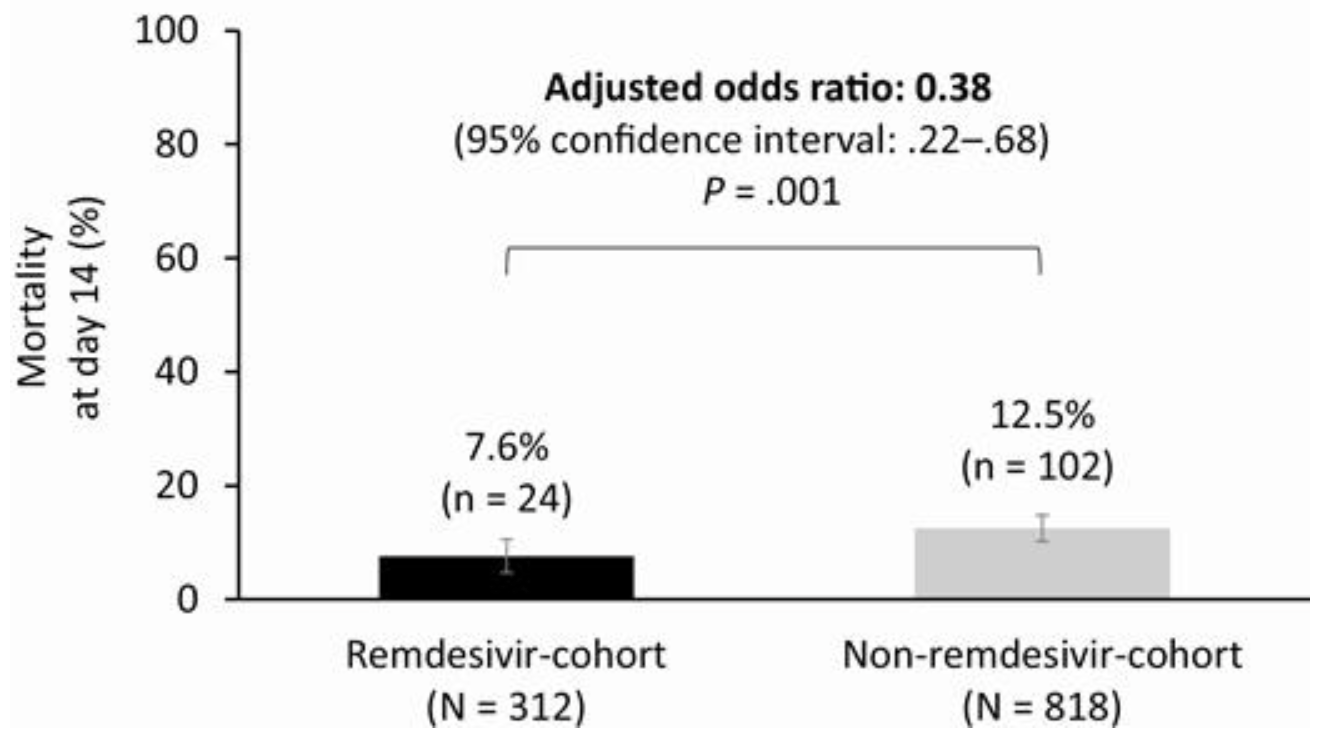
Remdesivir for Severe Coronavirus Disease 2019 (COVID-19) Versus a Cohort Receiving Standard of Care
et al., Clinical Infectious Diseases, doi:10.1093/cid/ciaa1041, Jul 2020
Comparative analysis between remdesivir trial GS-US-540-5773 and a retrospective SOC cohort with similar inclusion criteria, showing lower mortality and higher recovery at day 14 with remdesivir.
Gérard, Zhou, Wu, Kamo, Choi, Kim show increased risk of acute kidney injury, Leo, Briciu, Muntean, Petrov, Arch show increased risk of liver injury, Negru, Cheng, Mohammed, Kwok, Zhu show increased risk of cardiac disorders, and Kwok, Merches, Akinci, Tang, Bagheri show increased risk of mitochondrial toxicity with remdesivir.
Standard of Care (SOC) for COVID-19 in the study country,
the USA, is very poor with very low average efficacy for approved treatments21.
