Bovine lactoferrin-derived peptides as novel broad-spectrum inhibitors of influenza virus, Pathog Glob Health, doi:10.1179/2047773212Y.0000000004
Andrews, Disorders of iron metabolism, N Engl J Med, doi:10.1056/NEJM199912233412607
Baker, Baker, Lactoferrin and iron: structural and dynamic aspects of binding and release, Biometals, doi:10.1023/b:biom.0000027694.40260.70
Bartley, Zhou, Kuchel, Weinstock, Haynes, Impact of age, caloric restriction, and influenza infection on mouse gut microbiome: an exploratory study of the role of age-related microbiome changes on influenza responses, Front Immunol, doi:10.3389/fimmu.2017.01164
Bektas, Schurman, Sen, Ferrucci, Aging, inflammation and the environment, Exp Gerontol, doi:10.1016/j.exger.2017.12.015
Berlutti, Pantanella, Natalizi, Frioni, Paesano et al., Antiviral properties of lactoferrin-a natural immunity molecule, Molecules, doi:10.3390/molecules16086992
Bluard-Deconinck, Masson, Osinski, Heremans, Amino acid sequence of cysteic peptides of lactoferrin and demonstration of similarities between lactoferrin and transferrin, Biochim Biophys Acta, doi:10.1016/0005-2795(74)90002-6
Broz, Dixit, Inflammasomes: mechanism of assembly, regulation and signalling, Nat Rev Immunol, doi:10.1038/nri.2016.58
Budden, Gellatly, Wood, Cooper, Morrison et al., Emerging pathogenic links between microbiota and the gut-lung axis, Nat Rev Microbiol, doi:10.1038/nrmicro.2016.142
Campione, Cosio, Rosa, Lanna, Girolamo et al., Lactoferrin as protective natural barrier of respiratory and intestinal mucosa against coronavirus infection and inflammation, Int J Mol Sci, doi:10.3390/ijms21144903
Campione, Lanna, Cosio, Rosa, Conte et al., Lactoferrin against SARS-CoV-2 in vitro and in silico evidences, Front Pharmacol, doi:10.3389/fphar.2021.666600
Campione, Lanna, Cosio, Rosa, Conte et al., Lactoferrin as antiviral treatment in COVID-19 management: preliminary evidence, Int J Environ Res Public Health, doi:10.3390/ijerph182010985
Chang, Ng, Sun, Lactoferrin as potential preventative and adjunct treatment for COVID-19, Int J Antimicrob Agents, doi:10.1016/j.ijantimicag.2020.106118
Cheng, Li, Li, Liu, Yan et al., Ferritin in the coronavirus disease 2019 (COVID-19): a systematic review and meta-analysis, J Clin Lab Anal, doi:10.1002/jcla.23618
Coffey, Ganz, Iron homeostasis-an anthropocentric perspective, J Biol Chem, doi:10.1074/jbc.R117.781823
Cui, Chen, Li, Liu, Wang, Prevalence of venous thromboembolism in patients with severe novel coronavirus pneumonia, J Thromb Haemost, doi:10.1111/jth.14830
Cutone, Frioni, Berlutti, Valenti, Musci et al., Lactoferrin prevents LPS-induced decrease of the iron exporter ferroportin in human monocytes/macrophages, Biometals, doi:10.1007/s10534-014-9742-7
Cutone, Lepanto, Rosa, Scotti, Rossi et al., Aerosolized bovine lactoferrin counteracts infection, inflammation and iron dysbalance in a cystic fibrosis mouse model of Pseudomonas aeruginosa chronic lung infection, Int J Mol Sci, doi:10.3390/ijms20092128
Cutone, Rosa, Lepanto, Scotti, Berlutti et al., Lactoferrin efficiently counteracts the inflammation-induced changes of the iron homeostasis system in macrophages, Front Immunol, doi:10.3389/fimmu.2017.00705
Dai, Pan, Li, Liu, Zhang et al., The antimicrobial cathelicidin peptide hlF(1-11) attenuates alveolar macrophage pyroptosis induced by Acinetobacter baumannii in vivo, Exp Cell Res, doi:10.1016/j.yexcr.2018.01.035
Dai, Teng, Wu, The Antiviral roles of hydrogen sulfide by blocking the interaction between SARS-CoV-2 and its potential cell surface receptors, Oxid Med Cell Longev, doi:10.1155/2021/7866992
Denani, Real-Hohn, De Carvalho, Gomes, Gonçalves, Lactoferrin affects rhinovirus B-14 entry into H1-HeLa cells, Arch Virol, doi:10.1007/s00705-021-04993-4
Deriu, Boxx, He, Pan, Benavidez et al., Influenza virus affects intestinal microbiota and secondary Salmonella infection in the gut through Type I Interferons, PloS Pathog, doi:10.1371/journal.ppat.1005572
Di, Cutone, Polticelli, Rosa, Lepanto et al., The ferroportinceruloplasmin system and the mammalian iron homeostasis machine: regulatory pathways and the role of lactoferrin, Biometals, doi:10.1007/s10534-018-0087-5
Donovan, Lima, Pinkus, Pinkus, Zon et al., The iron exporter ferroportin/Slc40a1 is essential for iron homeostasis, Cell Metab, doi:10.1016/j.cmet.2005.01.003
Drakesmith, Prentice, Viral infection and iron metabolism, Nat Rev Microbiol, doi:10.1038/nrmicro1930
Ehsani, COVID-19 and iron dysregulation: distant sequence similarity between hepcidin and the novel coronavirus spike glycoprotein, Biol Direct, doi:10.1186/s13062-020-00275-2
Enaud, Prevel, Ciarlo, Beaufils, Wieërs et al., The gut-lung axis in health and respiratory diseases: a place for inter-organ and inter-kingdom crosstalks, Front Cell Infect Microbiol, doi:10.3389/fcimb.2020.00009
Fda, GRN 000465 Cow's Milk-Derived Lactoferrin
Fernandez-Musoles, Salom, Martínez-Maqueda, López-Díez, Recio et al., Antihypertensive effects of lactoferrin hydrolyzates: inhibition of angiotensin-and endothelin-converting enzymes, Food Chem, doi:10.1016/j.foodchem.2012.12.049
Figueroa-Lozano, Valk-Weeber, Van Leeuwen, Dijkhuizen, De Vos, Dietary N-Glycans from bovine lactoferrin and TLR modulation, Mol Nutr Food Res, doi:10.1002/mnfr.201700389
Fox, Akmatbekov, Harbert, Li, Brown et al., Pulmonary and cardiac pathology in African American patients with COVID-19: an Vol.: (0123456789) autopsy series from New Orleans, Lancet Respir Med, doi:10.1016/S2213-2600(20)30243-5
Frazer, Anderson, The orchestration of body iron intake: How and where do enterocytes receive their cues?, Blood Cells Mol Dis, doi:10.1016/s1079-9796(03)00039-1
Frioni, Conte, Cutone, Longhi, Musci et al., Lactoferrin differently modulates the inflammatory response in epithelial models mimicking human inflammatory and infectious diseases, Biometals, doi:10.1007/s10534-014-9740-9
Fujihara, Hayashi, Lactoferrin inhibits herpes simplex virus type-1 (HSV-1) infection to mouse cornea, Arch Virol, doi:10.1007/BF01322673
Furlund, Kristoffersen, Devold, Vegarud, Jonassen, Bovine lactoferrin digested with human gastrointestinal enzymes inhibits replication of human echovirus 5 in cell culture, Nutr Res, doi:10.1016/j.nutres.2012.06.006
Furmanski, Li, Fortuna, Swamy, Das, Multiple molecular forms of human lactoferrin. Identification of a class of lactoferrins that possess ribonuclease activity and lack iron-binding capacity, J Exp Med, doi:10.1084/jem.170.2.415
Giobbe, Bonfante, Jones, Gagliano, Luni et al., SARS-CoV-2 infection and replication in human gastric organoids, Nat Commun, doi:10.1038/s41467-021-26762-2
Grier, Mcdavid, Wang, Qiu, Java et al., Neonatal gut and respiratory microbiota: coordinated development through time and space, Microbiome, doi:10.1186/s40168-018-0566-5
Groot, Geijtenbeek, Sanders, Baldwin, Sanchez-Hernandez et al., Lactoferrin prevents dendritic cell-mediated human immunodeficiency virus type 1 transmission by blocking the DC-SIGN-gp120 interaction, J Virol, doi:10.1128/JVI.79.5.3009-3015.2005
Groves, Cuthbertson, James, Moffatt, Cox et al., Respiratory disease following viral lung infection alters the murine gut microbiota, Front Immunol, doi:10.3389/fimmu.2018.00182
Groves, The isolation of a red protein from milk, J Am Chem Soc
Gualdi, Mertz, Gomez, Ramilo, Wittke et al., Lack of effect of bovine lactoferrin in respiratory syncytial virus replication and clinical disease severity in the mouse model, Antiviral Res, doi:10.1016/j.antiviral.2013.05.013
Hanada, Pirzadeh, Carver, Deng, Respiratory viral infection-induced microbiome alterations and secondary bacterial pneumonia, Front Immunol, doi:10.3389/fimmu.2018.02640
Hartenian, Nandakumar, Ly, Tucker, Glaunsinger, The molecular virology of coronaviruses, J Biol Chem, doi:10.1074/jbc.REV120.013930
Hatmal, Alshaer, Al-Hatamleh, Hatmal, Smadi et al., Comprehensive structural and molecular comparison of spike proteins of SARS-CoV-2, SARS-CoV and MERS-CoV, and their interactions with ACE2, Cells, doi:10.3390/cells9122638
Hirashima, Orito, Ohba, Kondo, Sakamoto et al., A randomized controlled trial of consensus interferon with or without lactoferrin for chronic hepatitis C patients with genotype 1b and high viral load, Hepatol Res, doi:10.1016/j.hepres.2004.01.002
Hu, Meng, Zhang, Xiang, Wang, The in vitro antiviral activity of lactoferrin against common human coronaviruses and SARS-CoV-2 is mediated by targeting the heparan sulfate co-receptor, Emerg Microbes Infect, doi:10.1080/22221751.2021.1888660
Hunter, Fulton, Ganz, Vogel, The solution structure of human hepcidin, a peptide hormone with antimicrobial activity that is involved in iron uptake and hereditary hemochromatosis, J Biol Chem, doi:10.1074/jbc.M205305200
Hussain, Cher, Abid, Abid, Role of gut microbiome in COVID-19: an insight into pathogenesis and therapeutic potential, Front Immunol, doi:10.3389/fimmu.2021.765965
Ichinohe, Pang, Kumamoto, Peaper, Ho et al., Microbiota regulates immune defense against respiratory tract Influenza A virus infection, Proc Natl Acad Sci U S A, doi:10.1073/pnas.1019378108
Ishibashi, Takeda, Tsukidate, Miyazaki, Ohira et al., Randomized placebo-controlled trial of interferon alpha-2b plus ribavirin with and without lactoferrin for chronic hepatitis C, Hepatol Res, doi:10.1016/j.hepres.2005.03.018
Johansson, Isolation of an iron-containing red protein from human milk, Acta Chem Scand
Kalashnyk, Petrova, Lykhmus, Mikhalovska, Mikhalovsky et al., Expression, function and cooperating partners of protease-activated receptor type 3 in vascular endothelial cells and B lymphocytes studied with specific monoclonal antibody, Mol Immunol, doi:10.1016/j.molimm.2012.12.021
Ke, Oton, Qu, Cortese, Zila et al., Structures and distributions of SARS-CoV-2 spike proteins on intact virions, Nature, doi:10.1038/s41586-020-2665-2
Kell, Heyden, Pretorius, The Biology of lactoferrin, an iron-binding protein that can help defend against viruses and bacteria, Front Immunol, doi:10.3389/fimmu.2020.01221
Klok, Kruip, Van Der Meer, Arbous, Gommers et al., Incidence of thrombotic complications in critically ill ICU patients with COVID-19, Thromb Res, doi:10.1016/j.thromres.2020.04.013
Krause, Neitz, Magert, Schulz, Forssmann et al., LEAP-1, a novel highly disulfidebonded human peptide, exhibits antimicrobial activity, FEBS Lett, doi:10.1016/s0014-5793(00)01920-7
Kruzel, Actor, Boldogh, Zimecki, Lactoferrin in health and disease, Postepy Hig Med Dosw
Kruzel, Zimecki, Actor, Lactoferrin in a context of inflammation-induced pathology, Front Immunol, doi:10.3389/fimmu.2017.01438
Kuhara, Yamauchi, Tamura, Okamura, Oral administration of lactoferrin increases NK cell activity in mice via increased production of IL-18 and type I IFN in the small intestine, J Interferon Cytokine Res, doi:10.1089/jir.2006.26.489
Lang, Yang, Deng, Liu, Yang et al., Inhibition of SARS pseudovirus cell entry by lactoferrin binding to heparan sulfate proteoglycans, PLoS ONE, doi:10.1371/journal.pone.0023710
Latz, Xiao, Stutz, Activation and regulation of the inflammasomes, Nat Rev Immunol, doi:10.1038/nri3452
Lee, Peng, Gelbart, Wang, Beutler, Regulation of hepcidin transcription by interleukin-1 and interleukin-6, Proc Natl Acad Sci, doi:10.1073/pnas.0409808102
Legrand, Elass, Carpentier, Mazurier, Lactoferrin: a modulator of immune and inflammatory responses, Cell Mol Life Sci, doi:10.1007/s00018-005-5370-2
Legrand, Mazurier, A critical review of the roles of host lactoferrin in immunity, Biometals, doi:10.1007/s10534-010-9297-1
Lepanto, Rosa, Cutone, Conte, Paesano et al., Efficacy of lactoferrin oral administration in the treatment of anemia and anemia of inflammation in pregnant and non-pregnant women: an interventional study, Front Immunol, doi:10.3389/fimmu.2018.02123
Leveugle, Mazurier, Legrand, Mazurier, Montreuil et al., Lactotransferrin binding to its platelet receptor inhibits platelet aggregation, Eur J Biochem, doi:10.1111/j.1432-1033.1993.tb17871.x
Lin, Chu, Chiu, Lactoferrin inhibits enterovirus 71 infection of human embryonal rhabdomyosarcoma cells in vitro, J Infect Dis, doi:10.1086/343809
Liu, Philippou, Kolokotroni, Siakallis, Rahima et al., Gut and airway microbiota and their role in COVID-19 infection and pathogenesis: a scoping review, Infection, doi:10.1007/s15010-021-01715-5
Longstaff, Kolev, Basic mechanisms and regulation of fibrinolysis, J Thromb Haemost, doi:10.1111/jth.12935
Lu, Hangoc, Oliff, Chen, Shen et al., Protective influence of lactoferrin on mice infected with the polycythemia-inducing strain of Friend virus complex, Cancer Res
Mancinelli, Rosa, Cutone, Lepanto, Franchitto et al., Viral hepatitis and iron dysregulation: molecular pathways and the role of lactoferrin, Molecules, doi:10.3390/molecules25081997
Mangan, Olhava, Roush, Seidel, Glick et al., Targeting the NLRP3 inflammasome in inflammatory diseases, Nat Rev Drug Discov, doi:10.1038/nrd.2018.149
Marchetti, Ammendolia, Superti, Glycosaminoglycans are not indispensable for the anti-herpes simplex virus type 2 activity of lactoferrin, Biochimie, doi:10.1016/j.biochi.2008.04.015
Marchetti, Longhi, Conte, Pisani, Valenti et al., Lactoferrin inhibits herpes simplex virus type 1 adsorption to Vero cells, Antiviral Res, doi:10.1016/0166-3542(95)00840-3
Marchetti, Pisani, Antonini, Valenti, Seganti et al., Metal complexes of bovine lactoferrin inhibit in vitro replication of herpes simplex virus type 1 and 2, Biometals, doi:10.1023/a:1009217709851
Marchetti, Superti, Ammendolia, Rossi, Valenti et al., Inhibition of poliovirus type 1 infection by iron-, manganese-and zinc-saturated lactoferrin, Med Microbiol Immunol, doi:10.1007/s004300050093
Miller, Iron deficiency anemia: A common and curable disease, Cold Spring Harb Perspect Med, doi:10.1101/cshperspect.a011866
Miotto, Rienzo, Bò, Boffi, Ruocco et al., Molecular mechanisms behind anti SARS-CoV-2 action of lactoferrin, Front Mol Biosci, doi:10.3389/fmolb.2021.607443
Mirabelli, Wotring, Zhang, Mccarty, Fursmidt et al., Morphological cell profiling of SARS-CoV-2 infection identifies drug repurposing candidates for COVID-19, Proc Natl Acad Sci U S A, doi:10.1073/pnas.2105815118
Montreuil, Tonnelat, Mullet, Preparation and properties of lactosiderophilin (lactotransferrin) of human milk, Biochim Biophys Acta
Motoki, Mizuki, Tsukahara, Miyakawa, Kubo et al., Effects of lactoferrin-fortified formula on acute gastrointestinal symptoms in children aged 12-32 months: a randomized, double-blind, Placebo-Controlled Trial Front Pediatr, doi:10.3389/fped.2020.00233
Nai, Lorè, Pagani, Lorenzo, Modica et al., Hepcidin levels predict Covid-19 severity and mortality in a cohort of hospitalized Italian patients, Am J Hematol, doi:10.1002/ajh.26027
