Metformin Use in Relation to Clinical Outcomes and Hyperinflammatory Syndrome Among COVID-19 Patients With Type 2 Diabetes: A Propensity Score Analysis of a Territory-Wide Cohort
et al., Frontiers in Endocrinology, doi:10.3389/fendo.2022.810914, Mar 2022
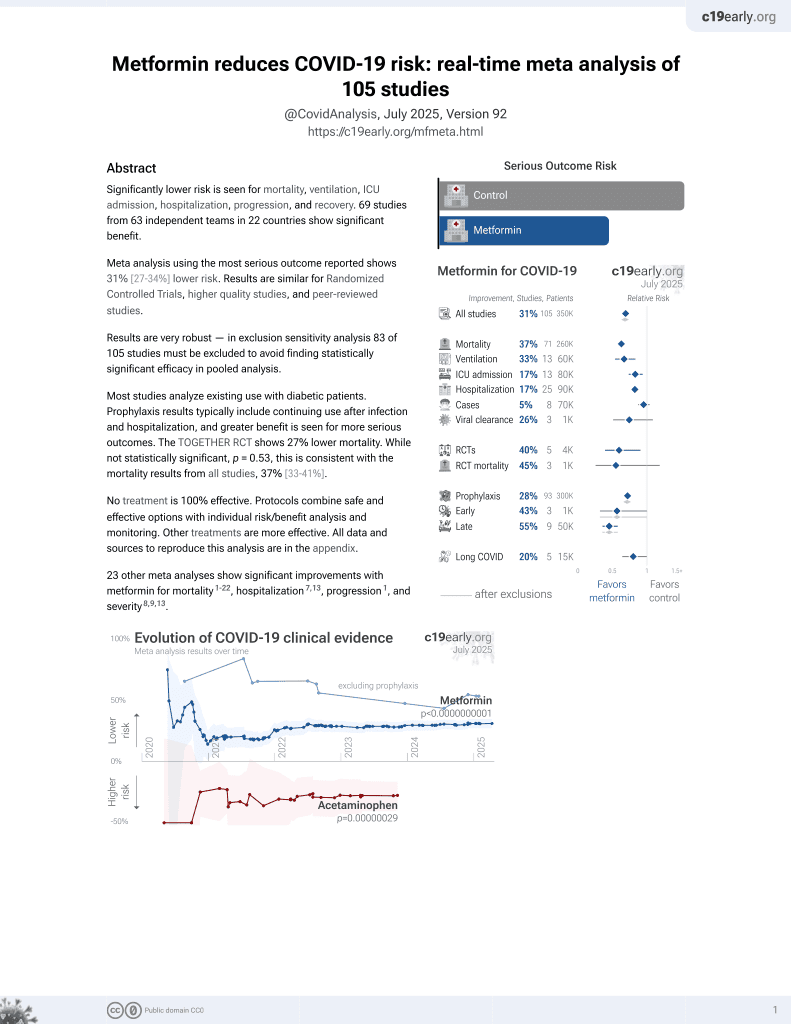
Metformin for COVID-19
3rd treatment shown to reduce risk in
July 2020, now with p < 0.00000000001 from 111 studies.
Lower risk for mortality, ventilation, ICU, hospitalization, progression, recovery, and viral clearance.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
Retrospective 1,214 COVID+ type 2 diabetes patients in Hong Kong, showing lower mortality and improved recovery with metformin use.
Standard of Care (SOC) for COVID-19 in the study country,
China, is poor with low average efficacy for approved treatments1.