Only expensive, high-profit treatments were approved for early treatment. Low-cost treatments were excluded, reducing the probability of early treatment due to access and cost barriers, and eliminating complementary and synergistic benefits seen with many low-cost treatments.
|
risk of death, 58.8% lower, RR 0.41, p = 0.001, treatment 24 of 312 (7.7%), control 102 of 818 (12.5%), odds ratio converted to relative risk, weighted multivariable logistic regression, day 14.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
1.
Gérard et al., Remdesivir and Acute Renal Failure: A Potential Safety Signal From Disproportionality Analysis of the WHO Safety Database, Clinical Pharmacology & Therapeutics, doi:10.1002/cpt.2145.
2.
Zhou et al., Acute Kidney Injury and Drugs Prescribed for COVID-19 in Diabetes Patients: A Real-World Disproportionality Analysis, Frontiers in Pharmacology, doi:10.3389/fphar.2022.833679.
3.
Wu et al., Acute Kidney Injury Associated With Remdesivir: A Comprehensive Pharmacovigilance Analysis of COVID-19 Reports in FAERS, Frontiers in Pharmacology, doi:10.3389/fphar.2022.692828.
4.
Kamo et al., Association of Antiviral Drugs for the Treatment of COVID-19 With Acute Renal Failure, In Vivo, doi:10.21873/invivo.13637.
5.
Choi et al., Comparative effectiveness of combination therapy with nirmatrelvir–ritonavir and remdesivir versus monotherapy with remdesivir or nirmatrelvir–ritonavir in patients hospitalised with COVID-19: a target trial emulation study, The Lancet Infectious Diseases, doi:10.1016/S1473-3099(24)00353-0.
6.
Kim et al., Investigating the Safety Profile of Fast‐Track COVID‐19 Drugs Using the FDA Adverse Event Reporting System Database: A Comparative Observational Study, Pharmacoepidemiology and Drug Safety, doi:10.1002/pds.70043.
7.
Leo et al., Hepatocellular liver injury in hospitalized patients affected by COVID-19: Presence of different risk factors at different time points, Digestive and Liver Disease, doi:10.1016/j.dld.2021.12.014.
8.
Briciu et al., Evolving Clinical Manifestations and Outcomes in COVID-19 Patients: A Comparative Analysis of SARS-CoV-2 Variant Waves in a Romanian Hospital Setting, Pathogens, doi:10.3390/pathogens12121453.
9.
Muntean et al., Effects of COVID-19 on the Liver and Mortality in Patients with SARS-CoV-2 Pneumonia Caused by Delta and Non-Delta Variants: An Analysis in a Single Centre, Pharmaceuticals, doi:10.3390/ph17010003.
10.
Petrov et al., The Effect of Potentially Hepatotoxic Medicinal Products on Alanine Transaminase Levels in COVID-19 Patients: A Case–Control Study, Safety and Risk of Pharmacotherapy, doi:10.30895/2312-7821-2025-458.
11.
Arch et al., Evaluation of the effectiveness of remdesivir in treating severe COVID-19 using data from the ISARIC WHO Clinical Characterisation Protocol UK: a prospective, national cohort study, medRxiv, doi:10.1101/2021.06.18.21259072.
12.
Negru et al., Comparative Pharmacovigilance Analysis of Approved and Repurposed Antivirals for COVID-19: Insights from EudraVigilance Data, Biomedicines, doi:10.3390/biomedicines13061387.
13.
Cheng et al., Cardiovascular Safety of COVID-19 Treatments: A Disproportionality Analysis of Adverse Event Reports from the WHO VigiBase, Infectious Diseases and Therapy, doi:10.1007/s40121-025-01225-z.
14.
Mohammed et al., Bradycardia associated with remdesivir treatment in coronavirus disease 2019 patients: A propensity score-matched analysis, Medicine, doi:10.1097/MD.0000000000044501.
15.
Kwok et al., Remdesivir induces persistent mitochondrial and structural damage in human induced pluripotent stem cell-derived cardiomyocytes, Cardiovascular Research, doi:10.1093/cvr/cvab311.
16.
Zhu et al., Cardiovascular Risks of COVID-19 Therapeutics: Integrated Analysis of FAERS, Electronic Health Records, and Transcriptomics, Pharmaceuticals, doi:10.3390/ph19040574.
17.