Naidu, Clemens, Pressman, Zaigham, Davies et al., COVID-19 during pregnancy and postpartum, J Diet Suppl, doi:10.1080/19390211.2020.1834047
Nemeth, Rivera, Gabayan, Keller, Taudorf et al., IL-6 mediates hypoferremia of inflammation by inducing the synthesis of the iron regulatory hormone hepcidin, J Clin Investig, doi:10.1172/JCI20945
Ng, Cheung, Wong, Wang, Ip et al., Antiviral activities of whey proteins, Appl Microbiol Biotechnol, doi:10.1007/s00253-015-6818-4
Oda, Wakabayashi, Tanaka, Yamauchi, Sugita et al., Effects of lactoferrin on infectious diseases in Japanese summer: a randomized, double-blinded, placebo-controlled trial, J Microbiol Immunol Infect, doi:10.1016/j.jmii.2020.02.010
Okada, Tanaka, Sato, Ueno, Saito et al., Dose-response trial of lactoferrin in patients with chronic hepatitis C, Jpn J Cancer Res, doi:10.1111/j.1349-7006.2002.tb02484.x
Paesano, Berlutti, Pietropaoli, Goolsbee, Pacifici et al., Lactoferrin efficacy versus ferrous sulfate in curing iron disorders in pregnant and non-pregnant women, Int J Immunopathol Pharmacol, doi:10.1177/039463201002300220
Paesano, Natalizi, Berlutti, Valenti, Body iron delocalization: the serious drawback in iron disorders in both developing and developed countries, Pathog Glob Health, doi:10.1179/2047773212Y.0000000043
Pan, Shen, Yu, Ge, Chen et al., SARS-CoV-2 N protein promotes NLRP3 inflammasome activation to induce hyperinflammation, J Biol Chem, doi:10.1074/jbc.M008922200
Pan, Zhang, Liu, Wang, Lao et al., Dengue virus M protein promotes NLRP3 Inflammasome activation to induce vascular leakage in mice, J Virol, doi:10.1128/JVI.00996-19
Perricone, Bartoloni, Bursi, Cafaro, Guidelli et al., COVID-19 as part of the hyperferritinemic syndromes: the role of iron depletion therapy, Immunol Res, doi:10.1007/s12026-020-09145-5
Pietrantoni, Ammendolia, Superti, Bovine lactoferrin: involvement of metal saturation and carbohydrates in the inhibition of influenza virus infection, Biochem Cell Biol, doi:10.1139/o11-072
Pietrantoni, Biase, Tinari, Marchetti, Valenti et al., Bovine lactoferrin inhibits adenovirus infection by interacting with viral structural polypeptides, Antimicrob Agents Chemother, doi:10.1128/AAC.47.8.2688-2691.2003
Pietrantoni, Dofrelli, Tinari, Ammendolia, Puzelli et al., Bovine lactoferrin inhibits influenza A virus induced programmed cell death in vitro, Biometals, doi:10.1007/s10534-010-9323-3
Puddu, Borghi, Gessani, Valenti, Belardelli et al., Antiviral effect of bovine lactoferrin saturated with metal ions on early steps of human immunodeficiency virus type 1 infection, Int J Biochem Cell Biol, doi:10.1016/s1357-2725(98)00066-1
Puddu, Latorre, Carollo, Catizone, Ricci et al., Bovine lactoferrin counteracts toll-like receptor mediated activation signals in antigen presenting cells, PLoS ONE, doi:10.1371/journal.pone.0022504
Puddu, Valenti, Gessani, Immunomodulatory effects of lactoferrin on antigen presenting cells, Biochimie, doi:10.1016/j.biochi.2008.05.005
Pérez-Cano, Marín-Gallén, Castell, Rodríguez-Palmero, Rivero et al., Supplementing suckling rats with whey protein concentrate modulates the immune response and ameliorates rat rotavirus-induced diarrhea, Nutr, doi:10.3945/jn.108.093856
Qian, Jollès, Migliore-Samour, Fiat, Isolation and characterization of sheep lactoferrin, an inhibitor of platelet aggregation and comparison with human lactoferrin, Biochim Biophys Acta, doi:10.1016/0304-4165(94)00126-i
Qiao, Sugianto, Fung, Del-Castillo-Rueda, Moran-Jimenez et al., Hepcidin-induced endocytosis of ferroportin is dependent Vol:. (1234567890) on ferroportin ubiquitination, Cell Metab, doi:10.1016/j.cmet.2012.03.018
Rosa, Cutone, Lepanto, Paesano, Valenti, Lactoferrin: a natural glycoprotein involved in iron and inflammatory homeostasis, Int J Mol Sci, doi:10.3390/ijms18091985
Rosa, Tripepi, Naldi, Aimati, Santangeli et al., Ambulatory COVID-19 patients treated with lactoferrin as a supplementary antiviral agent: a preliminary study, J Clin Med, doi:10.3390/jcm10184276
Sano, Nagai, Tsutsumi, Kuroki, Lactoferrin and surfactant protein a exhibit distinct binding specificity to F protein and differently modulate respiratory syncytial virus infection, Eur J Immunol, doi:10.1002/eji.200324218
Serrano, Kochergina, Albors, Diaz, Oroval et al., Liposomal lactoferrin as potential preventative and cure for COVID-19, Int J Res Health Sci, doi:10.5530/ijrhs.8.1.3
Sherman, Pritzl, Xia, Miller, Zaghouani et al., Lactoferrin acts as an adjuvant during influenza vaccination of neonatal mice, Biochem Biophys Res Commun, doi:10.1016/j.bbrc.2015.10.067
Shimizu, Matsuzawa, Okada, Tazume, Dosako et al., Lactoferrinmediated protection of the host from murine cytomegalovirus infection by a T-cell-dependent augmentation of natural killer cell activity, Arch Virol, doi:10.1007/BF01718201
Shin, Wakabayashi, Yamauchi, Teraguchi, Tamura et al., Effects of orally administered bovine lactoferrin and lactoperoxidase on influenza virus infection in mice, J Med Microbiol, doi:10.1099/jmm.0.46018-0
Sienkiewicz, Jaśkiewicz, Tarasiuk, Fichna, Lactoferrin: an overview of its main functions, immunomodulatory and antimicrobial role, and clinical significance, Crit Rev Food Sci Nutr, doi:10.1080/10408398.2021.1895063
Simpson, Aging and inflammation: directing traffic through physical activity, Brain Behav Immun, doi:10.1016/j.bbi.2016.05.015
Skendros, Mitsios, Chrysanthopoulou, Mastellos, Metallidis et al., Complement and tissue factor-enriched neutrophil extracellular traps are key drivers in COVID-19 immunothrombosis, J Clin Invest, doi:10.1172/JCI141374
Spik, Coddeville, Mazurier, Bourne, Cambillaut et al., Primary and three-dimensional structure of lactotransferrin (lactoferrin) glycans, Adv Exp Med Biol, doi:10.1007/978-1-4615-2548-6_3
Stefanescu, Hétu, Slaughter, Shea, Shetty, A pilot study of Biotene OralBalance® gel for oral care in mechanically ventilated preterm neonates, Contemp Clin Trials, doi:10.1016/j.cct.2013.03.010
Superti, Ammendolia, Valenti, Seganti, Antirotaviral activity of milk proteins: lactoferrin prevents rotavirus infection in the enterocyte-like cell line HT-29, Med Microbiol Immunol, doi:10.1007/s004300050049
Superti, Lactoferrin from bovine milk: a protective companion for Life, Nutrients, doi:10.3390/nu12092562
Superti, Siciliano, Rega, Giansanti, Valenti et al., Involvement of bovine lactoferrin metal saturation, sialic acid and protein fragments in the inhibition of rotavirus infection, Biochim Biophys Acta, doi:10.1016/s0304-4165(01)00178-7
Swart, Kuipers, Smit, Pauwels, Debéthune et al., Antiviral effects of milk proteins: acylation results in polyanionic compounds with potent activity against human immunodeficiency virus types 1 and 2 in vitro, AIDS Res Hum Retroviruses, doi:10.1089/aid.1996.12.769
Szabo, Petrasek, Inflammasome activation and function in liver disease, Nat Rev Gastroenterol Hepatol, doi:10.1038/nrgastro.2015.94
Taha, Mehrez, Sitohy, Dawood, Hamid et al., Effectiveness of esterified whey proteins fractions against Egyptian Lethal Avian Influenza A (H5N1), Virol J, doi:10.1186/1743-422X-7-330
Tanaka, Ikeda, Nozaki, Kato, Tsuda et al., Lactoferrin inhibits hepatitis C virus viremia in patients with chronic hepatitis C: a pilot study, Jpn J Cancer Res, doi:10.1111/j.1349-7006.1999.tb00756.x
Tang, Yang, Duan, Liao, Liu et al., Transferrin receptor is another receptor for SARS-CoV-2 entry, bioRxiv, doi:10.1101/2020.10.23.350348
Tian, Hu, Niu, Liu, Xu et al., Pulmonary pathology of early-phase 2019 novel Coronavirus (COVID-19) Pneumonia in two patients with lung cancer, J Thorac Oncol, doi:10.1016/j.jtho.2020.02.010
Tinari, Pietrantoni, Ammendolia, Valenti, Superti, Inhibitory activity of bovine lactoferrin against echovirus induced programmed cell death in vitro, Int J Antimicrob Agents, doi:10.1016/j.ijantimicag.2005.02.011
Toldo, Bussani, Nuzzi, Bonaventura, Mauro et al., Inflammasome formation in the lungs of patients with fatal COVID-19, Inflamm Res, doi:10.1007/s00011-020-01413-2
Ueno, Sato, Yamamoto, Tanaka, Ohkawa et al., Randomized, double-blind, placebo-controlled trial of bovine lactoferrin in patients with chronic
Valenti, Antonini, Lactoferrin: an important host defence against microbial and viral attack, Cell Mol Life Sci, doi:10.1007/s00018-005-5372-0
Valenti, Rosa, Capobianco, Lepanto, Schiavi et al., Role of Lactobacilli and Lactoferrin in the Mucosal Cervicovaginal Defense, Front Immunol, doi:10.3389/fimmu.2018.00376
Van Splunter, Perdijk, Fick-Brinkhof, Feitsma, Vollenbroek et al., Bovine lactoferrin enhances TLR7-mediated responses in plasmacytoid dendritic cells in elderly women: results from a nutritional intervention study with bovine lactoferrin, GOS Vitamin d Front Immunol, doi:10.3389/fimmu.2018.02677
Van Veen, Geerts, Van Berkel, Nuijens, The role of N-linked glycosylation in the protection of human and bovine lactoferrin against tryptic proteolysis, Eur J Biochem, doi:10.1111/j.1432-1033.2003.03965.x
Verga Falzacappa, Spasic, Kessler, Stolte, Hentze et al., STAT3 mediates hepatic hepcidin expression and its inflammatory stimulation, Blood, doi:10.1182/blood-2006-07-033969
Vitetta, Coulson, Beck, Gramotnev, Du et al., The clinical efficacy of a bovine lactoferrin/whey protein Ig-rich fraction (Lf/IgF) for the common cold: a double blind randomized study, Complement Ther Med, doi:10.1016/j.ctim.2012.12.006
Vlahakos, Marathias, Arkadopoulos, Vlahakos, Hyperferritinemia in patients with COVID-19: an opportunity for iron chelation?, Artif Organs, doi:10.1111/aor.13812
Wakabayashi, Kurokawa, Shin, Teraguchi, Tamura et al., Oral lactoferrin prevents body weight loss and increases cytokine responses during herpes simplex virus type 1 infection of mice, Biosci Biotechnol Biochem, doi:10.1271/bbb.68.537
Wakabayashi, Oda, Yamauchi, Abe, Lactoferrin for prevention of common viral infections, J Infect Chemother, doi:10.1016/j.jiac.2014.08.003
Wan, Du, Hong, Chen, Que et al., Neurological complications and infection mechanism of SARS-COV-2, Signal Transduct Target Ther, doi:10.1038/s41392-021-00818-7
Wang, Jiang, Yan, Gong, Han et al., RNA viruses promote activation of the NLRP3 inflammasome through a RIP1-RIP3-DRP1 signaling pathway, Nat Immunol, doi:10.1038/ni.3015
Wang, Li, Wei, Lian, Sun et al., Respiratory influenza virus infection induces intestinal immune injury via microbiota-mediated Th17 cell-dependent inflammation, J Exp Med, doi:10.1084/jem.20140625
Wang, Yue, Dang, Yang, Chen et al., Role of sialylated glycans on bovine lactoferrin against influenza virus, Glycoconjugate J, doi:10.1007/s10719-021-10029-5
Wang, Zhang, Wu, Niu, Song et al., Structural and functional basis of SARS-CoV-2 entry by using human ACE2, Cell, doi:10.1016/j.cell.2020.03.045
Welsh, Hwang, Boyd, Kruzel, Hunter et al., Influence of oral lactoferrin on Mycobacterium tuberculosis induced immunopathology, Tuberculosis, doi:10.1016/j.tube.2011.10.019
Wenzhong, Hualan, COVID-19: captures iron and generates reactive oxygen species to damage the human immune system, Autoimmunity, doi:10.1080/08916934.2021.1913581
Wisgrill, Wessely, Spittler, Förster-Waldl, Berger et al., Human lactoferrin attenuates the proinflammatory response of neonatal monocyte-derived macrophages, Clin Exp Immunol, doi:10.1111/cei.13108
Wrighting, Andrews, Interleukin-6 induces hepcidin expression through STAT3, Blood, doi:10.1182/blood-2006-06-027631
Wu, Guo, Tang, Hong, Zhou et al., Prolonged presence of SARS-CoV-2 viral RNA in faecal samples, Lancet Gastroenterol Hepatol, doi:10.1016/S2468-1253(20)30083-2
Wu, Monroe, Church, Characterization of the glycosaminoglycan-binding region of lactoferrin, Arch Biochem Biophys, doi:10.1006/abbi.1995.1139
Wu, Zhao, Yu, Chen, Song et al., A new coronavirus associated with human respiratory disease in China, Nature, doi:10.1038/s41586-020-2008-3
Xu, Fan, Liu, Cheng, Tu et al., Novel anticoagulant peptide from lactoferrin binding thrombin at the active site and Exosite-I, J Agric Food Chem, doi:10.1021/acs.jafc.9b08094
Xu, Shi, Wang, Zhang, Huang et al., Pathological findings of COVID-19 associated with acute respiratory distress syndrome, Lancet Respir Med, doi:10.1016/S2213-2600(20)30076-X
Yamamoto, Ura, Tanemura, Koyama, Takano et al., Inhibitory effect of bovine lactoferrin on human parainfluenza virus type 2 infection, J Health Sci, doi:10.1248/jhs.56.613
Yen, Chiu, Huang, Lin, Effects of lactoferrin-containing formula in the prevention of enterovirus and rotavirus infection and impact on serum cytokine levels: a randomized trial, Chang Gung Med J
Yi, Kaneko, Yu, Murakami, Hepatitis C virus envelope proteins bind lactoferrin, J Virol, doi:10.1128/JVI.71.8.5997-6002.1997
Yildiz, Mazel-Sanchez, Kandasamy, Manicassamy, Schmolke, Influenza A virus infection impacts systemic microbiota dynamics and causes quantitative enteric dysbiosis, Microbiome, doi:10.1186/s40168-017-0386-z
Zhu, Zhang, Li, Yang, Song et al., China Novel Coronavirus Investigating and Research Team (2020) A novel coronavirus from patients with pneumonia in China, 2019, N Engl J Med, doi:10.1056/NEJMoa2001017
Zimecki, Actor, Kruzel, The potential for Lactoferrin to reduce SARS-CoV-2 induced cytokine storm, Int Immunopharmacol, doi:10.1016/j.intimp.2021.107571
Zumerle, Mathieu, Delga, Heinis, Viatte et al., Targeted disruption of hepcidin in the liver recapitulates the hemochromatotic phenotype, Blood, doi:10.1182/blood-2014-01-550467
Zuo, Warnock, Harbaugh, Yalavarthi, Gockman et al., Plasma tissue plasminogen activator and plasminogen activator inhibitor-1 in hospitalized COVID-19 patients, Sci Rep, doi:10.1038/s41598-020-80010-z
Zwirzitz, Reiter, Skrabana, Ohradanova-Repic, Majdic et al., Lactoferrin is a natural inhibitor of plasminogen activation, J Biol Chem, doi:10.1074/jbc.RA118.003145
DOI record:
{
"DOI": "10.1007/s10534-022-00427-z",
"ISSN": [
"0966-0844",
"1572-8773"
],
"URL": "http://dx.doi.org/10.1007/s10534-022-00427-z",
"alternative-id": [
"427"
],
"assertion": [
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "Received",
"name": "received",
"order": 1,
"value": "14 April 2022"
},
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "Accepted",
"name": "accepted",
"order": 2,
"value": "26 July 2022"
},
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "First Online",
"name": "first_online",
"order": 3,
"value": "3 August 2022"
},
{
"group": {
"label": "Declarations",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 1
},
{
"group": {
"label": "Conflict of interest",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 2,
"value": "The authors declare no conflict of interest."