|
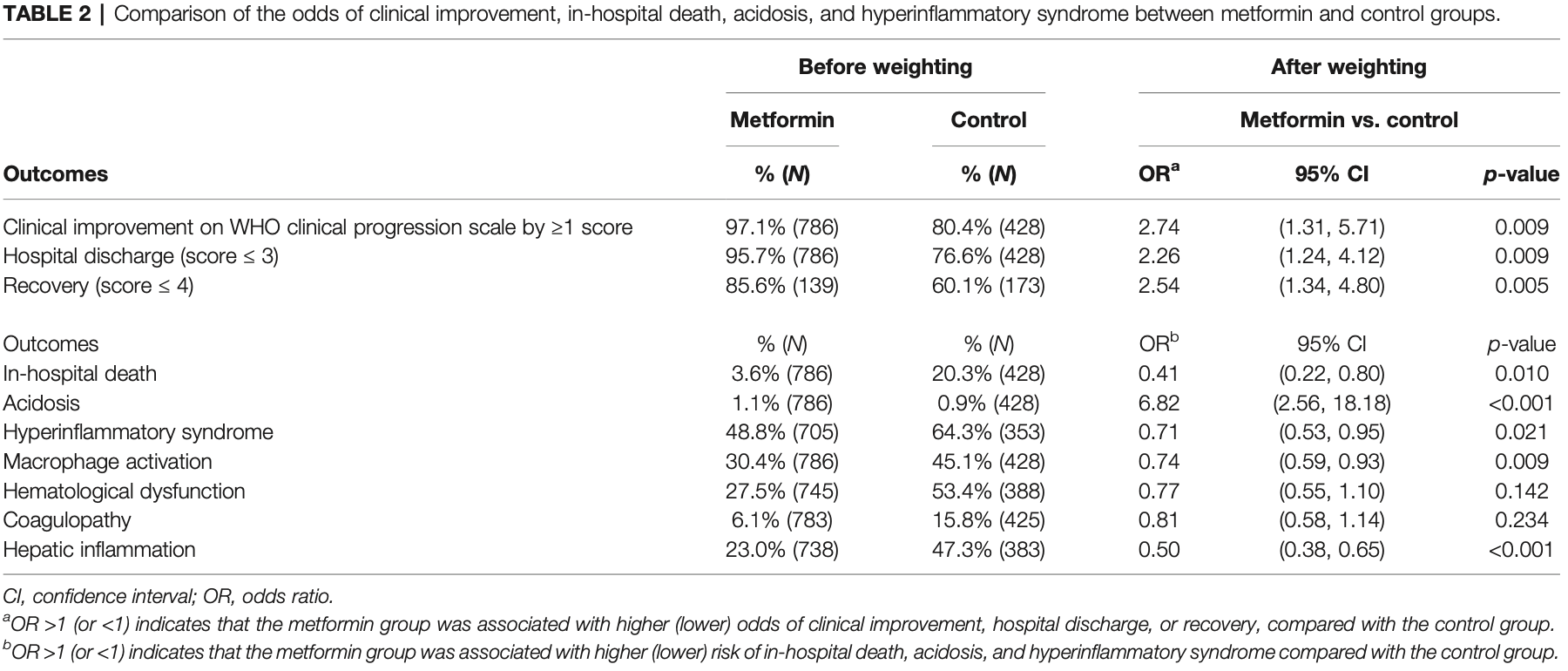
risk of death, 59.0% lower, OR 0.41, p = 0.01, treatment 786, control 428, adjusted per study, propensity score weighting, multivariable, RR approximated with OR.
|
|
risk of no recovery, 60.6% lower, OR 0.39, p = 0.005, treatment 786, control 428, adjusted per study, inverted to make OR<1 favor treatment, propensity score weighting, multivariable, RR approximated with OR.
|
|
clinical improvement, 63.5% better, OR 0.36, p = 0.009, treatment 786, control 428, adjusted per study, inverted to make OR<1 favor treatment, propensity score weighting, multivariable, RR approximated with OR.
|
|
risk of no hospital discharge, 55.8% lower, OR 0.44, p = 0.009, treatment 786, control 428, adjusted per study, inverted to make OR<1 favor treatment, propensity score weighting, multivariable, RR approximated with OR.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Wong et al., 7 Mar 2022, retrospective, China, peer-reviewed, 11 authors, study period 21 January, 2020 - 31 January, 2021.
Metformin Use in Relation to Clinical Outcomes and Hyperinflammatory Syndrome Among COVID-19 Patients With Type 2 Diabetes: A Propensity Score Analysis of a Territory-Wide Cohort
Frontiers in Endocrinology, doi:10.3389/fendo.2022.810914
This study was conducted in order to evaluate the association between metformin use and clinical outcomes in type 2 diabetes mellitus (T2DM) patients hospitalized with coronavirus disease 2019 . Methods: Patients with T2DM with confirmed diagnosis of COVID-19 and admitted between January 21, 2020, and January 31, 2021 in Hong Kong were identified in our cohort. Exposure was defined as metformin use within 90 days prior to admission until hospital discharge for COVID-19. Primary outcome was defined as clinical improvement of ≥1 point on the WHO Clinical Progression Scale (CPS). Other outcomes were hospital discharge, recovery, in-hospital death, acidosis, hyperinflammatory syndrome, length of hospitalization, and changes in WHO CPS score. Results: Metformin use was associated with greater odds of clinical improvement (OR = 2.74, p = 0.009), hospital discharge (OR = 2.26, p = 0.009), and recovery (OR = 2.54, p = 0.005), in addition to lower odds of hyperinflammatory syndrome (OR = 0.71, p = 0.021) and death (OR = 0.41, p = 0.010) than control. Patients on metformin treatment had a shorter hospital stay (−2.76 days, p = 0.017) than their control counterparts. The average WHO CPS scores were significantly lower in metformin users than non-users since day 15 (p < 0.001). However, metformin use was associated with higher odds of acidosis. Conclusions: Metformin use was associated with lower mortality and lower odds for hyperinflammatory syndrome. This provides additional insights into the potential mechanisms of the benefits of metformin use in T2DM patients with COVID-19.