Merches et al., The potential of remdesivir to affect function, metabolism and proliferation of cardiac and kidney cells in vitro, Archives of Toxicology, doi:10.1007/s00204-022-03306-1.
18.
Akinci et al., Elucidation of remdesivir cytotoxicity pathways through genome-wide CRISPR-Cas9 screening and transcriptomics, bioRxiv, doi:10.1101/2020.08.27.270819.
19.
Tang et al., Modeling drug-induced mitochondrial toxicity with human primary cardiomyocytes, Science China Life Sciences, doi:10.1007/s11427-023-2369-3.
Olender et al., 24 Jul 2020, retrospective, USA, peer-reviewed, 33 authors.
Remdesivir for Severe Coronavirus Disease 2019 (COVID-19) Versus a Cohort Receiving Standard of Care
Clinical Infectious Diseases, doi:10.1093/cid/ciaa1041
Background. We compared the efficacy of the antiviral agent, remdesivir, versus standard-of-care treatment in adults with severe coronavirus disease 2019 (COVID-19) using data from a phase 3 remdesivir trial and a retrospective cohort of patients with severe COVID-19 treated with standard of care. Methods. GS-US-540-5773 is an ongoing phase 3, randomized, open-label trial comparing two courses of remdesivir (remdesivircohort). GS-US-540-5807 is an ongoing real-world, retrospective cohort study of clinical outcomes in patients receiving standardof-care treatment (non-remdesivir-cohort). Inclusion criteria were similar between studies: patients had confirmed severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection, were hospitalized, had oxygen saturation ≤94% on room air or required supplemental oxygen, and had pulmonary infiltrates. Stabilized inverse probability of treatment weighted multivariable logistic regression was used to estimate the treatment effect of remdesivir versus standard of care. The primary endpoint was the proportion of patients with recovery on day 14, dichotomized from a 7-point clinical status ordinal scale. A key secondary endpoint was mortality. Results. After the inverse probability of treatment weighting procedure, 312 and 818 patients were counted in the remdesivirand non-remdesivir-cohorts, respectively. At day 14, 74.4% of patients in the remdesivir-cohort had recovered versus 59.0% in the non-remdesivir-cohort (adjusted odds ratio [aOR] 2.03: 95% confidence interval [CI]: 1.34-3.08, P < .001). At day 14, 7.6% of patients in the remdesivir-cohort had died versus 12.5% in the non-remdesivir-cohort (aOR 0.38, 95% CI: .22-.68, P = .001). Conclusions. In this comparative analysis, by day 14, remdesivir was associated with significantly greater recovery and 62% reduced odds of death versus standard-of-care treatment in patients with severe COVID-19. clinical Trials Registration. NCT04292899 and EUPAS34303.
Supplementary Data Supplementary materials are available at Clinical Infectious Diseases online. Consisting of data provided by the authors to benefit the reader, the posted materials are not copyedited and are the sole responsibility of the authors, so questions or comments should be addressed to the corresponding author. Acknowledgments. The authors extend their gratitude to the participants and their families, participating sites, site investigators, and study staff. The incredible work of the site investigators and their healthcare teams in times of enormous challenges and personal risk cannot be overstated. The full list of site investigators is provided in Supplemental Digital Content 1). The authors dedicate this work to those that are or have been ill with COVID-19 and to the memory of healthcare workers who have given their lives in the care of these patients. Medical writing support was provided by Ryan Woodrow and Emma McConnell from Aspire Scientific Ltd (Bollington, UK), and was funded by Gilead Sciences, Inc., Foster City, CA, USA. Gilead Sciences makes available anonymized individual patient data upon request or as required by law or regulation with external researchers. Approval of such requests is at Gilead Sciences's discretion and is dependent on the nature of the request, the merit of the research proposed, the availability of the data, and the intended use of the data. Data requests should be sent to datarequest@gilead.com. Data from GS-US-540-5807 can..
