Low Vitamin D Status at Admission as a Risk Factor for Poor Survival in Hospitalized Patients With COVID-19: An Italian Retrospective Study
et al., Journal of the American College of Nutrition, doi:10.1080/07315724.2021.1877580, Feb 2021
Vitamin D for COVID-19
8th treatment shown to reduce risk in
October 2020, now with p < 0.00000000001 from 137 studies, recognized in 18 countries.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
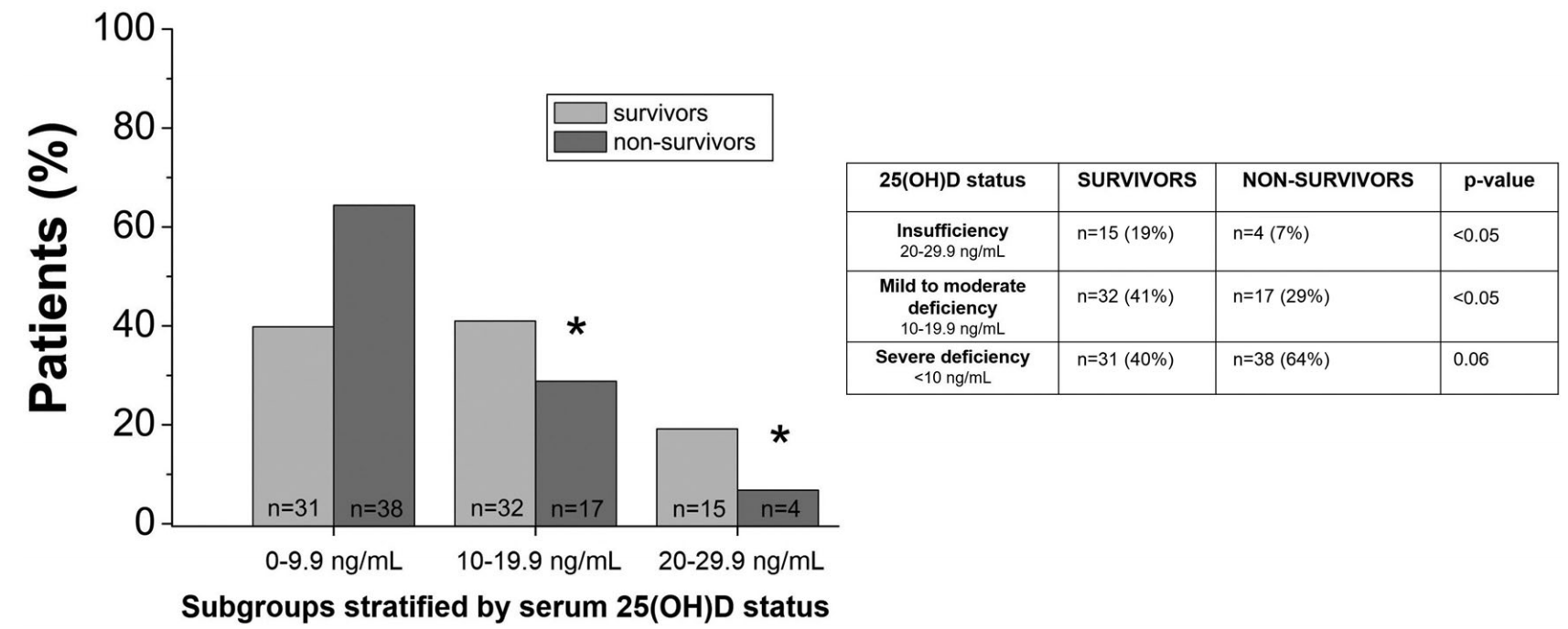
Retrospective 137 hospitalized patients in Italy. All patients had low vitamin D levels, and lower levels were associated with higher mortality.
In multivariate logistic regression, vitamin D levels were significantly inversely associated with mortality (OR 0.91 [0.85-0.98] p=0.01).
This is the 48th of 232 COVID-19 sufficiency studies for vitamin D, which collectively show higher levels reduce risk with p<0.0000000001.
|
risk of death, 54.8% lower, RR 0.45, p = 0.046, high D levels 4 of 19 (21.1%), low D levels 55 of 118 (46.6%), NNT 3.9, >20ng/mL.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Infante et al., 18 Feb 2021, retrospective, Italy, peer-reviewed, 11 authors.
Low Vitamin D Status at Admission as a Risk Factor for Poor Survival in Hospitalized Patients With COVID-19: An Italian Retrospective Study
Journal of the American Nutrition Association, doi:10.1080/07315724.2021.1877580
Objective: Preliminary findings suggest a relationship between lower serum 25-hydroxyvitamin D [25(OH)D] levels and incidence and severity of COVID-19. The aim of this study was to evaluate the relationship between vitamin D status at admission and different markers of inflammation, coagulation, and sepsis in hospitalized patients with COVID-19. Method: We conducted a retrospective study on 137 consecutive patients with SARS-CoV-2 infection and available data on serum 25(OH)D levels, who were admitted to our Institution between March 1 and April 30, 2020. Patients were divided into two groups: survivors (n ¼ 78; 57%) and non-survivors (n ¼ 59; 43%). Results: At admission, all patients showed hypovitaminosis D. Median total serum 25(OH)D levels at admission were significantly higher in survivors than non-survivors (12 ng/mL vs 8 ng/mL; p < 0.01). Non-survivors exhibited significantly higher median levels of white blood cell (WBC) count, neutrophil-to-lymphocyte count ratio (NLR), high-sensitivity C-reactive protein (hsCRP), ferritin, interleukin 6 (IL-6), D-dimer, fibrinogen, and procalcitonin (PCT) compared to survivors at three different time points during hospitalization. In a multivariate analysis performed by a logistic regression model, serum 25(OH)D levels were significantly inversely associated with risk of COVID-19-related in-hospital mortality (odds ratio, 0.91; 95% confidence interval, 0.85-0.98; p ¼ 0.01). According to receiver operating characteristic curve analysis, hsCRP, NLR, ferritin, and D-dimer were the best predictive biomarkers for poor prognosis of COVID-19, whereas IL-6, PCT, fibrinogen, 25(OH)D, WBC count, and tumor necrosis factor alpha (TNF-a) may serve as supportive biomarkers for worse clinical course of the disease. Conclusions: We found a markedly high prevalence (100%) of hypovitaminosis D in patients admitted to hospital with COVID-19, suggesting a possible role of low vitamin D status in increasing the risk of SARS-CoV-2 infection and subsequent hospitalization. The inverse association between serum 25(OH)D levels and risk of in-hospital mortality observed in our cohort suggests that a lower vitamin D status upon admission may represent a modifiable and independent risk factor for poor prognosis in COVID-19.