ETHICS STATEMENT The study protocol was approved by the Institutional Review Board of the University of Hong Kong/ Hospital Authority Hong Kong West Cluster (Reference No. . Given the extraordinary nature of the COVID-19 pandemic, individual patient informed consent was not required for this retrospective cohort study using anonymized data.
AUTHOR CONTRIBUTIONS CW reviewed the literature, designed the statistical analysis, conducted the analyses, and wrote the manuscript. DL, AL, AK, ML, and KL reviewed the literature, contributed to the
SUPPLEMENTARY MATERIAL The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fendo.2022.
810914/full#supplementary-material Conflict of Interest: The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. Publisher's Note: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
Apicella, Campopiano, Mantuano, Mazoni, Coppelli et al., COVID-19 in People With Diabetes: Understanding the Reasons for Worse Outcomes, Lancet Diabetes Endocrinol, doi:10.1016/S2213-8587(20)30238-2
Barron, Bakhai, Kar, Weaver, Bradley et al., Associations of Type 1 and Type 2 Diabetes With COVID-19-Related Mortality in England: A Whole-Population Study, Lancet Diabetes Endocrinol, doi:10.1016/S2213-8587(20)30272-2
Bode, Garrett, Messler, Mcfarland, Crowe et al., Glycemic Characteristics and Clinical Outcomes of COVID-19 Patients Hospitalized in the United States, J Diabetes Sci Tech, doi:10.1177/1932296820924469
Bornstein, Rubino, Khunti, Mingrone, Hopkins et al., Practical Recommendations for the Management of Diabetes in Patients With COVID-19, Lancet Diabetes Endocrinol, doi:10.1016/S2213-8587(20)30152-2
Bramante, Ingraham, Murray, Marmor, Hovertsen et al., Metformin and Risk of Mortality in Patients Hospitalised With COVID-19: A Retrospective Cohort Analysis, Lancet Healthy Longevity, doi:10.1016/S2666-7568(20)30033-7
Chen, Yang, Cheng, Chen, Peng et al., Clinical Characteristics and Outcomes of Patients With Diabetes and COVID-19 in Association With Glucose-Lowering Medication, Diabetes Care, doi:10.2337/dc20-0660
Cheng, Liu, Li, Zhang, Lei et al., Metformin Is Associated With Higher Incidence of Acidosis, But Not Mortality, in Individuals With COVID-19 and Pre-Existing Type 2 Diabetes, Cell Metab, doi:10.1016/j.cmet.2020.08.013
Gao, Liu, Zhong, Liu, Zhou et al., Risk of Metformin in Patients With Type 2 Diabetes With COVID-19: A Preliminary Retrospective Report, doi:10.1111/cts.12897
Hariyanto, Kurniawan, Metformin Use Is Associated With Reduced Mortality Rate From Coronavirus Disease 2019 (COVID-19) Infection, Obes Med, doi:10.1016/j.obmed.2020.100290
Hu, Huang, Yin, The Cytokine Storm and COVID-19, J Med Virol, doi:10.1002/jmv.26232
Ilyas, Wallis, Soilleux, Townsend, Zehnder et al., High Glucose Disrupts Oligosaccharide Recognition Function via Competitive Inhibition: A Potential Mechanism for Immune Dysregulation in Diabetes Mellitus, Immunobiology, doi:10.1016/j.imbio.2010.06.002
Katulanda, Dissanayake, Ranathunga, Ratnasamy, Wijewickrama et al., Prevention and Management of COVID-19
Kopec, Kowalski, Metformin-Associated Lactic Acidosis (MALA): Case Files of the Einstein Medical Center Medical Toxicology Fellowship, J? Med Toxicol, doi:10.1007/s13181-012-0278-3
Koufakis, Maltese, Metallidis, Zebekakis, Kotsa, Looking Deeper Into the Findings of DARE-19: Failure or an Open Door to Future Success?, Pharmacol Res, doi:10.1016/j.phrs.2021.105872
Koufakis, Metallidis, Zebekakis, Ajjan, Kotsa, Sodium-Glucose Cotransporter 2 Inhibitors in the Era of COVID-19 Pandemic: Is the Benefit to Risk Ratio Still Favorable?, J Diabetes Sci Tech, doi:10.1177/1932296820932155
Koufakis, Pavlidis, Metallidis, Kotsa, Sodium-Glucose Co-Transporter 2 Inhibitors in COVID-19: Meeting at the Crossroads Between Heart, Diabetes and Infectious Diseases, Int J Clin Pharmacy, doi:10.1007/s11096-021-01256-9
Kow, Hasan, Mortality Risk With Preadmission Metformin Use in Patients With COVID-19 and Diabetes: A Meta-Analysis, J Med Virol, doi:10.1002/jmv.26498