References
Beigel, Tomashek, Dodd, Remdesivir for the treatment of Covid-19-preliminary report, N Engl J Med, doi:10.1056/NEJMoa2007764
Borba, Val, Sampaio, Effect of high vs low doses of chloroquine diphosphate as adjunctive therapy for patients hospitalized with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection: a randomized clinical trial, JAMA Netw Open
Cao, Wang, Wen, A trial of lopinavir-ritonavir in adults hospitalized with severe Covid-19, N Engl J Med
Chen, Hu, Zhang, Efficacy of hydroxychloroquine in patients with COVID-19: results of a randomized clinical trial, medRxiv, doi:10.1101/2020.03.22.20040758
Choy, Wong, Kaewpreedee, Remdesivir, lopinavir, emetine, and homoharringtonine inhibit SARS-CoV-2 replication in vitro, Antiviral Res
Gao, Tian, Yang, Breakthrough: chloroquine phosphate has shown apparent efficacy in treatment of COVID-19 associated pneumonia in clinical studies, Biosci Trends
Gautret, Lagier, Parola, Hydroxychloroquine and azithromycin as a treatment of COVID-19: results of an open-label non-randomized clinical trial, Int J Antimicrob Agents
Goldman, Lye, Hui, Remdesivir for 5 or 10 days in patients with severe Covid-19, N Engl J Med, doi:10.1056/NEJMoa2015301
Gordon, Tchesnokov, Feng, Porter, Götte, The antiviral compound remdesivir potently inhibits RNA-dependent RNA polymerase from Middle East respiratory syndrome coronavirus, J Biol Chem
Grein, Ohmagari, Shin, Compassionate use of remdesivir for patients with severe Covid-19, N Engl J Med, doi:10.1056/NEJMoa2007016
Li, Xu, Yu, Risk factors for severity and mortality in adult COVID-19 inpatients in Wuhan, J Allergy Clin Immunol, doi:10.1016/j.jaci.2020.04.006
Liu, Xu, Zhang, Patients of COVID-19 may benefit from sustained lopinavir-combined regimen and the increase of eosinophil may predict the outcome of COVID-19 progression, Int J Infect Dis
Mehra, Desai, Ruschitzka, Patel, Hydroxychloroquine or chloroquine with or without a macrolide for treatment of COVID-19: a multinational registry analysis, Lancet, doi:10.1016/S0140-6736(20)31180â•fi6
Norrie, Remdesivir for COVID-19: challenges of underpowered studies, Lancet
Robins, Hernán, Brumback, Marginal structural models and causal inference in epidemiology, Epidemiology
Verity, Okell, Dorigatti, Estimates of the severity of coronavirus disease 2019: a model-based analysis, Lancet Infect Dis
Wang, Cao, Zhang, Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro, Cell Res
Wang, Zhang, Du, Remdesivir in adults with severe COVID-19: a randomised, double-blind, placebo-controlled, multicentre trial, Lancet
Weiss, Murdoch, Clinical course and mortality risk of severe COVID-19, Lancet
Xu, Ross, Raebel, Shetterly, Blanchette et al., Use of stabilized inverse propensity scores as weights to directly estimate relative risk and its confidence intervals, Value Health
Ye, Luo, Xia, Clinical efficacy of lopinavir/ritonavir in the treatment of coronavirus disease 2019, Eur Rev Med Pharmacol Sci
Zhang, Wang, Jia, Risk factors for disease severity, unimprovement, and mortality in COVID-19 patients in Wuhan, China, Clin Microbiol Infect
Zhou, Yu, Du, Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study, Lancet
DOI record:
{
"DOI": "10.1093/cid/ciaa1041",
"ISSN": [
"1058-4838",
"1537-6591"
],
"URL": "http://dx.doi.org/10.1093/cid/ciaa1041",
"abstract": "<jats:title>Abstract</jats:title>\n <jats:sec>\n <jats:title>Background</jats:title>\n <jats:p>We compared the efficacy of the antiviral agent, remdesivir, versus standard-of-care treatment in adults with severe coronavirus disease 2019 (COVID-19) using data from a phase 3 remdesivir trial and a retrospective cohort of patients with severe COVID-19 treated with standard of care.