},
{
"label": "Free to read",
"name": "free",
"value": "This content has been made available to all."
}
],
"author": [
{
"affiliation": [],
"family": "Rosa",
"given": "Luigi",
"sequence": "first"
},
{
"affiliation": [],
"family": "Cutone",
"given": "Antimo",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Conte",
"given": "Maria Pia",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Campione",
"given": "Elena",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Bianchi",
"given": "Luca",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-4354-3190",
"affiliation": [],
"authenticated-orcid": false,
"family": "Valenti",
"given": "Piera",
"sequence": "additional"
}
],
"container-title": "BioMetals",
"container-title-short": "Biometals",
"content-domain": {
"crossmark-restriction": false,
"domain": [
"link.springer.com"
]
},
"created": {
"date-parts": [
[
2022,
8,
3
]
],
"date-time": "2022-08-03T11:03:19Z",
"timestamp": 1659524599000
},
"deposited": {
"date-parts": [
[
2022,
8,
9
]
],
"date-time": "2022-08-09T16:07:54Z",
"timestamp": 1660061274000
},
"indexed": {
"date-parts": [
[
2022,
8,
9
]
],
"date-time": "2022-08-09T16:41:23Z",
"timestamp": 1660063283179
},
"is-referenced-by-count": 0,
"issued": {
"date-parts": [
[
2022,
8,
3
]
]
},
"language": "en",
"license": [
{
"URL": "https://www.springer.com/tdm",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2022,
8,
3
]
],
"date-time": "2022-08-03T00:00:00Z",
"timestamp": 1659484800000
}
},
{
"URL": "https://www.springer.com/tdm",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2022,
8,
3
]
],
"date-time": "2022-08-03T00:00:00Z",
"timestamp": 1659484800000
}
}
],
"link": [
{
"URL": "https://link.springer.com/content/pdf/10.1007/s10534-022-00427-z.pdf",
"content-type": "application/pdf",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://link.springer.com/article/10.1007/s10534-022-00427-z/fulltext.html",
"content-type": "text/html",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://link.springer.com/content/pdf/10.1007/s10534-022-00427-z.pdf",
"content-type": "application/pdf",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "297",
"original-title": [],
"prefix": "10.1007",
"published": {
"date-parts": [
[
2022,
8,
3
]
]
},
"published-online": {
"date-parts": [
[
2022,
8,
3
]
]
},
"publisher": "Springer Science and Business Media LLC",
"reference": [
{
"DOI": "10.1007/s00228-020-02942-9",
"author": "A Abobaker",
"doi-asserted-by": "publisher",
"first-page": "1619",
"issue": "11",
"journal-title": "Eur J Clin Pharmacol",
"key": "427_CR1",
"unstructured": "Abobaker A (2020) Can iron chelation as an adjunct treatment of COVID-19 improve the clinical outcome? Eur J Clin Pharmacol 76(11):1619–1620. https://doi.org/10.1007/s00228-020-02942-9",
"volume": "76",
"year": "2020"
},
{
"DOI": "10.2174/138161209788453202",
"author": "JK Actor",
"doi-asserted-by": "publisher",
"first-page": "1956",
"issue": "17",
"journal-title": "Curr Pharm Des",
"key": "427_CR2",
"unstructured": "Actor JK, Hwang SA, Kruzel ML (2009) Lactoferrin as a natural immune modulator. Curr Pharm Des 15(17):1956–1973. https://doi.org/10.2174/138161209788453202",
"volume": "15",
"year": "2009"
},
{
"DOI": "10.3390/medicina57080842",
"author": "FD Algahtani",
"doi-asserted-by": "publisher",
"first-page": "842",
"issue": "8",
"journal-title": "Medicina (kaunas)",
"key": "427_CR3",
"unstructured": "Algahtani FD, Elabbasy MT, Samak MA, Adeboye AA, Yusuf RA, Ghoniem ME (2021) The Prospect of lactoferrin use as adjunctive agent in management of SARS-CoV-2 patients: a randomized pilot study. Medicina (kaunas) 57(8):842. https://doi.org/10.3390/medicina57080842",
"volume": "57",
"year": "2021"
},
{
"DOI": "10.1016/j.clnesp.2021.08.019",
"author": "AS Ali",
"doi-asserted-by": "publisher",
"first-page": "26",
"journal-title": "Clin Nutr ESPEN",
"key": "427_CR4",
"unstructured": "Ali AS, Hasan SS, Kow CS, Merchant HA (2021) Lactoferrin reduces the risk of respiratory tract infections: a meta-analysis of randomized controlled trials. Clin Nutr ESPEN 45:26–32. https://doi.org/10.1016/j.clnesp.2021.08.019",
"volume": "45",
"year": "2021"
},
{
"DOI": "10.1182/blood.2020006520",
"author": "H Al-Samkari",
"doi-asserted-by": "publisher",
"first-page": "489",
"issue": "4",
"journal-title": "Blood",
"key": "427_CR5",
"unstructured": "Al-Samkari H, Karp Leaf RS, Dzik WH, Carlson JCT, Fogerty AE, Waheed A, Goodarzi K, Bendapudi PK, Bornikova L, Gupta S, Leaf DE, Kuter DJ, Rosovsky RP (2020) COVID-19 and coagulation: bleeding and thrombotic manifestations of SARS-CoV-2 infection. Blood 136(4):489–500. https://doi.org/10.1182/blood.2020006520",
"volume": "136",
"year": "2020"
},
{
"DOI": "10.1179/2047773212Y.0000000004",
"author": "MG Ammendolia",
"doi-asserted-by": "publisher",
"first-page": "12",
"issue": "1",
"journal-title": "Pathog Glob Health",
"key": "427_CR6",
"unstructured": "Ammendolia MG, Agamennone M, Pietrantoni A, Lannutti F, Siciliano RA, De Giulio B, Amici C, Superti F (2012) Bovine lactoferrin-derived peptides as novel broad-spectrum inhibitors of influenza virus. Pathog Glob Health 106(1):12–19. https://doi.org/10.1179/2047773212Y.0000000004",
"volume": "106",
"year": "2012"
},
{
"DOI": "10.1056/NEJM199912233412607",
"author": "NC Andrews",
"doi-asserted-by": "publisher",
"first-page": "1986",
"journal-title": "N Engl J Med",
"key": "427_CR7",
"unstructured": "Andrews NC (2000) Disorders of iron metabolism. N Engl J Med 341:1986–1995. https://doi.org/10.1056/NEJM199912233412607",
"volume": "341",
"year": "2000"
},
{
"DOI": "10.1023/b:biom.0000027694.40260.70",
"author": "HM Baker",
"doi-asserted-by": "publisher",
"first-page": "209",
"issue": "3",
"journal-title": "Biometals",
"key": "427_CR8",
"unstructured": "Baker HM, Baker EN (2004) Lactoferrin and iron: structural and dynamic aspects of binding and release. Biometals 17(3):209–216. https://doi.org/10.1023/b:biom.0000027694.40260.70",
"volume": "17",
"year": "2004"
},
{
"DOI": "10.3389/fimmu.2017.01164",
"author": "JM Bartley",
"doi-asserted-by": "publisher",
"first-page": "1164",
"journal-title": "Front Immunol",
"key": "427_CR9",
"unstructured": "Bartley JM, Zhou X, Kuchel GA, Weinstock GM, Haynes L (2017) Impact of age, caloric restriction, and influenza infection on mouse gut microbiome: an exploratory study of the role of age-related microbiome changes on influenza responses. Front Immunol 8:1164. https://doi.org/10.3389/fimmu.2017.01164",
"volume": "8",
"year": "2017"
},
{
"DOI": "10.1016/j.exger.2017.12.015",
"author": "A Bektas",
"doi-asserted-by": "publisher",
"first-page": "10",
"journal-title": "Exp Gerontol",
"key": "427_CR10",
"unstructured": "Bektas A, Schurman SH, Sen R, Ferrucci L (2017) Aging, inflammation and the environment. Exp Gerontol 105:10–18. https://doi.org/10.1016/j.exger.2017.12.015",
"volume": "105",
"year": "2017"
},
{
"DOI": "10.3390/molecules16086992",
"author": "F Berlutti",
"doi-asserted-by": "publisher",
"first-page": "6992",
"issue": "8",
"journal-title": "Molecules",
"key": "427_CR11",
"unstructured": "Berlutti F, Pantanella F, Natalizi T, Frioni A, Paesano R, Polimeni A, Valenti P (2011) Antiviral properties of lactoferrin–a natural immunity molecule. Molecules 16(8):6992–7018. https://doi.org/10.3390/molecules16086992",
"volume": "16",
"year": "2011"
},
{
"DOI": "10.1016/0005-2795(74)90002-6",
"author": "JM Bluard-Deconinck",
"doi-asserted-by": "publisher",
"first-page": "311",
"journal-title": "Biochim Biophys Acta",
"key": "427_CR12",
"unstructured": "Bluard-Deconinck JM, Masson PL, Osinski PA, Heremans JF (1974) Amino acid sequence of cysteic peptides of lactoferrin and demonstration of similarities between lactoferrin and transferrin. Biochim Biophys Acta 365:311–317. https://doi.org/10.1016/0005-2795(74)90002-6",
"volume": "365",
"year": "1974"
},
{
"DOI": "10.1007/s10534-018-0087-5",
"author": "MC Bonaccorsi di Patti",
"doi-asserted-by": "publisher",
"first-page": "399",
"issue": "3",
"journal-title": "Biometals",
"key": "427_CR13",
"unstructured": "Bonaccorsi di Patti MC, Cutone A, Polticelli F, Rosa L, Lepanto MS, Valenti P, Musci G (2018) The ferroportin-ceruloplasmin system and the mammalian iron homeostasis machine: regulatory pathways and the role of lactoferrin. Biometals 31(3):399–414. https://doi.org/10.1007/s10534-018-0087-5",
"volume": "31",
"year": "2018"
},
{
"DOI": "10.1038/nri.2016.58",
"author": "P Broz",
"doi-asserted-by": "publisher",
"first-page": "407",
"issue": "7",
"journal-title": "Nat Rev Immunol",
"key": "427_CR14",
"unstructured": "Broz P, Dixit VM (2016) Inflammasomes: mechanism of assembly, regulation and signalling. Nat Rev Immunol 16(7):407–420. https://doi.org/10.1038/nri.2016.58",
"volume": "16",
"year": "2016"
},
{
"DOI": "10.1038/nrmicro.2016.142",
"author": "KF Budden",
"doi-asserted-by": "publisher",
"first-page": "55",
"journal-title": "Nat Rev Microbiol",
"key": "427_CR15",
"unstructured": "Budden KF, Gellatly SL, Wood DLA, Cooper MA, Morrison M, Hugenholtz P, Hansbro PM (2017) Emerging pathogenic links between microbiota and the gut-lung axis. Nat Rev Microbiol 15:55–63. https://doi.org/10.1038/nrmicro.2016.142",
"volume": "15",
"year": "2017"
},
{
"DOI": "10.3390/ijms21144903",
"author": "E Campione",
"doi-asserted-by": "publisher",
"first-page": "4903",
"issue": "14",
"journal-title": "Int J Mol Sci",
"key": "427_CR16",
"unstructured": "Campione E, Cosio T, Rosa L, Lanna C, Di Girolamo S, Gaziano R, Valenti P, Bianchi L (2020) Lactoferrin as protective natural barrier of respiratory and intestinal mucosa against coronavirus infection and inflammation. Int J Mol Sci 21(14):4903. https://doi.org/10.3390/ijms21144903",
"volume": "21",
"year": "2020"
},
{
"DOI": "10.3389/fphar.2021.666600",
"author": "E Campione",
"doi-asserted-by": "publisher",
"first-page": "666600",
"journal-title": "Front Pharmacol",
"key": "427_CR17",
"unstructured": "Campione E, Lanna C, Cosio T, Rosa L, Conte MP, Iacovelli F, Romeo A, Falconi M, Del Vecchio C, Franchin E, Lia MS, Minieri M, Chiaramonte C, Ciotti M, Nuccetelli M, Terrinoni A, Iannuzzi I, Coppeda L, Magrini A, Bernardini S, Sabatini S, Rosapepe F, Bartoletti PL, Moricca N, Di Lorenzo A, Andreoni M, Sarmati L, Miani A, Piscitelli P, Valenti P, Bianchi L (2021a) Lactoferrin against SARS-CoV-2 in vitro and in silico evidences. Front Pharmacol 12:666600. https://doi.org/10.3389/fphar.2021.666600",
"volume": "12",
"year": "2021"
},
{
"DOI": "10.3390/ijerph182010985",
"author": "E Campione",
"doi-asserted-by": "publisher",
"first-page": "10985",
"issue": "20",
"journal-title": "Int J Environ Res Public Health",
"key": "427_CR18",
"unstructured": "Campione E, Lanna C, Cosio T, Rosa L, Conte MP, Iacovelli F, Romeo A, Falconi M, Del Vecchio C, Franchin E, Lia MS, Minieri M, Chiaramonte C, Ciotti M, Nuccetelli M, Terrinoni A, Iannuzzi I, Coppeta L, Magrini A, Bernardini S, Sabatini S, Rosapepe F, Bartoletti PL, Moricca N, Di Lorenzo A, Andreoni M, Sarmati L, Miani A, Piscitelli P, Squillaci E, Valenti P, Bianchi L (2021b) Lactoferrin as antiviral treatment in COVID-19 management: preliminary evidence. Int J Environ Res Public Health 18(20):10985. https://doi.org/10.3390/ijerph182010985",
"volume": "18",
"year": "2021"
},
{
"DOI": "10.1016/j.ijantimicag.2020.106118",
"author": "R Chang",
"doi-asserted-by": "publisher",
"journal-title": "Int J Antimicrob Agents",
"key": "427_CR19",
"unstructured": "Chang R, Ng TB, Sun WZ (2020) Lactoferrin as potential preventative and adjunct treatment for COVID-19. Int J Antimicrob Agents 56:106118. https://doi.org/10.1016/j.ijantimicag.2020.106118",
"volume": "56",
"year": "2020"
},
{
"DOI": "10.1002/jcla.23618",
"author": "L Cheng",
"doi-asserted-by": "publisher",
"issue": "10",
"journal-title": "J Clin Lab Anal",
"key": "427_CR20",
"unstructured": "Cheng L, Li H, Li L, Liu C, Yan S, Chen H, Li Y (2020) Ferritin in the coronavirus disease 2019 (COVID-19): a systematic review and meta-analysis. J Clin Lab Anal 34(10):e23618. https://doi.org/10.1002/jcla.23618",
"volume": "34",
"year": "2020"
},
{
"DOI": "10.1074/jbc.R117.781823",
"author": "R Coffey",
"doi-asserted-by": "publisher",
"first-page": "12727",
"issue": "31",
"journal-title": "J Biol Chem",
"key": "427_CR21",
"unstructured": "Coffey R, Ganz T (2017) Iron homeostasis—an anthropocentric perspective. J Biol Chem 292(31):12727–12734. https://doi.org/10.1074/jbc.R117.781823",
"volume": "292",
"year": "2017"
},
{
"DOI": "10.1111/jth.14830",
"author": "S Cui",
"doi-asserted-by": "publisher",
"first-page": "1421",
"issue": "6",
"journal-title": "J Thromb Haemost",
"key": "427_CR22",
"unstructured": "Cui S, Chen S, Li X, Liu S, Wang F (2020) Prevalence of venous thromboembolism in patients with severe novel coronavirus pneumonia. J Thromb Haemost 18(6):1421–1424. https://doi.org/10.1111/jth.14830",
"volume": "18",
"year": "2020"
},
{
"DOI": "10.1007/s10534-014-9742-7",
"author": "A Cutone",
"doi-asserted-by": "publisher",
"first-page": "807",
"issue": "5",
"journal-title": "Biometals",
"key": "427_CR23",
"unstructured": "Cutone A, Frioni A, Berlutti F, Valenti P, Musci G, Bonaccorsi di Patti MC (2014) Lactoferrin prevents LPS-induced decrease of the iron exporter ferroportin in human monocytes/macrophages. Biometals 27(5):807–813. https://doi.org/10.1007/s10534-014-9742-7",
"volume": "27",
"year": "2014"
},
{
"DOI": "10.3389/fimmu.2017.00705",
"author": "A Cutone",
"doi-asserted-by": "publisher",
"first-page": "705",
"journal-title": "Front Immunol",
"key": "427_CR24",