Lalau, Arnouts, Sharif, Broe, Metformin and Other Antidiabetic Agents in Renal Failure Patients, Kidney Int, doi:10.1038/ki.2014.19
Lukito, Pranata, Henrina, Lim, Lawrensia et al., The Effect of Metformin Consumption on Mortality in Hospitalized COVID-19 Patients: A Systematic Review and Meta-Analysis, Diabetes Metab Syndrome: Clin Res Rev, doi:10.1016/j.dsx.2020.11.006
Lumbers, Delforce, Pringle, Smith, The Lung, the Heart, the Novel Coronavirus, and the Renin-Angiotensin System; the Need for Clinical Trials, Front Med, doi:10.3389/fmed.2020.00248
Luo, Qiu, Liu, Liu, Zheng et al., Metformin Treatment was Associated With Decreased Mortality in COVID-19 Patients With Diabetes in a Retrospective Analysis, Am J Trop Med Hygiene, doi:10.4269/ajtmh.20-0375
Mirabelli, Chiefari, Puccio, Foti, Brunetti, Potential Benefits and Harms of Novel Antidiabetic Drugs During COVID-19
Pal, Banerjee, Mukherjee, Bhogal, Kaur et al., Dipeptidyl Peptidase-4 Inhibitor Use and Mortality in COVID-19 Patients With Diabetes Mellitus: An Updated Systematic Review and Meta-Analysis, Ther Adv Endocrinol Metab, doi:10.1177/2042018821996482
Papazafiropoulou, Antonopoulos, The COVID-19 Pandemic and Diabetes Mellitus, Arch Med Sci Atherosclerotic Dis, doi:10.5114/amsad.2020.97435
Riahi, Sombra, Lo, Chacko, Neto et al., Insulin Use, Diabetes Control, and Outcomes in Patients With COVID-19, Endocrine Res, doi:10.1080/07435800.2020.1856865
Sardu, Onofrio, Balestrieri, Barbieri, Rizzo et al., Outcomes in Patients With Hyperglycemia Affected by COVID-19: Can We do More on Glycemic Control?, Diabetes Care, doi:10.2337/dc20-0723
Sharif-Askari, Sharif-Askari, Mdkhana, Heialy, Ratemi et al., Effect of Common Medications on the Expression of SARS-CoV-2 Entry Receptors in Liver Tissue, Arch Toxicol, doi:10.1007/s00204-020-02869-1
Sharma, Ray, Sadasivam, Metformin in COVID-19: A Possible Role Beyond Diabetes, Diabetes Res Clin Practice, doi:10.1016/j.diabres.2020.108183
Suetrong, Walley, Lactic Acidosis in Sepsis: It's Not All Anaerobic: Implications for Diagnosis and Management, Chest, doi:10.1378/chest.15-1703
Tom, Mina, To Interpret the SARS-CoV-2 Test, Consider the Cycle Threshold Value, Clin Infect Dis, doi:10.1093/cid/ciaa619
Webb, Peltan, Jensen, Hoda, Hunter et al., Clinical Criteria for COVID-19-Associated Hyperinflammatory Syndrome: A Cohort Study, Lancet Rheumatol, doi:10.1016/S2665-9913(20)30343-X
Wong, Lui, Lui, Kwok, Low et al., Use of DPP4i Reduced Odds of Clinical Deterioration and Hyperinflammatory Syndrome in COVID-19 Patients With Type 2 Diabetes: Propensity Score Analysis of a Territory-Wide Cohort in Hong Kong, Diabetes Metab, doi:10.1016/j.diabet.2021.101307
Xiao, Sakagami, Miwa, ACE2: The Key Molecule for Understanding the Pathophysiology of Severe and Critical Conditions of COVID-19: Demon or Angel?, Viruses, doi:10.3390/v12050491
Yang, Sun, Zhang, Zhang, The Effect of Metformin on Mortality and Severity in COVID-19 Patients With Diabetes Mellitus, Diabetes Res Clin Practice, doi:10.1016/j.diabres.2021.108977
Zhang, Dong, Martin, He, Gongol et al., AMP-Activated Protein Kinase Phosphorylation of Angiotensin-Converting Enzyme 2 in Endothelium Mitigates Pulmonary Hypertension, Am J Respir Crit Care Med, doi:10.1164/rccm.201712-2570OC
Zhu, She, Cheng, Qin, Zhang et al., Association of Blood Glucose Control and Outcomes in Patients With COVID-19 and Pre-Existing Type 2 Diabetes, Cell Metab, doi:10.1016/j.cmet.2020.04.021
DOI record:
{
"DOI": "10.3389/fendo.2022.810914",
"ISSN": [
"1664-2392"
],
"URL": "http://dx.doi.org/10.3389/fendo.2022.810914",