</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Methods</jats:title>\n <jats:p>GS-US-540–5773 is an ongoing phase 3, randomized, open-label trial comparing two courses of remdesivir (remdesivir-cohort). GS-US-540–5807 is an ongoing real-world, retrospective cohort study of clinical outcomes in patients receiving standard-of-care treatment (non-remdesivir-cohort). Inclusion criteria were similar between studies: patients had confirmed severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection, were hospitalized, had oxygen saturation ≤94% on room air or required supplemental oxygen, and had pulmonary infiltrates. Stabilized inverse probability of treatment weighted multivariable logistic regression was used to estimate the treatment effect of remdesivir versus standard of care. The primary endpoint was the proportion of patients with recovery on day 14, dichotomized from a 7-point clinical status ordinal scale. A key secondary endpoint was mortality.</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Results</jats:title>\n <jats:p>After the inverse probability of treatment weighting procedure, 312 and 818 patients were counted in the remdesivir- and non-remdesivir-cohorts, respectively. At day 14, 74.4% of patients in the remdesivir-cohort had recovered versus 59.0% in the non-remdesivir-cohort (adjusted odds ratio [aOR] 2.03: 95% confidence interval [CI]: 1.34–3.08, P &lt; .001). At day 14, 7.6% of patients in the remdesivir-cohort had died versus 12.5% in the non-remdesivir-cohort (aOR 0.38, 95% CI: .22–.68, P = .001).</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Conclusions</jats:title>\n <jats:p>In this comparative analysis, by day 14, remdesivir was associated with significantly greater recovery and 62% reduced odds of death versus standard-of-care treatment in patients with severe COVID-19.</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Clinical Trials Registration</jats:title>\n <jats:p>NCT04292899 and EUPAS34303.</jats:p>\n </jats:sec>",
"author": [
{
"affiliation": [
{
"name": "Division of Infectious Diseases, Department of Internal Medicine, Columbia University Irving Medical Center, New York, New York, USA"
}
],
"family": "Olender",
"given": "Susan A",
"sequence": "first"
},
{
"affiliation": [
{
"name": "Houston Methodist, Houston, Texas, USA"
}
],
"family": "Perez",
"given": "Katherine K",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Division of Research, Kaiser Permanente Northern California, Oakland, California, USA"
}
],
"family": "Go",
"given": "Alan S",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Hackensack University Medical Center, Hackensack, New Jersey, USA"
}
],
"family": "Balani",
"given": "Bindu",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Ochsner Health System and Ochsner Clinical School, New Orleans, Louisiana, USA"
}
],
"family": "Price-Haywood",
"given": "Eboni G",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "NorthShore University Health System, Evanston, Illinois, USA"
}
],
"family": "Shah",
"given": "Nirav S",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Saint Barnabas Medical Center, RWJBarnabas Medical Group, Livingston, New Jersey, USA"
}
],
"family": "Wang",
"given": "Su",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Northwestern University Feinberg School of Medicine, Chicago, Illinois, USA"
}
],
"family": "Walunas",
"given": "Theresa L",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Rutgers New Jersey Medical School, Newark, New Jersey, USA"
}
],
"family": "Swaminathan",
"given": "Shobha",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Prime Healthcare Services, St Michael’s LLC, Newark, New Jersey, USA"
}
],
"family": "Slim",
"given": "Jihad",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "National Medical Center, Seoul, South Korea"
}
],
"family": "Chin",
"given": "BumSik",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "NEAT ID Foundation, CHU Saint Pierre, Brussels, Belgium"
}
],
"family": "De Wit",
"given": "Stéphane",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "NEAT ID Foundation, Chelsea and Westminster Hospital, London, United Kingdom"
},
{
"name": "School of Medicine, Moi University, Eldoret, Kenya"
}
],
"family": "Ali",
"given": "Shamim M",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Hospital Clinic de Barcelona, IDIBAPS, University of Barcelona, Barcelona, Spain"
}
],
"family": "Soriano Viladomiu",
"given": "Alex",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Hoag Memorial Hospital Presbyterian, Newport Beach, California, USA"
}
],
"family": "Robinson",