"unstructured": "Cutone A, Rosa L, Lepanto MS, Scotti MJ, Berlutti F, Bonaccorsi di Patti MC, Musci G, Valenti P (2017) Lactoferrin efficiently counteracts the inflammation-induced changes of the iron homeostasis system in macrophages. Front Immunol 8:705. https://doi.org/10.3389/fimmu.2017.00705",
"volume": "8",
"year": "2017"
},
{
"DOI": "10.3390/ijms20092128",
"author": "A Cutone",
"doi-asserted-by": "publisher",
"first-page": "2128",
"issue": "9",
"journal-title": "Int J Mol Sci",
"key": "427_CR25",
"unstructured": "Cutone A, Lepanto MS, Rosa L, Scotti MJ, Rossi A, Ranucci S, De Fino I, Bragonzi A, Valenti P, Musci G, Berlutti F (2019) Aerosolized bovine lactoferrin counteracts infection, inflammation and iron dysbalance in a cystic fibrosis mouse model of Pseudomonas aeruginosa chronic lung infection. Int J Mol Sci 20(9):2128. https://doi.org/10.3390/ijms20092128",
"volume": "20",
"year": "2019"
},
{
"DOI": "10.1016/j.yexcr.2018.01.035",
"author": "M Dai",
"doi-asserted-by": "publisher",
"first-page": "95",
"issue": "1",
"journal-title": "Exp Cell Res",
"key": "427_CR26",
"unstructured": "Dai M, Pan P, Li H, Liu S, Zhang L, Song C, Li Y, Li Q, Mao Z, Long Y, Su X, Hu C (2018) The antimicrobial cathelicidin peptide hlF(1–11) attenuates alveolar macrophage pyroptosis induced by Acinetobacter baumannii in vivo. Exp Cell Res 364(1):95–103. https://doi.org/10.1016/j.yexcr.2018.01.035",
"volume": "364",
"year": "2018"
},
{
"DOI": "10.1155/2021/7866992",
"author": "J Dai",
"doi-asserted-by": "publisher",
"first-page": "7866992",
"journal-title": "Oxid Med Cell Longev",
"key": "427_CR27",
"unstructured": "Dai J, Teng X, Jin S, Wu Y (2021) The Antiviral roles of hydrogen sulfide by blocking the interaction between SARS-CoV-2 and its potential cell surface receptors. Oxid Med Cell Longev 2021:7866992. https://doi.org/10.1155/2021/7866992",
"volume": "2021",
"year": "2021"
},
{
"DOI": "10.1007/s00705-021-04993-4",
"author": "CB Denani",
"doi-asserted-by": "publisher",
"first-page": "1203",
"issue": "4",
"journal-title": "Arch Virol",
"key": "427_CR28",
"unstructured": "Denani CB, Real-Hohn A, de Carvalho CAM, Gomes AMO, Gonçalves RB (2021) Lactoferrin affects rhinovirus B-14 entry into H1-HeLa cells. Arch Virol 166(4):1203–1211. https://doi.org/10.1007/s00705-021-04993-4",
"volume": "166",
"year": "2021"
},
{
"DOI": "10.1371/journal.ppat.1005572",
"author": "E Deriu",
"doi-asserted-by": "publisher",
"journal-title": "PloS Pathog",
"key": "427_CR29",
"unstructured": "Deriu E, Boxx GM, He X, Pan C, Benavidez SD, Cen L, Rozengurt N, Shi W, Cheng G (2016) Influenza virus affects intestinal microbiota and secondary Salmonella infection in the gut through Type I Interferons. PloS Pathog 12:e1005572. https://doi.org/10.1371/journal.ppat.1005572",
"volume": "12",
"year": "2016"
},
{
"DOI": "10.1016/j.cmet.2005.01.003",
"author": "A Donovan",
"doi-asserted-by": "publisher",
"first-page": "191",
"journal-title": "Cell Metab",
"key": "427_CR30",
"unstructured": "Donovan A, Lima CA, Pinkus JL, Pinkus GS, Zon LI, Robine S, Andrews NC (2005) The iron exporter ferroportin/Slc40a1 is essential for iron homeostasis. Cell Metab 1:191–200. https://doi.org/10.1016/j.cmet.2005.01.003",
"volume": "1",
"year": "2005"
},
{
"DOI": "10.1038/nrmicro1930",
"author": "H Drakesmith",
"doi-asserted-by": "publisher",
"first-page": "541",
"issue": "7",
"journal-title": "Nat Rev Microbiol",
"key": "427_CR31",
"unstructured": "Drakesmith H, Prentice A (2008) Viral infection and iron metabolism. Nat Rev Microbiol 6(7):541–552. https://doi.org/10.1038/nrmicro1930",
"volume": "6",
"year": "2008"
},
{
"DOI": "10.1186/s13062-020-00275-2",
"author": "S Ehsani",
"doi-asserted-by": "publisher",
"first-page": "19",
"issue": "1",
"journal-title": "Biol Direct",
"key": "427_CR32",
"unstructured": "Ehsani S (2020) COVID-19 and iron dysregulation: distant sequence similarity between hepcidin and the novel coronavirus spike glycoprotein. Biol Direct 15(1):19. https://doi.org/10.1186/s13062-020-00275-2",
"volume": "15",
"year": "2020"
},
{
"DOI": "10.3389/fcimb.2020.00009",
"author": "R Enaud",
"doi-asserted-by": "publisher",
"first-page": "9",
"journal-title": "Front Cell Infect Microbiol",
"key": "427_CR33",
"unstructured": "Enaud R, Prevel R, Ciarlo E, Beaufils F, Wieërs G, Guery B, Delhaes L (2020) The gut- lung axis in health and respiratory diseases: a place for inter-organ and inter-kingdom crosstalks. Front Cell Infect Microbiol 10:9. https://doi.org/10.3389/fcimb.2020.00009",
"volume": "10",
"year": "2020"
},
{
"DOI": "10.2903/j.efsa.2012.2701",
"author": "European Food Safety Authority",
"doi-asserted-by": "publisher",
"first-page": "2701",
"journal-title": "EFSA J",
"key": "427_CR34",
"unstructured": "European Food Safety Authority (2012) Scientific opinion on bovine lactoferrin. EFSA J 10:2701. https://doi.org/10.2903/j.efsa.2012.2701",
"volume": "10",
"year": "2012"
},
{
"key": "427_CR35",
"unstructured": "U.S. FDA (2014) GRN 000465 Cow’s Milk-Derived Lactoferrin; Morinaga Milk Industry Co., Ltd.: Tokyo, Japan; U.S. Food and Drug Administration (U.S. FDA), Center for Food Safety & Applied Nutrition (CFSAN), Office of Food Additive Safety: Silver Spring, MD, USA. http://www.accessdata.fda.gov/scripts/fdcc/index.cfm?set=GRASNotices&id=465 Accessed 06 April 2022"
},
{
"DOI": "10.1016/j.foodchem.2012.12.049",
"author": "R Fernandez-Musoles",
"doi-asserted-by": "publisher",
"first-page": "994",
"issue": "1–4",
"journal-title": "Food Chem",
"key": "427_CR36",
"unstructured": "Fernandez-Musoles R, Salom JB, Martínez-Maqueda D, López-Díez JJ, Recio I, Manzanares P, (2013) Antihypertensive effects of lactoferrin hydrolyzates: inhibition of angiotensin- and endothelin-converting enzymes. Food Chem 139(1–4):994–1000. https://doi.org/10.1016/j.foodchem.2012.12.049",
"volume": "139",
"year": "2013"
},
{
"DOI": "10.1002/mnfr.201700389",
"author": "S Figueroa-Lozano",
"doi-asserted-by": "publisher",
"first-page": "1700389",
"issue": "2",
"journal-title": "Mol Nutr Food Res",
"key": "427_CR37",
"unstructured": "Figueroa-Lozano S, Valk-Weeber RL, van Leeuwen SS, Dijkhuizen L, de Vos P (2018) Dietary N-Glycans from bovine lactoferrin and TLR modulation. Mol Nutr Food Res 62(2):1700389. https://doi.org/10.1002/mnfr.201700389",
"volume": "62",
"year": "2018"
},
{
"DOI": "10.1016/S2213-2600(20)30243-5",
"author": "SE Fox",
"doi-asserted-by": "publisher",
"first-page": "681",
"issue": "7",
"journal-title": "Lancet Respir Med",
"key": "427_CR38",
"unstructured": "Fox SE, Akmatbekov A, Harbert JL, Li G, Quincy Brown J, Vander Heide RS (2020) Pulmonary and cardiac pathology in African American patients with COVID-19: an autopsy series from New Orleans. Lancet Respir Med 8(7):681–686. https://doi.org/10.1016/S2213-2600(20)30243-5",
"volume": "8",
"year": "2020"
},
{
"DOI": "10.1016/s1079-9796(03)00039-1",
"author": "DM Frazer",
"doi-asserted-by": "publisher",
"first-page": "288",
"issue": "3",
"journal-title": "Blood Cells Mol Dis",
"key": "427_CR39",
"unstructured": "Frazer DM, Anderson GJ (2003) The orchestration of body iron intake: How and where do enterocytes receive their cues? Blood Cells Mol Dis 30(3):288–297. https://doi.org/10.1016/s1079-9796(03)00039-1",
"volume": "30",
"year": "2003"
},
{
"DOI": "10.1007/s10534-014-9740-9",
"author": "A Frioni",
"doi-asserted-by": "publisher",
"first-page": "843",
"issue": "5",
"journal-title": "Biometals",
"key": "427_CR40",
"unstructured": "Frioni A, Conte MP, Cutone A, Longhi C, Musci G, di Patti MC, Natalizi T, Marazzato M, Lepanto MS, Puddu P, Paesano R, Valenti P, Berlutti F (2014) Lactoferrin differently modulates the inflammatory response in epithelial models mimicking human inflammatory and infectious diseases. Biometals 27(5):843–856. https://doi.org/10.1007/s10534-014-9740-9",
"volume": "27",
"year": "2014"
},
{
"DOI": "10.1007/BF01322673",
"author": "T Fujihara",
"doi-asserted-by": "publisher",
"first-page": "1469",
"issue": "8",
"journal-title": "Arch Virol",
"key": "427_CR41",
"unstructured": "Fujihara T, Hayashi K (1995) Lactoferrin inhibits herpes simplex virus type-1 (HSV-1) infection to mouse cornea. Arch Virol 140(8):1469–1472. https://doi.org/10.1007/BF01322673",
"volume": "140",
"year": "1995"
},
{
"DOI": "10.1016/j.nutres.2012.06.006",
"author": "CB Furlund",
"doi-asserted-by": "publisher",
"first-page": "503",
"issue": "7",
"journal-title": "Nutr Res",
"key": "427_CR42",
"unstructured": "Furlund CB, Kristoffersen AB, Devold TG, Vegarud GE, Jonassen CM (2012) Bovine lactoferrin digested with human gastrointestinal enzymes inhibits replication of human echovirus 5 in cell culture. Nutr Res 32(7):503–513. https://doi.org/10.1016/j.nutres.2012.06.006",
"volume": "32",
"year": "2012"
},
{
"DOI": "10.1084/jem.170.2.415",
"author": "P Furmanski",
"doi-asserted-by": "publisher",
"first-page": "415",
"journal-title": "J Exp Med",
"key": "427_CR43",
"unstructured": "Furmanski P, Li ZP, Fortuna MB, Swamy CV, Das MR (1989) Multiple molecular forms of human lactoferrin. Identification of a class of lactoferrins that possess ribonuclease activity and lack iron-binding capacity. J Exp Med 170:415–429. https://doi.org/10.1084/jem.170.2.415",
"volume": "170",
"year": "1989"
},
{
"DOI": "10.1038/s41467-021-26762-2",
"author": "GG Giobbe",
"doi-asserted-by": "publisher",
"first-page": "6610",
"issue": "1",
"journal-title": "Nat Commun",
"key": "427_CR44",
"unstructured": "Giobbe GG, Bonfante F, Jones BC, Gagliano O, Luni C, Zambaiti E, Perin S, Laterza C, Busslinger G, Stuart H, Pagliari M, Bortolami A, Mazzetto E, Manfredi A, Colantuono C, Di Filippo L, Pellegata AF, Panzarin V, Thapar N, Li VSW, Eaton S, Cacchiarelli D, Clevers H, Elvassore N, De Coppi P (2021) SARS-CoV-2 infection and replication in human gastric organoids. Nat Commun 12(1):6610. https://doi.org/10.1038/s41467-021-26762-2",
"volume": "12",
"year": "2021"
},
{
"DOI": "10.1186/s40168-018-0566-5",
"author": "A Grier",
"doi-asserted-by": "publisher",
"first-page": "193",
"journal-title": "Microbiome",
"key": "427_CR45",
"unstructured": "Grier A, McDavid A, Wang B, Qiu X, Java J, Bandyopadhyay S, Yang H, Holden-Wiltse J, Kessler HA, Gill AL, Huyck H, Falsey AR, Topham DJ, Scheible KM, Caserta MT, Pryhuber GS, Gill SR (2018) Neonatal gut and respiratory microbiota: coordinated development through time and space. Microbiome 6:193. https://doi.org/10.1186/s40168-018-0566-5",
"volume": "6",
"year": "2018"
},
{
"DOI": "10.1128/JVI.79.5.3009-3015.2005",
"author": "F Groot",
"doi-asserted-by": "publisher",
"first-page": "3009",
"issue": "5",
"journal-title": "J Virol",
"key": "427_CR46",
"unstructured": "Groot F, Geijtenbeek TB, Sanders RW, Baldwin CE, Sanchez-Hernandez M, Floris R, van Kooyk Y, de Jong EC, Berkhout B (2005) Lactoferrin prevents dendritic cell-mediated human immunodeficiency virus type 1 transmission by blocking the DC-SIGN–gp120 interaction. J Virol 79(5):3009–3015. https://doi.org/10.1128/JVI.79.5.3009-3015.2005",
"volume": "79",
"year": "2005"
},
{
"DOI": "10.1021/ja01498a029",
"author": "ML Groves",
"doi-asserted-by": "publisher",
"first-page": "3345",
"journal-title": "J Am Chem Soc",
"key": "427_CR47",
"unstructured": "Groves ML (1960) The isolation of a red protein from milk. J Am Chem Soc 82:3345–3350",
"volume": "82",
"year": "1960"
},
{
"DOI": "10.3389/fimmu.2018.00182",
"author": "HT Groves",
"doi-asserted-by": "publisher",
"first-page": "182",
"journal-title": "Front Immunol",
"key": "427_CR48",
"unstructured": "Groves HT, Cuthbertson L, James P, Moffatt MF, Cox MJ, Tregoning JS (2018) Respiratory disease following viral lung infection alters the murine gut microbiota. Front Immunol 9:182. https://doi.org/10.3389/fimmu.2018.00182",
"volume": "9",
"year": "2018"
},
{
"DOI": "10.1016/j.antiviral.2013.05.013",
"author": "L Gualdi",
"doi-asserted-by": "publisher",
"first-page": "188",
"issue": "2",
"journal-title": "Antiviral Res",
"key": "427_CR49",
"unstructured": "Gualdi L, Mertz S, Gomez AM, Ramilo O, Wittke A, Mejias A (2013) Lack of effect of bovine lactoferrin in respiratory syncytial virus replication and clinical disease severity in the mouse model. Antiviral Res 99(2):188–195. https://doi.org/10.1016/j.antiviral.2013.05.013",
"volume": "99",
"year": "2013"
},
{
"DOI": "10.3389/fimmu.2018.02640",
"author": "S Hanada",
"doi-asserted-by": "publisher",
"first-page": "2640",
"journal-title": "Front Immunol",
"key": "427_CR50",
"unstructured": "Hanada S, Pirzadeh M, Carver KY, Deng JC (2018) Respiratory viral infection-induced microbiome alterations and secondary bacterial pneumonia. Front Immunol 9:2640. https://doi.org/10.3389/fimmu.2018.02640",
"volume": "9",
"year": "2018"
},
{
"DOI": "10.1074/jbc.REV120.013930",
"author": "E Hartenian",
"doi-asserted-by": "publisher",
"first-page": "12910",
"issue": "37",
"journal-title": "J Biol Chem",
"key": "427_CR51",
"unstructured": "Hartenian E, Nandakumar D, Lari A, Ly M, Tucker JM, Glaunsinger BA (2020) The molecular virology of coronaviruses. J Biol Chem 295(37):12910–12934. https://doi.org/10.1074/jbc.REV120.013930",
"volume": "295",
"year": "2020"
},
{
"DOI": "10.3390/cells9122638",
"author": "MM Hatmal",
"doi-asserted-by": "publisher",
"first-page": "2638",
"issue": "12",
"journal-title": "Cells",
"key": "427_CR52",
"unstructured": "Hatmal MM, Alshaer W, Al-Hatamleh MAI, Hatmal M, Smadi O, Taha MO, Oweida AJ, Boer JC, Mohamud R, Plebanski M (2020) Comprehensive structural and molecular comparison of spike proteins of SARS-CoV-2, SARS-CoV and MERS-CoV, and their interactions with ACE2. Cells 9(12):2638. https://doi.org/10.3390/cells9122638",
"volume": "9",
"year": "2020"
},
{
"DOI": "10.1016/j.hepres.2004.01.002",
"author": "N Hirashima",
"doi-asserted-by": "publisher",
"first-page": "9",