"abstract": "<jats:sec><jats:title>Aim</jats:title><jats:p>This study was conducted in order to evaluate the association between metformin use and clinical outcomes in type 2 diabetes mellitus (T2DM) patients hospitalized with coronavirus disease 2019 (COVID-19).</jats:p></jats:sec><jats:sec><jats:title>Methods</jats:title><jats:p>Patients with T2DM with confirmed diagnosis of COVID-19 and admitted between January 21, 2020, and January 31, 2021 in Hong Kong were identified in our cohort. Exposure was defined as metformin use within 90 days prior to admission until hospital discharge for COVID-19. Primary outcome was defined as clinical improvement of ≥1 point on the WHO Clinical Progression Scale (CPS). Other outcomes were hospital discharge, recovery, in-hospital death, acidosis, hyperinflammatory syndrome, length of hospitalization, and changes in WHO CPS score.</jats:p></jats:sec><jats:sec><jats:title>Results</jats:title><jats:p>Metformin use was associated with greater odds of clinical improvement (OR = 2.74, <jats:italic>p</jats:italic> = 0.009), hospital discharge (OR = 2.26, <jats:italic>p</jats:italic> = 0.009), and recovery (OR = 2.54, <jats:italic>p</jats:italic> = 0.005), in addition to lower odds of hyperinflammatory syndrome (OR = 0.71, <jats:italic>p</jats:italic> = 0.021) and death (OR = 0.41, <jats:italic>p</jats:italic> = 0.010) than control. Patients on metformin treatment had a shorter hospital stay (−2.76 days, <jats:italic>p</jats:italic> = 0.017) than their control counterparts. The average WHO CPS scores were significantly lower in metformin users than non-users since day 15 (<jats:italic>p</jats:italic> &lt; 0.001). However, metformin use was associated with higher odds of acidosis.</jats:p></jats:sec><jats:sec><jats:title>Conclusions</jats:title><jats:p>Metformin use was associated with lower mortality and lower odds for hyperinflammatory syndrome. This provides additional insights into the potential mechanisms of the benefits of metformin use in T2DM patients with COVID-19.</jats:p></jats:sec>",
"alternative-id": [
"10.3389/fendo.2022.810914"
],
"author": [
{
"affiliation": [],
"family": "Wong",
"given": "Carlos K. H.",
"sequence": "first"
},
{
"affiliation": [],
"family": "Lui",
"given": "David T. W.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Lui",
"given": "Angel Y. C.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Low",
"given": "Marshall C. H.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Kwok",
"given": "Ashley C. Y.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Lau",
"given": "Kristy T. K.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Au",
"given": "Ivan C. H.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Xiong",
"given": "Xi",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Chung",
"given": "Matthew S. H.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Lau",
"given": "Eric H. Y.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Cowling",
"given": "Benjamin J.",
"sequence": "additional"
}
],
"container-title": [
"Frontiers in Endocrinology"
],
"content-domain": {
"crossmark-restriction": true,
"domain": [
"frontiersin.org"
]
},
"created": {
"date-parts": [
[
2022,
3,
7
]
],
"date-time": "2022-03-07T05:34:50Z",
"timestamp": 1646631290000
},
"deposited": {
"date-parts": [
[
2022,
3,
7
]
],
"date-time": "2022-03-07T05:34:56Z",
"timestamp": 1646631296000
},
"funder": [
{
"DOI": "10.13039/501100005407",
"award": [
"COVID190210"
],
"doi-asserted-by": "publisher",
"name": "Food and Health Bureau"
}
],
"indexed": {
"date-parts": [
[
2022,
3,
7
]
],
"date-time": "2022-03-07T06:15:35Z",
"timestamp": 1646633735421
},
"is-referenced-by-count": 0,
"issn-type": [
{
"type": "electronic",
"value": "1664-2392"
}
],
"issued": {
"date-parts": [
[
2022,
3,
7
]
]
},
"license": [
{
"URL": "https://creativecommons.org/licenses/by/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2022,
3,
7
]
],
"date-time": "2022-03-07T00:00:00Z",
"timestamp": 1646611200000
}
}
],
"link": [
{
"URL": "https://www.frontiersin.org/articles/10.3389/fendo.2022.810914/full",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "1965",
"original-title": [],
"prefix": "10.3389",
"published": {
"date-parts": [
[
2022,
3,
7
]
]
},
"published-online": {
"date-parts": [
[
2022,
3,
7
]
]
},
"publisher": "Frontiers Media SA",
"reference": [
{
"key": "B1",
"unstructured": "Who Coronavirus (Covid-19) Dashboard"
},
{