"given": "Philip",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Baylor University Medical Center Dallas, Dallas, Texas, USA"
},
{
"name": "Baylor Scott and White Health, Dallas, Texas, USA"
}
],
"family": "Gottlieb",
"given": "Robert L",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Princess Margaret Hospital, Kwai Chung, Hong Kong"
}
],
"family": "Tsang",
"given": "Tak Yin Owen",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Gilead Sciences, Foster City, California, USA"
}
],
"family": "Lee",
"given": "I-Heng",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Gilead Sciences, Causeway Bay, Hong Kong"
}
],
"family": "Hu",
"given": "Hao",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Gilead Sciences, Foster City, California, USA"
}
],
"family": "Haubrich",
"given": "Richard H",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Gilead Sciences, Foster City, California, USA"
}
],
"family": "Chokkalingam",
"given": "Anand P",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Gilead Sciences, Foster City, California, USA"
}
],
"family": "Lin",
"given": "Lanjia",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Gilead Sciences, Foster City, California, USA"
}
],
"family": "Zhong",
"given": "Lijie",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Gilead Sciences, Foster City, California, USA"
}
],
"family": "Bekele",
"given": "B Nebiyou",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Gilead Sciences, Foster City, California, USA"
}
],
"family": "Mera-Giler",
"given": "Robertino",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Gilead Sciences, Stockley Park, Uxbridge, United Kingdom"
}
],
"family": "Phulpin",
"given": "Chloé",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Gilead Sciences, Stockley Park, Uxbridge, United Kingdom"
}
],
"family": "Edgar",
"given": "Holly",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Gilead Sciences, Foster City, California, USA"
}
],
"family": "Gallant",
"given": "Joel",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Gilead Sciences, Madrid, Spain"
}
],
"family": "Diaz-Cuervo",
"given": "Helena",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Gilead Sciences, Foster City, California, USA"
}
],
"family": "Smith",
"given": "Lindsey E",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Gilead Sciences, Foster City, California, USA"
}
],
"family": "Osinusi",
"given": "Anu O",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Gilead Sciences, Foster City, California, USA"
}
],
"family": "Brainard",
"given": "Diana M",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Hospital La Paz, IdiPAZ, Madrid, Spain"
}
],
"family": "Bernardino",
"given": "Jose I",
"sequence": "additional"
},
{
"affiliation": [],
"name": "GS-US-540–5773 and GS-US-540–5807 Investigators",
"sequence": "additional"
}
],
"container-title": "Clinical Infectious Diseases",
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2020,
7,
22
]
],
"date-time": "2020-07-22T19:22:47Z",
"timestamp": 1595445767000
},
"deposited": {
"date-parts": [
[
2021,
12,
10
]
],
"date-time": "2021-12-10T11:30:33Z",
"timestamp": 1639135833000
},
"funder": [
{
"DOI": "10.13039/100005564",
"doi-asserted-by": "publisher",
"name": "Gilead Sciences"
}
],
"indexed": {
"date-parts": [
[
2024,
3,
26
]
],
"date-time": "2024-03-26T12:00:42Z",
"timestamp": 1711454442830
},
"is-referenced-by-count": 101,
"issue": "11",
"issued": {
"date-parts": [
[
2020,
7,
24
]
]
},
"journal-issue": {
"issue": "11",
"published-online": {
"date-parts": [
[
2020,
7,
24
]
]
},
"published-print": {
"date-parts": [
[
2021,
12,
6
]
]
}
},
"language": "en",
"license": [
{
"URL": "https://creativecommons.org/licenses/by-nc-nd/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2020,
7,
24
]
],
"date-time": "2020-07-24T00:00:00Z",
"timestamp": 1595548800000
}
}
],
"link": [
{
"URL": "http://academic.oup.com/cid/advance-article-pdf/doi/10.1093/cid/ciaa1041/33776118/ciaa1041.pdf",
"content-type": "application/pdf",
"content-version": "am",
"intended-application": "syndication"
},
{
"URL": "https://academic.oup.com/cid/article-pdf/73/11/e4166/41607771/ciaa1041.pdf",
"content-type": "application/pdf",
"content-version": "vor",
"intended-application": "syndication"