Authors' contributions MI and MM designed the research project, wrote the paper, supervised the project, and equally contributed to the manuscript. AB collected and retrieved clinical data, analyzed results, and contributed to the research project. MP performed statistical analysis. SL and MN performed, collected, and retrieved biochemical data. SB, AF, MIa, MA, and VC supervised the research project and reviewed the manuscript. All authors edited the manuscript. No honorarium, grant, or other forms of payment were received by authors to write this manuscript.
Disclosure statement No author has potential conflicts of interest to disclose.
References
Annweiler, Corvaisier, Gautier, Dub Ee, Legrand et al., Vitamin D supplementation associated to better survival in hospitalized frail elderly COVID-19 patients: the GERIA-COVID quasi-experimental study, Nutrients, doi:10.3390/nu12113377
Baeke, Takiishi, Korf, Gysemans, Mathieu, Vitamin D: modulator of the immune system, Curr Opin Pharmacol, doi:10.1016/j.coph.2010.04.001
Baktash, Hosack, Patel, Shah, Kandiah et al., Vitamin D status and outcomes for hospitalised older patients with COVID-19
Berry, Hesketh, Power, Hypp€ Onen E, Vitamin D status has a linear association with seasonal infections and lung function in British adults, Br J Nutr, doi:10.1017/S0007114511001991
Boonstra, Barrat, Crain, Heath, Savelkoul et al., 1alpha,25-Dihydroxyvitamin d3 has a direct effect on naive CD4(þ) T cells to enhance the development of Th2 cells, J Immunol, doi:10.4049/jimmunol.167.9.4974
Bouillon, Lieben, Mathieu, Verstuyf, Carmeliet, Vitamin D action: lessons from VDR and Cyp27b1 null mice, Pediatr Endocrinol Rev
Brenner, Holleczek, Sch€ Ottker B, Vitamin D insufficiency and deficiency and mortality from respiratory diseases in a cohort of older adults: potential for limiting the death toll during and beyond the COVID-19 pandemic?, Nutrients, doi:10.3390/nu12082488
Cannell, Zasloff, Garland, Scragg, Giovannucci, On the epidemiology of influenza, Virol J, doi:10.1186/1743-422X-5-29
Caprio, Infante, Calanchini, Mammi, Fabbri, Vitamin D: not just the bone. Evidence for beneficial pleiotropic extraskeletal effects, Eat Weight Disord, doi:10.1007/s40519-016-0312-6
Caprio, Mammi, Rosano, Vitamin D: a novel player in endothelial function and dysfunction, Arch Med Sci
Carpagnano, Lecce, Vn Zito, Buonamico, Capozza, Vitamin D deficiency as a predictor of poor prognosis in patients with acute respiratory failure due to COVID-19, J Endocrinol Invest, doi:10.1007/s40618-020-01370-x
Cashman, Van Den Heuvel, Schoemaker, Dp, Macdonald et al., 25-Hydroxyvitamin D as a biomarker of vitamin D status and its modeling to inform strategies for prevention of vitamin D deficiency within the population, Adv Nutr, doi:10.3945/an.117.015578
Castillo, Costa, Barrios, Iaz, Opez Miranda et al., Effect of calcifediol treatment and best available therapy versus best available therapy on intensive care unit admission and mortality among patients hospitalized for COVID-19: a pilot randomized clinical study, J Steroid Biochem Mol Biol, doi:10.1016/j.jsbmb.2020.105751
Charoenngam, Holick, Immunologic effects of vitamin D on human health and disease, Nutrients, doi:10.3390/nu12072097
Chen, Sims, Chen, Gu, Chen et al., Modulatory effects of 1,25-dihydroxyvitamin D3 on human B cell differentiation, J Immunol, doi:10.4049/jimmunol.179.3.1634
D'avolio, Avataneo, Manca, Cusato, Nicol O et al., 25-Hydroxyvitamin D concentrations are lower in patients with positive PCR for SARS-CoV-2, Nutrients, doi:10.3390/nu12051359
Drincic, Armas, Van Diest, Heaney, Volumetric dilution, rather than sequestration best explains the low vitamin D status of obesity, Silver Spring), doi:10.1038/oby.2011.404
Drucker, Coronavirus infections and type 2 diabetes-shared pathways with therapeutic implications, Endocr Rev
Ebadi, Montano-Loza, Perspective: improving vitamin D status in the management of COVID-19, Eur J Clin Nutr, doi:10.1038/s41430-020-0661-0
Elkhwanky, Kummu, Piltonen, Laru, Morin-Papunen et al., Obesity represses CYP2R1, the vitamin D 25-hydroxylase, in the liver and extrahepatic tissues, JBMR Plus, doi:10.1002/jbm4.10397
Fabbri, Infante, Ricordi, Editorial -Vitamin D status: a key modulator of innate immunity and natural defense from acute viral respiratory infections, Eur Rev Med Pharmacol Sci, doi:10.26355/eurrev_202004_20876
Ferreira, Van Etten, Verstuyf, Waer, Overbergh et al., 1,25-Dihydroxyvitamin D3 alters murine dendritic cell behaviour in vitro and in vivo, Diabetes Metab Res Rev, doi:10.1002/dmrr.1275
Forget, Khalifa, Defour, Latinne, Van Pel et al., What is the normal value of the neutrophil-to-lymphocyte ratio?, BMC Res Notes, doi:10.1186/s13104-016-2335-5
Gan, Li, Li, Yang, Leucocyte subsets effectively predict the clinical outcome of patients with COVID-19 pneumonia: a retrospective case-control study, Front Public Health, doi:10.3389/fpubh.2020.00299