"issue": "1",
"journal-title": "Hepatol Res",
"key": "427_CR53",
"unstructured": "Hirashima N, Orito E, Ohba K, Kondo H, Sakamoto T, Matsunaga S, Kato A, Nukaya H, Sakakibara K, Ohno T, Kato H, Sugauchi F, Kato T, Tanaka Y, Ueda R, Mizokami M (2004) A randomized controlled trial of consensus interferon with or without lactoferrin for chronic hepatitis C patients with genotype 1b and high viral load. Hepatol Res 29(1):9–12. https://doi.org/10.1016/j.hepres.2004.01.002",
"volume": "29",
"year": "2004"
},
{
"DOI": "10.1080/22221751.2021.1888660",
"author": "Y Hu",
"doi-asserted-by": "publisher",
"first-page": "317",
"issue": "1",
"journal-title": "Emerg Microbes Infect",
"key": "427_CR54",
"unstructured": "Hu Y, Meng X, Zhang F, Xiang Y, Wang J (2021) The in vitro antiviral activity of lactoferrin against common human coronaviruses and SARS-CoV-2 is mediated by targeting the heparan sulfate co-receptor. Emerg Microbes Infect 10(1):317–330. https://doi.org/10.1080/22221751.2021.1888660",
"volume": "10",
"year": "2021"
},
{
"DOI": "10.1074/jbc.M205305200",
"author": "HN Hunter",
"doi-asserted-by": "publisher",
"first-page": "37597",
"issue": "40",
"journal-title": "J Biol Chem",
"key": "427_CR55",
"unstructured": "Hunter HN, Fulton DB, Ganz T, Vogel HJ (2002) The solution structure of human hepcidin, a peptide hormone with antimicrobial activity that is involved in iron uptake and hereditary hemochromatosis. J Biol Chem 277(40):37597–37603. https://doi.org/10.1074/jbc.M205305200",
"volume": "277",
"year": "2002"
},
{
"DOI": "10.3389/fimmu.2021.765965",
"author": "I Hussain",
"doi-asserted-by": "publisher",
"journal-title": "Front Immunol",
"key": "427_CR56",
"unstructured": "Hussain I, Cher GLY, Abid MA, Abid MB (2021) Role of gut microbiome in COVID-19: an insight into pathogenesis and therapeutic potential. Front Immunol 12:765965. https://doi.org/10.3389/fimmu.2021.765965",
"volume": "12",
"year": "2021"
},
{
"DOI": "10.1073/pnas.1019378108",
"author": "T Ichinohe",
"doi-asserted-by": "publisher",
"first-page": "5354",
"journal-title": "Proc Natl Acad Sci U S A",
"key": "427_CR57",
"unstructured": "Ichinohe T, Pang IK, Kumamoto Y, Peaper DR, Ho JH, Murray TS, Iwasaki A (2011) Microbiota regulates immune defense against respiratory tract Influenza A virus infection. Proc Natl Acad Sci U S A 108:5354–5359. https://doi.org/10.1073/pnas.1019378108",
"volume": "108",
"year": "2011"
},
{
"DOI": "10.1016/j.hepres.2005.03.018",
"author": "Y Ishibashi",
"doi-asserted-by": "publisher",
"first-page": "218",
"issue": "4",
"journal-title": "Hepatol Res",
"key": "427_CR58",
"unstructured": "Ishibashi Y, Takeda K, Tsukidate N, Miyazaki H, Ohira K, Dosaka-Akita H, Nishimura M (2005) Randomized placebo-controlled trial of interferon alpha-2b plus ribavirin with and without lactoferrin for chronic hepatitis C. Hepatol Res 32(4):218–223. https://doi.org/10.1016/j.hepres.2005.03.018",
"volume": "32",
"year": "2005"
},
{
"DOI": "10.3891/acta.chem.scand.14-0510",
"author": "B Johansson",
"doi-asserted-by": "publisher",
"first-page": "510",
"journal-title": "Acta Chem Scand",
"key": "427_CR59",
"unstructured": "Johansson B (1960) Isolation of an iron-containing red protein from human milk. Acta Chem Scand 14:510–512",
"volume": "14",
"year": "1960"
},
{
"DOI": "10.1016/j.molimm.2012.12.021",
"author": "O Kalashnyk",
"doi-asserted-by": "publisher",
"first-page": "319",
"issue": "3–4",
"journal-title": "Mol Immunol",
"key": "427_CR60",
"unstructured": "Kalashnyk O, Petrova Y, Lykhmus O, Mikhalovska L, Mikhalovsky S, Zhukova A, Gnatenko D, Bahou W, Komisarenko S, Skok M (2013) Expression, function and cooperating partners of protease-activated receptor type 3 in vascular endothelial cells and B lymphocytes studied with specific monoclonal antibody. Mol Immunol 54(3–4):319–326. https://doi.org/10.1016/j.molimm.2012.12.021",
"volume": "54",
"year": "2013"
},
{
"DOI": "10.1038/s41586-020-2665-2",
"author": "Z Ke",
"doi-asserted-by": "publisher",
"first-page": "498",
"issue": "7838",
"journal-title": "Nature",
"key": "427_CR61",
"unstructured": "Ke Z, Oton J, Qu K, Cortese M, Zila V, McKeane L, Nakane T, Zivanov J, Neufeldt CJ, Cerikan B, Lu JM, Peukes J, Xiong X, Kräusslich HG, Scheres SHW, Bartenschlager R, Briggs JAG (2020) Structures and distributions of SARS-CoV-2 spike proteins on intact virions. Nature 588(7838):498–502. https://doi.org/10.1038/s41586-020-2665-2",
"volume": "588",
"year": "2020"
},
{
"DOI": "10.3389/fimmu.2020.01221",
"author": "DB Kell",
"doi-asserted-by": "publisher",
"first-page": "1221",
"journal-title": "Front Immunol",
"key": "427_CR62",
"unstructured": "Kell DB, Heyden EL, Pretorius E (2020) The Biology of lactoferrin, an iron-binding protein that can help defend against viruses and bacteria. Front Immunol 11:1221. https://doi.org/10.3389/fimmu.2020.01221",
"volume": "11",
"year": "2020"
},
{
"DOI": "10.1016/j.thromres.2020.04.013",
"author": "FA Klok",
"doi-asserted-by": "publisher",
"first-page": "145",
"journal-title": "Thromb Res",
"key": "427_CR63",
"unstructured": "Klok FA, Kruip MJHA, van der Meer NJM, Arbous MS, Gommers DAMPJ, Kant KM, Kaptein FHJ, van Paassen J, Stals MAM, Huisman MV, Endeman H (2020) Incidence of thrombotic complications in critically ill ICU patients with COVID-19. Thromb Res 191:145–147. https://doi.org/10.1016/j.thromres.2020.04.013",
"volume": "191",
"year": "2020"
},
{
"DOI": "10.1016/s0014-5793(00)01920-7",
"author": "A Krause",
"doi-asserted-by": "publisher",
"first-page": "147",
"issue": "2–3",
"journal-title": "FEBS Lett",
"key": "427_CR64",
"unstructured": "Krause A, Neitz S, Magert HJ, Schulz A, Forssmann WG, Schulz-Knappe P, Adermann K (2000) LEAP-1, a novel highly disulfidebonded human peptide, exhibits antimicrobial activity. FEBS Lett 480(2–3):147–150. https://doi.org/10.1016/s0014-5793(00)01920-7",
"volume": "480",
"year": "2000"
},
{
"author": "ML Kruzel",
"first-page": "261",
"journal-title": "Postepy Hig Med Dosw",
"key": "427_CR65",
"unstructured": "Kruzel ML, Actor JK, Boldogh I, Zimecki M (2007) Lactoferrin in health and disease. Postepy Hig Med Dosw 61:261–267",
"volume": "61",
"year": "2007"
},
{
"DOI": "10.3389/fimmu.2017.01438",
"author": "ML Kruzel",
"doi-asserted-by": "publisher",
"first-page": "1438",
"journal-title": "Front Immunol",
"key": "427_CR66",
"unstructured": "Kruzel ML, Zimecki M, Actor JK (2017) Lactoferrin in a context of inflammation-induced pathology. Front Immunol 8:1438. https://doi.org/10.3389/fimmu.2017.01438",
"volume": "8",
"year": "2017"
},
{
"DOI": "10.1089/jir.2006.26.489",
"author": "T Kuhara",
"doi-asserted-by": "publisher",
"first-page": "489",
"issue": "7",
"journal-title": "J Interferon Cytokine Res",
"key": "427_CR67",
"unstructured": "Kuhara T, Yamauchi K, Tamura Y, Okamura H (2006) Oral administration of lactoferrin increases NK cell activity in mice via increased production of IL-18 and type I IFN in the small intestine. J Interferon Cytokine Res 26(7):489–499. https://doi.org/10.1089/jir.2006.26.489",
"volume": "26",
"year": "2006"
},
{
"DOI": "10.1371/journal.pone.0023710",
"author": "J Lang",
"doi-asserted-by": "publisher",
"issue": "8",
"journal-title": "PLoS ONE",
"key": "427_CR68",
"unstructured": "Lang J, Yang N, Deng J, Liu K, Yang P, Zhang G, Jiang C (2011) Inhibition of SARS pseudovirus cell entry by lactoferrin binding to heparan sulfate proteoglycans. PLoS ONE 6(8):e23710. https://doi.org/10.1371/journal.pone.0023710",
"volume": "6",
"year": "2011"
},
{
"DOI": "10.1038/nri3452",
"author": "E Latz",
"doi-asserted-by": "publisher",
"first-page": "397",
"issue": "6",
"journal-title": "Nat Rev Immunol",
"key": "427_CR69",
"unstructured": "Latz E, Xiao TS, Stutz A (2013) Activation and regulation of the inflammasomes. Nat Rev Immunol 13(6):397–411. https://doi.org/10.1038/nri3452",
"volume": "13",
"year": "2013"
},
{
"DOI": "10.1073/pnas.0409808102",
"author": "P Lee",
"doi-asserted-by": "publisher",
"first-page": "1906",
"issue": "6",
"journal-title": "Proc Natl Acad Sci USA",
"key": "427_CR70",
"unstructured": "Lee P, Peng H, Gelbart T, Wang L, Beutler E (2005) Regulation of hepcidin transcription by interleukin-1 and interleukin-6. Proc Natl Acad Sci USA 102(6):1906–1910. https://doi.org/10.1073/pnas.0409808102",
"volume": "102",
"year": "2005"
},
{
"DOI": "10.1007/s10534-010-9297-1",
"author": "D Legrand",
"doi-asserted-by": "publisher",
"first-page": "365",
"issue": "3",
"journal-title": "Biometals",
"key": "427_CR71",
"unstructured": "Legrand D, Mazurier J (2010) A critical review of the roles of host lactoferrin in immunity. Biometals 23(3):365–376. https://doi.org/10.1007/s10534-010-9297-1",
"volume": "23",
"year": "2010"
},
{
"DOI": "10.1007/s00018-005-5370-2",
"author": "D Legrand",
"doi-asserted-by": "publisher",
"first-page": "2549",
"issue": "22",
"journal-title": "Cell Mol Life Sci",
"key": "427_CR72",
"unstructured": "Legrand D, Elass E, Carpentier M, Mazurier J (2005) Lactoferrin: a modulator of immune and inflammatory responses. Cell Mol Life Sci 62(22):2549–2559. https://doi.org/10.1007/s00018-005-5370-2",
"volume": "62",
"year": "2005"
},
{
"DOI": "10.3389/fimmu.2018.02123",
"author": "MS Lepanto",
"doi-asserted-by": "publisher",
"first-page": "2123",
"journal-title": "Front Immunol",
"key": "427_CR73",
"unstructured": "Lepanto MS, Rosa L, Cutone A, Conte MP, Paesano R, Valenti P (2018) Efficacy of lactoferrin oral administration in the treatment of anemia and anemia of inflammation in pregnant and non-pregnant women: an interventional study. Front Immunol 9:2123. https://doi.org/10.3389/fimmu.2018.02123",
"volume": "9",
"year": "2018"
},
{
"DOI": "10.1111/j.1432-1033.1993.tb17871.x",
"author": "B Leveugle",
"doi-asserted-by": "publisher",
"first-page": "1205",
"issue": "3",
"journal-title": "Eur J Biochem",
"key": "427_CR74",
"unstructured": "Leveugle B, Mazurier J, Legrand D, Mazurier C, Montreuil J, Spik G (1993) Lactotransferrin binding to its platelet receptor inhibits platelet aggregation. Eur J Biochem 213(3):1205–1211. https://doi.org/10.1111/j.1432-1033.1993.tb17871.x",
"volume": "213",
"year": "1993"
},
{
"DOI": "10.1086/343809",
"author": "TY Lin",
"doi-asserted-by": "publisher",
"first-page": "1161",
"issue": "8",
"journal-title": "J Infect Dis",
"key": "427_CR75",
"unstructured": "Lin TY, Chu C, Chiu CH (2002) Lactoferrin inhibits enterovirus 71 infection of human embryonal rhabdomyosarcoma cells in vitro. J Infect Dis 186(8):1161–1164. https://doi.org/10.1086/343809",
"volume": "186",
"year": "2002"
},
{
"DOI": "10.1007/s15010-021-01715-5",
"author": "TFD Liu",
"doi-asserted-by": "publisher",
"first-page": "1",
"journal-title": "Infection",
"key": "427_CR76",
"unstructured": "Liu TFD, Philippou E, Kolokotroni O, Siakallis G, Rahima K, Constantinou C (2021) Gut and airway microbiota and their role in COVID-19 infection and pathogenesis: a scoping review. Infection 20:1–33. https://doi.org/10.1007/s15010-021-01715-5",
"volume": "20",
"year": "2021"
},
{
"DOI": "10.1111/jth.12935",
"author": "C Longstaff",
"doi-asserted-by": "publisher",
"first-page": "S98",
"issue": "Suppl 1",
"journal-title": "J Thromb Haemost",
"key": "427_CR77",
"unstructured": "Longstaff C, Kolev K (2015) Basic mechanisms and regulation of fibrinolysis. J Thromb Haemost 13(Suppl 1):S98-105. https://doi.org/10.1111/jth.12935",
"volume": "13",
"year": "2015"
},
{
"author": "L Lu",
"first-page": "4184",
"issue": "15",
"journal-title": "Cancer Res",
"key": "427_CR78",
"unstructured": "Lu L, Hangoc G, Oliff A, Chen LT, Shen RN, Broxmeyer HE (1987) Protective influence of lactoferrin on mice infected with the polycythemia-inducing strain of Friend virus complex. Cancer Res 47(15):4184–4188",
"volume": "47",
"year": "1987"
},
{
"DOI": "10.3390/molecules25081997",
"author": "R Mancinelli",
"doi-asserted-by": "publisher",
"first-page": "1997",
"issue": "8",
"journal-title": "Molecules",
"key": "427_CR79",
"unstructured": "Mancinelli R, Rosa L, Cutone A, Lepanto MS, Franchitto A, Onori P, Gaudio E, Valenti P (2020) Viral hepatitis and iron dysregulation: molecular pathways and the role of lactoferrin. Molecules 25(8):1997. https://doi.org/10.3390/molecules25081997",
"volume": "25",
"year": "2020"
},
{
"DOI": "10.1038/nrd.2018.149",
"author": "MSJ Mangan",
"doi-asserted-by": "publisher",
"first-page": "688",
"issue": "9",
"journal-title": "Nat Rev Drug Discov",
"key": "427_CR80",
"unstructured": "Mangan MSJ, Olhava EJ, Roush WR, Seidel HM, Glick GD, Latz E (2018) Targeting the NLRP3 inflammasome in inflammatory diseases. Nat Rev Drug Discov 17(9):688. https://doi.org/10.1038/nrd.2018.149",
"volume": "17",
"year": "2018"
},
{
"DOI": "10.1016/0166-3542(95)00840-3",
"author": "M Marchetti",
"doi-asserted-by": "publisher",
"first-page": "221",
"issue": "2–3",
"journal-title": "Antiviral Res",
"key": "427_CR81",
"unstructured": "Marchetti M, Longhi C, Conte MP, Pisani S, Valenti P, Seganti L (1996) Lactoferrin inhibits herpes simplex virus type 1 adsorption to Vero cells. Antiviral Res 29(2–3):221–231. https://doi.org/10.1016/0166-3542(95)00840-3",
"volume": "29",
"year": "1996"
},
{
"DOI": "10.1023/a:1009217709851",
"author": "M Marchetti",
"doi-asserted-by": "publisher",
"first-page": "89",
"journal-title": "Biometals",
"key": "427_CR82",
"unstructured": "Marchetti M, Pisani S, Antonini G, Valenti P, Seganti L, Orsi N (1998) Metal complexes of bovine lactoferrin inhibit in vitro replication of herpes simplex virus type 1 and 2. Biometals 11:89–94. https://doi.org/10.1023/a:1009217709851",
"volume": "11",
"year": "1998"
},
{
"DOI": "10.1007/s004300050093",
"author": "M Marchetti",