},
{
"URL": "https://academic.oup.com/cid/article-pdf/73/11/e4166/41607771/ciaa1041.pdf",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "286",
"original-title": [],
"page": "e4166-e4174",
"prefix": "10.1093",
"published": {
"date-parts": [
[
2020,
7,
24
]
]
},
"published-online": {
"date-parts": [
[
2020,
7,
24
]
]
},
"published-other": {
"date-parts": [
[
2021,
12,
1
]
]
},
"published-print": {
"date-parts": [
[
2021,
12,
6
]
]
},
"publisher": "Oxford University Press (OUP)",
"reference": [
{
"author": "Johns Hopkins University and Medicine",
"key": "2021121011165717000_CIT0001"
},
{
"DOI": "10.1074/jbc.AC120.013056",
"article-title": "The antiviral compound remdesivir potently inhibits RNA-dependent RNA polymerase from Middle East respiratory syndrome coronavirus",
"author": "Gordon",
"doi-asserted-by": "crossref",
"first-page": "4773",
"journal-title": "J Biol Chem",
"key": "2021121011165717000_CIT0002",
"volume": "295",
"year": "2020"
},
{
"author": "US Food & Drug Administration",
"key": "2021121011165717000_CIT0003"
},
{
"DOI": "10.1016/j.antiviral.2020.104786",
"article-title": "Remdesivir, lopinavir, emetine, and homoharringtonine inhibit SARS-CoV-2 replication in vitro",
"author": "Choy",
"doi-asserted-by": "crossref",
"first-page": "104786",
"journal-title": "Antiviral Res",
"key": "2021121011165717000_CIT0004",
"volume": "178",
"year": "2020"
},
{
"DOI": "10.1038/s41422-020-0282-0",
"article-title": "Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro",
"author": "Wang",
"doi-asserted-by": "crossref",
"first-page": "269",
"journal-title": "Cell Res",
"key": "2021121011165717000_CIT0005",
"volume": "30",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa2007016",
"article-title": "Compassionate use of remdesivir for patients with severe Covid-19",
"author": "Grein",
"doi-asserted-by": "crossref",
"journal-title": "N Engl J Med",
"key": "2021121011165717000_CIT0006",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa2015301",
"article-title": "Remdesivir for 5 or 10 days in patients with severe Covid-19",
"author": "Goldman",
"doi-asserted-by": "crossref",
"journal-title": "N Engl J Med",
"key": "2021121011165717000_CIT0007",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa2007764",
"article-title": "Remdesivir for the treatment of Covid-19—preliminary report",
"author": "Beigel",
"doi-asserted-by": "crossref",
"journal-title": "N Engl J Med",
"key": "2021121011165717000_CIT0008",
"year": "2020"
},
{
"DOI": "10.1016/S0140-6736(20)31022-9",
"article-title": "Remdesivir in adults with severe COVID-19: a randomised, double-blind, placebo-controlled, multicentre trial",
"author": "Wang",
"doi-asserted-by": "crossref",
"first-page": "1569",
"journal-title": "Lancet",
"key": "2021121011165717000_CIT0009",
"volume": "395",
"year": "2020"
},
{
"DOI": "10.1016/S0140-6736(20)31023-0",
"article-title": "Remdesivir for COVID-19: challenges of underpowered studies",
"author": "Norrie",
"doi-asserted-by": "crossref",
"first-page": "1525",
"journal-title": "Lancet",
"key": "2021121011165717000_CIT0010",
"volume": "395",
"year": "2020"
},
{
"DOI": "10.1097/00001648-200009000-00011",
"article-title": "Marginal structural models and causal inference in epidemiology",
"author": "Robins",
"doi-asserted-by": "crossref",
"first-page": "550",
"journal-title": "Epidemiology",
"key": "2021121011165717000_CIT0011",
"volume": "11",
"year": "2000"
},
{
"DOI": "10.1111/j.1524-4733.2009.00671.x",
"article-title": "Use of stabilized inverse propensity scores as weights to directly estimate relative risk and its confidence intervals",
"author": "Xu",
"doi-asserted-by": "crossref",
"first-page": "273",
"journal-title": "Value Health",
"key": "2021121011165717000_CIT0012",
"volume": "13",
"year": "2010"
},
{
"DOI": "10.1016/S0140-6736(20)30566-3",
"article-title": "Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study",
"author": "Zhou",
"doi-asserted-by": "crossref",
"first-page": "1054",
"journal-title": "Lancet",
"key": "2021121011165717000_CIT0013",
"volume": "395",
"year": "2020"
},
{
"DOI": "10.1016/S1473-3099(20)30243-7",
"article-title": "Estimates of the severity of coronavirus disease 2019: a model-based analysis",