Gauzzi, Purificato, Donato, Wang, Daniel et al., Suppressive effect of 1alpha,25-dihydroxyvitamin D3 on type I IFN-mediated monocyte differentiation into dendritic cells: impairment of functional activities and chemotaxis, J Immunol, doi:10.4049/jimmunol.174.1.270
Ginde, Mansbach, Camargo, Association between serum 25-hydroxyvitamin D level and upper respiratory tract infection in the Third National Health and Nutrition Examination Survey, Arch Intern Med, doi:10.1001/archinternmed.2008.560
Glinsky, Tripartite combination of candidate pandemic mitigation agents: Vitamin D, quercetin, and estradiol manifest properties of medicinal agents for targeted mitigation of the COVID-19 pandemic defined by genomics-guided tracing of SARS-CoV-2 targets in human cells, Biomedicines, doi:10.3390/biomedicines8050129
Gombart, Borregaard, Koeffler, Human cathelicidin antimicrobial peptide (CAMP) gene is a direct target of the vitamin D receptor and is strongly up-regulated in myeloid cells by 1,25-dihydroxyvitamin D3, FASEB J, doi:10.1096/fj.04-3284com
Gombart, The vitamin D-antimicrobial peptide pathway and its role in protection against infection, Future Microbiol, doi:10.2217/fmb.09.87
Grant, Lahore, Mcdonnell, Baggerly, French et al., Evidence that vitamin D supplementation could reduce risk of influenza and COVID-19 infections and deaths, Nutrients, doi:10.3390/nu12040988
Griffin, Xing, Kumar, Vitamin D and its analogs as regulators of immune activation and antigen presentation, Annu Rev Nutr, doi:10.1146/annurev.nutr.23.011702.073114
Hern Andez, Nan, Fernandez-Ayala, Garc Ia-Unzueta, Andez-Hern Andez et al., Vitamin D status in hospitalized patients with SARS-CoV-2 infection, J Clin Endocrinol Metab, doi:10.1210/clinem/dgaa733
Holick, Binkley, Bischoff-Ferrari, Gordon, Hanley et al., Evaluation, treatment, and prevention of vitamin D deficiency: an Endocrine Society clinical practice guideline, J Clin Endocrinol Metab, doi:10.1210/jc.2011-0385
Holick, The vitamin D deficiency pandemic: approaches for diagnosis, treatment and prevention, Rev Endocr Metab Disord, doi:10.1007/s11154-017-9424-1
Hu, Han, Pei, Yin, Chen, Procalcitonin levels in COVID-19 patients, Int J Antimicrob Agents, doi:10.1016/j.ijantimicag.2020.106051
Huang, Wang, Li, Ren, Zhao et al., Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China, Lancet, doi:10.1016/S0140-6736(20)30183-5
Infante, Ricordi, Caprio, Fabbri, Hydroxychloroquine in the COVID-19 pandemic era: in pursuit of a rational use for prophylaxis of SARS-CoV-2 infection, Expert Rev Anti Infect Ther
Infante, Ricordi, Padilla, Alvarez, Linetsky et al., The role of vitamin D and omega-3 PUFAs in islet transplantation, Nutrients, doi:10.3390/nu11122937
Infante, Ricordi, Sanchez, Salzler, Padilla et al., Influence of vitamin D on islet autoimmunity and beta-cell function in type 1 diabetes, Nutrients, doi:10.3390/nu11092185
Jain, Chaurasia, Sengar, Singh, Mahor et al., Analysis of vitamin D level among asymptomatic and critically ill COVID-19 patients and its correlation with inflammatory markers, Sci Rep, doi:10.1038/s41598-020-77093-z
Jeffery, Burke, Mura, Zheng, Qureshi et al., 1,25-Dihydroxyvitamin D3 and IL-2 combine to inhibit T cell production of inflammatory cytokines and promote development of regulatory T cells expressing CTLA-4 and FoxP3, J Immunol, doi:10.4049/jimmunol.0803217
Kaufman, Niles, Kroll, Bi, Holick, SARS-CoV-2 positivity rates associated with circulating 25-hydroxyvitamin D levels, PLoS One, doi:10.1371/journal.pone.0239252
Korf, Wenes, Stijlemans, Takiishi, Robert et al., 1,25-Dihydroxyvitamin D3 curtails the inflammatory and T cell stimulatory capacity of macrophages through an IL-10-dependent mechanism, Immunobiology, doi:10.1016/j.imbio.2012.07.018
Laaksi, Ruohola, Tuohimaa, Auvinen, Haataja et al., An association of serum vitamin D concentrations < 40 nmol/L with acute respiratory tract infection in young Finnish men, Am J Clin Nutr
Li, Liu, Zhang, Xu, Dai et al., SARS-CoV-2 and viral sepsis: observations and hypotheses, Lancet, doi:10.1016/S0140-6736(20)30920-X
Lips, Cashman, Lamberg-Allardt, Bischoff-Ferrari, Obermayer-Pietsch et al., Current vitamin D status in European and Middle East countries and strategies to prevent vitamin D deficiency: a position statement of the European Calcified Tissue Society, Eur J Endocrinol, doi:10.1530/EJE-18-
Liu, Li, Liu, Liang, Wang et al., Longitudinal characteristics of lymphocyte responses and cytokine profiles in the peripheral blood of SARS-CoV-2 infected patients, EBioMedicine, doi:10.1016/j.ebiom.2020.102763
Liu, Shen, Wang, Ge, Fei et al., Prognostic significance of neutrophil-to-lymphocyte ratio in patients with sepsis: a prospective observational study, Mediators Inflamm, doi:10.1155/2016/8191254
Liu, Stenger, Li, Wenzel, Tan et al., Toll-like receptor triggering of a vitamin D-mediated human antimicrobial response, Science, doi:10.1126/science.1123933
Maclaughlin, Holick, Aging decreases the capacity of human skin to produce vitamin D3, J Clin Invest, doi:10.1172/JCI112134