"doi-asserted-by": "publisher",
"first-page": "199",
"issue": "4",
"journal-title": "Med Microbiol Immunol",
"key": "427_CR83",
"unstructured": "Marchetti M, Superti F, Ammendolia MG, Rossi P, Valenti P, Seganti L (1999) Inhibition of poliovirus type 1 infection by iron-, manganese- and zinc-saturated lactoferrin. Med Microbiol Immunol 187(4):199–204. https://doi.org/10.1007/s004300050093",
"volume": "187",
"year": "1999"
},
{
"DOI": "10.1016/j.biochi.2008.04.015",
"author": "M Marchetti",
"doi-asserted-by": "publisher",
"first-page": "155",
"issue": "1",
"journal-title": "Biochimie",
"key": "427_CR84",
"unstructured": "Marchetti M, Ammendolia MG, Superti F (2009) Glycosaminoglycans are not indispensable for the anti-herpes simplex virus type 2 activity of lactoferrin. Biochimie 91(1):155–159. https://doi.org/10.1016/j.biochi.2008.04.015",
"volume": "91",
"year": "2009"
},
{
"DOI": "10.1101/cshperspect.a011866",
"author": "JL Miller",
"doi-asserted-by": "publisher",
"issue": "7",
"journal-title": "Cold Spring Harb Perspect Med",
"key": "427_CR85",
"unstructured": "Miller JL (2012) Iron deficiency anemia: A common and curable disease. Cold Spring Harb Perspect Med 3(7):a011866. https://doi.org/10.1101/cshperspect.a011866",
"volume": "3",
"year": "2012"
},
{
"DOI": "10.3389/fmolb.2021.607443",
"author": "M Miotto",
"doi-asserted-by": "publisher",
"journal-title": "Front Mol Biosci",
"key": "427_CR86",
"unstructured": "Miotto M, Di Rienzo L, Bò L, Boffi A, Ruocco G, Milanetti E (2021) Molecular mechanisms behind anti SARS-CoV-2 action of lactoferrin. Front Mol Biosci 8:607443. https://doi.org/10.3389/fmolb.2021.607443",
"volume": "8",
"year": "2021"
},
{
"DOI": "10.1073/pnas.2105815118",
"author": "C Mirabelli",
"doi-asserted-by": "publisher",
"issue": "36",
"journal-title": "Proc Natl Acad Sci U S A",
"key": "427_CR87",
"unstructured": "Mirabelli C, Wotring JW, Zhang CJ, McCarty SM, Fursmidt R, Pretto CD, Qiao Y, Zhang Y, Frum T, Kadambi NS, Amin AT, O’Meara TR, Spence JR, Huang J, Alysandratos KD, Kotton DN, Handelman SK, Wobus CE, Weatherwax KJ, Mashour GA, O’Meara MJ, Chinnaiyan AM, Sexton JZ (2021) Morphological cell profiling of SARS-CoV-2 infection identifies drug repurposing candidates for COVID-19. Proc Natl Acad Sci U S A 118(36):e2105815118. https://doi.org/10.1073/pnas.2105815118",
"volume": "118",
"year": "2021"
},
{
"DOI": "10.1016/0006-3002(60)91478-5",
"author": "J Montreuil",
"doi-asserted-by": "publisher",
"first-page": "413",
"journal-title": "Biochim Biophys Acta",
"key": "427_CR88",
"unstructured": "Montreuil J, Tonnelat J, Mullet S (1960) Preparation and properties of lactosiderophilin (lactotransferrin) of human milk. Biochim Biophys Acta 45:413–421",
"volume": "45",
"year": "1960"
},
{
"DOI": "10.3389/fped.2020.00233",
"author": "N Motoki",
"doi-asserted-by": "publisher",
"first-page": "233",
"journal-title": "Placebo-Controlled Trial Front Pediatr",
"key": "427_CR89",
"unstructured": "Motoki N, Mizuki M, Tsukahara T, Miyakawa M, Kubo S, Oda H, Tanaka M, Yamauchi K, Abe F, Nomiyama T (2020) Effects of lactoferrin-fortified formula on acute gastrointestinal symptoms in children aged 12–32 months: a randomized, double-blind. Placebo-Controlled Trial Front Pediatr 8:233. https://doi.org/10.3389/fped.2020.00233",
"volume": "8",
"year": "2020"
},
{
"DOI": "10.1002/ajh.26027",
"author": "A Nai",
"doi-asserted-by": "publisher",
"first-page": "E32",
"issue": "1",
"journal-title": "Am J Hematol",
"key": "427_CR90",
"unstructured": "Nai A, Lorè NI, Pagani A, De Lorenzo R, Di Modica S, Saliu F, Cirillo DM, Rovere-Querini P, Manfredi AA, Silvestri L (2021) Hepcidin levels predict Covid-19 severity and mortality in a cohort of hospitalized Italian patients. Am J Hematol 96(1):E32–E35. https://doi.org/10.1002/ajh.26027",
"volume": "96",
"year": "2021"
},
{
"DOI": "10.1080/19390211.2020.1834047",
"author": "SAG Naidu",
"doi-asserted-by": "publisher",
"first-page": "78",
"issue": "1",
"journal-title": "J Diet Suppl",
"key": "427_CR91",
"unstructured": "Naidu SAG, Clemens RA, Pressman P, Zaigham M, Davies KJA, Naidu AS (2022) COVID-19 during pregnancy and postpartum. J Diet Suppl 19(1):78–114. https://doi.org/10.1080/19390211.2020.1834047",
"volume": "19",
"year": "2022"
},
{
"DOI": "10.1172/JCI20945",
"author": "E Nemeth",
"doi-asserted-by": "publisher",
"first-page": "1271",
"issue": "9",
"journal-title": "J Clin Investig",
"key": "427_CR92",
"unstructured": "Nemeth E, Rivera S, Gabayan V, Keller C, Taudorf S, Pedersen BK, Ganz T (2004) IL-6 mediates hypoferremia of inflammation by inducing the synthesis of the iron regulatory hormone hepcidin. J Clin Investig 113(9):1271–1276. https://doi.org/10.1172/JCI20945",
"volume": "113",
"year": "2004"
},
{
"DOI": "10.1007/s00253-015-6818-4",
"author": "TB Ng",
"doi-asserted-by": "publisher",
"first-page": "6997",
"issue": "17",
"journal-title": "Appl Microbiol Biotechnol",
"key": "427_CR93",
"unstructured": "Ng TB, Cheung RC, Wong JH, Wang Y, Ip DT, Wan DC, Xia J (2015) Antiviral activities of whey proteins. Appl Microbiol Biotechnol 99(17):6997–7008. https://doi.org/10.1007/s00253-015-6818-4",
"volume": "99",
"year": "2015"
},
{
"DOI": "10.1016/j.jmii.2020.02.010",
"author": "H Oda",
"doi-asserted-by": "publisher",
"first-page": "566",
"issue": "4",
"journal-title": "J Microbiol Immunol Infect",
"key": "427_CR94",
"unstructured": "Oda H, Wakabayashi H, Tanaka M, Yamauchi K, Sugita C, Yoshida H, Abe F, Sonoda T, Kurokawa M (2021) Effects of lactoferrin on infectious diseases in Japanese summer: a randomized, double-blinded, placebo-controlled trial. J Microbiol Immunol Infect 54(4):566–574. https://doi.org/10.1016/j.jmii.2020.02.010",
"volume": "54",
"year": "2021"
},
{
"DOI": "10.1111/j.1349-7006.2002.tb02484.x",
"author": "S Okada",
"doi-asserted-by": "publisher",
"first-page": "1063",
"issue": "9",
"journal-title": "Jpn J Cancer Res",
"key": "427_CR95",
"unstructured": "Okada S, Tanaka K, Sato T, Ueno H, Saito S, Okusaka T, Sato K, Yamamoto S, Kakizoe T (2002) Dose-response trial of lactoferrin in patients with chronic hepatitis C. Jpn J Cancer Res 93(9):1063–1069. https://doi.org/10.1111/j.1349-7006.2002.tb02484.x",
"volume": "93",
"year": "2002"
},
{
"DOI": "10.1177/039463201002300220",
"author": "R Paesano",
"doi-asserted-by": "publisher",
"first-page": "577",
"issue": "2",
"journal-title": "Int J Immunopathol Pharmacol",
"key": "427_CR96",
"unstructured": "Paesano R, Berlutti F, Pietropaoli M, Goolsbee W, Pacifici E, Valenti P (2010) Lactoferrin efficacy versus ferrous sulfate in curing iron disorders in pregnant and non-pregnant women. Int J Immunopathol Pharmacol 23(2):577–587. https://doi.org/10.1177/039463201002300220",
"volume": "23",
"year": "2010"
},
{
"DOI": "10.1179/2047773212Y.0000000043",
"author": "R Paesano",
"doi-asserted-by": "publisher",
"first-page": "200",
"journal-title": "Pathog Glob Health",
"key": "427_CR97",
"unstructured": "Paesano R, Natalizi T, Berlutti F, Valenti P (2012) Body iron delocalization: the serious drawback in iron disorders in both developing and developed countries. Pathog Glob Health 106:200–216. https://doi.org/10.1179/2047773212Y.0000000043",
"volume": "106",
"year": "2012"
},
{
"DOI": "10.1128/JVI.00996-19",
"author": "P Pan",
"doi-asserted-by": "publisher",
"first-page": "e00996",
"issue": "21",
"journal-title": "J Virol",
"key": "427_CR98",
"unstructured": "Pan P, Zhang Q, Liu W, Wang W, Lao Z, Zhang W, Shen M, Wan P, Xiao F, Liu F, Zhang W, Tan Q, Liu X, Wu K, Liu Y, Li G, Wu J (2019) Dengue virus M protein promotes NLRP3 Inflammasome activation to induce vascular leakage in mice. J Virol 93(21):e00996-e1019. https://doi.org/10.1128/JVI.00996-19",
"volume": "93",
"year": "2019"
},
{
"DOI": "10.1038/s41467-021-25629-w",
"author": "P Pan",
"doi-asserted-by": "publisher",
"first-page": "5306",
"issue": "1",
"journal-title": "Nat Commun",
"key": "427_CR99",
"unstructured": "Pan P, Shen M, Yu Z, Ge W, Chen K, Tian M, Xiao F, Wang Z, Wang J, Jia Y, Wang W, Wan P, Zhang J, Chen W, Lei Z, Chen X, Luo Z, Zhang Q, Xu M, Li G, Li Y, Wu J (2021) SARS-CoV-2 N protein promotes NLRP3 inflammasome activation to induce hyperinflammation. Nat Commun 12(1):5306. https://doi.org/10.1038/s41467-021-25629-w",
"volume": "12",
"year": "2021"
},
{
"DOI": "10.1074/jbc.M008922200",
"author": "CH Park",
"doi-asserted-by": "publisher",
"first-page": "7806",
"issue": "11",
"journal-title": "J Biol Chem",
"key": "427_CR100",
"unstructured": "Park CH, Valore EV, Waring AJ, Ganz T (2001) Hepcidin, a urinary antimicrobial peptide synthesized in the liver. J Biol Chem 276(11):7806–7810. https://doi.org/10.1074/jbc.M008922200",
"volume": "276",
"year": "2001"
},
{
"DOI": "10.3945/jn.108.093856",
"author": "FJ Pérez-Cano",
"doi-asserted-by": "publisher",
"first-page": "2392",
"issue": "12",
"journal-title": "Nutr",
"key": "427_CR101",
"unstructured": "Pérez-Cano FJ, Marín-Gallén S, Castell M, Rodríguez-Palmero M, Rivero M, Castellote C, Franch AJ (2008) Supplementing suckling rats with whey protein concentrate modulates the immune response and ameliorates rat rotavirus-induced diarrhea. Nutr 138(12):2392–2398. https://doi.org/10.3945/jn.108.093856",
"volume": "138",
"year": "2008"
},
{
"DOI": "10.1007/s12026-020-09145-5",
"author": "C Perricone",
"doi-asserted-by": "publisher",
"first-page": "213",
"issue": "4",
"journal-title": "Immunol Res",
"key": "427_CR102",
"unstructured": "Perricone C, Bartoloni E, Bursi R, Cafaro G, Guidelli GM, Shoenfeld Y, Gerli R (2020) COVID-19 as part of the hyperferritinemic syndromes: the role of iron depletion therapy. Immunol Res 68(4):213–224. https://doi.org/10.1007/s12026-020-09145-5",
"volume": "68",
"year": "2020"
},
{
"DOI": "10.1128/AAC.47.8.2688-2691.2003",
"author": "A Pietrantoni",
"doi-asserted-by": "publisher",
"first-page": "2688",
"issue": "8",
"journal-title": "Antimicrob Agents Chemother",
"key": "427_CR103",
"unstructured": "Pietrantoni A, Di Biase AM, Tinari A, Marchetti M, Valenti P, Seganti L, Superti F (2003) Bovine lactoferrin inhibits adenovirus infection by interacting with viral structural polypeptides. Antimicrob Agents Chemother 47(8):2688–2691. https://doi.org/10.1128/AAC.47.8.2688-2691.2003",
"volume": "47",
"year": "2003"
},
{
"DOI": "10.1007/s10534-010-9323-3",
"author": "A Pietrantoni",
"doi-asserted-by": "publisher",
"first-page": "465",
"issue": "3",
"journal-title": "Biometals",
"key": "427_CR104",
"unstructured": "Pietrantoni A, Dofrelli E, Tinari A, Ammendolia MG, Puzelli S, Fabiani C, Donatelli I, Superti F (2010) Bovine lactoferrin inhibits influenza A virus induced programmed cell death in vitro. Biometals 23(3):465–475. https://doi.org/10.1007/s10534-010-9323-3",
"volume": "23",
"year": "2010"
},
{
"DOI": "10.1139/o11-072",
"author": "A Pietrantoni",
"doi-asserted-by": "publisher",
"first-page": "442",
"issue": "3",
"journal-title": "Biochem Cell Biol",
"key": "427_CR105",
"unstructured": "Pietrantoni A, Ammendolia MG, Superti F (2012) Bovine lactoferrin: involvement of metal saturation and carbohydrates in the inhibition of influenza virus infection. Biochem Cell Biol 90(3):442–448. https://doi.org/10.1139/o11-072",
"volume": "90",
"year": "2012"
},
{
"DOI": "10.1016/s1357-2725(98)00066-1",
"author": "P Puddu",
"doi-asserted-by": "publisher",
"first-page": "1055",
"journal-title": "Int J Biochem Cell Biol",
"key": "427_CR106",
"unstructured": "Puddu P, Borghi P, Gessani S, Valenti P, Belardelli F, Seganti L (1998) Antiviral effect of bovine lactoferrin saturated with metal ions on early steps of human immunodeficiency virus type 1 infection. Int J Biochem Cell Biol 30:1055–1062. https://doi.org/10.1016/s1357-2725(98)00066-1",
"volume": "30",
"year": "1998"
},
{
"DOI": "10.1016/j.biochi.2008.05.005",
"author": "P Puddu",
"doi-asserted-by": "publisher",
"first-page": "11",
"journal-title": "Biochimie",
"key": "427_CR107",
"unstructured": "Puddu P, Valenti P, Gessani S (2009) Immunomodulatory effects of lactoferrin on antigen presenting cells. Biochimie 91:11–18. https://doi.org/10.1016/j.biochi.2008.05.005",
"volume": "91",
"year": "2009"
},
{
"DOI": "10.1371/journal.pone.0022504",
"author": "P Puddu",
"doi-asserted-by": "publisher",
"journal-title": "PLoS ONE",
"key": "427_CR108",
"unstructured": "Puddu P, Latorre D, Carollo M, Catizone A, Ricci G, Valenti P, Gessani S (2011) Bovine lactoferrin counteracts toll-like receptor mediated activation signals in antigen presenting cells. PLoS ONE 6:e22504. https://doi.org/10.1371/journal.pone.0022504",
"volume": "6",
"year": "2011"
},
{
"DOI": "10.1016/0304-4165(94)00126-i",
"author": "ZY Qian",
"doi-asserted-by": "publisher",
"first-page": "25",
"issue": "1",
"journal-title": "Biochim Biophys Acta",
"key": "427_CR109",
"unstructured": "Qian ZY, Jollès P, Migliore-Samour D, Fiat AM (1995) Isolation and characterization of sheep lactoferrin, an inhibitor of platelet aggregation and comparison with human lactoferrin. Biochim Biophys Acta 1243(1):25–32. https://doi.org/10.1016/0304-4165(94)00126-i",
"volume": "1243",
"year": "1995"
},
{
"DOI": "10.1016/j.cmet.2012.03.018",
"author": "B Qiao",
"doi-asserted-by": "publisher",
"first-page": "918",
"journal-title": "Cell Metab",
"key": "427_CR110",
"unstructured": "Qiao B, Sugianto P, Fung E, Del-Castillo-Rueda A, Moran-Jimenez MJ, Ganz T (2012) Nemeth E (2012) Hepcidin-induced endocytosis of ferroportin is dependent on ferroportin ubiquitination. Cell Metab 15:918–924. https://doi.org/10.1016/j.cmet.2012.03.018",
"volume": "15",
"year": "2012"
},
{
"DOI": "10.3390/ijms18091985",
"author": "L Rosa",
"doi-asserted-by": "publisher",
"first-page": "1985",