"author": "Verity",
"doi-asserted-by": "crossref",
"first-page": "669",
"journal-title": "Lancet Infect Dis",
"key": "2021121011165717000_CIT0014",
"volume": "20",
"year": "2020"
},
{
"DOI": "10.5582/bst.2020.01047",
"article-title": "Breakthrough: chloroquine phosphate has shown apparent efficacy in treatment of COVID-19 associated pneumonia in clinical studies",
"author": "Gao",
"doi-asserted-by": "crossref",
"first-page": "72",
"journal-title": "Biosci Trends",
"key": "2021121011165717000_CIT0015",
"volume": "14",
"year": "2020"
},
{
"DOI": "10.1016/j.ijantimicag.2020.105949",
"article-title": "Hydroxychloroquine and azithromycin as a treatment of COVID-19: results of an open-label non-randomized clinical trial",
"author": "Gautret",
"doi-asserted-by": "crossref",
"first-page": "105949",
"journal-title": "Int J Antimicrob Agents",
"key": "2021121011165717000_CIT0016",
"volume": "56",
"year": "2020"
},
{
"article-title": "Efficacy of hydroxychloroquine in patients with COVID-19: results of a randomized clinical trial",
"author": "Chen",
"journal-title": "medRxiv",
"key": "2021121011165717000_CIT0017",
"year": "2020"
},
{
"DOI": "10.1016/j.ijid.2020.03.013",
"article-title": "Patients of COVID-19 may benefit from sustained lopinavir-combined regimen and the increase of eosinophil may predict the outcome of COVID-19 progression",
"author": "Liu",
"doi-asserted-by": "crossref",
"first-page": "183",
"journal-title": "Int J Infect Dis",
"key": "2021121011165717000_CIT0018",
"volume": "95",
"year": "2020"
},
{
"article-title": "Clinical efficacy of lopinavir/ritonavir in the treatment of coronavirus disease 2019",
"author": "Ye",
"first-page": "3390",
"journal-title": "Eur Rev Med Pharmacol Sci",
"key": "2021121011165717000_CIT0019",
"volume": "24",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa2001282",
"article-title": "A trial of lopinavir-ritonavir in adults hospitalized with severe Covid-19",
"author": "Cao",
"doi-asserted-by": "crossref",
"first-page": "1787",
"journal-title": "N Engl J Med",
"key": "2021121011165717000_CIT0020",
"volume": "382",
"year": "2020"
},
{
"DOI": "10.1001/jamanetworkopen.2020.8857",
"article-title": "Effect of high vs low doses of chloroquine diphosphate as adjunctive therapy for patients hospitalized with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection: a randomized clinical trial",
"author": "Borba",
"doi-asserted-by": "crossref",
"first-page": "e208857",
"journal-title": "JAMA Netw Open",
"key": "2021121011165717000_CIT0021",
"volume": "3",
"year": "2020"
},
{
"DOI": "10.1016/S0140-6736(20)30633-4",
"article-title": "Clinical course and mortality risk of severe COVID-19",
"author": "Weiss",
"doi-asserted-by": "crossref",
"first-page": "1014",
"journal-title": "Lancet",
"key": "2021121011165717000_CIT0022",
"volume": "395",
"year": "2020"
},
{
"DOI": "10.1016/j.jaci.2020.04.006",
"article-title": "Risk factors for severity and mortality in adult COVID-19 inpatients in Wuhan",
"author": "Li",
"doi-asserted-by": "crossref",
"journal-title": "J Allergy Clin Immunol",
"key": "2021121011165717000_CIT0023",
"year": "2020"
},
{
"DOI": "10.1016/j.cmi.2020.04.012",
"article-title": "Risk factors for disease severity, unimprovement, and mortality in COVID-19 patients in Wuhan, China",
"author": "Zhang",
"doi-asserted-by": "crossref",
"first-page": "767",
"journal-title": "Clin Microbiol Infect",
"key": "2021121011165717000_CIT0024",
"volume": "26",
"year": "2020"
},
{
"article-title": "Hydroxychloroquine or chloroquine with or without a macrolide for treatment of COVID-19: a multinational registry analysis",
"author": "Mehra",
"journal-title": "Lancet",
"key": "2021121011165717000_CIT0025",
"year": "2020"
}
],
"reference-count": 25,
"references-count": 25,
"relation": {},
"resource": {
"primary": {
"URL": "https://academic.oup.com/cid/article/73/11/e4166/5876045"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"Infectious Diseases",
"Microbiology (medical)"
],
"subtitle": [],
"title": "Remdesivir for Severe Coronavirus Disease 2019 (COVID-19) Versus a Cohort Receiving Standard of Care",
"type": "journal-article",
"volume": "73"
}