Maghbooli, Sahraian, Ebrahimi, Pazoki, Kafan et al., Vitamin D sufficiency, a serum 25-hydroxyvitamin D at least 30 ng/mL reduced risk for adverse clinical outcomes in patients with COVID-19 infection, PLoS One, doi:10.1371/journal.pone.0239799
Mangalmurti, Hunter, Cytokine storms: understanding COVID-19, Immunity, doi:10.1016/j.immuni.2020.06.017
Martineau, Forouhi, Vitamin D for COVID-19: a case to answer?, Lancet Diabetes Endocrinol, doi:10.1016/S2213-8587(20)30268-0
Martineau, Jolliffe, Greenberg, Aloia, Bergman et al., Vitamin D supplementation to prevent acute respiratory infections: individual participant data metaanalysis, Health Technol Assess, doi:10.3310/hta23020
Meltzer, Best, Zhang, Vokes, Arora et al., Association of vitamin D status and other clinical characteristics with COVID-19 test results, JAMA Netw Open, doi:10.1001/jamanetworkopen.2020.19722
Mercola, Grant, Wagner, Evidence regarding vitamin D and risk of COVID-19 and its severity, Nutrients, doi:10.3390/nu12113361
Merzon, Tworowski, Gorohovski, Vinker, Cohen et al., Low plasma 25(OH) vitamin D level is associated with increased risk of COVID-19 infection: an Israeli population-based study, Febs J, doi:10.1111/febs.15495
Monkawa, Yoshida, Hayashi, Saruta, Identification of 25-hydroxyvitamin D3 1alpha-hydroxylase gene expression in macrophages, Kidney Int, doi:10.1046/j.1523-1755.2000.00202.x
Moore, June, Cytokine release syndrome in severe COVID-19, Science, doi:10.1126/science.abb8925
Nikolich-Zugich, Knox, Rios, Natt, Bhattacharya et al., SARS-CoV-2 and COVID-19 in older adults: what we may expect regarding pathogenesis, immune responses, and outcomes, Geroscience, doi:10.1007/s11357-020-00186-0
Nisio, Toni, Sabovic, Rocca, Filippis et al., Impaired release of vitamin D in dysfunctional adipose tissue: new cues on vitamin D supplementation in obesity, J Clin Endocrinol Metab, doi:10.1210/jc.2016-3591
Overbergh, Decallonne, Valckx, Verstuyf, Depovere et al., Identification and immune regulation of 25-hydroxyvitamin D-1-alpha-hydroxylase in murine macrophages, Clin Exp Immunol, doi:10.1046/j.1365-2249.2000.01204.x
Overbergh, Decallonne, Waer, Rutgeerts, Valckx et al., 1alpha,25-dihydroxyvitamin D3 induces an autoantigen-specific T-helper 1/Thelper 2 immune shift in NOD mice immunized with GAD65 (p524-543), Diabetes, doi:10.2337/diabetes.49.8.1301
Penna, Adorini, 1 Alpha,25-dihydroxyvitamin D3 inhibits differentiation, maturation, activation, and survival of dendritic cells leading to impaired alloreactive T cell activation, J Immunol, doi:10.4049/jimmunol.164.5.2405
Piemonti, Monti, Sironi, Fraticelli, Leone et al., Vitamin D3 affects differentiation, maturation, and function of human monocyte-derived dendritic cells, J Immunol, doi:10.4049/jimmunol.164.9.4443
Prietl, Treiber, Pieber, Amrein, Vitamin D and immune function, Nutrients, doi:10.3390/nu5072502
Provvedini, Tsoukas, Deftos, Manolagas, 1,25-dihydroxyvitamin D3 receptors in human leukocytes, Science, doi:10.1126/science.6310748
Radujkovic, Hippchen, Tiwari-Heckler, Dreher, Boxberger, Vitamin d deficiency and outcome of COVID-19 patients, Nutrients, doi:10.3390/nu12092757
Rastogi, Bhansali, Khare, Suri, Yaddanapudi et al., Short term, high-dose vitamin D supplementation for COVID-19 disease: a randomised, placebo-controlled, study (SHADE study), Postgrad Med J, doi:10.1136/postgradmedj-2020-139065
Richardson, Hirsch, Narasimhan, Crawford, Mcginn et al., Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City Area, JAMA, doi:10.1001/jama.2020.6775
Roizen, Long, Casella, 'lear, Caplan et al., Obesity decreases hepatic 25-hydroxylase activity causing low serum 25-hydroxyvitamin D, J Bone Miner Res, doi:10.1002/jbmr.3686
Rottoli, Bernante, Belvedere, Balsamo, Garelli et al., How important is obesity as a risk factor for respiratory failure, intensive care admission and death in hospitalised COVID-19 patients? Results from a single Italian centre, Eur J Endocrinol, doi:10.1530/EJE-20-0541
Sabetta, Depetrillo, Cipriani, Smardin, Burns et al., Serum 25-hydroxyvitamin d and the incidence of acute viral respiratory tract infections in healthy adults, PLoS One, doi:10.1371/journal.pone.0011088
Sattar, Mcinnes, Mcmurray, Obesity a risk factor for severe COVID-19 infection: multiple potential mechanisms, Circulation, doi:10.1161/CIRCULATIONAHA.120.047659
Saul, Mair, Ivens, Brown, Samuel et al., 1,25-Dihydroxyvitamin D3 restrains CD4þ T cell priming ability of CD11c þ dendritic cells by upregulating expression of CD31, Front Immunol, doi:10.3389/fimmu.2019.00600
Sigmundsdottir, Pan, Debes, Alt, Habtezion et al., DCs metabolize sunlight-induced vitamin D3 to 'program' T cell attraction to the epidermal chemokine CCL27, Nat Immunol, doi:10.1038/ni1433
Sisnieguez, Espeche, Salazar, Arterial hypertension and the risk of severity and mortality of COVID-19, Eur Respir J, doi:10.1183/13993003.01148-2020
Smet, Smet, Herroelen, Gryspeerdt, Martens, Serum 25(OH)D level on hospital admission associated with COVID-19 stage and mortality, Am J Clin Pathol, doi:10.1093/ajcp/aqaa252