"issue": "9",
"journal-title": "Int J Mol Sci",
"key": "427_CR111",
"unstructured": "Rosa L, Cutone A, Lepanto MS, Paesano R, Valenti P (2017) Lactoferrin: a natural glycoprotein involved in iron and inflammatory homeostasis. Int J Mol Sci 18(9):1985. https://doi.org/10.3390/ijms18091985",
"volume": "18",
"year": "2017"
},
{
"DOI": "10.3390/jcm10184276",
"author": "L Rosa",
"doi-asserted-by": "publisher",
"first-page": "4276",
"issue": "18",
"journal-title": "J Clin Med",
"key": "427_CR112",
"unstructured": "Rosa L, Tripepi G, Naldi E, Aimati M, Santangeli S, Venditto F, Caldarelli M, Valenti P (2021) Ambulatory COVID-19 patients treated with lactoferrin as a supplementary antiviral agent: a preliminary study. J Clin Med 10(18):4276. https://doi.org/10.3390/jcm10184276",
"volume": "10",
"year": "2021"
},
{
"DOI": "10.1002/eji.200324218",
"author": "H Sano",
"doi-asserted-by": "publisher",
"first-page": "2894",
"issue": "10",
"journal-title": "Eur J Immunol",
"key": "427_CR113",
"unstructured": "Sano H, Nagai K, Tsutsumi H, Kuroki Y (2003) Lactoferrin and surfactant protein a exhibit distinct binding specificity to F protein and differently modulate respiratory syncytial virus infection. Eur J Immunol 33(10):2894–2902. https://doi.org/10.1002/eji.200324218",
"volume": "33",
"year": "2003"
},
{
"DOI": "10.5530/ijrhs.8.1.3",
"author": "G Serrano",
"doi-asserted-by": "publisher",
"first-page": "8",
"issue": "1",
"journal-title": "Int J Res Health Sci",
"key": "427_CR114",
"unstructured": "Serrano G, Kochergina I, Albors A, Diaz E, Oroval M, Hueso G, Serrano JM (2020) Liposomal lactoferrin as potential preventative and cure for COVID-19. Int J Res Health Sci 8(1):8–15. https://doi.org/10.5530/ijrhs.8.1.3",
"volume": "8",
"year": "2020"
},
{
"DOI": "10.1016/j.bbrc.2015.10.067",
"author": "MP Sherman",
"doi-asserted-by": "publisher",
"first-page": "766",
"issue": "4",
"journal-title": "Biochem Biophys Res Commun",
"key": "427_CR115",
"unstructured": "Sherman MP, Pritzl CJ, Xia C, Miller MM, Zaghouani H, Hahm B (2015) Lactoferrin acts as an adjuvant during influenza vaccination of neonatal mice. Biochem Biophys Res Commun 467(4):766–770. https://doi.org/10.1016/j.bbrc.2015.10.067",
"volume": "467",
"year": "2015"
},
{
"DOI": "10.1007/BF01718201",
"author": "K Shimizu",
"doi-asserted-by": "publisher",
"first-page": "1875",
"issue": "10",
"journal-title": "Arch Virol",
"key": "427_CR116",
"unstructured": "Shimizu K, Matsuzawa H, Okada K, Tazume S, Dosako S, Kawasaki Y, Hashimoto K, Koga Y (1996) Lactoferrin-mediated protection of the host from murine cytomegalovirus infection by a T-cell-dependent augmentation of natural killer cell activity. Arch Virol 141(10):1875–1889. https://doi.org/10.1007/BF01718201",
"volume": "141",
"year": "1996"
},
{
"DOI": "10.1099/jmm.0.46018-0",
"author": "K Shin",
"doi-asserted-by": "publisher",
"first-page": "717",
"issue": "Pt 8",
"journal-title": "J Med Microbiol",
"key": "427_CR117",
"unstructured": "Shin K, Wakabayashi H, Yamauchi K, Teraguchi S, Tamura Y, Kurokawa M, Shiraki K (2005) Effects of orally administered bovine lactoferrin and lactoperoxidase on influenza virus infection in mice. J Med Microbiol 54(Pt 8):717–723. https://doi.org/10.1099/jmm.0.46018-0",
"volume": "54",
"year": "2005"
},
{
"DOI": "10.1080/10408398.2021.1895063",
"author": "M Sienkiewicz",
"doi-asserted-by": "publisher",
"first-page": "1",
"journal-title": "Crit Rev Food Sci Nutr",
"key": "427_CR118",
"unstructured": "Sienkiewicz M, Jaśkiewicz A, Tarasiuk A, Fichna J (2021) Lactoferrin: an overview of its main functions, immunomodulatory and antimicrobial role, and clinical significance. Crit Rev Food Sci Nutr 8:1–18. https://doi.org/10.1080/10408398.2021.1895063",
"volume": "8",
"year": "2021"
},
{
"DOI": "10.1016/j.bbi.2016.05.015",
"author": "RJ Simpson",
"doi-asserted-by": "publisher",
"first-page": "10",
"journal-title": "Brain Behav Immun",
"key": "427_CR119",
"unstructured": "Simpson RJ (2016) Aging and inflammation: directing traffic through physical activity. Brain Behav Immun 56:10–11. https://doi.org/10.1016/j.bbi.2016.05.015",
"volume": "56",
"year": "2016"
},
{
"DOI": "10.1172/JCI141374",
"author": "P Skendros",
"doi-asserted-by": "publisher",
"first-page": "6151",
"issue": "11",
"journal-title": "J Clin Invest",
"key": "427_CR120",
"unstructured": "Skendros P, Mitsios A, Chrysanthopoulou A, Mastellos DC, Metallidis S, Rafailidis P, Ntinopoulou M, Sertaridou E, Tsironidou V, Tsigalou C, Tektonidou M, Konstantinidis T, Papagoras C, Mitroulis I, Germanidis G, Lambris JD, Ritis K (2020) Complement and tissue factor-enriched neutrophil extracellular traps are key drivers in COVID-19 immunothrombosis. J Clin Invest 130(11):6151–6157. https://doi.org/10.1172/JCI141374",
"volume": "130",
"year": "2020"
},
{
"DOI": "10.1007/978-1-4615-2548-6_3",
"author": "G Spik",
"doi-asserted-by": "publisher",
"first-page": "21",
"journal-title": "Adv Exp Med Biol",
"key": "427_CR121",
"unstructured": "Spik G, Coddeville B, Mazurier J, Bourne Y, Cambillaut C, Montreuil J (1994) Primary and three-dimensional structure of lactotransferrin (lactoferrin) glycans. Adv Exp Med Biol 357:21–32. https://doi.org/10.1007/978-1-4615-2548-6_3",
"volume": "357",
"year": "1994"
},
{
"DOI": "10.1016/j.cct.2013.03.010",
"author": "BM Stefanescu",
"doi-asserted-by": "publisher",
"first-page": "33",
"issue": "2",
"journal-title": "Contemp Clin Trials",
"key": "427_CR122",
"unstructured": "Stefanescu BM, Hétu C, Slaughter JC, O’Shea TM, Shetty AK (2013) A pilot study of Biotene OralBalance® gel for oral care in mechanically ventilated preterm neonates. Contemp Clin Trials 35(2):33–39. https://doi.org/10.1016/j.cct.2013.03.010",
"volume": "35",
"year": "2013"
},
{
"DOI": "10.3390/nu12092562",
"author": "F Superti",
"doi-asserted-by": "publisher",
"first-page": "2562",
"issue": "9",
"journal-title": "Nutrients",
"key": "427_CR123",
"unstructured": "Superti F (2020) Lactoferrin from bovine milk: a protective companion for Life. Nutrients 12(9):2562. https://doi.org/10.3390/nu12092562",
"volume": "12",
"year": "2020"
},
{
"DOI": "10.1007/s004300050049",
"author": "F Superti",
"doi-asserted-by": "publisher",
"first-page": "83",
"issue": "2–3",
"journal-title": "Med Microbiol Immunol",
"key": "427_CR124",
"unstructured": "Superti F, Ammendolia MG, Valenti P, Seganti L (1997) Antirotaviral activity of milk proteins: lactoferrin prevents rotavirus infection in the enterocyte-like cell line HT-29. Med Microbiol Immunol 186(2–3):83–91. https://doi.org/10.1007/s004300050049",
"volume": "186",
"year": "1997"
},
{
"DOI": "10.1016/s0304-4165(01)00178-7",
"author": "F Superti",
"doi-asserted-by": "publisher",
"first-page": "107",
"issue": "2–3",
"journal-title": "Biochim Biophys Acta",
"key": "427_CR125",
"unstructured": "Superti F, Siciliano R, Rega B, Giansanti F, Valenti P, Antonini G (2001) Involvement of bovine lactoferrin metal saturation, sialic acid and protein fragments in the inhibition of rotavirus infection. Biochim Biophys Acta 1528(2–3):107–115. https://doi.org/10.1016/s0304-4165(01)00178-7",
"volume": "1528",
"year": "2001"
},
{
"DOI": "10.1089/aid.1996.12.769",
"author": "PJ Swart",
"doi-asserted-by": "publisher",
"first-page": "769",
"issue": "9",
"journal-title": "AIDS Res Hum Retroviruses",
"key": "427_CR126",
"unstructured": "Swart PJ, Kuipers ME, Smit C, Pauwels R, deBéthune MP, de Clercq E, Meijer DK, Huisman JG (1996) Antiviral effects of milk proteins: acylation results in polyanionic compounds with potent activity against human immunodeficiency virus types 1 and 2 in vitro. AIDS Res Hum Retroviruses 12(9):769–775. https://doi.org/10.1089/aid.1996.12.769",
"volume": "12",
"year": "1996"
},
{
"DOI": "10.1038/nrgastro.2015.94",
"author": "G Szabo",
"doi-asserted-by": "publisher",
"first-page": "387",
"issue": "7",
"journal-title": "Nat Rev Gastroenterol Hepatol",
"key": "427_CR127",
"unstructured": "Szabo G, Petrasek J (2015) Inflammasome activation and function in liver disease. Nat Rev Gastroenterol Hepatol 12(7):387–400. https://doi.org/10.1038/nrgastro.2015.94",
"volume": "12",
"year": "2015"
},
{
"DOI": "10.1186/1743-422X-7-330",
"author": "SH Taha",
"doi-asserted-by": "publisher",
"first-page": "330",
"journal-title": "Virol J",
"key": "427_CR128",
"unstructured": "Taha SH, Mehrez MA, Sitohy MZ, Abou Dawood AG, Abd-El Hamid MM, Kilany WH (2010) Effectiveness of esterified whey proteins fractions against Egyptian Lethal Avian Influenza A (H5N1). Virol J 7:330. https://doi.org/10.1186/1743-422X-7-330",
"volume": "7",
"year": "2010"
},
{
"DOI": "10.1111/j.1349-7006.1999.tb00756.x",
"author": "K Tanaka",
"doi-asserted-by": "publisher",
"first-page": "367",
"issue": "4",
"journal-title": "Jpn J Cancer Res",
"key": "427_CR129",
"unstructured": "Tanaka K, Ikeda M, Nozaki A, Kato N, Tsuda H, Saito S, Sekihara H (1999) Lactoferrin inhibits hepatitis C virus viremia in patients with chronic hepatitis C: a pilot study. Jpn J Cancer Res 90(4):367–371. https://doi.org/10.1111/j.1349-7006.1999.tb00756.x",
"volume": "90",
"year": "1999"
},
{
"DOI": "10.1101/2020.10.23.350348",
"author": "X Tang",
"doi-asserted-by": "publisher",
"journal-title": "bioRxiv",
"key": "427_CR130",
"unstructured": "Tang X, Yang M, Duan Z, Liao Z, Liu L, Cheng R, Fang M, Wang G, Liu H, Xu J, Kamau PM, Zhang Z, Yang L, Zhao X, Peng X, Lai R (2020) Transferrin receptor is another receptor for SARS-CoV-2 entry. bioRxiv. https://doi.org/10.1101/2020.10.23.350348",
"year": "2020"
},
{
"DOI": "10.1016/j.jtho.2020.02.010",
"author": "S Tian",
"doi-asserted-by": "publisher",
"first-page": "700",
"issue": "5",
"journal-title": "J Thorac Oncol",
"key": "427_CR131",
"unstructured": "Tian S, Hu W, Niu L, Liu H, Xu H, Xiao SY (2020) Pulmonary pathology of early-phase 2019 novel Coronavirus (COVID-19) Pneumonia in two patients with lung cancer. J Thorac Oncol 15(5):700–704. https://doi.org/10.1016/j.jtho.2020.02.010",
"volume": "15",
"year": "2020"
},
{
"DOI": "10.1016/j.ijantimicag.2005.02.011",
"author": "A Tinari",
"doi-asserted-by": "publisher",
"first-page": "433",
"issue": "5",
"journal-title": "Int J Antimicrob Agents",
"key": "427_CR132",
"unstructured": "Tinari A, Pietrantoni A, Ammendolia MG, Valenti P, Superti F (2005) Inhibitory activity of bovine lactoferrin against echovirus induced programmed cell death in vitro. Int J Antimicrob Agents 25(5):433–438. https://doi.org/10.1016/j.ijantimicag.2005.02.011",
"volume": "25",
"year": "2005"
},
{
"DOI": "10.1007/s00011-020-01413-2",
"author": "S Toldo",
"doi-asserted-by": "publisher",
"first-page": "7",
"issue": "1",
"journal-title": "Inflamm Res",
"key": "427_CR133",
"unstructured": "Toldo S, Bussani R, Nuzzi V, Bonaventura A, Mauro AG, Cannatà A, Pillappa R, Sinagra G, Nana-Sinkam P, Sime P, Abbate A (2021) Inflammasome formation in the lungs of patients with fatal COVID-19. Inflamm Res 70(1):7–10. https://doi.org/10.1007/s00011-020-01413-2",
"volume": "70",
"year": "2021"
},
{
"DOI": "10.1111/j.1349-7006.2006.00274.x",
"author": "H Ueno",
"doi-asserted-by": "publisher",
"first-page": "1105",
"issue": "10",
"journal-title": "Cancer Sci",
"key": "427_CR134",
"unstructured": "Ueno H, Sato T, Yamamoto S, Tanaka K, Ohkawa S, Takagi H, Yokosuka O, Furuse J, Saito H, Sawaki A, Kasugai H, Osaki Y, Fujiyama S, Sato K, Wakabayashi K, Okusaka T (2006) Randomized, double-blind, placebo-controlled trial of bovine lactoferrin in patients with chronic hepatitis C. Cancer Sci 97(10):1105–1110. https://doi.org/10.1111/j.1349-7006.2006.00274.x",
"volume": "97",
"year": "2006"
},
{
"DOI": "10.1007/s00018-005-5372-0",
"author": "P Valenti",
"doi-asserted-by": "publisher",
"first-page": "2576",
"issue": "22",
"journal-title": "Cell Mol Life Sci",
"key": "427_CR135",
"unstructured": "Valenti P, Antonini G (2005) Lactoferrin: an important host defence against microbial and viral attack. Cell Mol Life Sci 62(22):2576–2587. https://doi.org/10.1007/s00018-005-5372-0",
"volume": "62",
"year": "2005"
},
{
"DOI": "10.3389/fimmu.2018.00376",
"author": "P Valenti",
"doi-asserted-by": "publisher",
"first-page": "376",
"journal-title": "Front Immunol",
"key": "427_CR136",
"unstructured": "Valenti P, Rosa L, Capobianco D, Lepanto MS, Schiavi E, Cutone A, Paesano R, Mastromarino P (2018) Role of Lactobacilli and Lactoferrin in the Mucosal Cervicovaginal Defense. Front Immunol 9:376. https://doi.org/10.3389/fimmu.2018.00376",
"volume": "9",
"year": "2018"
},
{
"DOI": "10.3389/fimmu.2018.02677",
"author": "M van Splunter",
"doi-asserted-by": "publisher",
"first-page": "2677",
"journal-title": "GOS Vitamin d Front Immunol",
"key": "427_CR137",
"unstructured": "van Splunter M, Perdijk O, Fick-Brinkhof H, Feitsma AL, Floris-Vollenbroek EG, Meijer B, Brugman S, Savelkoul HFJ, van Hoffen E, van Neerven RJJ (2018) Bovine lactoferrin enhances TLR7-mediated responses in plasmacytoid dendritic cells in elderly women: results from a nutritional intervention study with bovine lactoferrin. GOS Vitamin d Front Immunol 9:2677. https://doi.org/10.3389/fimmu.2018.02677",
"volume": "9",
"year": "2018"
},
{
"DOI": "10.1111/j.1432-1033.2003.03965.x",
"author": "HA van Veen",
"doi-asserted-by": "publisher",
"first-page": "678",
"issue": "4",
"journal-title": "Eur J Biochem",
"key": "427_CR138",
"unstructured": "van Veen HA, Geerts ME, van Berkel PH, Nuijens JH (2004) The role of N-linked glycosylation in the protection of human and bovine lactoferrin against tryptic proteolysis. Eur J Biochem 271(4):678–684. https://doi.org/10.1111/j.1432-1033.2003.03965.x",
"volume": "271",
"year": "2004"
},
{
"DOI": "10.1182/blood-2006-07-033969",