"DOI": "10.1016/S2213-8587(20)30272-2",
"article-title": "Associations of Type 1 and Type 2 Diabetes With COVID-19-Related Mortality in England: A Whole-Population Study",
"author": "Barron",
"doi-asserted-by": "publisher",
"journal-title": "Lancet Diabetes Endocrinol",
"key": "B2",
"volume": "8",
"year": "2020"
},
{
"DOI": "10.2337/dc20-0723",
"article-title": "Outcomes in Patients With Hyperglycemia Affected by COVID-19: Can We do More on Glycemic Control",
"author": "Sardu",
"doi-asserted-by": "publisher",
"journal-title": "Diabetes Care",
"key": "B3",
"volume": "43",
"year": "2020"
},
{
"DOI": "10.1016/S2213-8587(20)30152-2",
"article-title": "Practical Recommendations for the Management of Diabetes in Patients With COVID-19",
"author": "Bornstein",
"doi-asserted-by": "publisher",
"journal-title": "Lancet Diabetes Endocrinol",
"key": "B4",
"volume": "8",
"year": "2020"
},
{
"DOI": "10.5114/amsad.2020.97435",
"article-title": "The COVID-19 Pandemic and Diabetes Mellitus",
"author": "Papazafiropoulou",
"doi-asserted-by": "publisher",
"first-page": "e200",
"journal-title": "Arch Med Sci Atherosclerotic Dis",
"key": "B5",
"volume": "5",
"year": "2020"
},
{
"DOI": "10.1016/j.dsx.2020.11.006",
"article-title": "The Effect of Metformin Consumption on Mortality in Hospitalized COVID-19 Patients: A Systematic Review and Meta-Analysis",
"author": "Lukito",
"doi-asserted-by": "publisher",
"journal-title": "Diabetes Metab Syndrome: Clin Res Rev",
"key": "B6",
"volume": "14",
"year": "2020"
},
{
"DOI": "10.1016/j.cmet.2020.04.021",
"article-title": "Association of Blood Glucose Control and Outcomes in Patients With COVID-19 and Pre-Existing Type 2 Diabetes",
"author": "Zhu",
"doi-asserted-by": "publisher",
"first-page": "1068",
"journal-title": "Cell Metab",
"key": "B7",
"volume": "31",
"year": "2020"
},
{
"DOI": "10.1016/j.diabres.2020.108183",
"article-title": "Metformin in COVID-19: A Possible Role Beyond Diabetes",
"author": "Sharma",
"doi-asserted-by": "publisher",
"journal-title": "Diabetes Res Clin Practice",
"key": "B8",
"volume": "164",
"year": "2020"
},
{
"DOI": "10.4269/ajtmh.20-0375",
"article-title": "Metformin Treatment was Associated With Decreased Mortality in COVID-19 Patients With Diabetes in a Retrospective Analysis",
"author": "Luo",
"doi-asserted-by": "publisher",
"first-page": "69",
"journal-title": "Am J Trop Med Hygiene",
"key": "B9",
"volume": "103",
"year": "2020"
},
{
"DOI": "10.1016/j.cmet.2020.08.013",
"article-title": "Metformin Is Associated With Higher Incidence of Acidosis, But Not Mortality, in Individuals With COVID-19 and Pre-Existing Type 2 Diabetes",
"author": "Cheng",
"doi-asserted-by": "publisher",
"first-page": "537",
"journal-title": "Cell Metab",
"key": "B10",
"volume": "32",
"year": "2020"
},
{
"DOI": "10.1016/S2213-8587(20)30238-2",
"article-title": "COVID-19 in People With Diabetes: Understanding the Reasons for Worse Outcomes",
"author": "Apicella",
"doi-asserted-by": "publisher",
"journal-title": "Lancet Diabetes Endocrinol",
"key": "B11",
"volume": "8",
"year": "2020"
},
{
"DOI": "10.1111/cts.12897",
"article-title": "Risk of Metformin in Patients With Type 2 Diabetes With COVID-19: A Preliminary Retrospective Report",
"author": "Gao",
"doi-asserted-by": "publisher",
"journal-title": "Clin Trans Sci",
"key": "B12",
"volume": "13",
"year": "2020"
},
{
"key": "B13",
"volume-title": "Concise Advice on Inpatient Diabetes (COVID: Diabetes): Hyperglycaemia/Diabetes Guidance for People With Covid-19 Infections Managed in a Virtual Ward: a Guide for Healthcare Professionals",
"year": "2021"
},
{
"DOI": "10.1016/j.diabet.2021.101307",
"article-title": "Use of DPP4i Reduced Odds of Clinical Deterioration and Hyperinflammatory Syndrome in COVID-19 Patients With Type 2 Diabetes: Propensity Score Analysis of a Territory-Wide Cohort in Hong Kong",
"author": "Wong",
"doi-asserted-by": "publisher",
"journal-title": "Diabetes Metab",
"key": "B14",
"volume": "48",
"year": "2022"
},
{
"DOI": "10.1016/s1473-3099(20)30483-7",