Stoffels, Overbergh, Giulietti, Verlinden, Bouillon et al., Immune regulation of 25-hydroxyvitamin-D3-1alpha-hydroxylase in human monocytes, J Bone Miner Res, doi:10.1359/JBMR.050908
Takahashi, Nakayama, Horiuchi, Ohta, Komoriya et al., Human neutrophils express messenger RNA of vitamin D receptor and respond to 1alpha,25-dihydroxyvitamin D3, Immunopharmacol Immunotoxicol, doi:10.1081/iph-120014721
Tamara, Tahapary, Obesity as a predictor for a poor prognosis of COVID-19: a systematic review, Diabetes Metab Syndr, doi:10.1016/j.dsx.2020.05.020
Tang, Li, Wang, Sun, Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia, J Thromb Haemost, doi:10.1111/jth.14768
Valle, Kim-Schulze, Huang, Beckmann, Nirenberg et al., An inflammatory cytokine signature predicts COVID-19 severity and survival, Nat Med, doi:10.1038/s41591-020-1051-9
Wang, Nestel, Bourdeau, Nagai, Wang et al., Cutting edge: 1,25-dihydroxyvitamin D3 is a direct inducer of antimicrobial peptide gene expression, J Immunol, doi:10.4049/jimmunol.173.5.2909
White, Vitamin D metabolism and signaling in the immune system, Rev Endocr Metab Disord, doi:10.1007/s11154-011-9195-z
Wu, Mcgoogan, Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72 314 cases from the Chinese Center for Disease Control and Prevention, JAMA, doi:10.1001/jama.2020.2648
Xu, Soruri, Gieseler, Peters, 1,25-Dihydroxyvitamin D3 exerts opposing effects to IL-4 on MHC class-II antigen expression, accessory activity, and phagocytosis of human monocytes, Scand J Immunol, doi:10.1111/j.1365-3083.1993.tb03237.x
Yao, Liu, Tang, Prognostic role of neutrophil-lymphocyte ratio and platelet-lymphocyte ratio for hospital mortality in patients with AECOPD, COPD, doi:10.2147/COPD.S141760
Ye, Tang, Liao, Shaw, Deng et al., Does serum vitamin D level affect COVID-19 infection and its severity?-A case-control study, J Am Coll Nutr, doi:10.1080/07315724.2020.1826005
Ye, Wang, Mao, The pathogenesis and treatment of the 'Cytokine Storm' in COVID-19, J Infect, doi:10.1016/j.jinf.2020.03.037
Zaim, Chong, Sankaranarayanan, Harky, COVID-19 and multiorgan response, Curr Probl Cardiol, doi:10.1016/j.cpcardiol.2020.100618
Zdrenghea, Makrinioti, Bagacean, Bush, Johnston et al., Vitamin D modulation of innate immune responses to respiratory viral infections, Rev Med Virol
Zhang, Leung, Richers, Liu, Remigio et al., Vitamin D inhibits monocyte/macrophage proinflammatory cytokine production by targeting MAPK phosphatase-1, J Immunol, doi:10.4049/jimmunol.1102412
Zhang, Zhou, Guo, Song, Liu, 1,25-Dihydroxyvitamin D 3 promotes high glucose-induced M1 macrophage switching to M2 via the VDR-PPARc signaling pathway, Biomed Res Int
Zhou, Yu, Du, Fan, Liu et al., Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study, Lancet, doi:10.1016/S0140-6736(20)30566-3
DOI record:
{
"DOI": "10.1080/07315724.2021.1877580",
"ISSN": [
"2769-7061",
"2769-707X"
],
"URL": "http://dx.doi.org/10.1080/07315724.2021.1877580",
"alternative-id": [
"10.1080/07315724.2021.1877580"
],
"assertion": [
{
"label": "Peer Review Statement",
"name": "peerreview_statement",
"order": 1,
"value": "The publishing and review policy for this title is described in its Aims & Scope."
},
{
"URL": "http://www.tandfonline.com/action/journalInformation?show=aimsScope&journalCode=uacn21",
"label": "Aim & Scope",
"name": "aims_and_scope_url",
"order": 2,
"value": "http://www.tandfonline.com/action/journalInformation?show=aimsScope&journalCode=uacn21"
},
{
"group": {
"label": "Publication History",
"name": "publication_history"
},
"label": "Received",
"name": "received",
"order": 0,
"value": "2020-10-31"
},
{
"group": {
"label": "Publication History",
"name": "publication_history"
},
"label": "Revised",
"name": "revised",
"order": 1,
"value": "2021-01-08"
},
{
"group": {
"label": "Publication History",
"name": "publication_history"
},
"label": "Accepted",
"name": "accepted",
"order": 2,
"value": "2021-01-12"
},
{
"group": {
"label": "Publication History",
"name": "publication_history"
},
"label": "Published",
"name": "published",
"order": 3,
"value": "2021-02-18"
}
],
"author": [
{
"ORCID": "http://orcid.org/0000-0003-2032-8735",
"affiliation": [
{
"name": "Department of Systems Medicine, Diabetes Research Institute Federation (DRIF), CTO Hospital ASL Roma 2, University of Rome Tor Vergata, Rome, Italy"
},
{
"name": "UniCamillus, Saint Camillus International University of Health Sciences, Rome, Italy"
},
{
"name": "Network of Immunity in Infection, Malignancy and Autoimmunity (NIIMA), Universal Scientific Education and Research Network (USERN), Rome, Italy"
}
],
"authenticated-orcid": false,
"family": "Infante",
"given": "Marco",
"sequence": "first"
},
{
"ORCID": "http://orcid.org/0000-0001-9303-7993",
"affiliation": [
{
"name": "Department of Systems Medicine, University of Rome Tor Vergata, Rome, Italy"
},
{
"name": "Infectious Disease Clinic, Tor Vergata University Hospital (PTV), Rome, Italy"
}
],
"authenticated-orcid": false,
"family": "Buoso",
"given": "Andrea",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-3463-0268",
"affiliation": [