"author": "MV Verga Falzacappa",
"doi-asserted-by": "publisher",
"first-page": "353",
"issue": "1",
"journal-title": "Blood",
"key": "427_CR139",
"unstructured": "Verga Falzacappa MV, Vujic Spasic M, Kessler R, Stolte J, Hentze MW, Muckenthaler MU (2007) STAT3 mediates hepatic hepcidin expression and its inflammatory stimulation. Blood 109(1):353–358. https://doi.org/10.1182/blood-2006-07-033969",
"volume": "109",
"year": "2007"
},
{
"DOI": "10.1016/j.ctim.2012.12.006",
"author": "L Vitetta",
"doi-asserted-by": "publisher",
"first-page": "164",
"issue": "3",
"journal-title": "Complement Ther Med",
"key": "427_CR140",
"unstructured": "Vitetta L, Coulson S, Beck SL, Gramotnev H, Du S, Lewis S (2013) The clinical efficacy of a bovine lactoferrin/whey protein Ig-rich fraction (Lf/IgF) for the common cold: a double blind randomized study. Complement Ther Med 21(3):164–171. https://doi.org/10.1016/j.ctim.2012.12.006",
"volume": "21",
"year": "2013"
},
{
"DOI": "10.1111/aor.13812",
"author": "VD Vlahakos",
"doi-asserted-by": "publisher",
"first-page": "163",
"issue": "2",
"journal-title": "Artif Organs",
"key": "427_CR141",
"unstructured": "Vlahakos VD, Marathias KP, Arkadopoulos N, Vlahakos DV (2021) Hyperferritinemia in patients with COVID-19: an opportunity for iron chelation? Artif Organs 45(2):163–167. https://doi.org/10.1111/aor.13812",
"volume": "45",
"year": "2021"
},
{
"DOI": "10.1271/bbb.68.537",
"author": "H Wakabayashi",
"doi-asserted-by": "publisher",
"first-page": "537",
"issue": "3",
"journal-title": "Biosci Biotechnol Biochem",
"key": "427_CR142",
"unstructured": "Wakabayashi H, Kurokawa M, Shin K, Teraguchi S, Tamura Y, Shiraki K (2004) Oral lactoferrin prevents body weight loss and increases cytokine responses during herpes simplex virus type 1 infection of mice. Biosci Biotechnol Biochem 68(3):537–544. https://doi.org/10.1271/bbb.68.537",
"volume": "68",
"year": "2004"
},
{
"DOI": "10.1016/j.jiac.2014.08.003",
"author": "H Wakabayashi",
"doi-asserted-by": "publisher",
"first-page": "666",
"journal-title": "J Infect Chemother",
"key": "427_CR143",
"unstructured": "Wakabayashi H, Oda H, Yamauchi K, Abe F (2014) Lactoferrin for prevention of common viral infections. J Infect Chemother 20:666–671. https://doi.org/10.1016/j.jiac.2014.08.003",
"volume": "20",
"year": "2014"
},
{
"DOI": "10.1038/s41392-021-00818-7",
"author": "D Wan",
"doi-asserted-by": "publisher",
"first-page": "406",
"issue": "1",
"journal-title": "Signal Transduct Target Ther",
"key": "427_CR144",
"unstructured": "Wan D, Du T, Hong W, Chen L, Que H, Lu S, Peng X (2021) Neurological complications and infection mechanism of SARS-COV-2. Signal Transduct Target Ther 6(1):406. https://doi.org/10.1038/s41392-021-00818-7",
"volume": "6",
"year": "2021"
},
{
"DOI": "10.1084/jem.20140625",
"author": "J Wang",
"doi-asserted-by": "publisher",
"first-page": "2397",
"issue": "12",
"journal-title": "J Exp Med",
"key": "427_CR145",
"unstructured": "Wang J, Li F, Wei H, Lian ZX, Sun R, Tian Z (2014a) Respiratory influenza virus infection induces intestinal immune injury via microbiota-mediated Th17 cell-dependent inflammation. J Exp Med 211(12):2397–2410. https://doi.org/10.1084/jem.20140625",
"volume": "211",
"year": "2014"
},
{
"DOI": "10.1038/ni.3015",
"author": "X Wang",
"doi-asserted-by": "publisher",
"first-page": "1126",
"issue": "12",
"journal-title": "Nat Immunol",
"key": "427_CR146",
"unstructured": "Wang X, Jiang W, Yan Y, Gong T, Han J, Tian Z, Zhou R (2014b) RNA viruses promote activation of the NLRP3 inflammasome through a RIP1-RIP3-DRP1 signaling pathway. Nat Immunol 15(12):1126–1133. https://doi.org/10.1038/ni.3015",
"volume": "15",
"year": "2014"
},
{
"DOI": "10.1016/j.cell.2020.03.045",
"author": "Q Wang",
"doi-asserted-by": "publisher",
"first-page": "894",
"issue": "4",
"journal-title": "Cell",
"key": "427_CR147",
"unstructured": "Wang Q, Zhang Y, Wu L, Niu S, Song C, Zhang Z, Lu G, Qiao C, Hu Y, Yuen KY, Wang Q, Zhou H, Yan J, Qi J (2020) Structural and functional basis of SARS-CoV-2 entry by using human ACE2. Cell 181(4):894-904.e9. https://doi.org/10.1016/j.cell.2020.03.045",
"volume": "181",
"year": "2020"
},
{
"DOI": "10.1007/s10719-021-10029-5",
"author": "X Wang",
"doi-asserted-by": "publisher",
"first-page": "689",
"issue": "6",
"journal-title": "Glycoconjugate J",
"key": "427_CR148",
"unstructured": "Wang X, Yue L, Dang L, Yang J, Chen Z, Wang X, Shu J, Li Z (2021) Role of sialylated glycans on bovine lactoferrin against influenza virus. Glycoconjugate J 38(6):689–696. https://doi.org/10.1007/s10719-021-10029-5",
"volume": "38",
"year": "2021"
},
{
"DOI": "10.1016/j.tube.2011.10.019",
"author": "KJ Welsh",
"doi-asserted-by": "publisher",
"first-page": "S105",
"issue": "Suppl 1",
"journal-title": "Tuberculosis (edinb)",
"key": "427_CR149",
"unstructured": "Welsh KJ, Hwang SA, Boyd S, Kruzel ML, Hunter RL, Actor JK (2011) Influence of oral lactoferrin on Mycobacterium tuberculosis induced immunopathology. Tuberculosis (edinb) 91(Suppl 1):S105-113. https://doi.org/10.1016/j.tube.2011.10.019",
"volume": "91",
"year": "2011"
},
{
"DOI": "10.1080/08916934.2021.1913581",
"author": "L Wenzhong",
"doi-asserted-by": "publisher",
"first-page": "213",
"issue": "4",
"journal-title": "Autoimmunity",
"key": "427_CR150",
"unstructured": "Wenzhong L, Hualan L (2021) COVID-19: captures iron and generates reactive oxygen species to damage the human immune system. Autoimmunity 54(4):213–224. https://doi.org/10.1080/08916934.2021.1913581",
"volume": "54",
"year": "2021"
},
{
"DOI": "10.1111/cei.13108",
"author": "L Wisgrill",
"doi-asserted-by": "publisher",
"first-page": "315",
"issue": "3",
"journal-title": "Clin Exp Immunol",
"key": "427_CR151",
"unstructured": "Wisgrill L, Wessely I, Spittler A, Förster-Waldl E, Berger A, Sadeghi K (2018) Human lactoferrin attenuates the proinflammatory response of neonatal monocyte-derived macrophages. Clin Exp Immunol 192(3):315–324. https://doi.org/10.1111/cei.13108",
"volume": "192",
"year": "2018"
},
{
"DOI": "10.1182/blood-2006-06-027631",
"author": "DM Wrighting",
"doi-asserted-by": "publisher",
"first-page": "3204",
"issue": "9",
"journal-title": "Blood",
"key": "427_CR152",
"unstructured": "Wrighting DM, Andrews NC (2006) Interleukin-6 induces hepcidin expression through STAT3. Blood 108(9):3204–3209. https://doi.org/10.1182/blood-2006-06-027631",
"volume": "108",
"year": "2006"
},
{
"DOI": "10.1006/abbi.1995.1139",
"author": "HF Wu",
"doi-asserted-by": "publisher",
"first-page": "85",
"issue": "1",
"journal-title": "Arch Biochem Biophys",
"key": "427_CR153",
"unstructured": "Wu HF, Monroe DM, Church FC (1995) Characterization of the glycosaminoglycan-binding region of lactoferrin. Arch Biochem Biophys 317(1):85–92. https://doi.org/10.1006/abbi.1995.1139",
"volume": "317",
"year": "1995"
},
{
"DOI": "10.1038/s41586-020-2008-3",
"author": "F Wu",
"doi-asserted-by": "publisher",
"first-page": "265",
"issue": "7798",
"journal-title": "Nature",
"key": "427_CR154",
"unstructured": "Wu F, Zhao S, Yu B, Chen YM, Wang W, Song ZG, Hu Y, Tao ZW, Tian JH, Pei YY, Yuan ML, Zhang YL, Dai FH, Liu Y, Wang QM, Zheng JJ, Xu L, Holmes EC, Zhang YZ (2020a) A new coronavirus associated with human respiratory disease in China. Nature 579(7798):265–269. https://doi.org/10.1038/s41586-020-2008-3",
"volume": "579",
"year": "2020"
},
{
"DOI": "10.1016/S2468-1253(20)30083-2",
"author": "Y Wu",
"doi-asserted-by": "publisher",
"first-page": "434",
"issue": "5",
"journal-title": "Lancet Gastroenterol Hepatol",
"key": "427_CR155",
"unstructured": "Wu Y, Guo C, Tang L, Hong Z, Zhou J, Dong X, Yin H, Xiao Q, Tang Y, Qu X, Kuang L, Fang X, Mishra N, Lu J, Shan H, Jiang G, Huang X (2020b) Prolonged presence of SARS-CoV-2 viral RNA in faecal samples. Lancet Gastroenterol Hepatol 5(5):434–435. https://doi.org/10.1016/S2468-1253(20)30083-2",
"volume": "5",
"year": "2020"
},
{
"DOI": "10.1021/acs.jafc.9b08094",
"author": "S Xu",
"doi-asserted-by": "publisher",
"first-page": "3132",
"issue": "10",
"journal-title": "J Agric Food Chem",
"key": "427_CR156",
"unstructured": "Xu S, Fan F, Liu H, Cheng S, Tu M, Du M (2020a) Novel anticoagulant peptide from lactoferrin binding thrombin at the active site and Exosite-I. J Agric Food Chem 68(10):3132–3139. https://doi.org/10.1021/acs.jafc.9b08094",
"volume": "68",
"year": "2020"
},
{
"DOI": "10.1016/S2213-2600(20)30076-X",
"author": "Z Xu",
"doi-asserted-by": "publisher",
"first-page": "420",
"issue": "4",
"journal-title": "Lancet Respir Med",
"key": "427_CR157",
"unstructured": "Xu Z, Shi L, Wang Y, Zhang J, Huang L, Zhang C, Liu S, Zhao P, Liu H, Zhu L, Tai Y, Bai C, Gao T, Song J, Xia P, Dong J, Zhao J, Wang FS (2020b) Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med 8(4):420–422. https://doi.org/10.1016/S2213-2600(20)30076-X",
"volume": "8",
"year": "2020"
},
{
"DOI": "10.1248/jhs.56.613",
"author": "H Yamamoto",
"doi-asserted-by": "publisher",
"first-page": "613",
"journal-title": "J Health Sci",
"key": "427_CR158",
"unstructured": "Yamamoto H, Ura Y, Tanemura M, Koyama A, Takano S, Uematsu J, Kawano M, Tsurudome M, O’Brien M, Komada H (2010) Inhibitory effect of bovine lactoferrin on human parainfluenza virus type 2 infection. J Health Sci 54:613–617. https://doi.org/10.1248/jhs.56.613",
"volume": "54",
"year": "2010"
},
{
"author": "MH Yen",
"first-page": "395",
"issue": "4",
"journal-title": "Chang Gung Med J",
"key": "427_CR159",
"unstructured": "Yen MH, Chiu CH, Huang YC, Lin TY (2011) Effects of lactoferrin-containing formula in the prevention of enterovirus and rotavirus infection and impact on serum cytokine levels: a randomized trial. Chang Gung Med J 34(4):395–402",
"volume": "34",
"year": "2011"
},
{
"DOI": "10.1128/JVI.71.8.5997-6002.1997",
"author": "M Yi",
"doi-asserted-by": "publisher",
"first-page": "5997",
"issue": "8",
"journal-title": "J Virol",
"key": "427_CR160",
"unstructured": "Yi M, Kaneko S, Yu DY, Murakami S (1997) Hepatitis C virus envelope proteins bind lactoferrin. J Virol 71(8):5997–6002. https://doi.org/10.1128/JVI.71.8.5997-6002.1997",
"volume": "71",
"year": "1997"
},
{
"DOI": "10.1186/s40168-017-0386-z",
"author": "S Yildiz",
"doi-asserted-by": "publisher",
"first-page": "9",
"issue": "1",
"journal-title": "Microbiome",
"key": "427_CR161",
"unstructured": "Yildiz S, Mazel-Sanchez B, Kandasamy M, Manicassamy B, Schmolke M (2018) Influenza A virus infection impacts systemic microbiota dynamics and causes quantitative enteric dysbiosis. Microbiome 6(1):9. https://doi.org/10.1186/s40168-017-0386-z",
"volume": "6",
"year": "2018"
},
{
"DOI": "10.1056/NEJMoa2001017",
"author": "N Zhu",
"doi-asserted-by": "publisher",
"first-page": "727",
"issue": "8",
"journal-title": "N Engl J Med",
"key": "427_CR162",
"unstructured": "Zhu N, Zhang D, Wang W, Li X, Yang B, Song J, Zhao X, Huang B, Shi W, Lu R, Niu P, Zhan F, Ma X, Wang D, Xu W, Wu G, Gao GF, Tan W, China Novel Coronavirus Investigating and Research Team (2020) A novel coronavirus from patients with pneumonia in China, 2019. N Engl J Med 382(8):727–733. https://doi.org/10.1056/NEJMoa2001017",
"volume": "382",
"year": "2020"
},
{
"DOI": "10.1016/j.intimp.2021.107571",
"author": "M Zimecki",
"doi-asserted-by": "publisher",
"journal-title": "Int Immunopharmacol",
"key": "427_CR163",
"unstructured": "Zimecki M, Actor JK, Kruzel ML (2021) The potential for Lactoferrin to reduce SARS-CoV-2 induced cytokine storm. Int Immunopharmacol 95:107571. https://doi.org/10.1016/j.intimp.2021.107571",
"volume": "95",
"year": "2021"
},
{
"DOI": "10.1182/blood-2014-01-550467",
"author": "S Zumerle",
"doi-asserted-by": "publisher",
"first-page": "3646",
"issue": "23",
"journal-title": "Blood",
"key": "427_CR164",
"unstructured": "Zumerle S, Mathieu JR, Delga S, Heinis M, Viatte L, Vaulont S, Peyssonnaux C (2014) Targeted disruption of hepcidin in the liver recapitulates the hemochromatotic phenotype. Blood 123(23):3646–3650. https://doi.org/10.1182/blood-2014-01-550467",
"volume": "123",
"year": "2014"
},
{
"DOI": "10.1038/s41598-020-80010-z",
"author": "Y Zuo",
"doi-asserted-by": "publisher",
"first-page": "1580",
"issue": "1",
"journal-title": "Sci Rep",
"key": "427_CR165",
"unstructured": "Zuo Y, Warnock M, Harbaugh A, Yalavarthi S, Gockman K, Zuo M, Madison JA, Knight JS, Kanthi Y, Lawrence DA (2021) Plasma tissue plasminogen activator and plasminogen activator inhibitor-1 in hospitalized COVID-19 patients. Sci Rep 11(1):1580. https://doi.org/10.1038/s41598-020-80010-z",
"volume": "11",
"year": "2021"
},
{
"DOI": "10.1074/jbc.RA118.003145",
"author": "A Zwirzitz",
"doi-asserted-by": "publisher",
"first-page": "8600",
"issue": "22",
"journal-title": "J Biol Chem",
"key": "427_CR166",
"unstructured": "Zwirzitz A, Reiter M, Skrabana R, Ohradanova-Repic A, Majdic O, Gutekova M, Cehlar O, Petrovčíková E, Kutejova E, Stanek G, Stockinger H, Leksa V (2018) Lactoferrin is a natural inhibitor of plasminogen activation. J Biol Chem 293(22):8600–8613. https://doi.org/10.1074/jbc.RA118.003145",
"volume": "293",
"year": "2018"
}
],
"reference-count": 166,
"references-count": 166,
"relation": {},
"resource": {
"primary": {
"URL": "https://link.springer.com/10.1007/s10534-022-00427-z"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"Metals and Alloys",
"General Agricultural and Biological Sciences",
"General Biochemistry, Genetics and Molecular Biology",
"Biomaterials"
],
"subtitle": [],
"title": "An overview on in vitro and in vivo antiviral activity of lactoferrin: its efficacy against SARS-CoV-2 infection",
"type": "journal-article",
"update-policy": "http://dx.doi.org/10.1007/springer_crossmark_policy"
}