"article-title": "A Minimal Common Outcome Measure Set for COVID-19 Clinical Research",
"doi-asserted-by": "publisher",
"journal-title": "Lancet Infect Dis",
"key": "B15",
"volume": "20",
"year": "2020"
},
{
"DOI": "10.1016/S2665-9913(20)30343-X",
"article-title": "Clinical Criteria for COVID-19-Associated Hyperinflammatory Syndrome: A Cohort Study",
"author": "Webb",
"doi-asserted-by": "publisher",
"journal-title": "Lancet Rheumatol",
"key": "B16",
"volume": "2",
"year": "2020"
},
{
"DOI": "10.1093/cid/ciaa619",
"article-title": "To Interpret the SARS-CoV-2 Test, Consider the Cycle Threshold Value",
"author": "Tom",
"doi-asserted-by": "publisher",
"journal-title": "Clin Infect Dis",
"key": "B17",
"volume": "71",
"year": "2020"
},
{
"DOI": "10.1177/2042018821996482",
"article-title": "Dipeptidyl Peptidase-4 Inhibitor Use and Mortality in COVID-19 Patients With Diabetes Mellitus: An Updated Systematic Review and Meta-Analysis",
"author": "Pal",
"doi-asserted-by": "publisher",
"journal-title": "Ther Adv Endocrinol Metab",
"key": "B18",
"volume": "12",
"year": "2021"
},
{
"DOI": "10.1016/j.phrs.2021.105872",
"article-title": "Looking Deeper Into the Findings of DARE-19: Failure or an Open Door to Future Success",
"author": "Koufakis",
"doi-asserted-by": "publisher",
"journal-title": "Pharmacol Res",
"key": "B19",
"volume": "173",
"year": "2021"
},
{
"DOI": "10.1007/s11096-021-01256-9",
"article-title": "Sodium-Glucose Co-Transporter 2 Inhibitors in COVID-19: Meeting at the Crossroads Between Heart, Diabetes and Infectious Diseases",
"author": "Koufakis",
"doi-asserted-by": "publisher",
"first-page": "1",
"journal-title": "Int J Clin Pharmacy",
"key": "B20",
"year": "2021"
},
{
"DOI": "10.1177/1932296820932155",
"article-title": "Sodium-Glucose Cotransporter 2 Inhibitors in the Era of COVID-19 Pandemic: Is the Benefit to Risk Ratio Still Favorable",
"author": "Koufakis",
"doi-asserted-by": "publisher",
"journal-title": "J Diabetes Sci Tech",
"key": "B21",
"volume": "14",
"year": "2020"
},
{
"DOI": "10.1016/j.obmed.2020.100290",
"article-title": "Metformin Use Is Associated With Reduced Mortality Rate From Coronavirus Disease 2019 (COVID-19) Infection",
"author": "Hariyanto",
"doi-asserted-by": "publisher",
"first-page": "100290",
"journal-title": "Obes Med",
"key": "B22",
"volume": "19",
"year": "2020"
},
{
"DOI": "10.1002/jmv.26498",
"article-title": "Mortality Risk With Preadmission Metformin Use in Patients With COVID-19 and Diabetes: A Meta-Analysis",
"author": "Kow",
"doi-asserted-by": "publisher",
"journal-title": "J Med Virol",
"key": "B23",
"volume": "93",
"year": "2021"
},
{
"DOI": "10.1016/j.diabres.2021.108977",
"article-title": "The Effect of Metformin on Mortality and Severity in COVID-19 Patients With Diabetes Mellitus",
"author": "Yang",
"doi-asserted-by": "publisher",
"first-page": "108977",
"journal-title": "Diabetes Res Clin Practice",
"key": "B24",
"volume": "178",
"year": "2021"
},
{
"DOI": "10.1007/s00204-020-02869-1",
"article-title": "Effect of Common Medications on the Expression of SARS-CoV-2 Entry Receptors in Liver Tissue",
"author": "Sharif-Askari",
"doi-asserted-by": "publisher",
"journal-title": "Arch Toxicol",
"key": "B25",
"volume": "94",
"year": "2020"
},
{
"DOI": "10.1016/j.imbio.2010.06.002",
"article-title": "High Glucose Disrupts Oligosaccharide Recognition Function via Competitive Inhibition: A Potential Mechanism for Immune Dysregulation in Diabetes Mellitus",
"author": "Ilyas",
"doi-asserted-by": "publisher",
"journal-title": "Immunobiology",
"key": "B26",
"volume": "216",
"year": "2011"
},
{
"DOI": "10.1177/1932296820924469",
"article-title": "Glycemic Characteristics and Clinical Outcomes of COVID-19 Patients Hospitalized in the United States",
"author": "Bode",
"doi-asserted-by": "publisher",
"journal-title": "J Diabetes Sci Tech",
"key": "B27",
"volume": "14",
"year": "2020"
},
{
"DOI": "10.3390/ijerph17103664",
"article-title": "Potential Benefits and Harms of Novel Antidiabetic Drugs During COVID-19 Crisis",