{
"name": "Clinical Biochemistry and Molecular Biology, Department of Experimental Medicine, Faculty of Medicine, University of Rome Tor Vergata, Rome, Italy"
},
{
"name": "Clinical Biochemistry Department, Tor Vergata University Hospital (PTV), Rome, Italy"
}
],
"authenticated-orcid": false,
"family": "Pieri",
"given": "Massimo",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-6355-6987",
"affiliation": [
{
"name": "Clinical Biochemistry and Molecular Biology, Department of Experimental Medicine, Faculty of Medicine, University of Rome Tor Vergata, Rome, Italy"
}
],
"authenticated-orcid": false,
"family": "Lupisella",
"given": "Santina",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-8702-1116",
"affiliation": [
{
"name": "Clinical Biochemistry and Molecular Biology, Department of Experimental Medicine, Faculty of Medicine, University of Rome Tor Vergata, Rome, Italy"
}
],
"authenticated-orcid": false,
"family": "Nuccetelli",
"given": "Marzia",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0003-1984-6834",
"affiliation": [
{
"name": "Clinical Biochemistry and Molecular Biology, Department of Experimental Medicine, Faculty of Medicine, University of Rome Tor Vergata, Rome, Italy"
},
{
"name": "Clinical Biochemistry Department, Tor Vergata University Hospital (PTV), Rome, Italy"
}
],
"authenticated-orcid": false,
"family": "Bernardini",
"given": "Sergio",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0003-2269-1554",
"affiliation": [
{
"name": "Department of Systems Medicine, Diabetes Research Institute Federation (DRIF), CTO Hospital ASL Roma 2, University of Rome Tor Vergata, Rome, Italy"
}
],
"authenticated-orcid": false,
"family": "Fabbri",
"given": "Andrea",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-6938-8627",
"affiliation": [
{
"name": "Department of Systems Medicine, University of Rome Tor Vergata, Rome, Italy"
},
{
"name": "Infectious Disease Clinic, Tor Vergata University Hospital (PTV), Rome, Italy"
}
],
"authenticated-orcid": false,
"family": "Iannetta",
"given": "Marco",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-3205-9758",
"affiliation": [
{
"name": "Department of Systems Medicine, University of Rome Tor Vergata, Rome, Italy"
},
{
"name": "Infectious Disease Clinic, Tor Vergata University Hospital (PTV), Rome, Italy"
}
],
"authenticated-orcid": false,
"family": "Andreoni",
"given": "Massimo",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0003-0007-8580",
"affiliation": [
{
"name": "UNESCO Chair of Biotechnology & Bioethics, University of Rome Tor Vergata, Rome, Italy"
}
],
"authenticated-orcid": false,
"family": "Colizzi",
"given": "Vittorio",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-9023-1106",
"affiliation": [
{
"name": "Clinical Biochemistry and Molecular Biology, Department of Experimental Medicine, Faculty of Medicine, University of Rome Tor Vergata, Rome, Italy"
},
{
"name": "Clinical Biochemistry Department, Tor Vergata University Hospital (PTV), Rome, Italy"
}
],
"authenticated-orcid": false,
"family": "Morello",
"given": "Maria",
"sequence": "additional"
}
],
"container-title": "Journal of the American Nutrition Association",
"container-title-short": "Journal of the American Nutrition Association",
"content-domain": {
"crossmark-restriction": true,
"domain": [
"www.tandfonline.com"
]
},
"created": {
"date-parts": [
[
2021,
2,
20
]
],
"date-time": "2021-02-20T00:28:06Z",
"timestamp": 1613780886000
},
"deposited": {
"date-parts": [
[
2023,
10,
21
]
],
"date-time": "2023-10-21T15:04:10Z",
"timestamp": 1697900650000
},
"indexed": {
"date-parts": [
[
2024,
1,
26
]
],
"date-time": "2024-01-26T07:19:40Z",
"timestamp": 1706253580859
},
"is-referenced-by-count": 28,
"issue": "3",
"issued": {
"date-parts": [
[
2021,
2,
18
]
]
},
"journal-issue": {
"issue": "3",
"published-print": {
"date-parts": [
[
2022,
4,
3
]
]
}
},
"language": "en",
"link": [
{
"URL": "https://www.tandfonline.com/doi/pdf/10.1080/07315724.2021.1877580",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "301",
"original-title": [],
"page": "250-265",
"prefix": "10.1080",
"published": {
"date-parts": [
[
2021,
2,
18
]
]
},
"published-online": {
"date-parts": [
[
2021,
2,
18
]
]
},
"published-print": {
"date-parts": [
[
2022,
4,
3
]
]
},
"publisher": "Informa UK Limited",
"reference": [
{
"DOI": "10.1183/13993003.01148-2020",
"author": "Leiva Sisnieguez CE",
"doi-asserted-by": "crossref",
"first-page": "2001148",
"issue": "6",
"journal-title": "Eur Respir J",
"key": "CIT0001",
"volume": "55",
"year": "2020"
},
{
"DOI": "10.1001/jama.2020.2648",
"doi-asserted-by": "publisher",
"key": "CIT0002"
},
{
"DOI": "10.1001/jama.2020.6775",
"doi-asserted-by": "publisher",
"key": "CIT0003"
},
{
"DOI": "10.1161/CIRCULATIONAHA.120.047659",
"doi-asserted-by": "publisher",
"key": "CIT0004"
},
{
"DOI": "10.1210/endrev/bnaa011",
"doi-asserted-by": "publisher",
"key": "CIT0005"
},
{
"DOI": "10.1530/EJE-20-0541",
"author": "Rottoli M",
"doi-asserted-by": "crossref",
"first-page": "389",
"issue": "4",
"journal-title": "Eur J Endocrinol",