"author": "Mirabelli",
"doi-asserted-by": "publisher",
"first-page": "3664",
"journal-title": "Int J Environ Res Public Health",
"key": "B28",
"volume": "17",
"year": "2020"
},
{
"DOI": "10.2337/dc20-0660",
"article-title": "Clinical Characteristics and Outcomes of Patients With Diabetes and COVID-19 in Association With Glucose-Lowering Medication",
"author": "Chen",
"doi-asserted-by": "publisher",
"journal-title": "Diabetes Care",
"key": "B29",
"volume": "43",
"year": "2020"
},
{
"DOI": "10.1080/07435800.2020.1856865",
"article-title": "Insulin Use, Diabetes Control, and Outcomes in Patients With COVID-19",
"author": "Riahi",
"doi-asserted-by": "publisher",
"first-page": "45",
"journal-title": "Endocrine Res",
"key": "B30",
"volume": "46",
"year": "2021"
},
{
"DOI": "10.1016/S2666-7568(20)30033-7",
"article-title": "Metformin and Risk of Mortality in Patients Hospitalised With COVID-19: A Retrospective Cohort Analysis",
"author": "Bramante",
"doi-asserted-by": "publisher",
"journal-title": "Lancet Healthy Longevity",
"key": "B31",
"volume": "2",
"year": "2021"
},
{
"DOI": "10.3390/v12050491",
"article-title": "ACE2: The Key Molecule for Understanding the Pathophysiology of Severe and Critical Conditions of COVID-19: Demon or Angel",
"author": "Xiao",
"doi-asserted-by": "publisher",
"first-page": "491",
"journal-title": "Viruses",
"key": "B32",
"volume": "12",
"year": "2020"
},
{
"DOI": "10.1007/s00125-020-05164-x",
"article-title": "Prevention and Management of COVID-19 Among Patients With Diabetes: An Appraisal of the Literature",
"author": "Katulanda",
"doi-asserted-by": "publisher",
"journal-title": "Diabetologia",
"key": "B33",
"volume": "63",
"year": "2020"
},
{
"DOI": "10.3389/fmed.2020.00248",
"article-title": "The Lung, the Heart, the Novel Coronavirus, and the Renin-Angiotensin System; the Need for Clinical Trials",
"author": "Lumbers",
"doi-asserted-by": "publisher",
"journal-title": "Front Med",
"key": "B34",
"volume": "7",
"year": "2020"
},
{
"DOI": "10.1002/jmv.26232",
"article-title": "The Cytokine Storm and COVID-19",
"author": "Hu",
"doi-asserted-by": "publisher",
"journal-title": "J Med Virol",
"key": "B35",
"volume": "93",
"year": "2021"
},
{
"DOI": "10.1164/rccm.201712-2570OC",
"article-title": "AMP-Activated Protein Kinase Phosphorylation of Angiotensin-Converting Enzyme 2 in Endothelium Mitigates Pulmonary Hypertension",
"author": "Zhang",
"doi-asserted-by": "publisher",
"journal-title": "Am J Respir Crit Care Med",
"key": "B36",
"volume": "198",
"year": "2018"
},
{
"DOI": "10.1038/ki.2014.19",
"article-title": "Metformin and Other Antidiabetic Agents in Renal Failure Patients",
"author": "Lalau",
"doi-asserted-by": "publisher",
"journal-title": "Kidney Int",
"key": "B37",
"volume": "87",
"year": "2015"
},
{
"DOI": "10.1007/s13181-012-0278-3",
"article-title": "Metformin-Associated Lactic Acidosis (MALA): Case Files of the Einstein Medical Center Medical Toxicology Fellowship",
"author": "Kopec",
"doi-asserted-by": "publisher",
"journal-title": "J?Med Toxicol",
"key": "B38",
"volume": "9",
"year": "2013"
},
{
"DOI": "10.1378/chest.15-1703",
"article-title": "Lactic Acidosis in Sepsis: It’s Not All Anaerobic: Implications for Diagnosis and Management",
"author": "Suetrong",
"doi-asserted-by": "publisher",
"journal-title": "Chest",
"key": "B39",
"volume": "149",
"year": "2016"
}
],
"reference-count": 39,
"references-count": 39,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.frontiersin.org/articles/10.3389/fendo.2022.810914/full"
}
},
"score": 1,
"short-container-title": [
"Front. Endocrinol."
],
"short-title": [],
"source": "Crossref",
"subject": [
"Endocrinology, Diabetes and Metabolism"
],
"subtitle": [],
"title": [
"Metformin Use in Relation to Clinical Outcomes and Hyperinflammatory Syndrome Among COVID-19 Patients With Type 2 Diabetes: A Propensity Score Analysis of a Territory-Wide Cohort"
],
"type": "journal-article",
"update-policy": "http://dx.doi.org/10.3389/crossmark-policy",
"volume": "13"
}