"key": "CIT0006",
"volume": "183",
"year": "2020"
},
{
"DOI": "10.1016/j.dsx.2020.05.020",
"author": "Tamara A",
"doi-asserted-by": "crossref",
"first-page": "655",
"issue": "4",
"journal-title": "Diabetes Metab Syndr",
"key": "CIT0007",
"volume": "14",
"year": "2020"
},
{
"DOI": "10.1016/S0140-6736(20)30920-X",
"doi-asserted-by": "publisher",
"key": "CIT0008"
},
{
"DOI": "10.1016/j.immuni.2020.06.017",
"doi-asserted-by": "publisher",
"key": "CIT0009"
},
{
"DOI": "10.1016/S0140-6736(20)30183-5",
"doi-asserted-by": "publisher",
"key": "CIT0010"
},
{
"DOI": "10.1016/j.ebiom.2020.102763",
"doi-asserted-by": "publisher",
"key": "CIT0011"
},
{
"DOI": "10.1080/14787210.2020.1799785",
"doi-asserted-by": "publisher",
"key": "CIT0012"
},
{
"DOI": "10.1126/science.abb8925",
"doi-asserted-by": "publisher",
"key": "CIT0013"
},
{
"DOI": "10.1016/j.jinf.2020.03.037",
"doi-asserted-by": "publisher",
"key": "CIT0014"
},
{
"DOI": "10.1016/j.cpcardiol.2020.100618",
"doi-asserted-by": "publisher",
"key": "CIT0015"
},
{
"DOI": "10.1016/S0140-6736(20)30566-3",
"doi-asserted-by": "publisher",
"key": "CIT0016"
},
{
"DOI": "10.1111/jth.14768",
"doi-asserted-by": "publisher",
"key": "CIT0017"
},
{
"DOI": "10.1007/s40519-016-0312-6",
"doi-asserted-by": "publisher",
"key": "CIT0018"
},
{
"author": "Fabbri A",
"first-page": "4048",
"issue": "7",
"journal-title": "Eur Rev Med Pharmacol Sci",
"key": "CIT0019",
"volume": "24",
"year": "2020"
},
{
"DOI": "10.3390/nu11122937",
"doi-asserted-by": "publisher",
"key": "CIT0020"
},
{
"DOI": "10.3390/nu12072097",
"doi-asserted-by": "publisher",
"key": "CIT0021"
},
{
"DOI": "10.3390/nu11092185",
"doi-asserted-by": "publisher",
"key": "CIT0022"
},
{
"DOI": "10.4049/jimmunol.173.5.2909",
"doi-asserted-by": "publisher",
"key": "CIT0023"
},
{
"DOI": "10.1096/fj.04-3284com",
"doi-asserted-by": "publisher",
"key": "CIT0024"
},
{
"DOI": "10.2217/fmb.09.87",
"doi-asserted-by": "publisher",
"key": "CIT0025"
},
{
"DOI": "10.1126/science.1123933",
"doi-asserted-by": "publisher",
"key": "CIT0026"
},
{
"DOI": "10.1146/annurev.nutr.23.011702.073114",
"doi-asserted-by": "publisher",
"key": "CIT0027"
},
{
"DOI": "10.1111/j.1365-3083.1993.tb03237.x",
"doi-asserted-by": "publisher",
"key": "CIT0028"
},
{
"DOI": "10.4049/jimmunol.1102412",
"doi-asserted-by": "publisher",
"key": "CIT0029"
},
{
"DOI": "10.1046/j.1365-2249.2000.01204.x",
"doi-asserted-by": "publisher",
"key": "CIT0030"
},
{
"DOI": "10.1016/j.imbio.2012.07.018",
"doi-asserted-by": "publisher",
"key": "CIT0031"
},
{
"author": "Zhang X",
"first-page": "157834",
"journal-title": "Biomed Res Int",
"key": "CIT0032",
"volume": "2015",
"year": "2015"
},
{
"DOI": "10.4049/jimmunol.164.9.4443",
"doi-asserted-by": "publisher",
"key": "CIT0033"
},
{
"DOI": "10.4049/jimmunol.164.5.2405",
"doi-asserted-by": "publisher",
"key": "CIT0034"
},
{
"DOI": "10.4049/jimmunol.174.1.270",
"author": "Gauzzi MC",
"doi-asserted-by": "crossref",
"first-page": "270",
"issue": "1",
"journal-title": "J Immunol",
"key": "CIT0035",
"volume": "174",
"year": "2005"
},
{
"DOI": "10.1002/dmrr.1275",
"author": "Ferreira GB",
"doi-asserted-by": "crossref",
"first-page": "933",
"issue": "8",
"journal-title": "Diabetes Metab Res Rev",
"key": "CIT0036",
"volume": "27",
"year": "2011"
},
{
"DOI": "10.3389/fimmu.2019.00600",
"author": "Saul L",
"doi-asserted-by": "crossref",
"first-page": "600",
"journal-title": "Front Immunol",
"key": "CIT0037",
"volume": "10",
"year": "2019"
},
{
"DOI": "10.4049/jimmunol.0803217",
"doi-asserted-by": "publisher",
"key": "CIT0038"
},
{
"DOI": "10.2337/diabetes.49.8.1301",
"author": "Overbergh L",
"doi-asserted-by": "crossref",
"first-page": "1301",
"issue": "8",
"journal-title": "Diabetes",
"key": "CIT0039",
"volume": "49",
"year": "2000"
},
{
"DOI": "10.4049/jimmunol.167.9.4974",
"doi-asserted-by": "publisher",
"key": "CIT0040"
},
{
"author": "Bouillon R",
"first-page": "354",
"issue": "2",
"journal-title": "Pediatr Endocrinol Rev",
"key": "CIT0041",
"volume": "10",
"year": "2013"
},
{
"DOI": "10.1016/j.coph.2010.04.001",
"doi-asserted-by": "publisher",
"key": "CIT0042"
},
{
"DOI": "10.3390/nu5072502",
"doi-asserted-by": "publisher",
"key": "CIT0043"
},
{
"DOI": "10.1126/science.6310748",
"doi-asserted-by": "publisher",
"key": "CIT0044"
},
{
"DOI": "10.1081/IPH-120014721",
"doi-asserted-by": "publisher",
"key": "CIT0045"
},
{
"DOI": "10.1007/s11154-011-9195-z",
"doi-asserted-by": "publisher",
"key": "CIT0046"
},
{
"DOI": "10.1002/rmv.1909",
"doi-asserted-by": "publisher",
"key": "CIT0047"
},
{
"DOI": "10.1046/j.1523-1755.2000.00202.x",
"doi-asserted-by": "publisher",
"key": "CIT0048"
},
{
"DOI": "10.1038/ni1433",
"doi-asserted-by": "publisher",
"key": "CIT0049"
},
{
"DOI": "10.1359/JBMR.050908",
"doi-asserted-by": "publisher",
"key": "CIT0050"
},
{
"DOI": "10.4049/jimmunol.179.3.1634",
"doi-asserted-by": "publisher",
"key": "CIT0051"
},
{
"DOI": "10.1007/s11154-017-9424-1",
"doi-asserted-by": "publisher",
"key": "CIT0052"
},
{
"DOI": "10.1016/S2213-8587(20)30268-0",
"doi-asserted-by": "publisher",
"key": "CIT0053"
},
{
"DOI": "10.3390/nu12040988",
"doi-asserted-by": "publisher",
"key": "CIT0054"
},
{
"DOI": "10.3390/biomedicines8050129",
"author": "Glinsky GV.",
"doi-asserted-by": "crossref",
"first-page": "129",
"issue": "5",
"journal-title": "Biomedicines",
"key": "CIT0055",
"volume": "8",
"year": "2020"
},
{
"DOI": "10.3390/nu12113361",
"doi-asserted-by": "publisher",
"key": "CIT0056"
},
{
"DOI": "10.1038/s41430-020-0661-0",
"doi-asserted-by": "publisher",
"key": "CIT0057"
},
{
"DOI": "10.1155/2016/8191254",
"doi-asserted-by": "publisher",
"key": "CIT0058"
},
{
"DOI": "10.1186/s13104-016-2335-5",
"doi-asserted-by": "publisher",
"key": "CIT0059"
},
{
"DOI": "10.3945/an.117.015578",
"author": "Cashman KD",
"doi-asserted-by": "crossref",
"first-page": "947",
"issue": "6",
"journal-title": "Adv Nutr",
"key": "CIT0060",
"volume": "8",
"year": "2017"
},
{
"DOI": "10.1210/jc.2011-0385",
"doi-asserted-by": "publisher",
"key": "CIT0061"
},
{
"author": "Carpagnano GE",
"first-page": "1",
"journal-title": "J Endocrinol Invest",
"key": "CIT0062",
"volume": "9",
"year": "2020"
},
{
"DOI": "10.1111/febs.15495",
"doi-asserted-by": "publisher",
"key": "CIT0063"
},
{
"DOI": "10.1001/jamanetworkopen.2020.19722",
"doi-asserted-by": "publisher",
"key": "CIT0064"
},
{
"DOI": "10.3390/nu12051359",
"author": "D’Avolio A",
"doi-asserted-by": "crossref",
"first-page": "13591359",
"issue": "5",
"journal-title": "Nutrients",
"key": "CIT0065",
"volume": "12",
"year": "2020"
},
{
"DOI": "10.1371/journal.pone.0239252",
"doi-asserted-by": "publisher",
"key": "CIT0066"
},
{
"DOI": "10.1136/postgradmedj-2020-138712",
"doi-asserted-by": "publisher",
"key": "CIT0067"
},
{
"DOI": "10.3390/nu12092757",
"doi-asserted-by": "publisher",
"key": "CIT0068"
},
{
"author": "De Smet D",
"journal-title": "Am J Clin Pathol",
"key": "CIT0069",
"year": "2020"
},
{
"DOI": "10.1038/s41598-020-77093-z",
"doi-asserted-by": "publisher",
"key": "CIT0070"
},
{
"DOI": "10.1371/journal.pone.0239799",
"doi-asserted-by": "publisher",
"key": "CIT0071"
},
{
"author": "Hernández JL",
"journal-title": "J Clin Endocrinol Metab",
"key": "CIT0072",
"year": "2020"
},
{
"DOI": "10.1080/07315724.2020.1826005",
"doi-asserted-by": "publisher",
"key": "CIT0073"
},
{
"DOI": "10.1038/oby.2011.404",
"doi-asserted-by": "publisher",
"key": "CIT0074"
},
{
"DOI": "10.1210/jc.2016-3591",
"doi-asserted-by": "publisher",
"key": "CIT0075"
},
{
"DOI": "10.1002/jbm4.10397",
"author": "Elkhwanky M-S",
"doi-asserted-by": "crossref",
"first-page": "e10397",
"issue": "11",
"journal-title": "JBMR Plus",
"key": "CIT0076",
"volume": "4",
"year": "2020"
},
{
"DOI": "10.1002/jbmr.3686",
"author": "Roizen JD",
"doi-asserted-by": "crossref",
"first-page": "1068",
"issue": "6",
"journal-title": "J Bone Miner Res",
"key": "CIT0077",
"volume": "34",
"year": "2019"
},
{
"DOI": "10.1016/j.jsbmb.2020.105751",
"doi-asserted-by": "publisher",
"key": "CIT0078"
},
{
"DOI": "10.1136/postgradmedj-2020-139065",
"doi-asserted-by": "publisher",
"key": "CIT0079"
},
{
"DOI": "10.3390/nu12113377",
"doi-asserted-by": "publisher",
"key": "CIT0080"
},
{
"DOI": "10.1093/ajcn/86.3.714",
"doi-asserted-by": "publisher",
"key": "CIT0081"
},
{
"DOI": "10.1001/archinternmed.2008.560",
"doi-asserted-by": "publisher",
"key": "CIT0082"
},
{
"DOI": "10.1186/1743-422X-5-29",
"doi-asserted-by": "publisher",
"key": "CIT0083"
},
{
"DOI": "10.1017/S0007114511001991",
"doi-asserted-by": "publisher",
"key": "CIT0084"
},
{
"DOI": "10.3310/hta23020",
"doi-asserted-by": "publisher",
"key": "CIT0085"
},
{
"DOI": "10.3390/nu12082488",
"author": "Brenner H",
"doi-asserted-by": "crossref",
"first-page": "2488",
"issue": "8",
"journal-title": "Nutrients",
"key": "CIT0086",
"volume": "12",
"year": "2020"
},
{
"DOI": "10.1371/journal.pone.0011088",
"doi-asserted-by": "publisher",
"key": "CIT0087"
},
{
"DOI": "10.2147/COPD.S141760",
"doi-asserted-by": "publisher",
"key": "CIT0088"
},
{
"DOI": "10.3389/fpubh.2020.00299",
"doi-asserted-by": "publisher",
"key": "CIT0089"
},
{
"DOI": "10.1038/s41591-020-1051-9",
"doi-asserted-by": "publisher",
"key": "CIT0090"
},
{
"DOI": "10.1016/j.ijantimicag.2020.106051",
"doi-asserted-by": "publisher",
"key": "CIT0091"
},
{
"DOI": "10.1007/s11357-020-00186-0",
"doi-asserted-by": "publisher",
"key": "CIT0092"
},
{
"DOI": "10.1172/JCI112134",
"doi-asserted-by": "publisher",
"key": "CIT0093"
},
{
"DOI": "10.1530/EJE-18-0736",
"doi-asserted-by": "publisher",
"key": "CIT0094"
},
{
"DOI": "10.5114/aoms.2012.27271",
"doi-asserted-by": "publisher",
"key": "CIT0095"
}
],
"reference-count": 95,
"references-count": 95,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.tandfonline.com/doi/full/10.1080/07315724.2021.1877580"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subtitle": [],
"title": "Low Vitamin D Status at Admission as a Risk Factor for Poor Survival in Hospitalized Patients With COVID-19: An Italian Retrospective Study",
"type": "journal-article",
"update-policy": "http://dx.doi.org/10.1080/tandf_crossmark_01",
"volume": "41"
}
